Abstract
Background
Chronic hemiparetic stroke patients have very limited benefits from current therapies. Brain–computer interface (BCI) engaging the unaffected hemisphere has emerged as a promising novel therapeutic approach for chronic stroke rehabilitation.
Objectives
This study investigated the effectiveness of contralesionally-controlled BCI therapy in chronic stroke patients with impaired upper extremity motor function. We further explored neurophysiological features of motor recovery driven by BCI. We hypothesized that BCI therapy would induce a broad motor recovery in the upper extremity, and there would be corresponding changes in baseline theta and gamma oscillations, which have been shown to be associated with motor recovery.
Methods
Twenty-six prospectively enrolled chronic hemiparetic stroke patients performed a therapeutic BCI task for 12 weeks. Motor function assessment data and resting state electroencephalogram signals were acquired before initiating BCI therapy and across BCI therapy sessions. The Upper Extremity Fugl-Meyer assessment served as a primary motor outcome assessment tool. Theta–gamma cross-frequency coupling (CFC) was computed and correlated with motor recovery.
Results
Chronic stroke patients achieved significant motor improvement in both proximal and distal upper extremity with BCI therapy. Motor function improvement was independent of Botox application. Theta–gamma CFC enhanced bilaterally over the C3/C4 motor electrodes and positively correlated with motor recovery across BCI therapy sessions.
Conclusions
BCI therapy resulted in significant motor function improvement across the proximal and distal upper extremities of patients, which significantly correlated with theta–gamma CFC increases in the motor regions. This may represent rhythm-specific cortical oscillatory mechanism for BCI-driven rehabilitation in chronic stroke patients.
Trial Registration:
Advarra Study: https://classic.clinicaltrials.gov/ct2/show/NCT04338971 and Washington University Study: https://classic.clinicaltrials.gov/ct2/show/NCT03611855.
Introduction
Until recently, approaches for chronic stroke rehabilitation provided limited therapeutic benefits. Historically, motor improvements were thought to plateau at 3 months following stroke, leading to permanent motor deficits.1-3 However, recent studies employing brain–computer interface (BCI) strategies for motor rehabilitation have shown that significant functional improvements can be attained even during the chronic phase of stroke.4-7 Recently, the United States Food and Drug Administration (FDA) authorized the Neurolutions IpsiHand Upper Extremity Rehabilitation System (IpsiHand System), which is the first BCI therapy for chronic stroke-induced motor impairment. The IpsiHand System uses electroencephalographic (EEG) signals from the unaffected side of the brain to control a distal wearable exoskeleton around the affected hand. Early evidence has shown that regular use of the contralesionally-controlled BCI can facilitate motor recovery in chronic stroke patients.8-10
The involvement of the unaffected hemisphere in stroke recovery remains uncertain. Decreases in contralesional activity were associated with optimal recovery in some studies, which suggests that the contralesional hemisphere impedes recovery.11-14 Other studies have shown that increased activity in the contralesional hemisphere is associated with improved motor function.15,16 Given the inverse correlation between motor recovery and the extent of corticospinal tract transection, 17 we hypothesized that utilizing contralesional hemisphere activity to drive a BCI-controlled exoskeleton could potentially result in functional enhancements. The utilization of the uninjured motor cortex as the signal substrate for BCI motor rehabilitation has further reinforced the positive contribution of the unaffected hemisphere in motor recovery. 9 A BCI provides very precise temporal coupling of the cortical physiology associated with motor intention from the uninjured hemisphere to the sensory feedback provided to the hand. This BCI-enhanced cortico-sensory coupling is thought to create a Hebbian state to facilitate neuroplastic changes that can improve the brain’s control of the impaired hand. The cortical signals used by the IpsiHand System are low-frequency thalamocortical rhythms in the mu (8-12 Hz) and beta (13-29 Hz) bands that are thought to enable thalamic modulation of the cortex.18,19 Given that these rhythms have a broad cortical distribution, 20 it remains unclear to what extent the neuroplastic changes induced by the IpsiHand are specific to the hand or whether they have a wider functional effect. Given the emerging nature of this intervention, more data is necessary to define better the magnitude and extent of the benefit provided to these patients.
The coupling between various frequency bands could serve as a potential mechanism for motor learning. Recently, there has been growing interest in exploring complex neural oscillation properties, such as cross-frequency coupling (CFC), which involves synchronization between low-frequency oscillation phase and higher-frequency oscillation amplitude.21-24 The mechanisms of learning have been extensively investigated in the hippocampus, with theta–gamma CFC proposed as a pivotal mechanism related to learning,25-28 It has been shown that CFC similarly plays a role in learning across neocortical regions.24,29 Enhancement of theta–gamma coupling through transcranial alternating current stimulation over motor cortex during motor learning has shown significant improvements in motor skill acquisition. 30 Moreover, theta–gamma coupling has been demonstrated to correlate with functional improvements in chronic stroke rehabilitation. 31 These findings suggest a potential involvement of theta–gamma CFC in motor skill learning.
Here, we aimed to explore the effectiveness of the IpsiHand System for chronic stroke patients with impaired upper extremity motor function. Given that the IpsiHand System is controlled by thalamocortical motor rhythms, we hypothesized that the entire arm would demonstrate a functional improvement. We specifically aimed to assess the global improvement in upper extremity function and also to determine whether the functional benefit was regionally specific (distal limb only) or more general (distal and proximal limb). Further, we assessed the correlation between functional improvements and theta–gamma coupling, a potential mechanism previously implicated in motor recovery. In aggregate, this study demonstrates the robust benefit that the IpsiHand System provides to the entire upper limb and the associated cortical dynamics.
Methods
Study Participants
This study adopted a prospective design, wherein the registration date documented in the trial registry preceded the enrollment date as recorded in the trial registry. One hundred chronic stroke patients with upper-limb hemiparesis consented to participate in the study. Thirty patients completed BCI therapy for 12 weeks (see Supplemental Figure 1). The high number of dropouts in our study can be attributed to the trial requirements and the influence of the coronavirus disease 2019 (COVID-19) pandemic. Thirty-one patients did not meet inclusion/exclusion criteria. Fifteen patients were unable to consistently generate identifiable BCI control signals across 2 EEG screenings. The trial was heavily impacted by COVID-19, leading to the withdrawal of 24 patients. Inclusion criteria were stroke at least 6 months prior to this study; unilateral upper extremity weakness. We enrolled patients with varying degrees of hemiparesis, from mild to severe, to investigate the impact of BCI therapy on motor recovery based on the severity of baseline motor impairment. Exclusion criteria were severe visual, cognitive, or psychiatric impairment; severe aphasia, ataxia, unilateral neglect; impaired tactile; or proprioceptive sensation in the affected upper extremity. We additionally excluded 4 patients due to their enrollment before the official start date of the study. Twenty-six prospectively enrolled patients from 2 studies with the same experimental protocol were included in the manuscript (Advarra Study: ClinicalTrials.gov Identifier NCT04338971, approval number 201506124, 16 patients; Washington University Study: ClinicalTrials.gov Identifier NCT03611855, approval number 201701021, 10 patients). The Advarra and Washington University cohorts comprised chronic stroke patients, who were prospectively enrolled between May 5, 2017 and October 9, 2020, and between April 26, 2018 and March 18, 2020, respectively. Motor function outcomes were primarily evaluated using the validated Upper Extremity Fugl-Meyer (UEFM) assessment. We also assessed UEFM section scores to measure motor function improvements across the proximal and distal upper extremities (refer to Supplemental Table 1 for the methodology used to calculate UEFM section scores). 32 Secondary measures included the Action Research Arm Test (ARAT), Arm Motor Ability Test (AMAT), Motricity Index, Gross Grasp (Hand Grip Dynamometer), and Modified Ashworth Scale (MAS) at the wrist and elbow. Patients received Botox injections, which coincided with their BCI therapy (Supplemental Table 2). Thus, we further investigated if motor function improvement following BCI therapy was significantly influenced by Botox application. The written informed consent process followed Good Clinical Practice Guidelines. All questions were addressed, and patients verbalized understanding prior to signing informed consent.
BCI System Design
The IpsiHand System consisted of a wireless EEG headset with 6 active electrodes, EEG amplifier, and robotic hand orthosis (the FDA-approved IpsiHand Upper Extremity Rehabilitation System, Neurolutions, Santa Cruz, CA, USA; Figure 1A). Bluetooth-enabled touchscreen tablet was connected to the EEG headset to record brain signals. Local Wi-Fi network facilitated communication between the tablet and orthosis. The tablet guided patients through BCI tasks and converted changes in spectral power into control signals for the orthosis to open and close it in a 3-finger pinch grip. During the BCI task, patients were instructed to open the orthosis through motor imagery of their affected hand or to maintain it closed by resting quietly. The orthosis opened and closed in response to alterations in the power of the patient-specific control signal. Subjects who could partially move their affected arm were instructed to allow passive movements facilitated by the orthotic device.
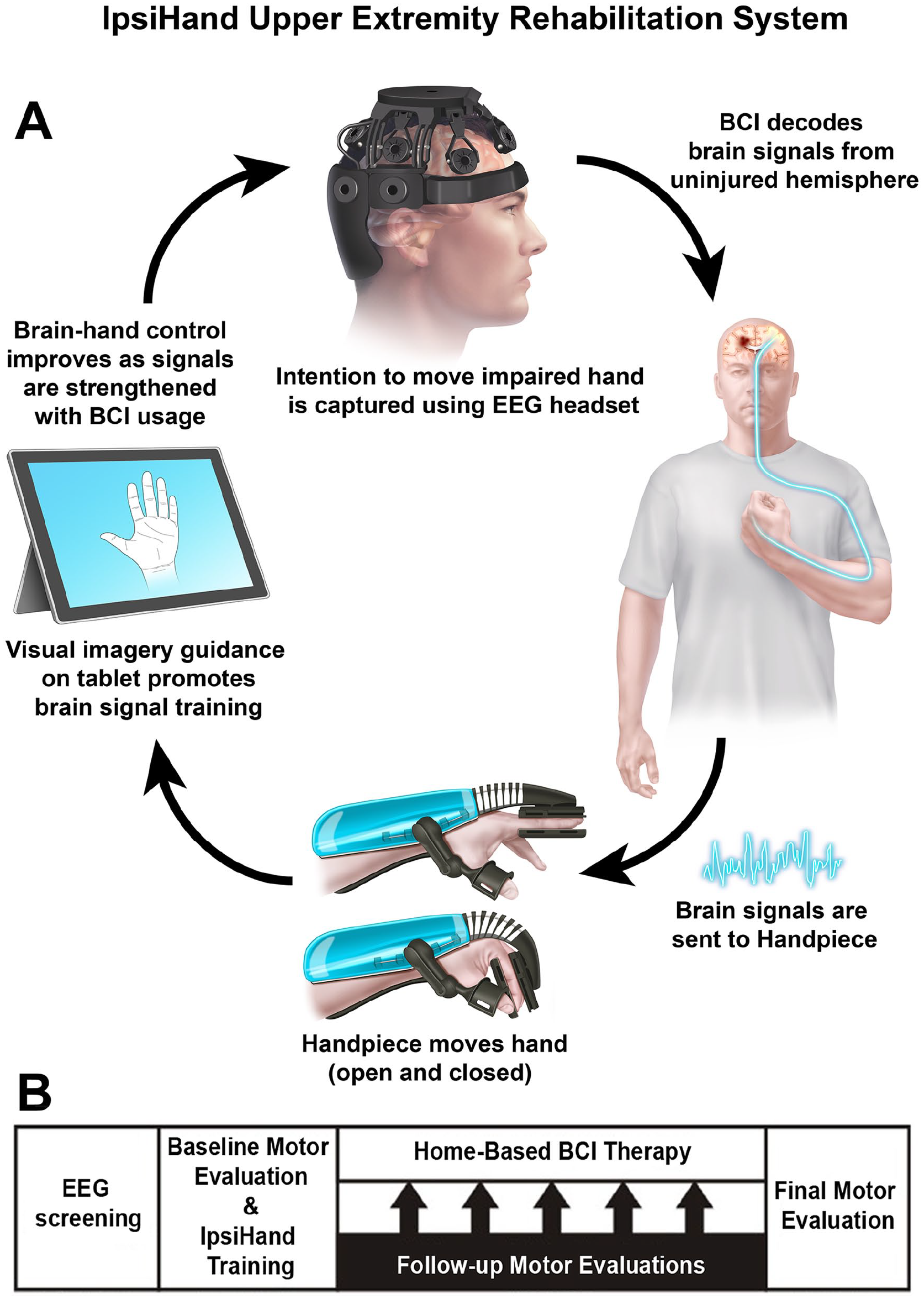
Experimental design. (A) BCI System Design. Patients performed motor imagery tasks. Contralesional EEG signals were decoded and translated into commands to open or close the orthosis. The orthosis provided proprioceptive sensory feedback. (B) 12-week BCI Therapy Protocol. Eligible patients underwent motor function assessments and resting EEG recordings prior to commencing BCI therapy and at 4-week intervals after completion of a BCI therapy session.
Intervention Protocol
Figure 1B illustrates the BCI therapy timeline. Prior to the therapeutic intervention, a screening session was conducted where patients performed a series of rest and motor imagery trials. The BCI device control signal was determined based on 1 Hz width frequency band with spectral power modulation that best aligned with the differences between rest and motor trials. The control signal was within the mu (8-12 Hz) or beta (13-29 Hz) frequency band and remained unchanged for each patient throughout BCI therapy. Only patients who exhibited a consistent feature frequency were included in the study. Baseline motor function was assessed by physical and occupational therapists before commencing the therapy. Research team members then provided training to patients on how to use the BCI system. Patients were instructed to use the device for 1 hour/day, 5 days/week, and over the course of 12 weeks. To avoid the observer bias, motor assessment was performed by the same clinician per patient before commencing BCI therapy and after completion of a BCI therapy session at 4-week intervals. Patients did not receive concomitant occupational or physical therapy while undergoing the BCI therapy over the 12-week period.
Each session of BCI therapy had a duration of approximately 1 hour and comprised 1 calibration phase followed by 5 BCI therapy runs. The pre-therapy calibration was employed to ensure data quality and identify motor imagery activity during the BCI task. During calibration, patients rested quietly and then proceeded to complete a series of task blocks and rest trials. Within the task blocks, patients were instructed to mentally envision moving their affected hand. The orthosis remained stationary during calibration. After the calibration phase, patients initiated the BCI therapy runs. Each run consisted of 30 motor imagery and 30 rest trials, with the trial order randomized. A 3-second “fidget” period was included between each 8-second trial, encouraging patients to blink or make minor physical adjustments. Upon completing a BCI therapy run, the system paused, allowing patients to rest before resuming their therapy. Resting state EEG data from pre-task calibration sessions were stored for subsequent analysis.
Electroencephalogram Recording and Processing
EEG data were acquired using 6 wireless dry electrodes (F3, F4, C3, C4, P3, and P4) mounted on an EEG headset in the International 10 to 20 System (Neurolutions, Santa Cruz, CA, USA). The EEG signals were sampled at 300 Hz, and ground electrode was placed on the forehead. Electrode impedance was maintained below 10 kΩ. EEG data obtained during the pre-therapy calibration rest period were analyzed across BCI therapy sessions (Pre-BCI, Post-BCI 4th, 8th, and 12th week; each 5 minutes long). The EEG recordings underwent band-pass filtering between 1 and 100 Hz. A 60 Hz notch filter was applied to eliminate environmental noise. The independent component analysis (ICA) was applied for artifact attenuation.33-35 Independent components associated with eye blinks, lateral eye movements, muscle-related activity, and cardiac artifacts were identified and eliminated. The EEG data were re-referenced using a common average reference.
Prior research has shown that non-invasive scalp EEG can detect alterations in high gamma power associated with motor tasks. 36 Scalp EEG was found to effectively record high gamma activity up to 100 Hz.22,37-40 We conducted time–frequency analysis to visualize resting high gamma cortical oscillations. 39 EEG was filtered offline using a FIR band pass filter from 65 to 100 Hz. Data were segmented into 5 seconds epochs. A Morlet wavelet convolution was computed using the channel time–frequency option. Thirty-five linearly spaced frequencies were computed between 65 and 100 Hz. For each patient, time–frequency data were averaged across all epochs per condition. The grand average time–frequency maps were obtained by averaging data across patients (see Supplemental Figure 2A, top panels). High gamma oscillations were then averaged across 65-100 Hz to visualize a single high gamma frequency wave (see Supplemental Figure 2A, bottom panels).
Cross-Frequency Coupling
CFC was calculated on resting EEG data, during which patients rested quietly and the orthosis was not active. The raw EEG recording was band-pass filtered in the theta and gamma frequency bands. The data were segmented into 5 seconds epochs. We applied a Hilbert transform to derive the complex-valued analytic signal. Estimates of theta band phase and gamma band amplitude were extracted from the analytic signal. The coupling between theta band (4-8 Hz) phase and gamma band (65-100 Hz) amplitude was calculated using the Mean Vector Length approach. 23 For each patient, CFC values were averaged across all epochs. The grand average CFC plot was obtained by averaging data across patients. Theta–gamma CFC was computed across BCI therapy sessions and compared between sessions.
As a complimentary tool, Canolty maps were computed to illustrate the theta–gamma coupling. 23 The phase troughs of the low frequency were specified at theta frequency band (5 Hz). A time window of 1 sec was extracted around each of these troughs. A time–frequency decomposition of these short epochs was performed. The power of all the time–frequency maps was averaged to obtain the final Canolty maps (see Supplemental Figure 2B). This approach enabled us to visualize whether high-frequency power systematically varied systematically with the phase of low frequencies, known as CFC.
Statistical Analyses
Changes in motor assessment scores with BCI therapy were assessed in a repeated-measures Analysis of Variance (ANOVA) with within-subjects factor Session (Pre-BCI, Post-BCI 4th, 8th, and 12th week). We hypothesized that BCI therapy would induce motor recovery in the upper extremity. Planned contrasts were used to decompose differences in motor scores between Pre-BCI versus Post-BCI sessions. Subsequent contrasts were employed to examine differences in motor scores between Post-BCI sessions. A nonparametric permutation test was performed to examine the differences in mean CFC values following BCI therapy. 41 Nonparametric Spearman rank correlation analyses were conducted between motor assessment scores and theta–gamma CFC values. Correlations were also computed between motor assessment scores and BCI usage/performance data. In all statistical tests, the significance thresholds were set at .05, and the P-values underwent adjustment using a Bonferroni correction.
Data Availability
The data will be made available upon reasonable request to the corresponding author.
Results
BCI Usage and Performance
BCI usage data and the success rates of BCI trials are detailed in Supplemental Table 3. Patients had to enter their usage on a provided tracking sheet, which assisted them in documenting therapy times. Therapy dosage for patients was estimated by summing the number of runs with at least 10% accuracy on both movement imagery and rest trials. The usage of BCI varied among patients, with an average of 41.6 ± 18.7 hours (mean ± standard deviation [SD]). There were no adverse events.
Patients generally used their BCI systems effectively, achieving median move and rest success rates of 71.8% and 39.8%, respectively. A definition of a successful trial was reaching the BCI activation threshold for at least 1 second for move trials or staying under the activation threshold for the entire trial duration for rest trials. Most patients achieved higher success rates with movement imagery trials due to the more stringent criteria for success in rest trials.
Patient Characteristics
Patient characteristics and primary motor assessment scores are shown in Table 1. The age was 58.4 ± 11.8 years (mean ± SD), 7 females and 19 males. Overall, 14 patients had left hemispheric stroke, and 12 patients had right hemispheric stroke. This study included patients with an average post-stroke duration of 69.1 ± 82.9 (mean ± SD) months.
Patient characteristics and primary motor assessment scores.

Abbreviations: LUE, left upper extremity; RUE, right upper extremity; UE, upper extremity; UEFM, upper extremity Fugl-Meyer.
Motor Assessment Scores
The UEFM served as a primary motor outcome assessment tool. Patients achieved an increase in UEFM score following BCI therapy. The mean increase in UEFM score was 8.1 points which exceeded the minimal clinically significant difference (MCID) threshold of 5.25 points increase. 42 A total of 18 out of the 26 patients reached the MCID. None of the patients exhibited a decline in motor function. Both the Advarra and Washington University studies demonstrated significant motor improvements with BCI intervention, and there were no significant differences in terms of motor recovery between the 2 studies (see Supplemental Figure 3). The final changes in UEFM scores for the Advarra and Washington University Studies were 8.8 and 7.1 points, respectively, with a P-value of .38.
Primary and secondary motor assessment scores across BCI therapy sessions were presented in Figures 2 and 3. UEFM: the main effect of session was significant, F(3,75) = 32.66, P < .001. Planned contrasts revealed significant increases in UEFM scores. ARAT: the main effect of session was significant, F(3,75) = 16.32, P < .001. Planned contrasts revealed significant increases in ARAT scores. AMAT: the main effect of session was significant, F(3,75) = 26.13, P < .001. Planned contrasts revealed significant increases in AMAT scores. Motricity Index: the main effect of session was significant, F(3,75) = 19.48, P < .001. Planned contrasts revealed significant increases in motricity index scores. Gross Grasp: the main effect of session was significant, F(3,75) = 5.06, P = .003. Planned contrasts revealed significant increases in motricity index scores. Elbow and Wrist MAS: the main effect of session did not prove significant, F(3,75) = 2.06 and 0.02, P = .11 and.92, respectively, excluding significant changes across BCI therapy sessions.
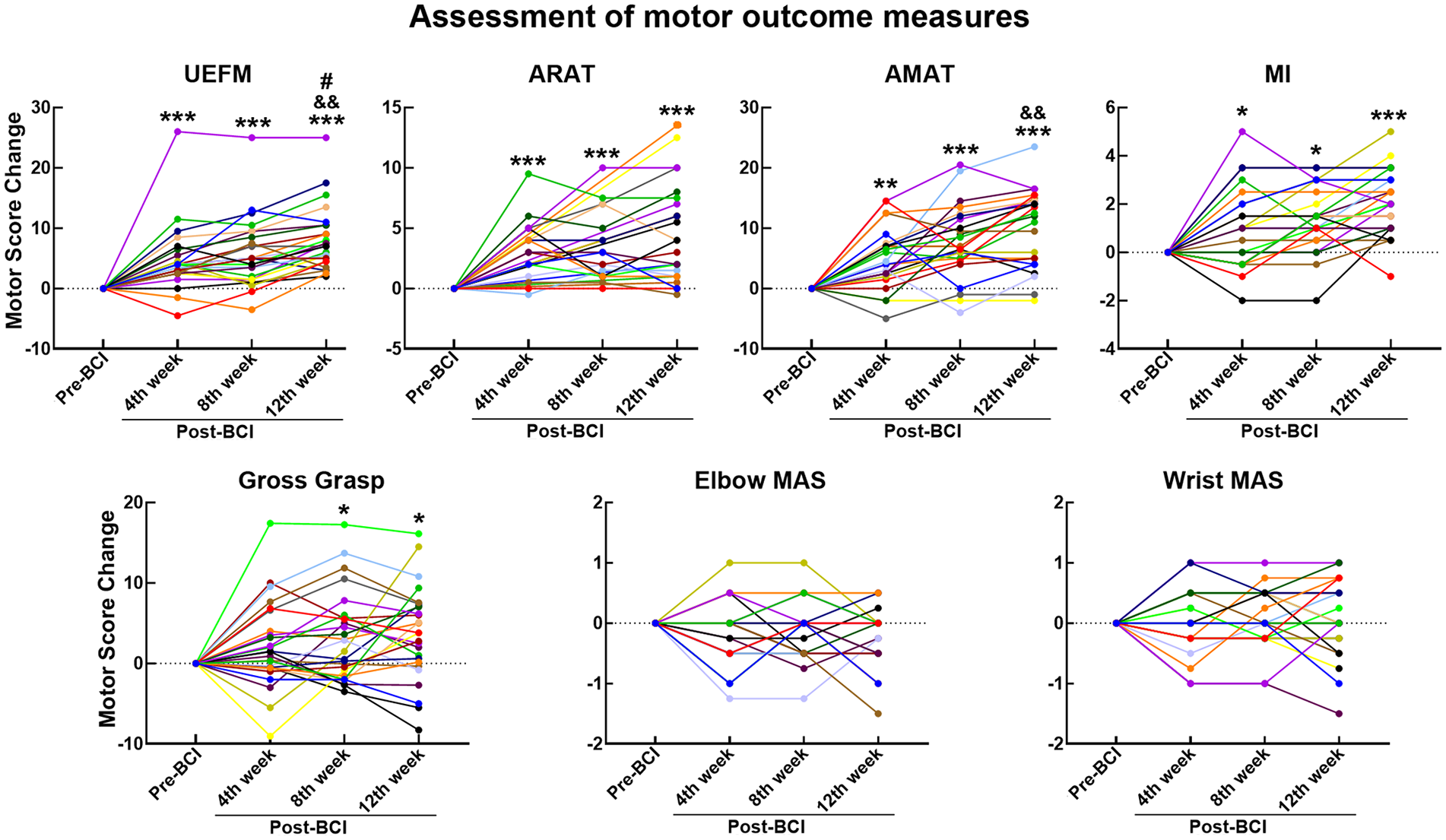
Motor assessment score changes. Y-axis, motor score change; X-axis, BCI therapy sessions.
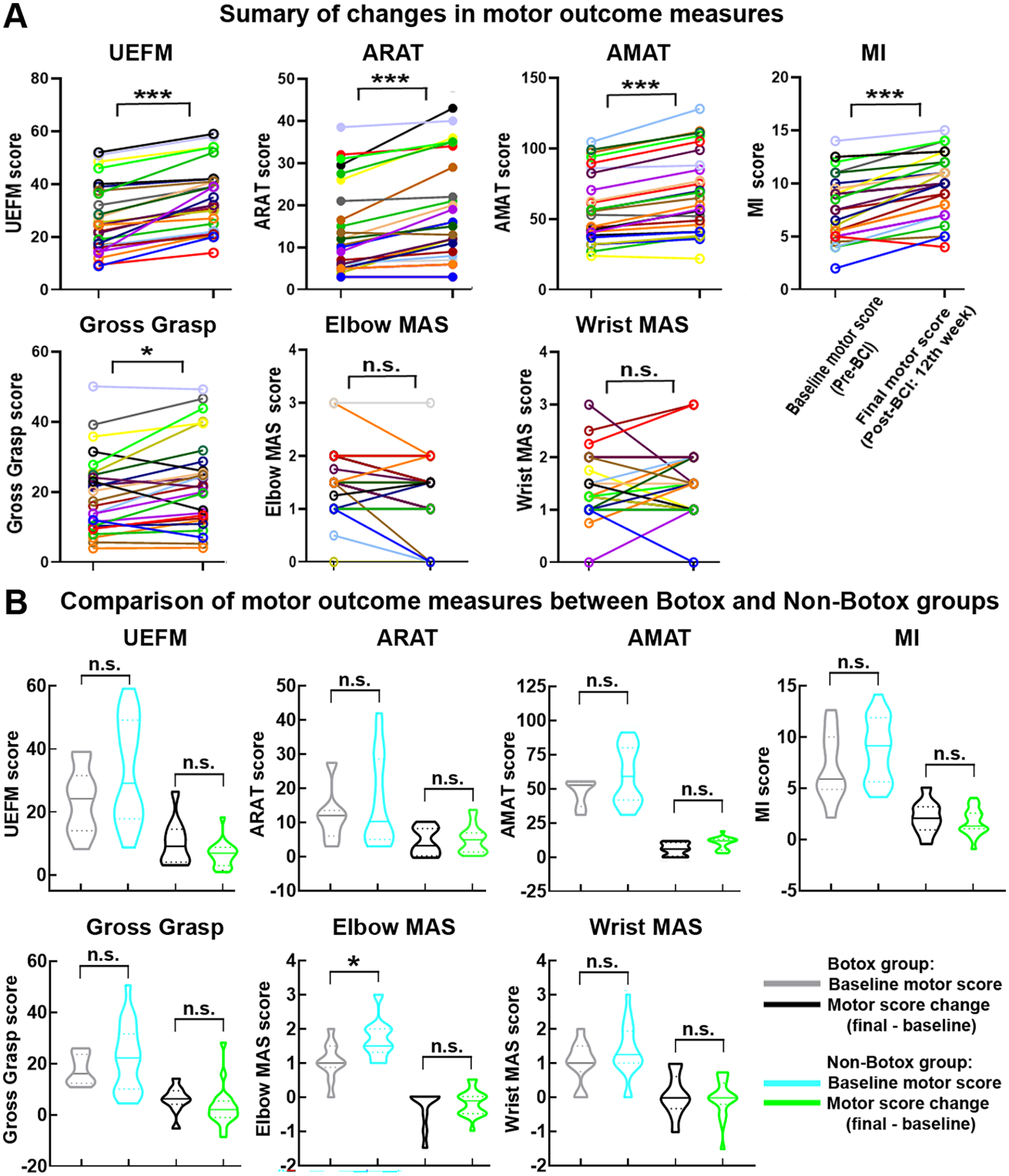
Summary of motor recovery measures. (A) Baseline and final motor scores. Patients are depicted in different colors. (B) Botox and Non-Botox groups. Baseline motor scores and final motor score changes. Y-axis, motor score; X-axis, BCI therapy sessions.
UEFM score changes showed significant positive correlations with BCI usage and move trial success rates (see Figure 4). There were no significant correlations between final UEFM score changes after 12 weeks of BCI treatment and baseline UEFM scores (r = -.22, P = .28), which excludes the differential effects of BCI therapy on motor recovery depending on the severity of baseline motor impairment.
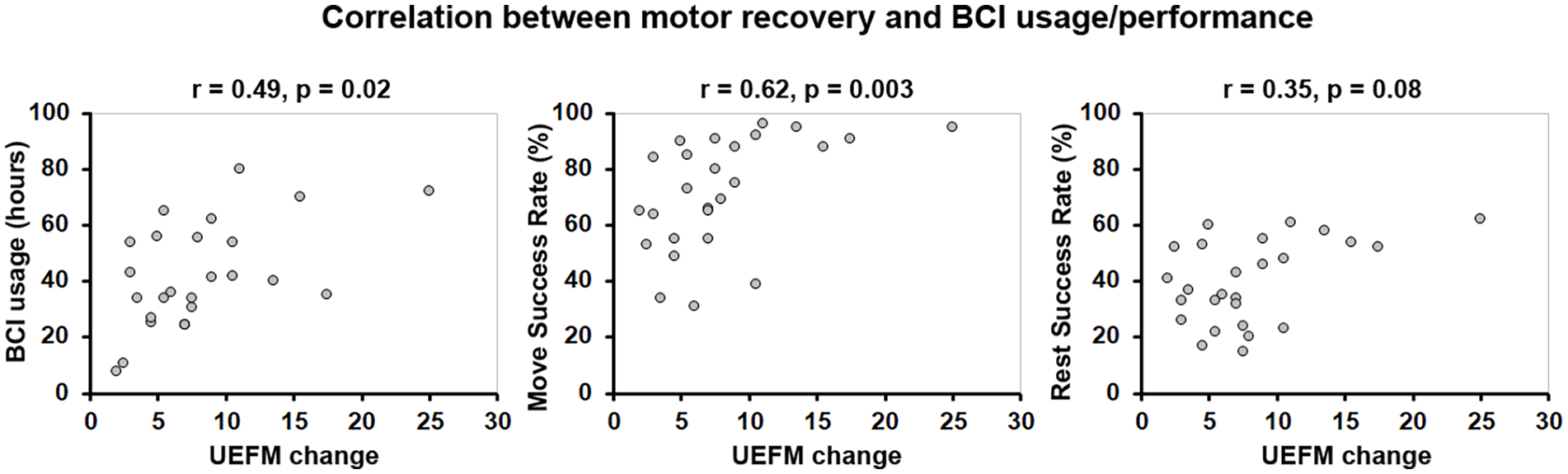
Correlation between UEFM scores and BCI usage/performance. Spearman rank correlations were run between changes in UEFM scores after 12 weeks of BCI treatment and BCI Usage, Move Success Rate, Rest Success Rate.
UEFM Section Scores
We evaluated changes in UEFM section scores following BCI therapy (see Supplemental Table 1 for UEFM section description). The results are summarized in Figure 5A. Section A1 (triceps/ biceps reflex): the main effect of session was not significant, F(3,75) = 2.45, P = .07, excluding significant changes across BCI therapy sessions. Sections A2, A3, and A4 (proximal upper extremity): the main effect of session was significant, F(3,75) = 15.99, 3.42, and 6.15, P < .001, .02 and .002, respectively. Planned contrasts revealed significant increases in UEFM section scores. Sections B and C (distal upper extremity): the main effect of session was significant, F(3,75) = 9.17 and 10.29, P < .001 and .001, respectively. Planned contrasts revealed significant increases in UEFM section scores. Section D (whole limb coordination): the main effect of session was significant, F(3,87) = 5.25, P = .003. Planned contrasts revealed significant increases in UEFM section scores.
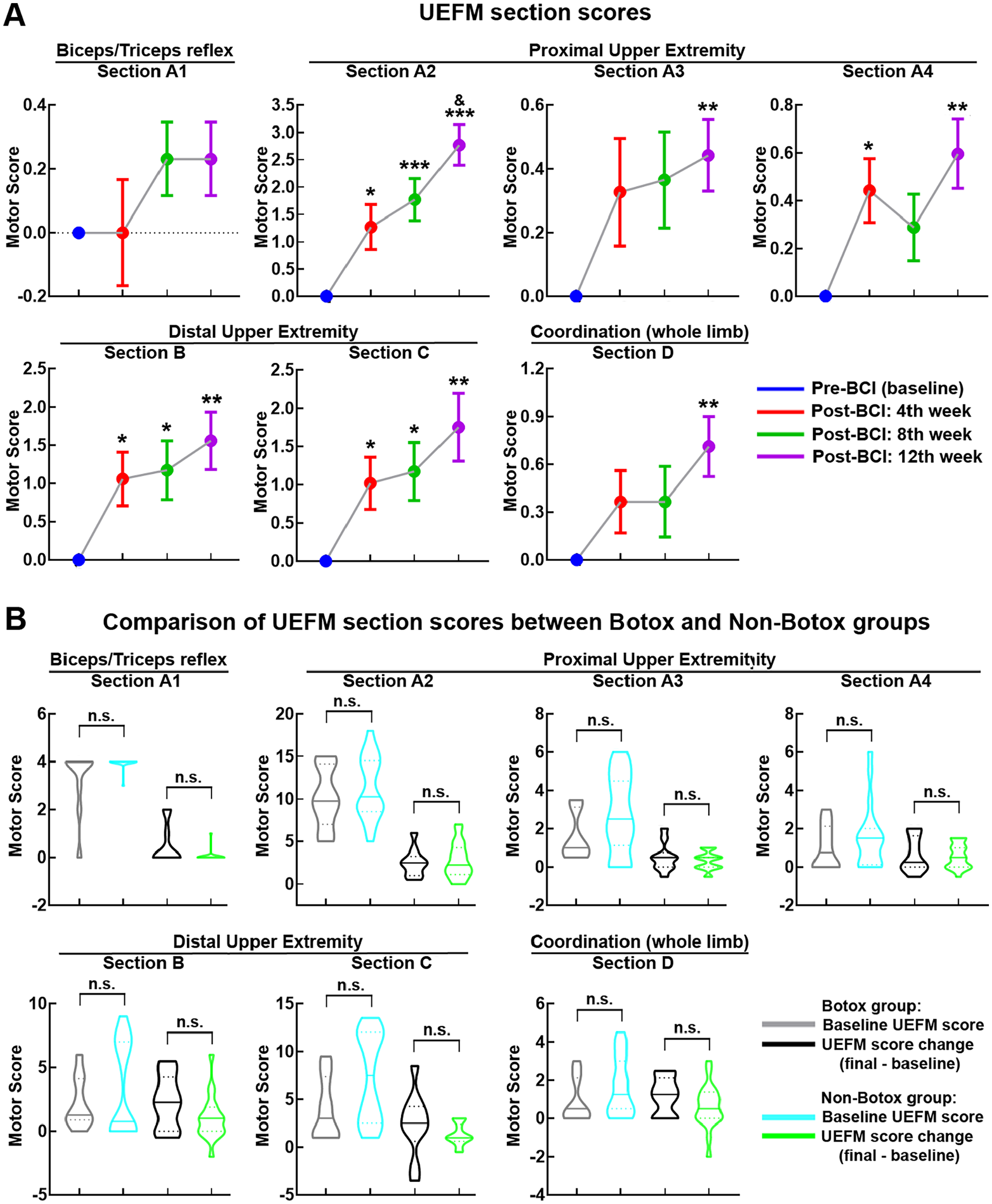
Section scores of UEFM. (A) UEFM section scores across BCI therapy sessions. (B) Botox and Non-Botox groups. Baseline UEFM section scores and final UEFM section score changes.
Comparison of Motor Recovery Between Botox and Non-Botox Groups
Both Botox and Non-Botox patients achieved greater than MCID gains in UEFM scores (10.7 and 6.9 points gain, respectively). The proportion of patients in each group who met the MCID for UEFM was similar: 67% (6/9) of Botox patients and 71% (12/17) of Non-Botox patients. Further statistical analyses were performed to assess if the influence of Botox application on motor recovery following BCI therapy was significant. Pre-BCI baseline motor scores and final motor score change (motor score Post-BCI 12th week—Pre-BCI baseline motor score) were compared between Botox (n = 9) and Non-Botox groups (n = 17; Figures 3B and 5B). The differences in baseline motor scores and final motor score change between groups were not statistically significant (all P’s > .05), except baseline Elbow MAS score. However, although baseline Elbow MAS score was significantly lower in Botox group compared with Non-Botox group (P = .03), groups did not significantly differ in final Elbow MAS score change (P = .51; Figure 3B). The Botox and Non-Botox groups were also compared for the UEFM section scores. The results presented in Figure 5B indicate that neither baseline motor scores nor final motor score changes were statistically different between groups for any section of the UEFM (all P’s > .05).
Modulation of Brain Activity
Theta–Gamma CFC
Supplemental Figure 4A depicts topographic representation of mean theta–gamma CFC values across BCI therapy sessions (Pre-BCI, Post-BCI 4th, 8th, and 12th week). We compared theta–gamma CFC values between Pre-BCI baseline and Post-BCI sessions as measured over all the electrode sites (Supplemental Figure 4B). During the earlier post-treatment session (Post-BCI fourth week), theta–gamma CFC increased at the C3 and C4 motor electrodes. This was in the expected direction but did not reach significance (both P’s > .05). The subsequent Post-BCI sessions were associated with significant bilateral enhancement of theta–gamma CFC over the motor electrodes (Post-BCI eighth week: significant effects at the C3 and C4 electrodes, both P’s < .05; Post-BCI 12th week: significant effects at the C3 and C4 electrodes, both P’s < .05). Post-BCI 12th week versus Post-BCI 8th week contrast did not reveal significant difference in theta–gamma CFC (all P’s > .05). Theta–gamma CFC over the frontal and parietal electrodes was not significantly modulated throughout BCI therapy (all P’s > .05).
Correlations Between Motor Recovery and Theta–Gamma CFC
Correlations between motor recovery and theta–gamma CFC change at the C3 and C4 electrodes across BCI therapy sessions relative to baseline are shown in Supplemental Figure 5. UEFM, AMAT, and Motricity Index score changes showed significant positive correlations with theta–gamma CFC change. ARAT, Gross Grasp, and Elbow and Wrist MAS score changes did not correlate significantly with theta–gamma CFC change. We also conducted correlation analyses for UEFM section scores (Supplemental Figure 6). The gains in both proximal and distal UEFM section scores correlated significantly with theta–gamma CFC change at the C3 and C4 electrodes.
Discussion
This study provides compelling evidence demonstrating that the use of a contralesionally-controlled BCI for chronic stroke therapy, the IpsiHand System, induced clinically significant motor function improvement. Despite being more than 6 months post-stroke, patients with chronic stroke achieved a significant increase in UEFM score. Importantly, we found improvement in both proximal and distal upper extremity section scores of UEFM. Additionally, improvements were observed in secondary measures of motor function. A significant increase was evident in ARAT, AMAT, Motricity Index, and Gross Grasp scores, which collectively assess proximal and distal upper extremity motor function. Conversely, the measurements of muscle spasticity, such as Elbow and Wrist MAS, did not exhibit significant changes following BCI intervention. These findings further corroborate previous evidence, reinforcing the notion that BCI-controlled rehabilitation systems can facilitate motor recovery.5,7 However, this study was unique in utilizing the unaffected hemisphere in a BCI rehabilitation system and demonstrating the benefit for improvement is widespread in the affected limb, setting it apart from previous research. Our findings provide support for the engagement of the unaffected hemisphere as an important substrate for motor rehabilitation.
According to a meta-analysis, 5 out of 9 BCI groups and 3 out of 9 control groups, which also included conventional therapy, showed motor improvements exceeding the minimal clinically important difference (MCID > 5.25). 10 However, the studies that reported clinically meaningful motor function gains had notably smaller sample size, ranging from as low as 7 to as high as 15 patients, in contrast to the 26 patients in our study. Furthermore, the time since stroke in those studies was considerably shorter, ranging from 2.2 ± 1.7 (mean ± SD) to 37.3 ± 43.9 (mean ± SD) months, whereas our study included patients with an average post-stroke duration of 69.1 ± 82.9 (mean ± SD) months. There is also heterogeneity among studies stemming from variations in patient characteristics, such as age, gender, and treatment adherence and duration, which complicates direct comparisons.
In this study, both Botox and Non-Botox groups achieved greater than MCID gains in UEFM scores. In addition, the proportion of patients in each group who met the MCID for UEFM was nearly the same. BCI therapy provided improvement in both proximal and distal upper extremity section scores of UEFM. The gains for each specific UEFM section were also not significantly different between Botox and Non-Botox groups. These results indicate that Botox application did not have a statistically significant impact on the benefit provided by the BCI therapy. The use of Botox to reduce spasticity may have functional effects that both facilitate and impair motor recovery. In a previous study, Botox application for post-stroke upper limb spasticity did not improve upper limb motor function, including activities such as grasping and reaching. 43 Botox has also been associated with increased motor impairment, including weakness of injected muscles. 44 Thus, while reducing muscle tone can enhance finer movements, it is important to acknowledge that it may also decrease strength and hinder overall motor function. Regardless, the presence of Botox did not facilitate or hinder the effect of the IpsiHand System’s functional benefits.
In addition to the functional gains associated with the use of the IpsiHand System, we also evaluated whether there were concomitant cortical electrophysiologic changes associated with these motor improvements. Given prior work that showed theta–gamma CFC was associated with BCI-driven motor rehabilitation, 31 we further evaluated the dynamic changes in theta–gamma CFC throughout BCI therapy to evaluate the neurophysiological mechanisms underlying motor recovery. A key electrophysiologic finding of our study was the bilateral enhancement of coupling between theta and gamma oscillations over the motor areas following BCI therapy. This effect showed a significant positive correlation with both proximal and distal upper limb motor improvement. While the exact functional significance of CFC remains uncertain, it is believed to play a crucial role in coordinating neuronal activity across brain regions.21,45 During the learning of item-context associations, theta–gamma coupling showed positive correlation with behavioral performance accuracy suggesting its involvement in learning process. 25 Theta–gamma coupling has been shown to emerge in the primary motor cortex when gamma-aminobutyric acidergic (GABAergic) activity is inhibited. 46 Reduced GABAergic activity in the primary motor cortex represents a central mechanism for motor plasticity. 47 This study suggests that enhanced synchronization of theta and gamma oscillations may play a mechanistic role in motor recovery.
Limitations
This study was conducted with the assumption that motor deficits would remain stable in the chronic stage of stroke. Indeed, previous research has shown limited improvement in motor deficits during the chronic stage of stroke.2,3,48,49 Thus, we did not have a control group for comparison with BCI therapy. Furthermore, in a separate study investigating motor rehabilitation in stroke patients, sham BCI therapy failed to facilitate motor recovery. 5 Consequently, we primarily attribute the observed improvement in motor function and associated electrophysiological findings in this study to the BCI intervention. However, we cannot currently determine whether enhanced theta–gamma coupling is specific to BCI techniques, or it represents a broader phenomenon associated with other methods utilized for chronic stroke rehabilitation. Additionally, because assessors were not blinded, there was a potential risk of assessor bias when evaluating the changes in motor outcomes over time.
Conclusions
IpsiHand BCI therapy resulted in significant improvement in both proximal and distal upper extremity motor function of chronic stroke patients, which was independent of Botox application. Motor recovery was paralleled with enhanced theta–gamma coupling over the motor regions, which may represent rhythm-specific cortical oscillatory mechanisms for BCI-driven motor rehabilitation.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241287731 – Supplemental material for IpsiHand Brain–Computer Interface Therapy Induces Broad Upper Extremity Motor Rehabilitation in Chronic Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683241287731 for IpsiHand Brain–Computer Interface Therapy Induces Broad Upper Extremity Motor Rehabilitation in Chronic Stroke by Nabi Rustamov, Lauren Souders, Lauren Sheehan, Alexandre Carter and Eric C. Leuthardt in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Nabi Rustamov: Formal analysis; Writing—original draft. Lauren Souders: Data curation; Writing—original draft. Lauren Sheehan: Data curation; Writing—original draft. Alexandre Carter: Funding acquisition; Investigation; Supervision; Writing—eview & editing. Eric Leuthardt: Conceptualization; Funding acquisition; Investigation; Supervision; Validation; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.C.L. owns stock in Neurolutions, Inner Cosmos, and Sora Neuroscience. Washington University owns stock in Neurolutions. This work and E.C.L. have had their conflict of interest rigorously evaluated and managed throughout this study and with the creation of this manuscript. Other authors have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health (NIH) R21NS102696 (E.C.L. and A.C.) and National Institute of Biomedical Imaging and Bioengineering (NIBIB) P41-EB018783 (E.C.L.).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
