Abstract
Gynecomastia is a common incidental finding on thoracic computed tomography (CT). This study aimed to retrospectively determine the prevalence, imaging characteristics, and possible causes of incidental gynecomastia on thoracic CT. Records of male patients who underwent thoracic CT in 2015 were reviewed. The size and morphologic types (nodular, dendritic, and diffuse) were recorded for patients with breast glandular tissue larger than 1 cm, and the cutoff value of gynecomastia was defined as 2 cm. Additionally, the possible causes of gynecomastia obtained by reviewing patients’ charts were recorded. CT-depicted gynecomastia was identified in 12.7% (650 of 5,501) of patients. The median size of the breast glandular tissue was 2.5 cm (interquartile range 2.2–3.1), and 36.8% of patients (239 of 650) had unilateral gynecomastia. The age distribution provided a bimodal pattern with two peaks in the age groups from 20 to 29 years old and greater than 70 years old. Chronic liver disease (CLD; p < .001), all stages of chronic kidney disease (CKD; p < .001), and medications (p = .002) were significantly associated with gynecomastia. Gynecomastia did not correlate with body mass index (p = .962). The size of breast glandular tissue was identified to be correlated with the morphologic type of breast tissue and the severity of CLD or CKD. The prevalence of incidental gynecomastia seen on thoracic CT was 12.7%. CT-depicted gynecomastia is not associated with obesity but with CLD, CKD, and medications. When gynecomastia is detected on CT, further evaluations and management might be required for patients with a treatable cause.
Gynecomastia is the benign proliferation of the male breast glandular tissue (Cuhaci et al., 2014; Johnson & Murad, 2009). It is the most common abnormality in the male breast (Cuhaci et al., 2014; Johnson & Murad, 2009) and believed to be caused by an imbalance between estrogen and testosterone that occurs through multiple mechanisms (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Mieritz et al., 2017; Nuttall, 2010; Sansone et al., 2017). Physiologic gynecomastia is quite common and appears with a trimodal age distribution in newborns, adolescents, and older men (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Mieritz et al., 2017; Nuttall, 2010; Sansone et al., 2017).
The prevalence of gynecomastia has not been established. Previously (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; Mieritz et al., 2017; Niewoehner & Nuttal, 1984; Nuttall, 1979; Williams, 1963) estimated prevalence rates for gynecomastia varied from 32% to 65% according to diagnostic modality, age, and lifestyle. These studies of gynecomastia have been limited by small patient numbers or because they were based on physical examination or autopsy studies (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; Niewoehner & Nuttal, 1984; Nuttall, 1979; Williams, 1963). Unlike mammography (MG) or ultrasonography (US), computed tomography (CT) is generally not intended for palpable breast masses in men. With the widespread use of thoracic CT for a variety of other indications, gynecomastia has been commonly reported as an incidental finding on thoracic CT (Gossner, 2019; Klang et al., 2017, 2018; Sonnenblick et al., 2016; Yi et al., 2008). The actual prevalence of gynecomastia initially detected on thoracic CT has, to date, not been well established owing to small patient numbers.
In addition, the clinical importance of CT-depicted gynecomastia is unknown. To our knowledge, patients with CT-depicted gynecomastia who complained of associated symptoms or had additional examinations or treatment has not been reported in prior studies. The purpose of this study was to retrospectively determine the prevalence, possible causes, and clinical implications of CT-depicted gynecomastia identified during thoracic CT in asymptomatic men.
Methods
Study Population
The institutional review board at Inha University Hospital (INHAUH 2019-04-025-002) approved the protocol for this retrospective study and the requirement for informed consent was waived. The initial study population was identified by searching the institutional Picture Archiving and Communications System database for 5,501 male patients who underwent thoracic CT examination at Inha University Hospital in South Korea during 2015. The exclusion criteria were patients with high-resolution CT (HRCT) scans with interscan gap (slice thickness of 1.25 mm using 2 mm interval) and patients without accurate medical records. HRCT scans were excluded because their goal is the assessment of lung parenchyma using an interscan gap and a high-resolution algorithm, which may render them nondiagnostic for the soft tissues (Worthy, 1995). If a patient underwent multiple examinations during the study period, only their first examination was used for analysis.
CT Imaging Technique
All thoracic CT studies were performed on one of the following CT scanners: a 16-slice CT scanner (SOMATOM Sensation 16; Siemens AG, Munich, Germany) or a 64-slice CT scanner (LightSpeed VCT or Optima 660; GE Healthcare, Milwaukee, WI). Examinations included images obtained with or without contrast. The area of coverage extended from the lower neck to the adrenal glands. Axial section data were reconstructed at a 3 mm thickness using a 3 mm slice interval for the SOMATOM Sensation 16 or at a 2.5 mm thickness using a 2.5 mm slice interval for the LightSpeed VCT. All images were processed with the standard mediastinal (width, 350 HU; level 20 HU) window setting.
Study Design and Subjects
An age-matched case-control study was conducted to evaluate the possible causes of gynecomastia. Subjects with gynecomastia were retrospectively selected through image analysis. Two radiologists (with 12 and 4 years of experience in thoracic imaging) retrospectively reviewed the presence of the breast glandular tissue on CT scans and reached consensus. Although the diagnostic cutoff value of gynecomastia was not established on CT, many studies used the presence of a 2 cm breast glandular tissue as a cutoff value for gynecomastia (Gossner, 2019; Niewoehner & Nuttal, 1984, Nuttall, 2010); the same criterion was also used in this study. The size of the glandular tissue in the right and left breasts was measured in millimeters using an electronic caliper tool at the level of the nipple on the axial CT image, as described previously (Gossner, 2019; Klang et al., 2018). Previous CT-based studies demonstrated that even in the general population, there were no males with no breast tissue at all; most men had a breast glandular tissue of mean 1.2 cm (Klang et al., 2018). For this reason, using software, an age-matched control group with the same number of participants as the subject group was selected among the patients with breast glandular tissue less than 1cm.
Data Collection
Breast glandular tissues were classified according to size and the three characteristic patterns (nodular, dendritic, and diffuse) established by previous studies (Appelbaum et al., 1999). Patients with breast glandular tissue of 1 cm or larger were screened and used for size comparisons among those with a morphologic type of breast glandular tissue, to prevent size restriction from underestimating subjects in the nodular type group. Both reviewers were blinded to the clinical data.
Medical records were reviewed to obtain patient demographics, indications for imaging, and medical history predisposing to gynecomastia, such as chronic liver disease (CLD), fatty liver, chronic kidney disease (CKD), medication, alcoholism, adrenal tumor, liver metastasis, HIV, thyrotoxicosis, germ cell tumor, and Klinefelter syndrome (Braunstein, 2007; Cuhaci et al., 2014; Deepinder & Braunstein, 2012; Gossner, 2019; Klang et al., 2017, 2018; Nuttall, 2010; Nuttall et al., 2015; Sonnenblick et al., 2016). From January 2015 through December 2018, any patients with symptoms associated with gynecomastia or who had undergone further imaging (such as MG or US) were also identified.
In South Korea, chronic hepatitis B and C, alcohol-induced chronic hepatitis, and liver cirrhosis (LC) account for more than 90% of hepatocellular carcinoma (HCC) cases (Trevisani et al., 2002; Zhang et al., 2004). For these high-risk groups, abdominal US and serum alpha-fetoprotein (AFP) are recommended at 6-month intervals (Korean Liver Cancer Association & National Cancer Center, 2019). For this study, reviewers identified findings of abdominal US in these high-risk groups and identified patients with CLD or LC.
Fatty liver was graded in four categories by reviewing the US performed on the same day as a CT or measuring the hepatic–splenic attenuation difference calculated as Δ = L – S, where L is the hepatic attenuation and S is the splenic attenuation. The grades of fatty liver, as depicted by US or CT, are defined qualitatively as Grade 0 (normal), Grade 1 (mild, characterized by a minimal increase in echogenicity of the liver, when compared with that of the renal cortex, and precise depiction of the hepatic and portal vein walls on US), Grade 2 (moderate, with increased liver echogenicity obscuring the hepatic and portal vein walls on US or a hepatic–splenic attenuation difference of −10 to 5 HU on CT), and Grade 3 (severe, with increased liver echogenicity and significant posterior shadowing that impairs evaluation of the deep liver parenchyma and diaphragm on US or a hepatic–splenic attenuation difference less than −10 HU on CT; Ma et al., 2009).
Renal function was estimated based on an estimated glomerular filtration rate (eGFR). The 2009 Chronic Kidney Disease Epidemiology Collaboration creatinine equation was used to calculate the eGFR (Levey et al., 2009), which was categorized as ≥90 (G1, normal), 60 to 89 (G2, mild), 30 to 59 (G3, moderate), 15 to 29 (G4, severe), and <15 ml/min/1.73 m2 (G5, kidney failure) according to the Kidney Disease Improving Global Outcomes 2012 Clinical Practice Guideline (Eknoyan et al., 2013).
Various medications or drugs are implicated in gynecomastia (Table 1). Any medication that might cause gynecomastia was classified into one of eight groups based on the guidance of previous literature (Braunstein, 2007; Cuhaci et al., 2014; Deepinder & Braunstein, 2012; Nuttall et al., 2015).
Medications Associated With Gynecomastia.

History of alcohol use was recorded; a high-risk drinking group was reviewed to investigate the correlation between alcohol and gynecomastia. The Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; American Psychiatric Association, 2013) diagnostic criteria are widely used for diagnosing alcohol abuse. However, due to the nature of the retrospective study, it was not possible to ask patients about their alcohol habits. Instead of the DSM-5 diagnostic criteria, “high-risk drinking” (as defined by the Korea Centers for Disease Control and Prevention) was used as a criterion for alcohol abuse. For males, the Korea Centers for Disease Control and Prevention (which conducts the Korea National Health and Nutrition Examination Survey) defines high-risk drinking as consuming seven or more alcoholic drinks at least twice a week (Korea Centers for Disease Control and Prevention, 2016).
Adrenal glands included in thoracic scans were reviewed to confirm the presence or absence of a mass. For patients with a history of malignancy including lymphoma, abdominal CT scans were also reviewed to check for liver metastases. Patients with a history of HIV infection, thyrotoxicosis, germ cell tumor, and Klinefelter syndrome were identified by a review of medical records.
Statistical Analysis
The Wilson method was used to calculate the 95% confidence intervals (CIs) of the prevalence of gynecomastia on thoracic CT (Newcombe, 1998). Analysis was conducted using the SPSS statistical software package (version 19.0, Chicago, IL) and dBSTAT for Windows (version 5.0, Seoul, Korea); p values <.05 were considered statistically significant.
Student’s t test was used to analyze continuous variables and the Fisher’s exact test or the chi-square test was used to identify correlations among categorical variables. Multivariable logistic regression analyses with the “Enter” method were performed to determine and obtain odds ratios (ORs) of possible causes affecting the presence of gynecomastia. Variables with p < .10 through unadjusted univariable analysis were used as input variables for logistic regression analyses. Age adjustment was only used to control for age effects.
Spearman’s correlation coefficients (rho) was used to analyze each correlation between size and multiple factors including body mass index (BMI), grade of CKD, and grade of CLD. BMI was classified into three groups based on the World Health Organization definition (WHO Expert Consultation, 2004). BMI values of 25.0 to 29.9 kg/m2 were classified as overweight and values above 30.0 kg/m2 and below 25.0 kg/m2 were classified as obesity and normal, respectively. The Cochran–Armitage test was used to investigate changes in risk factor trends based on age and assess linear trends regarding the presence of gynecomastia based on BMI and age (by decades) with possible causes. The Kruskal–Wallis test followed by Tukey’s post hoc test with size-rank was used to compare the sizes of gynecomastia among the morphologic types.
Results
A total of 5,501 patients who underwent a thoracic CT scan during the study period were included. Of them, 384 patients were excluded because they lacked precise medical records or because they underwent HRCT (Figure 1). Breast glandular tissues larger than 1 cm on thoracic CT were detected in 1,181 of 5,117 patients (23.1%; 95% CI [22.0, 24.3]). Of them, 671 were diagnosed as having gynecomastia with breast glandular tissue size ≥2 cm. Another 21 patients were excluded from the analysis because of incomplete medical records. A total of 650 patients (12.7%; 95% CI [11.8, 13.7]) were included. The mean age of the included patients was 56.44 years (±16.75 [SD], range 11–95). The clinical and radiographic characteristics of the study population are summarized in Table 2.
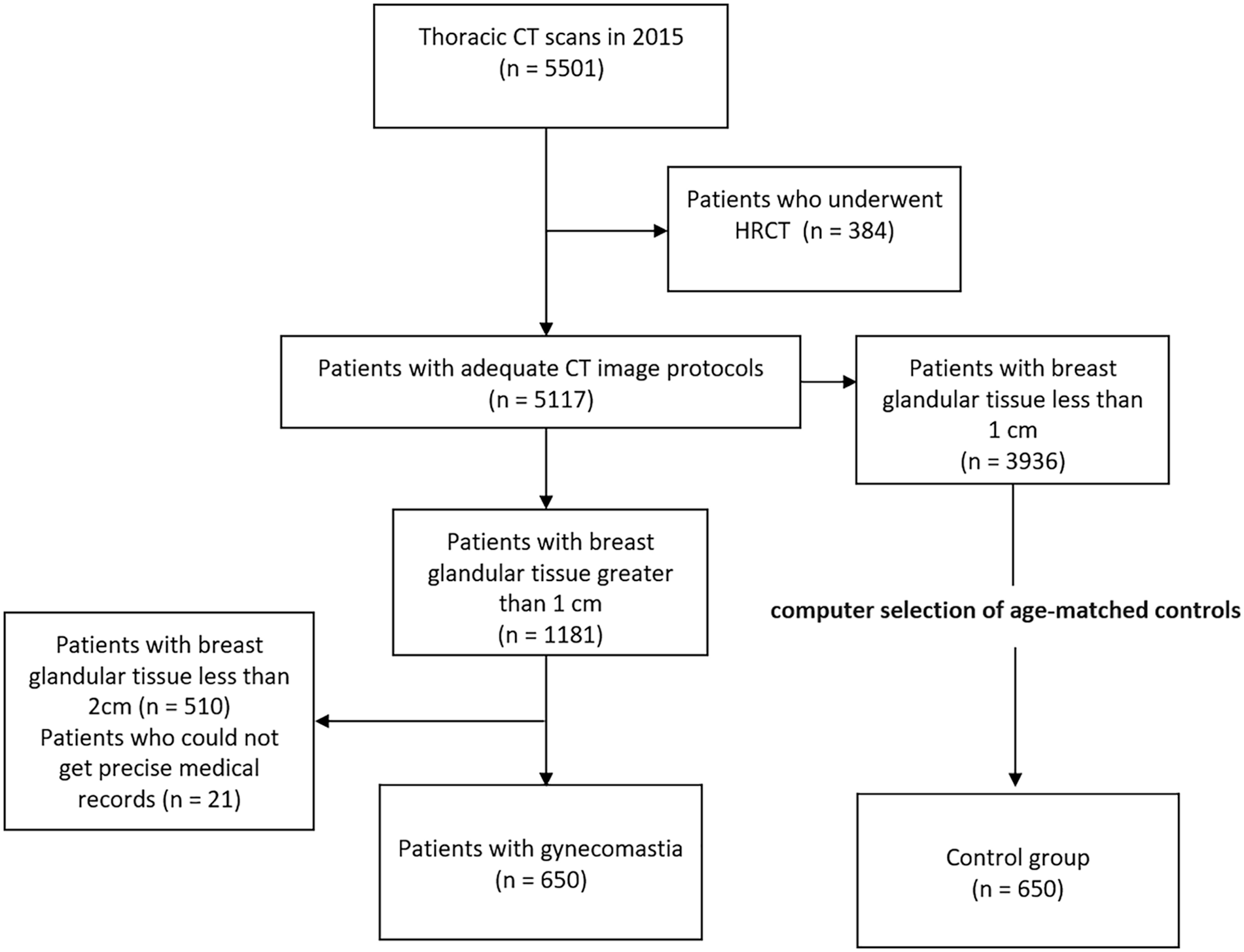
Flow chart of study inclusion and exclusion criteria. CT = computed tomography; HRCT = high-resolution CT.
Clinical Characteristics of Gynecomastia and Possible Causes.

Note. BMI = body mass index; CT = computed tomography; OR = odds ratio.
Multivariable logistic regression with age adjustment.
Six hundred and fifty patients without gynecomastia were selected as a control group with breast glandular tissue less than 1cm. The mean age of the control group was 56.74 years (±16.23, range 11–96). A few neonates were included in the study (five patients). Only one patient had breast glandular tissue larger than 1 cm (Table 3). However, the other four patients revealed prominent breast glandular tissues with a minimum of 0.5 cm and a maximum of 0.9 cm on both sides, all of which were the nodular type. The age distributions of gynecomastia and breast glandular tissue ≥1 cm are summarized in Table 3 (Figure 2 presents this data graphically). The highest increase in prevalence occurred among the over-90 age group. The second highest increase in prevalence was different between the two groups, occurring between 10 and 19 years in the gynecomastia group and 20 and 29 years in the ≥1 cm group. Between the ages of 30 and 69 years, the age distribution was flat and gradually increased, starting from over 70 years in both groups. A total of 2,277 lesions with breast glandular tissue >1 cm (mean diameter ± standard deviation, 2.09 ± 0.90 cm; median, 2.5; interquartile range [IQR], 2.2–3.1; range, 1.0–9.2 cm) were included in the study with 3.7% (85 of 1,181) of unilateral involvement. Of these lesions, the nodular, dendritic, and diffuse types were 35.6%, 32.9%, and 31.5%, respectively, based on CT. A total of 1,061 lesions were included in the study with breast glandular tissue ≥2 cm (mean diameter ± standard deviation, 2.76 ± 0.90 cm; median, 2.5; IQR, 2.2–3.1; range, 2.0–9.2 cm). Two hundred and thirty-nine of the 650 patients (36.8%) had unilateral gynecomastia. Of these lesions, the nodular, dendritic, and diffuse types were 2.5%, 36.5%, and 61.1%, respectively (Table 3). The breast glandular tissue size was significantly larger in the dendritic type than in the nodular type and in the diffuse type than in the dendritic type. The grades of CLD and CKD were significantly associated with breast glandular tissue size ≥1 cm (Spearman correlation coefficient [rho]; p = .002 and .006, respectively).
Age Distribution of Gynecomastia.


Prevalence according to age by decades.
The most common indication for a thoracic CT scan was pulmonary disease (250 of 650, 38.5%), followed by health checkup (172 of 650, 26.5%). Pulmonary disease and HCC were significantly more frequent indications for CT scan in the patient group than in the control group, and other malignancy (excluding HCC and lymphoma) and health checkup were significantly less frequent indications on multivariable analysis (Table 2).
CLD (p < .001), CKD (p < .001), and medication (p = .002) were associated with the presence of gynecomastia on univariable analysis. The presence of gynecomastia was not statistically associated with fatty liver, alcoholism, adrenal tumor, liver metastasis, HIV infection, thyrotoxicosis, germ cell tumor, or Klinefelter syndrome. LC among CLD (p = .002), all grades of CKD (G1; p = .003, G2; p = .017, G3; p = .013, and G4; p = .001), and antipsychotic drugs (p = .012) was associated with gynecomastia on multivariable regression analysis adjusted for patient age. There was no clear trend in the presence of gynecomastia according to BMI groups (p = .962). Table 4 presents trends in risk factors stratified by age. The prevalence of overweight or obesity (p = .002), CKD (p < .001), CLD (p = .023), and medications (p < .001) was significantly higher in older patients; however, idiopathic (p < .001) gynecomastia revealed a decreasing trend (p = .02). The grades of CLD and CKD were significantly associated with increasing breast glandular tissue size using the Spearman correlation coefficient (rho; p = .002 and p = .006, respectively). The correlation of BMI and size of breast tissue was not significant based on the Spearman correlation coefficient (rho; p = .068).
Characteristics of the Breast Glandular Tissue.
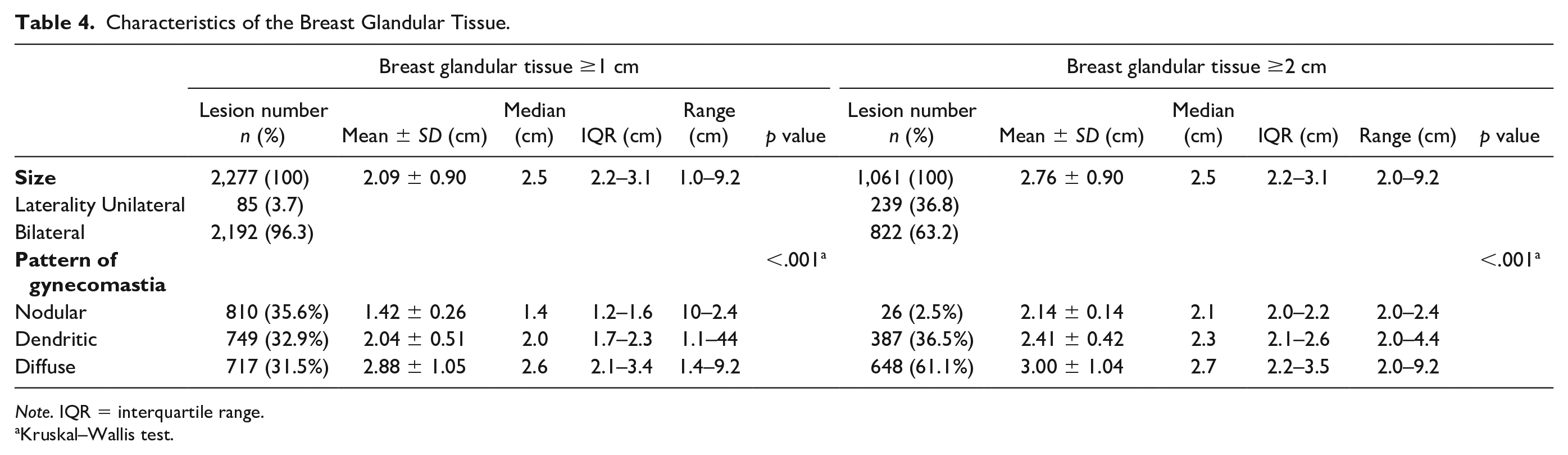
Note. IQR = interquartile range.
Kruskal–Wallis test.
Two of the 5,117 patients had mastectomy for breast cancer. They underwent regular follow-up with breast USs every year, with no gynecomastia in the contralateral breast. During 4 years of follow-up, only 4 of the 650 patients with gynecomastia underwent further imaging; 1 of them underwent both MG and US and 3 patients underwent only US. All of them were cancer patients and underwent further imaging workup with complaints of a palpable mass. Their mean age was 74.5 ± 9.11 years. The mean size of the breast glandular tissue was 2.88 ± 0.62 and the tissue was either of a dendritic or a diffuse type. One patient taking goserelin was diagnosed with gynecomastia as a side effect of the medication but was not checked with further imaging.
Discussion
Gynecomastia is defined as the benign proliferation of the ductal epithelium in the retroareolar region (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Nuttall, 2010). The pathophysiology of gynecomastia involves an imbalance in estrogen action relative to androgen action at the level of the breast tissue (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Nuttall, 2010). Gynecomastia is usually idiopathic (physiologic) but can be secondary to the effects of certain medications and systemic diseases (Braunstein, 2007; Cuhaci et al., 2014; Deepinder & Braunstein, 2012; He et al., 2019; Johnson & Murad, 2009; Mieritz et al., 2017; Nuttall, 2010; Nuttall et al., 2015; Polat et al., 2019; Sansone et al., 2017). Patients with suspected gynecomastia are usually diagnosed by physical examination (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Nuttall, 2010) or, if necessary, may be referred for confirmatory imaging with MG or US (Appelbaum et al., 1999; Chen & Slanetz, 2014; Iuanow et al., 2011; Madhukar & Chetlen, 2013; Tangerud et al., 2016). CT is not usually used as a diagnostic for gynecomastia. However, with the increased use of CT imaging for a variety of other conditions, incidental gynecomastia is frequently detected (Gossner, 2019; Klang et al., 2017, 2018; Sonnenblick et al., 2016; Yi et al., 2008).
The prevalence of gynecomastia varies widely depending on the population studied, specific imaging modality, and diagnostic criteria applied, ranging from 32% to 65% (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; Niewoehner & Nuttal, 1984; Nuttall, 1979; Williams, 1963), and earlier studies yielded a much higher prevalence than this study. Most of the data on the prevalence of gynecomastia have been derived from physical examination or autopsy (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; Niewoehner & Nuttal, 1984; Nuttall, 1979; Williams, 1963). The prevalence of gynecomastia was reported to be around 40% in an unselected autopsy series consisting of 447 cases (Williams, 1963). The prevalence of gynecomastia, based on thoracic CT, is rarely reported. One recent study reported a prevalence of 25.6% in a smaller cohort of hospitalized men (n = 82) undergoing thoracic CT (Gossner, 2019). In this study, gynecomastia was present in 12.7% of the study population, which is considerably lower than that seen in previous studies (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; Gossner, 2019; Niewoehner & Nuttal, 1984; Nuttall 1979; Williams, 1963). The discrepancy might be for the following reasons: (a) The study population in the present study were mostly Asian and included a significant proportion (45.6%); (b) only patients with actual breast glandular tissue on the CT were included in the subject group. Studies based on physical examination did not directly identify breast glandular tissue; pseudogynecomastia may sometimes be misdiagnosed as gynecomastia, which may result in overestimation of prevalence; and (c) differences in imaging protocol or modality may affect the results.
Asymptomatic gynecomastia is widespread and has a trimodal age distribution, occurring in neonatal (60%–90%), pubertal (50%–60%), and elderly males (up to 70%; Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; McKiernan & Hull, 1981; Niewoehner & Nuttal, 1984; Nordt & DiVasta, 2008; Nuttall 1979; Williams, 1963). Maternal estrogen is known to cause varying degrees of gynecomastia in newborns (Cuhaci et al., 2014). The number of neonatal patients in this study was very few. Thus, discussing the prevalence of neonatal gynecomastia is not meaningful for this study. However, all five neonatal patients had bilateral breast glandular tissue of the nodular type, although most of them were smaller than 1 cm. Usually the size of the breast grows to 1–2 cm in the first few weeks of life (Jayasinghe et al., 2010). In newborns with a much smaller body size than adults, it is difficult to apply the same standards. Further studies are needed to define the cutoff value of image-based gynecomastia in neonates. The present study demonstrates a high prevalence of gynecomastia in older patients over 70 years and adolescents from 10 to 19 years old. These findings corroborate those of previous studies (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; McKiernan & Hull, 1981; Niewoehner & Nuttal, 1984; Nordt & DiVasta, 2008; Nuttall 1979; Williams, 1963). With increasing age, the slope of prevalence of gynecomastia increased more steeply than the prevalence of the breast glandular tissue more than 1 cm (Figure 2), probably due to a higher proportion of patients with chronic diseases such as CKD or LC or those who take multiple medications (Table 5). The prevalence of breast glandular tissue in patients 20–29 years old is as high as that in the adolescent group, which is a unique result regardless of the health checkup group or inpatient/outpatient group status (Table 3). Even when the cutoff value of breast glandular tissue was 1 cm, the prevalence was slightly higher than that of patients 10–19 years old. According to a study by Costanzo et al., among 237 men 18–85 years old with gynecomastia, those 21–30 years old accounted for 31.2% of the total and had the highest prevalence (Costanzo et al., 2018). Although the study population is different, this finding is similar to the results from the present study, suggesting that gynecomastia in young adults is more common than previously known. Gynecomastia in young adult males is either idiopathic or persistent pubertal gynecomastia. Idiopathic gynecomastia is closely correlated with generalized obesity and reduced LH and testosterone levels, which may be the result of increased conversion of testosterone to estradiol in increased adipose tissue mass (Costanzo et al., 2018; Kurtoglu & Tor, 2002).
Possible Causes of Gynecomastia by Age.

Note. CKD = chronic kidney disease; FL= fatty liver; Liver meta = liver metastasis; GCT = germ cell tumor; Mx = medication.
Statistically significant increase. bStatistically significant decrease. cNumber of patients with two or more risk factors.
Multivariable analysis revealed that CLD, CKD, and medications were significantly associated with gynecomastia. In another previous CT-based study (Klang et al., 2017), LC and end-stage renal disease were significantly associated with gynecomastia. Although not precisely the same, both CKD and CLD are associated with both gonadal and a hypothalamic–pituitary dysfunction leading to a decrease in the total plasma testosterone levels (Carrero et al., 2011; Cuhaci et al., 2014; Edey, 2017; Foresta et al., 2008; Iglesias et al., 2012; Schmidt et al., 2002). The development of hypogonadism depends on the severity of liver disease (Foresta et al., 2008). In the present study, sex hormone levels in patients with gynecomastia could not be measured due to the retrospective nature of the study. Nevertheless, gynecomastia is one of the findings suggestive of hypogonadism in patients with LC (Foresta et al., 2008). Therefore, gynecomastia could develop in the advanced stage of CLD. The current study demonstrates that gynecomastia was not statistically associated with early CLD but was with LC. In contrast to CLD, changes in androgen synthesis and metabolism are known to develop “early” after the onset of renal insufficiency (Iglesias et al., 2012). However, no study has researched which stage of CKD specifically defines “early.” The present study demonstrates that all stages of CKD were associated with gynecomastia. These findings suggested that gynecomastia might be associated with gonadal dysfunction from the very early stages of CKD. In both diseases, disease progression was related to the increase in the size of breast glandular tissue with a tendency for the progression to increase with age. This might be a natural outcome, given the chronic nature of both diseases.
Gynecomastia can be caused by a variety of medications via different mechanisms (Deepinder & Braunstein, 2012; He et al., 2019; Nuttall et al., 2015). Medications account for 10%–25% of the causes of gynecomastia (Deepinder & Braunstein, 2012). Multivariable analysis in this study provided a significant association between antipsychotic drugs and gynecomastia. Antipsychotic drugs are known to be associated with gynecomastia (Deepinder & Braunstein, 2012; Grigg et al., 2017; Storch, 1997). There is insufficient evidence because there are no extensive studies and only a few case reports have been documented (Deepinder & Braunstein, 2012). The exact pathophysiology also remains unknown. Spironolactone, antiandrogens, gonadotropin-releasing hormone analogs, 5α-reductase inhibitors, α1-adrenergic receptor, and antiulcer drugs are medications definitely associated with the onset of gynecomastia (Deepinder & Braunstein, 2012; Nuttall et al., 2015). The androgen receptor antagonists or blockers interfere with the biosynthesis, metabolism, or the action of endogenous androgens (Deepinder & Braunstein, 2012; Nuttall et al., 2015). None of these were associated with gynecomastia upon multivariable analysis. These results may be due to the small number of patients and because the duration or dose of the medication was not considered. Chemotherapeutic agents, calcium channel blockers, angiotensin converting enzyme inhibitors, opioids, and anabolic steroids are likely associated with gynecomastia, but not definitely (Deepinder & Braunstein, 2012; Nuttall et al., 2015). In the present study, these drugs were not associated with gynecomastia.
No previous study has implicated fatty liver as one of the causes of gynecomastia. Fatty liver was included in the assessment because it represents a spectrum that can lead to progressive nonalcoholic steatohepatitis, fibrosis, and ultimately LC (Hardy et al., 2016). In this study, fatty liver before the chronic stage was not statistically associated with gynecomastia.
Excessive alcohol consumption is known to be one of the causes of gynecomastia. Alcohol abuse can disrupt testicular enzyme activity and the hypothalamic–pituitary–testicular axis, resulting in secondary hypogonadism with damage to the testes itself (Bell et al., 1990; Braunstein, 2007; Chiao et al., 1981; Cuhaci et al., 2014; Deepinder & Braunstein, 2012; Gavaler & Van Thiel, 1988; Johnson & Murad, 2009; Rachdaoui & Sarkar, 2017). In contrast, feminization including gynecomastia occurs late in the natural progression of alcohol abuse and only if LC has developed (Gavaler & Van Thiel, 1988; Van Thiel, 1979). The present study demonstrates that alcohol abuse was not correlated with gynecomastia and this finding supports previous studies (Gavaler & Van Thiel, 1988; Van Thiel, 1979). Adrenal tumor, liver metastasis, HIV, thyrotoxicosis, germ cell tumor, and Klinefelter syndrome were not statistically associated with gynecomastia. No definitive conclusion could be drawn because of the small number of patients.
Earlier research has reported the association between BMI and gynecomastia in adolescents and young adults (Georgiadis et al., 1994; Johnson & Murad, 2009; Niewoehner & Nuttal, 1984; Rosen et al., 2010; Yazici et al., 2010). However, in the most recent of those studies, pubertal gynecomastia was not associated with body fat percentage but with the growth hormone–insulin-like growth factor axis (Mieritz et al., 2014). In adults, obesity may cause mild hypogonadism due to insulin-resistance-associated reductions in sex-hormone-binding globulin and suppression of the hypothalamic–pituitary–thyroid axis. (Cuhaci et al., 2014; Fui et al., 2014; Johnson & Murad, 2009; Santen et al., 2009). However, studies aimed at determining whether obesity-induced hypogonadism is severe enough to cause gynecomastia are lacking. A recent CT-based study (Klang et al., 2017) has reported that obesity (BMI and subcutaneous fat measurements) does not significantly contribute to gynecomastia. Similarly, the current study demonstrates that there was no statistical correlation between BMI and gynecomastia, and BMI and the size of breast tissue. Moreover, as opposed to gynecomastia, obesity tended to decrease with age in the patient group. As already mentioned, subareolar fat and true breast glandular tissue can be distinguished by a physical examination in patients with breast enlargement to a limit. There is a possibility that BMI may have been mistaken for one of the causes of gynecomastia due to pseudogynecomastia misdiagnosed as gynecomastia.
The distribution of the breast glandular tissue in patients with gynecomastia centered around 2.5 cm with diameters of 2.3 cm and 3.1 cm representing the 25th and 75th percentiles, respectively. Most often, gynecomastia is bilateral, although it may sometimes be unilateral (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009; Nuttall, 2010; Sansone et al., 2017). Two hundred and thirty-nine of the 650 patients (36.8%) had unilateral gynecomastia in the study population. This is much higher than that seen in other research. The prevalence of unilateral gynecomastia in patients was 14% (8 of 55) in a previous MG-based study (Appelbaum et al., 1999) and 5% (1 of 21) and 19% (11 of 59) in CT-based studies (Gossner, 2019; Sonnenblick et al., 2016). However, reducing the baseline for breast glandular tissue to 1 cm lowered the proportion of patients with unilateral gynecomastia to 7.5% (85 of 1,181), similar to that seen in previous studies (Appelbaum et al., 1999; Gossner, 2019; Sonnenblick et al., 2016). Three mammographic patterns of gynecomastia have been described: nodular, dendritic, and diffuse (Appelbaum et al., 1999). The nodular pattern is thought to be the acute phase of gynecomastia (less than 1 year). It correlates with the pathologic classification of florid gynecomastia, which is characterized by hyperplasia of the intraductal epithelium with loose cellular stroma and surrounding edema. The dendritic pattern correlates with the pathologic classification of fibrous gynecomastia, which is thought to occur over a prolonged period. In this phase, fibrosis becomes the dominant process and is irreversible. Diffuse glandular gynecomastia is similar to a heterogeneously dense female breast (Appelbaum et al., 1999; Chen & Slanetz, 2014; Iuanow et al., 2011). The CT patterns of gynecomastia are not well described, but a recent study suggested that the appearance of gynecomastia on CT scans and mammograms was well correlated (Sonnenblick et al., 2016). The present study demonstrates that there were few nodular types in patients with glandular tissue larger than 2 cm. In patients with glandular tissue larger than 1 cm, the size of the glandular tissue significantly increased in the order of the nodular, dendritic, and diffuse type. Given that the nodular type is a pathologically early form of gynecomastia and the dendritic or diffuse types are more chronic forms, an increase in size is expected.
Only four of the patients in the present study with CT-depicted gynecomastia underwent further imaging. Despite the presence of gynecomastia on thoracic CT, very few patients received proper evaluation or management. Gynecomastia can lead to an increase in anxiety and cause cosmetic issues (Braunstein, 2007; Cuhaci et al., 2014; Johnson & Murad, 2009). All the patients who received further imaging in this study were cancer patients, which potentially increased the anxiety of these patients. In addition, the current study demonstrates that 81.5% of patients have one or more possible causes of gynecomastia, other than idiopathic gynecomastia, and older patients tended to have more than one possible cause (p < .001). Previous reports demonstrated considerable variation in gynecomastia, ranging from 43% to 95%, with a possible underlying cause (Ikard et al., 2011; Klang et al., 2018; Mieritz et al., 2017). Based on those findings, it is assumed that the prevalence of gynecomastia is higher in the inpatient/outpatient group than in the health checkup group, which is consistent with the results of this study except for patients aged 20–29 years (Table 3). Gynecomastia may be correlated with relevant underlying clinical conditions, which highlights the need to identify idiopathic gynecomastia and provide appropriate clinical and laboratory assessment of these patients, which is essential to avoid unnecessary tests and relieve patient anxiety.
Study Limitations
This study had several limitations. First, this was a retrospective and single-center study that was performed in a large tertiary hospital and may not be reflective of patients in community settings. Further, this study did not include patients with detailed diagnostic workup for gynecomastia. However, the retrospective study design had advantages in that the formation of a large-scale cohort was possible. Future prospective studies involving a larger diverse community setting would be helpful to validate the findings of the present study. Second, this study lacks a correlation with patients’ symptoms or physical examinations. Through the present study, a large number of undiagnosed gynecomastia were found. However, this study suggested that the various appearances of gynecomastia on thoracic CT are enough so that unnecessary additional breast imaging can be avoided. Third, because the size of breast glandular tissue was measured in only the axial image, volume information may not be reflected. Although volume is more accurate than diameter measurement, most studies do not suggest diagnostic criteria for volume (Gossner, 2019; Klang et al., 2017, 2018; Sonnenblick et al., 2016) and a strong correlation between diameter and volume was observed. It would be most reasonable to measure the glandular tissue in the axial image, because the length in the axial image is the largest. Fourth, BMI was used to investigate the correlation between obesity and gynecomastia. However, on average, Asians have a higher percentage of body fat than Westerners of the same age, sex, and BMI. Moreover, because of the controversy over appropriate BMI cutoff values for Asians, overweight and obesity according to WHO standards were divided in the present study (WHO Expert Consultation, 2004). These facts may have led to an underestimation of patients’ total body fat and an incorrect correlation between gynecomastia and body fat. Further studies may be necessary to conclude a causal association between total body fat and gynecomastia with more objective indicators such as dual-energy X-ray absorptiometry and CT-based measurement of body fat. Fifth, this study investigated the association between gynecomastia and specific drugs reported in previous studies. However, there are newer drugs available now that were not used in the past and these are not reflected in this study. Sixth, some studies regarding both the prevalence and possible causes of gynecomastia have been published between the 1960s and 1990s (Andersen & Gram, 1982; Carlson, 1980; Georgiadis et al., 1994; McKiernan & Hull, 1981; Niewoehner & Nuttal, 1984; Nuttall 1979; Williams, 1963). Changes such as patients with chronic diseases living longer could affect the prevalence of gynecomastia. In addition, the present study’s population included patients examined for routine health checkups, yet routine testing is not performed in all countries. Also, most of the patients in this study, undergoing such screening, might be older and healthier than groups with other indications, which might affect the analysis of prevalence and etiologic factors. Finally, the intraobserver and interobserver variability of breast glandular tissue measurement was not calculated. However, a simple method to measure the breast glandular tissue was used in this study and it was identified to be highly reproducible.
Conclusion
As the widespread use of thoracic CT with variable indications increases, it is increasingly likely that gynecomastia is incidentally found in patients. This study determined the prevalence of incidental gynecomastia detected on CT, and the morphologic features, in a large population. LC, CKD, and some medications are possible causes of gynecomastia. Recognizing these findings helps clinicians understand the etiology of gynecomastia and guide the management of this disease by understanding treatable underlying causes. CT-depicted gynecomastia is not associated with obesity, but studies with large cohorts may be required to delineate the true association between gynecomastia in adolescents and obesity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Inha University Hospital Research Grant (2019). The study sponsor had no involvement in the conduct of the study or writing of the article.
