Abstract
Erectile dysfunction has been a concern for men since the beginning of written history. For many men it can lead to severe psychological distress and humiliation. The treatment of erectile dysfunction has advanced significantly over the past 200 years. Men today are presented with many more viable therapy options leading to improved efficacy and more satisfactory sex lives. The objective of this article is to explore historical options for the treatment of erectile dysfunction, with particular emphasis on the development and progression of the inflatable penile prosthesis.
Introduction
Erectile dysfunction (ED) is defined as the persistent inability to achieve or maintain an erection sufficient for satisfactory sexual performance (National Institute of Health, 1992). It has been described since the beginning of written history with reports being found in Egyptian tombs, on Greek cup paintings, and in the Old Testament (Carson, 2002). It affects men of all races and across all social classes (McLaren, 2007). Both paupers and kings have been reported to have ED. King Louis XVI of France was rumored to have ED as early as age 15 (Fogg & Boorjian, 2010; McLaren, 2007). The objective of this article is to briefly review the historical treatments for ED, with a focus on the history and progression of the inflatable penile prosthesis (IPP).
Materials and Methods
A literature review was conducted using PubMed and EBSCO host academic search complete. For researching the historical elements, the search terms used include: “history of erectile dysfunction,” “history of erectile dysfunction treatment,” and “history of penile prosthesis.” Twenty-one articles met criteria for inclusion in this historical review. Other reference articles were cited for describing the natural history of the disease as well as treatment options.
Etiology of Erectile Dysfunction
The normal physiologic response to sexual stimulation is an increase in nitric oxide (NO) production by nerve endings and vascular endothelial cells, leading to an increase in the enzyme guanylyl cyclase. This enzyme increases levels of cyclic guanosine monophosphate (cGMP), which regulates calcium channels. The calcium channels close in the presence of cGMP, which then decreases intracellular calcium and results in smooth muscle relaxation (Figure 1). This causes vasodilation of penile arteries and allows engorgement of the corpora cavernosa with blood (Archer, Gragasin, Webster, Bochinski, & Evangelos, 2005). This engorgement causes compression of venules against the tunica albuginea, restricting venous outflow. This occurs within the paired corpora cavernosa, resulting in an erection.
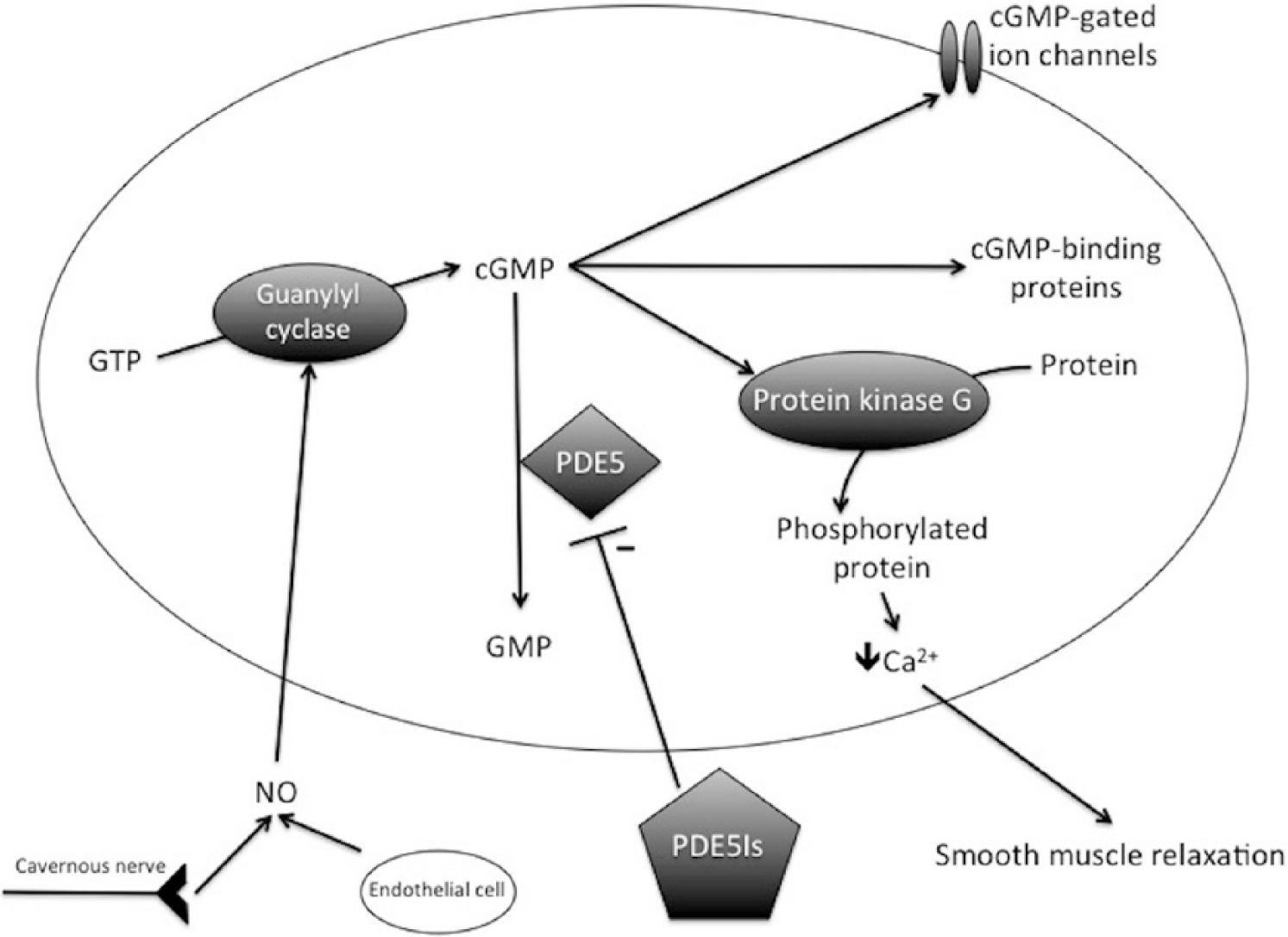
Molecular physiology of erections.
The Massachusetts Male Aging Study revealed that in a population of otherwise healthy men aged 40 to 70 years, 52% of these men had ED (O’Donnell, Araujo, & McKinlay, 2004). However, age alone does not cause ED. Instead, ED is secondary to chronic diseases that increase in incidence with increasing age (Mulligan, Retchin, Chinchilli, & Bettinger, 1988). Most notable of these conditions is cardiovascular disease (CVD). As men age the incidence of CVD increases, which in turn increases the risk for ED (Jousilahti, Vartiainen, Tuomilehto, & Puska, 1999). CVD causes impaired vasodilatory reserve, endothelial and smooth muscle dysfunction, and vascular inflammation (Kumar et al., 2013). All these factors impair the ability to increase blood flow sufficiently to overcome venous outflow and maintain an erection (Lue, 2013). ED shares many of the same risk factors as CVD, including hyperlipidemia, diabetes, obesity, hypertension, and alcohol and tobacco use. In fact, the relationship is so intertwined that ED itself is a risk factor for development of future CVD (Grant, Jackson, Baig, & Quin, 2013). Additionally, as the severity of ED increases so does the prevalence of multivessel CVD (Kumar et al., 2013). Diabetes is also a risk factor for ED. Bacon et al. (2002) report that men with diabetes have a relative risk of 1.3 for having ED compared with men without diabetes. They also noted the risk of ED increases with increasing time from diagnosis of diabetes (Bacon et al., 2002). Another potential cause of ED is hypogonadism. As men age, testosterone levels fall by 1.6% per year starting at age 40 (Buvat, Maggi, Guay, & Torres, 2013). This decline can result in decreased libido and decreased sexual function (Nieschlaq, 2014; Singh, 2013). Less common causes of ED include anatomical disorders such as Peyronie’s Disease, neurogenic disorders such as multiple sclerosis and spinal cord trauma, and psychiatric disorders. Aghighi, Grigoryan, and Delavar (2014) report that among middle-aged men, psychological factors can indeed contribute to ED even when medical or lifestyle risk factors for ED are absent. Their study reported somatization and interpersonal sensitivity to be major contributors (Aghighi et al., 2014). ED can also be a side effect of some medications such as antihypertensives, antidepressants, and antipsychotics (Gregorian et al., 2002; Hatzimouratidis et al., 2010).
Noninvasive Treatments
Treatments for ED date back to as early as the eighth century, when men of Ancient Rome and Greece wore talismans of rooster and goat genitalia. They believed these talismans would serve as an aphrodisiac and promote sexual function (McLaren, 2007). Remedies such as ingesting roasted wolf penis were suggested by the 13th century friar Albertus Magnus (McLaren, 2007). Romans would consume the genitalia of animals with high libidos, such as rabbits, and drink the semen of hawks and eagles (Taberner, 1985). By the end of the 18th century, Dr. Samuel Solomon created his cordial balm of gilead (Grant et al., 2013). During this time, impotence was thought to be a consequence of masturbation and voluntary loss of semen leading to male sexual dysfunction (Darby, 2005). Dr. Solomon’s balm was devised to prevent a man’s desire to masturbate and therefore maintain semen reserves (Helfand, 1989). The balm was composed of cardamom, brandy, and cantharides, and was applied to the testicles in a cold-water bath (McLaren, 2007).
In the 19th century, with the realization of testosterone’s role in erectile function, French neurologist Dr. Charles Edouard Brown-Séquard began injecting himself with extract from the testicles of dogs and guinea pigs (Brown-Séquard, 1889). This progressed to Russian surgeon Dr. Serge Voronoff performing testicular graft transplantations from apes into human testes (Voronoff, 1925). While these were unsuccessful at increasing testosterone levels, the transplantations were a step toward the artificial synthesis of testosterone from cholesterol by Adolf Butenandt in 1935. Butenandt was ultimately awarded the Nobel Prize, shared with Leopold Ruzica, for this work in 1939 (Payne, 2004.)
In 1998, Pfizer (New York, NY) introduced the first effective Food and Drug Administration–approved oral treatment for ED, sildenafil (Viagra; Frumkin, 1944; Payne, 2004). Today, sildenafil and other phosphodiesterase type 5 (PDE5) inhibitors are the first-line treatment for ED (Satyanarayana & Vickers, 2002). The mechanism of action of these drugs is to amplify the previously described NO-cGMP pathway via competitive inhibition of the breakdown mechanism (Lue, 2013). PDE5 breaks down cGMP and allows for penile detumescence. Inhibition of PDE5, and thus inhibition of the cGMP breakdown mechanism, promotes the effects of cGMP and thus smooth muscle relaxation and increased penile blood flow (Lue, 2013). The majority of NO is broken down via oxidation in red blood cells to its nitrate and nitrite metabolites (Kelm, 1999). Because of this, PDE5 inhibitors are contraindicated in men taking nitrates, as a build up of these products can cause fatal hypotension.
Because oral and topical therapies are neither universally effective nor appropriate for all patients, more invasive treatments have been developed (Lue et al., 2004). In the late 17th century, Dr. William Hammond recommended using galvanism in the treatment of ED (McLaren, 2007). This involved placing electrodes on the spine, perineum, testicles, and penis, and administering electric current (McLaren, 2007). Hammond himself admitted that this process was a rather unpleasant experience (McLaren, 2007). Similar methods have been explored in the 21st century, including percutaneous perineal electrostimulation to treat neurogenic ED. Effectiveness and practicality of this technique have not been fully established (Shafik, Shafik, Shafik, & El Sibai, 2008). Devices such as Joseph Lowenstein’s “Coitus Training Apparatus” were designed to treat psychological causes of impotence and involved a ring placed on each end of the penis with a rubber rod connecting them (Loewenstein, 1947), similar to a splint. In a randomized, double-blind, controlled study by Vardi, Appel, Kilchevsky, and Gruenwald (2012), low-intensity extracorporeal shock wave therapy to the penile shaft can have positive effects on erectile function in men who respond to pharmacotherapy. This study was limited by a small patient population as well as short follow-up. More studies are needed to determine long-term effects as well as treatment protocols.
Another noninvasive mechanical method used to treat ED is the vacuum erection device. It was first developed by Vincent Marie Mondat in the early 1800s (McLaren, 2007) and is still in wide use 200 years later (Lue et al., 2004). The pump works by applying negative pressure to the penis to increase corporal blood flow and result in an erection (Jonas, 2001). A more effective, yet also more invasive, method commonly used is the intracorporal injection or intraurethral placement of prostaglandin E1 (PGE1, alprostadil). PGE1 stimulates adenyl cyclase (AC) to increase intracellular cyclic adenosine monophosphate (cAMP) and lower levels of intracellular calcium. This relaxes vascular smooth muscle, resulting in increased penile blood flow (Lue, 2013).
Penile Implants
In 1936, Nicolos Bogoras fashioned the first penile implant using rib cartilage and bone, which was reabsorbed by the body a few years after implantation (Jonas, 2001). This idea was quickly modified by Russian surgeon Dr. Anatolii Frumkin. He used a similar method to reconstruct the genitalia of war victims who suffered amputations (Frumkin, 1944). However, he encountered similar problems with reabsorption. Next, in the early 1950s, acrylic rods were used as penile implants to address reabsorption (Henry, 2009). Drs. Willard Goodwin and William Scott were the first to use implants made of this material (Goodwin & Scott, 1952). Unfortunately, these prostheses had high rates of infection and erosion (Henry, 2009). In the 1960s, silicone rubber was used in an attempt to avoid reabsorption and to decrease infection risk. In 1964, Drs. H. Lash, D. C. Zimmerman, and R. A. Loeffler implanted the first single-rod rubber prosthesis made of silicone (Simmons & Montague, 2008). This single rod was implanted beneath Buck’s fascia but outside of the corpora cavernosum, which led to problems with erosion and extrusion (Henry, 2009). These recurring issues led Dr. Robert Pearman to place a solitary rod of silicone beneath the tunica albuginea inside of the corpora cavernosum in 1967 (Henry, 2009).
While these silicone devices did offer rigidity, concealment was a concern (Henry, 2009). For this reason, devices such as the Flexirod by Dr. Roy P. Finney were invented (Henry, 2009). The device consisted of paired semi-rigid silicone rods placed in each cavernosal body (Simmons & Montague, 2008). Additional features included a soft hinge placed beneath the pubis for concealment, a tapered distal end that allowed for better glans stability, and a trimmable tail for size adjustment (Finney, 1984).
The Jonas prosthesis (Jonas & Jacobi, 1980) was the first malleable device introduced in 1980 and resembles the malleable device available today (Simmons & Montague, 2008). This device was composed of paired silicone rods with an embedded silver wire core. This design gave men the ability to voluntarily bend the penis when they desired concealment or urination and to straighten the penis when they desired an erection (Simmons & Montague, 2008). Current semi-rigid devices are made by American Medical Systems® (AMS, Minnetonka, MN) and Coloplast® (Minneapolis, MN, formerly Mentor). The AMS 600® and AMS 650® are made of a core of braided stainless steel. The Coloplast Genesis® (Figure 2) uses a single spiral sliver wire core (Wilson & Delk, 2000) to employ this malleable mechanism. Both of these models are available today.

Coloplast® Genesis®.
Inflatable Penile Prosthesis
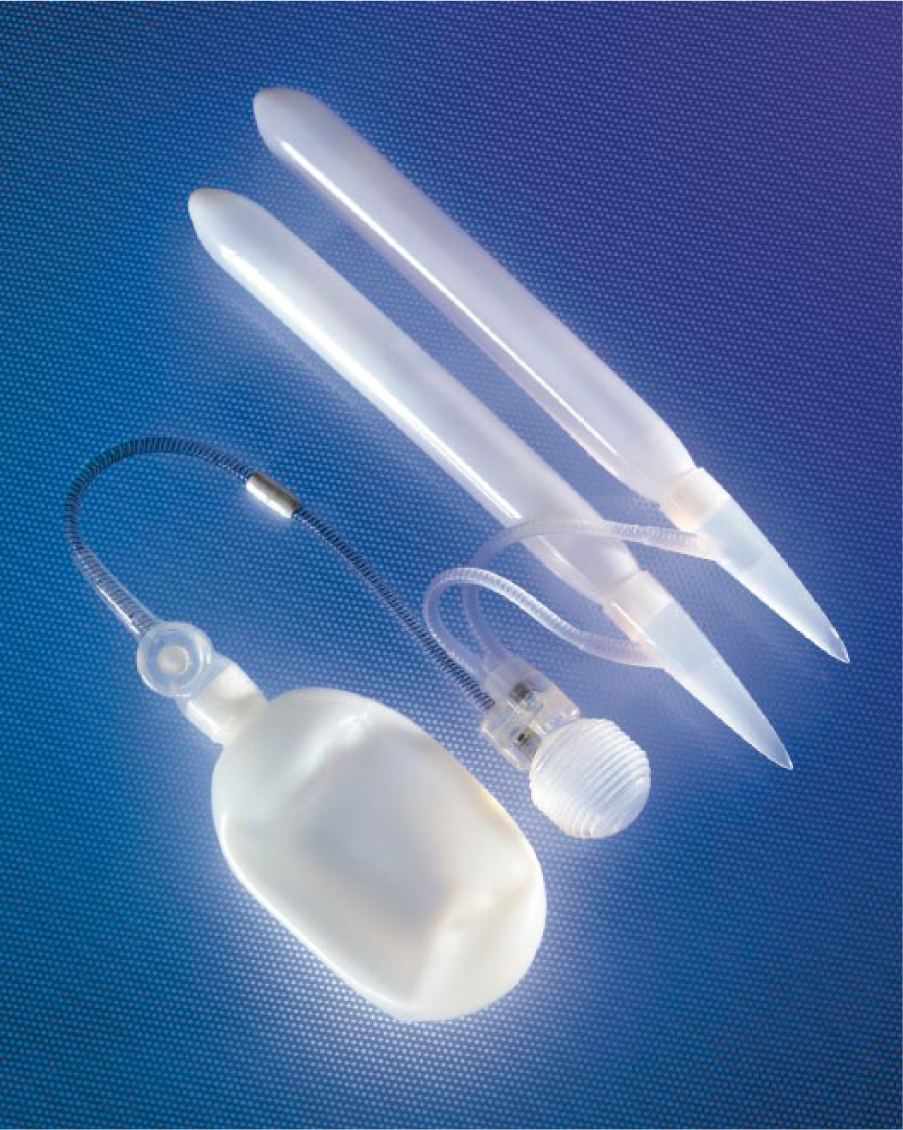
While the malleable devices offered ease of implantation and a user-friendly mechanism, many patients were still not satisfied with the level of concealment and desired a more natural appearance in the flaccid state (Sadeghi-Nejad, 2007). In 1973, Dr. F. Brantley Scott (Scott, Bradley, & Timm, 1973), a urologist at Baylor College of Medicine, became the pioneer of the IPP. His invention of the first three-piece IPP (Henry, 2009) consisted of sponge filled silicone cylinders (Wilson & Delk, 2000). The three-piece device is composed of a pair of inflatable intracorporeal cylinders connected to a small scrotal pump and an intra-abdominal fluid reservoir (Simmons & Montague, 2008; Figure 3). The IPP functions by manual compression of the scrotal pump, which transfers fluid from the intra-abdominal reservoir to the penile cylinders (Lakin & Wood, 2012). When the cylinders inflate, they become rigid to allow penetration (Lakin & Wood, 2012). When the deflation valve is pressed (also located in the scrotal pump), the cylinders empty and fluid returns to the intra-abdominal reservoir (Wilson & Delk, 2000). With the IPP, men can experience an erection when desired, but maintain a normal appearance when deflated. The first three-piece IPP that resembles current models was the AMS 700® (Figure 4), which was introduced in 1983 (Henry, 2009). Because expansion was limited only by the properties of the patient’s corpora cavernosa and because the cylinders were composed of only a single layer of silicone (Garber, 2005), cylinder aneurysms and leakage were a frequent complication (Henry, 2009). These cylinders were too elastic to withstand inflation pressures and would allow asymmetric ballooning of the cylinder (Garber, 2005). The AMS 700 CX® model addressed this by introducing a triple-ply design. The inner layer is a silicone tube that fills with fluid, the middle layer is composed of fabric that governs expansion of the inner tube, and the outermost layer is composed of silicone (Simmons & Montague, 2008). Expansion is limited by the middle fabric layer and not limited by corporal tissue (Henry, 2009). The silicone-woven fabric is made of monofilament-knitted polypropylene and spandex synthetic fiber (Sadeghi-Nejad, 2007). The 700 CXR® is used in patients with a narrow penis or those with corporal fibrosis (those undergoing revision surgery; Hellstrom et al., 2010). This model has fabric that expands unidirectionally allowing only an increase in girth and no increase in length (Garber, 2005).
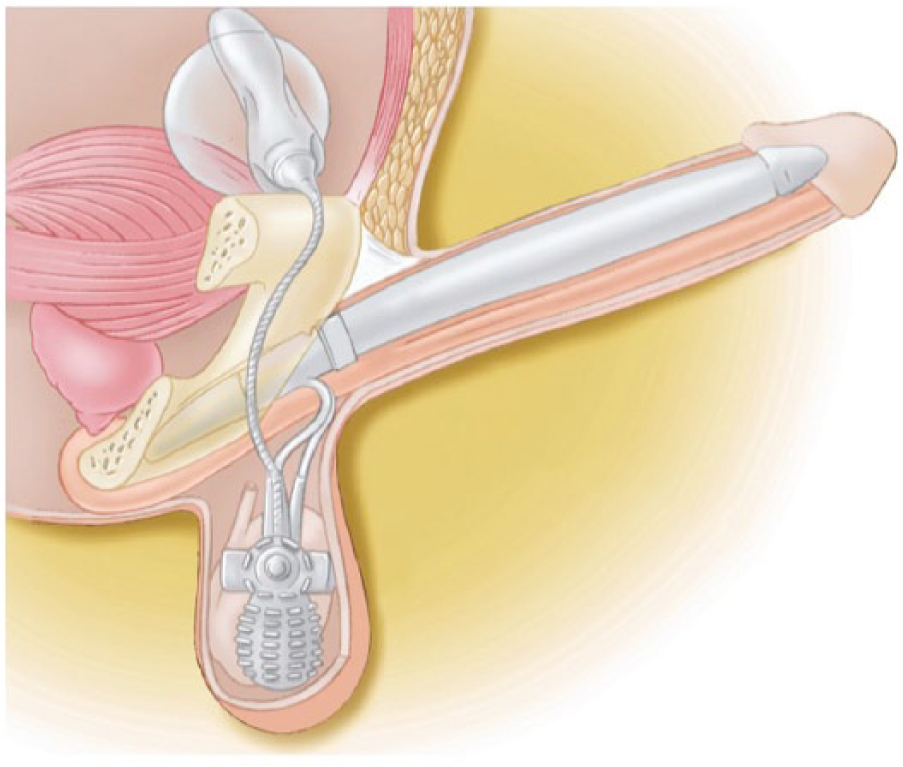
Three-piece inflatable penile prosthesis. Courtesy of American Medical Systems.

American Medical Systems 700®.
A three-piece IPP has also been developed by Coloplast using their Bioflex® technology (Henry, 2009). With seven times the tensile strength of silicone, this material resists aneurysmal dilation and is more durable than previously used materials (Sadeghi-Nejad, 2007). The exact composition of Bioflex is unknown to the public. However, it is reported to be an aromatic polyurethane elastomer with hard and soft segments (Garber, 2005). Other three-piece Coloplast models include the Alpha-1® (Figure 5), the first connector-less single pump-cylinder IPP, which reduced the risk of connector leakage (Henry, 2009).
Coloplast Alpha-1®.
In an attempt to combine the ease of implantation of the semi-rigid devices with the concealment of an inflatable device, the first unitary inflatable device (Surgitek’s Flexi-Flate® and AMS Hydroflex®, later replaced by the two-piece Dynaflex®; Simmons & Montague, 2008) was developed in 1985 (Wilson & Delk, 2000). The advantage of this device was ease of implantation like malleable devices, with the cosmetic appeal of inflatables (Sadeghi-Nejad, 2007). To inflate the device, a small pump located at the distal end of the prosthesis is compressed and fluid transfers from a self-contained fluid reservoir into the nondistensible inner chamber (Simmons & Montague, 2008). Complications arose due partial inflation of the device, which gradually caused tissue expansion by compressing the corporal tissue. Over time, this led to the cylinders being of inadequate size for a rigid erection (Wilson & Delk, 2000). This device has been abandoned.
In another attempt to make implantation easier, two-piece models have been developed including the AMS Ambicor® (Figure 6; Sadeghi-Nejad, 2007) and the Coloplast GFS 2-piece®. Only the AMS Ambicor device is available today. In these models the paired cylinders are connected by tubing to the scrotal component, which serves as both the pump and fluid reservoir (Simmons & Montague, 2008). There was a high failure rate of the tubing in this model, leading to the development of the Mark II®, which employs a connector-less system (Wilson & Delk, 2000). These devices were developed for patients in whom intra-abdominal reservoir placement may need to be avoided (Wilson & Mulcahy, 2006). However, rigidity and expansion are less with a 2-piece device than with a three-piece device.
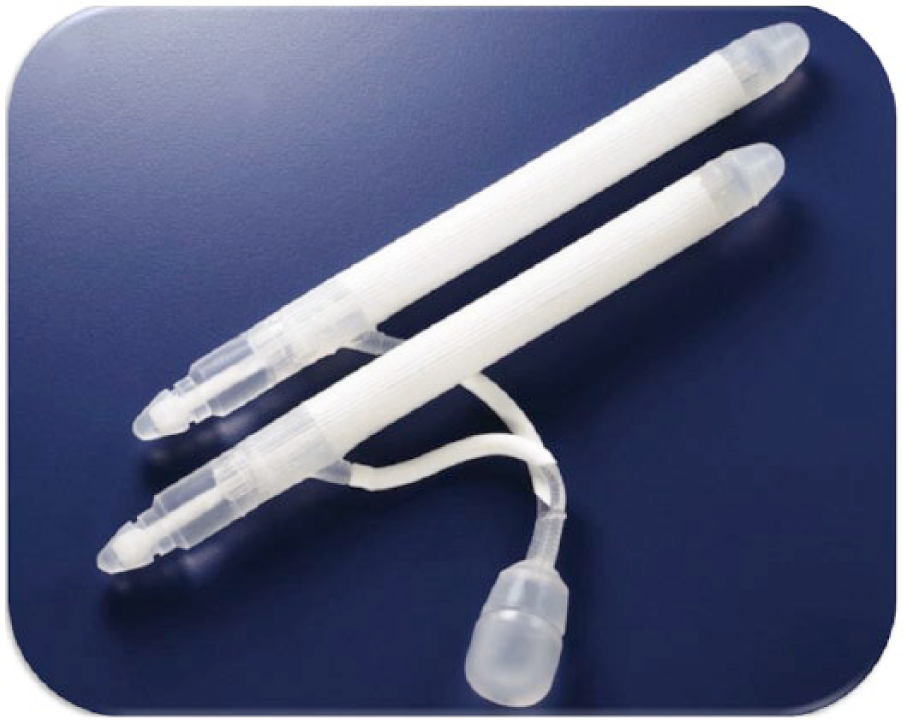
American Medical Systems Ambicor®.
The IPP comes with a small yet extremely morbid risk of infection (Sadeghi-Nejad, 2007). In the case of any infection, all prosthetic material must be removed. This can lead to further complications with revision surgeries (Hellstrom et al., 2010) such as scarring and fibrosis of corporal bodies, penile shortening, and recurrent infection (Mulcahy et al., 2004). The introduction of both InhibiZone® by AMS® in 2000 and Coloplast’s hydrophilic coating in 2002 has attempted to combat this issue (Henry, 2009). InhibiZone uses minocycline and rifampin embedded into the external silicone surface to prevent bacterial growth and the formation of biofilms (Henry, 2009). The Coloplast Titan® hydrophilic coating allows the cylinders to be immersed in antibiotics of the surgeon’s choice at the time of implantation (Henry, 2009). In a retrospective review of 1,337 implantations over 7 years, it was reported that the infection rate for first time implantations was 3% and the infection rate for revision surgeries was 10% (Wilson & Delk, 1995) before the introduction of antibiotic coated devices. Further studies have reported that antibiotic coatings have decreased infection rates by 50% in first time implantations (Mandava, Serefoglu, Freier, Wilson, & Hellstrom, 2012). In a retrospective review, the AMS InhibiZone was reported to decrease infection rates 180 days post implant from 1.61% to 0.68% in initial implantations (Carson, 2004). Similarly, in a study comparing Coloplast hydrophilic coated Titan model with noncoated Alpha-1 model, infection rates were seen to be 2.07% for the Alpha-1 model compared with 1.06% for Titan models at 1-year follow-up after initial implantation (Wolter & Hellstrom, 2004).
An uncommon complication with IPPs is autoinflation, in which the device inflates without the pump being compressed. For this reason AMS created the Momentary Squeeze® deflate button. This button must be compressed for longer to promote deflation and also has a one-way valve to prevent autoinflation (Henry, 2009). Similarly, Coloplast’s Lock-out Valve® was introduced in 2001 to the Alpha 1 model and was carried over to Titan models (Hakky, Wang, & Henry, 2014). This is a one-way valve that does not allow fluid to exit the reservoir during an increase in intra-abdominal pressure (Valsalva, coughing, sneezing, laughing), which reduces the incidence of autoinflation (Henry, 2009). Other rare complications associated with penile prostheses include mechanical failure, reservoir displacement, distal cylinder erosion or extrusion, and cavernosal fibrosis (particularly in revision cases; Hellstrom et al., 2010).
Future Developments
The evolution of the IPP continues. Development in the areas of mechanized cylinder inflation via battery-operated pumps could eliminate problems associated with manual manipulation of the scrotal pump and allow for more rapid fluid transfer (Simmons & Montague, 2008). Additionally, the development of more compact, simpler devices that eliminate the need for connector systems and tubing may lead to lower mechanical failure and infection rates (Simmons & Montague, 2008). Research into these areas is promising and will hopefully lead to better functioning prostheses with lower rates of complications and higher rates of patient satisfaction.
Conclusion
ED has been documented dating back to the beginning of written history. In the days of Louis XVI, few effective options were available, and ED was often associated with public humiliation and ridicule. In the 21st century, men have many more effective options for treatment with evidence-based results to support their use. As research continues and new advances in treatment are made, we can potentially offer men even more efficacious and less morbid options to treat ED. Although the subject may be difficult to approach for some patients, it is not a problem that men have to endure quietly; Treatment options are available to help men and their partners maintain satisfactory sex lives.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
