Abstract
As robotics in spine surgery has progressed over the past 2 decades, studies have shown mixed results on its clinical outcomes and economic impact. In this review, we highlight the evolution of robotic technology over the past 30 years, discussing early limitations and failures. We provide an overview of the history and evolution of currently available spinal robotic platforms and compare and contrast the available features of each. We conclude by summarizing the literature on robotic instrumentation accuracy in pedicle screw placement and clinical outcomes such as complication rates and briefly discuss the future of robotic spine surgery.
History of Robotic Technology
Spine surgery has made significant advancements over the past 3 decades, largely driven by innovation in operative techniques, instrumentation, implant design, biomaterials, biologics, and intraoperative imaging. Many of these advances allowed for smaller incisions, higher fusion rates, and improved accuracy, all of which may contribute to decreased complications and improved patient-reported outcomes [13,16,26,46]. Perhaps the most significant progress occurred in the field of intraoperative imaging, with developments such as real-time navigation and 3-dimensional (3D) spinal mapping. In addition to facilitating some spine procedures, these innovations have directly driven the advancement of spinal robotic technology.
Robotic-assisted surgery has been used in urology, gynecology, and general surgery since the early 1990s. However, spine surgery has been a relatively late adopter. In general, surgical robots can be classified into 3 categories: telesurgical, supervisory-controlled, and shared-control. Telesurgical systems allow the surgeon to directly control the robotic instruments from a remote location. Supervisory-controlled systems allow the surgeon to preoperatively plan the surgery; once the plan is set, the robot executes it under close supervision by the surgeon. Shared-control systems allow the surgeon and robot to function simultaneously—both the robot and the surgeon control instruments and motions. Historically and currently, available spine robots are shared-control systems.
Early attempts to use robotic technology in spine surgery occurred in the 1990s but were met with varying degrees of success and significant workflow challenges. The Miro system (German Aerospace Center, Cologne, Germany) used an optical tracking system with central control to position a drill guide [39]. The SpineBot (Centre for Intelligent Surgery Systems, Hanyang University, South Korea) used preoperative computed tomography (CT) to plan pedicle screw instrumentation, and an intraoperative robotic arm and holder were used to localize the trajectory [28]. Two initial telesurgical systems—one made by Georgetown University that used joystick control and fluoroscopic-based imaging and a second, the Spine Bull’s-Eye Robot (First Affiliated Hospital, Zhengzhou University, Henan Province, China)—demonstrated initial promise with regard to accurate placement of needles or guidewires in targeted spine locations, but neither reached significant commercial success [5,56].
In the early 2000s, with improvements in intraoperative CT imaging and navigation, there was a resurgence of interest in robotic-assisted spine surgery. Ultimately, improved intraoperative navigation led to the development of several spine robots, now commercially available. However, early iterations of these robots were fraught with challenges, including software crashes, errors in synchronizing intraoperative fluoroscopic images with preoperative 3D imaging, problems deflecting the robotic arm resulting in decreased accuracy, and challenges with intraoperative user interfaces [48]. Technical or clinical errors were noted in more than 50% of spine procedures performed using robotic assistance during this time [2]. For example, pedicle screws had to be placed over guidewires, resulting in decreased workflow efficiency. In addition, platforms did not allow for real-time visual feedback for instrument localization, which meant surgeons were forced to trust the robot’s accuracy and precision. Considering these challenges, this technology was not widely adopted.
Currently Available Robots
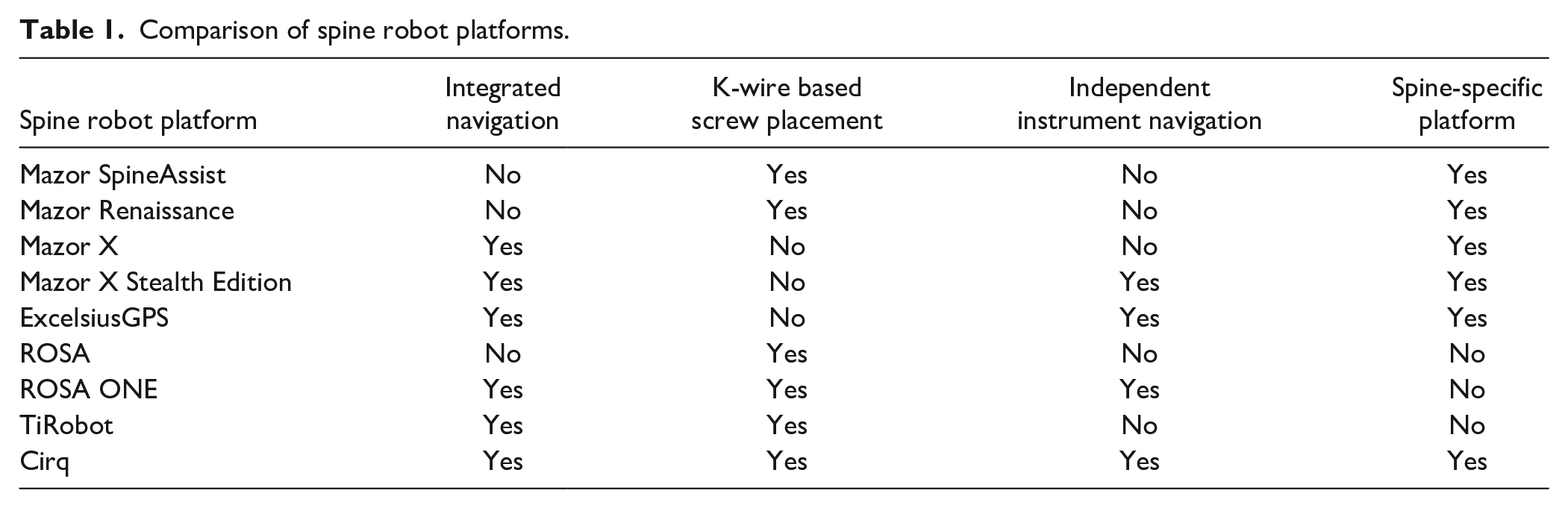
An overview of spinal robot platforms is provided in Table 1.
Comparison of spine robot platforms.
Mazor
The Mazor robotic platform (Medtronic Navigation, Louisville, CO, USA; Medtronic Spine, Memphis, TN, USA) was the first robotic platform cleared by the US Food and Drug Administration (FDA) for use in spine surgery. The SpineAssist, the first-generation Mazor robot, obtained FDA clearance in 2004 and continues to be one of the most widely used systems worldwide. It uses a patient-mounted track attached to an anatomical landmark and relies on preoperative and/or intraoperative CT imaging to allow for planning screw trajectories. The SpineAssist also allows for the pairing of preoperative CT with intraoperative fluoroscopy, as opposed to intraoperative CT imaging. The Renaissance, the second-generation Mazor robot, was released commercially in 2011. The Renaissance is smaller and lighter than the SpineAssist, and software improvements allow for 10 times faster processing speeds and enhanced image recognition algorithms. It also provides surgeons the ability to flatten bone at desired pedicle screw start points to minimize the risk of skiving. Both the SpineAssist and Renaissance Mazor robots require pedicle screws to be placed over guidewires.
The Mazor X, the third-generation Mazor spine robot, launched in 2016, and upgrades include a fully automated robotic arm that no longer requires a patient-mounted track (as seen with first- and second-generation Mazor robots). Software upgrades allow for the registration of each vertebral body independently, providing multiplanar imaging datasets to assist with surgical planning. Last, an optic camera allows for real-time volumetric assessment of the operating room environment to self-detect its location within space and avoid collision intraoperatively. The acquisition of Mazor Robotics by Medtronic resulted in the integration of Medtronic StealthStation navigation technology, and the 2019 release of the Mazor X Stealth Edition, the latest generation Mazor robot (Fig. 1). The Mazor X Stealth Edition provides for fully independent navigation capability and real-time instrumentation position feedback.
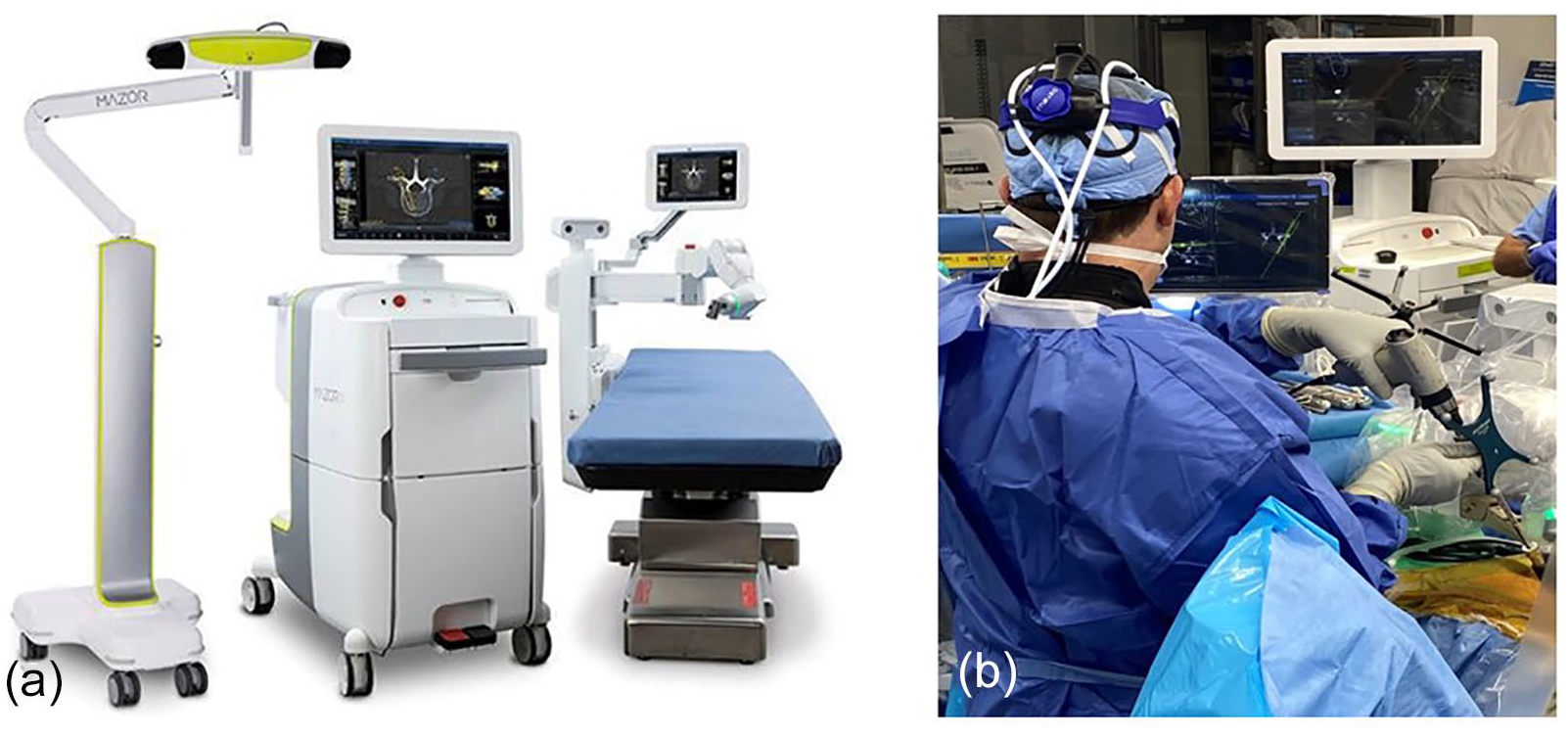
(a) Mazor X Stealth Edition Spinal Robotic Platform (Medtronic Navigation Louisville, CO, USA; Medtronic Spine, Memphis, TN, USA) and (b) Intraoperative image demonstrating utilization of the Mazor Robotic platform to place lumbar pedicle screws in the prone position.
Excelsius
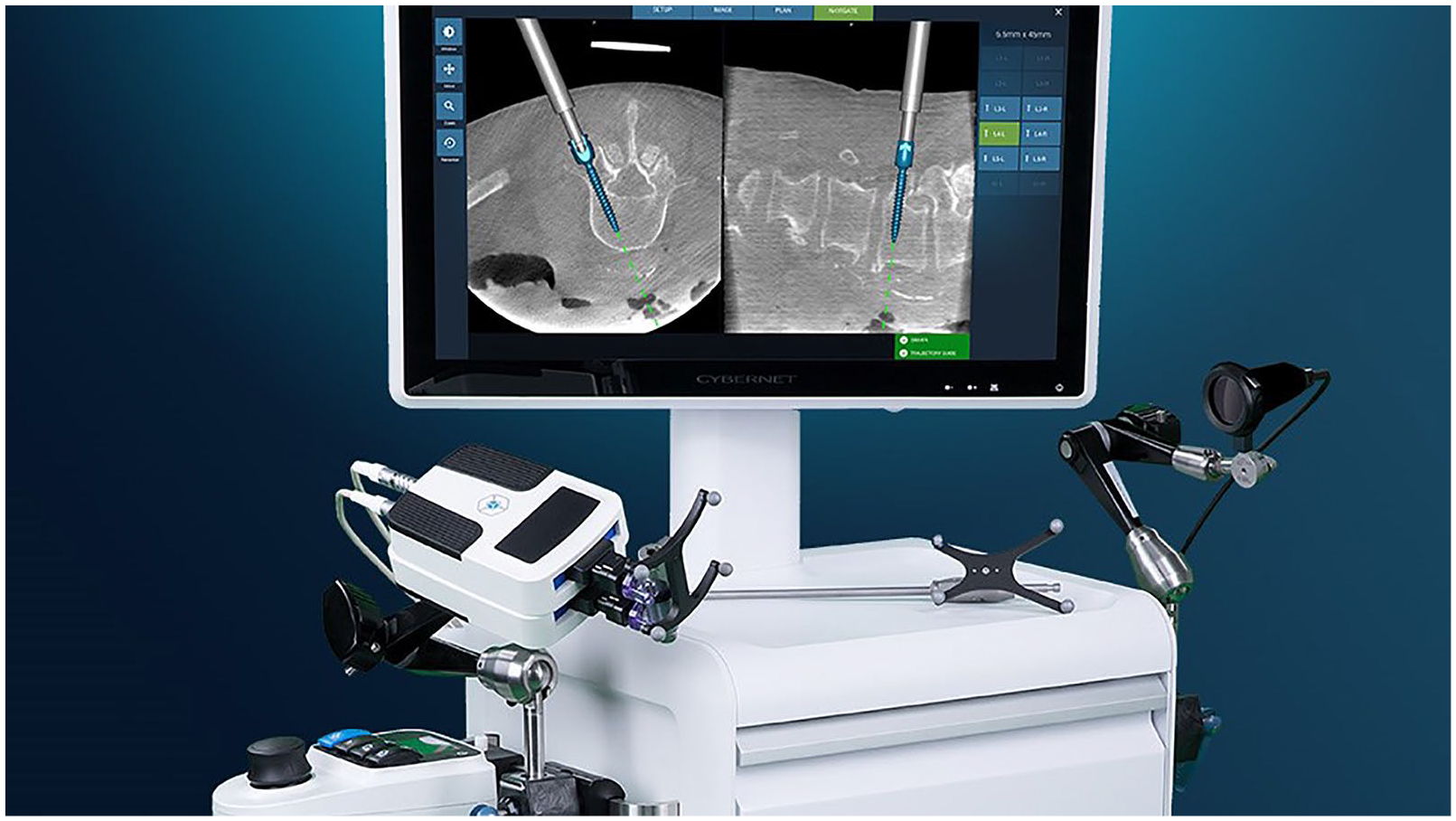
The ExcelsiusGPS (Globus Medical, Inc., Audubon, PA, USA) was released in 2017 and is marketed as the first spine robot with a fully integrated navigation platform, allowing for real-time instrument tracking and pedicle screw placement without guidewires (Fig. 2). The robot is anchored to a floor-mounted base station, as opposed to the operative table. The navigation platform is compatible with multiple intraoperative 2-dimensional (2D) and 3D imaging modalities and can also merge a preoperative CT scan with intraoperative fluoroscopic imaging, similar to the latest generation Mazor robots. The ExcelsiusGPS robotic arm is rigid and capable of maintaining less than 1 mm of deflection when exposed to lateral forces up to 200 N, and excessive lateral force can be detected with a unique sensor [6]. Its independent navigation capability allows for localization of manufacturer-specific instrumentation, similar to the Mazor X Stealth Edition, and its modular robotic arm allows for additional instruments to be mounted to the robot.
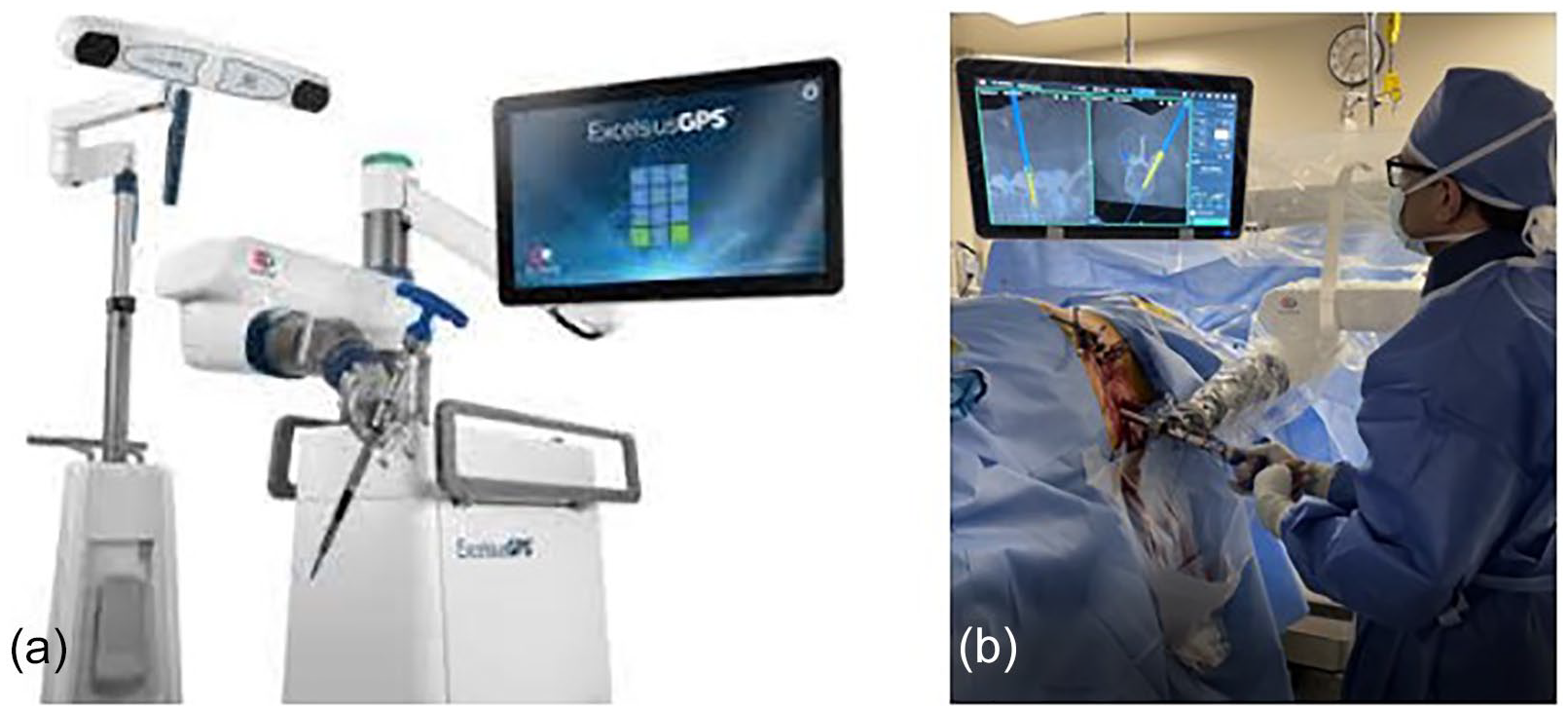
(a) ExcelsiusGPS Spinal Robotic Platform (Globus Medical, Inc.) and (b) intraoperative image demonstrating utilization of the ExcelsiusGPS spine robot to place lumbar pedicle screws in the lateral position.
ROSA
The ROSA Spine Robot (Zimmer Biomet, Warsaw, IN, USA), FDA clearance in 2016, is a freestanding robotic system that uses preoperative or intraoperative CT imaging to plan screw trajectories and has a fully automated robotic arm, allowing for screw placement over guidewires. A recent upgrade, known as the ROSA ONE, received FDA clearance in 2019 (Fig. 3) and features a fully integrated navigation system compatible with manufacturer-specific instrumentation, similar to the Mazor X Stealth Edition and Globus ExcelsiusGPS. The ROSA platform allows for navigation and instrumentation across spine, cranial, and knee arthroplasty procedures. This unique capability makes it a potentially more attractive option for hospitals or ambulatory surgery centers that have needs beyond spine-specific robotic platforms.

The ROSA ONE Spinal Robotic Platform (Zimmer Biomet).
Recent Spine Robots
The Cirq spinal robot (Brainlab AG, Munich, Germany) received FDA clearance for spine procedures in September 2019, and its accuracy for placing cervical instrumentation has recently been assessed (Fig. 4) [10,21]. More recently, the Fusion Robotics spinal navigation and robotics system (Boulder, CO) received FDA clearance in February 2021 (Fig. 5). Finally, the TiRobot (TINAVI Medical Technologies, Beijing, China) was approved in China in 2016 but not in the United States. Like the ROSA platform, the TiRobot can also be used for other orthopedic applications outside of spine surgery.
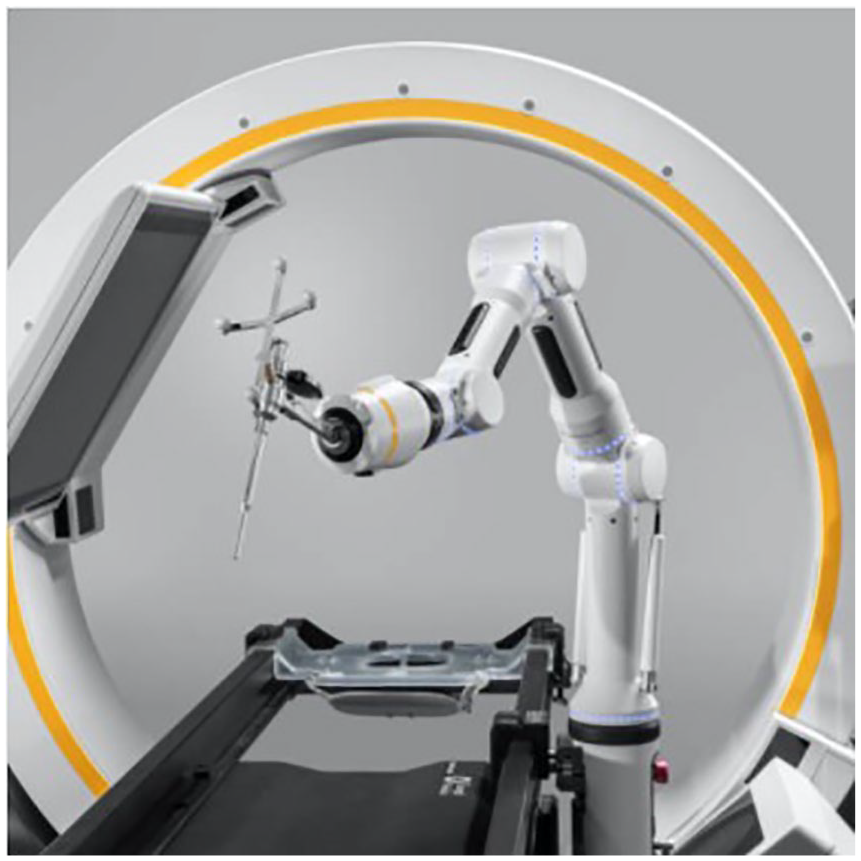
The Cirq spinal robotic platform (Brainlab AG).
Fusion Robotics LLC spinal robotic platform.
Advantages of Robotic Technology
Safe and efficient spine surgery relies upon meticulous fine motor skills to navigate narrow corridors, particularly when placing pedicle screw instrumentation. These procedures can be arduous; surgeons are susceptible to fatigue, and this may compromise accuracy and operative efficiency. Spinal robotic platforms offer a number of benefits, including reducing surgeon fatigue and tremors, while providing stability for instrumentation through a fixed working angle that may increase accuracy and precision. Ultimately, it is hoped that robotic platforms will increase operating room efficiency, result in more consistent and improved outcomes, decrease complications, and decrease overall health care costs. While much of the evidenced-based benefit of robotic technology in spine surgery has yet to be established, there is a growing body of evidence on accuracy and radiation exposure.
Pedicle Screw Placement
Several studies investigating the accuracy of robotic-assisted pedicle screw placement have demonstrated 91% to 98% accuracy [7,22,23,29,40,49,50]. Studies comparing robotic-assisted pedicle screw placement versus freehand techniques have demonstrated varying results. Two studies demonstrated significantly higher accuracy with robot-assisted placement [9,54], while a prospective study demonstrated lower accuracy of robotic-assisted screws versus fluoroscopic-guided screws [42]. Given the inconsistencies in the literature, high-quality meta-analyses have been performed, 2 of which demonstrated equivalent accuracy between the 2 techniques [12,41], and a third demonstrated superior accuracy with robotic assistance [32]. Compared with 3D navigation techniques, robotic-assisted pedicle screw placement has demonstrated a slightly higher accuracy in some retrospective studies [30,43]. A recent meta-analysis demonstrated that compared with freehand techniques, both robotic-assisted and navigated screw placement resulted in lower intraoperative and postoperative screw revision risk [47].
Aside from potentially improved accuracy secondary to minimizing pedicle cortical breaches, robotic-guided screws may allow for less proximal facet violation, thereby potentially decreasing the risk of adjacent segment disease that can compromise clinical outcomes [1,44,53]. Several prospective studies and 2 meta-analyses have demonstrated a reduced risk of proximal facet joint violation when using robotic technology as compared with freehand or fluoroscopically based pedicle screw placement [15,19,27,32,57,58].
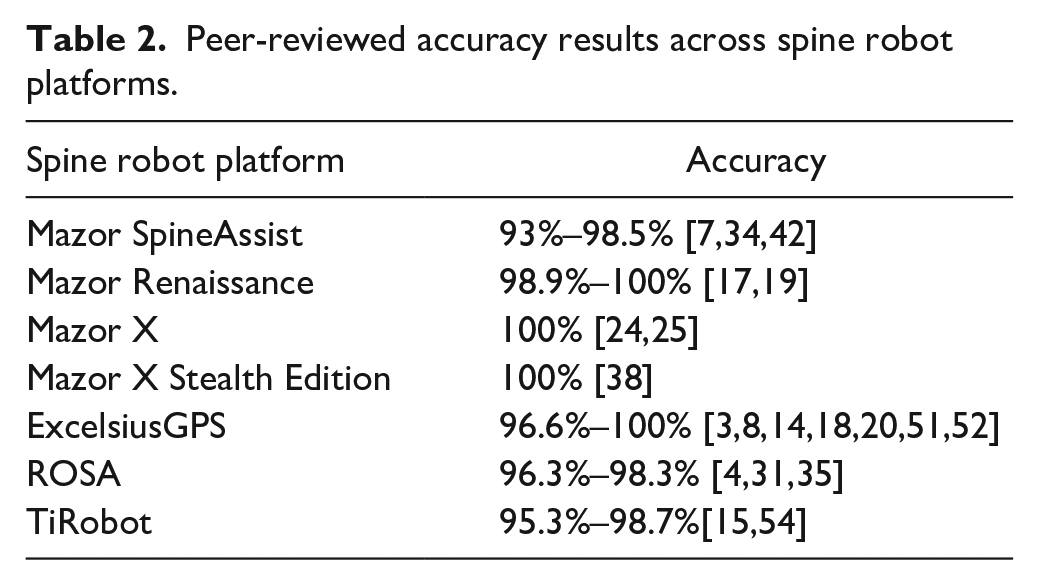
There are currently no available peer-reviewed studies directly comparing the accuracy of the available spinal robotic systems. The majority of the published literature on instrumentation accuracy is based on the Mazor SpineAssist and Renaissance platforms, as these were the earliest platforms with advanced navigation technology to come to market. Table 2 highlights the accuracy results based on the literature for the Mazor SpineAssist, Mazor Renaissance, Mazor X, Mazor X Stealth Edition, ExcelsiusGPS, ROSA, and TiRobot robotic platforms.
Peer-reviewed accuracy results across spine robot platforms.
Radiation
When comparing robotic-assisted surgery to freehand pedicle screw placement, many studies have demonstrated significantly decreased radiation exposure using the former approach [22,23,34,43], while 2 studies demonstrated no significant difference in radiation exposure between the 2 techniques [30,42]. However, some studies suggest that with robot-assisted pedicle screw placement, radiation exposure may be higher to the patient [42] and lower to the surgeon [54]. We need studies comparing radiation exposure resulting from the use of both robotic platforms and traditional 3D navigation-based techniques in pedicle screw placement.
Clinical Outcomes
Studies examining clinical outcomes of robotic technology in spine surgery are difficult to interpret due to study design. Most of the available literature compares traditional open procedures to robot-assisted minimally invasive spine surgery (MISS). Therefore, it is not clear if it is the robotic component of the procedure or the less invasive aspect of MISS that is contributing to lower complication rates, decreased length of stay, less postoperative opioid consumption, and faster postoperative recovery [19,22,23,36,54]. To better understand the additive benefit of robotic technology in a similar cohort of spine procedures, well-designed studies are needed to compare clinical outcomes in traditional open versus robot-assisted open spine surgery or MISS using fluoroscopic or real-time navigation versus robot-assisted MISS.
Complications of Robotic Technology
We have focused mostly on the potential benefits of robotic technology in spine surgery, but several papers have demonstrated contrasting results. For example, when compared with freehand techniques of pedicle screw placement, the accuracy of robotic-assisted pedicle screw placement has not been found to be consistently higher. In fact, 2 of the meta-analyses mentioned earlier failed to demonstrate higher accuracy of robotic-assisted pedicle screw placement [12,41], while Ringel et al found the approach demonstrated lower accuracy [42].
Two recent database studies that compared robot-assisted lumbar spinal fusion with conventional lumbar spinal fusion demonstrated increased short-term complications and revisions associated with robotic assistance. Yang et al found that higher risk of revision, 30-day readmission, and total 30-day complications were associated with robotic assistance during lumbar spinal fusion [55]. A study by Lieber et al also demonstrated a higher inpatient complication rate associated with robotic assistance during lumbar spinal fusion [33]. One possible cause of such increased complications may be the longer operative time associated with robotic-assisted surgery—a longer operative time can lead to increases in perioperative complications, particularly infection. Another explanation may be the learning curve associated with adopting robotic technology, which may result in higher initial rates of screw malposition that ultimately require early revision. However, the limitation of both studies is the use of large national databases that limit patient identification to procedure codes, not allowing for the further stratification of MISS versus open procedures. In addition, both studies are subject to the standard limitations associated with any database study, such as selection bias and coding error.
Costs
The use of robotics in spine surgery has increased 169% over the past decade and has linearly increased each year [55]. This has led to significant concerns about expenditures associated with robotic technology, as it can require significant capital acquisition costs—for some platforms, costs near $1,000,000. In order for robotic technology in spine surgery to be economically pragmatic, there needs to be a proven benefit demonstrated through decreased operative time, lower postoperative lengths of stay and revision rates, and improved postoperative patient-reported outcomes.
While there is no current consensus on the economic practicality of robotic technology in spine surgery, studies citing decreased revision rates suggest that this may be the most likely justification for robotic technology in spine surgery from a cost standpoint; lower revision rates decrease direct health care expenditure related to additional surgery and also minimize the indirect societal cost of prolonged disability [11,45]. In examining retrospective data from 1 academic center, Menger et al calculated a cost benefit of approximately $600,000 over 1 year due to shorter operative times and lower revision rates associated with robotic spine surgery [37]. Further prospective studies are needed to evaluate the overall costs of robotic technology in spine surgery, but it may become more favorable as acquisition and maintenance costs continue to decrease and robotic technology advances.
Future Advances
The latest generation of spine robot platforms have made significant advances in real-time navigation, improved intraoperative user interface, and workflow efficiency. More research is needed to increase the use of robotics in spine surgery. Studies are needed to assess whether the potentially improved outcomes, reduced complications, and decreased health care costs justify the capital expense of a robotic platform. In addition, robotic technology needs to evolve beyond just facilitating pedicle screw placement. Some platforms currently allow surgeons to plan tubular retractor trajectories and can stabilize retractors with the robotic arm. In addition, some surgeons are using the integrated navigation platform to plan interbody placement and execute navigated disk preparation. However, a minority of surgeons make use of these relatively novel advancements. As these features become more streamlined, more surgeons will likely integrate them into the surgical workflow.
Future spine robots may have burrs that are compatible with the robotic platform and allow for precise intraoperative execution of preoperatively planned bony decompression. Haptic feedback must also improve for this feature to evolve. Finally, it is possible that pedicle screw placement becomes fully automated and evolves from the current shared-controlled platform. As spinal robotic technology continues to advance, capital cost decreases, and further research is conducted that demonstrates the potential clinical and economic benefits of robotics in spine surgery, it is more likely that spine surgeons will adopt it in their operating rooms.
In conclusion, spinal robotic technology has made marked advancement over the past 2 decades, and the latest generation of robots is just starting to penetrate the spine market. Along with expansions in the coming years will come increased competition and innovation.
Footnotes
CME Credit
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Darren R. Lebl, MD, reports relationships with Medtronic, Nuvasive, Stryker, Depuy Synthes, Guidepoint, Remedy Logic, ISPH II, Vestia Ventures, MiRUS, Woven Orthopedic Technologies, Integrity Implants, and HS2. Sheeraz A. Qureshi, MD, MBA, reports relationships with Stryker K2M, Globus Medical, Paradigm Spine, AMOpportunities, RTI Surgical, Integrity Implants, Medical Device Business Services, Medtronic USA, Nuvasive, Avaz Surgical, Simplify Medical, Tissue Differentiation Intelligence, Vital 5, Spinal Simplicity, ![]() , Healthgrades, Society of Minimally Invasive Spine Surgery, Simplify Medical, North American Spine Society, Minimally Invasive Spine Study Group, Lumbar Spine Research Society, International Society for the Advancement of Spine Surgery, Contemporary Spine Surgery, Cervical Spine Research Society, Association of Bone and Joint Surgeons, and Annals of Translational Medicine. Ram K. Alluri, MD, Fedan Avrumova, BS, Ahilan Sivaganesan, MD, and Avani S. Vaishnav, MBBS, declare no potential conflicts of interest.
, Healthgrades, Society of Minimally Invasive Spine Surgery, Simplify Medical, North American Spine Society, Minimally Invasive Spine Study Group, Lumbar Spine Research Society, International Society for the Advancement of Spine Surgery, Contemporary Spine Surgery, Cervical Spine Research Society, Association of Bone and Joint Surgeons, and Annals of Translational Medicine. Ram K. Alluri, MD, Fedan Avrumova, BS, Ahilan Sivaganesan, MD, and Avani S. Vaishnav, MBBS, declare no potential conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
