Abstract
Background:
The most common cancer in women worldwide, breast cancer most often metastasizes to the bone. Improved chemo- and radiotherapies and novel molecular therapies have prolonged survival in women with osseous metastatic breast cancer, but spinal metastases often cause cord compression that degrades their functional independence.
Purpose:
In women with breast cancer metastasized to the spine, we sought to (1) identify independent predictors of a functional deficit 3 months after surgical management and (2) assess the utility of existing metrics at highlighting patients at risk of a postoperative functional deficit.
Methods:
We performed a single-institution, retrospective analysis of 92 patients meeting our inclusion criteria between 2004 and 2021. Patients were classified by 3-month postoperative Eastern Cooperative Oncology Group (ECOG) scores into good/independent (ECOG 0 to 2) and poor/dependent (ECOG 3 to 5) functional outcome groups. Univariate and multivariate analyses were performed to identify patient and tumor factors associated with good vs. poor 3-month ECOG scores.
Results:
Preoperative use of selective estrogen receptor modulators (SERMs) was significantly associated with good postoperative functional outcomes. Poor preoperative function, the presence of visceral metastases at the time of surgery, and triple-negative primary or metastatic tumor status were independently associated with poor 3-month postoperative function. Host characteristics, sociodemographic factors, and indicators of surgical complexity, including estimated blood loss, front/back surgery, and corpectomy reconstruction, were not associated with 3-month ECOG score. A multivariate model including these significant univariate associations and normalized for patient demographics identified preoperative SERM use, poor preoperative function (ECOG score), and triple-negative primary or metastatic tumor status as independently associated with functional status 3 months after surgery.
Conclusions:
Our retrospective analysis found that preoperative SERM use was significantly associated with improved postoperative functional outcomes, while poor preoperative function and triple-negative tumor status were significantly associated with poor function 3 months after surgery. These factors may serve as indicators of function and independence after surgery for patients with metastatic breast cancer to the spine.
Level of Evidence:
Level IV: Prognostic Study
Keywords
Introduction
Breast cancer is the most common cancer in women worldwide, and the second most common cancer overall. 1 It most often metastasizes to bone; 6.4% to 9.4% of all breast cancer patients develop osseous metastases at some point in their lives. 2 Improved chemo and radiotherapies and the introduction of novel molecular therapies have prolonged the survival of women with osseous metastatic breast cancer—in 1990 to 1994—their survival was 24.2 months, and by 2005 to 2009 it had increased to 37.8 months. 3 Improved survival has also been observed in patients who required surgery for metastatic breast cancer to the spine, from a median of 21 months in 2007 to 26.8 months in 2014. 4
However, discussing therapeutic advancements in terms of survival alone provides an incomplete picture of the impact on patients with metastatic breast cancer, since not all patients may view improved survival in a debilitated state as an “improvement.” This is critical when discussing surgery with patients with metastatic breast cancer to the spine, as spinal metastases often result in cord compression that can severely and permanently degrade the patient’s functional independence and quality of life, but not necessarily indicate imminent death.5,6 This emphasis on restoration or preservation of function is inherent to the neurologic, oncologic, mechanical, and systemic decision framework that guides treatment decisions for these patients. 7 Several works have identified independent risk factors for poor function after surgery for metastatic spine disease.8 –10 However, factors including small sample size, failure to consider changes in primary versus metastatic disease histology, and limited detail regarding the surgery performed limit the quality of this analysis. Current works have also not evaluated the ability of known metrics that characterize spinal instability, cord compression, or mortality risk to predict postoperative functional recovery.
We evaluated a large single-institution cohort of patients with breast cancer who had spinal metastases and underwent surgical treatment. We sought to (1) identify independent predictors of a functional deficit 3 months after surgery and (2) assess the utility of existing predictive metrics at highlighting patients at risk of a postoperative functional deficit.
Methods
This was an institutional review board-approved single-institution retrospective study of adult female and male patients (>18 years old) diagnosed with breast cancer spinal metastases treated surgically between 2004 and 2021. Patients were excluded if they underwent spinal surgery for a non-oncologic reason, such as trauma or infection; if they underwent multiple spinal surgeries; or if they were lost to follow-up (return visit) <3 months postoperatively.
Of the 202 patients who underwent surgery for metastatic breast cancer to the spine, 92 met our inclusion criteria; of these, 91 (99%) were female. Of the patients with information available for race, 81 (88.04%) were white. Mean age was 57.07 ± 11.8 years (median 56.36, 95% CI [54.6, 59.5]), and mean body mass index (BMI) at OR was 28.5 ± 7.1 (91 of 92, median 27.3, [27, 30]). Mean follow-up time postoperatively was 2.5 ± 2.84 years (median 1.475, range 0.03–16.7, [1.9, 3.1]). Mean time from symptoms to diagnosis (in months) for the 53 patients with available data was 6.32 months ± 14.75 (range 0–96, [2.25, 10.3], median of 2 months).
All pertinent available records—including patient visits, operative notes, imaging, laboratory results, and pathology reports—were accessed from the electronic medical record (EMR) to identify patients who met our inclusion criteria. The patient list was generated from a pre-existing repository of patients who underwent spinal surgery for metastatic cancer. They were then screened for breast cancer. The senior author had access to the database, which allowed for the development of the cohort. All variables were curated by the authors upon chart review. Data cleaning was performed by manual curation. Demographic data (age, weight, historical BMI, and BMI at OR), social determinant data (race, insurance status, zip code; all gathered from EMR), and smoking status were collected. Comorbidities were measured and quantified using the age-adjusted Charlson Comorbidity Index. 11 Cancer history, including the date of diagnosis, primary and metastatic tumor hormone status, primary tumor grade and stage at diagnosis, primary tumor histology (infiltrating ductal carcinoma (IDC) vs. non-IDC), multimodal treatment status for primary and metastatic disease, primary recurrence, and additional sites of metastatic disease were extracted. Operative data derived from surgical documentation included levels operated upon and decompressed, front/back surgeries, estimated blood loss, corpectomy reconstruction, separation surgery, postoperative ICU care, and perioperative complications (including infections, wound complications, venous thromboembolic complications, and <90-day readmissions and reoperations; Table 1).
Demographic parameters. Univariate analysis of demographic parameters of patients separated by post-operative ECOG scores.
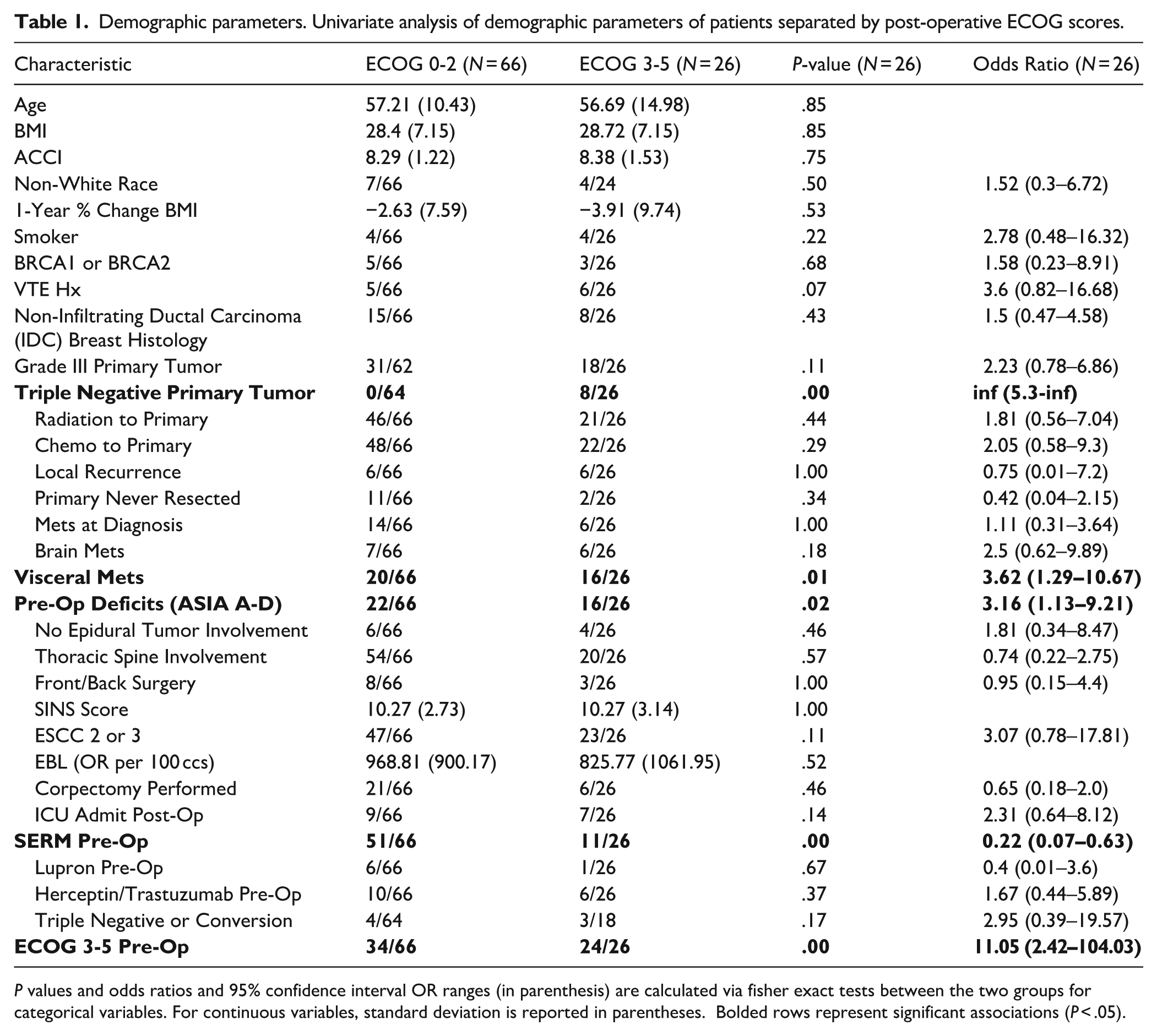
P values and odds ratios and 95% confidence interval OR ranges (in parenthesis) are calculated via fisher exact tests between the two groups for categorical variables. For continuous variables, standard deviation is reported in parentheses. Bolded rows represent significant associations (P < .05).
Eastern Cooperative Oncology Group (ECOG) functional scores were recorded preoperatively and 90 days postoperatively. 12 We defined ECOG scores of 0 to 2 as functionally independent (good function), while ECOG scores of 3 to 5 were considered functionally disabled (poor function). There were 66 patients with postoperative ECOG scores of 0 to 2 and 26 patients with postoperative ECOG scores of 3 to 5.
Additional metrics of interest included Spinal Instability Neoplasia score (SINS), 13 epidural spinal cord compression (ESCC) scale, 14 and Skeletal Oncology Research Group 30-day mortality, 90-day survival, and 1-year survival likelihoods. 15 We used the SINS score as a continuous variable, while we defined an ESCC score of 2 as severe cord compression.
Statistical Analysis
Statistical analysis was performed using SPSS 21 (IBM) and Python version 3.8.18 16 using Jupyter Notebooks 17 (Matplotlib version 3.7.2, 18 NumPy version 1.24.3, 19 pandas version 2.0.3, 20 SciPy version 1.10.1, 21 seaborn version 0.12.2, 22 and statsmodels version 0.14.0 23 ). Patient demographic data (age, BMI, and race), comorbidities, tumor characteristics, oncologic treatment history, metastasis characteristics, surgical treatment history, and functional scoring were reported as frequencies for categorical data and mean with standard deviation (SD) for continuous data. Analyses were performed, including and excluding patients who died within 90 days of surgery. Characteristics were compared across functional status groups, defined by ECOG score. Associations were assessed using Fisher’s exact test (for categorical variables) or independent t-tests (for continuous variables) as indicated. We then performed a stepwise multivariate regression that included factors found to have significant univariate comparisons and was normalized for age, BMI at OR, and race. Any missing data for categorical variables is noted in the population size in Table 1.
Data were reported, analyzed, and organized according to the RECORD checklist. 24
Results
Triple-negative primary tumors were universally associated with a poor functional outcome 90 days after surgery (P = .004, perfect separation). Visceral metastases were also significantly correlated with a poor functional outcome (P < .05, odds ratio (OR): 3.68). Preoperative motor deficits were significantly associated with poor postoperative functional outcomes (P = .02); 9 of the 13 patients at ECOG 3 to 4 postoperatively had preoperative American Spinal Injury Association Impairment Scale scores of A to D. While the SINS score did not significantly differ between groups when the full cohort was considered, there was a trend between elevated SINS score and worse postoperative function when patients with a 90-day mortality were removed (P = .08). Finally, preoperative selective estrogen receptor modulator (SERM) treatment was associated with better postoperative function (OR: 0.22 of ECOG 3 to 5 at 3 months postoperatively, P = .002). Fifty-one of the 66 (83.3%) patients who had a postoperative ECOG score of 0 to 2 were previously on SERMs, as opposed to only 11 of the 26 (42.3%) patients with ECOG 3 to 5.
Hormone receptor status had a substantial impact on patient function both before and after surgery. Patients whose primary tumors were positive for the estrogen receptor (ER) were 26 times more likely to have good function 3 months postoperatively. ER positivity within the metastatic disease appeared to have a protective correlation from a 90-day mortality standpoint; 8 of the 14 ER-negative patients died within 90 days of surgery, whereas only 5 of the 57 ER-positive patients died (OR: 13.8; Fisher’s exact P: .0002). Progesterone-receptor positivity appeared to have a mildly beneficial correlation with low functional impairment 3 months postoperatively (OR: 4.16; Fisher’s exact P: .005) but showed no association with 90-day mortality. Her2-neu status was not significantly correlated with either functional status or mortality.
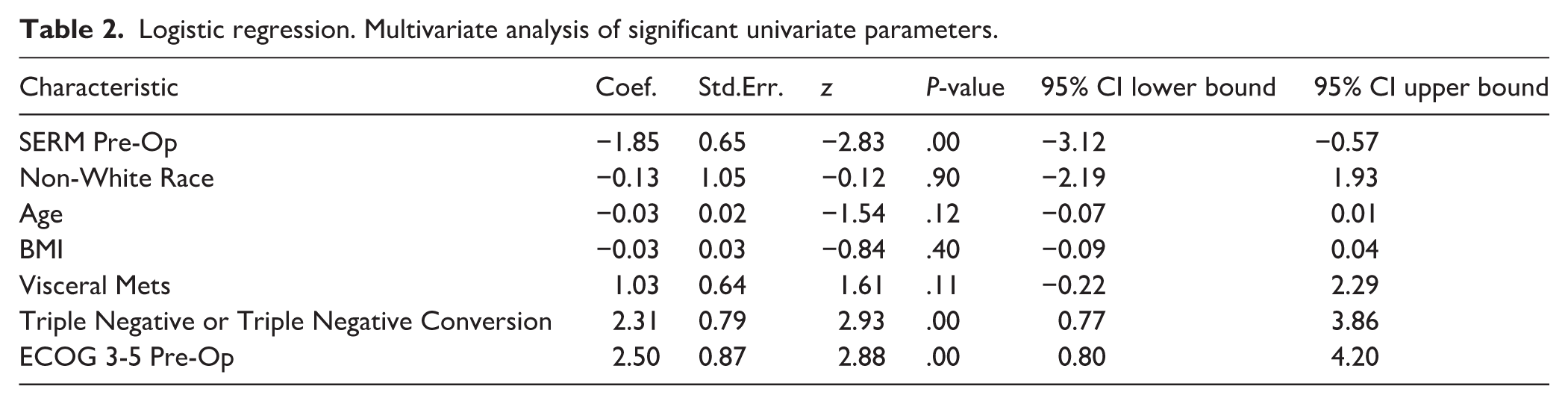
Our multivariate model included visceral metastasis, preoperative SERM treatment (univariate OR: 0.22, 95% CI [0.07, 0.63]), preoperative ECOG > 2 (univariate OR: 11.05 [2.42, 04.03]), and triple-negative primary tumor or conversion to triple-negative metastatic disease (univariate OR: 10.97 [2.78, 54.04]). Preoperative SERM therapy was found to be independently associated with better postoperative function, while a preoperative ECOG > 2 and triple-negative disease were independently associated with worse postoperative function (Figure 1, Table 2).
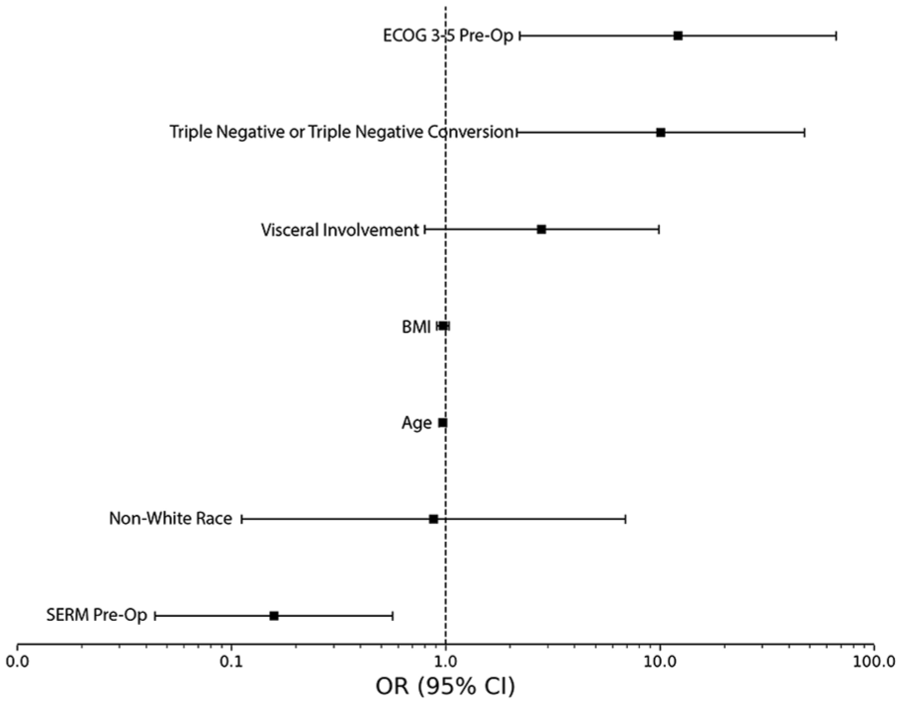
Multivariate model predicting functional outcomes postoperatively | Logistic regression including significant univariate associations as well as demographic variables of age, BMI, and race in predicting postoperative ECOG 3-5.
Logistic regression. Multivariate analysis of significant univariate parameters.
Discussion
Given the growing number of patients projected to have metastatic breast cancer to the spine, it is important to identify preoperative predictors not only of survival but also of satisfactory postoperative function.25,26 This study aimed to identify factors that correlated with functional outcome 90 days after surgery for patients with metastatic breast cancer to the spine. We found that neurologic deficits, triple-negative primary tumors, and visceral metastases prior to surgery were independently associated with a worse functional outcome, while a history of SERM therapy was independently associated with a better functional outcome. We hope that our findings can serve as an aid to clinicians in providing preoperative guidance and managing expectations for patients with metastasis to the spine who require surgical intervention.
This study has multiple limitations, including those intrinsic to retrospective studies. We could not assess the effect of gender or race on functional independence, given that the majority of patients (81/90) were white and the majority (91/92) were female, although non-white race was evaluated. The region of analysis has universal Medicaid, so government insurance status may not be as strongly associated with socioeconomic deprivation as in other areas of the United States. As ECOG 5 is assigned to patients who have died, our inclusion of deaths may have skewed the analysis of living patients as well. However, we believe that dependent function and mortality are equivalent, and we chose to include these patients in our analysis. Patients who died (ECOG 5) did not die intraoperatively but rather died sometime within the 3-month postoperative period, which, for the purposes of this study, renders the patients functionally equivalent to someone who is totally disabled from a functional perspective. While SERM use was included in our analysis, no specific medication was evaluated. However, the overwhelming proportion of our cohort received tamoxifen. Further, it is unclear if the improved outcome in the SERM group was due to SERM use or if it was a proxy for improved medical control of the tumor. Finally, surgical decision-making between surgeons at the same institution may vary. While there are generally reliable indications for surgery for metastatic breast cancer, we did not normalize between individual surgeons.
The goals of managing metastasis to the spine are to restore and prolong functional independence. 27 Function is only partially conflated with survival. A large-scale population-based study of survival after spinal metastases across different primary cancer types found that 13.4% of breast cancer patients undergoing operations for spinal metastases died within 90 days of surgery; another 35.1% died within 1 year of surgery. 28 However, the functional outcomes of radiation alone for spinal cord compression are variable.29 –32 A recent study identified patients who had been preoperatively irradiated or with triple-negative metastatic tumor histology as at a higher risk for adverse events. 33 However, surgical morbidity does not necessarily inform function, which should be measured independently. The novelty of our study is that it focuses on functional independence, which, while just one component of morbidity and mortality, can help us understand if treatments are helpful.
A strength of the present work is that the factors that were independently associated with independent versus dependent functional status can easily be identified by both surgeons and non-surgeons. This permits broad pre-emptive engagement of the oncologic team at the earliest time point. Our findings are also largely intuitive and, therefore, easy for the patient and family to understand. Patients who are independent at the time of surgery are more likely to be independent after surgery. Patients with a disease with a positive ER status may have broader and less morbid systemic treatment options, which can lead to less functional dependence.34,35 Those with triple-negative primary tumors or aggressive metastatic disease may fare worse, a finding more likely exacerbated given that an ECOG score of 5 indicates death. Markers for expectation setting are vital to engaging in meaningful discussions with patients and family and may also help to build trust with healthcare providers.36,37
Almost every patient who was severely impaired (ECOG 3 to 4) at the time of surgery for spinal metastases had poor function or died within 3 months of surgery. This same patient group was more likely to have triple-negative tumors (8 of 26 with ECOG 3 to 5, vs. 0 of 64 with ECOG 0 to 2), and visceral disease involvement (16 of 26 vs. 20 of 66). These parameters are non-modifiable attributes of patients and their disease that are highly predictive of functional outcomes. This conclusion is supported by recent work, which shows that the incorporation of molecular subtype features into spinal metastasis risk tools improves their ability to predict prognosis. 38 While molecular subtyping of breast cancer tumors has been assessed in works evaluating radiotherapy outcomes 39 and overall breast cancer survival,40 –42 the present work is the first to our knowledge to focus on the functional independence of patients who undergo surgery for metastatic breast cancer.
Interestingly, having advanced disease at the time of diagnosis, SINS score, and ESCC were not predictive of ultimate functional outcome. This suggests that the degree of epidural compression or structural instability alone should not disqualify a patient from open surgery, which is an area that remains actively investigated.14,43
In conclusion, in a retrospective analysis of a large single-institution cohort of patients who underwent surgery for metastatic breast cancer to the spine, preoperative function and a history of SERM use were associated with functional independence 3 months after surgery, while triple-negative disease status was associated with functional dependence. These factors were independent of surgical technique and complexity. These findings may inform anticipatory guidance for the patient and their family during the perioperative period. They may also serve as easy red flags for patients who may require aggressive rehabilitation treatment during the postoperative period.
Supplemental Material
sj-docx-1-hss-10.1177_15563316251377721 – Supplemental material for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer
Supplemental material, sj-docx-1-hss-10.1177_15563316251377721 for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer by Layla Siraj, Julia B. Duvall, Elie Massaad, Mitchell S. Fourman and John H. Shin in HSS Journal®
Supplemental Material
sj-docx-2-hss-10.1177_15563316251377721 – Supplemental material for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer
Supplemental material, sj-docx-2-hss-10.1177_15563316251377721 for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer by Layla Siraj, Julia B. Duvall, Elie Massaad, Mitchell S. Fourman and John H. Shin in HSS Journal®
Supplemental Material
sj-docx-3-hss-10.1177_15563316251377721 – Supplemental material for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer
Supplemental material, sj-docx-3-hss-10.1177_15563316251377721 for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer by Layla Siraj, Julia B. Duvall, Elie Massaad, Mitchell S. Fourman and John H. Shin in HSS Journal®
Supplemental Material
sj-docx-4-hss-10.1177_15563316251377721 – Supplemental material for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer
Supplemental material, sj-docx-4-hss-10.1177_15563316251377721 for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer by Layla Siraj, Julia B. Duvall, Elie Massaad, Mitchell S. Fourman and John H. Shin in HSS Journal®
Supplemental Material
sj-docx-5-hss-10.1177_15563316251377721 – Supplemental material for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer
Supplemental material, sj-docx-5-hss-10.1177_15563316251377721 for Preoperative Function, Previous SERM Treatment, and Triple-Negative Tumor Status are Independently Associated With 3-Month Postoperative Function After Surgical Decompression of Metastatic Breast Cancer by Layla Siraj, Julia B. Duvall, Elie Massaad, Mitchell S. Fourman and John H. Shin in HSS Journal®
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by award numbers T32GM007753 and T32GM144273 from the National Institute of General Medical Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of General Medical Sciences or the National Institutes of Health.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LS discloses a relationship with the National Institutes of Health. MSF discloses relationships with Johnson & Johnson and Astura Medical. JHS discloses relationships with ATEC, Augmedics, Globus, iCotec, and Evolution Spine. The other authors disclose no potential conflicts of interest.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration.
Consent to Participate
Informed consent was waived from all participants included in this study by the IRB at Massachusetts General Hospital (approval #2015P001837).
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as Supplemental Material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
