Abstract
When hospitals ask broad consent for the secondary use of patient data for scientific research, it is unknown for which studies the data will be used. We investigated what patients at a cancer hospital consider to be an adequate level and most suitable method of information provision using questionnaires (n = 71) and interviews (n = 24). A part of the respondents indicated that they would feel sufficiently informed by either being notified about potential further use, or by receiving a general brochure before being asked for consent. Others stated that additional information would be interesting and appreciated. Yet, when discussing required resources needed to provide additional information, interviewees lowered the bar of what they considered minimally required, voicing the importance of spending resources on research.
Keywords
Introduction
Patient data gathered during diagnosis, treatment and follow up are an extremely valuable resource for research, innovation, and improvement of care. Generally, patients support secondary use of data from their electronic health records for research (Aitken et al., 2016; Grande et al., 2013; Richter et al., 2021). However, data processing will always entail small risks for patients, most notably privacy risks (Piasecki et al., 2021; Rocher et al., 2019). With the advancement of technology (e.g., artificial intelligence, data science) and the upsurge of personalized medicine, data of even more patients are systematically collected, linked, and analyzed. The future applications are seemingly endless, and impossible to foresee.
There is an ethical duty to inform patients about research with their data, as laid down in, for example, the Belmont Report and the Declaration of Helsinki (National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research, 1978; World Medical Association, 2013). Patients also voice the wish to be informed (Aitken et al., 2016; Hill et al., 2013; Kalkman et al., 2019; Page et al., 2016; Spencer et al., 2016). Informing patients increases trust, makes patients feel valued and gives the patients a feeling of control over their data. In addition, the General Data Protection Regulation provides a legal duty to inform, irrespective of the legal basis used to process the data for research. In this paper however, we do not focus on how patients should be informed based on the legal basis, we focus on the patients’ wishes. Informing patients benefits research as well: the high level of trust in research increases the patients’ support for using health data in research (Aitken et al., 2016; Kraft et al., 2018), which may increase data availability and research quality.
However, informing patients about every research project separately can be expensive, time consuming, and patients can only absorb a limited amount of information at a time (Dankar et al., 2019). Re-contacting patients may even be impossible, e.g., when the data were gathered a long time ago and the patient is deceased or has moved. Asking consent for unknown future research within a certain range, i.e., broad consent, is a way to inform patients without overwhelming them and is considered acceptable by patients (Garrison et al., 2016; Platt et al., 2014; Richter et al., 2018).
But, at the time of asking broad consent it is often unknown for exactly what specific studies the data will be used. One way to inform patients about the specific studies without having to contact the patients about every research project separately, is to provide the information about several projects combined, when the research is completed (post-research information). Providing post-research information has the additional advantage that it can include research results. Previous research shows that the majority of patients is interested in learning the results (Cook et al., 2020; Franklin et al., 2020; Long et al., 2016; Purvis et al., 2017). Patients indicate that receiving the results enhances a feeling of being respected and involved and is seen by patients as a reciprocal action from the researcher (Beskow et al., 2012).
Although receiving research results has often been investigated, those studies mostly focused on patients that had already been explicitly informed or had to participate actively in clinical trials or prospective and longitudinal studies (Cook et al., 2020; Purvis et al., 2017; Sacristán et al., 2016). Because of active participation, patients may remember study details better than when electronic health record data are re-used. In addition, data can be used often and potentially for a long time (depending on the retention period), even when patients do not visit the hospital anymore. This ‘passive’ participation might alter the informational needs. Therefore, these results might not be generalizable to secondary use of electronic health records. Other research regarding dissemination of results focused on individual results (e.g., Richter et al., 2018; Thorogood et al., 2014), on a specific type of health data, for example genetic data (e.g., Taylor et al., 2021), or on research participants in general, without taking into account which (if any) disease the participant has (e.g., Beskow et al., 2012; Milne et al., 2022).
It is still unknown what patients consider to be an adequate level and type of information about research that will be conducted with their medical data, including the consideration of information provided prior to research (during the broad consent process) and post-research. Our study, therefore, aimed at investigating what patients consider to be an adequate level and most suitable method of information provision and transparency. We focused on patients who suffer from cancer. We will not elaborate on the legal framework, though it goes without saying that the legal parameters will have to be considered in implementing the most suitable method of informing patients. We investigated which topics patients would like to receive information about during the consent procedure, how tailored to the patients’ disease or treatment the post-research information should be, and whether the resources (time, money) providing this information would take alter the patients’ opinion of what is considered minimally required. Additionally, since research with electronic health records often involves data linkage, we zoomed in on the informational needs regarding linkage.
Methods
Patient Inclusion
This study was conducted at the Radiotherapy department of the Antoni van Leeuwenhoek hospital (hereafter: AVL), a specialized cancer hospital and research institute in The Netherlands. Radiotherapy generates a substantial amount of complex and diverse data that can be used in research. Together, patients with prostate carcinoma, mamma carcinoma and rectum carcinoma, that are treated with radiotherapy in the AVL hospital, cover a wide age range and a variety of patient profiles. Patients of these groups who had an appointment at the radiotherapy department of the AVL hospital in the months January till March 2020, were selected. Patients with WHO performance status 2 or higher (i.e., unable to carry out any work; up and more than 50% of waking hours), patients who did not speak the Dutch language sufficiently, and patients who were already enrolled in a questionnaire study were excluded. The goal was to include 125 patients to receive 63 completed questionnaires (expected response rate 50%). Inclusion was halted prematurely due to the first COVID-19 lockdown in the Netherlands, after 117 patients had been included. We refer to Supplement 1 for more details.
Patients received an invitation letter signed by their treating physician, that provided an explanation of the study and a link and login code to an online questionnaire. After three weeks, non-responders received a reminder letter. Both letters mentioned the possibility to request the questionnaire on paper, which three patients did.
Broad Consent in the AVL Hospital
The AVL uses a broad consent procedure for research with medical data and residual biosamples. Before patients are asked to give broad consent, they are informed through a brochure with general information about medical research. The hospital website presents several frequently asked questions and examples of completed studies. If patients want more information, they can click on a link that leads them to the website of the research institute, where the research groups and research themes are explained, and where a list of publications and a scientific annual report can be found.
When a researcher desires to start a new study with health data from AVL patients, they must submit their research proposal to the institutional review board. The institutional review board reviews each protocol and considers whether principles such as purpose limitation and data minimization are adhered to.
Questionnaire
The questionnaire was administered online, using Exploratio. An English translation of the questionnaire is included in Supplement 2. The questionnaire consisted of 38 questions on (factors that might influence) information preferences (see Supplement 1 for details).
This research distinguishes information given prior to the start of the research and information given after research has been completed. This information can be either general or specific (Figure 1).
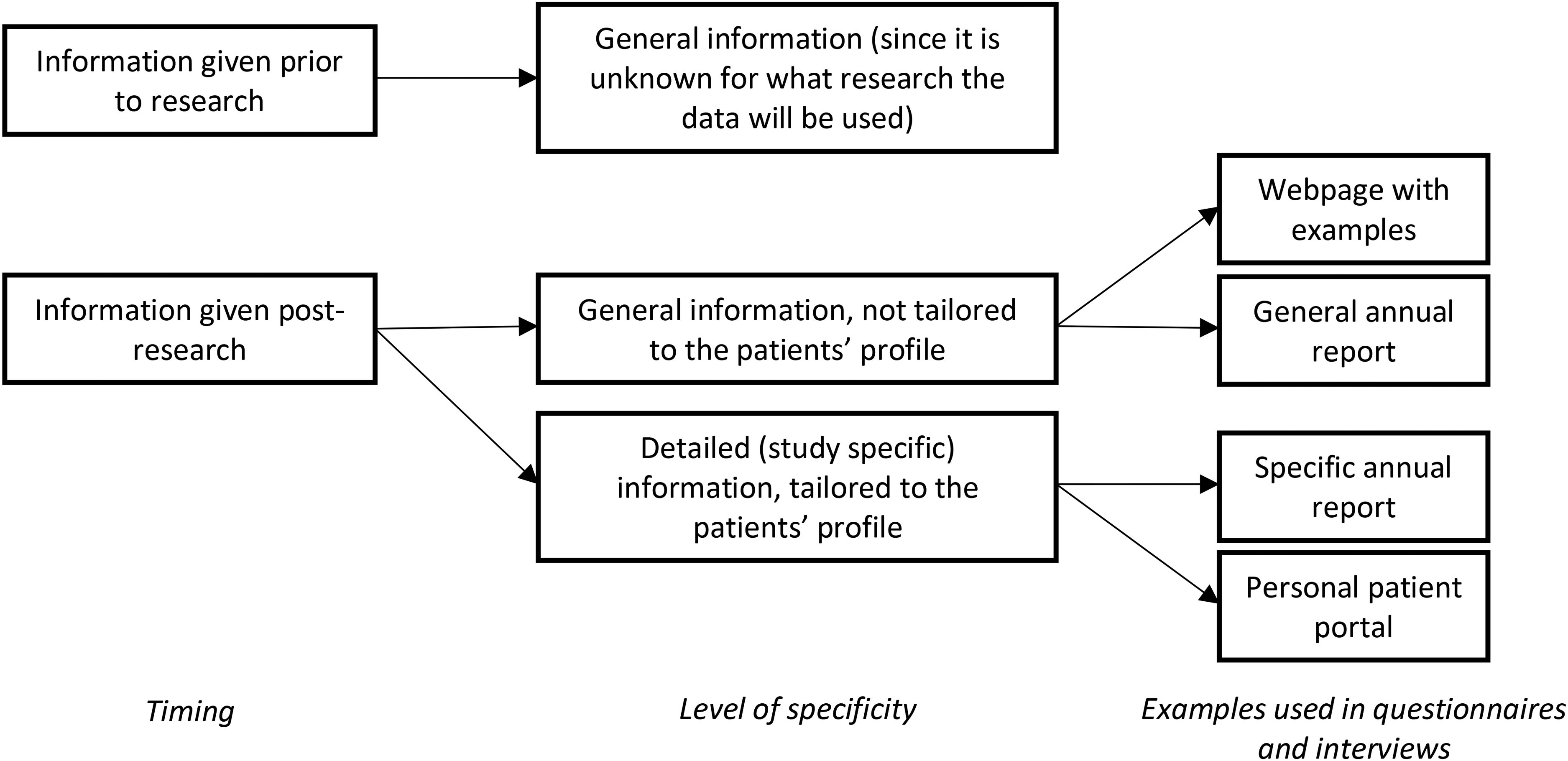
Timing, level of specificity and concrete examples of information provision presented to the respondents in the questionnaires and interviews.
Four different methods of providing information post-research were introduced: (1) a webpage with examples of completed research projects; (2) a general annual report, containing examples of research projects that were completed in the past year; (3) a specific annual report, containing research projects with data of patients similar to the respondent (e.g., the same diagnosis or treatment); (4) adding information to the patients’ personal portal, indicating exactly which research projects have been carried out with the patients' data.
Interviews
To gain more in-depth insight into the patients’ reasoning and thoughts about the subject, (semi) structured interviews were conducted. Topics discussed during interviews were several aspects of information provision: timing, the breadth, level of specificity and content of the information provided, methods of information provision, costs of the methods. Most of these topics were also present in the questionnaire. The costs of information provision and the specific topic of data linkage were only discussed in the interviews, each for their own reasons. Regarding the costs, we wanted to investigate whether patients would take this aspect (on their own initiative) into account when explaining their preferred method of information provision. Regarding data linkage, we assumed this would benefit from a dialogue, to enable giving a more extensive explanation about what data linkage entails, answering questions, and to identify any potential misunderstandings about this complex topic. Consent was also discussed during interviews, however, these results are not described in this paper. An English translation of the interview guide can be found in Supplement 3. The goal was to conduct a maximum of 30 telephone interviews. In the questionnaire, 38 respondents indicated that they wanted to participate in an interview, of which 30 were randomly selected to be interviewed.
Audio recordings of the interviews were made. Interviews were transcribed verbatim (pseudonyms replaced all interviewees’ names) and coded with R using the RQDA package (version 3.5.0). The transcripts were analyzed using standard inductive qualitative methods: a set of codes was derived from the interview guide and literature, which was refined and expanded using ‘in vivo coding’, meaning that the codes were based on themes that arose from multiple readings of the transcripts (Thomas, 2006). A small sample was coded independently by MB and SR and compared to check agreement and to establish the final set of codes. MB read and coded the remaining transcripts.
Ethics Approval and Consent
The IRB of the AVL hospital reviewed and approved the study (reference 19.103/IRBd19064). Data were processed in accordance with privacy regulations (COREON, 2022; Dutch Implementation Act GDPR, 2018; General Data Protection Regulation, 2016; Medical Treatment Contracts Act, 1994) and stored in a pseudonymized way. The introduction of the questionnaire stated that by filling in the questionnaire, respondents consented for the use of their data for this study. Audio recordings of the interviews were made, for which verbal consent by the interviewee was given beforehand.
Access to data (without direct identifiers) gathered in this study may be granted, please contact the corresponding author for conditions. Transcripts will not be shared because of potential re-identifiability.
Results
Respondents’ Characteristics
In total, 71 respondents filled in the questionnaire (response rate 62%) between January and June 2020. Most patients were between 60 and 69 years old (33.8%), married or living together as a couple (77.5%), born in the Netherlands (93%), and retired (46.5%) or employed (42.3%), as shown in Table 1. A comparison of the demographic characteristics of all (N = 63025) patients who came to the AVL for the first time between May 2018 and December 2020 showed that the characteristics were similar to that of the hospital population regarding age, country of birth and marital status, except for gender, since 57.1% of the hospital population is female (hospital population data not shown).
Demographic Characteristics of the Questionnaire Respondents (N = 71).

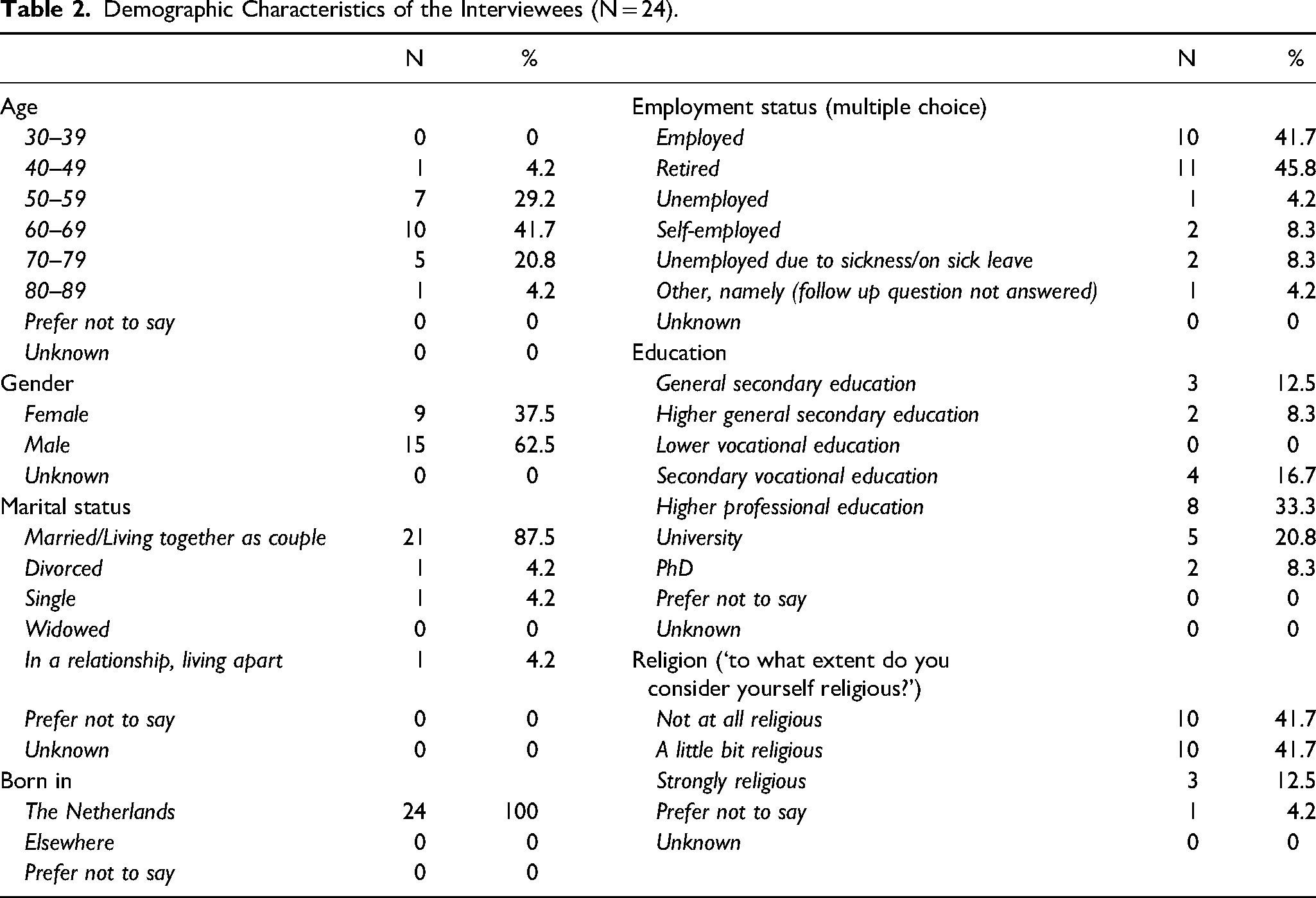
In total, 24 semi-structured telephone interviews were conducted between January and March 2020. Demographic characteristics are shown in Table 2.
Demographic Characteristics of the Interviewees (N = 24).
Interviews were halted in March 2020 due to the COVID-19 lockdown to prevent influencing the results and the goal of 30 interviews was not reached. However, saturation was achieved (i.e., no new topics emerged during the last couple of interviews). The duration of the interviews ranged from 10 to 30 min with an average of 20 min. Figure 2 visualizes the themes and subthemes that arose when interviewees described their wishes regarding information provision.
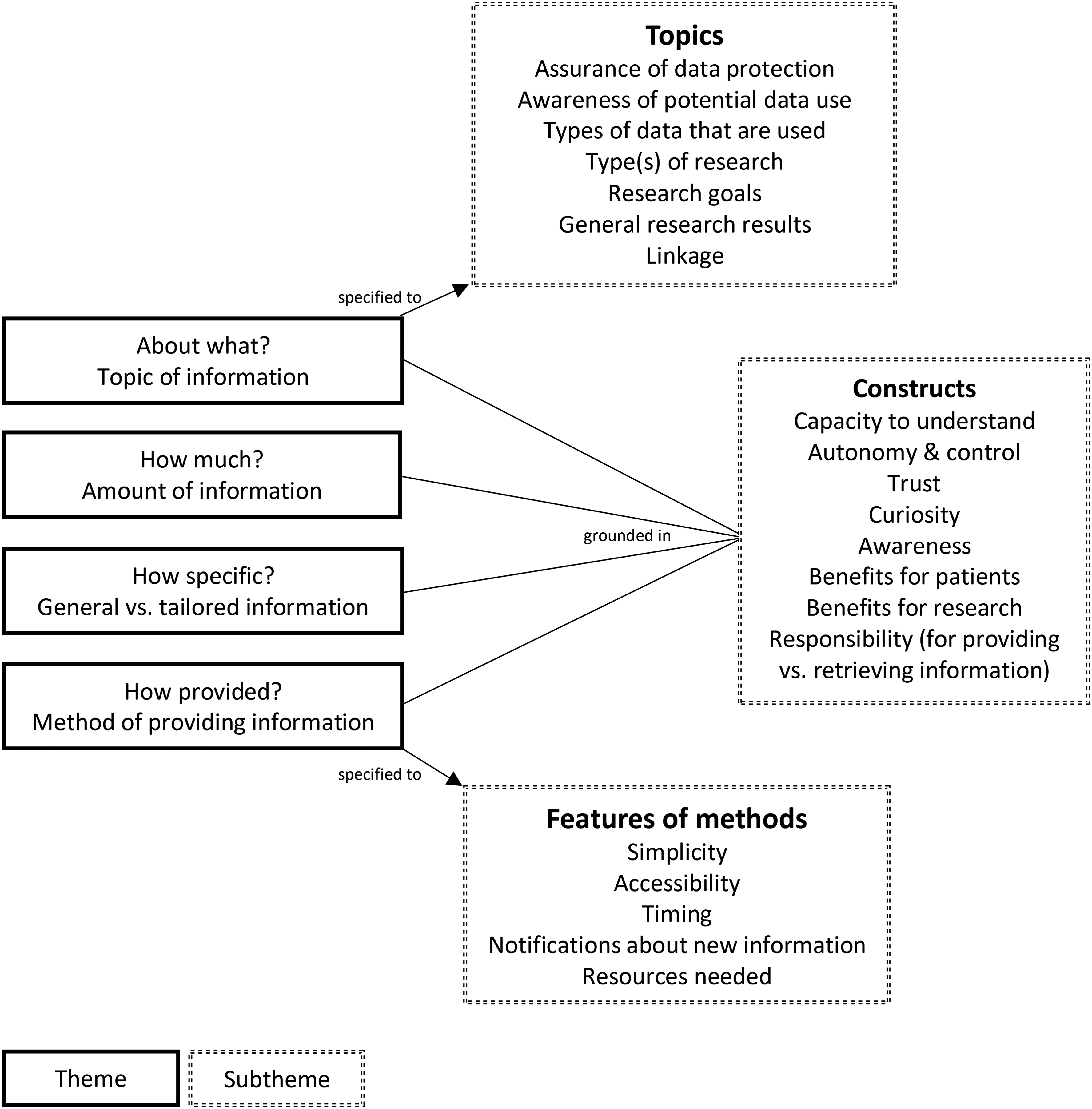
Visualization of inductive qualitative methods: themes and subthemes that arose when interviewees voiced their wishes regarding information given about research.
Below, results of the questionnaire and interviews are described, structured according to topics in the questionnaire and interview guide. For every topic, the first paragraph discusses the questionnaire results and the following paragraph(s) discusses the interview results. For the last two topics, only interview data are available. Quotes are given to illustrate the themes that arose during interviews. Quotes were selected carefully to maximize the representation of the interview content.
Information Given Prior to Research
In the questionnaire, respondents were asked to rate how important it is to receive information about the topics listed (Table 3). Although information about all topics was considered important by the majority of the questionnaire respondents, data protection was most often considered ‘very important’.
Overview of Questionnaire Respondents Scoring the Importance of Receiving Information About Certain Topics (N = 71).

During interviews, data protection and research purposes were considered important topics. Interviewee 23 (female, 50–59): ‘I just want to be assured that my privacy is guaranteed. And that's the only thing for me actually.’. Interviewee 22 (female, 60–69): ‘That there is no name, no address, no zip code, something like that. Well, zip code is okay, but never a name and address. I think that's important.’. Interviewee 24 (female, 50–59) noticed the advantages of mentioning specific examples of research and the achievements of research: ‘Then it becomes a bit more concrete and a bit more attractive to participate’.
Interviewees differed with respect to the amount of information they would like to receive. While some would like to receive information regarding which data are used and who gets access to data, others found it sufficient to only state that research is carried out, for instance because they felt patients already receive a lot of information and they might not understand all of it. Interviewee 3 (male, 60–69): ‘And what research and how and how profound, well… Look, my knowledge and my capabilities do not go that far, to oversee it.’. It was suggested that patients should have a choice in how much information they would like to receive. Interviewee 7 (female, 50–59): ‘In any case, people should have the choice: what do you want to know and what not?’.
Information Given Post-Research
The proportion of questionnaire respondents that indicated that the information given prior to research is sufficient (39.4%) was almost equal to the proportion of questionnaire respondents that would like to receive post-research information (38%) (Questionnaire data, Table 4).
Overview of Questionnaire Respondents Indicating Whether They Would Like to Receive Post-Research Information (N = 71).

During interviews, some of the interviewed patients indicated that they would like to receive post-research information. The majority of interviewees thought post-research information would be nice, interesting, and appreciated, but not required. Interviewee 1 (male, 60–69): ‘It would be nice, but I’m not going to miss it, I guess.’. Interviewee 16 (female, 50–59): ‘Well, I think ‘must’ is a strong word, but I find it interesting myself. So, I would like or appreciate it if they do.’. Other patients indicated that they did not need to receive post-research information. Interviewee 24 (female, 50–59): ‘You give consent, and then you have to be able to hand over control.’. Interviewees explicitly mentioned downsides. Interviewee 17 (male, 70–79): ‘I think it would be far too specific for me to be personally informed. […] My normal life should continue as well.’ Interviewee 24 (female, 50–59): ‘And perhaps there are people who say: ‘I don't want to hear about that’, who want to forget that entire period, that should also be possible.’. On the other hand, interviewee 8 (female, 40–49) emphasized she expected a potential personal benefit: ‘It's going to be on the top of my mind for the next ten years. So, I find it very pleasant to know: what else is possible if it [the cancer] comes back?’. It was suggested that it would be optimal if the hospital takes the patients’ preference into account: Interviewee 17 (male, 70–79): ‘Assess the patients’ needs and provide a targeted response’.
Post-Research Information: General vs. Tailored to Patient Profile (Disease/Treatment)
Post-research information can be general (not tailored to the patient, e.g., examples of completed research on a website) or tailored (e.g., information about research relevant to the patients' disease or treatment), see Figure 1. Table 5 shows that 47.9% of the questionnaire respondents indicated that general information would be sufficient and 35.2% of the questionnaire respondents would like to receive tailored information.
Overview of Questionnaire Respondents Indicating Whether They Would Like to Receive Specific Information (N = 71).

Although the question about general versus tailored information was not meant to focus on the amount of information (just on how tailored to the patient), some patients perceived ‘general information’ as a lot more information than tailored information, namely information about all the research projects that are conducted in the hospital. This assumption about the amount of information showed to be a potential reason to prefer specific information. Interviewee 11 (male, 60–69): ‘The specific information about myself, the colon cancer that I have, with metastases, is relevant to me. I like all of it, the scientific research that the AVL does, but 90% is not important to me [personally].’. Interviewee 14 (female, 60–69): ‘Right now, it is of no use to look at other studies that do not relate to you and may never relate to you.’. Interviewee 16 (female, 50–59) explicitly indicated that having to search for relevant research in the large amount of information would be difficult.
Reasons for deeming general information sufficient were divers. Interviewee 9 (male, 80–89): ‘I trust that it [data] will be handled properly. And even more that you, the hospital, are of course a very good, reliable place to share data.’. Interviewee 3 (male, 60–69) felt he did not have the capacity to understand what specific information would mean for his treatment. He would prefer the experts to decide whether treatment should be adjusted or not. Interviewee 5 (male, 60–69) indicated that it would be sufficient to know that data are used: ‘So just the fact that you know that something will be done with it. I don't need to know what is done, and I don't need to know what the outcomes are’.
Providing Post-Research Information: Four Methods
In the questionnaire, respondents were provided with four potential methods to provide post-research information (see Figure 1). A webpage with examples, a general annual report, a specific annual report and providing additional information about research projects to the patients’ personal portal were considered certainly sufficiently informative by respectively 37.7%, 18.8%, 36.2%, and 60.9% of questionnaire respondents. Therefore, the personal portal is expected to be most informative, with the website as a second-best option. A general annual report is most often considered to be ‘probably not sufficient’ (Table 6).
Overview of Questionnaire Respondents Indicating Whether the Described Method Would be Sufficiently Informative (N = 71).

During interviews, the website and portal were methods preferred, particularly because patients considered the website the easiest method (for the hospital and for the patient) while still being sufficiently informative, and the portal to provide more detailed, tailored information (because this shows research that actually used the patients’ electronic health records).
The methods were designed to be increasingly specific and tailored (with method 1 being the least specific and method 4 being the most specific), but some interviewees based their preference for a method on the type of medium mentioned in the method (website, report, portal), instead of on the level of specificity. For example, interviewee 18 (female, 70–79) said: ‘Well, annual reports are often so tough and so technical. Simple, please (laughs).’. Interviewee 18 (female, 70–79) illustrated an advantage of the website: ‘Anytime you want, you can go there.’. Interviewees pointed out that information could also be of interest to family and friends. Interviewee 7 (female, 50–59) expressed that annual reports implicate having to wait a year; therefore, the website is more accessible.
During interviews, it was mentioned that interviewees would like to receive a notification when there is new information available. Interviewee 20 (female, 50–59): ‘Then it would be nice if I got a notification, like: ‘hello, there is news’. But I assume that is quite labor intensive so that is also a disadvantage. Such an annual report would be nice, then you know: it will be released after the end of the year, or once a year.’. However, others considered that it is not the hospitals’ responsibility to update patients with post-research information. Interviewee 23 (female, 50–59): ‘If you want more information about ‘what happened to my data?’, then you can request it. But that should be an action taken by the patient, in my opinion, and not the hospital.’. Interviewee 13 (male, 60–69): ‘You can expect from people: if you are interested, come, and get that information. Then you [the hospital] just make sure it is findable.’.
One of the differences between the tailored annual report and the personal portal, is that the portal only includes research in which data of the patients themselves is used. Interviewees indicated that this difference did not matter much. Interviewee 6 (male, 70–79): ‘It is still about the same condition that you have yourself, so that is just as interesting.’.
Costs of Informing Patients
This topic was not in the questionnaire, therefore, only interview data are available. Some interviewees mentioned on their own initiative (without the interviewer prompting them) that they took the costs and time into consideration when choosing their preferred method. Interviewee 8 (female, 40–49): ‘I am very fond of logging into the portal […]. So, I would be happy to be able to find information there, but I do not know to what extent that will take a lot of effort, to realize that as an extra feature, for patients. Because I think that requires quite a few extra administrative actions. And I’d rather have the energy to go to the actual research.’.
In some interviews, the interviewer brought up the resources (time, money) involved (see supplement 1 and 3). These patients expressed that research is more important than informing patients. Interviewee 18 (female, 70–79): ‘If it is going to be very expensive, I would say: ‘never mind’. I'd rather have you do research with it, that is what it's ultimately intended for.’. However, two patients indicated that, even though research is more important, this does not mean that patients do not have to be informed. Interviewee 6 (male, 70–79): ‘You see, if it is true that it would take up quite a lot of time and money, it wouldn't really be necessary for me. But I expect, once the research is done, that it is actually quite easy to inform the patient about it and make it known, like you can read the report here.’. Interviewee 16 (female, 50–59): ‘Research is of course paramount, but I think personal information is also very important. That is part of it, so I think it is important that resources are made available for it. Because people ultimately want to know.’.
When asked which of the four methods was minimally required, most patients indicated the website. Interviewee 20 (female, 50–59): ‘The website is minimally required, I think. And everything that is provided additionally, is added value.’. A few patients indicated that an annual report of the research is minimally required. A different method of providing additional information was also suggested (interviewee 8, female, 40–49): ‘I am sure, you also publish internally and in certain journals. You publish there and that is registered. I think it takes relatively less energy and time to make that publicly accessible. So I would really appreciate that.’.
Data Linkage with Other Institutes
Since research with electronic health records often involves data linkage, which may increase privacy risks, patients were asked how they feel about linking their data to other databases. This topic was not in the questionnaire, therefore, only interview data are available. Patients illustrated several benefits, e.g., more knowledge, being able to learn from each other, more complete data, and the advancement of science. The disadvantages that patients indicated were privacy related risks and losing track of where the data are, especially when data are shared internationally. Interviewees stated that the guarantee of data protection is a precondition, for example interviewee 22 (female, 60–69): ‘I find it a bit scary that all this is possible, but on the other hand, it's also good that it is possible. And as long as it is all heavily secured and not used improperly, then, yes, I have no problem with it.’.
The majority of the interviewees wanted to be informed about linkage once. To some, it was essential to be informed, others indicated that it would have been nice, but not essential. Some interviewees indicate that they do not need to be informed. The wish to be informed each time linkage occurred was never voiced during interviews.
Discussion
Our study investigated what patients consider to be an adequate level and most suitable method of information provision and transparency regarding retrospective research with patients’ electronic health records. This study adds to the existing literature by taking into account the whole information procedure, from information given prior to research (during the broad consent process) to information given post-research.
Level of Information – General Versus Specific and Tailored to the Patients’ Profile
The most notable finding is that most patients in this study considered general information about research, not tailored to the patients’ profile (e.g., same disease or treatment), to be sufficient in order to provide broad consent (under certain conditions and for specific types of research) for research with data. However, specific and tailored information was seen as interesting and was highly appreciated. Interviewees differed with respect to the amount of information they were interested in. Therefore, a one-size-fits-all approach is not sufficient, as also indicated by previous research (Taylor et al., 2021). If an institute decides to provide tailored information, it is recommended to use a layered system: actively provide the majority's need to know (general information) and make sure additional nice to know (tailored and specific information) is easily findable.
Previous studies that investigated the desired level of information, often focused on patients that had already been informed explicitly or had to participate actively in clinical trials or prospective studies (Cook et al., 2020; Purvis et al., 2017; Sacristán et al., 2016). Our research indicates that participating in a study in which no active participation is required, patients’ wishes regarding information provision are different. For example, previous research on clinical trials or prospective studies showed that participants are interested in individualized information, to understand how the research affects them, and to be able to compare their results to others (Cook et al., 2020). In our study, although individual research results were out of scope, it did not seem to matter much whether the data of the patients themselves was actually used in a study. General research results of research with patients like them was considered equally interesting.
Most Suitable Method of Information Provision
The Belmont Report states that researchers must provide sufficient information in order to conduct research in accordance with the ethical principles of respect for persons, beneficence, and justice (National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research, 1978). The Belmont Report does not define what ‘sufficient’ is, since this depends on the context and changes over time. According to the majority of the patients with cancer included in this study, a general brochure and website with lay summaries of several examples of completed research is sufficiently informative.
A patient portal that exactly indicates in which research studies the patients’ data is used, was considered most informative by our study participants. However, setting up and running the portal and the infrastructure behind it would require more resources (time and money). These scarce resources cannot be used for research anymore, which is a disadvantage for all patients. Our interviewees indicated that providing information should not take up too much budget that could otherwise be spent on research. This shows that they preferred the benefit for a larger number of people over their own benefit of receiving all the information they were interested in. This shows solidarity and seems to follow the utilitarian perspective by striving for the highest net benefit. Other research showed that respondents preferred a relatively low-cost dissemination method as well (Courbier et al., 2019; Long et al., 2016; Mester et al., 2015; Richards et al., 2016; Taylor et al., 2021). For example, Taylor et al., (2021) showed that very few participants found it acceptable to spend less money on research so that this could be spent on sending research results updates to participants. Therefore, we consider the website, a relatively low-cost method that is sufficiently informative, the best suitable method to inform cancer patients with cancer about research with data from their electronic health records.
Another advantage of a website over a patient portal is the accessibility. Some interviewees stressed that information should be publicly accessible, enabling their relatives and friends to read the information as well. In addition, information should be easily accessible through time, because patients can change opinion when they had time to make up their mind. One interviewee in this study seemed to increase the amount of preferred information during the interview. This might be the result of higher level of consciousness about research because of the interview. Other research also reported a shift in opinion during the study, in several directions (Hill et al., 2013; Jagsi et al., 2019; Willison et al., 2008). This shows it is important to enable patients to take in the information at any moment.
Topics That Patients Should be Informed About Prior to Research
Our results indicated that privacy and data protection are important topics to provide information about. Previous research showed that the assurance of individuals’ confidentiality is a crucial condition for trust, leading to more support for research (Aitken et al., 2016; Franklin et al., 2020; Hill et al., 2013; Hutchings et al., 2020; Kraft et al., 2018; Nobile et al., 2016; Piasecki et al., 2021; Stockdale et al., 2018). This corresponds with our finding that as long as the data are de-identified, patients supported sharing data for research.
In addition, our study participants wanted to be informed about the research purpose, to learn what researchers want to achieve with the research. Informing patients about this is not only useful because patients are interested, but it also prevents the therapeutic misconception (Appelbaum et al., 1987). This is important, considering some interviewees in this study seemed to expect a personal benefit from learning about the research results. It is essential to keep informing patients that there is no direct personal benefit.
Limitations
In the questionnaire and during the interviews, the methods of providing information (website, general or specific annual report, portal) were hypothetical and described concisely, without showing or illustrating the full method. This might have led to different interpretations of what the methods would entail and therefore lowering the validity of the findings. The interviewers were aware of this issue and used interview techniques to enhance validity (Arksey & Knight, 1999). For example, patients were prompted to illustrate and elaborate on their initial responses to get insight into how the patient perceives the method. However, it is likely that not all interpretation differences were surfaced.
The second limitation is generalizability. First, this study was conducted with patients with cancer and is therefore not necessarily generalizable to the broader patient population of a general hospital. Patients with cancer might be more willing to share data for research, compared to the general patient population (Beckjord et al., 2011; Grande et al., 2015), and providing consent is sometimes considered a minor detail compared to receiving the diagnosis of cancer (Williams et al., 2016). In addition, the interviewees volunteered to be interviewed, potentially inducing selection bias. These patients might be more research-minded and therefore not representative for the total patient population of this hospital. Therefore, we did not exactly quantify the interview results, but we illustrated the themes that were identified during interviews, yet did not mention the amount of times an opinion was given. Sometimes, we indicated the proportion, e.g., we explicitly mentioned that the majority of interviewees thought post-research information would be nice, interesting, and appreciated, but not required. We deemed it useful to know that this nuanced view seemed to play a role for a substantial amount of interviewees. However, because our sample might not have been representative, we do not know whether the majority of the patient population feel this way. This could be further investigated in a representative patient sample.
Best Practices
Currently, in the Netherlands, patients sometimes receive limited information about that data from their electronic health records might be used in observational research and how their data are protected. Our results show that patients desire more information, including post-research information with general results. Therefore, we pose the following recommendations based on this study.
Prior to research (during the consent process), patients find data protection an important topic to be informed about. This enhances trust and increases support for research. An example is to explain that research databases do not contain the patients' name. Provide the information in a layered way since patients differ with respect to the desired amount of information. Actively provide the need to know and make sure the nice to know is easily findable. This information should be easily accessible through time, preferably not only for patients, but also for their relatives and the public. A relatively non-expensive and sufficiently informative way to inform patients is a publicly accessible website with a lay summary of several research projects and their general results.
Research Agenda
More research is needed to investigate the desired amount of information in different contexts. This study was conducted with patients with cancer and is therefore generalizable to a limited extent to the broader patient population of a general hospital. Patients with conditions or symptoms that are relatively easy to cure might need more information to see the value of scientific research and provide consent. However, for the same reason, they might be less interested in research.
In addition, this study investigated information preferences based on a short description of hypothetical methods. Future research might benefit from detailed illustrations. Ideally, one or several methods of information provision could be developed together with patients (co-creation). Lastly, future research should investigate whether patient wishes correspond to legal demands, e.g., given by the General Data Protection Regulation and national legislation.
Educational Implications
In our study, patients want to receive post-research information about research (including general research results) in addition to the information provided prior to research. Lay summaries are a potential way to inform patients about the research that is conducted. Awareness of the importance of lay summaries is increasing, and even mandatory for clinical trials since the Clinical Trials Regulation came into force on 31 January 2022. However, awareness is mostly still limited to specific research of which patients are already explicitly informed (see for example Fernandez et al., 2003; Shalowitz & Miller, 2008). Creating a good lay summary is complex (Long et al., 2019). Researchers need guidance, training and collaboration with the communication department and science communicators in translating their results into an easy-to-understand format (e.g., written summaries or animated videos). Research institutes and hospitals should stimulate and facilitate this training and collaboration. Communication departments and science communicators should gather the lay summaries and translate this into a layered system with easily findable information for patients (e.g., a website).
Supplemental Material
sj-docx-1-jre-10.1177_15562646231181439 - Supplemental material for Health Research with Data in a Time of Privacy: Which Information do Patients Want?
Supplemental material, sj-docx-1-jre-10.1177_15562646231181439 for Health Research with Data in a Time of Privacy: Which Information do Patients Want? by Miriam Beusink, Folkert Koetsveld, Sonja van Scheijen, Tomas Janssen, Maarten Buiter, Marjanka K Schmidt and Susanne Rebers in Journal of Empirical Research on Human Research Ethics
Supplemental Material
sj-docx-2-jre-10.1177_15562646231181439 - Supplemental material for Health Research with Data in a Time of Privacy: Which Information do Patients Want?
Supplemental material, sj-docx-2-jre-10.1177_15562646231181439 for Health Research with Data in a Time of Privacy: Which Information do Patients Want? by Miriam Beusink, Folkert Koetsveld, Sonja van Scheijen, Tomas Janssen, Maarten Buiter, Marjanka K Schmidt and Susanne Rebers in Journal of Empirical Research on Human Research Ethics
Supplemental Material
sj-docx-3-jre-10.1177_15562646231181439 - Supplemental material for Health Research with Data in a Time of Privacy: Which Information do Patients Want?
Supplemental material, sj-docx-3-jre-10.1177_15562646231181439 for Health Research with Data in a Time of Privacy: Which Information do Patients Want? by Miriam Beusink, Folkert Koetsveld, Sonja van Scheijen, Tomas Janssen, Maarten Buiter, Marjanka K Schmidt and Susanne Rebers in Journal of Empirical Research on Human Research Ethics
Supplemental Material
sj-docx-4-jre-10.1177_15562646231181439 - Supplemental material for Health Research with Data in a Time of Privacy: Which Information do Patients Want?
Supplemental material, sj-docx-4-jre-10.1177_15562646231181439 for Health Research with Data in a Time of Privacy: Which Information do Patients Want? by Miriam Beusink, Folkert Koetsveld, Sonja van Scheijen, Tomas Janssen, Maarten Buiter, Marjanka K Schmidt and Susanne Rebers in Journal of Empirical Research on Human Research Ethics
Footnotes
Acknowledgements
We acknowledge the support provided by the Trial Bureau of the radiotherapy department of the Antoni van Leeuwenhoek hospital, we thank them for assisting with inclusion of study participants. This study would have not been possible without the support provided by the physicians of the Antoni van Leeuwenhoek hospital, we thank them for inviting their patients for this study. We sincerely thank the patients that participated in the study. We also thank Irith Kist for her review on English grammar and structure of sentences.
Authors’ Contributions
Miriam Beusink, Folkert Koetsveld and Susanne Rebers designed the study with input from the other authors. Miriam Beusink and Sonja van Scheijen gathered the data, Miriam Beusink analyzed the data, all authors contributed to the interpretation of the results and provided comments on the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Antoni van Leeuwenhoek hospital, an institutional grant of the Dutch Cancer Society and of the Dutch Ministry of Health, Welfare and Sport (grant number not applicable).
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
