Abstract
Botulinum toxin A (BoNT/A) injections into the bulbospongiosus muscle have emerged as a novel treatment for lifelong premature ejaculation (PE), though efficacy remains controversial. This study evaluates BoNT/A’s efficacy and safety through a comprehensive search of PubMed, Embase, Cochrane Library, Web of Science, ClinicalTrials.gov, and ISRCTN registry. We included randomized controlled trials, prospective, and retrospective studies, focusing on intravaginal ejaculation latency time (IELT) and adverse events. Four studies (three meta-analyzed) involving 263 patients were analyzed. BoNT/A increased IELT by 37.87 s at 1 month (MD, 37.87; 95% CI, −2.86 to 78.59; p = .07; I2 = 96%), 11.52 s at 3 months (MD, 11.52; 95% CI, −16.91 to 39.94; p = .43; I2 = 95%), and 2.41 s at 6 months (MD, 2.41; 95% CI, −9.19 to 14.00; p = 0.68; I2 = 77%). Short-term IELT improvement was observed but lacked statistical significance (p > .05), with high heterogeneity (I2 = 96%). Long-term effects diminished, suggesting declining efficacy. Adverse events occurred in 10.9% of patients, primarily erectile dysfunction (n = 5), and urinary disorders (n = 5). While current evidence does not definitively support BoNT/A’s efficacy, limited studies and methodological heterogeneity suggest that further research is warranted. Future studies should employ larger, multicenter designs, optimize injection methods, doses, and protocols, and identify suitable patient populations to validate BoNT/A’s clinical benefits.
Keywords
Introduction
Premature ejaculation (PE) is a prevalent male sexual dysfunction that not only impacts a man’s sense of masculinity but also diminishes sexual satisfaction and emotional intimacy between partners. According to the International Society for Sexual Medicine guidelines, PE is characterized by three key features: (a) ejaculation that consistently or almost always occurs within approximately 1 min of vaginal penetration during a man’s first sexual experience (lifelong PE) or a significant and distressing reduction in ejaculation time, typically around 3 min or less (acquired PE); (b) an inability to delay ejaculation during nearly all instances of vaginal penetration; and (c) negative personal consequences, such as distress, frustration, or avoidance of sexual intimacy (Serefoglu, McMahon, et al., 2014). Reports from various countries and regions suggest that 20% to 30% of men are affected by this condition (Corona et al., 2021; Hanafy et al., 2019; Porst et al., 2007).
Currently, on-demand oral selective serotonin reuptake inhibitors (SSRIs) and topical anesthetics are commonly employed for the treatment of PE. However, these therapies offer only temporary delays in ejaculation latency and are associated with a high likelihood of PE recurrence upon discontinuation (Gul et al., 2022). Furthermore, the side effects of SSRIs and topical anesthetics contribute to patient dissatisfaction with these treatment modalities (Dinsmore & Wyllie, 2009; Tram et al., 2024). Consequently, a definitive cure for PE remains elusive, and ongoing research continues to explore the most effective treatments for this condition (Gul et al., 2022).
Observations of the ejaculatory physiological process have shown that during the semen expulsion phase, rhythmic contractions of the bulbospongiosus, ischiocavernosus, and levator ani muscles propel semen antegradely from the posterior urethra (Gerstenberg et al., 1990). Consequently, inhibiting the contraction of the bulbospongiosus muscle to prolong ejaculatory latency is regarded as a potentially effective treatment option.
Botulinum toxin A (BoNT/A) selectively blocks the release of acetylcholine from nerve endings. When injected into muscle, it inhibits neural transmission, thereby preventing rhythmic muscle contractions (Huang et al., 2000). BoNT/A has been used in urology to treat neurogenic detrusor overactivity, detrusor-sphincter dyssynergia, motor and sensory urgency, and chronic prostatic pain, demonstrating both favorable therapeutic efficacy and safety (Leippold et al., 2003).
In 2010, Professor Serefoglu first proposed the hypothesis of utilizing BoNT/A injections into the bulbospongiosus muscle as a potential treatment for lifelong PE (Serefoglu & Silay, 2010). Subsequent animal studies have demonstrated that this method effectively prolongs ejaculatory latency time in rats, reduces the integrated electromyographic activity of the bulbospongiosus muscle, and does not adversely affect other dimensions of sexual function (Ongün et al., 2019; Serefoglu, Hawley, et al., 2014). Over recent years, there has been growing interest in BoNT/A as a therapeutic option for lifelong PE. Several randomized controlled trials (RCTs) have demonstrated that intramuscular BoNT/A injections into the bulbospongiosus muscle can significantly extend intravaginal ejaculatory latency time (IELT) in patients with lifelong PE (Li et al., 2018; Shaher et al., 2024). However, another study has reported no substantial improvements with this intervention (Almekaty et al., 2024). Thus, the present study aims to further assess the efficacy and safety of BoNT/A injections into the bulbospongiosus muscle for the treatment of lifelong PE.
Materials and Methods
Study Protocol
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Page et al., 2021). All data used in this meta-analysis were sourced from previously published studies; therefore, ethical approval was not required.
Search Strategy
Two reviewers (CL and YJ) conducted a comprehensive search of the PubMed, the Embase, the Cochrane Library, the Web of Science, the ClinicalTrials.gov, and the ISRCTN registry to identify studies on BoNT/A treatment for PE. The search, conducted in September 2024, had no language restrictions. Medical Subject Headings terms provided by PubMed were used during the search, and the detailed search strategy can be found in the supplementary materials.
Study Inclusion and Exclusion Criteria
Article screening was conducted by two additional reviewers (DS and ZC), who first reviewed the titles and abstracts. For articles with unclear titles or abstracts, the full texts were thoroughly reviewed to determine eligibility for inclusion. This systematic review included RCTs, preliminary clinical trials, prospective studies, and retrospective studies. Reviews, animal studies, letters to the editor, comments, studies with no valid data, conference papers, and non-peer-reviewed studies were excluded. In the meta-analysis, we only selected RCTs comparing the efficacy of 100U BoNT/A intramuscular injections into the bulbospongiosus muscle with a placebo.
Data Extraction
From the included studies, we extracted the first author’s name, publication year, country, sample size, type of PE, intervention, placebo, injection site, injection method, follow-up duration, and pre-treatment IELT. The primary outcome was post-treatment IELT, while the secondary outcome was adverse events.
Risk of Bias Assessment
The risk of bias was assessed using Review Manager 5.3.0 and the Cochrane Risk of Bias Assessment Tool, taking into account factors such as random sequence generation, allocation concealment, blinded randomization, blinding of outcome assessors, data completeness, selective outcome reporting, and other sources of bias. Each study was categorized as having a “low,” “high,” or “unclear” risk of bias (Higgins et al., 2011). In cases of disagreement, all authors participated in discussions to resolve the issues.
Statistical Analyses
The mean difference (MD) with a 95% confidence interval (CI) was used to estimate the effect size for continuous outcomes, while the odds ratio (OR) with a 95% CI was employed for dichotomous outcomes. Heterogeneity among studies was assessed using the I2 statistic. An I2 value greater than 50% indicated substantial heterogeneity, in which case a random-effects model was applied. If heterogeneity was low (I2 ≤ 50%), a fixed-effects model was used. Sensitivity analysis was performed by omitting one study in each turn. Publication bias was assessed using the funnel plot and Egger’s test to evaluate potential bias in the included studies. Due to insufficient data, subgroup analysis was not performed in this study. The pooled results are presented in a forest plot. All statistical analyses were performed using Review Manager 5.3.0 and Stata 15.1, with statistical significance defined as p < .05 for two-tailed tests.
Results
Study Selection
A search across six public databases initially identified a total of 49 records. After removing duplicates, 19 unique records remained. These comprised four review articles, three animal studies, two studies with unpublished results, two studies deemed irrelevant, one letter to the editor, one conference paper, and two studies with incomplete data. Among these, four RCTs specifically investigated the efficacy of BoNT/A for the treatment of lifelong PE (Almekaty et al., 2024; Elshahid et al., 2024; Li et al., 2018; Shaher et al., 2024). However, one RCT involving BoNT/A injections into the ischiocavernosus muscle, penile base, and glans lacked a placebo control group and was therefore excluded from this meta-analysis (Elshahid et al., 2024). Ultimately, only three studies met the inclusion criteria for the meta-analysis (Almekaty et al., 2024; Li et al., 2018; Shaher et al., 2024). Figure 1 illustrates the PRISMA flowchart, and the supplementary materials provide detailed information regarding the literature search process.

Study Selection Flowchart Following PRISMA Guidelines.
Systematic Review
Li et al. published the first RCT investigating the efficacy of BoNT/A for the treatment of lifelong PE. In this study, researchers administered multipoint injections of BoNT/A (100 U, concentration 10 U/mL) into the bilateral bulbospongiosus muscles based on clinical experience. At the 4-week follow-up, the IELT in patients with lifelong PE significantly increased from 44.4 ± 16.2 s at baseline to 141 ± 109.8 s (p = .000). Compared to the placebo group, the BoNT/A group demonstrated a significantly greater improvement in IELT (141 ± 109.8 s vs. 47.4 ± 12.6 s, p = .001). Additionally, the BoNT/A group showed superior outcomes in other measures, including the Premature Ejaculation Profile (PEP) and female satisfaction (Table 1). Furthermore, this study reported changes in ejaculatory function before and after treatment, as well as comparisons with the control group. The results indicated that following BoNT/A treatment, there were no statistically significant differences in ejaculatory force, semen volume, sexual pleasure, or scores related to ejaculation-related distress among the patients. Adverse events were reported in six cases (17.65%) in the BoNT/A group, including four cases (11.76%) of self-reported reduced erectile hardness and two cases (5.88%) of incomplete urination sensation. These adverse events occurred 3 to 4 days post-injection. Notably, patients with reduced erectile hardness were still able to complete sexual intercourse, and no additional treatment was required, as all symptoms resolved spontaneously within 3 weeks (Li et al., 2018).
Characteristic of Studies Included in the Analysis.

Note. BoNT/A
The study by Shaher et al. is the only RCT utilizing a double-blind design, where researchers performed extensive multipoint injections of BoNT/A (100 U, concentration 10 U/mL) into the bulbospongiosus muscle under ultrasound guidance. Compared to the baseline IELT of 35.79 ± 14.23 s, the postoperative IELT at 1, 3, and 6 months was 70.00 ± 47.64 s (p < .001), 60.36 ± 40.08 s (p < .001), and 42.64 ± 30.35 s (p = .162), respectively. Significant differences were observed between the BoNT/A group and the placebo group at 1 month (70.00 ± 47.64 s vs. 34.76 ± 15.25 s, p < .001) and 3 months (60.36 ± 40.08 s vs. 33.73 ± 14.11 s, p = .002), but no significant difference was noted at 6 months (42.64 ± 30.35 s vs. 33.64 ± 13.94 s, p = .386). Similarly, other metrics, such as the PEP and the Premature Ejaculation Diagnostic Tool (PEDT), demonstrated significant improvements at 1 and 3 months post-treatment, but no significant differences were observed at 6 months (Table 1). Adverse events were reported in four cases (8.5%) within the treatment group. Among these, two patients experienced post-micturition dribbling, one developed an infection that was managed with appropriate antibiotics, and one patient required Non-Steroidal Anti-Inflammatory Drugs for 1 day due to pain (Shaher et al., 2024).
In contrast to the findings of the previous two studies, the research conducted by Almekaty et al. concluded that BoNT/A treatment is ineffective for lifelong PE. Under ultrasound guidance, the researchers administered fan-shaped injections of BoNT/A (100 U, concentration 10 U/mL) into the bilateral bulbospongiosus muscles of patients with lifelong PE. Compared to the baseline IELT of 31.4 ± 9 s, the IELT at 1, 3, and 6 months post-treatment was 33.1 ± 11 s (p = .6), 33 ± 7 s (p = .6), and 31.9 ± 10 s (p = 0.5), respectively. No statistically significant differences were observed between the treatment group and the placebo group at 1 month (33.1 ± 11 s vs. 35.6 ± 7 s, p = .6), 3 months (33 ± 7 s vs. 35.4 ± 8 s, p = .5), or 6 months (31.9 ± 10 s vs. 34.8 ± 12 s, p = .5). Similarly, other metrics, such as the PEP and female satisfaction, also showed no statistically significant improvements (Table 1). Adverse events were reported in only two cases (6.9%) within the treatment group. One patient experienced mild erectile dysfunction (ED), characterized by reduced rigidity, which persisted for 1 month but did not preclude penetrative intercourse. Another patient reported post-micturition dribbling, which began on day 4 post-injection and lasted for 2 months (Almekaty et al., 2024).
The fourth study was conducted by Elshahid et al., who randomly assigned 45 patients with lifelong PE into three groups, with 15 patients in each group. Each group received 75 U of Dysport (equivalent to 25 U of Botox) injected respectively into the root of the penis, glans penis, and ischiocavernosus muscles. At the 1-month follow-up, the IELT in the three groups increased from baseline values of 31.90 ± 8.76 s, 41.46 ± 7.93 s, and 37.90 ± 10.28 s to 53.93 ± 18.46 s (p = .0001), 56.03 ± 8.47 s (p = .00002), and 76.60 ± 35.30 s (p = .0001), respectively. The PEDT scores at baseline were 12.9 ± 3.3, 11.4 ± 2.0, and 11.5 ± 2.4 for the three groups, and at 1 month post-treatment, the scores were 9.7 ± 4.3 (p = .03), 11.0 ± 1.9 (p = .5736), and 8.0 ± 5.1 (p = .02), respectively. Adverse events were reported as follows: 51% of patients experienced severe pain during injection (12 at the muscles, 8 at the root, and 3 at the glans). Additionally, 11% of patients reported localized edema that lasted for about 1 day and was relieved by an ice pack (three at the root and two at the glans). Furthermore, 7% of patients reported decreased rigidity during erection, but they were still able to engage in penetrative intercourse without additional treatment; this side effect lasted for approximately 3 weeks (two at the muscles and one at the root) (Elshahid et al., 2024).
Finally, a total of 263 patients with valid data were included in the four studies, all of whom had lifelong PE. Among them, 110 patients received bilateral injections of 100 U BoNT/A into the bulbospongiosus muscle. None of the four studies received any funding. Table 1 summarizes the detailed characteristics of the included studies.
Quality of the Study
Among the four included RCTs, one study was rated as having a low risk of bias (Shaher et al., 2024), one as moderate (Elshahid et al., 2024), and two as high (Almekaty et al., 2024; Li et al., 2018). The primary sources of bias stemmed from issues with blinding and incomplete outcome data. Figure 2 provides an overview of the risk of bias across all included studies.

Risk of Bias Assessment: (A) Risk of bias graph; (B) Risk of bias summary.
Efficacy in Improving IELT
Based on three placebo-controlled RCTs (Almekaty et al., 2024; Li et al., 2018; Shaher et al., 2024), bulbospongiosus muscle injections of 100 U of BoNT/A improved IELT by 37.87 s at 1 month (MD, 37.87; 95% CI, −2.86 to 78.59; p = .07; I2 = 96%; Figure 3a). However, since the 95% CI includes zero (−2.86 to 78.59), this result is not statistically significant (p = .07), indicating that the treatment effect could be either positive or negative. Therefore, no conclusion can be drawn regarding a significant improvement in IELT with BoNT/A.

Forest Plots Comparing Botulinum Toxin A 100U with Placebo in Terms of Intravaginal Ejaculatory Latency Time for Patients with Lifelong Premature Ejaculation at Different Follow-Up Time Points: (A) 1 Month, (B) 3 Months, and (C) 6 Months.
At 3 months, two RCTs (Almekaty et al., 2024; Shaher et al., 2024) showed a smaller improvement of 11.52 s (MD, 11.52; 95% CI, −16.91 to 39.94; p = .43; I2 = 95%; Figure 3b). Again, as the 95% CI includes zero (−16.91 to 39.94), this result did not reach statistical significance (p = .43).
At 6 months, the treatment effect diminished further, with a 2.41 s difference in IELT (MD, 2.41; 95% CI, −9.19 to 14.00; p = .68; I2 = 77%; Figure 3c). Since the 95% CI includes zero (−9.19 to 14.00), this difference is also not statistically significant (p = .68).
Sensitivity Analysis and Publication Bias Analysis
To assess the robustness of the meta-analysis results, we conducted a sensitivity analysis by sequentially excluding each study to observe changes in heterogeneity (I2) and p-values. When the study by Almekaty et al. was excluded, the I2 value decreased to 88%, and the p-value changed to .03 (Figure 4), indicating a reduction in heterogeneity and rendering the overall effect statistically significant (p < .05). This change suggests that the study by Almekaty et al. had a substantial impact on the overall effect estimate, particularly in the context of high heterogeneity. The significant decrease in heterogeneity and the shift to statistical significance after its removal imply that the results of Almekaty et al. may differ notably from those of other studies, potentially due to differences in sample characteristics, study design, or other methodological factors.
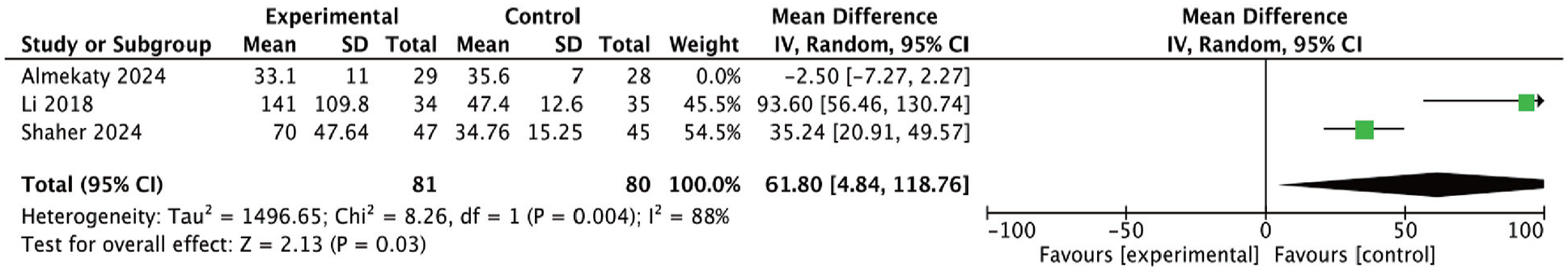
Sensitivity Analysis Conducted by excluding Studies with Heterogeneity.
The funnel plot exhibited some asymmetry, which may suggest potential publication bias (Figure 5a); however, this interpretation is limited by the small number of included studies (only three), reducing the reliability of the assessment. To further evaluate publication bias, Egger’s test was performed to examine the relationship between effect size and its standard error. The regression analysis revealed that the slope coefficient was not statistically significant (Slope = −17.82, p = .133), indicating no strong evidence of a linear relationship between effect size and standard error. Furthermore, the bias coefficient showed a marginally significant result (Bias = 6.48, p = .093), suggesting a potential but non-definitive bias, as it did not reach the conventional significance threshold of 0.05. The 95% confidence interval for the bias coefficient (−5.60 to 18.55) spanned both negative and positive values, further highlighting the uncertainty regarding the presence and magnitude of publication bias. In summary, while there may be some indication of potential publication bias, the evidence remains inconclusive due to the limited number of studies and the marginal significance of the statistical tests (Table 2 and Figure 5b).

Publication Bias Analysis: (A) Funnel Plot; (B) Eggerãs Publication Bias Plot.
Egger’s Test for Publication Bias.

Adverse Events
The meta-analysis of adverse events included data from three studies (Almekaty et al., 2024; Li et al., 2018; Shaher et al., 2024), involving a total of 218 individuals—110 in the treatment group and 108 in the placebo group. The incidence of adverse events in the treatment group was 10.9%, including five cases of ED, three cases of post-micturition dribbling, and two cases of incomplete urination. Additionally, there was one case of pain and one case of infection. The analysis revealed a significant difference in the occurrence of adverse events between the treatment and placebo groups, with an OR of 7.04 (95% CI, 1.57 to 31.59; p = .01; I2 = 0%; Figure 6).

Forest Plots Showing the Comparison of Adverse Events Between Botulinum Toxin A and Placebo.
Discussion
Ejaculation is a complex spinal reflex involving multiple neurophysiological pathways and a variety of neurochemical mediators within both the peripheral and central nervous systems (Clement & Giuliano, 2016). Although the exact etiology of lifelong PE is not fully understood, the prevailing theory in the academic community is Waldinger’s neurobiological hypothesis. This theory suggests that lifelong PE in men may result from a combination of polymorphisms in serotonergic transporters and receptors, as well as other neurotransmitters and/or receptors (Waldinger, 2002). A recent study found that patients with lifelong PE exhibit hyperexcitability in the “posterior urethra–bulbospongiosus muscle” reflex (Zhou et al., 2010). During the emission phase, semen accumulates in the posterior urethra, and the increased pressure in this area more easily triggers the rhythmic contractions of the bulbospongiosus muscle, leading to rapid ejaculation.
BoNT/A consists of a 100
Injecting BoNT/A into the bulbospongiosus muscle is considered a potential localized treatment aimed at inhibiting muscle contractions during the expulsion phase, thereby prolonging ejaculatory latency (Serefoglu & Silay, 2010). For patients with a hyperexcitable “posterior urethra–bulbospongiosus muscle” reflex, this therapy may offer promising outcomes. However, this technique currently lacks standardized operating procedures and dosing guidelines. The injection can be performed either under ultrasound guidance to target the muscle precisely or by blind injection in the lithotomy position, based on the operator’s experience. Both multiple-site and fan-shaped injection techniques have been applied in clinical practice. Regarding dosage, the published data generally report a dosage of 100 U. Based on currently published data and meta-analysis results, the efficacy of BoNT/A in the treatment of PE appears to decrease over time. Although there may be some improvement in the short term (1 month) (MD, 37.87; 95% CI, −2.86 to 78.59; p = .07; I2 = 96%), the therapeutic effect remains unclear. In the long term (6 months), the efficacy significantly diminished (MD, 2.41; 95% CI, −9.19 to 14.00; p = .68; I2 = 77%), suggesting that the long-term clinical value of BoNT/A may be limited. Similar results have been reported in other urological conditions, where BoNT/A has demonstrated difficulty in maintaining long-term efficacy, with therapeutic effects typically lasting between 2 and 6 months (Hamid et al., 2021; Hoelscher et al., 2024).
The current studies exhibit considerable heterogeneity, with published research showing contrasting trends, a concern that has also been raised by Professor Serefoglu. Recently, his team published a commentary addressing this issue (Gul et al., 2025). First, the heterogeneity may stem from differences in patient populations. For instance, the baseline IELT in Li et al.’s cohort was 44.4 ± 16.2 s, while in Shaher et al.’s cohort, it was 35.79 ± 14.23 s, and in Almekaty et al.’s cohort, it was the lowest at 31.4 ± 9 s. This suggests that the patients in Almekaty et al.’s cohort had more severe PE. The ejaculation reflex is mediated by a complex interaction between spinal reflexes and higher spinal centers (Clement & Giuliano, 2016). Increased excitability of these pathways, particularly in the lumbosacral spinal cord and the nerves surrounding the genitalia, may contribute to PE (Gul et al., 2025). In patients with more severe PE, the neural circuits responsible for ejaculatory control may be more active (Gul et al., 2025). However, BoNT-A primarily works by inhibiting the release of acetylcholine at the neuromuscular junction (Huang et al., 2000), and its therapeutic effect may be insufficient to address more severe neurophysiological imbalances. Therefore, the poorer outcomes observed in Almekaty et al.’s cohort may be attributed to deeper neural dysfunction at both the central and peripheral levels. Second, the injection technique may also contribute to variability in therapeutic outcomes. Both Li et al. and Shaher et al. employed a widely used multiple-site injection technique, whereas Almekaty et al. utilized a fan-shaped injection technique. The multiple-site injection allows BoNT-A to be distributed over a larger area of the bulbospongiosus muscle, which may lead to more effective muscle relaxation. This broader distribution could help reduce the rhythmic contractions of the bulbospongiosus muscle, thereby extending the IELT. In contrast, the fan-shaped injection technique may result in a less uniform distribution of BoNT-A within the bulbospongiosus muscle, potentially impairing the ability to modulate muscle activity during ejaculation.
In terms of adverse events, the most frequently reported issues associated with BoNT/A injections into the bulbospongiosus muscle are ED and urinary disturbances. The coordinated contraction of the bulbospongiosus muscle, along with the ischiocavernosus and superficial transverse perineal muscles, compresses the proximal corpora cavernosa and pulls the penile crura, thereby compressing the erectile vessels at the penile crura to restrict blood flow. This mechanism increases intracavernosal pressure, sustaining penile rigidity (Juárez & Cruz, 2014). Additionally, contraction of the bulbospongiosus muscle plays a supporting role in urination and ejaculation (Juárez & Cruz, 2014). The inhibition of bulbospongiosus muscle contraction by BoNT/A may interfere with these functions, potentially resulting in insufficient contraction strength, inadequate pressure on the erectile vessels, and impairments in urination, ejaculation, and erectile rigidity. However, these effects are reversible and tend to resolve as synaptic transmission is gradually restored.
Limitations and Prospects
According to current research, the application of BoNT/A for the treatment of lifelong PE remains in the early exploratory stages. Existing studies exhibit several limitations, including small sample sizes, single-center designs, and difficulties in implementing effective double-blind protocols, all of which may undermine statistical power. To address these limitations, future research should prioritize multicenter, large-scale, double-blind clinical trials to more rigorously establish the true efficacy of BoNT/A in PE management.
Furthermore, the absence of standardized injection techniques, optimal drug concentrations, and well-defined patient populations likely contributes to significant variability in treatment outcomes. There is a critical need for further investigation to develop standardized protocols, including comparative studies of different injection techniques, identification of the most effective drug concentrations, and establishment of clear patient selection criteria. Additionally, the limited evidence regarding the long-term durability of BoNT/A treatment raises concerns about the cost-effectiveness of repeated injections compared to on-demand therapies. Consequently, conducting robust cost-effectiveness analyses within clinical practice frameworks is imperative. Comprehensive safety evaluations are also warranted to assess both short-term adverse effects and potential long-term consequences associated with repeated treatments.
Finally, future research would benefit significantly from the adoption of standardized, internationally recognized, and validated assessment tools and questionnaires. Such an approach would enhance the credibility, reliability, and generalizability of findings, thereby facilitating the broader clinical application of BoNT/A in the management of PE.
Conclusion
Although the current meta-analysis results do not provide definitive evidence supporting the efficacy of BoNT/A for the treatment of lifelong PE, the limited number of studies and significant methodological heterogeneity highlight the need for further high-quality research before drawing conclusive judgments. These limitations suggest that prematurely dismissing BoNT/A as a potential treatment option may be unwarranted at this stage. As suggested in the prospects, future research should prioritize large-scale, well-designed RCTs to further confirm the efficacy of BoNT/A in treating lifelong PE.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883251328312 – Supplemental material for Clinical Efficacy and Safety of Botulinum Toxin A Injection into the Bulbospongiosus Muscle for Treating Lifelong Premature Ejaculation
Supplemental material, sj-docx-1-jmh-10.1177_15579883251328312 for Clinical Efficacy and Safety of Botulinum Toxin A Injection into the Bulbospongiosus Muscle for Treating Lifelong Premature Ejaculation by Dawei Gao, Chuyu Li, Yihan Jin, Dalin Sun, Zifeng Chen, Bo Tang, Weiping Chen and Baofang Jin in American Journal of Men's Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
