Abstract
Previous studies have reported an enhancement of central cholinergic signal cascade by shilajit. For the present study, it was hypothesized that parasympathomimetic effect of shilajit accounting for relaxation of rat corpus cavernosum may be one of the major mechanisms attributing to its traditional role as an aphrodisiac. To test this hypothesis, the acute peripheral effect of standard acetylcholine (ACh), shilajit, and their combination was evaluated on cardiorespiratory parameters such as mean arterial blood pressure (MABP), heart rate (HR), respiratory rate (RR), and neuromuscular transmission (NMT). Furthermore, in vitro effect of standard ACh, shilajit, and their combination was tested on the rat corpus cavernosum. Six groups were used for the in vivo study (N = 5): Group I (control-saline), Group II (ACh), Group III (Sh), Group IV (Sh followed by ACh), Group V (Atropine followed by ACh), and Group VI (Atropine followed by Sh). The in vitro study included four groups: Group I (control-saline), Group II (ACh), Group III (Sh), and Group IV (Sh followed by ACh). The results of the in vivo study confirmed the peripheral parasympathomimetic effect of shilajit (400 µg/mL). The in vitro results revealed that shilajit (400 and 800 µg/mL) relaxed cavernous strips’ concentration dependently and enhanced ACh-mediated relaxations. The peripheral parasympathomimetic effects of shilajit were confirmed by blockade of shilajit-induced relaxations (in vitro) and shilajit-induced lowering of MABP and HR (in vivo) by atropine.
From prehistoric times, sexuality has been considered as a cornerstone for quality life, and man has always been on a quest for a remedy that can enhance sexual function and/or treat erectile dysfunction (ED). ED is defined as the inability to attain and/or maintain penile erection sufficient for satisfactory sexual performance (NIH Consensus Conference, 1993) and adversely affects the quality of life (Althof, 2002). ED is a multifaceted disorder involving cardiovascular, metabolic, psychological, neurological, and hormonal factors affecting more than 100 million men worldwide (Hannan, Maio, Komolova, & Adams, 2009).
Longevity and vitality, two important hallmarks of rasayana and vajikarana therapy of ayurveda, prevent ageing and diseases and enhance sexual vigor in men (Sharma & Dash, 2000). As per the literature, a person devoid of sexual potency regains vitality through vajikarana therapies, and it makes a person capable of performing sex like a strong horse even in old age (Sharma & Dash, 2000). Beautiful woman, nature, fragrant flowers, garlands and ornaments, comfort, and pleasing sounds are considered vaajikarans (Sharma & Dash, 2000). Predisposing men to these vaajikarans seems to reduce physical and psychological stress, enhancing sexual motivation. It is well established that during stress and fear, activation of sympathetic nervous system inhibits penile erection by enhancing the contractile tone of penis, whereas during mental relaxation, activation of parasympathetic nerves elicits erection by enhancing the relaxant tone (E. D. Andersson & Wagner, 1995; Simonsen, Garcia-Sacristan, & Prieto, 2002). Several epidemiological studies suggest the involvement of psychological stress in a number of cases of ED, either alone or in combination with organic causes (Ahn et al., 2007; Araujo, Durante, Feldman, Goldstein, & McKinlay, 1998; Melman & Gingell, 1999; Quek, Sallam, Ng, & Chua, 2008). Several rasayana and vaajikaran drugs such as shilajit, Withania somnifera, Tinospora cordifolia, Tribulus terrestris, Asparagus racemosus, Sida cordifolia, Mucuna pruriens, and so on, mentioned in Ayurvedic texts, are deemed to enhance longevity, sexual strength, and potency in men (Sharma & Dash, 2000).
Shilajit, a pale-brown to blackish-brown exudation of variable consistencies, found worldwide in sedimentary rocks at altitudes between 1,000 and 5,000 m by the unique interaction of marine invertebrate fossils/plants, land plants, and microorganisms, is a multicomponent naturally occurring mineral substance referred to as rasayana in ayurveda and prevents ailments and enhances quality of life (Ghosal, 1990, 2006a, 2006b; Wilson et al., 2011). In ayurveda (Charak Samhita, Susruta Samhita, Rasaratna Samucchya, Bhava Prakash), as quoted by Chopra, Chopra, Handa, and Kapoor (1958) and Tiwari, Tiwari, and Joshi (1973), shilajit is claimed to be a maharasa and is recommended for the treatment of diabetes, anemia, ulcers, bronchial asthma, gastrointestinal infections, liver diseases, geriatric problems, sexual dysfunctions, and in wound healing. Traditionally used for its rejuvenating and aphrodisiac effects, shilajit enhances sexual strength and vitality in men (Sharma & Dash, 2000).
An earlier histochemical study has suggested an enhan cement of the cholinergic signal transduction cascade in cortical and basal forebrain, which accounts for the memory enhancing effects of shilajit (Schliebs et al., 1997). For the present study, it was hypothesized that one of the ways by which shilajit can be effective against ED is by its parasympathomimetic effects, because the activation of parasympathetic nerves to the penis contribute to the relaxation of blood vessels and corpus cavernosum smooth muscles, leading to erection (Simonsen et al., 2002). To test this hypothesis, the acute effect of standard acetylcholine (ACh) and the test drug, shilajit, as well as their combination was evaluated on cardiorespiratory parameters such as mean arterial blood pressure (MABP), heart rate (HR), respiratory rate (RR), and neuromuscular transmission (NMT). Parameters such as MABR, HR, and RR are potentiated by sympathetic activation, whereas they are depressed by parasympathetic activation, and NMT is potentiated by parasympathetic activation. This in vivo experiment provided us a preliminary assumption regarding the peripheral parasympathomimetic effects of shilajit. Furthermore, to justify whether parasympathomimetic effects of shilajit account for relaxation of rat corpus cavernosum or not, the in vitro effects of standard ACh, test drug shilajit, and a combination of ACh and shilajit was evaluated on rat corpus cavernosum in tissue organ bath.
Materials and Method
Animals
Sexually matured male Wistar albino rats (300-350 g) randomly bred and maintained in the Animal Facility Division, Defence Research and Development Establishment, were used for the study. The animals were housed in polypropylene cages on dust-free and steam-autoclaved rice husk as the bedding material and were provided with pellet diet (Ashirwad Brand, India) and water ad libitum. The animals were maintained in a 12-hour light/dark cycle with ambient temperature regulation at 23 ± 2°C. The study was approved by the Institutional Ethical Committee (IAEC No. 01/10/87) of Dr. H. S. Gour University, Sagar, India. The quarantine procedures and animal maintenance were according to the recommendations of the Canadian Council Guide to the Care and Use of Experimental Animals (Olfert, Cross, & McWilliam, 1993) and the Committee for the Purpose of Supervision and Control of Experiments on Animals (India).
Chemicals
A standardized extract of shilajit supplied by Indian Herbs Ltd, Saharanpur, India, constituting 0.4% to 1% w/w, oxygenated dibenzo-α-pyrones, and 65% to 70%, w/w low- to medium-molecular-weight fulvic acids were used for the study (U.S. Patent No. US 6,869,612 B2). The constituents for the Krebs solution (NaCl, 118 mM; KCl, 4.5 mM; KH2PO4, 1 mM; NaHCO3, 25 mM; glucose, 6 mM; CaCl2, 2.5 mM; MgSO4, 1 mM) were purchased from Merck India Ltd. (Mumbai). Noradrenaline (NAd), ACh, atropine sulfate, and sodium nitroprusside (SNP) were purchased from Sigma-Aldrich (St. Louis, MO). All the drugs/chemicals were freshly prepared by dissolving in distilled water.
Regimen to Animals
In vivo study on cardiorespiratory parameters and NMT in anesthetized rats
Thirty sexually matured male Wistar rats were randomized into six groups of five animals each: Group I, 0.9% saline (control); Group II, ACh at concentrations of 1, 2, and 4 µg/mL; Group III, shilajit at concentrations of 100, 200, and 400 µg/mL; Group IV, shilajit at a concentration of 400 µg/mL, followed by successive concentration of ACh (1, 2, and 4 µg/mL); Group V, atropine (10 mg/kg) followed by successive concentration of ACh (1, 2, and 4 µg/mL); Group VI, atropine (10 mg/kg) followed by successive concentration of shilajit (100, 200, and 400 µg/mL). The animals were allowed a stabilization period of 15 minutes between successive treatments in each group.
In vitro studies in tissue organ bath
Twenty sexually matured male rats were randomized into four groups of five animals each as follows: Group I, control (distilled water); Group II, standard ACh (10−8 to 10−4 M); Group III, shilajit (100, 200, 400, and 800 µg/mL); Group IV, shilajit (400 µg/mL) followed after 10 minutes by ACh (10−8 to 10−4 M). At least 2 minutes were allowed between successive doses, to establish a stable response at each concentration. After washout and another 45 minutes of equilibration, atropine hydrochloride (10−6 M) was added at stable tension, and at the end of each experiment, maximum relaxation was induced with SNP (10−4 M).
Procedure
For recording the effects of ACh and shilajit on cardiorespiratory parameters and NMT in male rats
MABP, HR, RR, and NMT were recorded using an 8-channel polygraph (model 7D, Grass Instruments, West Warwick, RI). The rats were anesthetized with urethane (1.6 gm/kg i.p.), and the neck region was dissected and exposed. Trachea was cannulated and connected to a pneumotachograph (Hugo Sachs Electronic, Hugstetten, Germany) which was connected to a Grass PT-5 differential transducer and connected to a preamplifier (low-level DC; Grass Instruments), and inspiration was recorded as an upward deflection and expiration as a downward deflection. Right jugular vein was cannulated for intravenous administration of dose. Left carotid artery was cannulated with a thin polypropylene tube connected to a pressure transducer (Statham P-23 DC), filled with heparinized normal saline. The pressure transducer was connected to a preamplifier (low-level DC; Grass instruments) and arterial blood pressure was recorded on the polygraph. Mean arterial pressure was calculated from the recorded blood pressure. The signals from the DC driver amplifier recording blood pressure were taken from J6 output and fed into the external triggering (high) input of the EKG Tachograph preamplifier (Grass Instruments) for recording HR. The gastrocnemius muscle and sciatic nerve were dissected and opened. The distal end of the muscle was cut and connected to a transducer (FT03). The sciatic nerve was lifted and bipolar silver electrode was placed. The nerve was stimulated with supramaximal voltage (1-10 V) of 0.2 ms duration at a frequency of 0.2 Hz using a Grass Stimulator (Model S-88). The resultant muscle twitch responses were recorded. The animals were allowed to stabilize for 60 minutes after the surgical procedures following which MABP, HR, RR, and NMT were recorded.
In vitro preparation for recording the effect of ACh and shilajit on contraction and relaxation of corpus cavernosum from rat penis
The method described by Cartledge, Minhas, Eardley, and Morrison (2000) was followed for the isolation of strips of corpora cavernosa from rat penis. Briefly, rats were sacrificed by cervical dislocation and the penis was rapidly dissected and placed in oxygenated Krebs solution at 4°C. With the aid of a 12× binocular dissecting microscope, loose connective tissue and adventitia were removed. All skeletal muscles were removed from the proximal crura to expose the corpus spongiosum surrounding the urethra. The corpus spongiosum was reflected off the underlying tunica albuginea in a proximal to distal direction before division at the base of the glans penis. The penis was then turned over to expose the dorsal venous complex, which was also excised. Sharp incision of the thick tunica albuginea of the crus penis exposed the underlying corpora cavernosa. A strip of corpus cavernosum was dissected by separating it from the thick, midline, tunica albuginea, leaving the lateral, thin, tunica intact and adherent to the corpus. This process was repeated on the contralateral side to produce two identical strips with dimensions of approximately 3 × 3 × 12 mm.
For all in vitro experiments, the strips of corpora cavernosa were attached between a fixed point by a loose silk suture and an isometric force transducer (Grass FT03) by a hooked glass rod. The signal from the force transducer was passed through a bridge amplifier (Grass Instruments). At the beginning of each experiment, the bridge amplifier was balanced and the pen recorder was calibrated so that active tension between 0 and 500 mg could be recorded following the application of optimal resting tension of 1 g. The strip was suspended in an organ bath containing Krebs solution gassed with 95% O2/5% CO2 to maintain pH 7.4. A temperature of 37°C was maintained in the organ bath by circulating water through a surrounding jacket. Tissue strips were allowed to equilibrate for 90 minutes at a resting tension of 1 g. Krebs solution within the organ bath was exchanged completely every 15 minutes during equilibration. Whenever washout of the organ bath was performed it drew warmed, aerated Krebs solution from the stock reservoir. Following the equilibration period of 90 minutes, strips developed oscillating spontaneous activity at a mean tension of 50 mg. Strips were then precontracted with NAd (10−6 M), and the strips that reached a maximal tension of less than 300 mg were excluded from the study. ACh was added cumulatively in intervals to allow tension to reach a stable plateau. Isometric tension was recorded continuously. After washout and another 45 minutes of equilibration, NAd (10−6 M) was added as described above. Later, atropine hydrochloride (10−6 M) was added at stable tension. At the end of each experiment, relaxation was induced with SNP (10−4 M). Reliability of all the instruments were ensured by using conventional calibration procedures, in addition to the method described in “Instrument Operation Manuals.” Briefly, the pressure transducer (Statham P-23DC) was calibrated using a mercury manometer. The EKG Tachograph preamplifier was calibrated by generating mild pressure air pulses of known frequency using a rodent ventilator (UGO Basile, Model 7025, Comerio, Italy), connected with the pressure transducer. The same ventilator was also used to calibrate the pneumotachograph by adjusting and generating air pulses of known volume at different frequencies to closely mimic the rat’s respiration. Similarly, force displacement transducer (Grass FT 03) was calibrated by hanging weight of known value on its sensing probe. The same calibration procedures were randomly repeated after termination of a few experiments to doubly confirm instruments’ reliability.
Statistical Analysis
Data are expressed as mean ± SEM. All data were analyzed by one-way analysis of variance (ANOVA). If the ANOVA p value was <.05, multiple paired comparison were made using Student’s t test. Paired comparisons were considered to be significantly different if p ≤ .05. The statistical analyses were carried out using SigmaStat (IBM SPSS Inc., Chicago, IL).
Results
In Vivo Studies
The results for the in vivo studies are depicted in Table 1. During the 4-hour monitoring period, the control animals exhibited a steady response. In Group II, administration of ACh caused a dose-dependent decrease in MABP and HR at the successive doses of 1, 2, and 4 µg/mL of ACh. The depth of breathing and NMT increased at different doses of ACh (Figure 1). In Group III, although intravenous administration of shilajit exhibited no change in MABP, HR, depth of respiration, and NMT at doses of 100 and 200 µg/mL, but a dose of 400 µg/mL caused a significant decrease in the MABP, HR, and increase in depth of breathing (Hypernea) and NMT compared with the control (Figure 2). Pretreatment with shilajit (400 µg/mL) synergistically decreased MABP and HR and increased depth of breathing for ACh dose dependently at 1, 2, and 4 µg/mL in Group IV compared with Group II and the control group. However, NMT in Group IV remained unchanged. In Groups V and VI, following intravenous administration of atropine (10 mg/kg), responses for ACh and shilajit remained unchanged and was comparable with the control group.
Effect of ACh and Shilajit on Cardiorespiratory Parameters and Neuromuscular Transmission in Anesthetized Rats
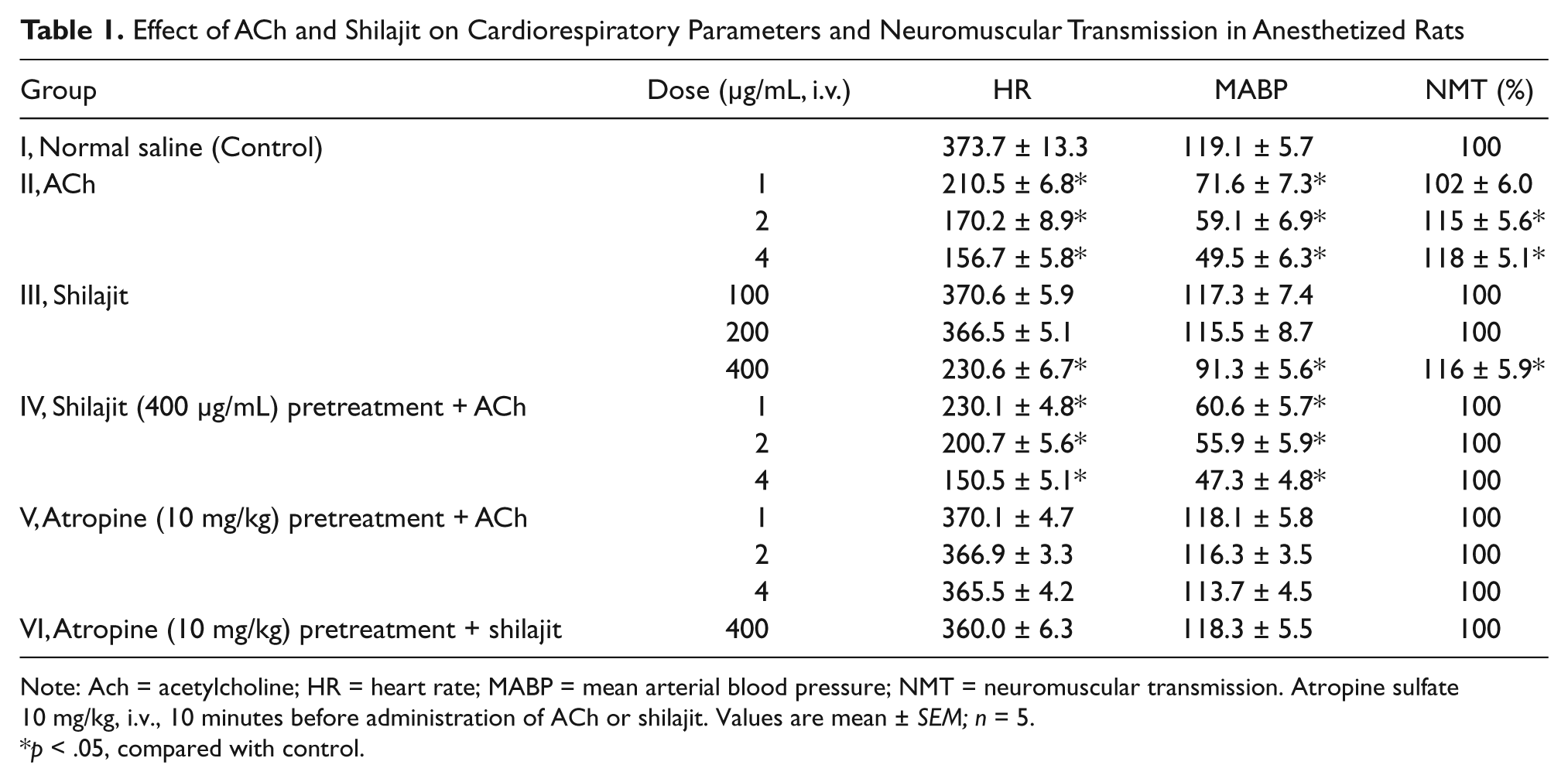
Note: Ach = acetylcholine; HR = heart rate; MABP = mean arterial blood pressure; NMT = neuromuscular transmission. Atropine sulfate 10 mg/kg, i.v., 10 minutes before administration of ACh or shilajit. Values are mean ± SEM; n = 5.
p < .05, compared with control.

Effect of standard ACh at successive doses of 1, 2, and 4 µg/mL on cardiorespiratory parameters and neuromuscular transmission

Effect of shilajit (400 µg/mL) on cardiorespiratory parameters and neuromuscular transmission
In Vitro Studies
On contraction of tissue with noradrenaline (NAd, 10−6 M), tension increased to 300 mg (Figures 3 and 4). Cumulative addition of ACh (10−8 to 10−4 M) reduced NAd-induced tension in a dose-dependent fashion to 51.6 ± 7.9% of the maximum (N = 5; Figures 3A and 6A). The effects of ACh were transient: fast initial relaxation was followed by contraction, and the result was tension almost comparable with that before addition of ACh (Figure 3A). Addition of shilajit at doses of 100 and 200 µg/mL did not reduce the tension of NAd-precontracted tissue strips; however, at doses of 400 and 800 µg/mL, shilajit reduced NAd-induced tension to 68.3 ± 8.9% of the maximum (Figures 4A and 6A). Atropine sulfate (10−6 M) increased NAd-induced tensions slightly to 109.5 ± 3.5% and almost abolished the effects of cumulatively applied ACh (Figures 3B and 6B) and shilajit (Figures 4B and 6A) in Group II and Group III, respectively. In Group IV, shilajit (400 µg/mL) pretreatment followed by cumulatively applied ACh (10−8 to 10−4 M) synergistically reduced the NAd-induced tension to 23.5 ± 5.7% of the maximum (Figures 5 and 6B).

Effects of ACh (10−8 to 10−4 M) on tension of NAd-precontracted rat cavernous smooth muscle strips (A) and after inhibition by incubation with atropine (B)

Effects of Shilajit (100, 200, 400, and 800 µg/mL) on tension of NAd-precontracted cavernous smooth muscle strips (A) and after inhibition with atropine (B)

The effects of ACh and shilajit combination on tension of NAd-precontracted cavernous smooth muscle strips
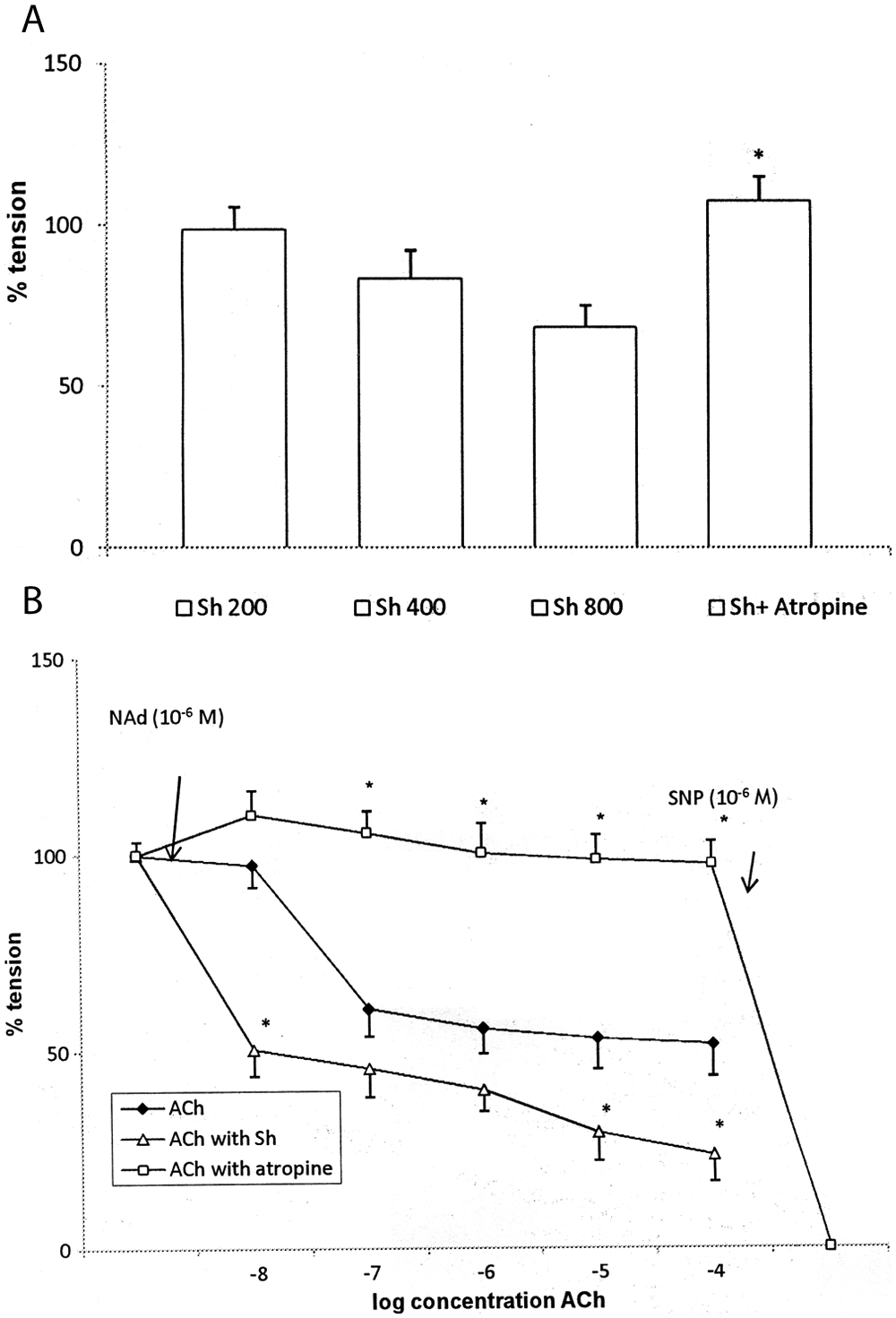
Effect of Shilajit (Sh) and ACh on tension of NAd-precontracted corpus cavernosum strips
Discussion
An earlier study has reported an enhancement of central cholinergic signal transduction by shilajit (Schliebs et al., 1997). As the stimulation of parasympathetic impulses from the sacral portion of spinal cord causes coordinated relaxation of the cavernous vessels and trabecular smooth muscles via the release of ACh and nitric oxide, enhancing the blood supply to the sinusoids, thus culminating in erection (Simonsen et al., 2002), the present research focused on evaluating the peripheral parasympathomimetic effects of shilajit. The results revealed parasympathomimetic effects of shilajit at a dose of 400 µg/mL. Comparable with standard ACh (1, 2, and 4 µg/mL), shilajit (400 µg/mL) also caused a decrease in MABP, HR, hypernea, and an increase in NMT. Lower doses of shilajit (100 and 200 µg/mL), however, did not change MABP, HR, RR, and NMT when compared with the control. Shilajit at a dose of 400 µg/mL also potentiated ACh-mediated lowering of MABP and HR and enhancement of depth of breathing. However, NMT remained unaltered. Pretreatment with atropine blocked the response of ACh and shilajit on the above-mentioned cardiorespiratory parameters and NMT, thereby confirming the parasympathetic effects of shilajit. As a sequel to this study, more in vitro studies were carried out to evaluate whether parasympathomimetic effects of shilajit account for relaxation of rat corpus cavernosum smooth muscle or not.
For the preparation of corpus cavernosum from rat penis, the dissection technique developed by Cartledge et al. (2000) was followed, which offered following three advantages: First, the cavernosal tissue specimen is covered by the minimum of tissue, allowing maximum penetration of drugs under experimental conditions. Second, it reduces the handling of cavernosal tissue, preventing the disruption of endothelium during tissue handling. Third, the cavernosal tissue strip mounted in vitro included the crus penis, which consist mainly of smooth muscle cells, whereas distally connective tissue predominates, with a reduced smooth muscle distribution. As the rat corpus cavernosum differs from that of humans by lesser amounts of smooth muscle cells and greater amounts of collagen (Pinheiro, Costa, Cardoso, & Sampaio, 2000), to minimize the differences in studies on erection with rat penis, rat crus was included in the tissue specimen. It is well accepted that ACh contributes to the relaxation of cavernosal smooth muscle directly by inhibition of contractile adrenergic inputs and indirectly through its action on the endothelium, via the generation of nitric oxide (E. D. Andersson & Wagner, 1995; K. E. Andersson, 2001). The results of our first study, confirming the peripheral parasympathomimetic effects of shilajit, motivated us to focus our second part (in vitro) of study on endothelium-dependant relaxation rather than neurogenic-dependant relaxation where NO released by electrical stimulation of nonadrenergic and noncholinergic nerves contributes to the relaxation of corpus cavernous smooth muscles. The results of the in vitro study confirmed that shilajit induce an endothelium-dependant relaxation and enhance ACh-induced relaxation of rat corpus cavernosum, which can be attributed to the parasympathomimetic effects of shilajit. The rat corpus cavernosum tissue displayed a characteristic biphasic response exhibiting rhythmically both contraction and relaxation phases, which have been reported in previous studies on rabbit corpus cavernosum (Knispel, Goessl, & Beckmann, 1991).
The biological effects of Shilajit are attributed to a combination of two broad groups of compounds: (a) DCPs (oxygenated dibenzo-α-pyrone chromoproteins), comprising metal ions, oxygenated dibenzo-α-pyrones, chromo-constituents, lipoproteins, and apoproteins and (b) low- and medium-molecular-weight fulvic acids (FAs) and fusoms, acting as a systemic carrier/facilitator of the bioactive molecules (Ghosal, 2006a, 2006b). Shilajit is considered as a maharasa (rasayan of highest order) in ayurveda because of its strong antioxidant properties (Ghosal, 2006a, 2006b). The potentiality of antioxidants in the treatment of ED is evidenced from several clinical and preclinical studies (Azadzoi, Schulman, Aviram, & Siroky, 2005; Jones et al., 2002). As per Bhattacharya, Sen, and Ghosal (1995), processed shilajit containing DCPs and FA (20 and 50 mg/kg/day, i.p., for 21 days) induce a dose-related increase in antioxidant enzymes superoxide dismutase, catalase, and glutathione peroxidase in frontal cortex and striatum of rats. Interaction of soft-spin semiquinone radicals of shilajit with oxygen-, sulfur-, and nitrogen-centered free radicals account for the potent free radical scavenging and antioxidant effects of shilajit (Ghosal, 2006a, 2006b). Thus, apart from parasympathomimetic effects, antioxidant potential of shilajit can account for its role against oxidative stress–induced ED.
This research shed light on the key mechanism behind the traditional use of shilajit in the enhancement of sexual vitality in men. The parasympathomimetic effect of key constituents of shilajit accounts for endothelium-dependent relaxation of corpus cavernosum smooth muscles. One limitation of this exploratory study is the inability of the in vitro model to commensurate the various psychological, environmental, social, and health factors responsible for developing ED in humans. In conclusion, the findings of our study provide a new mechanistic approach supporting the traditional claim of shilajit as a sexual vitalizer and provide potential signals for future research. Further in vivo studies exploring the beneficial effect of shilajit on sexual behavior in ED animal models as well as on markers of oxidative stress and cholinergic signal cascade in corpus cavernosum will lend credence to the potential of this maharasa for the treatment of ED in men.
Footnotes
Acknowledgements
The authors would like to acknowledge Indian Herbs Pvt. Ltd., Saharanpur, India, for providing a standardized extract of shilajit as a gift.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge the All India Council for Technical Education, New Delhi, India, for providing a national doctoral fellowship to Ms. Sarabjeet Kaur.
