Abstract
Stroke is a major cause of functional limitation in older adults. Although patients with stroke improve in functional ability through spontaneous recovery and rehabilitation, recovery in most patients plateaus approximately 6 months after onset.1,2 Studies have shown that approximately 35% of patients with chronic stroke require assistance in performing daily activities.3-5 Patients with stroke in a community-living setting reported dissatisfaction with functional recovery 1 year after onset because of hindered participation in life events.5-7 Although 60% of community-living patients with stroke can visit places of interest (eg, shopping malls) after being discharged from hospital, approximately one-third cannot leave home unattended. 8
Because of high costs, many patients with stroke often depend on outpatient care for rehabilitation after being discharged from hospitals. 9 However, compliance with rehabilitation programs is frequently compromised because of financial strain or logistical difficulties or for social and psychological reasons. 10 Thus, home-based programs have emerged as an attractive alternative for stroke rehabilitation. Numerous studies have shown that home-based rehabilitation programs can improve the mobility and functional performance of patients with acute or subacute stroke and reduce health care costs.11-15 Anderson et al 11 observed that early hospital discharge, coupled with a home-based rehabilitation scheme, was less costly than conventional hospital care. Widen Holmqvist et al 12 found that early supported discharge with continually provided home-based rehabilitation services for patients with stroke in the first 3 months after an acute stroke was no less beneficial than routine rehabilitation. Home-based rehabilitation programs were observed to reduce initial hospitalization significantly and had no adverse effect on mortality or the number of falls. These studies suggest that home-based rehabilitation programs are a viable option in certain health care systems. 13
However, the cost of services provided by a home-based rehabilitation team, even a physical therapist, might still be prohibitive for many patients with stroke or their families. 9 Thus, additional cost-effective and flexible rehabilitation models for patients with chronic stoke need to be developed.
Patients with chronic stroke rely on caregivers to perform daily activities.14,15 Current clinical practice guidelines recommend that family caregivers of stroke patients become active members of their rehabilitation teams by participating in goal setting and decision making to facilitate functional recovery and community reintegration.16,17 Family members are often the closest persons to, and the most vital environmental component for, patients with stroke.18,19 Family-mediated exercise therapy has been shown to be effective in improving the functional recovery of patients with acute stroke, 20 and a family-mediated and home-based program supervised once a week by a physical therapist was as effective as outpatient or day hospital therapy. 21 Caregiver training has been demonstrated to be cost-effective in improving the physical functioning of patients with stroke in rehabilitation settings. 22 The International Classification of Functioning, Disability, and Health (ICF) conceptual framework of the World Health Organization was recently applied to a rehabilitation strategy.23,24 It asserts that functioning encompasses “body functions and structures and activities and participation” and is viewed in relation to the health condition as well as personal factors (patients with stroke) and environmental factors (family members and life situation). The model enables patients with stroke conditions to achieve and maintain optimal functioning in interacting at home and in a community environment. However, to our knowledge, the effectiveness of a caregiver-mediated, home-based intervention (CHI) based on the ICF conceptual framework has not been examined in patients with chronic stroke in community-living settings. Hence, this study was undertaken to examine whether CHI based on the ICF conceptual framework was effective in improving the physical functioning of patients with chronic stroke.
Methods
Study Design and Participant Recruiting
A 2-arm, single-blind (the evaluator), randomized controlled trial was conducted (Figure 1). Participants were recruited from the rehabilitation and neurology departments of 3 teaching hospitals in Southern Taiwan. Patients were qualified to participate in this study if they (1) experienced a single ischemic or hemorrhagic stroke in the cerebral hemisphere, as determined through computed tomography or magnetic resonance imaging; (2) were > 6 months postonset; (3) exhibited mild to moderate disability (Brunnstrom recovery stages III-V); (4) were undergoing rehabilitation activities 2 or fewer times per week; (5) were home dwelling; (6) had family members, friends, or paid workers as caregivers; and (7) still required assistance to accomplish everyday activities. Patients were excluded if they required use of a nasogastric feeding, urine, or tracheal tube or exhibited 1 of the following conditions: recurring stroke, dementia (based on medical records), global or receptive aphasia, severe orthopedic disability, or an unstable medical condition (eg, severe acute myocardial infarction). We defined the caregiver as a person who was most responsible for patient’s daily care and who lived with the patient. Caregivers were excluded if they were in poor physical health, had mental or behavioral disorders (eg, alcohol abuse, severe orthopedic disability, uncontrolled diabetes, and hypertension), or were unable to provide to the patient at least two 60- to 90-minute sessions of rehabilitation training per week.
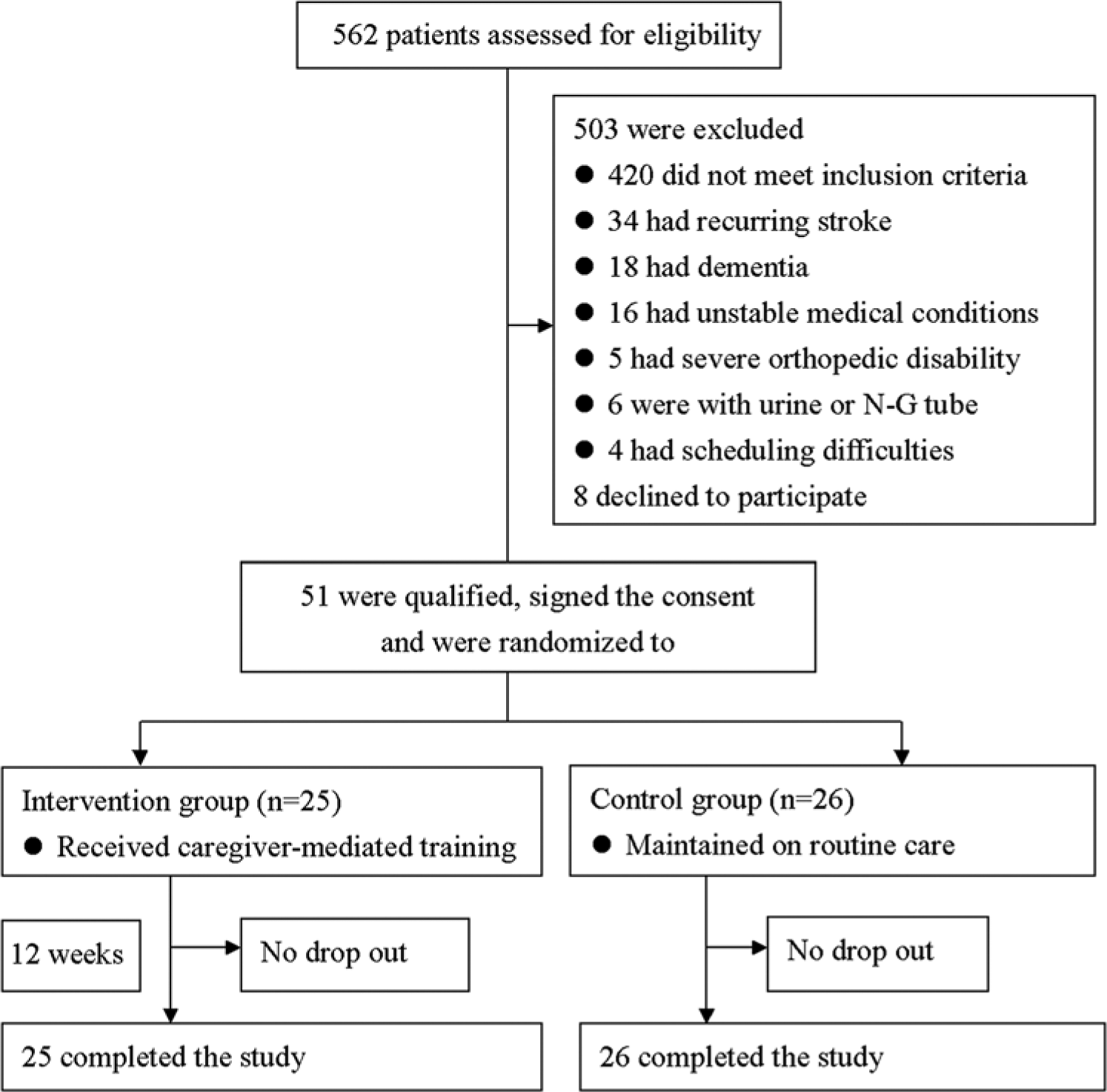
Study design and sample flowchart.
Prospective participants were presented an oral and written explanation of the objective and procedure of the study in plain language. Those who agreed to participate were asked to sign an informed consent form and were entered into the study on a rolling basis. The Asia University Institutional Review Board approved the study protocol (No. 09901002), and the study was conducted according to ethical standards set forth in the Helsinki Declaration.
Randomization
Each approved patient was asked to draw a folded piece of paper marked with a computer-generated random number from a bag. Patients were assigned to groups per the number drawn. The trial was initiated immediately following group assignment.
Intervention
A physical therapist outlined a personalized weekly training schedule for each patient according to the CHI program (Figure 2). The program was roughly divided into 3 phases: phase 1 (weeks 1-4), to improve patients’ body functions and structural components; phase 2 (weeks 5-8), to improve patients’ ability to undertake everyday activities within their living environments using task-specific restorative and compensatory training methods; and phase 3 (weeks 9-12), to help the patients reintegrate into the society by participating in restorative outdoor leisure activities (Figure 2).
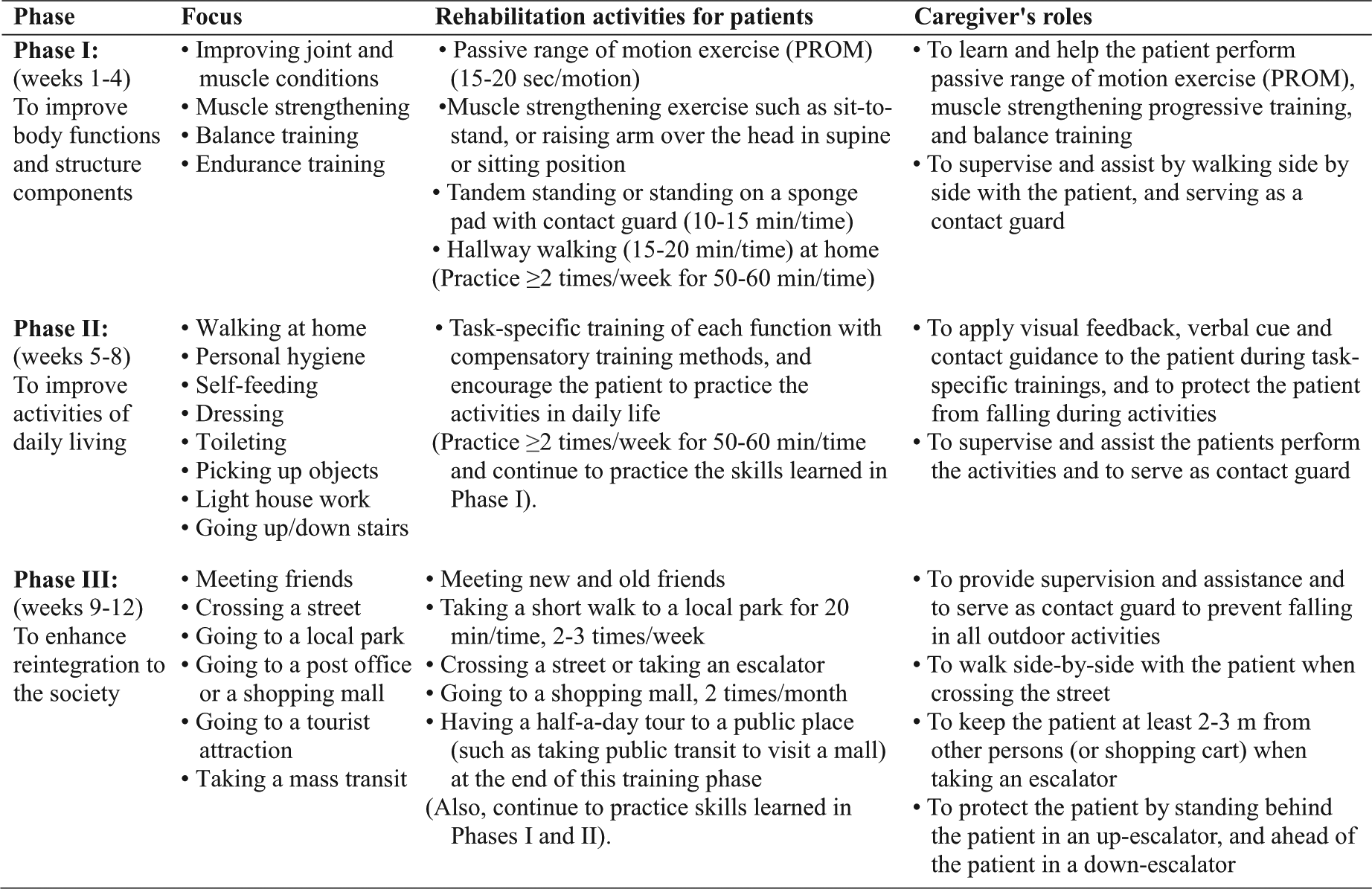
The 12-week caregiver-mediated, home-based intervention program for patients with chronic stroke designed according to the International Classification of Functioning, Disability, and Health conceptual framework.
A physical therapist visited each patient in the intervention group once weekly for approximately 90 minutes to teach patients personalized rehabilitation skills and to teach the caregivers the skills necessary to assist the patients in performing the planned tasks during trial period. In each visit, the physical therapist explained and demonstrated the tasks to the patient and the caregiver and requested that they practice the tasks to ensure correctness. The physical therapist wrote individualized training guidelines and simple illustrations for the caregiver and requested him or her to record the frequency of training and tasks completed each week. The caregiver was asked to encourage (and help, if necessary) the patient to perform the planned activities at least twice weekly and, if possible, every day. During each follow-up visit, the physical therapist examined the activities practiced, the frequency of practice, and the overall progress of the patient during the past week. The therapist also inquired about any experience of adverse events and planned new weekly activities accordingly.
Patients in the control group maintained their everyday routines but also received weekly visits or telephone calls by the therapist to talk about their rehabilitation progress, daily activities, and general health conditions. However, patients in the control group were not given specific instructions or guidance related to rehabilitation skills.
Outcome Measures
At baseline (after randomization) and the endpoint of the trial, all participants were evaluated with the Berg Balance Scale, the 10-Meter Walk Test, and the 6-Minute Walk Test (6MWT) in a hospital setting and the Stroke Impact Scale (SIS) and the Barthel Index at participants’ homes. All caregivers were evaluated for burden with the Caregiver Burden Scale. The SIS used in this study was a stroke-specific outcome measurement tool developed by Duncan et al. 25 The scale has been widely used in numerous clinical rehabilitation studies.26-28 The current version (SIS 3.0) 29 is a revised version of the original SIS, 25 and its reliability and validity have been established. 30 The SIS 3.0 contains 59 items measuring 8 domains: strength, hand function, activities of daily living/instrumental activities of daily living (ADL/IADL), mobility, communication, emotion, memory/thinking, and social participation. Factor analysis of the SIS revealed that 4 domains (strength, hand function, mobility, and ADL/IADL) could be combined to create a composite physical dimension score. The other domains (emotion, communication, memory, and social participation) were scored individually, and a single item was used to assess the perceived general recovery of patients with stroke. Each item was assessed with a 5-point scale (5, not difficult at all; 1, extremely difficult or cannot do at all). A summed score was generated for each domain (score range, 0-100), and a high total score indicated satisfactory functional recovery.25,29,30
The Berg Balance Scale evaluated the patients’ balance control based on 14 common tasks performed in everyday life. Each task was rated 0 to 4 points, with a maximal score of 56. The scale was determined to provide a valid and reliable measurement of balance control ability. 31 The original 0- to 100-point Barthel Index was used to measure performance in daily activities. A high score suggested a greater ability to live independently at home. 32 For the 10-Meter Walk Test, the free (habitual) and maximal walking velocities of each patient were measured 3 times on 2 occasions in a 10-m corridor; mean speeds on each occasion were analyzed. 33 The 6MWT was used to measure functional walking capacity. Each patient was asked to walk along a 25-m corridor for 6 minutes, and the total distance walked was recorded. This measure was found to be a reliable submaximal test of the cardiovascular fitness of patients with stroke. 34
In addition, the Caregiver Burden Scale 35 assessed the subjective burden of each caregiver. The scale consists of 22 items covering caregivers’ general strain, isolation, disappointment, emotional involvement, and environmental aspects. Each item was scored on a scale of 1 to 4 (1, not at all; 4, often). Scores ranged from 22 (no burden) to 88 (high burden). All outcome measurements were evaluated by an independent physical therapist who was blinded from knowing the treatment assignment.
Sample Size Calculation
Following Rosner, 36 we estimated that a sample size between 16 and 36 patients per group would be necessary by assuming an improvement of 10 points (SD = 10-15 points) 25 on the physical domains of the SIS, an effect size of 0.67 to 1.0, and to achieve 80% power at α = .05.
Statistical Analysis
All statistical analyses were performed with SPSS 19.0 (IBM Corp, Chicago, IL). Simple statistics were used for descriptive data. The Mann-Whitney
Results
This randomized controlled trial was conducted from October 2011 to January 2013. For this trial, the 3 hospitals referred to us 562 patients who had had a stroke. Among them, 59 met the inclusion criteria, and 51 agreed to participate in this study. Figure 1 depicts the flowchart of the study sample and the reasons for disqualifications. All participants completed the 12-week trial, and none reported falling or having any other major adverse events. Table 1 shows the characteristics of participants. Overall, 58% (30 of 51) were men; 47% (24 of 51) had right hemiplegia; and 75% (38 of 51) lived in homes equipped with mobility barriers, such as raised doorsteps between rooms. The majority of the caregivers (71%, 36 of 51) were the patients’ spouses.
Participant Characteristics.
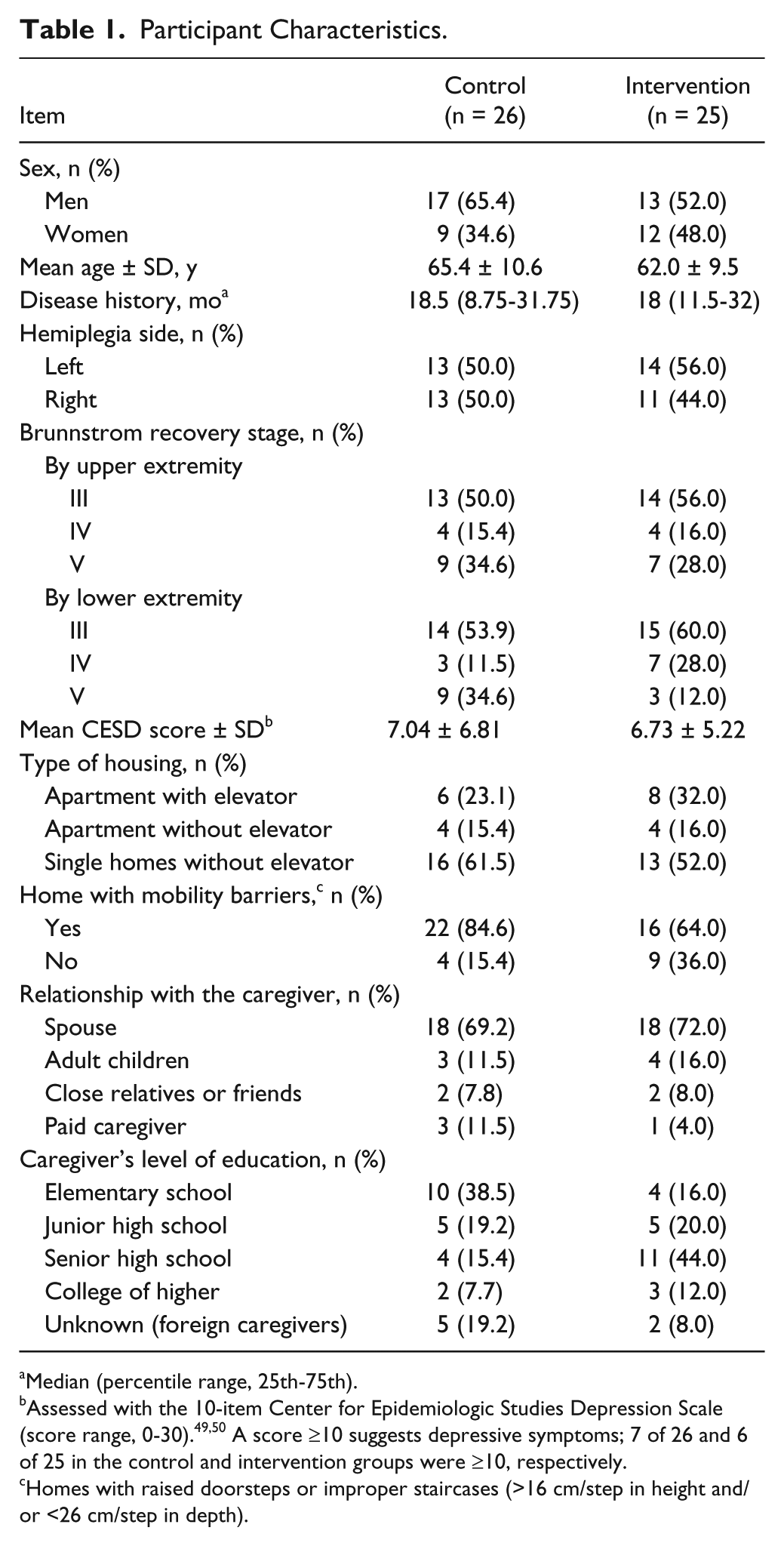
Median (percentile range, 25th-75th).
Assessed with the 10-item Center for Epidemiologic Studies Depression Scale (score range, 0-30).49,50 A score ≥10 suggests depressive symptoms; 7 of 26 and 6 of 25 in the control and intervention groups were ≥10, respectively.
Homes with raised doorsteps or improper staircases (>16 cm/step in height and/or <26 cm/step in depth).
The effects of CHI on SIS domains are shown in Table 2. CHI significantly improved the following domain scores: strength (net change of control group vs intervention group, respectively: 1.4 vs 15.5;
Effects of a 12-Week Caregiver-Mediated, Home-Based Intervention on Stroke Impact Scale Domain Scores in Patients With Chronic Stroke.
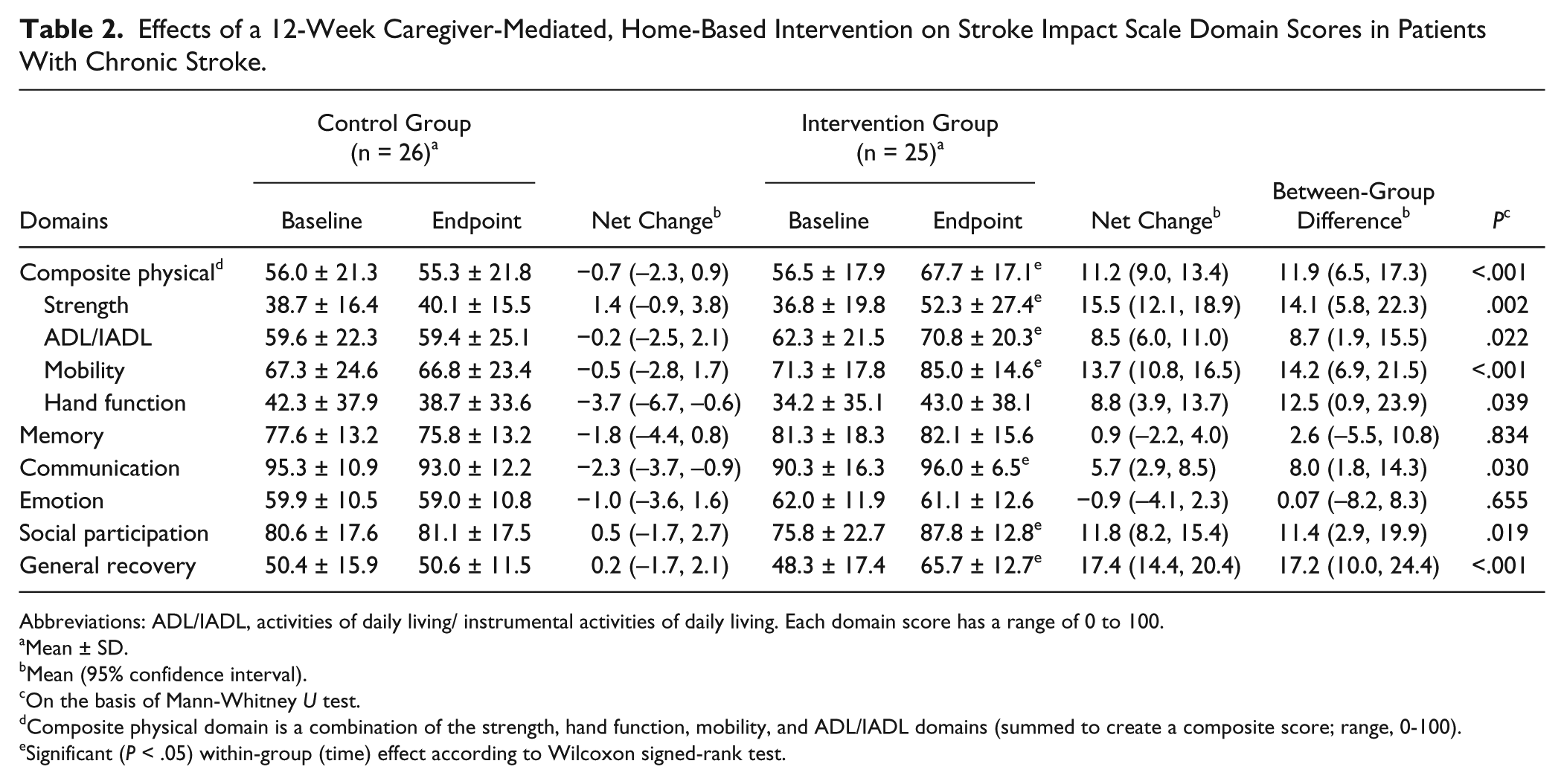
Abbreviations: ADL/IADL, activities of daily living/ instrumental activities of daily living. Each domain score has a range of 0 to 100.
Mean ± SD.
Mean (95% confidence interval).
On the basis of Mann-Whitney
Composite physical domain is a combination of the strength, hand function, mobility, and ADL/IADL domains (summed to create a composite score; range, 0-100).
Significant (
Table 3 shows the effects of CHI on walking velocity, 6MWT, Berg Balance Scale, and Barthel Index. CHI significantly improved the following scores: free-walking velocity (–1.4 vs 7.5 cm/s), 6MWT (–10.5 vs 15.8 m), Berg Balance Scale (–0.8 vs 4.5), and Barthel Index (0.6 vs 7.2) (all
Effects of a 12-Week Caregiver-Mediated, Home-Based Intervention on Walking Velocity, 6MWT, Berg Balance Scale, and Barthel Index in Patients With Chronic Stroke.
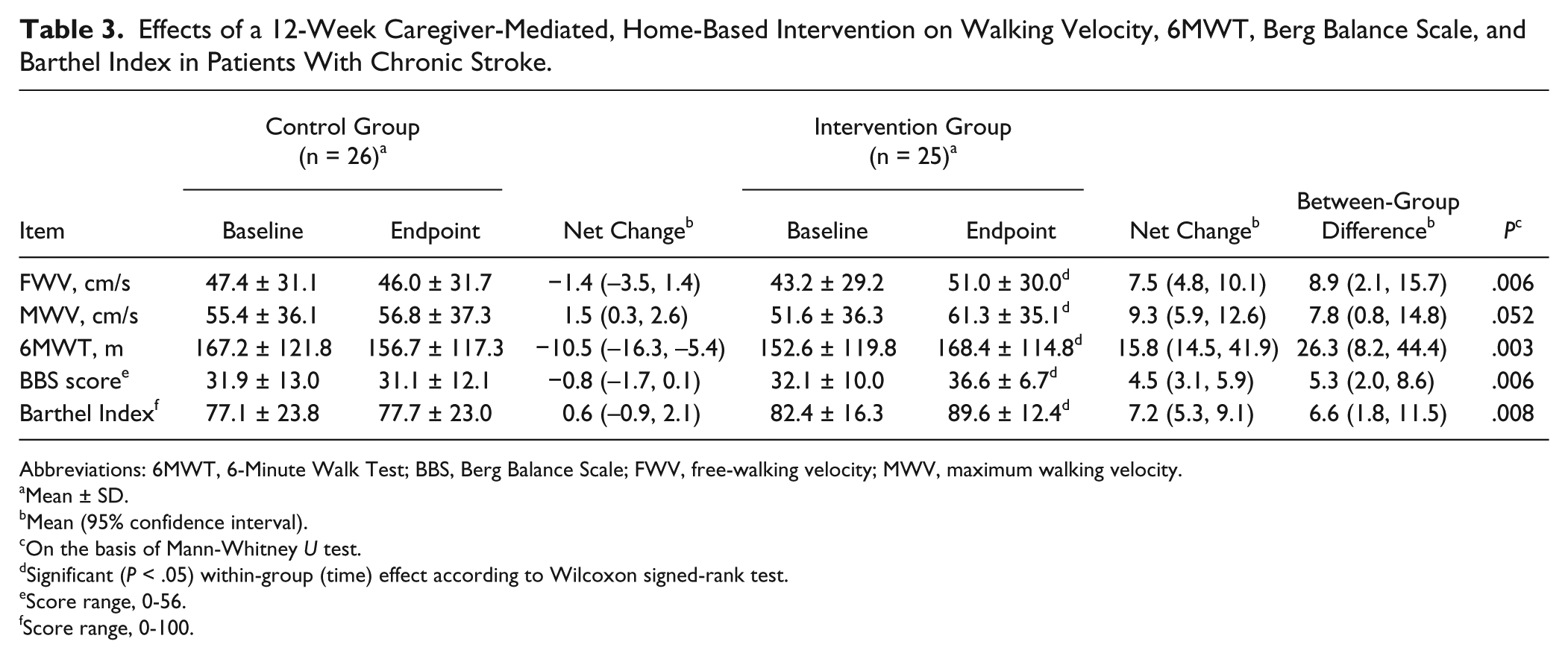
Abbreviations: 6MWT, 6-Minute Walk Test; BBS, Berg Balance Scale; FWV, free-walking velocity; MWV, maximum walking velocity.
Mean ± SD.
Mean (95% confidence interval).
On the basis of Mann-Whitney
Significant (
Score range, 0-56.
Score range, 0-100.
Effects of a 12-Week Caregiver-Mediated, Home-Based Intervention on Caregiver Burden Scores in the Major Caregivers of Patients With Chronic Stroke.
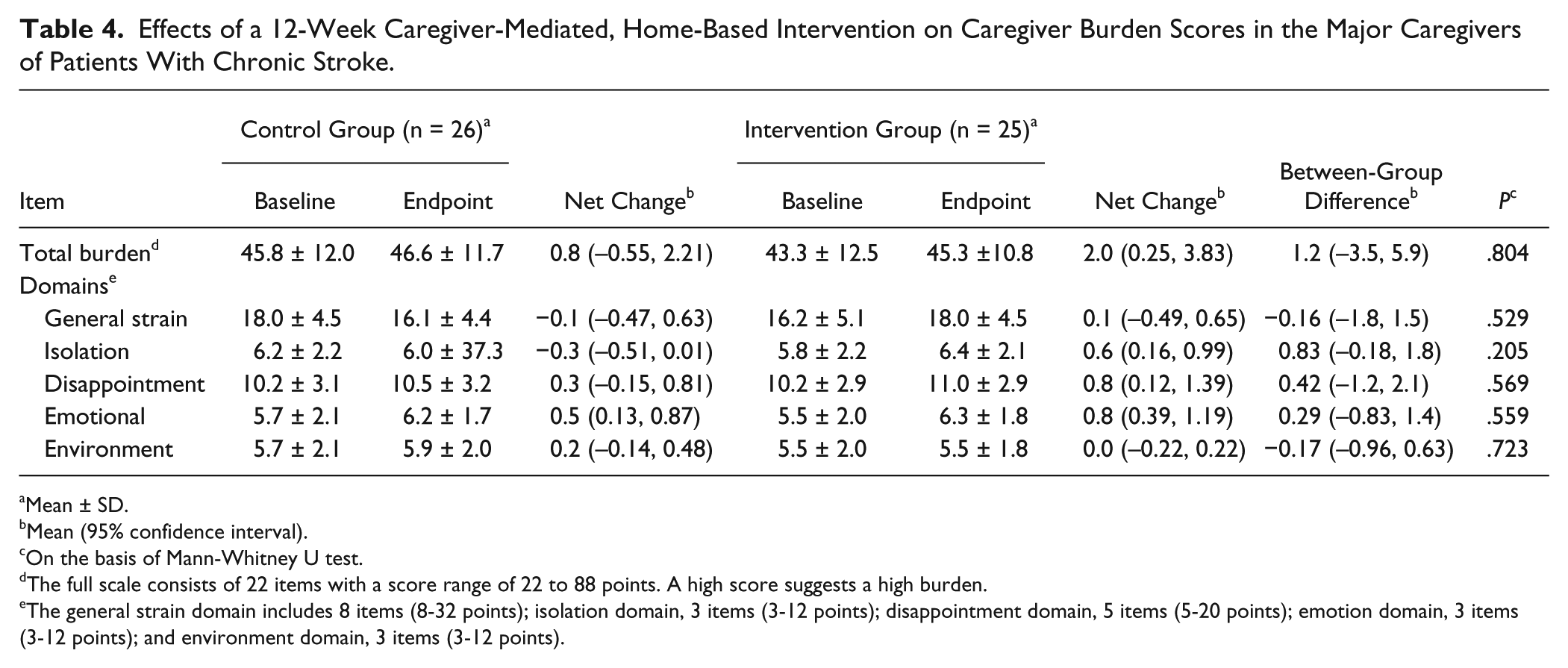
Mean ± SD.
Mean (95% confidence interval).
On the basis of Mann-Whitney U test.
The full scale consists of 22 items with a score range of 22 to 88 points. A high score suggests a high burden.
The general strain domain includes 8 items (8-32 points); isolation domain, 3 items (3-12 points); disappointment domain, 5 items (5-20 points); emotion domain, 3 items (3-12 points); and environment domain, 3 items (3-12 points).
Discussion
Results of this study suggest that CHI can be an effective rehabilitation strategy for patients with chronic stroke. The 12-week CHI resulted in significant improvements in the composite physical ability, general recovery, and physical functioning of patients with chronic stroke. CHI increased walking velocity, 6MWT, Berg Balance Scale, and Barthel Index. These results suggest that CHI can improve physical functional recovery in patients with stroke beyond the first 6 months after onset. Significant improvements were even observed in individuals with a 30-month-long history of stroke.
Functional Recovery of People With Chronic Stroke
CHI improved most SIS domains by more than 10 points (range, 8.5-17.4), except the memory and emotion domains. Lin et al observed that SIS scores that showed clinically important differences in strength, ADL/IADL, mobility, and hand function domains were at least 9.2, 5.9, 4.5, and 17.8 points, respectively. 37 Duncan et al also suggested that the clinically meaningful changes in each SIS domain fall in the range of 10 to 15 points. 25 Thus, the score improvements observed in the present study suggest significant improvements in physical functioning in the respective domains.
CHI improved free-walking velocity from 43.2 ± 29.2 to 51.0 ± 30.0 cm/s. Such an improvement is likely to increase the confidence and ability of many patients to walk across most city streets. Most city streets in Taiwan are 6-15 m wide, and most traffic light systems set a 30-second period for pedestrian crossing. Thus, CHI is likely to have increased patients’ confidence to participate in community activities and reintegrate with the society.
We also observed a slight time-related decline in 6MWT in the control group, suggesting that certain patients might have stopped routine therapy. Previous studies have reported that patients with stroke who do not receive appropriate rehabilitation or care support generally exhibit a considerable decline in physical functioning.38,39 These results suggest that patients with chronic stroke need to include continual physical recovery training in their formal/routine rehabilitation programs.
Advantages of Home-Based Rehabilitation
Previous studies have demonstrated that patients with chronic stroke can achieve and maintain physical functional improvement through outpatient or home-based rehabilitation after hospital discharge. 17 The home environment drastically differs from the hospital environment. The hospital environment has minimal barriers and readily available professionals and therapies to protect patients with stroke from injuries and to provide assistance when needed. 40 Yet, the home environment is typically filled with obstacles and barriers, 11 particularly in Taiwan, where rooms in traditional homes are divided by raised doorsteps approximately 10 to 20 cm high. Thus, once returning home, most patients lack the ability and support to manage this “new” environment, 41 and the caregiver, most often a spouse or close relative, is generally not well trained to solve these problems. Thus, patients’ mobility is often hindered by their physical impairments and inability to manage barriers in the home.42,43 In this study, repeating daily activities in the home environment seemed to have resulted in marked improvements in patients’ mobility, balance, and self-care ability.
Benefits of Caregiver-Mediated Rehabilitation
Family-mediated rehabilitation programs have been determined to enhance the functional recovery of patients with stroke and reduce the care burden on their caregivers.20-22 Traditionally, most caregiver training and education programs for patients with acute or subacute stroke are conducted in hospital or rehabilitation settings.20-22,44 Most of these programs instruct participants about stroke-related problems, skills for assisting the patient in everyday activities, how to play a supervising role, and how to prevent disease complications.21,22,44 Galvin et al 20 determined that family-mediated exercise intervention improved walking ability and integration into the community and decreased caregiver strain in patients with acute stroke. In the present study, a CHI program involving only 1 physical therapist was effective in facilitating physical functional recovery in patients with chronic stroke. A key contributing factor in this regard was likely the adherence to the ICF conceptual framework, 24 which emphasizes family involvement, repetitive task training, and reintegration into the society. Several clinical reports have suggested that the most effective approach to learn an activity is to practice it. Repetitive-task training can be effective in facilitating physical functional improvement in patients with stroke.45,46 CHI enabled the patients to repetitively practice specific daily tasks at home. Most caregivers were the patients’ spouses or close relatives and thus lived in the same environment and likely had the same living patterns. As such, they were most likely to serve as facilitators for the patients, providing the most needed care and rehabilitation skills. Patients’ physical functioning can therefore be more effectively maintained or enhanced with improvement of the caregivers’ ability to administer care, coupled with their high accessibility.
Caregiver Burden
Caregiver burden is primarily associated with the caregiver’s psychological distress, the amount of care provided, and the patient’s level of physical impairment.47,48 In this study, CHI did not significantly affect caregiver’s burden, neither on a per domain basis nor on a total burden basis. This lack of impact could be attributable to 2 factors: first, the impact was evaluated at the endpoint rather than at an earlier stage of the intervention; second, the study sample was relatively small. CHI may have exerted additional burdens on the caregiver, particularly during the earlier stage. Confirmation of this possible impact would probably require a larger study sample.
Strengths of the Study
The study has some strengths. First, to our knowledge, this is the first study to adopt the ICF conceptual framework to implement CHI. Second, the program was home based and patient centered. Thus, the patient and the caregiver can practice the rehabilitating skills in a familiar and comfortable home environment. Third, implementing CHI eliminated the need for patients to commute for rehabilitation. Transportation is often a major problem for these patients. Frequent trips are time-consuming and may not be affordable for many of them. Fourth, the patients learned the exact skills that are needed in their everyday lives. Finally, the training program emphasized social participation to facilitate patients’ reintegration into the society.
Limitations of the Study
This study also has limitations. First, CHI is unsuitable for patients without a suitable caregiver or for those with more severe disabilities. Patients with stroke of Brunnstrom recovery stage I or II may have muscle tone or spasticity but not voluntary movement control. These patients would require higher-level care, and their caregivers would require higher-level training. Second, the frequency of daily training varied among patients, which might have had some impact on the outcome. Third, despite our effort, the sample size was relatively small. Confirmation of these findings with a study involving a larger sample is desired. Fourth, the control group received a home visit or telephone call by a therapist to balance the therapist-patient contact time. Although no intervention was involved, the visit/call might have a slight effect on the recovery of the control patients. However, the exact effect could not be easily determined. Fifth, the data used in this study did not cover the posttraining period. Thus, how much of the learned skill was retained in the long-term is unknown. Sixth, only 10% of the patients with chronic stroke who were screened met the inclusion criteria. Some patients who met the stroke severity criterion were excluded because they were undergoing outpatient rehabilitation therapy 3 or more times per week. Finally, the generalizability of this study is limited to patients who met the inclusion criteria. A study is warranted to determine whether the intervention benefits patients undergoing outpatient rehabilitation therapy 3 or more times per week.
Conclusion
A 12-week caregiver-mediated rehabilitation program designed based on the ICF conceptual framework emphasizing home-based repetitive training, outdoor activities, and social reintegration is effective in improving the physical functional recovery of patients with chronic stroke. The program may have practical value for many patients with chronic stroke.
Footnotes
Acknowledgements
The authors wish to express their sincere appreciation to the participating hospitals, patients with stroke, and caregivers for their cooperation during the course of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by a grant from the National Science Council of Taiwan (NSC99-2314-B-468-001).
