Abstract
Backgrounds. Transcranial direct current stimulation (tDCS) is a non-invasive brain stimulation technique for the treatment of several psychiatric disorders, eg, mood disorders and schizophrenia. Although tDCS provides a promising approach, its neurobiological mechanisms remain to be explored. Objectives. To provide a systematic review of animal studies, and consider how tDCS ameliorates psychiatric conditions. Methods. A literature search was conducted on English articles identified by PubMed. We defined the inclusion criteria as follows: (1) articles published from the original data; (2) experimental studies in animals; (3) studies delivering direct current transcranially, ie, positioning electrodes onto the skull. Results. 138 papers met the inclusion criteria. 62 papers deal with model animals without any dysfunctions, followed by 52 papers for neurological disorder models, and 12 for psychiatric disorder models. The most studied category of functional areas is neurocognition, followed by motor functions and pain. These studies overall suggest the role for the late long-term potentiation (LTP) via anodal stimulation in the therapeutic effects of tDCS. Conclusions. tDCS Anodal stimulation may provide a novel therapeutic strategy to particularly enhance neurocognition in psychiatric disorders. Its mechanisms are likely to involve facilitation of the late LTP.
Introduction
Transcranial direct current stimulation (tDCS) is a non-invasive method that modulates neural activities in the brain by delivering low-amplitude currencies (usually no more than 2 mA) over a short time period (no more than 30 min) between electrodes, ie, anode and cathode. At least, one of the electrodes is placed on the scalp, through which electronic currents penetrate the skull to enter the brain and facilitate or inhibit spontaneous neural activities in the vicinity of electrodes. 1 In recent years, the potential ability of tDCS to treat psychiatric disorders and its mechanisms of action have been extensively studied.1–4 The effectiveness of multi-sessions, rather than a single session tDCS has been reported to ameliorate several symptoms of psychiatric disorders. 2 For example, meta-analyses have shown small to moderate effects on depressive symptoms in patients with acute depression (Hedges’g = 0.37), 5 as well as moderate to large effects on hallucinations (0.86), 6 negative symptoms (0.41), 6 and working memory (0.49) 7 in patients with schizophrenia.
Improvements of psychiatric symptoms by multi-session tDCS may be mediated through several mechanisms, including long-term potentiations (LTPs), continuous enhancement of signal transduction between neurons, and related neural events. 2 Specifically, LTPs have been shown to enhance the synthesis of various proteins, such as neurotransmitter synthases, receptors, ion channels, and intracellular signal proteins, thus facilitating the efficiency of neurotransmissions in cortical circuits. 2
Direct investigations of the cellular and histologic modifications in using animals are useful for exploring the mechanisms of tDCS, 8 especially for those involving LTPs. Animal studies are also useful for testing a variety of parameters of stimulation, eg, safety range, threshold for brain damage, and so on, which would help plan effective clinical research. 8 In addition, basic experiments allow us to test various disease model animals to discover novel indications for tDCS. In view of increasing considerations on preclinical evidence, we herein provide a systematic review of animal experiments to explain the ability of tDCS to ameliorate some cardinal symptoms of psychiatric disorders, eg, cognitive impairment.
Materials and Methods
Data Source and Search Terms
From inception to April 8, 2021, YY and TS independently examined the PubMed databases. They conducted manual searches of reference lists in relevant publications. The following search terms were used as keywords: (“transcranial direct current stimulation” OR “tDCS”) AND (animal OR rat OR rodent). The discrepancies were resolved by consensus of both authors.
Eligibility Criteria
Studies were chosen according to the following inclusion criteria: (1) articles published from the original data; (2) experimental studies in animals; (3) studies delivering direct current transcranially, ie, positioning electrodes onto the skull; and (4) studies reported in English. Exclusion criteria were: (1) experiments applying electric currents invasively, ie, cortical and epidural stimulation; (2) experiments using other forms of electric stimulation, ie, transcranial magnetic stimulation (TMS), alternating current (AC) stimulation, and other forms of non-constant current stimulation; (3) experiments applying electric currents in vitro; (4) studies in humans; and (5) reviews, editorials, protocols, or reporting data extracted from original articles.
Procedures and Data Extraction
Initially, titles and abstracts were screened to identify eligible studies. Full-text articles were obtained for all the studies considered compatible based on the abstract screening and were further reviewed for eligibility.
Results
The initial search provided a total of 598 papers. After screening the title and abstract, 460 articles were excluded, as they were reviews, editorials, or studies in inappropriate fields (299 articles); studies in humans (67); other forms of electric stimulation (59); stimulation in vitro (23); invasive stimulation (8); and articles not in English (4). As shown in Figure 1, 138 articles were further retrieved for full-text assessment. The species of laboratory animals were as follows; rats (99 articles), mice (27), cats (6), monkeys (4), dogs (1), and rabbits (1) (Figure 1).
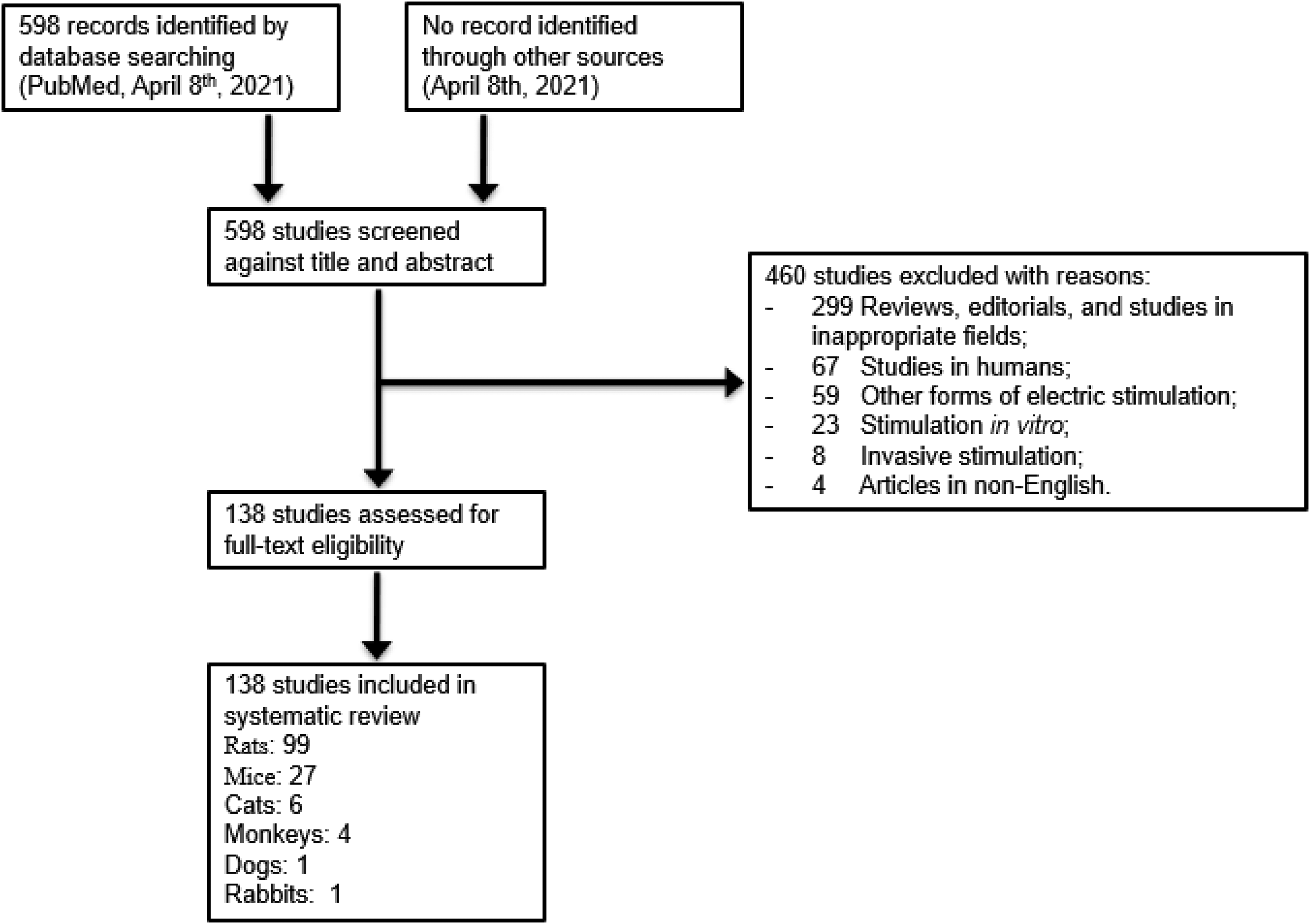
Study selection flowchart.
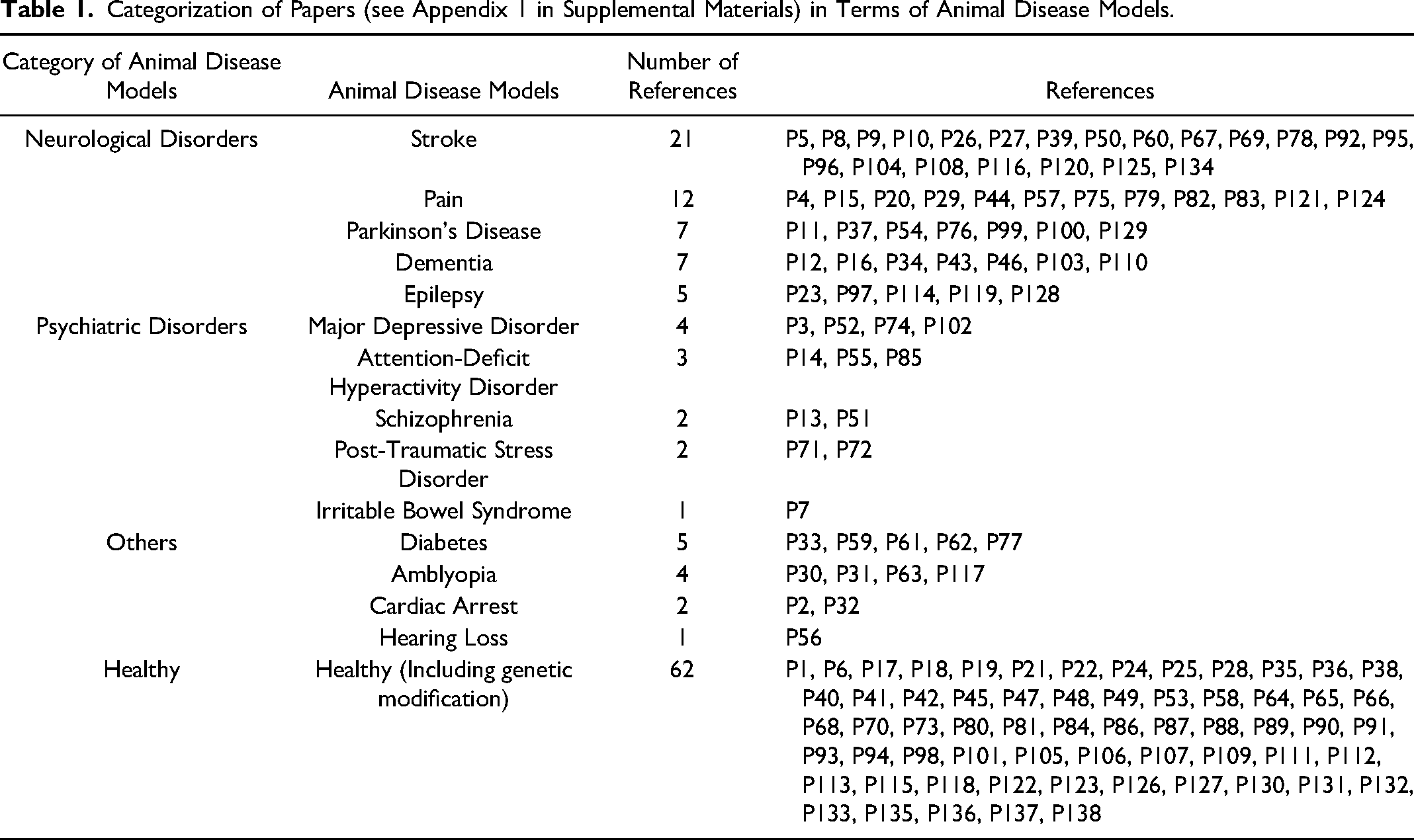
As to the type of models, the most studied was model animals without any manipulations (including genetic modifications) for 62 papers, followed by 52 papers for neurological disorder models, and 12 for psychiatric disorder models (Table 1) (see Appendix 1, in Supplemental Materials). In neurological disorder models, the most studied is stroke was 21 papers, followed by 12 for pain models, 7 for Parkinson's models, 7 for dementia, and 5 for epilepsy. In psychiatric disorder models, the most studied was major depressive disorder with 4 papers, followed by 3 for attention-deficit hyperactivity disorder (ADHD), and 2 for schizophrenia, 2 for post-traumatic stress disorder, and 1 for irritable bowel syndrome.
Categorization of Papers (see Appendix 1 in Supplemental Materials) in Terms of Animal Disease Models.
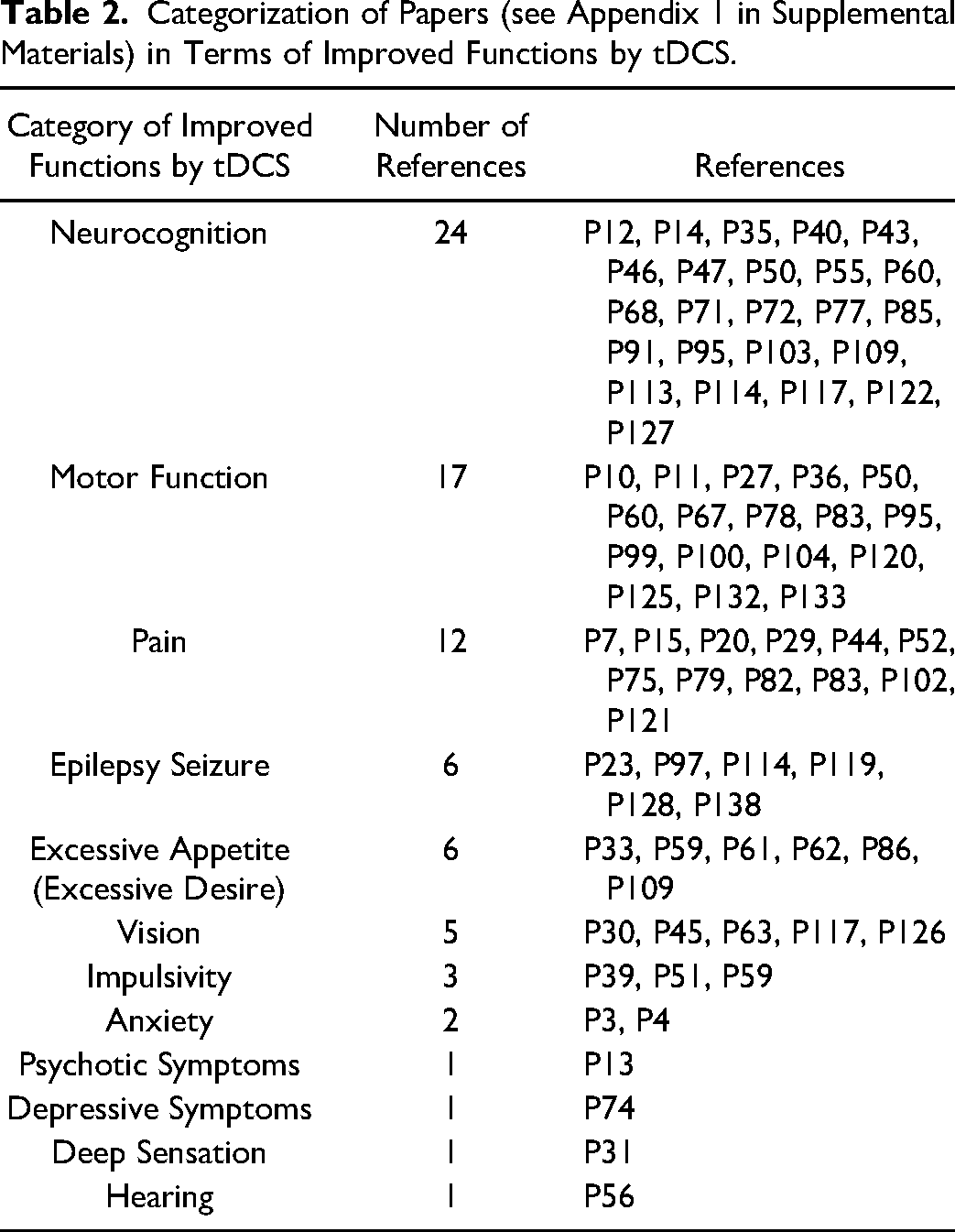
The most studied category of functional areas was neurocognition with 24 papers, followed by 17 for motor function, 12 for pain, 6 for epileptic seizure, 6 for excessive appetite, 5 for vision problems, 3 for impulsivity, 2 for anxiety, and 1 for psychotic symptoms, depressive symptoms, deep sensation, or hearing (see Table 2) (Appendix 1, in Supplemental Materials).
Categorization of Papers (see Appendix 1 in Supplemental Materials) in Terms of Improved Functions by tDCS.
Anodal stimulation enhances glutamatergic transmissions, 9 which leads to insertion of AMPA receptors, usually stored in endosome vesicles, into the postsynaptic membrane. 10 Moreover, Ca2+ flows facilitated by glutamatergic transmissions 9 activate signal cascades, and phosphorylate CREB,11,12 which promotes protein synthesis9,11–15 and establishes the late LTP.9,15–17 LTPs are considered to improve functional connectivity between brain regions18–20 and efficiency of information processing12,13,16,20,21 (see Table 3).
Long-Term Potentiation by Anodal tDCS.
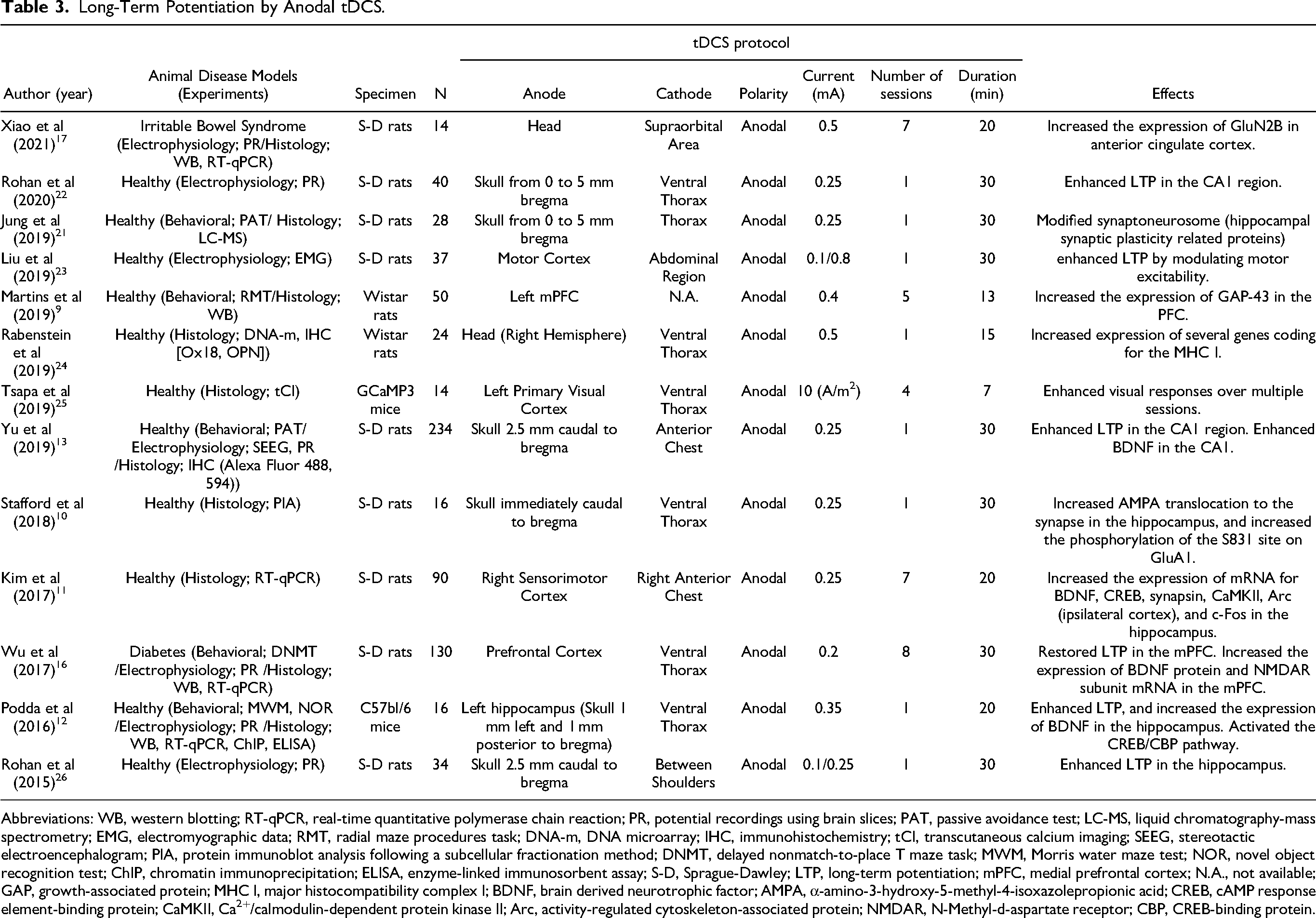
Abbreviations: WB, western blotting; RT-qPCR, real-time quantitative polymerase chain reaction; PR, potential recordings using brain slices; PAT, passive avoidance test; LC-MS, liquid chromatography-mass spectrometry; EMG, electromyographic data; RMT, radial maze procedures task; DNA-m, DNA microarray; IHC, immunohistochemistry; tCI, transcutaneous calcium imaging; SEEG, stereotactic electroencephalogram; PIA, protein immunoblot analysis following a subcellular fractionation method; DNMT, delayed nonmatch-to-place T maze task; MWM, Morris water maze test; NOR, novel object recognition test; ChIP, chromatin immunoprecipitation; ELISA, enzyme-linked immunosorbent assay; S-D, Sprague-Dawley; LTP, long-term potentiation; mPFC, medial prefrontal cortex; N.A., not available; GAP, growth-associated protein; MHC I, major histocompatibility complex I; BDNF, brain derived neurotrophic factor; AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; CREB, cAMP response element-binding protein; CaMKII, Ca2+/calmodulin-dependent protein kinase II; Arc, activity-regulated cytoskeleton-associated protein; NMDAR, N-Methyl-d-aspartate receptor; CBP, CREB-binding protein.
138 papers met the inclusion criteria for the current review. Most studies used rats (71.7%) or mice (19.6%) as laboratory animals. In the classification of animal models, the majority showed non-dysfunctional models (44.9%) and neurological disorder models (37.7%). Among the functional areas improved in the dysfunction models, the most studied was neurocognition (30.4%).
Discussion
In experiments with rats, the electric field of anodal stimulation becomes maximum just below the anode, and rapidly diminishes with the vertical/horizontal distance of the electrodes. 27 These electric fields enhance the excitability of the cortex,28,29 and increase cerebral blood flow.30,31
Cortical regions excited by the electric field also affect neurotransmitters, especially those in the hippocampus. This is because the hippocampus is a hub of neural circuits consisting of the cerebral cortex, striatum, and amygdala. Information flows via these neural connections are mediated by neurotransmitters, such as amino acids and monoamines. 32 Thus, anodal stimulation enhances glutamatergic transmissions, which increase the density of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors 10 and N-methyl-D-aspartate (NMDA) receptors 16 in the hippocampus. In Alzheimer's disease model rats, anodal stimulation increases acetylcholine concentrations in the cerebral cortex and hippocampus. 33 On the other hand, anodal stimulation on the visual cortex decreases gamma-aminobutyric acid (GABA) levels in cats. 34 Since acetylcholine is an excitatory neurotransmitter while GABA is inhibitory, these changes of neurotransmitter concentrations may facilitate excitatory synaptic transmissions.
Anodal stimulation has also been found to regulate serotonin and dopamine transmissions in the reward system of obesity model rats. 35 Since increased dopamine levels in the hippocampus and striatum have been reported in ADHD model rats, 36 anodal stimulation may ameliorate symptoms of some psychiatric disorders by modulating activities of these monoamine neurotransmitters.
Several animal experiments with tDCS have targeted cognitive function (Table 2), some of which report facilitation of long-term memory.12,16,21,37 Accordingly, anodal stimulation has been shown to produce LTPs in recent studies (Table 3). There are two types of LTPs, ie, early LTP and late LTP. The former type lasts for 1 to 3 h, whereas the latter type lasts as long as several weeks. 38 The establishment of early LTP by tDCS may be accompanied with a cascade of events, including release of glutamate into the synaptic cleft to activate AMPA receptors. Subsequent depolarization of postsynaptic membranes attenuates blockade of the NMDA receptors by Mg2+, and facilitates Ca2+ flows into the postsynaptic cells. This leads to activation of the biochemical cascade and insertion of AMPA receptors, usually stored in endosome vesicles, into the postsynaptic membrane, leading to enhancement of glutamatergic transmissions. 10 Further, some researchers have argued that astrocytes 39 and microglia 40 are also involved in the induction of LTP by increasing intracellular Ca2+ concentrations. For example, early LTP has been found to be induced by astrocytic Ca2+ surges across the entire cortex, which depends on activation of α1 adrenergic receptors 39 (Figure 2).
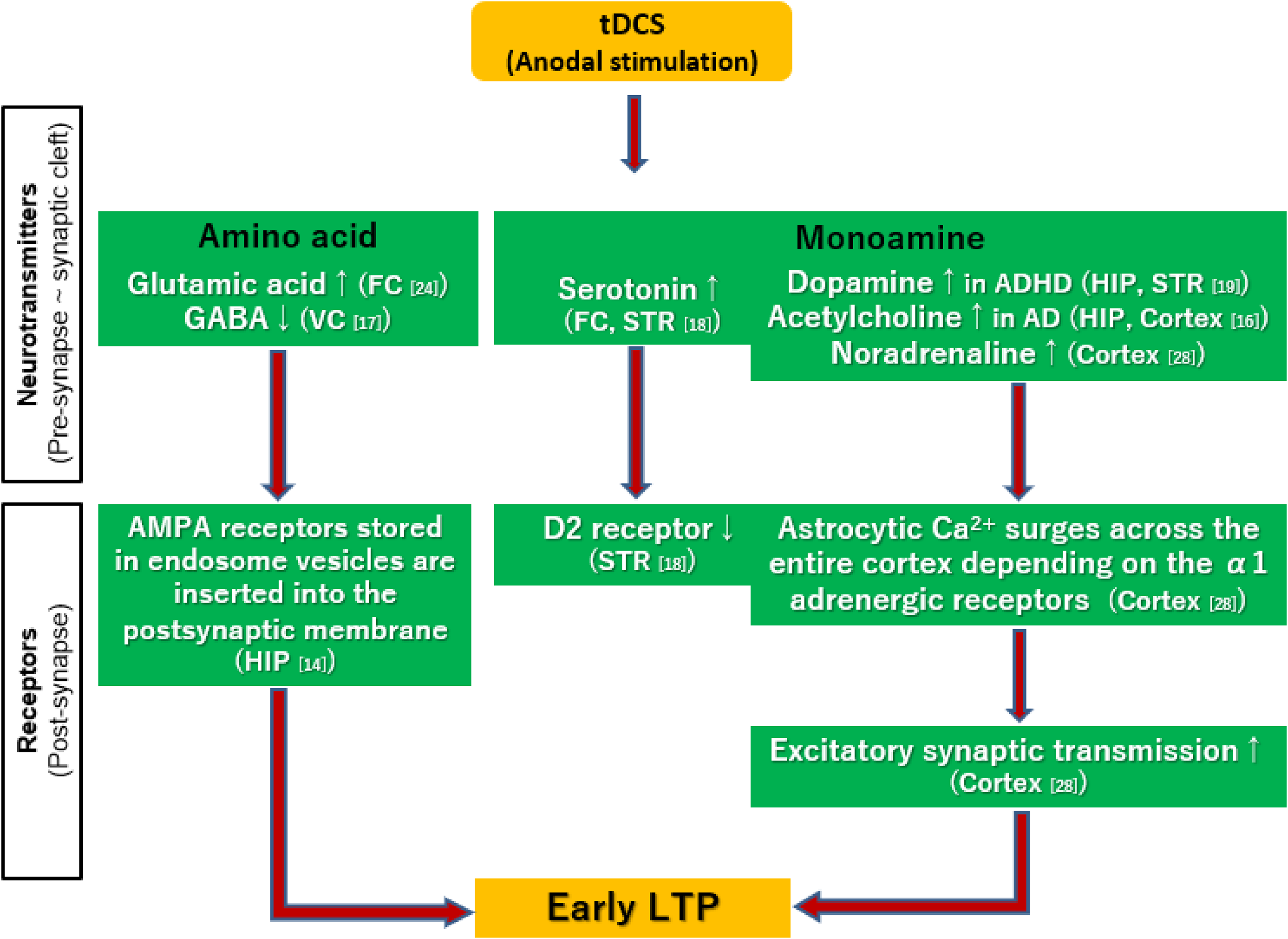
Putative mechanisms of the early long-term potentiation (LTP) induced by tDCS. Anodal stimulation enhances glutamate, ref. 9 serotonin, 35 dopamine, 36 acetylcholine, 33 and noradrenaline transmissions. 39 On the other hand, anodal stimulation decreases GABA levels. 34 The establishment of early LTP by tDCS may be accompanied with insertion of AMPA receptors, usually stored in endosome vesicles, into the postsynaptic membrane. 10 Moreover, astrocytic Ca2+ surges across the entire cortex depending on α1 adrenergic receptors may also induce early LTP. 39
Unlike the case with early LTPs, the establishment of late LTP by tDCS requires synthesis of various proteins. 38 The late LTP is accompanied with signal cascades; Ca2+ flows into postsynaptic cells, 16 and phosphorylates cAMP-responsive element binding protein (CREB) through activation of Ca2+/calmodulin-dependent protein kinase II (CaMKII) and protein kinase A (PKA). The CREB then activates transcription of the target gene to synthesize new proteins,11,12 eg, AMPA receptors, 9 NMDA receptors,16,17 ion channels and intracellular signal proteins,12,13,15 which enhances synaptic binding.11,12
The establishment of the late LTP has been shown to be accompanied with the induction of brain-derived neurotrophic factor (BDNF),12,13,15 glial cell line derived neurotrophic factor (GDNF), 15 nerve growth factor (NGF), 15 as well as increased expressions of growth-related proteins, such as growth-associated protein-5 (GAP-5) 14 and GAP-43. 9 Recent investigations also report tDCS produces hippocampal neurogenesis in rat models of ADHD 15 or cerebral infarction.41,42 Also, anodal stimulation may increase the permeability of the blood-brain barrier, 43 and enhance the substance transport capacity in brain tissues 44 (Figure 3).
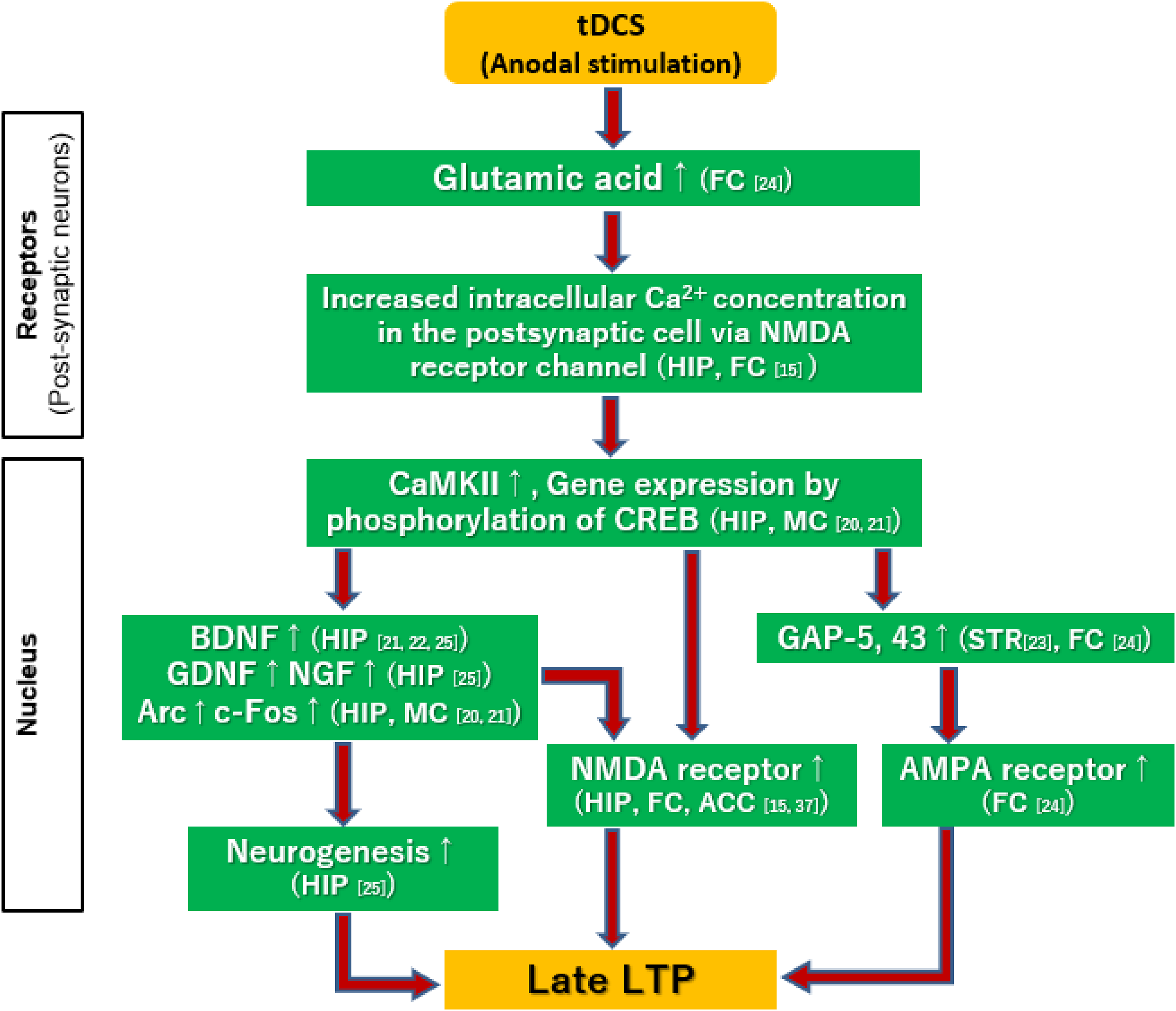
Putative mechanisms of the late long-term potentiation (LTP) induced by tDCS. First, Ca2+ flows into postsynaptic cells, ref. 16 and phosphorylates CREB through activation of CaMKII. The CREB then activates transcription of the target gene to synthesize new proteins, eg, AMPA receptors, 9 NMDA receptors,16,17 which are mediated by BDNF,12,13,15 GDNF, NGF, 15 Arc, and c-Fos.11,12
Enhancement of synaptic transmissions by LTPs is considered to exert a wide range of effects not only locally but also throughout brain networks. Thus, enhancement of connectivity has been reported 1) in cortical sensorimotor networks, 18 2) between the frontal cortex and nucleus accumbens, 19 and 3) between brain regions governing associative learning accompanied with low-frequency oscillations in the underlying tissue. 20 Specifically, anodal stimulation has been found to correct the activity of the striato-thalamo-cortical circuit regulating sensorimotor gating in rats overexpressing dopamine transporters. 45 These effects through brain networks improve learning and memory performance12,13,16,20,21 (Figure 4).
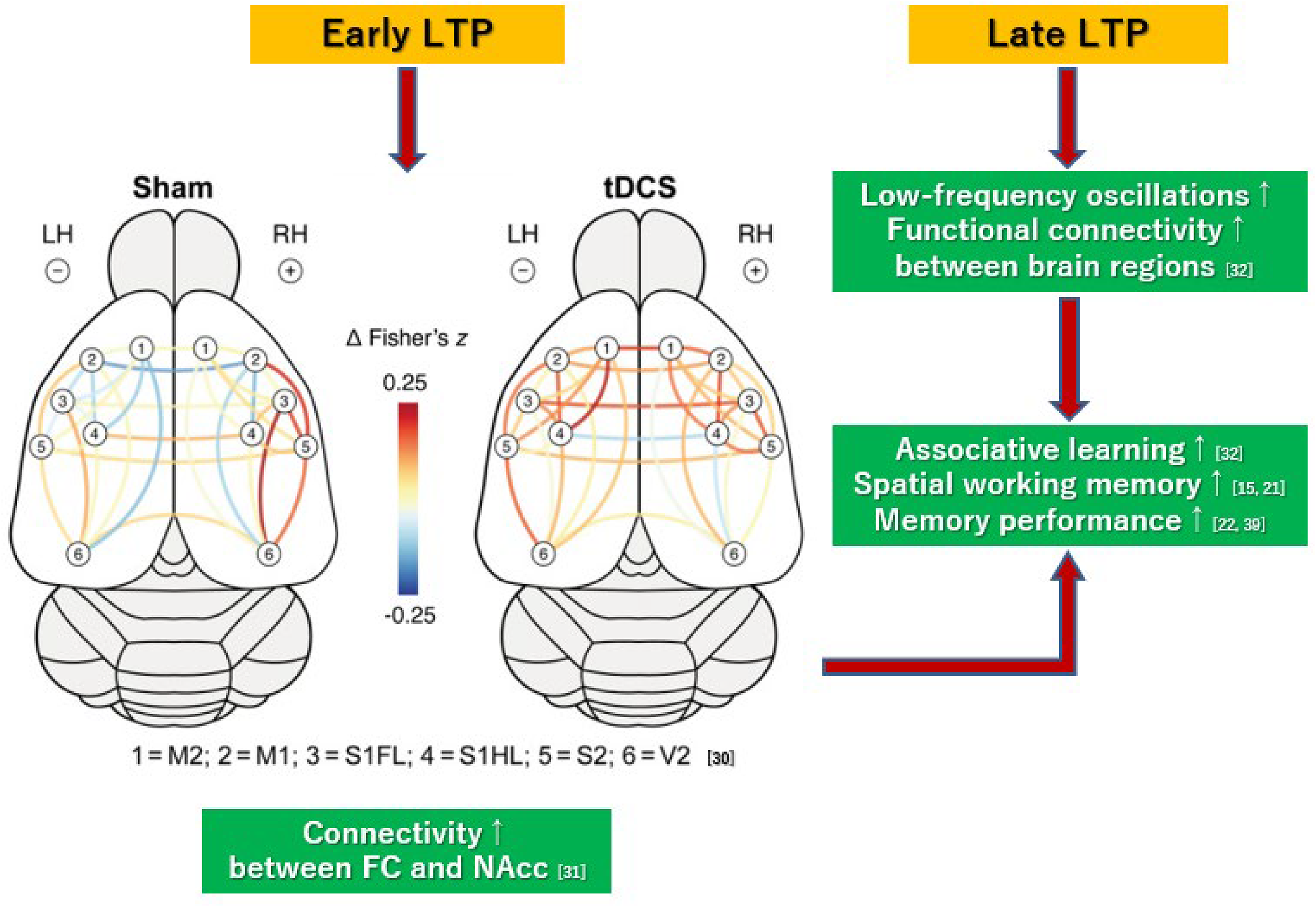
Enhancement of connectivity through brain networks by tDCS. Color-coded lines represent tDCS or sham stimulation-induced changes in functional connectivity (estimated by Fisher's z correlation) between regions of interest within the sensorimotor and visual cortices.ref. 18 Anodal stimulation also enhances connectivity between the frontal cortex and nucleus accumbens. 19 Moreover, multi-session tDCS induces low-frequency oscillations in the underlying brain regions and alters functional connectivity between them. 20 These effects on brain networks may improve performance on learning 20 and memory.12,13,16,21
The above neural events are summarized in Figure 5. Anodal stimulation enhances glutamatergic transmissions, 9 and facilitates Ca2+ flows into the postsynaptic cells, 38 which leads to insertion of AMPA receptors, usually stored in endosome vesicles, into the postsynaptic membrane. 10 Moreover, astrocytic Ca2+ surges, depending on activation of α1 adrenergic receptors, induce early LTP. 39 On the other hand, Ca2+ flows facilitated by glutamatergic transmissions 9 activate signal cascades, and phosphorylate CREB,11,12 which promotes protein synthesis9,11–15 and establishes late LTP.9,15–17 Finally, LTPs are considered to improve functional connectivity between brain regions18–20 and efficiency of information processing.12,13,16,20,21 LTPs are thought to play a main role in the action of tDCS on psychiatric symptoms. Further exploration is warranted to examine the effects of tDCS on specific symptoms in patients with psychiatric disorders.
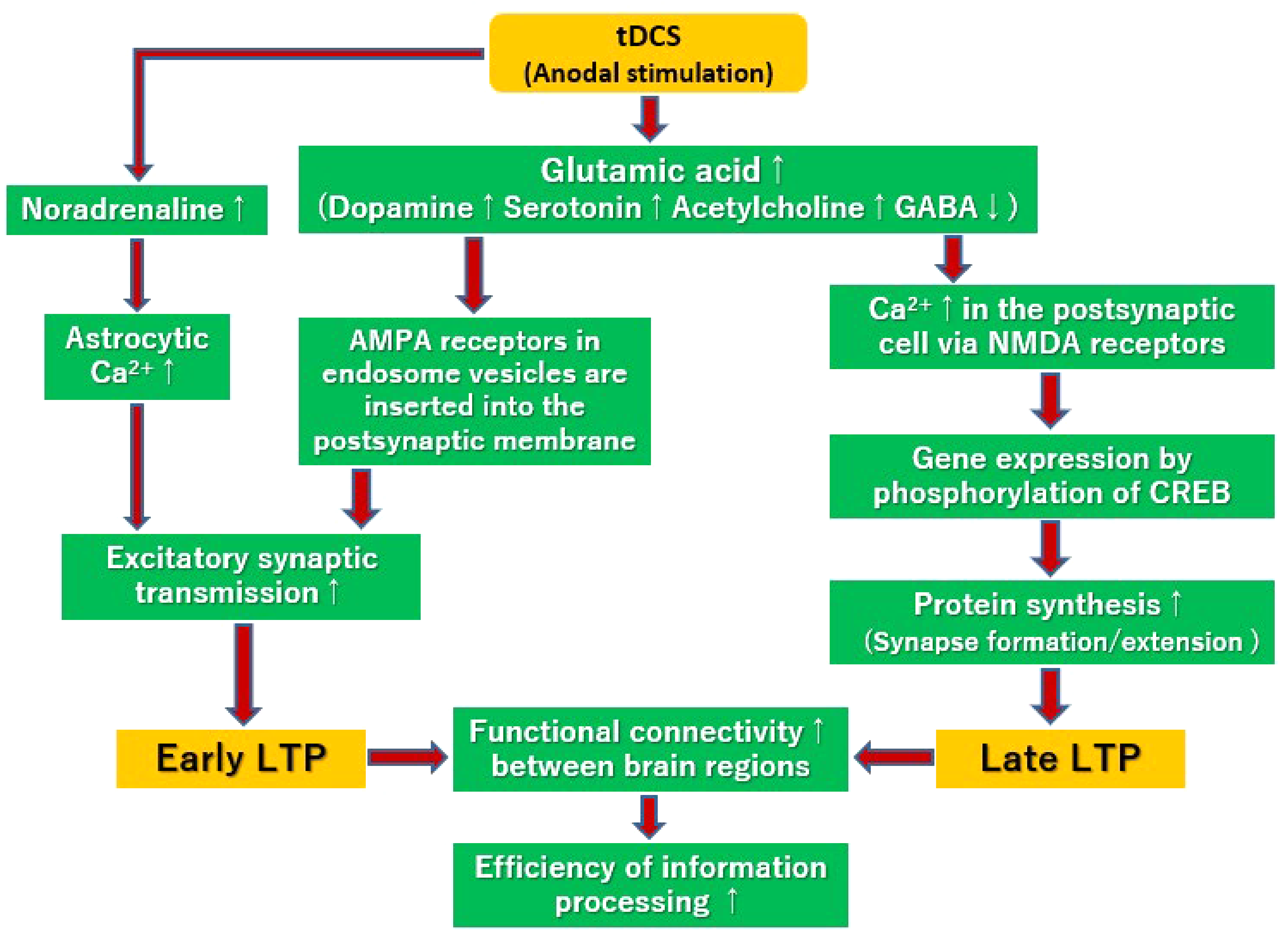
Putative mechanisms for the enhancement of long-term potentiation (LTP) by tDCS. Anodal stimulation enhances glutamatergic transmissions, ref. 9 and facilitates Ca2+ flows into the postsynaptic cells. 38 This mobilizes AMPA receptors stored in endosome vesicles to postsynaptic membranes. 10 Moreover, astrocytic Ca2+ surges mediated by α1-adrenergic receptors may also induce early LTP. 39 On the other hand, Ca2+ flows facilitated by glutamatergic transmissions 9 activate signal cascades, and phosphorylate CREB,11,12 enhancing protein synthesis,9,11–15 and establishment of late LTP.9,15–17 These LTPs are considered to improve functional connectivity between brain regions,18–20 and efficiency of information processing.12,13,16,20,21
This systematic review discusses the reduction of oxidative stress and inflammation induced by tDCS. For example, anodal stimulation prevents neurotoxicity by modulating autophagy and ameliorates behavioral deficits in a mouse model of Parkinson's disease.46,47 Moreover, in vascular dementia model rats, tDCS stimulation ameliorates cognitive impairments via modulating oxidative stress, inflammation, and autophagy. 48 In model rats of ADHD, anodal stimulation improves long-term memory deficits by modulating oxidative and inflammatory parameters. 49
The ability of tDCS to ameliorate various symptoms of psychiatric conditions has been mainly reported in studies using anodal stimulation over the frontal cortex. On the other hand, where the cathodal electrode is placed has not been uniform, indicating that the mechanism of tDCS has been more strongly associated with anodal stimulation of frontal cortical regions. 50 Nevertheless, there are reports of inflammation reduction and neuroprotective effects by cathodal stimulation. In the rat, cathodal tDCS after acute ischemic stroke promotes rehabilitation of motor function deficits51–54 via neurogenesis modulated by the Notch1 signaling pathway. 55 In epilepsy model animals, cathodal stimulation prevents seizures56,57 and improves cognitive performance. 58
In conclusion, data from animal experiments overall indicate tDCS facilitates LTPs, which may be mediated by several neural mechanisms, including AMPA receptors, NMDA receptors, BDNF, neurogenesis, and glia cells. These considerations may explain the ability of tDCS to alleviate several symptoms (eg, cognitive impairment) in psychiatric disorders. 32
Supplemental Material
sj-docx-1-eeg-10.1177_15500594211066151 - Supplemental material for Preclinical Evidence for the Mechanisms of Transcranial Direct Current Stimulation in the Treatment of Psychiatric Disorders; A Systematic Review
Supplemental material, sj-docx-1-eeg-10.1177_15500594211066151 for Preclinical Evidence for the Mechanisms of Transcranial Direct Current Stimulation in the Treatment of Psychiatric Disorders; A Systematic Review by Yuji Yamada and Tomiki Sumiyoshi in Clinical EEG and Neuroscience
Footnotes
Acknowledgments
We would like to thank Drs. Naotsugu Hirabayashi, Shinsuke Kito, and Kazuyuki Nakagome at the National Center of Neurology and Psychiatry for valuable supports.
Author Contributions
YY and TS planned the study. YY designed it and drafted the first manuscript. YY and TS independently searched and assessed the literature, and approved the final list of included studies. TS critically reviewed the draft and revised it. Both authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was partially supported by the Japan Society for the Promotion of Science KAKENHI Grant No.20K16635 to YY, as well as JSPS KAKENHI Grant No.20H03610, Health and Labour Sciences Research Grants for Comprehensive Research on Persons with Disabilities, AMED (20dk0307099 and 21he2202007), Intramural Research Grants for Neurological and Psychiatric Disorders of NCNP (2-3, 3-1), and Japan Health Research Promotion Bureau Grants (2020-B-08, 2021-B-01) to TS.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
