Abstract
Background
Public involvement enhances the relevance, quality, and impact of research. There is some evidence that public involvement in Australian research lags other countries, such as the United Kingdom. The purpose of the systematic review was to establish the rates and describe the characteristics of public involvement in Australian clinical trials.
Methods
We reviewed evidence of public involvement in all Australian randomised controlled trials published in the first 6 months of 2021. To determine the quality of public involvement, we used the five-item short-form version of the Guidance of Reporting Involvement Patients and the Public, version 2.
Results
In total, 325 randomised controlled trials were included, of which 17 (5%) reported any public involvement. Six trials reported public involvement in setting the research aim and seven in developing study methods. The authors of one study reflected on the overall role and influence of public involvement in the research.
Conclusion
Rate of public involvement in Australian clinical trials is seemingly substantially lower than those reported in countries with similar advanced public health care systems, notably the United Kingdom. Our observations may be explained by a lack of researcher skills in how to involve the public and the failure by major funding agencies in Australia to mandate public involvement when deciding on how to award grant funding.
Keywords
Introduction
There is a consensus that involving the public in research can improve the relevance, quality, and impact of the work.1–3 Public involvement in research can be defined as the work carried out ‘with’ or ‘by’ the members of the public rather than ‘to’, ‘about’ or ‘for’ them.1–4 In public involvement, people who would usually be the target population of the intervention are active partners in all aspects of research, from generating the research question to co-authoring publications. Major research funding bodies, such as the National Institute for Health Care Research (NIHR) 4 in the United Kingdom, mandate public involvement in the research they support. In Australia, the National Health and Medical Research Council and Medical Research Future Fund emphasise the importance of public involvement in the research they support but have not made it a requirement of funding. The authors of a recent Medical Research Future Fund Consumer Reference Panel 5 suggest that meaningful public involvement will be necessary for future funding to align Australian research with international best practice. Despite policy directives encouraging public involvement, studies have shown low levels of participation. For example, Owyang et al. 6 reviewed 475 articles in orthopaedic research and found that only two studies involved the public. Similar results were found in the studies by Jones et al., 7 Patel et al., 8 Gray et al., 9 and Price et al., 2 where almost no reviewed studies had evidence of public involvement.
We identified five studies where there were consistently higher rates of public involvement in Europe (including the United Kingdom) and the United States of America as compared to Australia. For example, Camelo Castillo et al. 10 reviewed 11 pharmacoepidemiology studies that had involved the public and found that most of the work was conducted in Europe and North America (n = 10), with only one study from the Asia–Pacific region. In another review, Pawson et al. 11 examined 71 realist reviews where there was evidence of public involvement. Half of the included studies were from the United Kingdom, 7% were from Australia. Similarly, Zhao et al. 12 reviewed 23 best practice reports related to transitions from hospital to home and found that half of the included reports described some form of public involvement, with only one from Australia. Aguayo et al. 13 reviewed 97 epidemiology studies with public involvement published between 2000 and 2020. Almost two-thirds of included studies were from the United Kingdom, while only 6% were from Australia. Finally, Rouncefield-Swales et al. 14 reviewed 38 studies involving children and young people’s involvement in health research, where over 80% were from the United Kingdom, and only 8% were from Australia.
The objective of this systematic review was to estimate the extent (number of studies that included public involvement in the sample) and quality (calibre and nature of the studies) of public involvement in Australian Clinical Trials published in the most recent 6-month period that the search was conducted – between the 1 January and 31 July 2021.
Methods
Search strategy and eligibility
All sources in this study were identified through MEDLINE (OVID platform) and CINAHL (EBSCO platform). Both were searched on 18 August 2021. Full MEDLINE and CINHAL search strategies are available in Supplementary Document 1. Searches were conducted by an information scientist. We adopted a two-stage approach to searching databases. Stage 1 focused on identifying all clinical trials published between 1 January and 31 July 2021, using Cochrane filters typically employed to refine a subject-specific search to identify randomised controlled trials. 15 At Stage 2, we applied filters so that studies were only included if the author information in the manuscript stated they were affiliated with an Australian institution.
We included Australian randomised controlled clinical trials that we defined using the following criteria: the research was consistent with the World Health Organisation 16 definition of a clinical trial (prospective assignment of human participants or groups of humans (…) to a health-related intervention to evaluate the effects on health outcomes); participants or groups were randomly allocated to two or more interventions using any recognised true randomisation procedure; at least 50% of the trial participants were recruited from research institutions (e.g. hospitals, universities, and institutes) in Australia; the first or corresponding author and the trial coordinating centre was an Australian institution; the paper was in English and published in a peer-reviewed journal. We excluded any study testing an intervention in a healthy population. Finally, we excluded studies reporting secondary analysis of clinical trial data, where an analysis of the dataset had been previously published.
Ethical considerations
Ethical approval for this study was not required because data were extracted from publicly available sources or published research. The study was registered on 22 July 2021 prior to searches being conducted (https://osf.io/87qfv/). Registration was with the Open Science Framework which is an extensively used repository of the Centre for Open Science. After registering our review, we amended our definition of an Australian clinical trial, clarifying that we would not include studies involving healthy volunteers. In addition, we revised our study protocol to extract published trial protocols (that were cited in included manuscript) as an additional source of information about public involvement in included studies.
Lived experience academic involvement
All authors in this study have a commitment to the inclusion of lived expertise in research, consistent with the Medical Research Future Fund perspective that different perspectives and lived experiences that consumers bring to research improve its quality, relevance, and impact. 5 The study was instigated by R.G., who does not identify as a lived experience researcher. A.H.N., T.Z., and C.B. are employed as teaching and research academics and have lived experience of medical conditions. All four research team members were involved in all stages of the project and held decision-making roles.
Data collection process
The results of our searches were exported by T.Z. to ENDNOTE and then uploaded into COVIDENCE. Title and abstract and full-text screening were undertaken by two researchers (T.Z., R.G., A.H.N., or C.B.). Where there were conflicts, either a third researcher reviewed the paper or the two original reviewers met to reach a consensus decision. A full bibliography of included studies is available as Supplementary Document 3. We used the short-form version of the Guidance for Reporting Involvement of Patients and the Public, version 2 18 to determine the quality of public involvement in included trials. The Guidance for Reporting Involvement of Patients and the Public, version 2 comprises five areas: (1) the aim of public involvement in the study, (2) methods used to involve the public in the study, (3) the result of public involvement in the study, including both positive and negative outcomes, (4) the extent to which public involvement influenced the study overall, and (5) reflections and critical review of public involvement.
Public involvement requirement of funding bodies
We generated a list of funding bodies that had supported included studies (Supplementary Document 4). We manually checked the website of each to determine if public involvement was a recommendation or requirement of funding applications.
Journal discipline coding and author guidelines
We used the Journal Citation Reports 19 categories to code journals into broad discipline areas. The author guidelines for publishing journals (for the primary manuscript only) were searched to determine if the journal had any requirement for reporting public involvement.
Results
Study selection
Figure 1 shows the flow of papers through the study. A total of 3595 articles were screened. However, 325 trials met our criteria and were included in the study (Supplementary Document 2). A list of studies excluded at full-text screening is available as Supplementary Document 5. Supplementary Document 2 also shows extracted data for all included studies.
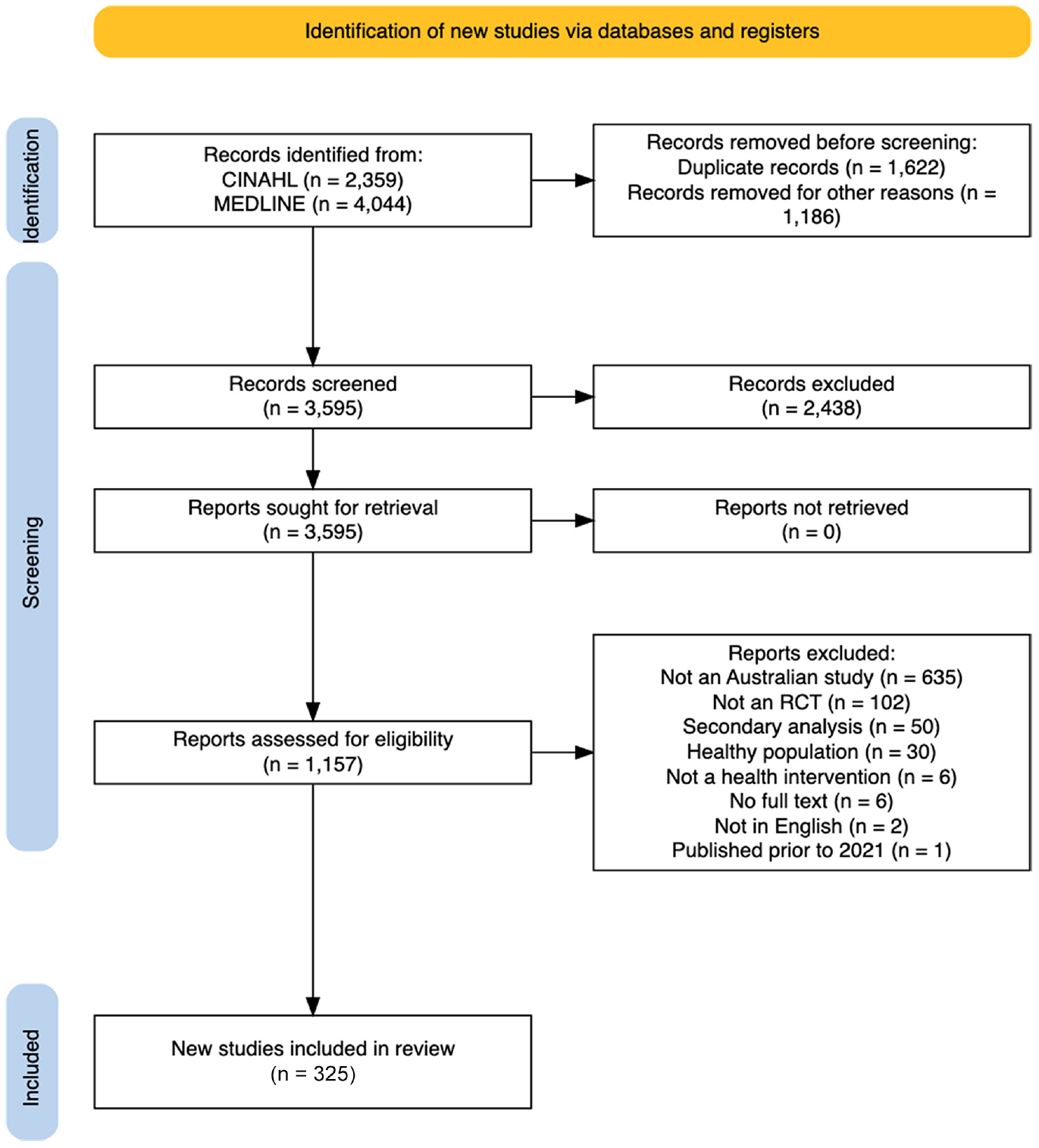
The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA).
Public involvement in trials
Table 1 provides details of the studies examined and the extent of study involvement in terms of various study characteristics. As the aim of this study is to assess the extent of public involvement in randomised controlled trials, we focused on studies that had public involvement and analysed these as a sub-section of the broader sample. Detailed information is available in Supplementary Document 2.
Summary of included studies.
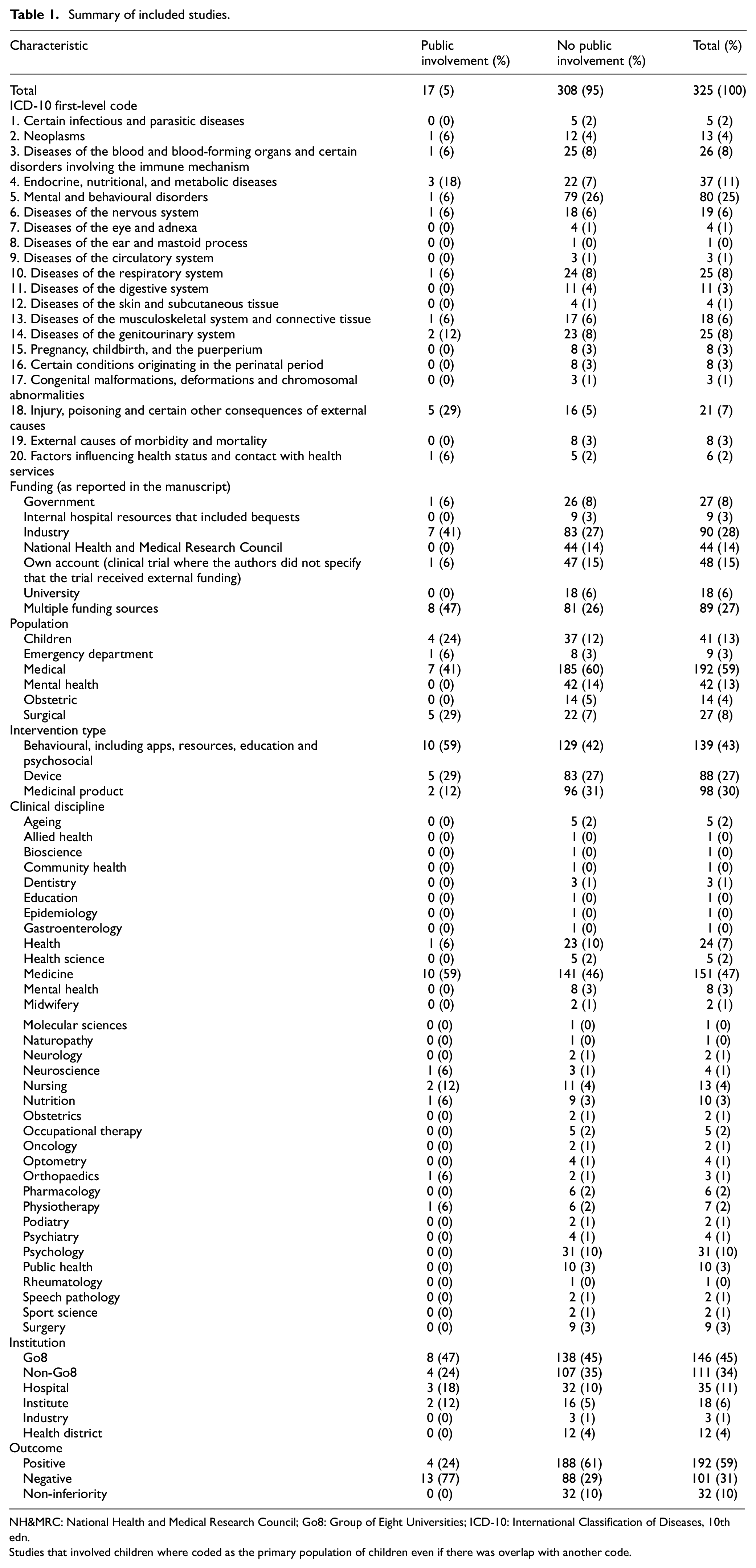
NH&MRC: National Health and Medical Research Council; Go8: Group of Eight Universities; ICD-10: International Classification of Diseases, 10th edn.
Studies that involved children where coded as the primary population of children even if there was overlap with another code.
We did not identify any trials where the lead investigator identified as a lived experience researcher. Participants in included clinical trials were thanked for their contribution to the research in roughly a third of reports (118, 36%). Any form of public involvement in research was reported in 17 (5%) studies. Three trials (1%) had a co-produced intervention.20–22 Three trials specifically indicated that there was no public involvement.23–25
Guidance for reporting involvement of patients and the public, version 2 – short-form guidelines
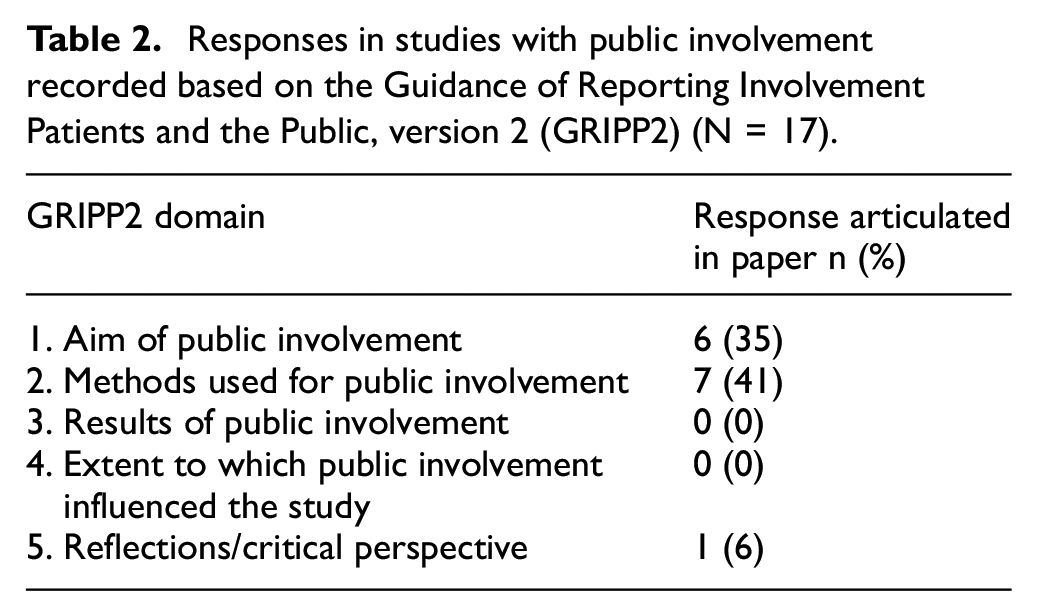
Table 2 shows the Guidance for Reporting Involvement of Patients and the Public, Version 2 for the 17 trials where there was public involvement. The authors of six trials reported the aim of public involvement in the research. Seven trials described the methods used to involve the public in the research. The results of public involvement in the study and the extent to which public involvement influenced the study overall were not described in any of the included trials. There was reflection and a critical perspective of public involvement reported in one trial.
Responses in studies with public involvement recorded based on the Guidance of Reporting Involvement Patients and the Public, version 2 (GRIPP2) (N = 17).
The low number of studies with public involvement and the lack of detailed reporting in the 17 studies that did use it has limited the capacity for aggregate analysis. For example, Franklin et al. 26 reported having public involvement in their study. Under the heading ‘Patient and public involvement’, the authors report that parents were asked to provide consent for their children to take part in the study – this is not an example of public involvement, which refers to engaging members of the public as experts on the health conditions being studied. 5 That said, this section of the manuscript goes on to indicate that parents of children affected by the medical condition in question were consulted prior to the start of the study, and ‘their comments and feedback’ were integrated into ‘the design of the study and how it would be performed’. The manuscript does not include any information on how this information was collected (e.g. via a consumer steering group or through informal consultation with one to two members of the public), what suggestions were made by members of the public, or how these were incorporated. In addition, this publication did not include the appropriate information to address any of the five criteria in the Guidance for Reporting Involvement of Patients and the Public, version 2 (GRIPP2).
A study by Janda et al. 27 did not have a public involvement section in the manuscript; however, the acknowledgements section includes reference to a consumer researcher who provided ‘assistance in protocol development and preparation of information material to patients’. This person did not meet the requirements for authorship. More information on the extent of this assistance would be required to understand how consumer knowledge impacted the conduct of the study and to satisfy GRIPP2.
A paper by McIntyre et al. 22 was one of the three included studies that had a co-produced intervention. Members of the public were engaged to identify content for the intervention and were contacted through an existing consumer group. In the section on public involvement in the manuscript, the authors include information about randomisation, which is not consistent with GRIPP2.
These examples show that papers that did have public involvement had a paucity of clarity of what the public involvement consisted of. Despite these gaps in practice and reporting, all authors that did have public involvement – noting that it was just 17 authors out of 325 included studies – should be commended.
Public involvement requirements of funding bodies
There were 308 funding agencies that supported the research included in this study. Supplementary Document 4 is a list of all funding bodies and any requirements for public involvement as part of the funding application. Public involvement was not a requirement for any funding agency. The Allied Health Professions’ Office of Queensland, Alzheimer’s Australia Dementia Collaborative Research Centre, Bupa Foundation, Cancer Australia, Heart Foundation Australia, Kidney Health Australia, Liaison Committee for Education, Research and Innovation, Central Norway, and the Victorian Cancer Agency requested or recommended public involvement should be part of any application to them. The website of 201 (65%) funding agencies did not provide sufficient information to determine if public involvement was a requirement for funding.
Journals
Included trials were published across 244 journals from 54 Journal Citation Report categories (Supplementary Document 6). Seven journals included in their author guidelines (BMJ Open, Emergency Medicine Journal, British Journal of Sports Medicine, Heart, Journal of Orthopaedic & Sports Physical Therapy, Nursing in Critical Care, Developmental Medicine, and Child Neurology) a statement that public involvement in the research should be described in the published manuscript. All journals that requested a statement about public involvement indicated that they would still publish articles where there was no public involvement. Nursing in critical care stated that public involvement was essential, but only for quality improvement initiatives and service evaluations. All other journals had no requirement for public involvement reporting.
Discussion
The aim of this systematic review was to determine the extent and quality of public involvement in Australian randomised clinical trials published in 2021. We found evidence of public involvement in roughly 1 in 20 studies. Where there was evidence, this tended to focus on statements reporting that the public was involved in the research but included few specific details about how their involvement had directly impacted trial design, conduct, or reporting.
Our observation confirms that rates of public involvement in Australian research are low compared to countries with similarly advanced health systems.12,13,28 By the way of comparison, Lang et al. 29 reported that of 591 papers from the United Kingdom and published in BMJ Open– where reporting of public involvement is a requirement of publication – almost half (45%) included some degree of public involvement.
In the United Kingdom, NIHR has a positive position on the importance of public involvement in the research that they fund. NIHR review panels that arbitrate on the allocation of research funds also actively include members of the public. 4 In Australia, the National Health and Medical Research Council, while positive about public involvement, is seemingly more nuanced in tone and does not require evidence of public involvement in the research they support. 30 The emergence, in 2015, of the Medical Research Futures Fund in providing substantial funding for medical research has resulted in more positive and directive messages around the importance of public involvement. For example, the Medical Research Future Fund consumer reference panel 31 recently published ‘Principles for Consumer Involvement in Research’. 5 The Medical Research Future Fund also has a specific funding stream for consumer-led research. It will, however, take several funding cycles for these strategy changes to be evidenced in published research.
It is plausible that institutional and researcher ambivalence as to the benefits of involving the public in research may also be an important influence on the rates of public involvement. For example, a qualitative study of 36 health researchers by Boylan et al. 32 found that many were reticent to engage in patient involvement.
A paucity of training on how to involve the public in research may contribute to the reported low rates of public involvement in Australian clinical trials. Researchers that state that they are generally positive about involving public in research also say that they struggle with how to do this in practice.32–34 A survey of clinical trial networks and coordinating centres found that only a quarter who responded to a survey provided training to investigators of how to involve the public in their research. 35
Limitations
Work on included studies was likely started 4–5 years prior to the results being published. Consequently, we cannot claim to be reporting a snapshot of current practice. A more up-to-date indicator of levels of public involvement could be obtained by reviewing protocols for funded clinical trials; however, these are not always published and can be challenging to identify. Our work is predicated on an assumption that if authors involved the public in their research, they reported that they did this in the outputs from the study (trial protocol and results paper). It is plausible that authors did involve the public but did not report this; most journal author guidelines do not require that public involvement was reported. It is also not a requirement within current CONSORT guidelines 36 to report public involvement.
Conclusion
Despite widespread recognition of the importance of public involvement in clinical trials, Australia lags some considerable way behind other countries. This is likely explained by a combination of factors that include the failure of funding agencies to mandate public involvement and hesitancy and a lack of skills among researchers. We recommend that funding bodies should have a clear statement about requirements for public involvement in the work they support. Research institutions should provide training to research about how to authentically involve the public in their research. As a nation, we need to embrace public involvement to enhance the relevance, quality, and impact of the research we do.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-1-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Supplemental Material
sj-docx-2-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-2-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Supplemental Material
sj-docx-3-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-3-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Supplemental Material
sj-docx-4-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-4-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Supplemental Material
sj-docx-5-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-5-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Supplemental Material
sj-docx-6-ctj-10.1177_17407745231224533 – Supplemental material for Public involvement in Australian clinical trials: A systematic review
Supplemental material, sj-docx-6-ctj-10.1177_17407745231224533 for Public involvement in Australian clinical trials: A systematic review by Tessa-May Zirnsak, Ashley H Ng, Catherine Brasier and Richard Gray in Clinical Trials
Footnotes
Acknowledgements
The authors thank Elizabeth Lawrence, Information Scientist, for assisting with search strategy and early data organisation.
Author contributions
R.G., C.B., and A.H.N. contributed in conception and design. R.G., T.Z., C.B., and A.H.N. involved in data extraction and analysis. R.G. wrote a first draft of the article. C.B., T.Z., and A.H.N. edited and contributed to revisions of the article. All authors read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval for this study was not required.
Consent for publication
Not applicable.
Transparency statement
The Lead author affirms that the article is an honest, accurate, and transparent account of the study being reported, that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Availability of data,code and other materials
All data related to this study are available in the article or as supplementary document.
Availability of data and materials
A list of included studies and the data extraction table are available as supplementary documents.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
