Abstract
Background
Investigating brain activation during motor imagery (MI) tasks in people with multiple sclerosis (pwMS) can increase the knowledge of the neural mechanisms underlying motor dysfunction in MS and, hopefully, aid in developing improved rehabilitation strategies.
Objective
To investigate brain activation in the supplementary motor area and premotor cortex via functional near-infrared spectroscopy (fNIRS) during a hand manipulation task, and comparing MI with actual practice (AP) in pwMS.
Methods
Each subject completed a sequence of 4 consecutive manual dexterity trials wearing an fNIRS device. The tasks included the following conditions: AP dominant hand, MI dominant hand, AP non-dominant hand, and MI non-dominant hand.
Results
Twenty pwMS (mean Expanded Disability Status Scale = 4.75 [3.0-6.5]) and 20 healthy controls (HC) participated in the study. According to the fNIRS timeline course, a similar increase (compared with baseline) was observed in the relative oxygenated hemoglobin (HbO) concentration during the MI and AP tasks, which was immediately followed by a decrease (for either hand) in the pwMS and the HC groups. A difference in the relative HbO concentration between the HC and pwMS was detected solely when the 2 groups mentally replicated the manual dexterity task movements in the MI condition (dominant hand). The increase was higher in the HC group (P = .030).
Conclusions
Despite exhibiting manual dexterity difficulties, pwMS demonstrated comparable neural activation patterns as the HCs during MI tasks in regions associated with motor planning and complex movement control, thus, suggesting that deficits in manual dexterity among pwMS may not solely originate from impairments in the motor planning processes.
Introduction
Multiple sclerosis (MS) is an autoimmune disease affecting the central nervous system’s white and gray matter, and is characterized by chronic disease progression and a wide range of neurological symptoms. 1 MS occurs most commonly in young adults with a higher onset incidence between ages 20 and 40 years, and a double incidence in females. This disease is characterized by an unpredictable course with a wide range of neurological symptoms. 2
Upper limb (UL) function, including manual dexterity, is one of the most affected domains in people with MS (pwMS). Holper et al 3 highlighted the fact that 50% of pwMS reported self-perceived UL dysfunction. Despite this, dysfunction of the UL has often been considered less debilitating than lower limb impairment in pwMS. However, it was found to be associated with a loss of independence in activities of daily living, reduced quality of life, and limitations on participation.4,5
The supplementary motor area (SMA) and premotor cortical (PMC) areas are essential in planning, initiating, and coordinating voluntary motor actions crucial for precise hand movements. The SMA activation comprises various aspects of voluntary movement behaviors, particularly self-initiated actions, whereas the PMC contributes mainly to early movement planning and task selection processes.6,7 Lesions in the SMA and PMC can significantly impair manual dexterity, disrupting hand movement coordination, sequencing, and motor planning. Consequently, pwMS afflicted with these lesions experience weakness, clumsiness, and impaired hand movement control. 8 Interestingly, Fillipi et al 9 found that in pwMS, during a simple hand manipulation task, there is evidence of widespread recruitment of brain regions. Some of these regions are involved in processing the sensory information needed to accurately perform the movement. As the difficulty of the manipulation task increases, the role of cross-modal neurons and the frontal lobe becomes more prominent.
Motor imagery (MI) requires mentally simulating or rehearsing physical movements without actual execution. 10 Neuroimaging studies have demonstrated that MI activates brain neural circuits similar to when performed in real-time (with active movements), including activation in the SMA and PMC regions.11,12 By activating the SMA and PMC brain regions via MI, it is possible to facilitate motor learning, improve movement performance, and enhance functional recovery across various neurological populations.13,14
Notwithstanding its promise, only a limited number of studies have investigated the impact of MI on motor recovery in pwMS. According to a recent systematic review, MI interventions positively affected walking speed, distance, perceived fatigue, and quality of life in pwMS with mild disability. Moreover, MI has significantly improved dynamic balance and perceived walking ability, 15 however, caution is warranted due to the limited number (n = 5) and heterogeneity of the studies analyzed.
According to our literature search, no study has as yet examined the effect of MI on brain cortical activation in pwMS. This is in contrast with the extensive research performed on this topic across various neurological populations, especially, stroke survivors.16,17 Investigating brain activation during MI tasks in pwMS can increase the knowledge of the neural mechanisms underlying motor dysfunction in MS and, hopefully, aid in developing new or improved rehabilitation strategies.
Therefore, the current study objective was to investigate brain activation via functional near-infrared spectroscopy (fNIRS) during a hand manipulation task, and comparing MI with actual practice (AP) in pwMS. Age- and gender-matched healthy adults served as controls. Given their significant role in manual dexterity, the brain region of interest (ROI) was the SMA and PMC. We hypothesized that the relative oxy and deoxyhemoglobin concentrations in the SMA and PMC would differ between AP and MI in both groups, with more significant differences observed in pwMS.
Materials and Methods
Study Design and Participants
This observational fNIRS case–control study was conducted at the Multiple Sclerosis Center, Sheba Medical Center, Tel-Hashomer, Israel, comprising a convenience sample of 20 pwMS (48.8 ± 11.2 years, 55.0% female) and 20 healthy controls (42.7 ± 15.4 years, 55.0% female). Due to a lack of comparable studies, no sample size calculation was performed. Nevertheless, our sample size was similar to other fNIRS gait studies researching pwMS.18 -22 pwMS were enrolled in the study according to the following inclusion criteria: (1) a neurologist-confirmed diagnosis of definite MS according to the revised McDonald criteria 23 ; (2) the Expanded Disability Status Scale (EDSS) ranging from 3.0 to 6.5 24 ; and (3) age range between 20 and 60 years. Exclusion criteria included: (1) corticosteroid treatment within 90 days prior to examination; (2) other significant neurological or psychiatric illnesses; (3) alcohol or drug abuse; (4) orthopedic disorders that could negatively affect UL movement; and (5) severe cognitive impairment (Mini-Mental State Examination ≤23). 25 The objectives and requirements of our study were explained to all participants who provided informed consent before their participation. Approval was obtained from the Tel-Aviv University Ethics Committee before the commencement of the study (Ethics Ref: 0005462-1) and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Experimental Procedure
The experiment was conducted in a single session. Following the acquisition of informed consent, the participants completed the Symbol Digit Modalities Test (SDMT), a measure of cognitive information processing, 26 the Nine-Hole Peg Test (NHPT), a measure of manual dexterity 27 ; and the Kinesthetic and Visual Imagery Questionnaire (KVIQ), assessing MI. 28 Each subject completed a sequence of 4 consecutive manual dexterity trials with a 1 minute rest interval between trials. All manual dexterity trials were performed with the subject wearing an fNIRS device (NIRSport2, NIRx Medical Technologies LLC, New York, USA). Each trial was repeated 3 times (30 seconds each trial, with a rest interval of 40 seconds).
The trials consisted of the following conditions performed in an identical order:
AP: all participants sat comfortably adjacent to a standard-sized table and instructed to perform a manual dexterity task (detailed separately) with their dominant hand.
MI: Similar conditions to the AP. Each participant was asked to mentally replicate the manual dexterity task movements as performed in the AP condition. MI was applied using a kinesthetic modality where participants were asked to focus on sensations caused by the actual movements and feelings of muscular activity during imagination. This ability was a priori checked since subjects with a KVIQ score of ≥12 points for the kinesthetic subscale had been previously enrolled. 29
Identical to AP performed with the non-dominant hand.
Identical to MI performed with the non-dominant hand.
The experiment was coded by the Python PsychoPy® software (Open Science Tools Ltd). 30 PsychoPy® (https://psychopy.org/index.html), an application used for creating experiments in behavioral sciences with precise spatial control and timing of stimuli. The coded experiment, including triggers for the walking trials was synced with the recording signal of the fNIRS system.
Equipment and Outcome Measures
Herein, a portable fNIRS system (NIRSport, NIRx Medical Technologies, NY, USA) was attached to a standardized cap (EasyCap GmBH, Herrsching, Germany), 58 cm in circumference, and equipped with 8 sources and 8 detectors with 8 short separation channels according to the International 10-20 system for EEG in order to cover the brain ROI. The average source-detector separation distance was 30 to 40 mm. The arrangement of the optodes was performed by the fNIRS Optodes’ Location Decider (fOLD) toolbox. 31 Additional information as to the sensitivity of the channels according to the fOLD toolbox is provided in the Supplementary Material. The cap was placed in the middle of the scalp between the nasion and inion and between the left preauricular and right preauricular point (reference point Cz). The applied fNIRS system operates at 2 different wavelengths (760/850 nm) and at a fixed sampling frequency of 7.81 Hz. The cortical areas captured were the SMA and PMC (Brodmann area 6).
FNIRS Data Processing
Homer3 (version 1.32.4) processed and converted the fNIRS data. 32 Non-existing values were replaced by spline interpolation (function hmrR PreprocessIntensity NAN). Channels with a too-weak or too-strong signal, as well as a too-high standard deviation, were excluded (function: hmrR PruneChannels: data range = 1 × 10−2 to 1 × 107; signal-to-noise threshold = 2; source-detector separation range: 0.0-45.0 mm). The preprocessed raw data were then converted to optical density data (function: hmR Intensity2OD). 32 The spline interpolation and a digital Savitzky-Golay filter motion removed artifacts (function: hmR MotionCorrectSplineSG: P = .99; frame size = 15 seconds). 33 The third order Butterworth bandpass filter was applied to diminish physiological artifacts (function: hmrR BandpassFilt: Bandpass Filter OpticalDensity), therefore, the high-pass filter was set at 0.01 Hz to minimize the proportion of oscillations associated with the vascular endothelial function. 34 The low-pass filter was set at 0.09 Hz to primarily filter out the Mayer waves. 35 Subsequently, the optical density data were converted to concentration data according to the Beer-Lambert law, adapting the differential path length factor to the age of each participant. 36 Finally, the individual hemodynamic response function (HRF) was calculated via the ordinary least squared deconvolution method by utilizing a general linear model approach (function: hmrR GLM). 37 The HRF was based on a consecutive sequence of Gaussian functions (width of the Gaussian 0.5 and temporal spacing between the consecutive Gaussian 0.5). The short separation regression was performed with the nearest short separation channel. The third-order polynomial drift baseline correction was applied. Subsequently, the data were processed by the MATLAB program (version R2020b, The MathWorks, Natick, MA, USA).
Initially, the early phase of task onset (5 seconds) was eliminated for each subject in order to avoid the transient effects of movement initiation on the hemodynamic response. 38 Subsequently, the last 5 seconds were eliminated to minimize the impact of the expected ending of the manual dexterity trial. Accordingly, data recorded during the 5- to 25-second interval from each manual dexterity trial were analyzed. Each channel’s relative oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) concentration data during this time interval were then averaged for each subject. Finally, the channels were merged into the cortical ROI as described above.
NHPT and Manual Dexterity Task
The NHPT tool was employed in 2 different ways: (1) assessment of manual dexterity (standard mode of use), and (2) the manual dexterity task for both AP and MI. In terms of assessment, the NHPT is recommended as a gold standard for measuring manual dexterity in pwMS. 27 It has excellent psychometric properties as to its reliability, discriminant, concurrent, and ecological validity; detects progression over time; is sensitive to treatment and is recommended for inclusion in clinical trials. 39 Briefly explained, the NHPT requires participants to repeatedly place 9 pegs into 9 holes, 1 at a time, as quickly as possible and quickly remove them from the holes. The total time needed to complete the task is then recorded. Two consecutive trials with the dominant hand are immediately followed by 2 consecutive trials with the non-dominant hand.
As for the manual dexterity task, the participants were instructed to place and remove the pegs from the holes identically as performed for the NHPT. However, in contrast with the standard mode of use in which the individual stops once all pegs are removed from the holes, the participants were instructed to continue performing the hand movements for 30 seconds in the task phase. Thus, in cases where the participant finished placing and removing the pegs in and out of the holes within the 30-second time period, they were instructed to repeat the task until the end of the 30-second time period, which was declared out loud by the tester. The rationale of this design was to keep a fixed and equal time frame for the AP and MI tasks in sync with the recording of the fNIRS system.
Statistical Analysis
Descriptive statistics determined the study sample’s demographics, clinical characteristics, and hemodynamic measures. Normal distribution was verified using the Kolmogorov–Smirnov test. The relative HbO and HbR concentration data were also examined using boxplots for the SMA and PMC. In the event of outlier identification, the data were removed. Study groups were compared for age and gender. A general linear model performed a repeated measures analysis of variance on the HbO, HbR, and HbT concentration data.
All analyses were performed using the SPSS software (version 28.0 for Windows; SPSS Inc., Chicago, IL, USA). All reported P-values were 2-tailed. The level of significance was set at P < .05. The figures illustrating the relative HbO concentration change in the cortical ROI during each group’s 4 manual dexterity trials were created using the R software program (R studio version 1.4.1717). The Ggplot2 library was utilized to create the plots by implementing the geom_smooth function, thus, producing a curve estimating the conditional mean function. The generalized additive model method was applied.
Results
Clinical characteristics of the 40 participants are summarized in Table 1. No differences were observed between pwMS and healthy controls in terms of age and gender. The median EDSS score of the pwMS was 4.75 [3.0-6.5], indicating moderate disability; the mean disease duration from diagnoses was 13.2 (SD = 11.3) years; 18 had the relapsing-remitting disease type, and 2, the progressive type. All participants were stable in terms of their disease-modifying drugs. PwMS presented with a slower cognitive processing speed (based on the SDMT) and poorer manual dexterity in both the dominant and non-dominant hands (based on the NHPT), compared with the healthy controls. The imagery was similar amongst the pwMS and the healthy controls (based on the KVIQ).
Demographical and Clinical Data of the Study Sample.
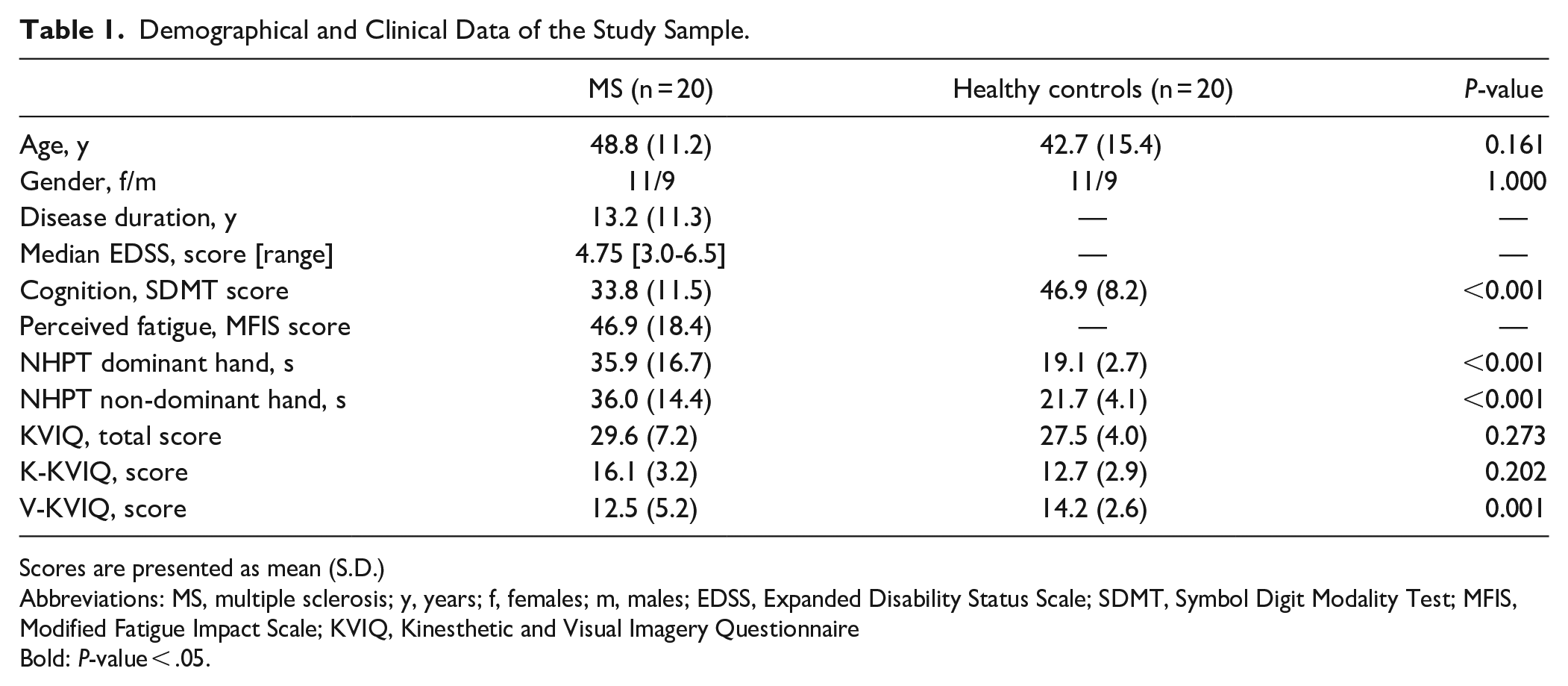
Scores are presented as mean (S.D.)
Abbreviations: MS, multiple sclerosis; y, years; f, females; m, males; EDSS, Expanded Disability Status Scale; SDMT, Symbol Digit Modality Test; MFIS, Modified Fatigue Impact Scale; KVIQ, Kinesthetic and Visual Imagery Questionnaire
Bold: P-value < .05.
Based on the FOLD software toolbox we calculated the relative HbO, HbR, and HbT concentration in the SMA and PMC according to the values of 3 main channels (4, 5, and 6), each with a specificity score above 80%. Scores from channel 5 were evident in 38 (out of 40) participants, scores from channel 4 were evident in 34 subjects, and scores from channel 6 were evident in 31 participants. All participants scored at least 2 (out of the 3) key channels.
The relative hemodynamic concentration in the SMA and PMC according to the 4 manual dexterity conditions, dominant/non-dominant hand, and study groups are presented in Table 2. A similar increase in the relative HbO or HbT concentration was observed during the MI and AP tasks (for either hand) in the pwMS and the HC groups. This observation is shown in Figure 1, presenting the mean relative HbO concentration time course in both the AP and MI groups. According to the timeline course illustration, there was a similar increase (compared with baseline) in the relative HbO concentration during the 30-second period, immediately followed by a decrease when the task was completed. A difference in the relative HbO and HbR concentrations between the HC and pwMS groups was observed only when the 2 groups mentally replicated the manual dexterity task movements in the MI condition. The increase in the relative HbO was higher in the HC group when performing with the dominant hand (P = .030; Table 3). Similarly, the HbR concentration was lower in the HC group compared to the pwMS group when performing with the non-dominant hand (P = .018). Table 4 presents the hemodynamic measures during the AP and MI tasks with the performance of both hands combined for each study group. The relative HbO and HbT concentration increase was similar between the AP and MI task conditions in both groups. However, a significant difference was found in the HbR reduction, which was more prominent during the AP task.
Mean (S.D.) of the Relative Oxy and Deoxyhemoglobin Concentrations in the SMA and PMC Cortical Region According to Task Condition in the Study Groups.
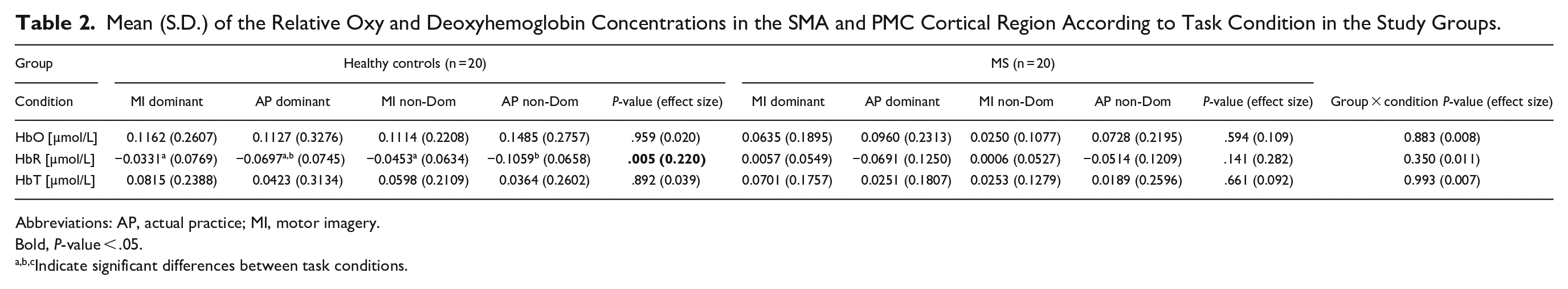
Abbreviations: AP, actual practice; MI, motor imagery.
Bold, P-value < .05.
Indicate significant differences between task conditions.
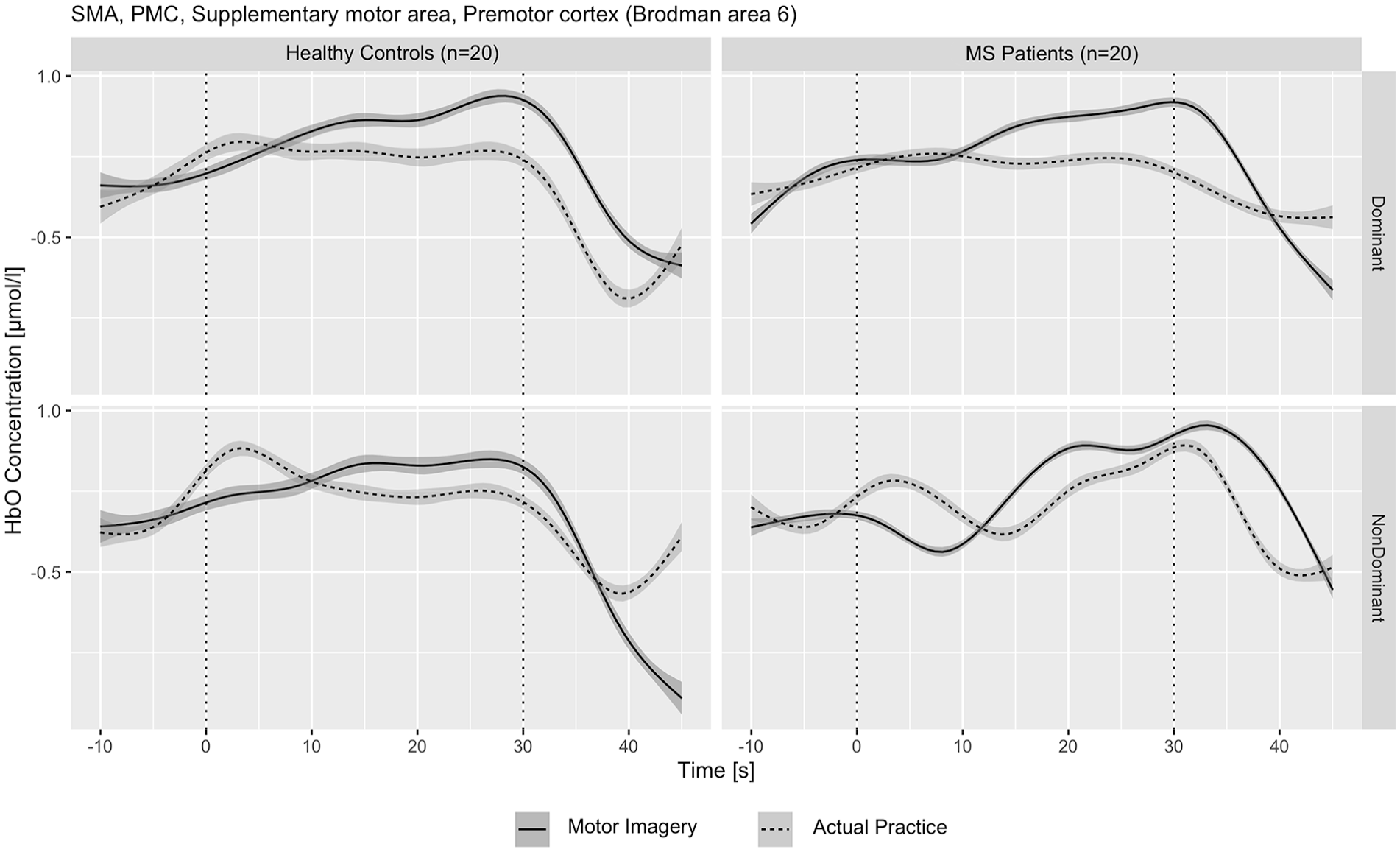
HbO concentration in the SMA and PMC according to task condition (MI/AP) and hand (Dominant/Non-dominant) in pwMS (MS) and healthy controls (HC).
Relative Oxy and Deoxyhemoglobin Concentrations Comparison Between MS and Healthy Controls According to Task Condition.
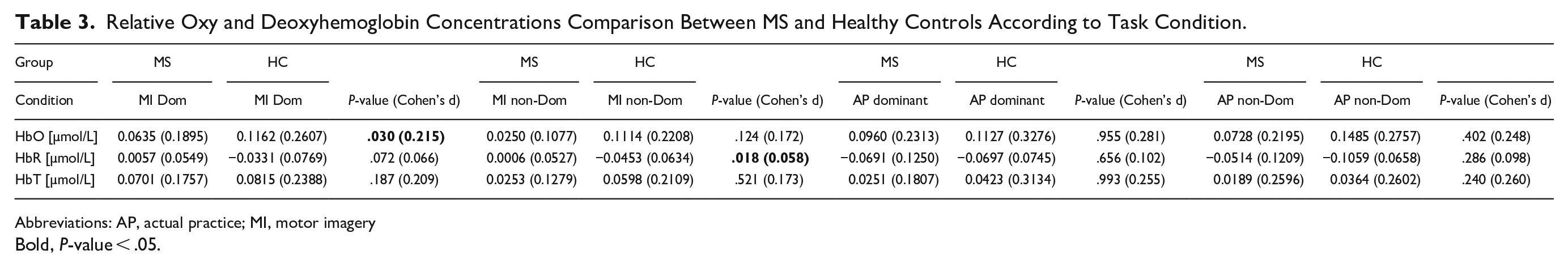
Abbreviations: AP, actual practice; MI, motor imagery
Bold, P-value < .05.
Comparison of Hemodynamic Values in the SMA and PMC According to Task Condition Within Each Study Group.
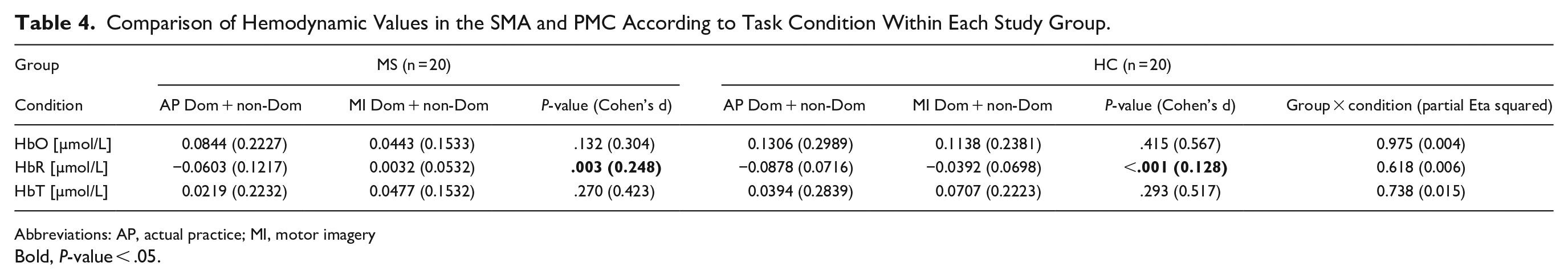
Abbreviations: AP, actual practice; MI, motor imagery
Bold, P-value < .05.
Figure 2 illustrates the relative HbO concentration according to task condition, separately for each hand, in the total group (n = 40). According to the time course, there was a similar increase in the HbO concentration during the first half of the trial (second 0-15) for both the AP and MI tasks. However, the increase in the HbO concentration was significantly higher in the MI task compared with the AP task during the second half of the trial (15-30 seconds).
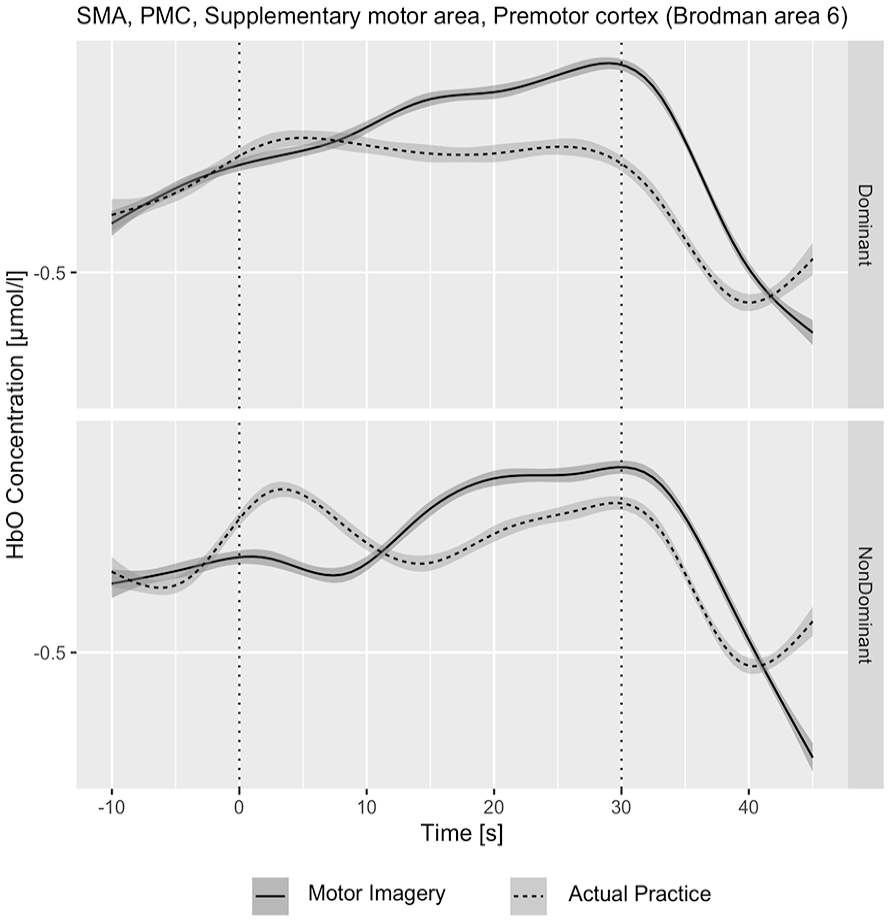
HbO concentration in the SMA and PMC according to task condition (MI/AP) and hand (Dominant/Non-dominant) in the total sample (n = 40).
Discussion
The primary aim of this study was to investigate cortical neural activity, specifically in the SM and PMC, during an MI task involving manual dexterity in pwMS. Our main finding confirmed that the relative concentration of HbO in the SMA and PMC similarly increases during a manual dexterity task, whether executed with actual movements or by MI, in the HC and pwMS. Moreover, the change in relative HbO concentration in the SMA and PMC during AP and MI was comparable between the pwMS and the HC, regardless of the fact that only pwMS exhibited manual dexterity difficulties.
Our research was motivated by the widespread use of MI in neurological rehabilitation and the limited information on its application in pwMS. Only a few studies have examined MI in pwMS, with most focusing on behavioral aspects, particularly gait.14,15 According to a systematic review on the efficacy of MI in pwMS, Gil-Bermejo-Bernardez-Zerpa et al 15 found that MI significantly improves walking speed, reduces fatigue, and enhances quality of life. Additional benefits have been observed in dynamic balance and perceived walking ability. 15 Regarding the UL, the impact of MI on hand function in pwMS is not well-documented. However, research on stroke survivors provides some insights. A recent systematic review found that MI can improve UL function in stroke survivors, but, only when combined with standard treatment. 40
MI is based on the theory of mirror neurons, demonstrating that individuals can induce plastic changes in the functionality of hand movements. 41 Several studies have identified patterns of brain activation that occur during MI, revealing that MI largely activates multiple brain regions. These include the frontoparietal network, subcortical and cerebellar regions, anterior intraparietal cortex, primary motor cortex, bilateral SMA, and PMC. 42 Furthermore, MI has been suggested as a valuable tool for accessing the brain’s motor network. 13 The impact of MI on brain activity in pwMS has been scarcely documented. Our literature search identified a single study that examined brain activation patterns via fMRI during MI in pwMS. 43 This study involved a simple squeeze-ball movement and found that pwMS exhibited different patterns of neural engagement during MI, with brain activation varying depending on whether the dominant or non-dominant hand was used for executing or imagining the movement.
Our study offers new insights into the effects of MI on brain activity in pwMS. Despite poor manual dexterity abilities, our findings indicated that pwMS exhibited comparable neural activation patterns in the HC during MI in brain regions related to motor planning and control of complex movements. This observation prompts several interpretations. Firstly, it suggests that deficits in manual dexterity among pwMS may not primarily stem from impairments in motor planning processes. Secondly, it underscores the potential resilience of certain motor-related brain regions, such as the PMC and SMA, due to MS relative to other components of the motor network. Nevertheless, it is crucial to acknowledge the restricted scope of our data due to equipment constraints, which precluded the assessment of additional brain regions relevant to voluntary movements, such as the primary sensorimotor cortex. In this context, a previous study examined cortical functional changes during a simple hand motor task in pwMS. 44 The authors found that compared to patients without pyramidal tract lesions, those with such lesions exhibited more significant activation in the contralateral primary sensorimotor cortex, secondary sensorimotor cortex, inferior central sulcus, and cingulate motor area. 45 Unfortunately, our study includes data only on the PMC and SMA, which limits our ability to make a comprehensive comparison. We, therefore, advocate for future fNIRS studies to address this gap in knowledge by exploring activity in other brain areas within the motor circuit beyond the PMC and SMA. For instance, recent research, including a study employing a 7 Tesla MRI, demonstrated a significant relationship between lesions in the primary sensorimotor cortex and hand disability in pwMS. 45 Interestingly, a previous fMRI study demonstrated alterations in connectivity patterns within the sensorimotor network during hand movement tasks in pwMS patients, exhibiting increased effective connectivity between the PMC and SMA regions, as well as reduced connectivity between the primary sensorimotor cortex and the cerebellum, relative to the controls. 46 These findings underscore the complexity of the neural mechanisms underlying manual dexterity deficits in pwMS, highlighting the need for continued investigation into this topic.
Another novel finding, was that MI produced more cortical activation compared with AP (in the dominant hand) but, solely in the healthy controls. No differences were observed between the MI and AP tasks in the MS group. We present several explanations for this comparison. In healthy adults, MI often activates premotor cortical areas more intensely than actual movement due to the complex cognitive processes involved in simulating movement. 12 Imagining a movement requires detailed cognitive rehearsal, heavily engaging the premotor cortex in planning and adjustment. This mental simulation, combined with reliance on internal movement models and the absence of sensory feedback, leads to heightened premotor cortex activation. 47
In contrast, no significant differences between MI and AP were observed in the MS group. This lack of differentiation may result from impaired connectivity within the motor pathway network, including the premotor cortex, primary motor cortex, basal ganglia, and cerebellum, leading to suboptimal engagement of the PMC and SMA. Moreover, the increased cognitive load and fatigue in pwMS can reduce activation of the PMC and SMC during MI. Altered neuroplasticity, which affects the brain’s ability to reorganize and form new connections, may also lead to atypical recruitment of the PMA and SMA during MI. 48
The study has several strengths; however, some limitations should be mentioned. One general limitation of this study was that only 1 cap size (58 cm circumference) was used for all subjects, which may have led to inaccuracies in the measurement results. Furthermore, due to the explanatory nature of the study, our data do not include measures of the MS brain lesions. Surprisingly, according to our literature search, MS brain lesion metrics are missing in fNIRS studies performed on pwMS. Future studies are encouraged to supplement this data as it will improve the validity of fNIRS studies investigating MS. Another limitation is the absence of information regarding additional brain structure and regions involved within the motor system (eg, cerebellum, primary motor cortex, and posterior parietal cortex). This limitation can be attributed to the fNIRS system in use, which could not cover additional motor brain regions with an acceptable level of sensitivity. Future studies are encouraged to complete this information with better-equipped fNIRS systems.
Conclusions
In conclusion, our study provides valuable insights into the neural mechanisms underlying MI in pwMS. Despite exhibiting manual dexterity difficulties, pwMS demonstrated comparable neural activation patterns as the HCs during MI tasks in regions associated with motor planning and complex movement control, suggesting that deficits in manual dexterity among pwMS may not solely originate from impairments in motor planning processes. Moreover, our findings illuminate the potential resilience of certain motor-related brain regions, such as the PMC and SMA in MS pathology. Future research should further enhance our understanding of the complex interplay between brain pathology, motor function, and MI in pwMS, paving the way to the development of more effective rehabilitation strategies for individuals with this condition.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241298260 – Supplemental material for Supplementary and Premotor Cortical Activation During Manual Dexterity Involving Motor Imagery in Multiple Sclerosis: A Functional Near-Infrared Spectroscopy Study
Supplemental material, sj-docx-1-nnr-10.1177_15459683241298260 for Supplementary and Premotor Cortical Activation During Manual Dexterity Involving Motor Imagery in Multiple Sclerosis: A Functional Near-Infrared Spectroscopy Study by Shaked Sadot, Sapir Dreyer-Alster and Alon Kalron in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Shaked Sadot: Data curation; Formal analysis; Visualization; Writing—review & editing. Sapir Dreyer-Alster: Data curation; Visualization; Writing—review & editing. Alon Kalron: Conceptualization; Formal analysis; Visualization; Writing—original draft
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
