Abstract
Public health measures for medical emergencies generate methodological and ethical challenges for human research. Using Emanuel et al.'s framework, we assessed the ethical integrity of the research methods used in a men's alcohol and other drug (AOD) use disorder study following their revision due to COVID-19 restrictions in South Africa. Following the amendments, the study's social value, favorable risk-benefit ratio, and respect for participants increased. Collaborative partnership, scientific validity, fair participant selection, independent review, and informed consent improved in terms of successful stakeholder engagements and interviewing procedures, but were compromised due to a cellphone access eligibility criterion and complicated consenting procedures. Methodological and ethical challenges of research during health emergencies can be navigated with flexibility and innovation.
Keywords
Introduction
Alcohol and other drug (AOD) research has numerous and sometimes unique ethical challenges as it often involves collection of sensitive information from vulnerable, marginalized and stigmatized populations (Fry et al., 2006a; Souleymanov et al., 2016). Such challenges are compounded during times of crises, such as the COVID-19 pandemic. Yet, as with health policy generally, effective AOD policy demands a sound evidence base for planning interventions (Louie et al., 2020).
South Africa's (SA) response to the COVID-19 pandemic was particularly assertive. A national lockdown was introduced on 27 March 2020 under a National State of Disaster (Department of Cooperative Governance and Traditional Affairs, 2020a). Restrictions to mitigate the spread of the pandemic included a total ban on alcohol sales, closure of on- and off-consumption alcohol outlets, prohibition of the transportation of alcoholic beverages, and severe restrictions on people's movements except to provide essential services (Department of Cooperative Governance and Traditional Affairs, 2020b).
Considering the case of a multi-site study on AOD use disorder treatment among men in SA, which commenced just before the onset of COVID-19, this paper describes the methodological changes that were necessitated by the COVID-19-related restrictions that were implemented in SA and the ethical implications of those methodological changes for the study. We applied an ethical analysis drawing on the framework of Ezekiel Emanuel and colleagues (Emanuel et al., 2004, 2008), identified lessons that could be learned, and proposed recommendations on how AOD and other researchers might manage such challenges during future public health crises. Given the World Health Organization's (WHO) call for increased preparedness for future similar pandemics (WHO, 2022) and the impacts of climate change on health systems (WHO, 2020), achieving responsive adaptations in research approaches that retain ethical integrity during such system stresses would be a significant benefit for maintaining evidence-based health policy. This paper focuses on research ethics issues and challenges during COVID-19 from the perspective of the researcher, in contrast to other papers from South Africa (e.g., Burgess et al., 2023, 2024; Rossouw et al., 2024) and internationally (e.g., Salamanca-Buentello et al., 2024) that have examined ethical issues more from the perspective of the research ethics reviewer/review committee.
The Emanuel et al. Ethical Framework for Biomedical Research
Emanuel and colleagues (2004, 2008) developed a framework of eight ethical principles for biomedical research with a set of 31 benchmarks to aid the evaluation of research studies’ adherence to the principles (Table 1). The principles are argued to be interrelated, necessary and sufficient for ensuring ethical conduct of research (Emanuel et al., 2004, 2008). We selected this framework due to its proposed universal applicability (Emanuel et al., 2004, 2008), its relevance not only to biomedical research, but also to epidemiological and social/behavioural science studies (Wassenaar & Mamotte, 2012), and its incorporation of many principles of the major ethical codes (e.g., the Nuremburg code and Belmont Report). We briefly outline the framework here, which is henceforth referred to as the Emanuel framework.
Eight Ethical Principles for Assessing Biomedical Research by Emanuel et al. (2004, 2008).

The first principle - Collaborative partnerships - involves including beneficiaries and stakeholders, such as policy makers, advocacy groups and community advisory boards (CABs), in decisions concerning different stages of research planning, implementation and dissemination. Such stakeholder involvement increases the chances that the research will benefit communities and be of social value and scientific validity (discussed below). The value of stakeholder involvement is being recognized increasingly for health research, particularly in low- and middle-income countries (LMICs; Zhao et al., 2019), as well as for AOD research specifically (e.g., Fry et al., 2006b). In the case of AOD studies, relevant CAB members often include people with lived experience of AOD use, AOD service users and providers, and community members and leaders who may serve as gatekeepers (among other roles) to research.
The social value principle requires that studies address important and/or widespread problems and is considered particularly important in contexts of scarce resources for studying and addressing societal problems, such as LMICs (Emanuel et al., 2004, 2008). Ensuring both collaborative partnerships and social value in research in LMICs can also help to enhance distributive and social justice and can be valuable in contexts where there are few local researchers available to conduct or provide culturally competent interpretations of research data (Zingela et al., 2023). To realize social value, AOD research should ideally benefit people and communities affected by and involved in addressing AODs.
The principle of scientific validity relates to the methodological rigor of research. Research of high scientific validity (and social value) can potentially benefit communities, whereas research with limited scientific validity would lack social value and is considered an unnecessary burden of time on participants (Emanuel et al., 2004, 2008).
The fair participant selection principle requires that participants are selected such that research findings can be valid and reliable; risks associated with participation are minimized and shared equally, while benefits are maximized; vulnerable participants are protected; and participants stand to benefit from the research. For AOD studies, fair participant selection is often difficult to achieve due to stigma, people's marginal living arrangements and the often illegal behaviors under study (Maestre et al., 2018; Millum et al., 2019), but various means to overcome these challenges exist.
The principle of favorable risk-benefit ratio involves balancing study-related risks to individual participants against benefits to society (Emanuel et al., 2008). Risk-benefit assessments underpinned the SA government's banning of alcohol sales during the COVID-19 pandemic; the benefits to society of drastically reduced alcohol availability and hence consumption (e.g., freeing up hospital beds from people with alcohol-related injuries to those needing COVID-related treatment and hospitalization; Barron et al., 2020, 2024) were judged to outweigh possible risks to people with alcohol use disorders (AUDs), such as possible alcohol withdrawal symptoms following abrupt cessation of alcohol consumption.
To achieve independent review, research ethics committee (REC) members must be competent to review studies according to agreed ethical principles and make independent decisions free of conflicts of interest (Emanuel et al., 2004, 2008). Conflicts of interest may arise where REC members are recipients of industry funding on a topic under review (Saban & London, 2022). Also, REC members’ personal opinions and judgements concerning AODs may hinder unbiased and independent review of AOD studies (Bell & Salmon, 2012).
The informed consent principle requires that, before enrolment
Finally, the respect for participants principle, which is a core ethical principle, requires researchers to honor commitments made during the consenting process throughout the study's duration, by monitoring possible study-related harms and providing requisite relief, where possible (Emanuel et al., 2004, 2008). For AOD studies, this may entail keeping information about illegal drug-related practices confidential, in the absence of requirements for disclosure that may be set by legal or ethics review bodies.
The Multi-Site Study
In 2019 our team, comprising researchers from five universities and institutions in SA and one outside of SA, initiated a multi-site study in four provinces in SA. Our primary goal was to examine the availability and nature of AOD treatment provision and factors associated with limited treatment seeking for AOD use disorders among men. This study focused on men as they are disproportionately affected by AOD use disorders (WHO, 2019) and often their masculinities attitudes contribute to alcohol-related harm (Giusto & Puffer, 2018; Rich et al., 2015) and a failure or reluctance to access health services (Dworkin et al., 2015; Dzinamarira & Mashamba-Thompson, 2019).
The first phase of the project (2019–2021) was designed to involve qualitative exploratory in-person in-depth interviews (IDIs) and focus group discussions (FGDs). However, following the imposition of COVID-19 lockdown restrictions in March 2020, and given the study's non-therapeutic nature, we were required to cease all in-person research activities. In response, we applied for and were granted ethics clearance for three major modifications to our research methods to allow our research to continue with minimal risk (see Table 2). Firstly, we conducted all our research activities via telephonic or web-based virtual/remote formats instead of in person. Consequently, access to a cellphone became a study inclusion criterion for all participants, and prospective participants were given cellphone data to enable them to take part in the study. Secondly, we dropped the FGDs from among the study methods as we deemed them impractical and infeasible via telephonic or virtual platforms. To compensate for this change, we increased the target number of participants for the in-depth interviews (IDIs). Finally, we added questions about AOD use and treatment during COVID-19 to our data collection tools.
Pre- and During COVID Research Activities.
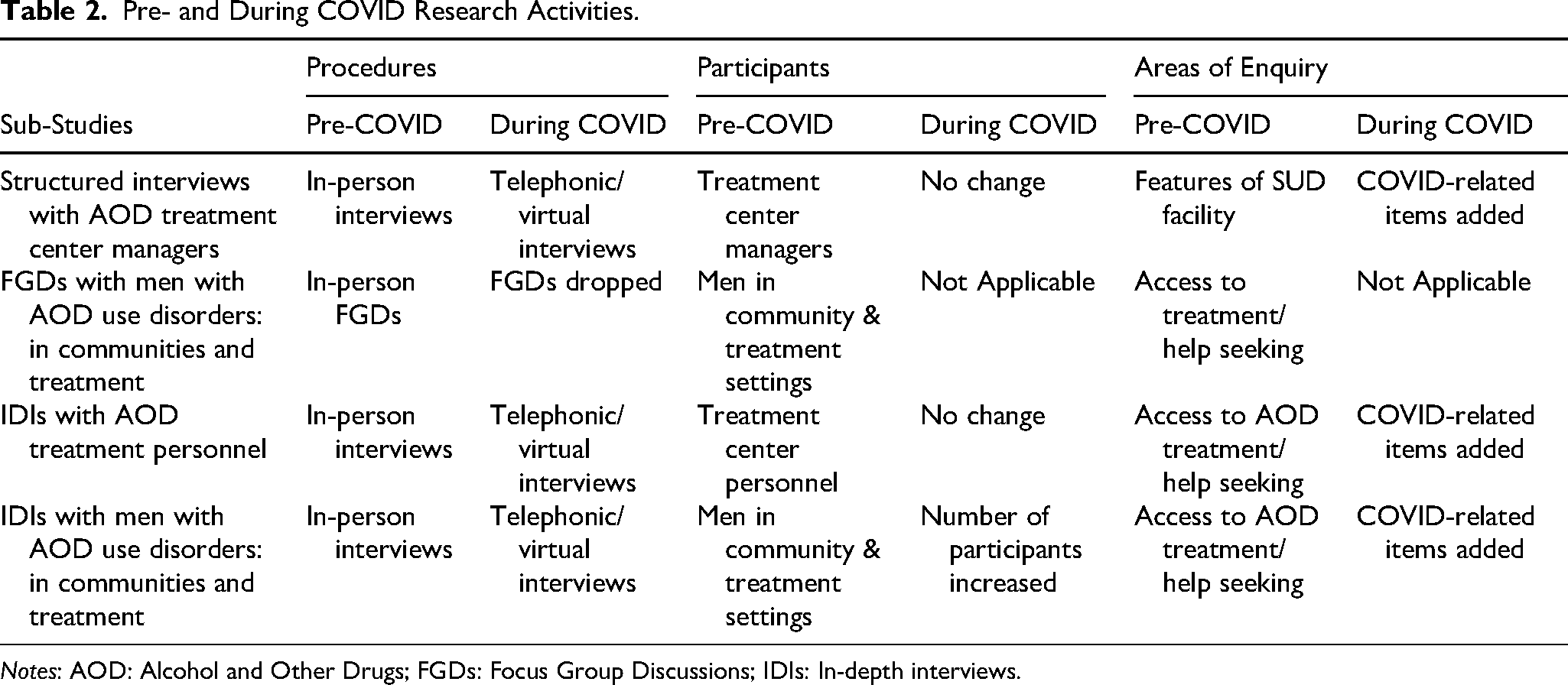
Notes: AOD: Alcohol and Other Drugs; FGDs: Focus Group Discussions; IDIs: In-depth interviews.
Method
The research was granted ethics approval by the relevant research ethics committees (RECs) of all the participating institutions.
We applied the Emanuel framework, and, for each ethical principle, we asked the following three questions to assess the impact of the methodological changes on the study: (a) In what ways was this ethical principle affected by the methodological changes?; (b) How did we address the ethical shortcomings that were brought about by the methodological changes, if any?; and (c) What are the lessons learned from conducting AOD research during the COVID-19 pandemic?
We answered these questions based on two main sets of information: (a) advice received from some of our RECs, and to a greater extent (b) our deliberations during our regular investigators’ meetings and ad hoc meetings that were held to discuss the COVID-related challenges. For example, with the team of investigators being based in different provinces, we had somewhat different COVID-19-related risk perceptions which we attributed to the regional differences in prevailing COVID-19 infection rates and AOD patterns (for example Gauteng's COVID-19 rates were generally higher than those of the Eastern Cape province). Furthermore, investigators’ personal circumstances also influenced our decisions. For example, one of the investigators was infected with COVID-19 at the time of the deliberations and was particularly risk averse relative to some of the other team members. Initially, we recommended site-specific approaches to address the COVID-19 challenges, but in the end, we decided to adopt the same approach of suspending in-person data collection across all sites in order to preserve a favourable risk-benefit ratio, and to apply equivalent study methods across sites.
Results
In this section we describe how our adherence to each of the eight principles was affected by the methodological changes that were required as a result of the COVID-19 restrictions. Table 3 summarizes the results in terms of the eight principles.
How Adherence to the Ethical Principles was Affected Following Changes in Study Methods.
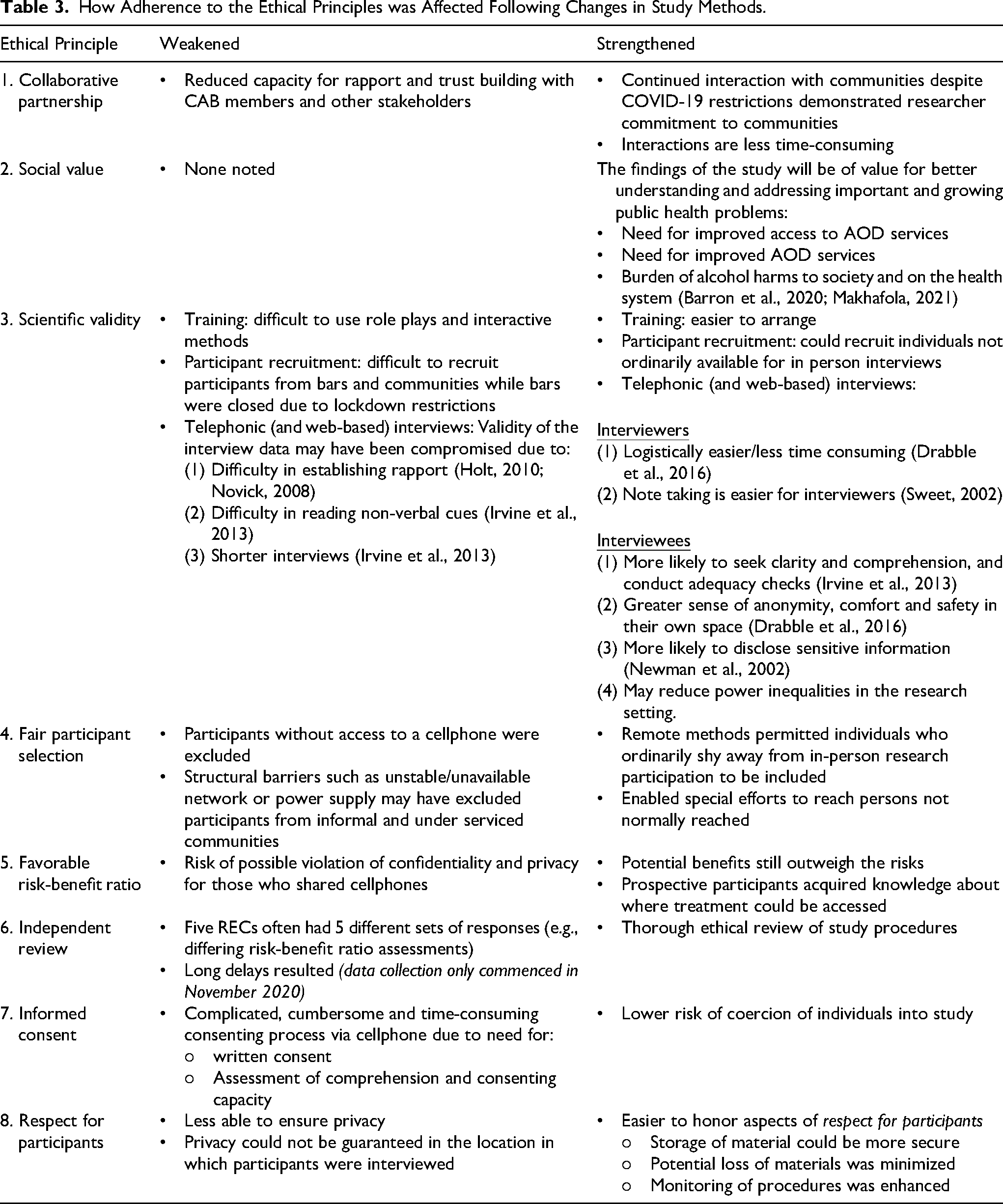
Collaborative Partnership
In keeping with the collaborative partnership principle, in 2019 when the study commenced, we formed a national advisory committee (NAC) comprising policy makers and researchers, as well as CABs from all four sites. After the imposition of lockdown restrictions, we interacted with the NAC and CAB members remotely by cellphone or other devices. Continued meeting with the CABs helped to maintain trust and demonstrate our consistent commitment to hearing community voices despite the major social disruption imposed by COVID-19 restrictions. We provided CAB members with data bundles to facilitate virtual meeting attendance. In one site (WC) we had established a good rapport prior to the pandemic and so responding as we did was important for maintaining trust. Although we considered seeking approval to depart from isolation protocols and hold some CAB meetings in-person (with full personal protections), this strategy was not needed in the end as we managed to maintain good relationships with the CABs and target communities despite the mostly virtual format of engagement.
Virtual and telephonic methods were not ideal, on the other hand, for recruiting men with AODs through bar managers (a key gatekeeper group), since bars were closed due to lockdown restrictions. Some bar managers indicated their preference to interact with our project staff in-person in order to be able to verify their authenticity, and in one site they reported that people masquerading as researchers had been robbing alcohol serving establishments in their areas. Being from the study communities, the CAB members were very helpful in facilitating the process of recruitment as in some instances we would give them the names of the selected facilities, and they then made the first contact with the bar managers on our behalf.
The main lessons learned from this experience are that virtual meetings with community members who had access to appropriate devices and data bundles were feasible and worked well during the pandemic. Holding regular virtual meetings also contributed to maintaining the trust that had been established before the pandemic. Advantages and disadvantages of different community engagement approaches that can be adopted while employing physical distancing during data collection have been discussed for other contexts in sub-Saharan Africa (e.g., Myers, van der Westhuizen, et al., 2021; Tindana et al., 2020). Our study complements the work of Rossouw and colleagues (2024) in highlighting the important role of CABs and other community stakeholders in helping researchers navigate the additional ethical complexities imposed by the COVID-19 pandemic.
Social Value
The study's social value, given its topic of access to AOD treatment, increased during COVID-19. The lockdown restrictions exposed the vast inequalities in access to health care and the need for improved AOD service provision and better understanding of treatment access in SA. Public awareness of alcohol-related harms was greatly amplified during COVID-19 (Matzopoulos et al., 2020) and the SA government demonstrated an unprecedented level of commitment to addressing alcohol harm, often against the interests of the alcohol industry. During the most restrictive periods when COVID-19 infections were high, only limited movement was allowed, the sale of alcohol was prohibited, and most places of trade were closed. This resulted in some people accessing commercially produced alcohol illegally from existing supplies or supplies that were smuggled across the border, while some people brewed their own alcoholic beverages at home, with evidence of sporadic cases of negative consequences (Smit et al., 2021; Theron et al., 2023). A study among people with TB noted heavier consumption of alcohol compared to their pre-pandemic levels among those who were able to access it (Myers, Carney, et al., 2021) despite the widespread benefits for the public health system. Such benefits included marked decreases in alcohol-related trauma and emergency cases which assisted the burden hospital workers were carrying while managing COVID-19 infections and admissions (Chu et al., 2022; Van Hoving et al., 2021). The lockdown restrictions thus highlighted how health services were chronically impacted by alcohol consumption (Barron et al., 2020).
Our study was also of increased social value given that many unintended consequences of the lockdown restrictions – job losses, stress and mental distress – exacerbated AOD use and the risk of AOD use disorders (Spagnolo et al., 2020). Our inclusion of CABs whose members were drawn from the study communities and well informed about the study processes also provided added advantages of distributive and social justice.
Scientific Value
The implications of the amended research methods for the study's scientific value were variable and concerned four main aspects of the study: (a) the dropping of FGDs; (b) staff training; (c) the recruitment of participants remotely; and (d) the remote interviewing.
The dropping of the FGDs is likely to have reduced the richness and completeness of the information collected from the target population. This is because group interactions during FGDs can unearth rich information on community and societal norms about health issues that is less likely to be elicited via individual interviews (Acocella, 2012).
Although the training of study personnel via a remote format proved feasible and acceptable to trainers and trainees, this format was not entirely ideal for conducting role plays and other interactive activities particularly since most of the staff attended the training using cellphones. As cellphones have small screens, and training interactions involved multiple individuals, this may have prevented field staff from engaging fully and hence diluted their training experience.
Accessing and recruiting participants via cellphone was challenging and risked yielding a skewed sample since individuals who did not have access to a functioning cellphone were excluded from participation. Although the rate of cellphone ownership is high in SA (ICASA, 2020), cellphone ownership is still associated positively with socio-economic status (SES) (Miyajima, 2020), and cellphones may sometimes be a useful commodity to sell or exchange for drugs by some persons with AOD use disorders. Hence the study may have excluded the views and experiences of men of lower SES and men with more severe AOD use disorders; people for whom treatment access is generally most difficult. Additionally, the validity of the information received could have been reduced if confidentiality and privacy were compromised for those who used shared cellphones.
Conducting qualitative interviews remotely was generally feasible and acceptable, and our experience confirmed many of the advantages and disadvantages of telephonic interviewing compared with face-to-face interviewing that have been noted in the literature. Among advantages for interviewees are that they may (a) be more inclined to seek clarity and comprehension, and conduct adequacy checks over the telephone than during in-person interviews (Irvine et al., 2013); (b) have a greater sense of anonymity and comfort when answering questions in their own space (Drabble et al., 2016; Tremblay et al., 2021); and (c) be more likely to disclose sensitive information (Newman et al., 2002). For interviewers, note-taking of non-verbal factors that cannot be audio-recorded may be easier telephonically since they do not need to maintain eye contact with interviewees (Sweet, 2002). Furthermore, virtual interviews may reduce power inequalities between participants and researchers (Surmiak et al., 2022; Tremblay et al., 2021) and allow both interviewers and interviewees to feel safer (Nel et al., 2017).
Conversely, conducting interviews telephonically can have at least three disadvantages compared to in-person interviews. Firstly, not being in the same physical space can hinder rapport-building with participants (Holt, 2010; Myers, Carney, et al., 2021; Myers, van der Westhuizen, et al., 2021; Novick, 2008), which is particularly important for research among vulnerable participants whose behaviors may be stigmatized. Secondly, interviewers are less able to read non-verbal cues that may let them know when to prompt, probe or rephrase questions for clarity and comprehension (Irvine et al., 2013). Telephonic interviews also require more “effortful listening” due to limited availability of non-verbal feedback (Irvine et al., 2013). Lastly, compared to face-to-face interviews, telephonic interviews tend to be shorter due to respondent fatigue and their more formal mode of interaction (Irvine et al., 2013).
Fair Selection of Participants
Although a concern, digital exclusion due to reduced ownership of cellphones among men from lower SES groups was likely to be minimal in our study, given that 95% of households in SA are reported to have a cellphone and smartphone penetration is estimated at 90% (ICASA, 2020). As discussed above, only a minority (about 10%) of prospective participants referred into our study reported that they did not have cellphone access. We reduced digital exclusion by providing data bundles and airtime to prospective participants who had access to cellphones. Positively, the virtual mode of recruitment and data collection allowed us to include people who would ordinarily choose to not participate in studies in-person and is consistent with the call to include people who use AODs in research (Striley et al., 2008). However, some participants from lower income and under-serviced communities may have faced structural barriers such as an inability to charge their device in areas with inadequate power supplies or unstable cellular networks (Hoeflich et al., 2022).
Favorable Risk-Benefit Ratio
Originally the study's main potential risks to participants involved primarily emotional discomfort or distress when answering sensitive questions, while the main benefits were increased knowledge about men's access to AOD treatment in SA. With the emergence of COVID-19, the possibility of COVID-19 infection from in-person contact with the study personnel presented a new risk which was heightened for our target group given that individuals with AOD use disorders have an increased risk for COVID-19 infection and illness severity (e.g., Bailey et al., 2022; Dubey et al., 2020; Forsyth et al., 2022; Hasin et al., 2022). However, this risk was mitigated as we were compelled to suspend in-person data collection from March 2020 until the ban was lifted completely in order to preserve a favorable risk-benefit ratio.
A key benefit that emerged for participants was better knowledge about where to access treatment. In one under-served site, many prospective participants indicated that one of their main reasons for participating in the research was their desire to know where to access AOD treatment. All participants were provided with resource cards with contact details of AOD counselling and treatment services in their areas.
Independent Review
Our multi-site study underwent independent ethics review from the participating institutions’ RECs. These RECs had somewhat differing risk-benefit assessments of our amendments in response to the COVID-19 pandemic, which may have resulted from the RECs’ lack of guidance from the national body (NHREC) on how to handle research studies during the earlier days of the pandemic and their prioritisation of research protocols involving pandemic-related studies (Burgess et al., 2023, 2024). The updated guidelines published in 2024 (National Health Research Ethics Council, 2024) now include a section with guidance for RECs during public health emergencies.
There were delays in final ethics approval being granted for our ethics amendments. These lengthy delays were compounded by the need for parallel approvals from all the sites’ RECs. A preferable approach would be to use a single REC for multi-site studies, in line with the NHREC policy which permits reciprocal recognition of review decisions (National Health Research Ethics Council, 2024). However, in this study, some institutions’ RECs preferred to retain the responsibility of conducting ethics reviews of their own researchers’ studies rather than handing over the ethics review to other HRECs during the COVID-19 period. One REC did grant reciprocity to another REC involved in the study. The National Health Research Ethics Council (2024) guidelines suggest that reciprocal review is useful under pandemic conditions (p. 50), and can be implemented in a systematic manner with appropriate standard operating procedures (SOPs) in place to guide the processes.
Informed Consent
The remote informed consent process was complicated and time-consuming. Specifically, in compliance with national requirements for written acknowledgement of informed consent in SA (Department of Health, 2004) which were in effect at the time, and before the publication of the 2024 National Health Research Ethics Council guidelines on ethics in health research, we were compelled by one of our RECS to ask individuals who had provided verbal consent to participate in the study to also send a cellphone text message confirming their consent to participate in the study. We also assessed individuals’ capacity to consent telephonically by using the University of California, San Diego Brief Assessment of Capacity to Consent (UBACC; Jeste et al., 2007). This process was particularly important for prospective participants who may have been under the influence of AODs, and although this assessment over the phone was cumbersome, like others (e.g., De Vries et al., 2020), we found it to be feasible.
Respect for Participants
The remote format of recruiting and engaging with participants presented new ways of honoring and monitoring respect for participants. For example, since we audio-recorded most staff-participant interactions, we were able to verify whether the interviews were conducted respectfully. In addition, remote working minimized the chances of confidentiality breaches through the loss of study materials that may occur when study personnel transport research materials and devices from study sites to the research offices. In our study, the field staff were required to store the study materials in locked cabinets, and password protected electronic devices. On the downside, the remote format did not allow us to ensure that participants and research staff were in private spaces during the interviews, as would be possible with face-to-face interactions; a limitation identified in the literature as a potential obstacle to participation (Hoeflich et al., 2022).
Discussion
This paper uses a multi-site study on access to AOD treatment among men in four regions of SA to illustrate ethical and methodological challenges that arose from the COVID-19-related lockdown restrictions and discusses the implications of the changes for our adherence to the eight ethical principles for biomedical research outlined by Emanuel and colleagues (2004, 2008). The lockdown restrictions and our study's amended methods had both negative and positive effects on our study's ethical integrity as assessed in terms of Emanuel et al.'s ethical principles (2008).
Despite the changes we were able to maintain high levels of ethical integrity in terms of three ethical principles in particular: social value, favorable risk-benefit ratio, and respect for participants. The potential societal benefits of the study increased during COVID-19 given (a) the increased need for AOD treatment services that was precipitated by COVID-19 lockdowns (where access to substances, particularly alcohol was limited), and (b) the likely increased use of AODs and emergence of AOD use disorders as a result of pandemic-related stressors (Spagnolo et al., 2020). Potential personal benefits accrued to some individuals as they learned for the first time through our study where to access AOD treatment. The virtual or telephonic mode of data collection, with improved recording potential, facilitated ethical compliance by enabling closer monitoring of study procedures, better security of research materials, and increased physical safety for both field staff and participants.
In terms of the remaining five principles, the effects of these procedural changes were mixed. While the use of remote communication methods prevented optimal rapport building with prospective CAB members (collaborative partnership) and participants, adaptation enabled us to consolidate trust relationships with existing stakeholders, a very important element of trust-building in AOD (Hoeflich et al., 2022) and other research (Rossouw et al., 2024) during the pandemic. This is particularly important for interpreting vulnerability in a research context so as to avoid under- or over-estimating risks based on assumptions or stereotypes about the group and ignoring community capacity and agency (Peter & Friedland, 2017).
We put substantial effort into establishing rapport with the community partners in order that we could get to know them and earn their trust. Ironically, with the shared experience of dealing with COVID (a shared universal emergency) and the consequences of COVID restrictions that affected everyone, the pandemic may have helped to build trust by breaking down barriers between the community, people who used substances and the research team. During COVID, the usual social norms and ways of communicating that enhance trust relationships, such as greeting through a handshake or hugs or other non-verbal communication aids or cues, were all affected. However, we were able to create other means of building trust through authentic gestures of care like enabling space for expressing grief after the loss of a CAB member to COVID. Our experience was that trust could be built by research with “heart”, and if we engaged with “people as people”, thereby allowing for authentic engagement.
For study participants, the remote format eliminated the need for travel, lessened participant burden and provided them with a greater sense of anonymity than would have been possible with face-to-face methods. Accordingly, those who may have been hard to reach in-person may have become easier to reach. Study personnel who did not have to travel to sites with high crime rates were also at reduced risk of harm (Nel et al., 2017). The scientific validity and fair selection of participants were likely impacted by exclusion of participants who did not have access to a cellphone or other mobile devices, but evidence suggests that those numbers are relatively low (ICASA, 2020).
Our study highlighted some additional ethical considerations that are not necessarily addressed by the Emanuel framework. For instance, while the framework may propose universality of the principles, there may be a need for context-specific guidelines to support the implementation of the framework in contexts with differing social norms and values (e.g., Sutrop et al., 2020). Furthermore, the relative importance of the eight Emanuel et al. principles may vary across contexts and study designs. In our study there was a heightened need for the respect for persons principle to be adhered to, given the stigmatized nature of SUDs in general, and during the pandemic in particular. In South Africa, measures to address alcohol withdrawal following the sudden termination of alcohol availability were not put in place for people with AUDs. Similarly, little was done to assist people whose access to drug supplies and drug services (such as needle exchange programmes and other harm reduction services) was limited by the lockdown restrictions (Harker et al., 2022).
Finally, the COVID-19 lockdown restrictions led to serious cases of deprivation, social isolation and stress, and reduced opportunities for income generation (as discussed above). These factors also had an impact on how we were able to adhere to some of the ethical principles. For example, while the social value of the research was increased as we were able to inform people about resources for SUD treatment, in reality, such treatment resources were limited during the pandemic, especially in marginalised communities.
Best Practices
The learnings from this study can guide improvements in ethical research practice during future health emergencies beyond cases of AOD research during the recent COVID-19 pandemic. When facing major health system challenges in the future, researchers should be able to adapt their methods, using flexibility and innovation, without compromising ethical standards. Our experience was that with appropriate and carefully thought-through responses, many of the anticipated difficulties, such as telephonic interviewing of men in impoverished communities, did not actually materialize. Surmiak and colleagues (2022) have highlighted how an opportunity-orientated approach during COVID-19 enabled social researchers in Poland to increase the innovativeness and flexibility of their research while at the same time ‘democratising’ the research process by giving voice to the participants.
To minimize the limitations of using remote methods to conduct research, the following are recommended. Firstly, in a context of a highly socially disruptive pandemic, it is important to invest in community advisory structures in order to address ethical challenges, since grounded community feedback is particularly critical to making ethical decisions under conditions of crisis. In order to consolidate rapport with community members when establishing CABs, it may be advisable to depart from blanket isolation protocols so as to conduct some meetings in-person while abiding by pandemic-related safety protocols. Secondly, to address the challenges of remote participant recruitment and data collection, a hybrid method may be advisable, involving in-person recruitment (while adhering to pandemic-related safety protocols) followed by virtual methods for subsequent procedures (e.g., Sturges & Hanrahan, 2004). Thirdly, data collection/interviewing via a remote method among some groups (such as the treatment personnel in this study) may be feasible and possibly even preferred (Archibald et al., 2019). Fourthly, use of digital technology for research in low-income settings during situations of public health emergencies given its value and feasibility, and further research on web-based methods for conducting research in LMICs is recommended. Lastly, jurisdictions where informed consent is prioritized in written form may need to develop less rigid methods for respecting participant autonomy.
The COVID-19 lockdown restrictions affected our ability to conduct research as normal, but with innovation and flexibility, AOD research can and should continue to be conducted ethically during major health system stresses such as the COVID-19 pandemic (Brown et al., 2021; Hoeflich et al., 2022). Funders of research should also be flexible in accommodating revised study methods and deliverables (Surmiak et al., 2022).
Research Agenda
As the world enters a phase of pandemic preparedness in response to both climate impacts and anticipated future pandemics (Stern et al., 2021), the need for evidence to inform policy and practices timeously is accentuated. Some have argued that, “rather than treating adaptive evidence-making and decision making as a break from the routine, … this should be a defining feature of an ‘evidence-making intervention’ approach to health.” (Lancaster et al., 2020, p. 477). Hence, having the capacity to be nimble and innovative in overcoming ethical and methodological challenges in research during future crises is crucial. We suggest that a level of meta-research to determine the ethical rigor of research under conditions of public health crises should be conducted to answer questions such as: How valid is informed consent obtained if there is no in-person contact? How do participants experience the impact of changed methods imposed by a public health crisis and can their preferences and understanding influence ethical decision making? How can the institutionalisation of community participation through CABs be made more flexible and responsive to building and maintaining trust through times of rapid change?
Commentators have pointed to the need for research prioritization to address the multiple ethical issues raised for health policy and services by the pandemic and its control measures (Ulrich et al., 2020). To this list, we add the need to study how researchers can still maintain the ethical integrity and scientific rigor required for evidence-based policy and program development under conditions where methodological adaptations are imposed on research, whether related to COVID or otherwise. In South Africa, many pandemic preparedness initiatives continue to be underway for public health research, and the incorporation of a new section specifically dedicated to research under pandemic conditions in the National Department of Health research ethics guidelines (NDOH, 2024) should help to minimize the challenges that caught us (and the research community in general) off-guard during the COVID-19 pandemic. Other South African publications have also highlighted how RECs might offer better and more appropriate support and guidance during a future pandemic (e.g., Burgess et al., 2023, 2024; Rossouw et al., 2024), as has an international review paper by Salamanca and colleagues (2024). There are also at least two other local or regional initiatives on pandemic preparedness that may have relevance to health research ethics during pandemics. These are the National Pandemic Preparedness Initiative funded by the Department of Trade, Industry and Competition (DTIC) involving all universities in South Africa (Simon, 2024), and the African Pandemic Sciences Collaborative (Science for Africa Foundation, n.d).
Educational Implications
The educational implications of the considerations above are that enhancing training for REC members and researchers on how community responsiveness mechanisms should be strengthened by research would be useful for protocol review processes. Additionally, examples of adaptations to COVID-19 (such as those discussed in this paper), may be used to stimulate ethical reflection on how best to maximize the risk-benefit ratio and meet other ethical requirements on one's research more generally. Training curricula on research protocol writing may also encourage people to make back-up plans that can allow quick adaptations to suit possible unexpected pandemic conditions, should they arise in the future.
Footnotes
Acknowledgements
We are most grateful to Prof Sphiwe Madiba, and Drs Jabulani Ncayiyana and Ziyanda Vundle for collaborating on this project. We also gratefully acknowledge the contributions of Dr Feyi Odunitan-Wayas, Mzamile Zweni and Hedwick Masomera for project management; Lesiba Masango, Lungisa Ndzamela and Naomi Hlongwane for research coordination; and all the interviewers on this project.
ORCID iDs
Ethical Considerations
The research was granted ethics approval by the relevant research ethics committees (RECs) of the participating universities: University of Johannesburg Faculty of Health Sciences (REC-01-78-2019); University of Cape Town Faculty of Health Sciences (HREC REF 301/2019); South African Medical Research Council (EC003-3/2019); Walter Sisulu University Faculty of Health Sciences (023/2019); and the London School of Hygiene and Tropical Medicine (19170-1).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this paper was supported by the South African Medical Research Council with funds received from the South African National Department of Health, and the UK Medical Research Council with funds received from the UK Government's Newton Fund. Dr Sebenzile Nkosi's time for writing this paper was supported by the Fogarty International Center of the National Institute of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
