Abstract
Infertility is among the most serious medical problems worldwide. Male factors contribute to 40%–50% of all infertility cases, and approximately 7% of men worldwide are affected by infertility. Spermatozoa are extremely vulnerable to oxidative insult. Oxidative stress results in axonemal damage and increased midpiece sperm morphological defects, which lead to reduced sperm motility. The aim of the study is to evaluate the association between sperm motility and the levels of selected antioxidants, cytokines, and markers of oxidative damage in the seminal plasma.
The study group included 107 healthy males, who were split into two subgroups based on the percentage of motile spermatozoa after 1 hr: low motility (LM, n = 51) and high motility (HM, n = 56).
The glucose-6-phosphate dehydrogenase (G6PD) activity was 52% lower in the LM group compared to that in the HM group. The level of malondialdehyde (MDA) was 12% higher in the LM group compared to that in the HM group. Similarly, the median values of interleukin (IL)-1β, IL-10, IL-12, and tumor necrosis factor alpha (TNF-α) were higher in the LM group than those in the HM group. Results of the present study revealed that the percentage of motile spermatozoa after 1 hr correlated positively with the levels of IL-1β, IL-10, IL-12, and TNFα.
The lower motility of spermatozoa in healthy men is associated with a decreased activity of G6PD and increased levels of cytokines, which may be related to increased oxidative stress in seminal plasma that manifests as an increased level of MDA.
Infertility is usually defined in the biomedical context as the inability to conceive after 12 months of regular unprotected intercourse (Greil et al., 2010). Infertility is among the most serious medical problems worldwide (Dobrakowski et al., 2018; Mascarenhas et al., 2012). Male factors contribute to 40%–50% of all infertility cases, and approximately 7% of men worldwide are affected by infertility (Agarwal and Bui, 2017). Male infertility could be a result of anatomical abnormalities, such as semen outflow tract obstruction, neurological disorders of ejaculation, and urogenital trauma; however, most of the cases are caused by impaired spermatogenesis and failure in sperm function. As male fertility can be influenced by a variety of factors, one possible explanation for the declining trend would be that there are environmental and/or occupational factors along with lifestyle practices that contribute to the deterioration of semen quality. The lifestyle factors that are associated with male infertility are as follows: smoking cigarettes, alcohol intake, use of illicit drugs, obesity, psychological stress, advanced paternal age (APA), dietary practices, and coffee consumption (Durairajanayagam, 2018).
Semen analysis is routinely performed to assess the reproductive potential of the male partner in infertile couples, even though there are no standard parameters to diagnose infertility (Guzick et al., 2001). Sperm motility is considered the most important parameter of a semen specimen during the evaluation of fertility (Amelar et al., 1980).
Sperm quality is particularly altered by oxidative stress (Walczak-Jedrzejowska et al., 2013), which is defined as an imbalance between the generation of reactive oxygen species (ROS) and the protective action of antioxidant systems responsible for their neutralization and removal (Sikka, 2001). Imbalance between production and utilization of ROS leads to damage of many cell structures, especially phospholipids of cellular membranes. Oxidative damage to lipids, called lipid peroxidation, triggers, in turn, signaling cascades of the inflammatory processes which promote peroxidation of lipids, resulting in intracellular oxidative burden. The sequence of events involves lipid peroxidation, loss of membrane integrity with increased permeability, reduced sperm motility, structural DNA damage, and apoptosis (Alahmar, 2019). ROS as by-products are released during the physiological metabolic processes of cells in semen (Ko et al., 2014). Results of earlier studies indicate that the cytokine levels in seminal plasma have been correlated with semen quality parameters (Kopa et al., 2005). A number of studies have reported that ROS and cytokines possess a complex and exquisite interplay (Chyra-Jach et al., 2018). Although ROS can promote the expression and production of cytokines, some cytokines can modulate prooxidant and antioxidant systems and the generation of ROS (Jiang et al., 2016; Seshadri et al., 2009).
Spermatozoa are extremely vulnerable to oxidative insult because they lack the necessary repair systems and are unable to restore the oxidative damages. Besides, spermatozoa membranes are rich in polyunsaturated fatty acids (PUFAs), which makes them highly susceptible to lipid peroxidation. Oxidative stress results in axonemal damage, decreased sperm viability, and increased midpiece sperm morphological defects. These dysfunctions may contribute to decreased sperm motility (Agarwal et al., 2014). In light of this, the present study was undertaken to examine the associations between sperm motility and the levels of selected antioxidants, cytokines, and oxidative damage markers.
Materials
The study was approved by the Bioethics Committee of the Medical University of Silesia in Katowice (KNW/0022/KB1/I/13/09). A total of 107 fertile males living in Upper Silesia (Poland), who attended the andrology clinic for the diagnosis of infertility, were enrolled as the study group. All patients were healthy and fertile with normal sperm parameters according to the World Health Organization (WHO) standards. Written informed consent was obtained from all the study subjects. A questionnaire about the participants’ medical history was also collected. Subjects who reported drug consumption, smoking habits, alcohol abuse, and a history of any chronic disease were excluded from the study. Also, any diseases that may cause secondary infertility were included in the exclusion criteria.
Semen specimens were collected via masturbation after 3 days of abstinence, in the morning before the first meal. According to the percentage of motile spermatozoa after 1 hr, the patients were divided into two groups based on the median: low motility group with less than or equal to than 57% motile spermatozoa (LM group, n = 51) and high motility group with more than 57% motile spermatozoa (HM group, n = 56).
Methods
Semen specimens were analyzed according to the WHO standards (WHO, 2010). The seminal plasma and spermatozoa lysate were prepared according to Kasperczyk et al. (2015).
The activity of antioxidant enzymes was measured in the seminal plasma according to the methods described in Kasperczyk et al. (2015). The method of Oyanagui (1984) was used to measure the activity of superoxide dismutase (SOD) in seminal plasma. The enzymatic activity of SOD was expressed in nitric units (NU). The activity of SOD is equal to 1 NU when it inhibits nitric ion production by 50%. Activity of SOD was expressed in NU/mg of protein. The glutathione peroxidase (GPx) activity was measured by the kinetic method of Paglia and Valentine (1967). The GPx activity was expressed as U/g of protein. The activity of glutathione-S-transferase (GST) was measured according to the kinetic method of Habig and Jakoby (1981). The GST activity was expressed as the µmoles of thioether produced per minute per gram of protein (mU/g protein). The activities of glucose-6-phosphare dehydrogenase (G6PD) and glutathione reductase (GR) were measured according to Richterich (1971). G6PD and GR activity were expressed as µmoles of NADPH produced and utilized, respectively, per minute per gram of protein (U/min/g protein). Catalase (CAT) activity was measured by the method of Johansson and Borg (1988). The activity of catalase was expressed as U/g of protein.
Malondialdehyde (MDA) was measured fluorometrically as a 2-thiobarbituric acid-reactive substance (TBARS) in seminal plasma according to Ohkawa et al. (1979) with modifications according to Kasperczyk et al. (2015). Concentrations were given in μmol/L.
The levels of interleukin (IL)-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12, IL-13, IL-17, tumor necrosis factor alpha (TNF-α), and interferon gamma (IFN-γ) were measured in seminal plasma using a Bio-Plex 200 System (Bio-Rad Laboratories Inc., USA) according to the manufacturer’s instructions. Data were presented in pg/ml.
The results are expressed as median values and interquartile ranges (IQRs) or mean and standard deviation (SD). The statistical analyses were performed using nonparametric tests for data without a normal distribution (Mann–Whitney U test) or parametric tests for data with a normal distribution (Student’s t test). Normality and homogeneity of variances were verified using Shapiro–Wilk’s and Levene’s test, respectively. Statistical analysis of the impact of age in the examined groups was done using the one-way analysis of variance (ANOVA). Additionally, regression analysis was performed. The level of statistical significance was assumed to be p < .05. The statistical analyses were carried out using Stat-Soft software (Statistica 10.0 PL).
Results
Table 1 reports the age, semen parameters, and antioxidant enzyme activities in the enrolled patients, according to the study groups. Patients in the LM group were older than those in the HM group. Semen volume, sperm concentration in 1 ml, total sperm count, normal morphology (%), and motile spermatozoa after 24 hr (%) were similar between the examined groups. Other parameters characterizing motility differed between the examined groups due to the criterion of study group division.
Age, Semen Analysis, and Oxidative Stress Intensity and Antioxidant Reserves in Seminal Plasma.

Note. Data are expressed as mean ± SD. Statistical differences were analyzed by Student’s t test. CAT = catalase; GPx = glutathione peroxidase; GR = glutathione reductase; GST = glutathione-S-transferase; HM = high motility; LM = low motility; SOD = superoxide dismutase.
p < .05.
The activity of G6PD was 52% lower in the LM group compared to that in the HM group, while the level of MDA was 12% higher in LM group compared to that in the HM group (Figure 1). Similarly, the median values of IL-1β, IL-10, IL-12 (p70), and TNF-α were higher in the LM group than those in the HM group (Figures 2 and 3). The level of other antioxidant enzyme activities and interleukins was similar in both groups (Tables 1 and 2.)

Glucose-6-phosphate dehydrogenase (G6PD) activity and malondialdehyde (MDA) concentration in seminal plasma. Data are shown as mean and SD. * p < .05.

Concentrations of cytokines IL-1β and IL-10 in seminal plasma. Data are shown as median, lower and upper quartiles. * p < .05.

Concentrations of cytokines IL-12 (p70) and TNF-α in seminal plasma. Data are shown as median, lower and upper quartiles. * p < .05.
Concentrations of Cytokines in Seminal Plasma.
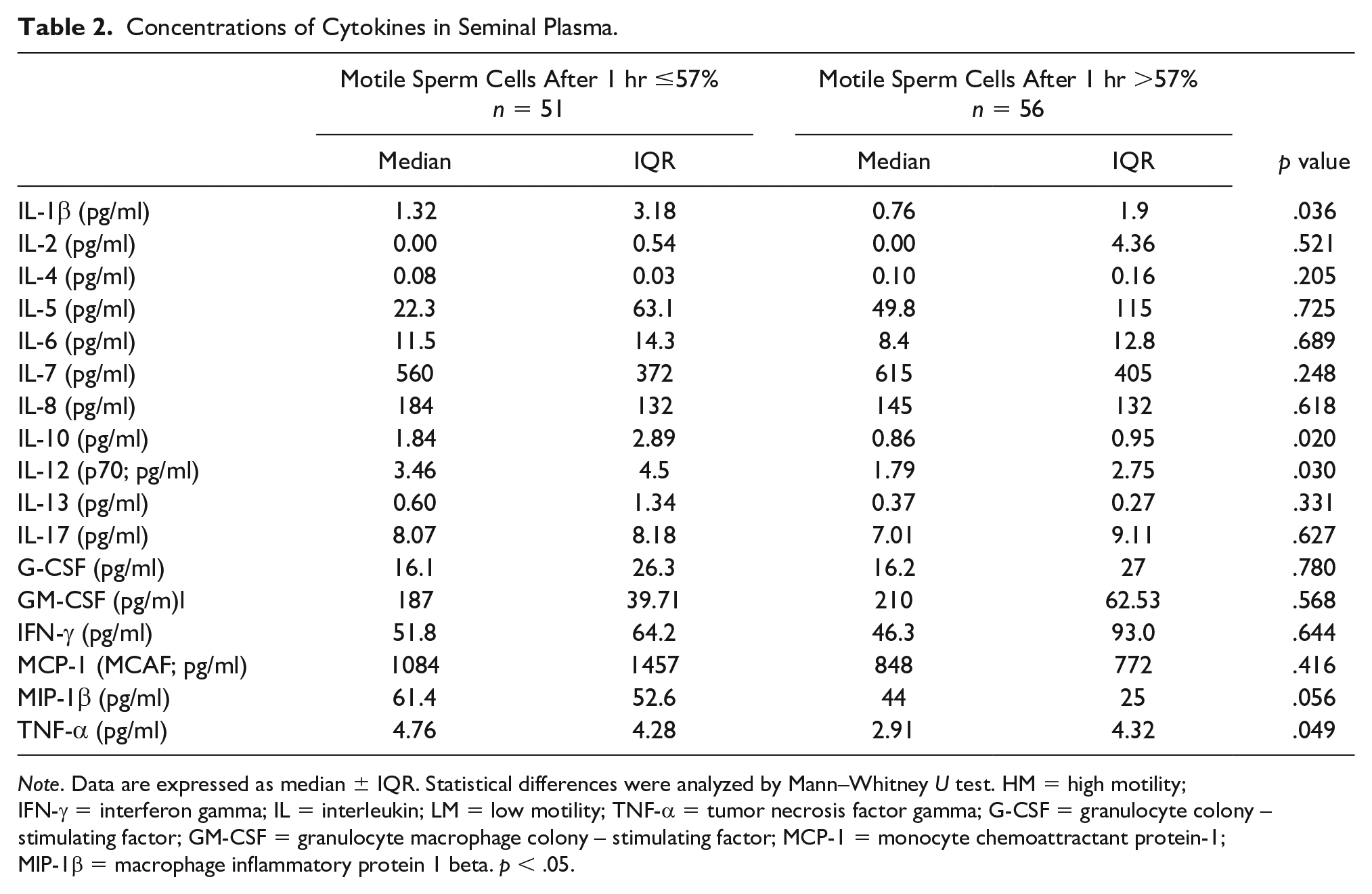
Note. Data are expressed as median ± IQR. Statistical differences were analyzed by Mann–Whitney U test. HM = high motility; IFN-γ = interferon gamma; IL = interleukin; LM = low motility; TNF-α = tumor necrosis factor gamma; G-CSF = granulocyte colony – stimulating factor; GM-CSF = granulocyte macrophage colony – stimulating factor; MCP-1 = monocyte chemoattractant protein-1; MIP-1β = macrophage inflammatory protein 1 beta. p < .05.
The percentage of motile spermatozoa after 1 hr was negatively correlated to the levels of IL-1β (R = −0.37, p = 0.015), IL-10 (R = −0.31, p = .040), IL-12 (R = −0.30, p = .047), and TNFα (R = −0.32, p = .042; data not reported). The activity of G6PD had a tendency toward positive correlation to the percentage of motile spermatozoa after 1 hr (R = 0.26 p = .057). There was no significant correlation between age and motile sperm (p = .248). Moreover, the analysis of the impacts of age in the examined groups did not obtain any significant differences (data not shown).
Discussion
The effect of age on fertility in men is not as explicit as that in women. Since the average age of paternity has been rising in Western countries, the issue of defining APA is becoming an important issue. The semen analysis seems to be crucial for assessing the fertility potential of males. The comparisons of semen parameters of younger and older men in individual studies demonstrated inconsistent findings, while meta-analyses proved that age has a negative impact on semen quality, including volume, morphology, and motility (Mazur and Lipshultz, 2018). The present study showed that study participants with higher motility were significantly younger than those with lower motility. These findings are in concordance with the study of Beguería et al. (2014) who reported that the percentage of motile spermatozoa significantly decreased with age by 1.2% every 5 years.
The enzymatic and nonenzymatic compounds that interact with each other to protect spermatozoa against ROS build up the protective antioxidant system in semen. The triad of main antioxidant enzymes includes SOD, CAT, and GPx. SOD neutralizes the superoxide anion, while CAT and GPx remove the hydrogen peroxide produced by SOD. GPx catalyzes the reduction of organic peroxides (Walczak-Jedrzejowska et al., 2013). The bioavailability of the reduced glutathione (GSH) is indispensable for GPx, which oxidizes GSH to its disulfide form (GSSG). The recycling of GSH from GSSG depends on GR, which needs NADPH. One of the major sources of NADPH is glucose-6-phosphate dehydrogenase (G6PD), the enzyme of the pentose phosphate pathway that transforms glucose-6-phosphate into phospho-6-glucono-lactone (Griveau et al., 1995).
An in vitro study by Williams and Ford (2004) reported that the pentose phosphate pathway in human spermatozoa responds dynamically to oxidative stress and, along with GPx and GR, serves as a functional effective antioxidant defense. In a study on goat semen, Qiu et al. (2016) reported that the pentose phosphate pathway is more important than glycolysis to maintain sperm motility. The present study did not identify any association between sperm motility and the activities of SOD, CAT, GPx, and GR. Nevertheless, the activity of G6PD was significantly lower in the LM group than that in the HM group. The level of MDA was significantly higher in the LM group. Such data suggest the existence of additional mechanisms that connect the activity of G6PD, oxidative stress and motility. According to the study of Fernandez and O’Flaherty (2018), peroxiredoxins (PRDXs) may serve as key components of such mechanism. This family of antioxidant enzymes is present in all subcellular compartments of human spermatozoa and can scavenge a wide variety of ROS, thanks to the cysteine (Cys) residue at their active sites. Oxidation of the Cys thiol group inactivates PRDXs by forming a disulfide bond. Similarly to glutathione, the reduction of oxidized PRDXs is catalyzed by the thioredoxin- (TRX) thioredoxin reductase (TRD) that needs NADPH produced by G6PD. This cascade of reactions indicates how decreased G6PD activity may lead to an elevated MDA level, even though the activity of GPx and GR remains unchanged. On the contrary, Roshankhah et al. (2016) reported that G6PD deficiency does not increase the susceptibility of sperm to oxidative stress induced by hydrogen peroxide; however, the small sample group represents a substantial limitation of this study. The authors suggest an existence of alternative mechanisms for the generation of NADPH, such as NADPH-linked malic enzyme, NADPH-linked isocitrate dehydrogenase, isocitrate dehydrogenase and glutamate dehydrogenase, nicotinamide nucleotide transhydrogenase, and NADH kinase.
MDA is widely measured to assess the oxidative damage to lipids in spermatozoa, even though the results of the association between the seminal MDA level and sperm quality are controversial (Ammar et al., 2019). Tomar et al. (2017) reported that MDA levels in normospermic men were significantly lower than those in asthenoteratospermic and oligoasthenoteratospermic men and had a negative correlation with sperm count, motility, and morphology. A negative correlation between MDA and motility was also identified in studies by Subramanian et al. (2018) and Atig et al. (2012). In contrast, Ammar et al. (2019) and Suleiman et al. (1996) reported no correlation between MDA and motility. In the present study, despite a significant difference between MDA levels in the studied groups, there was no correlation between MDA level and motility, which suggests a possible nonlinear dependence between these parameters in the semen of fertile males. Our previous study demonstrated that the level of MDA in seminal plasma was higher in males with asthenospermia than in controls; however, the level of MDA in spermatozoa lysate was significantly lower in males with oligospermia, asthenospermia, and oligoasthenospermia than that in the fertile controls (Dobrakowski et al., 2018).
The proliferation of germ cells and differentiation of mesenchymal cells are regulated by cytokines produced by testes. These polypeptide regulatory factors in seminal plasma are correlated with semen quality parameters. Genital infection is related to elevated levels of several cytokines, which could play a role in the immune defense of the male genital tract. Besides, many reports indicate that there are complex interactions between ROS and cytokines. ROS can induce the expression and production of cytokines, while cytokines can modulate the generation and utilization of ROS (Jiang et al., 2016). It has been proposed that poor sperm quality related to oxidative stress and lipid peroxidation may be partially due to the action of proinflammatory cytokines such as IL-1β, IL-6, IL-8, and TNF-α (Martínez et al., 2007); however, studies on this topic are not conclusive. The present study reports that IL-1β and TNF-α levels in the seminal plasma are higher in males with low sperm mobility, with negative correlation between the percentage of motile spermatozoa after 1 hr and the levels of these cytokines. Furthermore, we showed similar results for IL-10 and IL-12. Both cytokines are associated with T-cell function and are detected at low concentration in physiological semen. Increased levels of these cytokines were detected in the semen of men with genital infections (Politch et al., 2007). Naz and Evans (1998) reported a positive correlation between IL-12 level and the number and normal morphology of sperm. A positive association between IL-6 and lipid peroxidation, as well as infertility, was identified by Camejo et al. (2011); however, the authors of this study did not find such association for TNF-α. Nevertheless, Fraczek et al. (2007) reported that proinflammatory cytokines per se are unable to induce lipid peroxidation due to oxidative stress. Such results must be considered with caution, because the levels of proinflammatory cytokines vary between fertile men and fluctuate over time in seminal fluid, in response to duration of sexual abstinence and other unidentified factors.
The results of the conducted tests may indicate that in normal semen, factors that increase sperm motility are high G6PD activity and low level of oxidative stress as well as low values of some cytokines. Impact on the above-mentioned factors, for example, through the use of drugs and supplements can contribute to the improvement of sperm motility.
The results of the present research should be evaluated in the context of its limitations. The possible confounding role of lifestyle and additional disease states linked with ROS. Moreover, it is possible that the study was underpowered to detect significant differences in measured parameters.
Conclusions
Lower motility of spermatozoa of healthy men is associated with decreased activity of G6PD and increased levels of IL-1β, IL-10, IL-12, and TNF-α, which may be related to elevated oxidative stress in seminal plasma.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical University of Silesia KNW-1-167/N/9/K.
