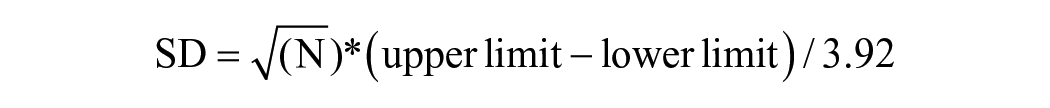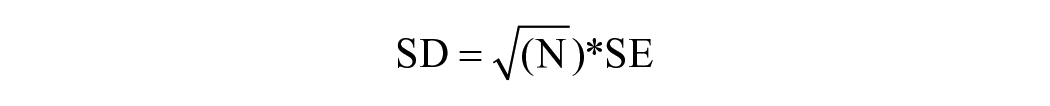Abstract
Background. Non-invasive brain stimulation (NIBS) is sometimes used alongside medication to alleviate motor symptoms in people with Parkinson’s disease (PD). However, the evidence supporting NIBS’s effectiveness for improving motor function in PD patients is uncertain. Objective. This umbrella review aims to synthesize recent systematic reviews and meta-analyses that have evaluated the effectiveness of NIBS in improving motor function in people with PD, with a key focus being to examine the quality of the evidence presented. Methods. The review protocol was registered in PROSPERO (CRD42022380544) and conducted per Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. The search strategy was guided by the Population, Intervention, Comparison, and Outcome framework, focusing on individuals with idiopathic PD (Hoehn and Yahr stages 1-4). The review included studies comparing various NIBS techniques (eg, repetitive transcranial magnetic stimulation and transcranial direct current stimulation) to sham or alternative treatments, targeting motor and cognitive regions. Six databases were searched up to June 2024. Methodological quality was assessed using Assessment of Multiple Systematic Reviews 2 (AMSTAR2), and random-effects meta-analyses were performed to pool standardized mean differences (SMDs). Results. The final analysis included 31 meta-analyses and 10 systematic reviews. Overall, the reviews were rated as moderate quality (54% average for AMSTAR2). NIBS showed a small-to-moderate effect on motor function (Unified Parkinson’s Disease Rating Scale-Section III scores; SMD = −0.80), functional mobility (gait speed and timed-up-and-go; SMD = −0.39), and freezing of gait (SMD = −0.58), but no significant effect on balance. Conclusion. NIBS offers small-to-moderate benefits for motor symptoms and functional movement in PD, though it does not significantly impact balance. Practitioners should consider the variety of techniques and treatment parameters before application.
Background
Parkinson’s disease (PD) affects between 1% and 3% of the older adult population worldwide and is the second most common neurodegenerative disease behind Alzheimer’s.1,2 Compared to healthy age-matched counterparts, people with PD have a significantly higher mortality rate. 3 While pharmacotherapies (eg, Levodopa) may improve quality of life and some motor functions, certain dopamine-resistant motor symptoms progressively worsen over time (ie, postural instability and gait difficulties [PIGD]) and is of continuing concern. Specifically, individuals with PD that have the PIGD subtype are at an increased risk of falls and freezing of gait (FoG) episodes compared to those with the tremor-dominant PD subtype. 4 Hence, there is a need for non-pharmacotherapeutic interventions that alleviate PIGD symptoms and decrease falls and FoG risk.
Non-invasive brain stimulation (NIBS) includes the use of transcranial direct current stimulation (tDCS), transcranial alternating current stimulation (tACS), transcranial random noise stimulation (tRNS), or transcranial magnetic stimulation (TMS) to modulate cortical excitability. Detailed discussion of the physiological impacts of these techniques has been described elsewhere,5 -7 but in brief, tDCS applies a direct weak electrical current (typically between 1 and 2 mA) to underlying, adjacent cortical tissue and relies on polarity-specific membrane potential alterations. The electrical current applied via tDCS therefore aims to shift the resting membrane potential state toward either depolarization or hyperpolarization depending on the site being targeted. 5 On the other hand, the electrical current applied via tACS enhances the spontaneous oscillatory activity of cortical neurons, and at certain frequency bands can alter cortical excitability both during and post stimulation. 8 Similarly, tRNS potentially modulates cortical excitability by using low-intensity biphasic alternating current. 9 In contrast to these direct current approaches though, TMS passes a brief electrical current through magnetic coils, causing a changing magnetic field that penetrates the scalp resulting in neuromodulation of cortical neurons in proximity to the stimulation site. Stimulation intensities typically range from 80% to 120% of motor threshold, 10 with intensities greater than motor threshold triggering action potentials in cortical neurons. 11
Independent of the mode, NIBS is relatively easy to administer, cost effective, and can be applied with few side effects provided the correct treatment guidelines are adhered to. 12 As such, the use of NIBS, and in particular tDCS and rTMS, has become a tenable clinical treatment option for numerous neurodegenerative and neuropsychiatric populations.13 -16 In individuals with PD for example, NIBS has received widespread attention as an adjuvant therapy used concurrently alongside other treatments such as exercise training or medication, or as a standalone tool, in an attempt to restore neurological and physical function17 -20 and reduce FoG episodes. 21 However, there are notable inter- and intra-individual variability in NIBS outcomes, 22 as well as considerable PD study and population heterogeneity.20,23 Therefore, questions about the effectiveness of NIBS as a physical rehabilitative tool for people with PD remain.
Recently, there has been a growing number of systematic reviews and meta-analyses focussed on the use of NIBS to improve physical outcomes in people with PD, often with conflicting findings. Therefore, the primary aim of this umbrella review is to summarize and map the overarching NIBS evidence on its effectiveness for improving physical outcomes in people with PD.
Materials and Methods
The protocol was published in June 2024 24 and was registered a priori in the International Registry PROSPERO (ID: CRD42022380544). We followed the Joanna Briggs Institute reviewer’s manual 25 and adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 26 Additionally, we employed the Population, Intervention, Comparison, and Outcomes (PICO) framework to guide our search strategy and inclusion/exclusion criteria.
We included reviews of studies with individuals diagnosed with idiopathic PD based on the Movement Disorder Society Clinical Diagnostic Criteria for PD (MDS-PD Criteria), 27 regardless of sex, with participants ranging from stage 1 (unilateral involvement only) to stage 4 (severe disability, but still able to walk and stand unassisted) according to the Hoehn and Yahr (H&Y) 28 scale (Population). We considered reviews of studies where NIBS was applied for either a single session or more, targeting the primary motor cortex (M1), dorsolateral prefrontal cortex (DLPFC), dorsal premotor cortex, cerebellum, or supplementary motor area (SMA; Intervention). We included reviews of studies that compared NIBS techniques as standalone therapies or in combination with exercise training or medication, with comparisons made against sham stimulation, no treatment (ie, regular care), or alternative treatments such as exercise only without sham stimulation (Comparison). Each of the reviews needed to examine the effect of NIBS on physical outcomes in people with PD (Outcome measure).
Inclusion Criteria
We included systematic reviews and meta-analyses of randomized and clinical controlled studies published from 2013 onwards that compared various forms of NIBS techniques, with no treatment, exercise, or sham stimulation in people with PD. Selecting reviews from the past decade was chosen to avoid overlap and overrepresentation of single studies. Included systematic reviews (with and without meta-analysis) focused on populations that (1) had a clinical diagnosis of PD established via the MDS-PD criteria and (2) had stages 1 to 4 H&Y disease severity. 28 No exclusions were applied based on the presence of co-morbidities. Given the wide scope of physical outcomes, we separated the physical outcome measures across systematic reviews into (1) motor function (ie, Unified Parkinson’s Disease Rating Scale [UPDRS]-Section III scores), (2) functional movement (ie, gait speed, or dynamic balance like timed-up-and-go [TUG]), and (3) static balance or postural control measures. A fourth outcome of disease severity (using the FoG questionnaire [FoG-Q]) was also examined.
Exclusion Criteria
We excluded all systematic reviews (with and without meta-analyses) of randomized controlled trials or other experimental studies that were published before November 2013 (ie, past 10 years of evidence), or were not written in English. For articles where the full-text was unavailable following an online search, attempts were made to contact the authors directly to retrieve the full-text copy. Articles were excluded if the authors could not be contacted or if they could not provide the full-text copy of the article.
Data Sources and Search Strategy
Six separate databases were searched: PubMed/MEDLINE, PEDro, Scopus, CINHL, EBSCO (ie, PsychInfo, PsychTherapy, and SPORTDiscus), and Cochrane Library, for systematic reviews (with or without meta-analyses) that were written in English up to June 2024. Various arrangements of the keywords in “Parkinson’s disease” AND “non-invasive brain stimulation or transcranial*” AND “physical function or strength or walking or gait” was used within the title and/or abstract searches. Where a filter was absent, the key words in “systematic review,” “meta-analysis,” or “practice guidelines” was also used within the title and/or abstract searches. A full list of MeSH terms and Boolean operators used for the searches can be found in Supplemental Table 1. In addition, the reference lists of each of the included reviews were manually checked to retrieve articles that were not covered by the database searches.
Methodological Quality
Methodological quality were recorded using the Joanna Briggs Institute critical appraisal checklist for Systematic Reviews and Research Syntheses. 29 Two members of the research team (SJO and CL) screened the titles and abstracts of the studies and extracted the data using an online systematic screening tool (Covidence). Any conflicts in screening were resolved by a third member of the research team (DMH). The methodological quality of the included reviews was assessed using the Assessment of Multiple Systematic Reviews 2 (AMSTAR2) instrument, which provides an empirical evaluation of their methodological rigor. The AMSTAR2 instrument uses a scoring system that ranges from 0 to 16, with higher scores indicating better quality. 30 It includes 16 items that assess various aspects of systematic reviews and meta-analyses, such as the thoroughness of the search strategy and the evaluation of publication bias. Each item is scored dichotomously as either 0 or 1. Based on the AMSTAR2 scores, reviews are categorized as high quality (satisfying at least 80% of the items), moderate quality (satisfying between 40% and 79% of the items), or low quality (satisfying less than 40% of the items).30,31 To ensure consistency, all extracted review information and quality rankings were compared among raters (DMH and NT) to establish inter-rater reliability.
Meta-analysis
We performed random-effects pooled meta-analyses using the included effect sizes for studies reporting standardized mean difference (SMD). By pooling the SMD’s and applying 95% confidence intervals (CIs) weighted for sample size, we derived an aggregated effect size that encompassed all the reviews. 32 The estimated SMD’s were interpreted using the description presented by Hopkins et al 33 ; an SMD of >4.0 = an extremely large clinical effect; between 2.0 and 4.0 = a very large effect; 1.2 to 1.9 = a large effect; 0.6 to 1.1 = a moderate effect; 0.2 to 0.59 = a small effect; and 0.0 to 0.19 = a trivial effect. Heterogeneity among the reviews was assessed using the I2 statistic, which quantifies the degree of variability between study results (<25% = negligible; 25%-50% = low; 50% to 75% = moderate to high; >75% = very high). 34 Additionally, we assessed publication bias using the Egger test, a statistical method that evaluates the presence of bias in the reporting of study outcomes. 35
When the results from meta-analyses were reported as mean difference (MD) or weighted MD (WMD), they were re-expressed as SMD. As a form of sensitivity analysis, we conducted a separate meta-analysis including these studies to ensure that conversion to SMD did not influence the overall results. If necessary, CIs and standard error (SE) where converted to standard deviation (SD) using the formulas recommended by the Cochrane Handbook for Systematic Reviews of Interventions version 6.2 36 :
OR
Where N is the number of participants (sample size), and upper and lower bound limits refer to the 95% CI’s, respectively.
Analysis of Degree of Overlap in Studies
Citation matrices were generated to determine overlap in studies across reviews, while the corrected covered areas (CCAs) were also calculated (CCA = 0%-5%, slight; 6%-10%, moderate; 11%-15%, high; and >15%, very high overlap) 37 to assess the impact of overlap in reviews on the findings of this umbrella review.
Results
Search Results
A comprehensive search of 6 databases yielded a total of 61 reviews that were potentially eligible for inclusion in this umbrella review. After careful examination, 18 reviews were excluded as they did not meet the predefined eligibility criteria (Figure 1). The reasons for exclusion varied and are listed in Supplemental Table 2. This left a total of 38 reviews that were deemed eligible and included in this umbrella review for further analysis. Three additional studies published in 2022, 38 2023, 39 and in 2024 40 were included subsequent to the initial search and screening process, owing to their relevance to our research objectives (n = 41 reviews in total included). Of these included reviews, 31 were systematic review with meta-analysis, 10 were either systematic or scoping reviews only (without meta-analysis).
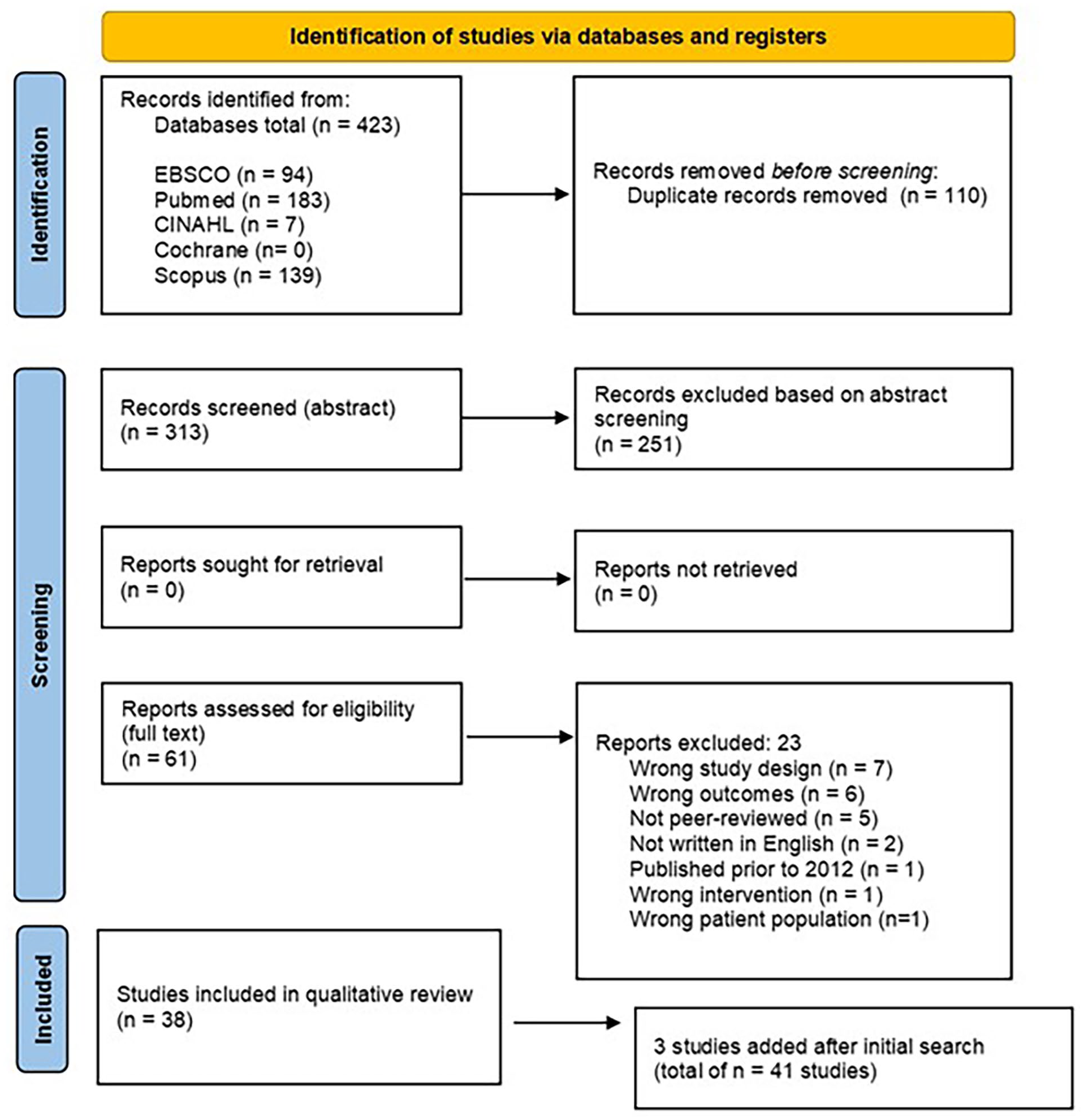
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow chart of review.
Summary of the Included Reviews
The number of studies included in each review ranged from 7 to 33 (Table 1) and the number of participants ranged from 40 to 1596. NIBS techniques mainly included rTMS only (19 studies, 46%), tDCS only (13 studies, 32%), or mix of rTMS and tDCS (8 studies, 20%), with 1 study investigating galvanic vestibular stimulation. Of the meta-analyses, the more common physical outcomes included assessing the effects of NIBS on motor disability (measured via the UPDRS-III; 18 studies, 58%), functional mobility including gait speed and TUG tests (11 studies, 35%) and balance (6 studies, 19%). Nine meta-analyses examined the effects of NIBS alone versus sham stimulation, while 18 meta-analyses examined the effects of NIBS versus a control group. The stimulation parameters and sites varied considerably across the reviews (Supplemental Table 3).
Summary of the Characteristics and Main Findings of the Included Reviews.
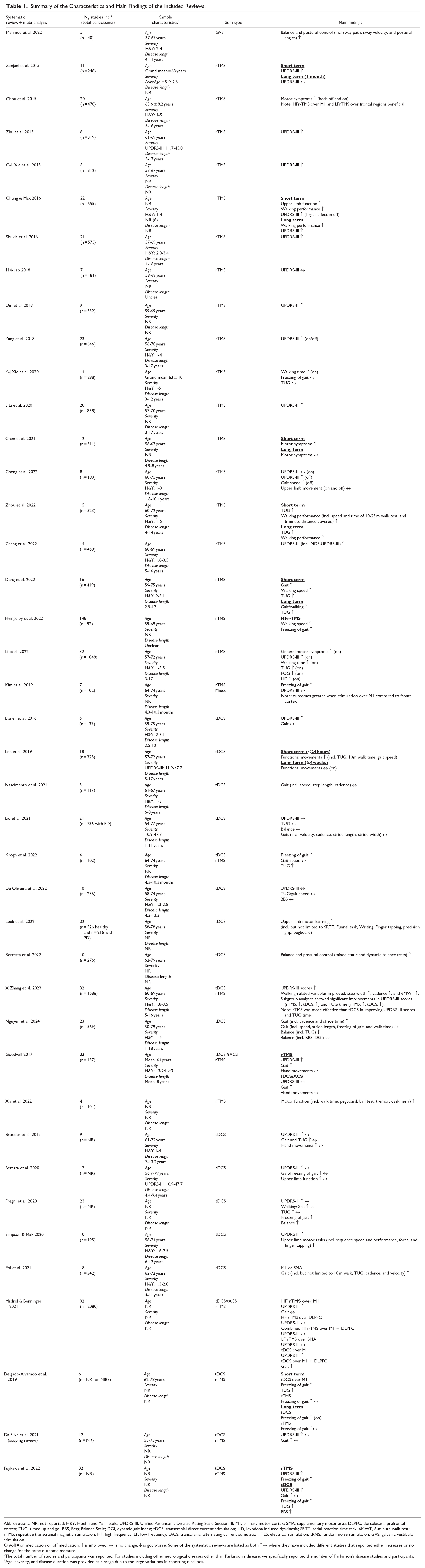
Abbreviations: NR, not reported; H&Y, Hoehn and Yahr scale; UPDRS-III, Unified Parkinson’s Disease Rating Scale-Section III; M1, primary motor cortex; SMA, supplementary motor area; DLPFC, dorsolateral prefrontal cortex; TUG, timed up and go; BBS, Berg Balance Scale; DGI, dynamic gait index; tDCS, transcranial direct current stimulation; LID, levodopa induced dyskinesia; SRTT, serial reaction time task; 6MWT, 6-minute walk test; rTMS, repetitive transcranial magnetic stimulation; HF, high frequency; LF, low frequency; tACS, transcranial alternating current stimulation; TES, electrical stimulation; tRNS, random noise stimulation; GVS, galvanic vestibular stimulation.
On/off = on medication or off medication. ↑ is improved, ↔ is no change, ↓ is got worse. Some of the systematic reviews are listed as both ↑↔ where they have included different studies that reported either increases or no change for the same outcome measure.
The total number of studies and participants was reported. For studies including other neurological diseases other than Parkinson’s disease, we specifically reported the number of Parkinson’s disease studies and participants.
Age, severity, and disease duration was provided as a range due to the large variations in reporting methods.
Methodological Quality and Quality of Evidence
There was a moderate level of agreement between reviewer scores for the AMSTAR2 (κ = .75). For the AMSTAR2, the average score across all the included reviews was 8.7 out of 16 (54%), and so the average quality of the reviews was rated as moderate. A total of 12 out of the 41 reviews were rated as low quality, and only 4 reviews were rated as high quality.40 -43 Many reviews failed to include a comprehensive list of excluded studies (item 7, 88%) and half of the reviews (54%) did not discuss the likely impact of publication bias on their results (item 12). This involves reviews failing to perform a sensitivity analysis to determine how many missing “null” studies would be needed to invalidate the results they obtained. Only 39% of reviews designed their research questions and inclusion criteria using the components of PICO (item 1), and 37% stated that their search methodology was designed a priori and pre-registered (item 2). Also, 63% of the reviews did not report the sources of funding of the individual studies that were included within their results (item 10; Figure 2).
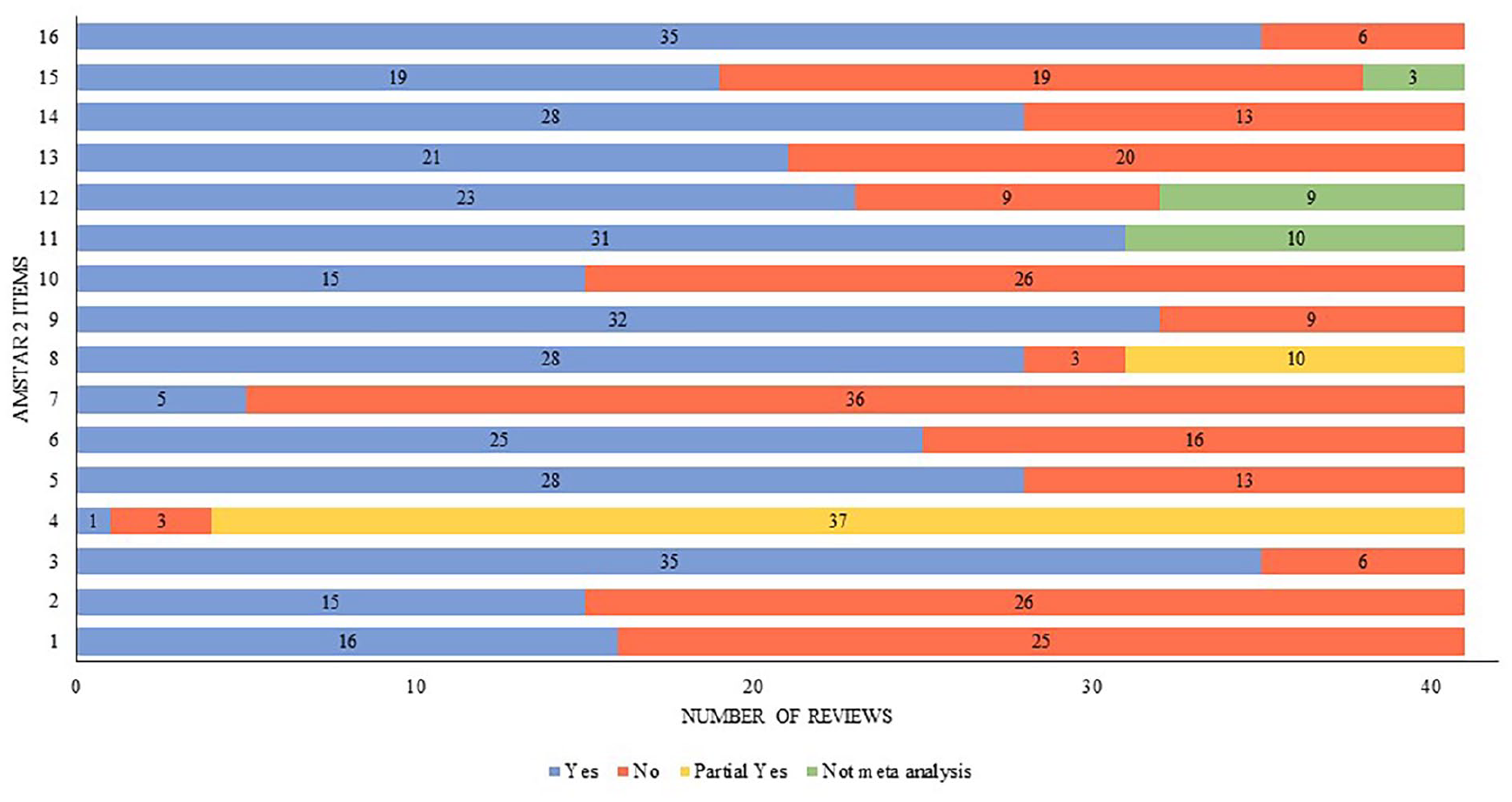
Quality assessment (AMSTAR2).
NIBS Effects
Motor Function (UPDRS-III)
Our review included 18 meta-analyses that examined the effects of NIBS on motor function (UPDRS-Section III) in people with PD, irrespective of co-morbidities. About 13 of these meta-analyses reported SMD’s, and our pooled results indicated that NIBS had a moderate positive effect on UPDRS-III scores (SMD = −0.80; 95% CI −1.37 to −0.23; n = 7043; P = .006; I2 = 98.47%; Figure 3). This result was unaffected when including studies which reported MD instead of SMD (SMD = −0.73, 95% CI −1.23 to −0.23; n = 7106; P = .004; I2 = 97.7%). In addition, when 1 study was removed due to separating groups into “on” versus “off” medication, 44 there was still a moderate positive effect on UPDRS-III scores (SMD = −0.89; 95% CI −1.55 to −0.24; n = 6884; P = .007; I2 = 98.71%).
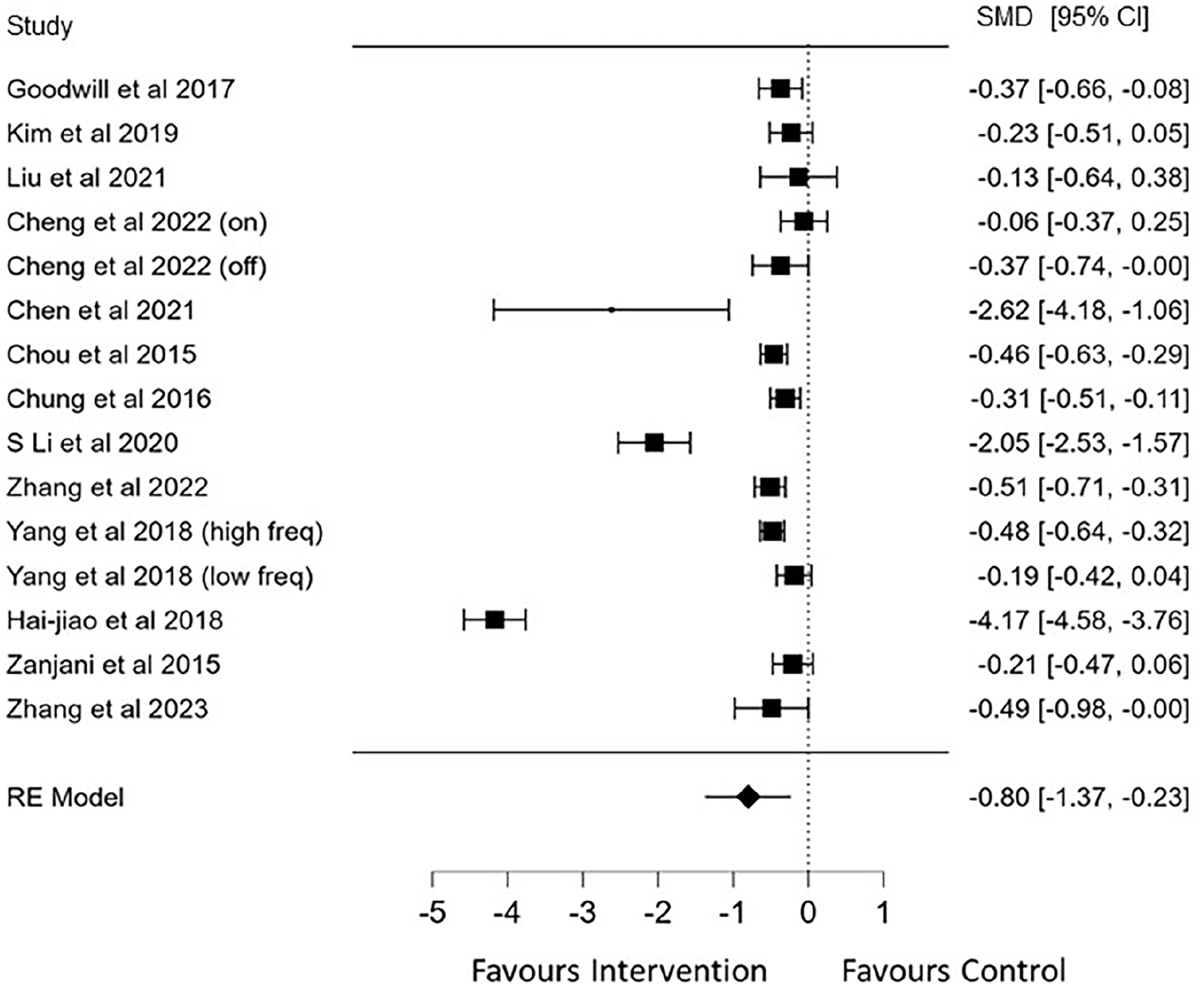
Pooled meta-analysis of the effects of non-invasive brain stimulation on Unified Parkinson’s disease Rating Scale (UPDRS) motor function (section III) scores.
Our review included 2 meta-analyses that examined the effects of NIBS on motor function of people with PD with clinical depression. Due to the wide variety of outcomes used to assess clinical depression, which included instruments such as the Hamilton Rating Scale for Depression, the Montgomery-Åsberg Depression Rating Scale, and the Beck Depression Inventory, we opted not to pool the results. Qin et al 45 proposed that the motor benefits after treatment with high frequency (HF)-rTMS might be better than sham-rTMS (MD = −2.80, 95% CI −5.45 to −0.15), and this was also supported by Chen et al 46 who reported that rTMS could alleviate motor symptoms of people with PD with clinical depression (WMD = −2.617, 95% CI −4.183, −1.051).
Functional Mobility: Gait Speed and TUG
Our review included 17 meta-analyses that examined the effects of NIBS on functional movement in people with PD. Of these, 11 meta-analyses reported SMD’s. Our pooled results indicated that NIBS had a small positive effect on functional mobility (SMD = −0.39, 95% CI −0.51 to −0.28; n = 4852; P < .001; I2 = 51.4%; Figure 4), which was unaffected when including studies for which MD was reported instead of SMD (SMD = −0.35, 95% CI −0.46 to −0.24; n = 5509; P < .001; I2 = 52.2%). When 1 study was removed due to separating groups into “on” versus “off” medication, 44 there was still a small positive effect on functional mobility (SMD = −0.35, 95% CI −0.47 to −0.24; n = 4693; P < .001; I2 = 57.1%).
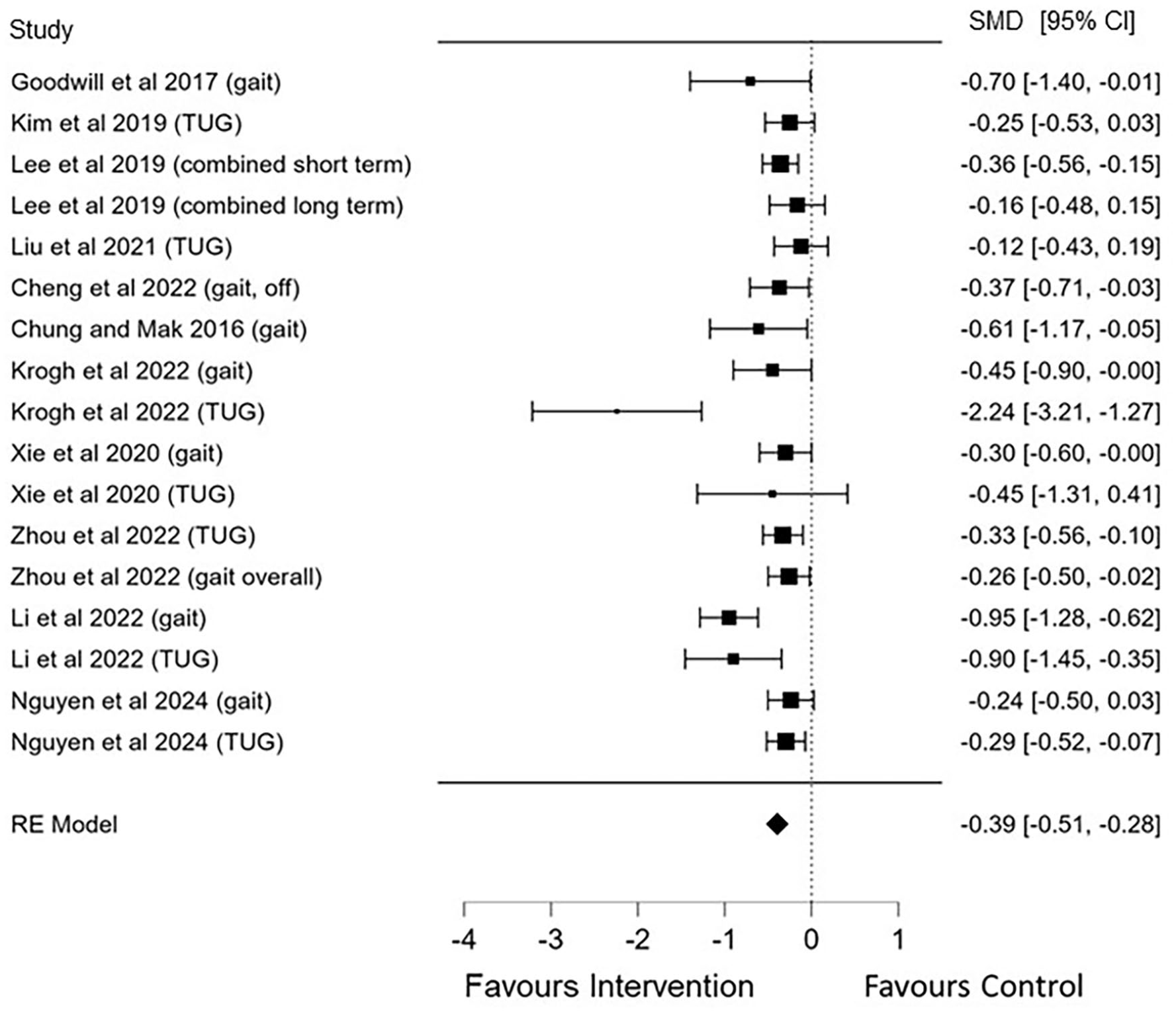
Pooled meta-analysis of the effects of non-invasive brain stimulation on functional mobility.
Two sub-analyses were performed separating gait speed from TUG results. Nine studies examined the effects of NIBS on gait speed only in people with PD and reported SMD’s. The pooled results revealed that NIBS had a small positive effect on gait speed (SMD = −0.43, 95% CI −0.61 to −0.25, P < .001; n = 2239; I2 = 50.2%; Supplemental Figure 1). This result was not different when 1 study was removed due to separating groups into “on” versus “off” medication 44 (SMD = −0.44, 95% CI −0.65 to −0.24, P < .001; n = 2080; I2 = 55.8%). In addition, another 9 studies examined the effects of NIBS on TUG performance only in people with PD. The pooled results also revealed that NIBS had a small positive effect on TUG scores (SMD = −0.36, 95% CI −0.58 to −0.13, P = .002; n = 3189; I2 = 77.1%; Supplemental Figure 2).
Freezing of Gait
Our review included 5 meta-analyses that examined the effects of NIBS on FoG in people with PD. Our pooled results indicated that NIBS had a moderate and significant positive effect on reducing FoG (SMD = −0.58, 95% CI −0.91 to −0.26, P < .001; n = 1183; I2 = 78.3%; Figure 5).
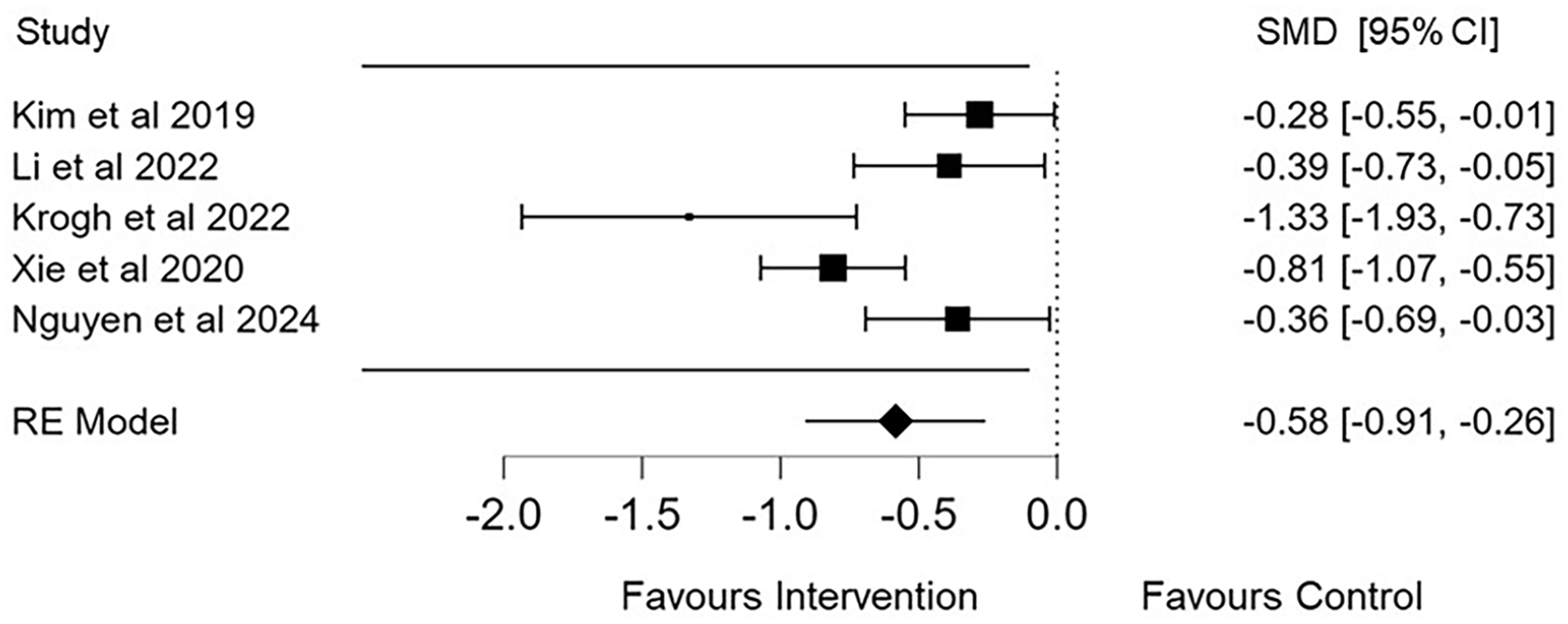
Pooled meta-analysis of the effects of non-invasive brain stimulation on freezing of gait (FoG) scores.
Static Balance/Postural Control Measures
Our review included 6 meta-analyses that examined the effects of NIBS on static balance/postural control outcomes in people with PD. Five meta-analyses used the Berg Balance Scale (BBS) as the primary balance outcome (1 combined BBS with dynamic posturography 47 ) and 1 explored dynamic posturography (ie, body sway) in isolation. 38 Pooled results indicated that NIBS had no effect on static balance/postural control outcomes (SMD = −0.16, 95% CI −0.40 to 0.08, P = .196; n = 1175; I2 = 47.6; Figure 6).
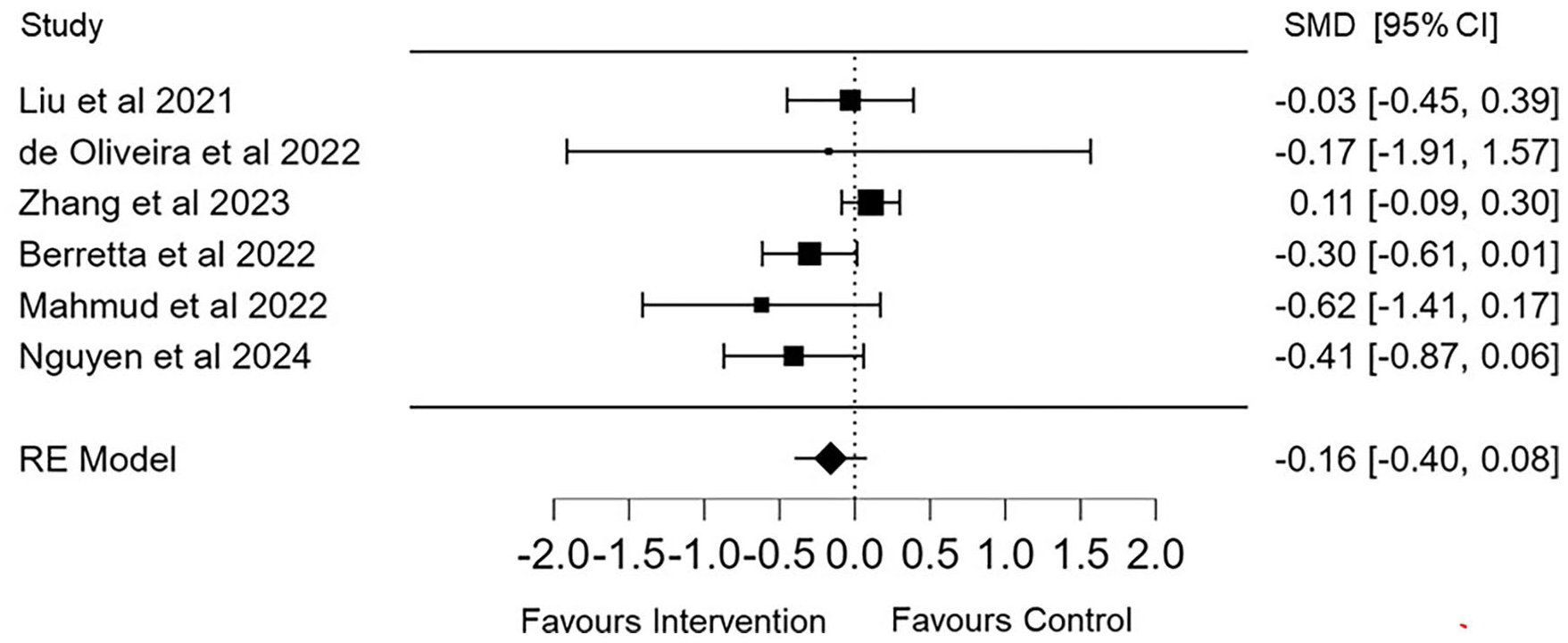
Pooled meta-analysis of the effects of non-invasive brain stimulation on balance.
As part of our sensitivity analysis, we examined the funnel and Q–Q plots for outcomes with very high heterogeneity (>75%), including UPDRS-III (I2 = 98.47%), TUG sub-analysis (I2 = 77.1%), and FoG (I2 = 78.3%) outcomes. For UPDRS-III, 3 studies were excluded based on visual inspection of the funnel and Q–Q plots, as well as a high likelihood of bias.46,48,49 After their removal, a small but significant effect remained (SMD = −0.37, 95% CI = −0.45 to −0.28, P < .001), and heterogeneity was substantially reduced (I2 = 23.93%; Egger’s test P = .118). For the TUG outcome, 1 study was removed following the same approach 41 . A small but significant positive effect of NIBS on TUG scores was still observed (SMD = −0.24, 95% CI = −0.33 to −0.14, P < .001), with heterogeneity effectively eliminated (I2 = 0.02%; Egger’s test P = .143). Lastly, for FoG, removing the same study 41 resulted in a moderate and significant positive effect on FoG scores (SMD = −0.50, 95% CI = −0.83 to −0.17, P = .003), although heterogeneity dropped slightly from very high to high (I2 = 74.15%; Egger’s test P = .661).
Degree of Overlap
Overall, there were 161 primary studies that were examined across the 41 reviews included in this umbrella review. The CCA across all the included reviews was 6.58%, suggesting moderate overlap of primary studies (Supplemental Figure 3). For the reviews focussing on motor function (UPDRS-III) specifically, the CCA was 2.16%, suggesting slight overlap of primary studies (Supplemental Figure 4). For the reviews focussing on functional movement, including TUG and gait speed assessments, the CCA was 1.81%, suggesting a slight overlap of primary studies (Supplemental Figure 5). Due to the limited number of reviews focussing on specific balance and FoG outcomes, the CCA for these groups of reviews also suggest a slight overlap in primary studies (Supplemental Figures 6 and 7).
Evidence Map
A visual bubble plot of the meta-analyses included in this umbrella review was designed, based on previous maps 50 and in-line with our published methodology. 24 Generally, most meta-analysis rated as high-quality reported a trivial to small positive effect of NIBS on motor outcomes in people with PD (Figure 7).
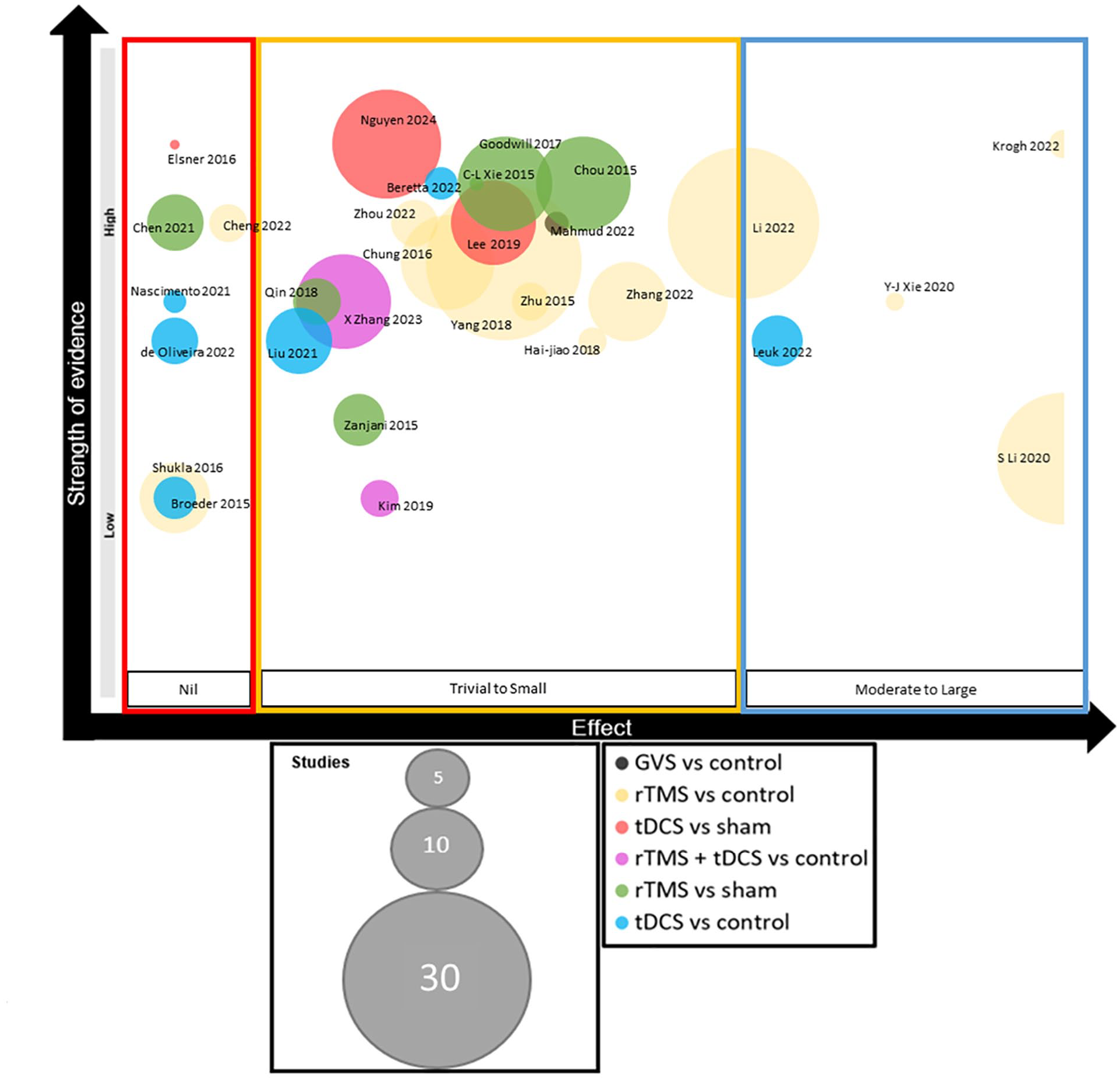
Evidence visual map synthesis. The meta-analyses are grouped into 3 categories on the x-axis, depending on the effect of the interventions for their primary outcome (“nil,” “trivial to small,” and “moderate to large”). Along the y-axis, studies are ranked based on the AMSTAR 2 score. The bubble size represents the number of studies included in each review. The bubble color represents the type of noninvasive brain stimulation explored, and if the comparison was a control or sham stimulation.
Discussion
Methodological Quality of Reviews
This umbrella review is the first to consolidate and evaluate the quality of evidence from NIBS systematic reviews and meta-analyses in PD. The average methodological quality score using AMSTAR2 for the included reviews was “moderate,” with various inconsistencies present in the rigor and reporting standards. The updated PRISMA 2020 guidelines revised several reporting elements to enhance clarity in systematic reviews, including requiring authors to report on data preparation, synthesis, heterogeneity and sensitivity analyses, and citing excluded studies and explaining reasons for exclusion. 26 Our findings reveal that half of the reviews did not address how publication bias may have impacted the findings (54%), and many reviews failed to provide a comprehensive list of excluded studies (88%). Thus, at a minimum, future reviews should aim to incorporate these methodological approaches to improve quality and ensure compliance with all items outlined in the PRISMA 2020 guidelines.
Despite the combined corrected coverage area for the included reviews being moderate at 6.58%, only slight overlap was observed across the motor outcomes when analyzed separately, with ranges between 1.8% and 2.2%. This suggests a sufficient spread of primary studies, along with considerable complexity and diversity among the reviews, indicating that the evidence can be trusted for various motor outcomes in PD. This finding is not unexpected, as we exclusively included reviews published in the past 10 years (from 2013 onward) to minimize overlap in primary studies. It also implies that over the past decade reviews are becoming increasingly targeted in addressing specific questions related to NIBS and motor outcomes for PD.
A broader critique of the meta-analyses included within our review is that the various methodological approaches used to analyze trial data likely lead to diverse and often non-standardized effects. Using pooled standard deviation of change scores, post-intervention scores, or pre- and post-intervention scores, when conducting a meta-analysis can often result in biased and difficult-to-interpret results. 51 Whilst it is difficult to determine which statistical approaches were used by the meta-analyses included herein, authors who wish to perform future meta-analysis on the effects of NIBS techniques on physical function in PD should consider alternative approaches like using log transformation of factor effects, and using minimum clinically important differences for standardization after meta-analysis. 51
NIBS on Motor Function and Functional Movement
Our results suggests that NIBS can benefit motor function in PD. This aligns with previous work suggesting that various modes of NIBS may enhance motor cortex excitability and facilitate motor performance in PD.18,43,44,52 -55 In our review, NIBS also exhibited a small yet significant positive effect on functional movement, including gait speed and TUG performance. This finding is particularly relevant as gait difficulties significantly impair quality of life 56 and increase falls risk in people with PD. 4 When applied to the motor or prefrontal cortex, NIBS (especially rTMS and tDCS) can modulate the release of dopamine in the caudate nucleus 57 and putamen, 58 and their cortico-striatal projections 59 which may, at least partially, explain the short term effects of NIBS on motor function and functional movement in PD.
In our review, NIBS demonstrated small to moderate significant effects on UPDRS-III scores, suggesting that NIBS may enhance overall motor function in PD. This finding aligns with evidence-based guidelines on the therapeutic use of NIBS.60,61 However, it is important to determine whether this effect reaches the minimal clinically meaningful difference threshold. Moderate to large clinically important differences in UPDRS-III scores lies between −5.2 and −10.8 points difference from baseline scores. 62 Comparing to a previous example, 63 our result translates to a moderate clinically important difference. Individual differences in pathophysiology likely influence NIBS treatment outcomes, and so future studies should aim to determine optimal stimulation protocols for various clinical subtypes and symptoms. Tailoring NIBS therapies to address specific symptoms may result in greater, and more clinically meaningful, improvements in motor function.
As PD progresses, balance while standing, walking with a narrow base, and turning actions become more unstable.64 -67 As such, NIBS has commonly been proposed as an effective treatment option to attenuate the postural instability experienced by people with PD.38,47,68 However, our review showed that NIBS had a non-significant trivial effect on static balance and postural control outcomes. Zhang et al 39 suggested that there was no effect of NIBS (ie, both rTMS and tDCS) on balance (measured via the BBS; MD = 1.39; 95% CI −1.13 to 3.91; P = .28), which is akin to the findings of Nguyen et al 40 (SMD = 0.406; 95% CI −0.059 to 0.87; P = .087). In contrast, other meta-analyses have reported significant improvements in balance and dynamic posturography following tDCS applied over the M1 (SMD = −0.30; −0.61 to 0.01; P = .07) 47 and following GVS (SMD = 0.16; 0.01 to 0.31; P < .05). 38 These discrepancies may result from variations in stimulation methods, differences in the location and frequency of treatments, and differences between study cohorts. In addition, the complexity of postural instability in PD, which involves multiple neural pathways and sensorimotor integration processes,69,70 may also be difficult to treat using a single stimulation paradigm/location. It must also be acknowledged that the heterogeneity in study designs and balance assessment methods used across the reviews could also account for these mixed results.
FoG episodes are debilitating for people with PD, often resulting in poor quality of life for the patient and their carer. 71 Our review demonstrated that NIBS had a moderate positive effect on reducing FoG. However, some careful consideration is needed when interpreting our results. Firstly, we included 5 meta-analyses that explored the effects of NIBS on FoG, using the FoG questionnaire. Of these, 1 meta-analysis included just 2 randomized trials with low sample sizes in their analysis 41 and showed a large and significant effect in favor of rTMS for improving FoG (N = 38 real and 20 sham patients; SMD = −1.33; −1.94 to 0.73; P < .0001). Another meta-analysis included 4 randomized trials, and showed no effect of rTMS improving FoG (N = 45 real and 45 sham patients; SMD = −0.81; −1.68 to 0.06; P = .07). 72 Given the heterogeneity in findings, it remains uncertain whether NIBS is effective in the treatment of FoG at this time, despite our positive results. Further clinical trials, with larger samples, and more homogenized techniques for stimulation location, dosage, and follow-up are needed.
Additional Considerations
Exploring NIBS timing, location, dosage, and effectiveness across different PD stages is crucial, yet we were unable to assess this for several reasons. First, to our knowledge, no studies have directly compared NIBS in early onset versus advanced PD. Second, prospective studies typically include a range of PD severities in a treatment/comparison group. A suggestion for future research is to include multi-arm trials where the same treatment is delivered across groups ranging from mild to severe disease.
The interaction between NIBS and PD medications is another crucial consideration that has been poorly investigated. To date, only 1 meta-analysis has explored the effects of NIBS during “on” versus “off” therapy 44 and so it is currently unclear if NIBS effectiveness is influenced by the medication state. Cheng et al 44 among 6 studies (n = 159 patients) found that rTMS applied over the SMA could significantly improve the UPDRS-III score for PD patients in the “off,” not in the “on,” medicine state (“off” medication SMD = −0.37, 95% CI −0.65 to −0.09, P < .01; “on” medication SMD = −0.06, 95% CI −0.37 to 0.25; P = .69).
A limitation of the meta-analyses included in this review is that most studies either combine data from populations “on” and “off” medication or focus solely on patients in the “on” medication state. Additionally, these reviews often pool outcomes from patients taking a combination of medications, such as Levodopa, dopamine agonists, catechol-O-methyltransferase inhibitors, and anticholinergics. Consequently, further primary research is needed to better assess the effects of NIBS when used alongside combination medications, and to compare its efficacy in the “on” versus “off” medication states for improving motor outcomes. This research is crucial because certain symptoms, such as FoG, do not respond well to dopamine-based treatments, suggesting that NIBS could offer potential benefits. Moreover, dopamine medications can lead to side effects like dyskinesia and neuropsychiatric issues, which may make higher doses intolerable. 73 Investigating the use of NIBS with lower medication doses could help reduce these side effects while maintaining motor improvements, but this needs thorough exploration before any therapeutic recommendations can be made.
To our knowledge there has been 3 network meta-analysis exploring the relative effectiveness of different NIBS modes and brain sites on motor outcomes in people with PD.74 -76 These were excluded from our pooled results due to the difficulty of unpacking analyses to make direct pairwise comparisons. Nevertheless, the results of these network meta-analyses warrant discussion. Across all network meta-analyses, high-frequency TMS consistently improved UPDRS-III scores in the short term compared to sham,74 -76 with effectiveness observed across multiple brain sites (SMA, DLPFC, prefrontal cortex, and M1). 74 . In addition, the surface under the cumulative ranking curve value, which is often used to rank the treatment outcome scores, suggested that high-frequency repetitiveTMS (HFr-TMS) had the highest probability of being effective in enhancing motor outcomes in the short-term (values of between 77% 75 and 95.1% 74 ) or dual-mode therapy with HFr-TMS and tDCS (88.5%). 76 However, some limitations warrant caution when interpreting these findings. These include the low number of studies investigating NIBS techniques other than HFr-TMS and the scarcity of direct comparisons between different NIBS modes across various brain sites in people with PD.
Strengths and Limitations
The strengths of this umbrella review include, first and foremost, successfully achieving our primary goal of evaluating and summarizing the scope and quality of systematic reviews that report the effects of NIBS on physical function in PD. We achieved this by thoroughly reviewing the literature to: (1) map the evidence using a novel bubble plot for visual inspection, (2) assess the quality of the included reviews using the AMSTAR2 scale, and (3) analyze the number of primary studies to determine the degree of overlap between reviews. Additionally, we extracted results from 31 meta-analyses and conducted an aggregate meta-analysis focused on motor function, mobility, FoG, and balance. This approach provides a more comprehensive and robust overview of the latest NIBS evidence on physical outcomes in PD.
Though we followed standardized criteria for conducting this review and reporting the findings, there are certain limitations which should be acknowledged. For instance, even though we performed a comprehensive search of multiple databases, we might have missed potential reviews by limiting the search to English-language studies. However, Cochrane recommendations are unclear about including only English-language studies and do not indicate that this would likely bias the review results. In addition, we relied on the meta-analyses results reported in the included reviews and did not extract or verify data from the original studies, or re-run a meta-analysis based on original data, as this was beyond the scope of this umbrella review.
Conclusion
This umbrella review includes 31 meta-analyses (41 reviews in total) involving 14 253 participants. While the overall quality of the systematic reviews needs improvement, NIBS therapy shows potential benefits for motor function (UPDRS-Section III) and functional mobility (TUG and gait speed) in PD patients, along with some positive effects on reducing FoG. However, the effects on static balance and postural control are limited. To strengthen recommendations, future primary clinical studies should focus on determining optimal NIBS parameters, including stimulation intensity, frequency, and duration, as well as effective combinations with pharmacotherapy and exercise. More rigorous systematic reviews and meta-analyses that follow the PRISMA 2020 reporting criteria are essential to establish precise guidelines for the “optimal dose” and “location” of NIBS therapy in PD.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-docx-1-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-docx-2-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-docx-3-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-10-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-10-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-4-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-4-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-5-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-5-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-6-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-6-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-7-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-7-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-8-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-8-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Supplemental Material
sj-JPG-9-nnr-10.1177_15459683241310984 – Supplemental material for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses
Supplemental material, sj-JPG-9-nnr-10.1177_15459683241310984 for Exploring Non-invasive Brain Stimulation Effects on Physical Outcomes in People With Parkinson’s Disease: An Umbrella Evidence Mapping Review With Meta-analyses by Dale M. Harris, Christopher Latella, Nicholas Tripodi and Steven J. O’Bryan in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
None.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Author Contributions
Dale M. Harris: Conceptualization; Data curation; Formal analysis; Methodology; Project administration; Writing—original draft; Writing—review & editing. Christopher Latella: Data curation; Methodology; Writing—review & editing. Nicholas Tripodi: Data curation; Formal analysis; Writing—review & editing. Steven J. O’Bryan: Data curation; Formal analysis; Methodology; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.