Abstract
Background
Brain–computer interface (BCI) is a procedure involving brain activity in which neural status is provided to the participants for self-regulation. The current review aims to evaluate the effect sizes of clinical studies investigating the use of BCI-based rehabilitation interventions in restoring upper extremity function and effective methods to detect brain activity for motor recovery.
Methods
A computerized search of MEDLINE, CENTRAL, Web of Science, and PEDro was performed to identify relevant articles. We selected clinical trials that used BCI-based training for post-stroke patients and provided motor assessment scores before and after the intervention. The pooled standardized mean differences of BCI-based training were calculated using the random-effects model.
Results
We initially identified 655 potentially relevant articles; finally, 16 articles fulfilled the inclusion criteria, involving 382 participants. A significant effect of neurofeedback intervention for the paretic upper limb was observed (standardized mean difference = .48, [.16-.80], P = .006). However, the effect estimates were moderately heterogeneous among the studies (I2 = 45%, P = .03). Subgroup analysis of the method of measurement of brain activity indicated the effectiveness of the algorithm focusing on sensorimotor rhythm.
Conclusion
This meta-analysis suggested that BCI-based training was superior to conventional interventions for motor recovery of the upper limbs in patients with stroke. However, the results are not conclusive because of a high risk of bias and a large degree of heterogeneity due to the differences in the BCI interventions and the participants; therefore, further studies involving larger cohorts are required to confirm these results.
Introduction
Stroke is a major cause of disability in survivors, often resulting in permanent motor or cognitive impairment. In addition, the number of people requiring assistance with activities of daily life has drastically increased and will continue to escalate in the coming decades due to demographic factors. 1 In particular, recovery from upper limb disability is vital as it is highly prevalent and vastly reduces independence in activities of daily living and social participation for patients.
Various types of rehabilitation training have been applied to facilitate motor recovery in patients with paretic stroke, including constraint-induced therapy, 2 robot-based therapy, 3 and functional electric stimulation. 4 In addition to these types of training, recent advances in the technology for measuring neural activity have led to the development of neurofeedback. Neurofeedback is a type of biofeedback, and the measured brain activity is presented as visual or auditory information, or using other modes of representation to users/patients to facilitate self-regulation of the putative neural substrates. The behavioral changes resulting from self-manipulation of neural activity indicate that physiological regulation by neurofeedback may be considered a form of endogenous neural stimulation. 5 In a typical neurofeedback trial for rehabilitation, patients are instructed to self-regulate their electroencephalogram (EEG) activity, which electrically records the activity of the brain through motor imagery (MI). Moreover, by extracting relevant features in recent studies, the brain activity could be used as a trigger for contingent sensory feedback. The feedback can be delivered in an abstract form (moving cursor on a monitor) or as an embodied method (somatosensory representations delivered through robotic, haptic, or neuromuscular electrical stimulation [NMES] systems) that reproduces the intended movement.
Brain–computer interface (BCI) techniques are mainly being explored in clinical applications, such as in assistive technologies and rehabilitative strategies. Assistive technologies aim to compensate for lost functions (communication in locked-in syndrome or movements in paralysis) by controlling robotic actuators or functional electrical stimulation by detecting bio-signals. Rehabilitation strategies aim to enhance neuroplasticity through manipulation or self-regulation of neurophysiological activity, facilitating motor recovery.
The original BCI-based rehabilitation for patients with stroke was conducted as a single case study in 2009 and reported the feasibility of the BCI technique combined with functional electrical stimulation (FES). 6 Subsequently, the effects of BCI-based rehabilitation on motor recovery in paretic limbs have been suggested in many well-designed clinical studies, and a recent meta-analysis also supported the immediate effects of BCI training on upper extremity motor function in stroke.7–9 Lesioned brain networks after stroke rely on neural activity,10,11 and coincident activation of presynaptic and postsynaptic neurons can reinforce synaptic strength, resulting in increased and more reliable communication between the activated neurons. BCI-based rehabilitation has predominantly focused on cortical reorganization of the lesioned hemisphere through the potential relevance of this concept. 12 Specifically, BCI training has encompassed controlling the neural activity of the ipsilesional sensorimotor area (sensorimotor rhythm [SMR], also referred to as Rolandic alpha or mu-rhythm). Regarding the SMR, the power is decreased while attempting to move the limbs and is associated with an increase in the excitability of the motor cortex, 13 disinhibition of GABAergic inhibitory interneurons, 14 and increased excitability of the corticospinal tract. 15 On the other hand, several previous studies have reported a significantly reduced SMR in the lesioned hemisphere in stroke patients with poor recovery.16,17 Therefore, rehabilitation training using a feedback system associated with the SMR focuses on increasing the neural activity by purposefully modulating sensorimotor oscillations through Hebbian neuroplasticity and error/reward-based learning. 18
Recent studies have reported neurological and behavioral improvements through BCI training,19–21 and several systematic reviews recently indicated that BCI-based rehabilitative intervention is a promising approach in patients with stroke.8,9,22,23 However, BCI algorithms for motor recovery through re-established neural activities are designed to detect and decode various brain signals in each study; thus, an effective method of BCI training has not been identified so far. Therefore, the purpose of the current study was to summarize the evidence regarding effective methods to detect brain activity and conditions for motor recovery from the relevant clinical studies.
Materials and Methods
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement, 24 the PRISMA checklist (Supplementary Materials 1), and the Cochrane Handbook for Systematic Reviews of Interventions. The review protocol was prepared according to the PRISMA protocols (PRISMA-P) 25 and was registered with the International Prospective Register of Systematic Reviews (PROSPERO: CRD42021238795).
Search Strategy and Eligibility Criteria
We searched MEDLINE, Cochrane Central Register of Controlled Trials (CENTRAL), Web of Science, and Physiotherapy Evidence Database (PEDro). Searches used combined key terms, including “brain computer/machine interface,” “neurofeedback,” and “stroke,” which are Medical Subject Headings terms. These keywords include those used in previous studies7,8 (Supplementary Materials 2).
The inclusion criteria for this study were as follows: (1) published in a peer-reviewed journal; (2) a randomized controlled trial (RCT), quasi-experimental, and within-subject repeated design; (3) involving neurofeedback training using brain signals as rehabilitative intervention; (4) involving BCI intervention for the upper limb; and (5) written in English. No restrictions were imposed on study dates, follow-up duration, target electrodes, or participant characteristics to minimize publication bias. For each electronic database, the end date was April 2021. We excluded studies in which BCI therapy was applied in both the experimental and control groups, and studies that did not provide motor impairment assessment scores before and after the intervention (Supplementary Materials 3).
Data Extraction and Risk of Bias Assessment
A standardized data extraction form was used to search for study characteristics, patient characteristics, intervention and control details, and outcome data before and after the intervention. The Cochrane Collaboration risk of bias (RoB) 2.0 assessment was used to rate the quality of the included studies. 26 Assessment of risk of bias was conducted by 2 reviewers (NI and SH) using the following criteria: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) selective outcome assessment, (5) incomplete outcome data, and (6) selective outcome reporting, and other potential sources of bias.
Meta-Analysis Method
All data were combined using Review Manager (RevMan) 5.4.1, released by the Cochrane Library and RStudio (version 1.1.463). Two reviewers, who are content experts, assessed eligibility, according to the Cochrane Handbook, for each article. The retrieved articles were screened by reading the titles and abstracts. Potentially eligible studies were then analyzed in full. Studies providing motor performance scores in the upper limb before and after BCI training were considered for the meta-analysis. The same reviewers collected the data using a standardized data extraction form regarding authors, publication years, study design, subject population, feedback (ie, perceptual stimulus by visual or auditory stimulus, passive movement by robotic or orthotic devices, and NMES), decoding methods of brain activity, adverse events, and funding sources.
The intervention effect for each study was calculated as the standardized mean difference (SMD) of change in the outcome measure between the experimental and control groups. We used the mean change scores and SD of each included study for the current meta-analysis 8 (Supplementary Materials 4-1). When mean and SD values were not directly reported in the selected article, we contacted the corresponding author, or they were calculated from other available data. The SMD was calculated for paired samples based on Hedge’s equation with a correction for small studies (Supplementary Materials 4-2-3). For studies with multiple evaluation points, data from the available evaluation points immediately after the intervention were used. A forest plot was used to represent the results of the meta-analysis, similar to a previous study.7,8
Given the clinical heterogeneity of the selected articles, random-effects models were used. Heterogeneity in the intervention effect was inevitable because of the different study designs. Heterogeneity across studies was evaluated using Higgins’ 26 I 2 statistic and Q statistics, quantitative measure of inconsistency across studies (0%: homogeneity; 50%: moderate heterogeneity; 100%: heterogeneity) indicated the percentage of variance. The pooled estimate of the effect of the intervention and the associated 95% confidence interval (CI) were calculated using the DerSimonian–Laird method for the random-effects model using unadjusted means and measures of dispersion. 27 This approach is widely applied because of its relative simplicity, and further, it does not require the assumption of normality for the random effect. The methods of feedback combined with BCI (eg, passive movement, NMES, and perceptual stimulus), time since stroke (eg, subacute (<6 months since stroke onset) and chronic (>6 months), and training characteristics (training duration (long: >4 weeks, short: <4 weeks) or training time (long: >60 min/days, short: <60 min/days) were used as random-effects models for subgroup analyses. Moreover, the method for feature value detection from brain activity (eg, development of a classifier for judgment or sensorimotor rhythm) was examined in a subgroup analysis to help identify an effective method to analyze brain activity for BCI training. Random-effects meta-regression was conducted using the following parameters: (1) training sessions (number of training sessions during the experiment) and (2) training length (training time per day). These factors were chosen as moderators because of their potential association with estimating the effects of BCI training.
Two reviewers also assessed the possibility of publication bias by plotting the SMD against its precision (funnel plot asymmetry), measured as the standard error of the SMD. Subsequently, a funnel plot and Egger’s test 28 were performed (Supplementary Materials 4-4). Funnel plot asymmetry examines whether the association between estimated intervention effects and a measure of study size is greater than might be expected to occur by chance.
Results
Search Results
We obtained 655 articles from the database search and additional records, and after adjusting for duplicated articles, the titles and abstracts of 593 publications were screened. Of these, 51 articles were assessed for eligibility by full-text screening, and 35 articles were excluded based on the following exclusion criteria: (1) study design without control groups (n = 22; 43.1%), (2) outcome measure excluding motor performance in the upper limb (n = 3; 5.9%), (3) effect verification of the difference intervention as a main objective (n = 2; 3.9%), (4) study not including patients with stroke (n = 5, 9.8%), and (5) publication in a language other than English (n = 2; 3.9%). Ramos–Murguialday published 2 articles, one of which was a follow-up study involving the same patients in 2013
29
and 2019,
30
so we included it in the first article for this study. The remaining 16 articles were retained for qualitative synthesis. Finally, 16 articles fulfilled the inclusion criteria and were included in the meta-analysis, of which 12 involved RCTs. A flowchart depicting the selected studies is shown in Figure 1. The review process was confirmed using a checklist in the PRISMA statement for reviews (Supplementary Materials 1). Flow diagram.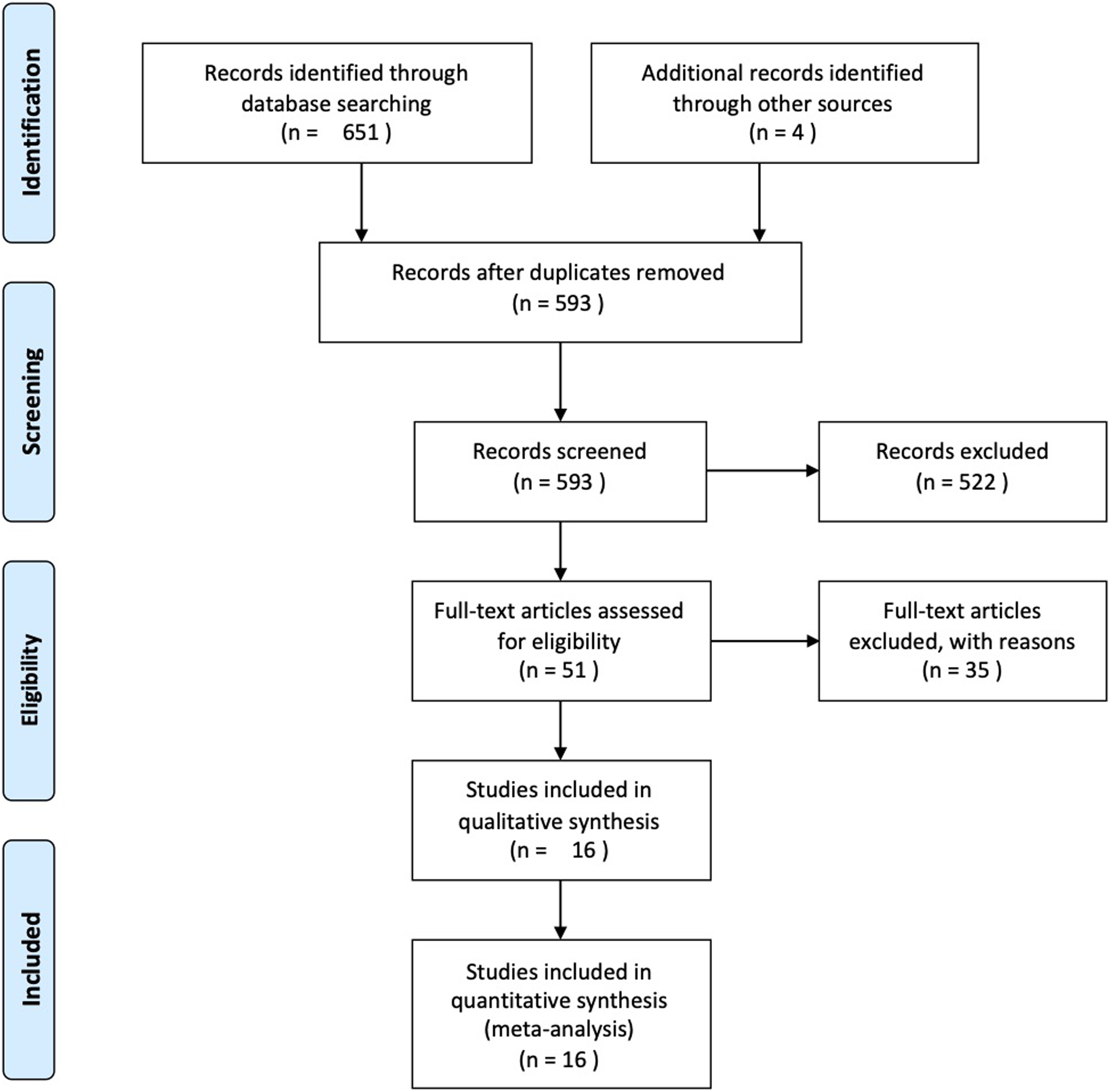
Characteristics of the Studies Included in the Meta-analysis
Data is reported as means (SD).
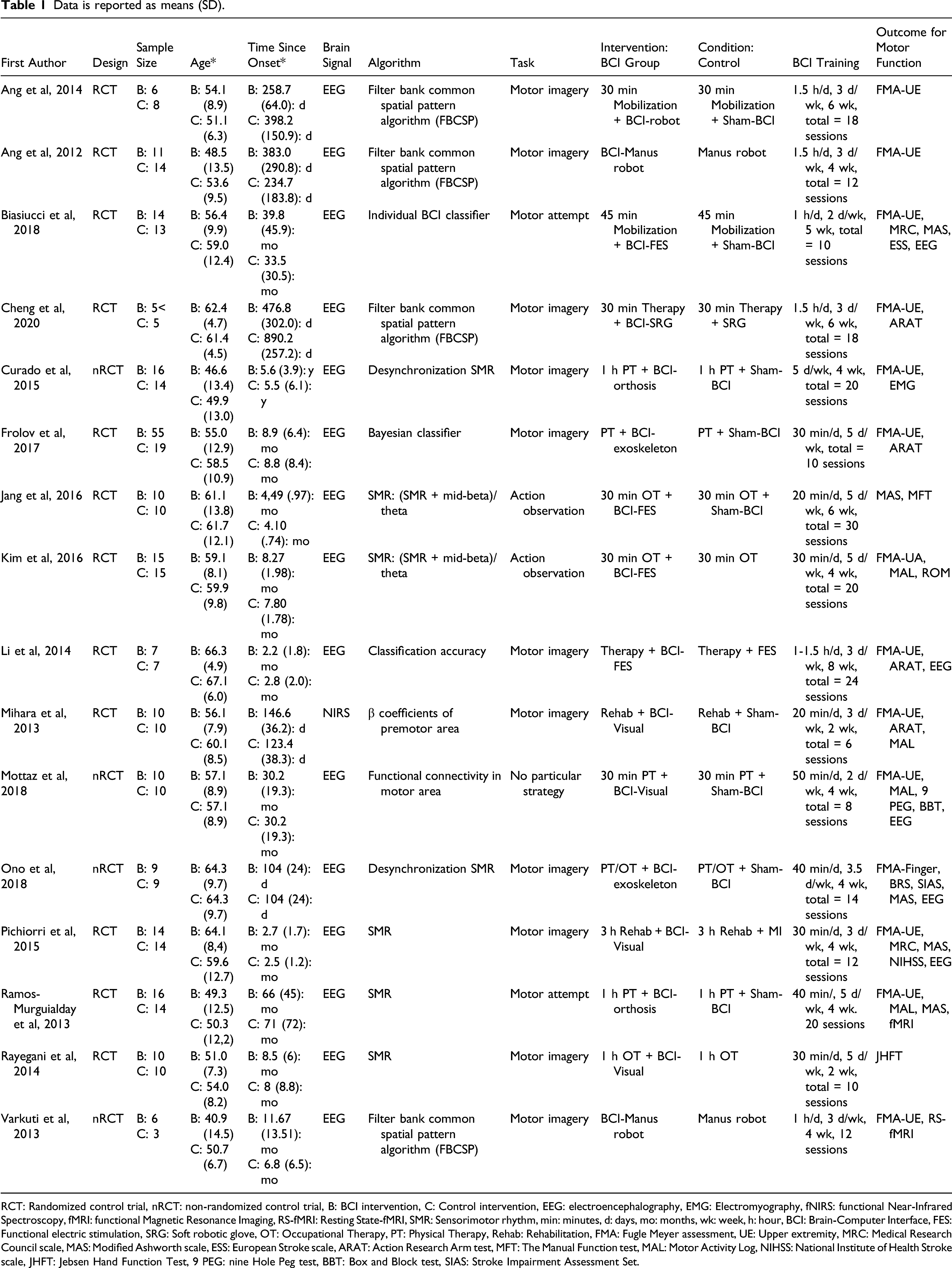
RCT: Randomized control trial, nRCT: non-randomized control trial, B: BCI intervention, C: Control intervention, EEG: electroencephalography, EMG: Electromyography, fNIRS: functional Near-Infrared Spectroscopy, fMRI: functional Magnetic Resonance Imaging, RS-fMRI: Resting State-fMRI, SMR: Sensorimotor rhythm, min: minutes, d: days, mo: months, wk: week, h: hour, BCI: Brain-Computer Interface, FES: Functional electric stimulation, SRG: Soft robotic glove, OT: Occupational Therapy, PT: Physical Therapy, Rehab: Rehabilitation, FMA: Fugle Meyer assessment, UE: Upper extremity, MRC: Medical Research Council scale, MAS: Modified Ashworth scale, ESS: European Stroke scale, ARAT: Action Research Arm test, MFT: The Manual Function test, MAL: Motor Activity Log, NIHSS: National Institute of Health Stroke scale, JHFT: Jebsen Hand Function Test, 9 PEG: nine Hole Peg test, BBT: Box and Block test, SIAS: Stroke Impairment Assessment Set.
Although most studies revealed no serious adverse events associated with BCI training for patients with stroke, several studies31,34 have reported mild discomfort after BCI training. Moreover, Ang reported that one subject in the BCI group discontinued due to a transient mild seizure. 32
Neurofeedback Protocols
The most common method of brain activity to control BCI systems is event-related desynchronization (ERD) related to motor imagery or voluntary intention of movement, and patients were encouraged to learn to upregulate ipsilesional SMR by reinforcing successful ERD.15,29,33,35,36,38,40,44 In contrast, 5 studies recorded whole brain activity using the developed filter systems.31,32,34,41,42 One study used a near-infrared spectroscopy (NIRS) system, which can measure task-related regional hemodynamic changes in levels of oxygenated and deoxygenated hemoglobin from the sensory-motor cortices. 37 The detected target signals were then used to trigger sensory or motor feedback provided by external devices. The duration of BCI training ranged from 2 to 8 weeks, and the total time (minutes) per week ranged from 40 to 200 min. In the control group, the nature differed across studies: 7 studies provided sham-feedback using random unrelated brain activity, 4 studies used conventional therapy, 3 studies used robot-assisted training, one study used NMES, and one study used motor imagery. Six different factors proposed by the Cochrane Organization were analyzed to assess the RoB in each study.
The Effects of BCI Training on Upper Limb Intervention
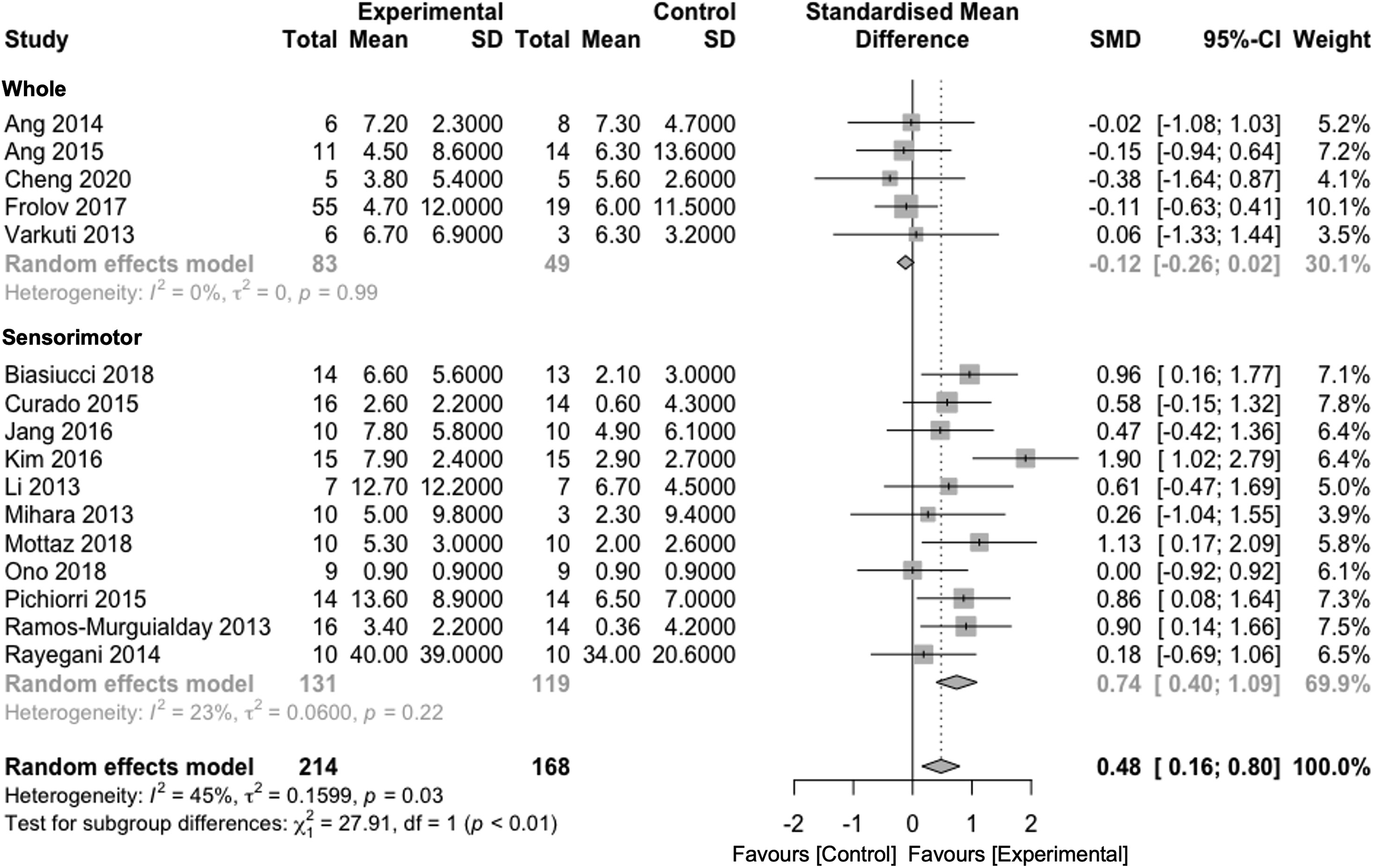
Individual studies and effect sizes for the selected studies are presented in Figure 2. Overall, BCI training (n = 214) achieved moderate beneficial effects on motor performance of the paretic upper limb (SMD = .48, 95% CI [.16-.80], P = .006). In 5 studies, the lower boundary for the 95% CI was above the no-effect vertical line. The weights of the studies ranged from 3.9% to 10.1%; the contributions of each study to the results are mostly comparable. Although the funnel plot appeared to be generally symmetrical (Figure 2), we observed an I
2
coefficient of 45%, indicating a moderate level of heterogeneity among the studies (P = .03). (A) Comparison of the effects of BCI and control training on the upper extremity in patients with stroke. Thirteen selected studies were pooled, and a standard mean difference (SMD) with 95% confidence intervals was computed as the overall effect of training. The results revealed that BCI training was significantly effective at improving motor recovery for the upper extremity (SMD = 0.48; 95% CI = 0.16-0.80, I2 = 45%, P = 0.03). B) The funnel plot representing publication bias showed a comparison of the effects between BCI and control training on motor recovery in patients with stroke. Egger’s regression test was negative (P = 0.765). Two dashed diagonal lines represent pseudo 95% confidential limits around the summary effect for each standard error on the vertical axis.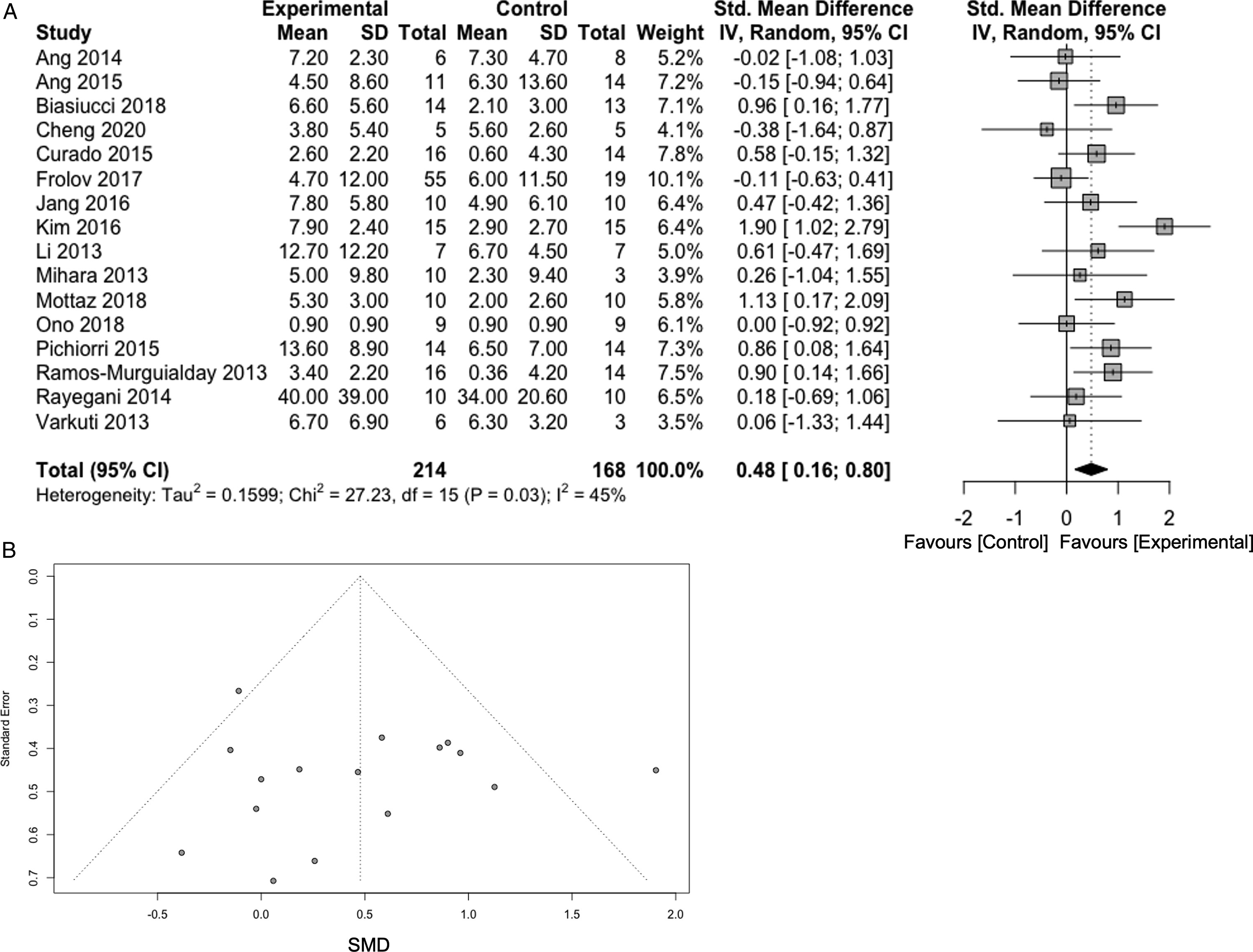
Two reviewers assessed the risk as low (“+”), high (“−”), or unclear (“?”) according to the Cochrane guidelines. A summary of the risk of bias for the 6 factors is presented in Supplementary Figure 1. Most studies used random generation sequences to select participants, but 3 studies showed some concerns regarding blinding in the randomization procedure and dealing with incomplete data.
Regarding publication bias, we observed no evidence of asymmetry in the distribution of study findings (Egger’s test, P = .765) and the effect size using a funnel plot. Further, 2 of the 16 studies overhang the region delimitated by the pseudo 95% CIs, indicating high heterogeneity between studies (Figure 2).
Subgroup analysis was conducted regarding the method of feedback on the following stimuli: (1) passive movement by robot or orthosis (n = 8), (2) NMES (n = 4), and (3) perceptual stimulus by visual/auditory sensation (n = 4). The NMES group (SMD = 1.01 [−.03-2.04]) and perceptual stimulus group (SMD = .66 [−.05-1.37]) tended to show superior clinical effects, compared with the passive movement group (SMD = .15 [−.21-.51]) with regard to the improvement in motor function (Figure 3). For the time since stroke onset, patients in the subacute phase achieved an SMD of .48 ([.05-.92]), and patients in the chronic phase had an SMD of .48 ([.02-.95]). However, no statistically significant differences were found between the BCI and control groups in the subgroup analysis (P = 1.00, Supplementary Figure 2). For the measurement of brain activity, SMR using electrodes over sensorimotor areas was commonly used, followed by specific developed filtering systems to classify brain activity using multiple electrodes. A subgroup analysis of the analysis methods indicated that simple BCI systems targeted with SMR activity, focused on SMR (SMD = .74 [.40-1.09]), had a significantly larger effect on motor function recovery, compared with classifier systems with a sophisticated filter (SMD = −.12 [−.26-.02]) (Figure 4). There was a significant difference among the groups (P < .01). In terms of training duration, the long-term group showed an SMD of .51 ([.15-.87]), whereas the short-term group achieved an SMD of .23 ([−.23-.64]). Subgroup analysis for training time produced an SMD of .25 ([−.32-.81]) in the long-time group, whereas SMD in the short-time group was .39 ([.11-1.11]). However, the training duration (P = .08, Supplementary Figure 3) and time (P = .47, Supplementary Figure 4) exhibited no statistically significant differences. The effects of different devices combined with BCI training in the subgroup analysis. Although BCI training combined with NMES and perception as the trigger stimulus tended toward a superior effect on motor recovery as compared to that of passive stimulus in patients with stroke, the effects of these feedback stimulations were not statistically significant. The effects of different measurement methods to detect brain signals in BCI training in the subgroup analysis. The results showed that the focusing on SMR from electrodes over sensorimotor areas provided a greater effect on motor recovery in patients with stroke, compared with the whole brain measurements (P < 0.01).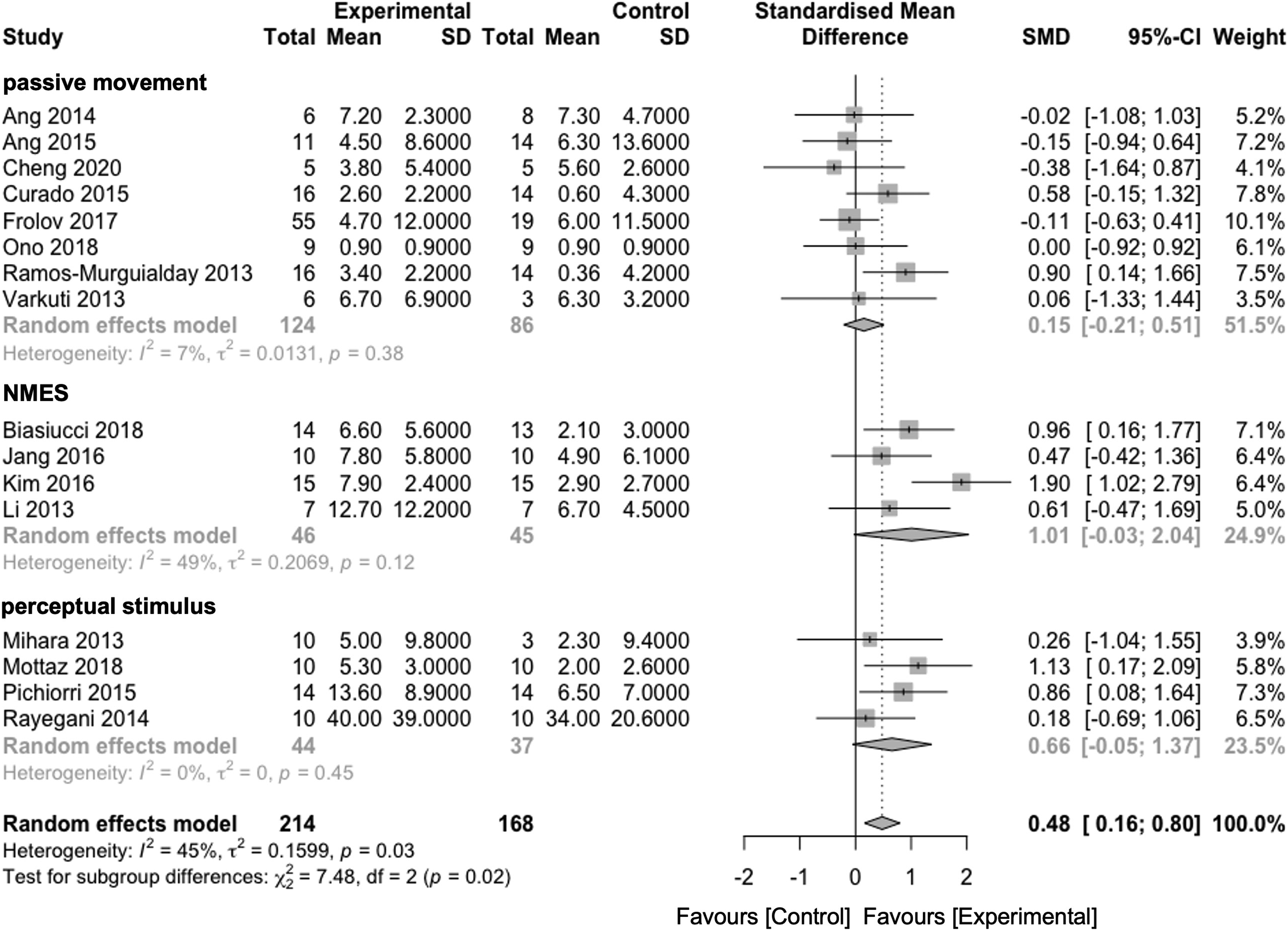
Meta-Regression
We performed a random-effects meta-regression analysis to evaluate the effect of 2 covariates (eg, training sessions and duration) on the effect size. However, there was no significant association between the training effects of the upper limb and training sessions (F1,14 = .034, P = .856) duration (F1,14 = .092, P = .766) (Figure 5). Meta-regression indicated the relationship between the effect size of SMD and the number of training sessions (A) and training length per week. (B) The size of each circle is inversely proportional to weight to correspond to a random-effects analysis. The straight line shows the regression line.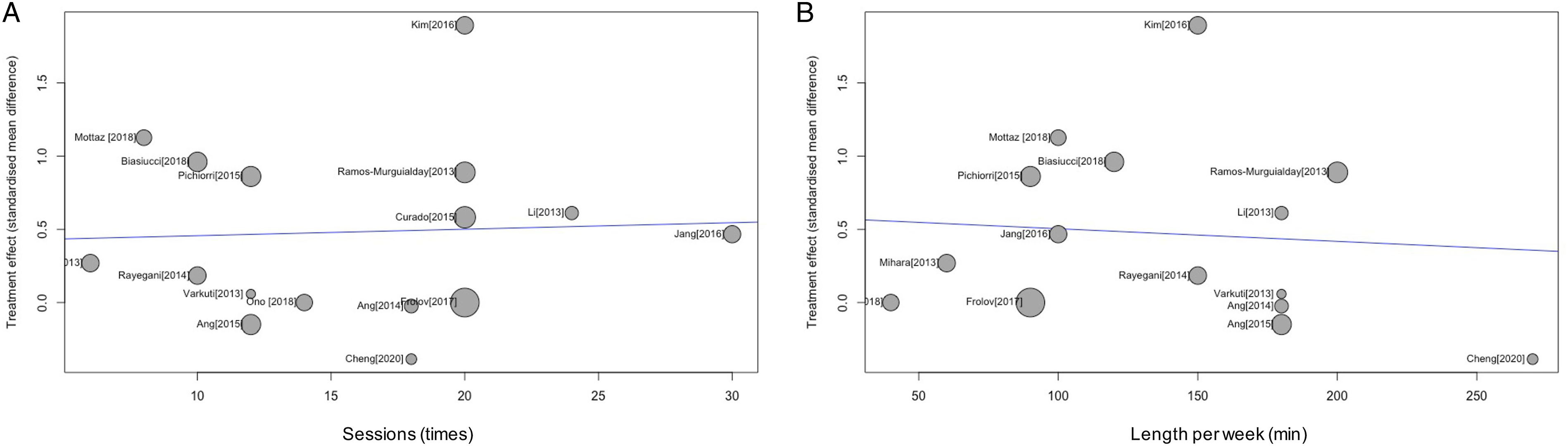
Discussion
The current meta-analysis summarizes the clinical effectiveness and effect size of the BCI training. BCI training has been recognized as a self-regulation method that leads to behavioral improvement due to the voluntary modulation of brain activity. The subgroup analyses indicated a higher intervention effect for NMES and perceptual stimulus than passive movement by robot or orthosis group, and a simple BCI algorithm by measurement of SMR over sensorimotor areas was more effective than a sophisticated classifier. Moreover, none of the included studies revealed severe adverse events after receiving BCI training, although symptoms of discomfort such as transient nausea, fatigue, and headaches were reported in a few studies.31,34
Improvement of Motor Performance Through BCI Training
This meta-analysis showed an SMD of .48 (95% CI: .16-.80) for all included studies. Other rehabilitative approaches for the paretic upper limb reported that an SMD of .34 was achieved for constraint-induced movement therapy (CIMT), 45 .49 for mirror therapy, 46 .59 for mental practice, 47 and .21 for robot rehabilitation. 48 These results showed that in a clinical setting, the effect of BCI training is comparable to or larger than that of other widely applied therapies. Furthermore, we found that 8 out of 14 studies assessed by FMA achieved improvements that exceeded the MCID, 49 demonstrating a clinically important difference in the BCI group. These results might fulfill the criterion of effectiveness of BCI training on motor recovery based on the modulation of brain activity.
Subgroup analysis of feedback methods revealed a higher SMD for NMES and perceptual stimulus than for passive movement by robotic devices or orthosis. Electrical stimulation is commonly used as a rehabilitative intervention to re-educate and facilitate voluntary contraction, and it has been suggested that electrical administration of somatosensory stimulation promotes recovery of motor function. 50 The possible mechanism of greater motor recovery by using NEMS as feedback could be explained by the plastic change in the sensorimotor cortex, such as unmasking of connections 51 and release of the neurotransmitter by electric stimulation. 52 Previous studies also reported that a sustained period of somatosensory electric stimulation exerts a modulatory effect on motor excitability, predominantly in the motor cortex. 51 Moreover, Biasiucci et al 33 have reported that motor function improves through learning, which progressively modifies the neural activity using contingent feedback and reward sharing by BCI intervention with electrical stimulation. BCI training provides a closed-loop pathway in which brain signals interact with external devices. Therefore, the sensorimotor interaction through sensory feedback induced by NEMS leading to the plastic change could be enhanced.
Several systematic review articles regarding neurofeedback systems have been published7,8,22,23; however, none of these studies presented an in-depth description and comparison of the various types of BCI-controlled methods adopted for stroke rehabilitation. Hence, subgroup analyses of methods to measure brain activity were comprehensively presented and compared in the present review and revealed an important finding. The effect size of the BCI algorithm using SMR over the sensorimotor area was significant, but that of classifier systems using a specific filter was not. Various signal processing methods have been used to control brain activity. The first clinically relevant method was the use of slow cortical potentials (SCP) to translate purposeful modulation for communication of locked-in patients. 53 Moreover, some have reported being able to achieve reliable control of robotic arms solely using brain activity after implantation of a sensor over the cortex in severely paralyzed patients.54,55 These devices have been developed as invasive or non-invasive assistive technologies to support patients with disabilities. Impressive demonstrations suggest that these devices, which use brain activity, could become a realistic option for improving the living conditions of patients. On the other hand, non-invasive rehabilitative strategies using BCI techniques have been developed, based on the theoretical concept that operant conditioning of neural activity can change behavior. Among several brain signals, SMR (8-15 Hz, also termed mu-rhythm or Rolandic alpha) 56 is the frequency band commonly used over the sensorimotor cortex for rehabilitative neurofeedback training. SMR is inversely related to sensorimotor cortex activity and is generated by a thalamo-cortical network 57 ; it displays no specific contingency to actual motor output. This subgroup analysis indicated that the BCI algorithm using SMR was a better method to enhance motor recovery through self-regulation of brain activity compared to the classifier for whole brain activity using sophisticated filters. Therefore, SMR appears to be an ideal candidate for brain signals in non-invasive BCI training for stroke neurorehabilitation. 58 On the other hand, the BCI algorithm that makes individual classifiers tended to provide less motor improvement. Ang reported the necessity to perform subject-specific calibration as pre-processing using their developed filtering system. Therefore, this may require considerable setup-time from patient-to-patient and complicated control for their brain activity.31,59 Fatigue is a common symptom in patients with stroke (ie, central fatigue) and is a widespread issue for patients with stroke because of debilitating complications. 60 The psychological state influences the attainment of a high level of self-regulation because BCI training requires concentration on the instructions and feedback. Indeed, Frolov reported that the majority of patients reported fatigue after 20-30 min of training. 34 Therefore, the management of subjective fatigue during BCI training may be crucial to improve motor function in patients with stroke. Nevertheless, the meta-regression revealed that training sessions and training length were not significant predictors of effect size. These might show the importance of concentrated training in controlling brain activity in each session while avoiding fatigue despite the number of weeks or duration of intervention. Patients with stroke might generally have difficulty controlling the devices; thus, simple BCI systems (algorithm, device, and introduction) may be appropriate to improve their motor function. On the other hand, one of the advantages of non-invasive techniques is that they allow the feedback of neural activation to be measured over an entire network of distributed brain regions that are involved in a specific function from a spatiotemporal pattern of brain activation. 61 Therefore, the ability to modulate neural dynamics at the network level using BCI may be a more effective method of neural regulation; it focuses on re-establishing connectivity in damaged neural areas by BCI training.8,23
Underlying Neural Mechanisms of BCI Training
To understand the neural mechanisms underlying motor recovery induced by BCI training, we need to elucidate the general theoretical and experimental bases of learning. Learning in operant conditioning states that the appearance of target brain activity is paired with an external stimulus and proceeds when correct responses are reinforced by contingent feedback or reward. 62 Buch reported that sensory feedback contingent upon ipsilesional SMR activity in BCI systems based on operant conditioning volitionally led to modulation of the cortical activity pattern, 63 thereby suggesting that BCI training might strengthen the ipsilesional sensorimotor loop and foster neuroplasticity that facilitates motor recovery by re-establishing contingency. The neurophysiological association between operant learning and synaptic changes through BCI systems could strengthen the targeted sensorimotor loop and lead to neuroplasticity that facilitates motor recovery. 64 Moreover, Hebbian plasticity driving functional connectivity following the simultaneous activation of pre- and postsynaptic neurons is also hypothesized to be the basis of neural plasticity in BCI training.58,61 This coincident activation of the motor cortex and sensory feedback loops might reinforce previously dormant cortical connections by Hebbian plasticity, thus supporting functional recovery.9,65
Other suggested potential mechanisms involve rebalancing of the interhemispheric activity shift from the contralesional hemisphere toward the ipsilesional hemisphere.29,31 A longitudinal case study reported the correlation between functional motor improvements and increased activation in the ipsilesional hemisphere. 66 Neurofeedback training for healthy individuals also enhanced the laterality of event-related sensorimotor oscillations. 67 In contrast, a follow-up study reported that laterality changes between hemispheres before and after intervention did not correlate with motor recovery, indicating that activity changes observed during the intervention may represent motor learning rather than motor recovery. 30 A previous review demonstrated the importance of personalized interventions through a neurotechnology-aided upper limb 9 ; clarification of the mechanisms of single-BMI intervention is required to provide adequate evidence for the long and complex process of stroke rehabilitation. Although these might show that BCI closed-loop systems could focus on gaining motor recovery by facilitating greater physiological recruitment of the stroke-affected hemisphere, the exact mechanisms of recovery and the factors influencing rehabilitation success using BCI are yet to be fully elucidated.
Potential Clinical Applications of Neurofeedback Systems
EEG-mediated BCI training is commonly based on brain rhythms and the ratio of a specific frequency over the ipsilesional sensorimotor cortex. Another method for neurofeedback, coherence of neural activity between brain regions, has been implicated as a new neurofeedback approach for self-regulation, and a recent review has focused on the effectiveness of this approach for improving the link between the brain and the paralyzed muscle. 22 It has been reported that the BCI approach using coherence could also enhance connectivity in a target frequency band between the motor cortex and the rest of the brain. 68 Moreover, use of cortico-muscular coherence (CMC) calculating the amount of synchronization between cortical and muscle activity69,70 has been attempted to facilitate functional connectivity. 71 Movement control involves the interaction between the central nervous system and the periphery, and enhanced coupling is believed to facilitate large-scale interaction in the nervous system. 72 Synchronization between the oscillatory activities of the sensorimotor cortex and the spinal motor neuron pool has been the focus of research in human motor control.73,74 Beta-band oscillatory synchronization is considered an indicator of neural functional connectivity for the control of movement. CMC-mediated neurofeedback training in healthy patients was shown to be able to voluntarily modify CMC strength in the target frequency range. 71 However, it is known that ∼15-30% of participants are unable to learn volitional control of brain activity using different neuronal processes. 75 Although visualization of neural coupling could provide useful information for performance gain, further detailed research to uncover the neural mechanisms of neural coupling is required for clinical application.
Conclusions
The current review demonstrated the effectiveness of BCI training combined with conventional therapy on the upper limb in patients with stroke. Although heterogeneity was revealed in the participants and the interventions conducted in the included studies, adding BCI training might be potentially beneficial in improving motor outcome measures. Regarding the feedback method, the NMES and perceptual stimulus might be a useful stimulus triggered by BCIs for functional recovery. Simple methods that enable recording over sensorimotor areas may have a significant, positive effect on motor recovery and serve as effective methods to detect the feature value based on brain activity.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211062895 – Supplemental Material for Brain–Computer Interface Training Based on Brain Activity Can Induce Motor Recovery in Patients With Stroke: A Meta-Analysis
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211062895 for Brain–Computer Interface Training Based on Brain Activity Can Induce Motor Recovery in Patients With Stroke: A Meta-Analysis by Ippei Nojima, Hisato Sugata, Hiroki Takeuchi and Tatsuya Mima in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-2-nnr-10.1177_15459683211062895 – Supplemental Material for Brain–Computer Interface Training Based on Brain Activity Can Induce Motor Recovery in Patients With Stroke: A Meta-Analysis
Supplemental Material, sj-pdf-2-nnr-10.1177_15459683211062895 for Brain–Computer Interface Training Based on Brain Activity Can Induce Motor Recovery in Patients With Stroke: A Meta-Analysis by Ippei Nojima, Hisato Sugata, Hiroki Takeuchi and Tatsuya Mima in Neurorehabilitation and Neural Repair
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Scientific Research (B) 21H03304 (to HS), Grant-in-Aid for Scientific Research (C) 20K09448 (to HT), Grant-in-Aid for Scientific Research (A) 19H01091, Grant-in-Aid for Challenging Research (Exploratory) 21K19745 (to TM).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
