Abstract
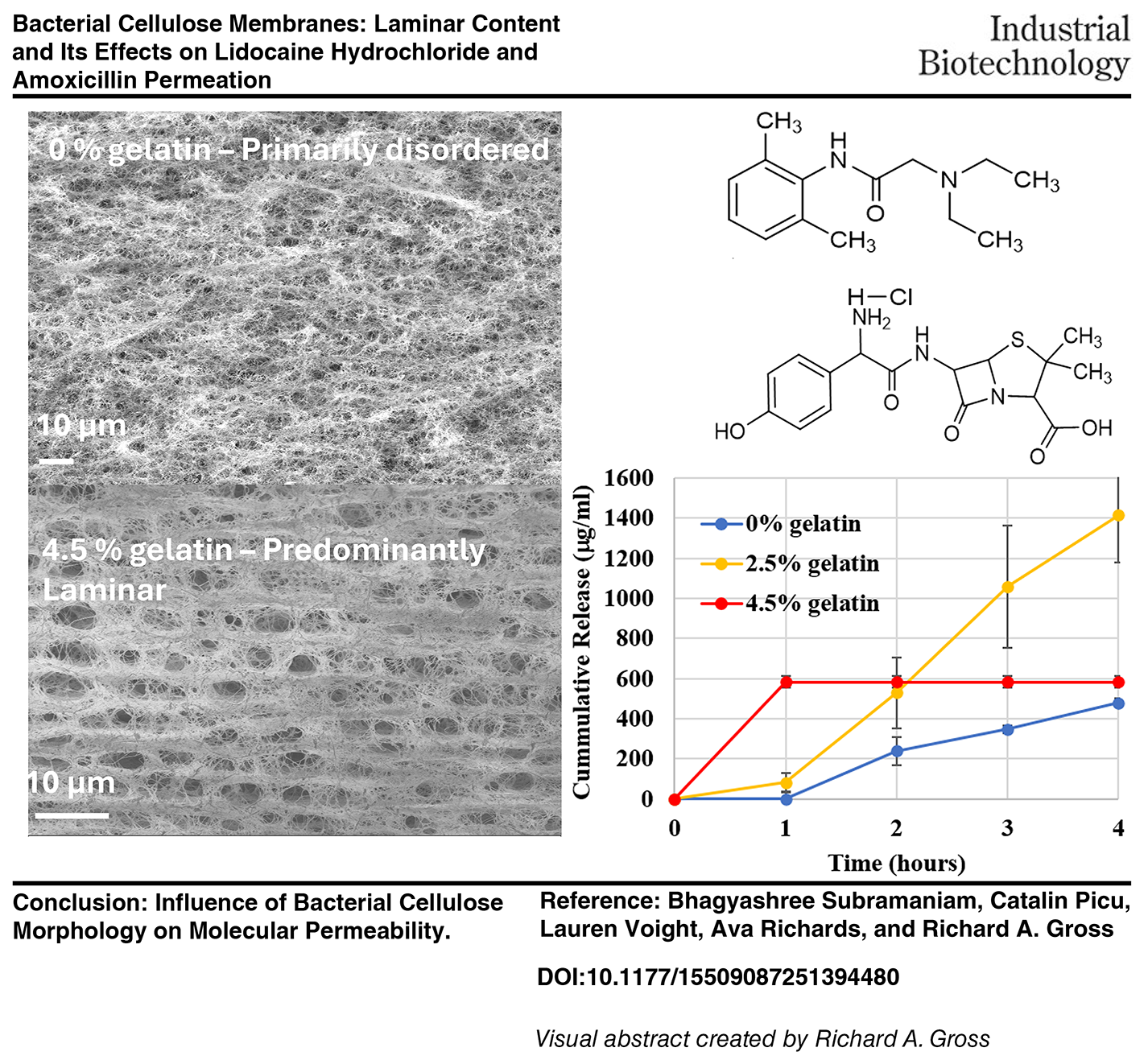
Bacterial cellulose (BC) chains are extruded through pores in cell membranes that self-assemble into nanofiber ribbons that form 3D microporous cellulose networks. A challenge during BC manufacturing is the control of nanofiber morphology that determines BC network properties. Generally, BC is characterized by measuring fiber density, water holding capacity, mechanical strength, porosity, and crystallinity, each of which is a cumulative average of contributions from multiple regions within the matrix. Also, it is common practice to define BC matrix morphology by scanning electron microscopy images taken from one or a few selected matrix regions. Consequently, information on the extent of matrix heterogeneity is absent. In this work, the order of BC dense layer deposition was altered by including additives in the growth media during BC formation by Komagataeibacter xylinus ATCC 700178. Gelatin, known for impacting the directional uniformity of dense layers, was included in culture media at concentrations of 0%, 2.5%, and 4.5% v/v, creating differences in matrix viscosities and corresponding changes in matrix order. The full area of 2-by-1 mm cross-sections was analyzed from three replicate BC matrices. Random selections of 36 300 × 500-μm areas within cross-sections at upper, middle, and bottom matrix regions were analyzed for dense-layer directional uniformity by ImageJ. Laminarity ranged from 15% to over 90% for 0% and 4.5% gelatin, respectively. The diffusion of lidocaine hydrochloride and amoxicillin across BC membranes was analyzed using a Franz cell. Plots were constructed for cumulative release (µg/mL) versus time, and the flux was determined from the slope when release reached the steady state. Unlike the 0 and 2.5 BC membranes, after the first hour of flow, no further flow of lidocaine hydrochloride and amoxicillin into the receptor compartment was observed. This is attributed to the high laminar content in 4.5 BC membranes. The large effects of BC morphological characteristics and laminar content on molecular diffusion highlight the importance of defining matrix morphology.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
