Abstract
Background
The prognosis of prolonged disorders of consciousness (pDoC) in children has consistently posed a formidable challenge in clinical decision-making.
Objective
This study aimed to develop a machine learning (ML) model based on conventional structural magnetic resonance imaging (csMRI) to predict outcomes in children with pDoC.
Methods
A total of 196 children with pDoC were included in this study. Based on the consciousness states 1 year after brain injury, the children were categorized into either the favorable prognosis group or the poor prognosis group. They were then randomly assigned to the training set (n = 138) or the test set (n = 58). Semi-quantitative visual assessments of brain csMRI were conducted and Least Absolute Shrinkage and Selection Operator regression was used to identify significant features predicting outcomes. Based on the selected features, support vector machine (SVM), random forests (RF), and logistic regression (LR) were used to develop csMRI, clinical, and csMRI-clinical-merge models, respectively. Finally, the performances of all models were evaluated.
Results
Seven csMRI features and 4 clinical features were identified as important predictors of consciousness recovery. All models achieved satisfactory prognostic performances (all areas under the curve [AUCs] >0.70). Notably, the csMRI model developed using the SVM exhibited the best performance, with an AUC, accuracy, sensitivity, and specificity of 0.851, 0.845, 0.844, and 0.846, respectively.
Conclusions:
A csMRI-based prediction model for the prognosis of children with pDoC was developed, showing potential to predict recovery of consciousness 1 year after brain injury and is worth popularizing in clinical practice.
Keywords
Introduction
Prolonged disorders of consciousness (pDoC) refer to conditions where individuals experience disturbances in consciousness for over 28 days following a coma caused by severe acquired brain injury, including traumatic brain injury (TBI) and non-TBI. 1 pDoC generally encompasses the vegetative state/unresponsive wakefulness syndrome (VS/UWS) and the minimally conscious state (MCS). VS/UWS is characterized by wakefulness without awareness or reflexive/non-purposeful behavioral responses,2,3 whereas MCS may exhibit minimal and inconsistent but replicable voluntary behaviors. 4 The prognosis of individuals diagnosed with pDoC, including the possibility of recovery from DoC (favorable prognosis [FP]), persistence in a pDoC state, or mortality (poor prognosis [PP]), has received significant attention from medical professionals, family members, and society. Uncertain or incorrect prognostic information often leads to many important issues, such as medical management, ethical considerations, social implications, and even legal matters. In contrast, an accurate prognosis is beneficial for clinicians and the patients’ family members in making informed decisions and formulating an effective rehabilitation treatment plan, which is essential for the outcomes of patients with pDoC.
Currently, bedside neurobehavioral measures remain the primary approach for predicting the prognosis of individuals with pDoC despite their inherent misdiagnosis rate.5,6 With rapid advancements in neuroimaging techniques, it has emerged as a valuable complementary tool for bedside assessments.1,3 However, most neuroimaging studies of pDoC prognosis have primarily focused on functional brain imaging, which requires sophisticated data processing and occasionally yields inconsistent findings. Conversely, structural magnetic resonance imaging (MRI), particularly conventional structural MRI (csMRI), is widely used to assess the extent of brain damage in individuals with pDoC. Additionally, existing prognostic studies on pDoC have predominantly been conducted at the group level, and significant differences obtained at the group level cannot be directly applied to predict prognosis at the single-subject level. Machine learning (ML) offers a promising approach for quantifying neural response variability at the individual level and has been utilized to predict the diagnosis and outcomes of individuals with pDoC. 7 Furthermore, most studies involving pDoC participants have focused on adults rather than children, whereas pediatric patients tend to exhibit more favorable prognoses than adults. 8
Given the abovementioned facts and the evidence of the correlation between structural abnormalities and consciousness recovery in individuals with pDoC,9-11 we hypothesized that there may be brain structural disparities between the FP group (FPG) and PP group (PPG), which could be effectively detected using ML techniques. This study aimed to develop an ML prediction model for the automatic prognostic categorization of pediatric pDoC and identify the neuroimaging features derived from csMRI that may significantly contribute to the prognosis decisions in children with pDoC.
Materials and Methods
Ethics Statement
This study was approved by the Human Ethics Committee of the Children’s Hospital of Chongqing Medical University (File No. 2022078) and conducted according to the principles outlined in the Declaration of Helsinki. Due to the retrospective nature of the study, patient informed consent was waived. However, all patient data were anonymized to ensure confidentiality.
Participants
Data from 1553 children with DoC from January 2013 to December 2022 were extracted from the hospital-specific electronic medical record system. The preliminary screening of the data was performed based on the following inclusion and exclusion criteria. The inclusion criteria were as follows: (1) age <18 years; (2) participants who fulfilled the DoC diagnostic criteria based on the repetitive Coma Recovery Scale-Revised (CRS-R) and the duration of DoC was at least 28 days from the incident; and (3) participants who had an available outcome of consciousness recovery at 1-year post-injury. The exclusion criteria were as follows: (1) participants who did not undergo MRI Examination within 4 to 12 weeks after injury and (2) MRI data of the pDoC were incomplete or had serious artifact interference, which did not meet the subsequent analysis. After screening, 196 children with pDoC were enrolled. All participants were then randomly assigned in a 7:3 ratio to the training set (138/196) and test set (58/196), respectively. Based on the consciousness status recorded in the electronic medical records or via telephone follow-up 1 year after onset, all included children with pDoC were categorized into 2 groups: FP and PP. If a child with pDoC can use an object or intelligible verbalization, indicating that he or she is in a state of emergence from MCS (EMCS), this is considered as regaining consciousness and has a FP. Conversely, if a child with pDoC remained in the state of VS/UWS or MCS, he or she was considered to have a PP. Finally, the FP group included 67 individuals in the training set, and the PP group included 71 individuals. The FP group included 32 individuals in the testing set, and the PP group included 26 individuals. Supplemental Figure S1 shows the participants’ screening flowchart.
Imaging Protocol
The enrolled children underwent multi-sequence cranial MRI examinations using a 1.5 Tesla MR scanner (GE, Signa Propeller HD, USA) or a 3.0 Tesla MR scanner (Achieva, Philips Medical Systems, Holland), both equipped with an 8-channel head coil. Supine and head-advanced positions were used for all participants. The sequences and parameters used are listed as follows: (1) axial T1-weighted image (WI) with inversion recovery (IR) sequence, TR/TE (ms) = 2000/20, TI = 800 ms; (2) axial T2W fluid-attenuated inversion recovery (T2W-FLAIR), TR/TE = 8000/125, TI = 2500 ms; and (3) axial and sagittal T2WI with turbo spin echo (TSE) sequence, TR/TE = 3500/80; other parameters included field of vision of 220 mm × 220 mm to 240 mm × 240 mm, slice thickness of 6.5 mm, and 20 slices.
Procedure of csMRI Model Establishment
Based on the existing literature on pDoC,9,12,13 43 features were extracted from csMRI images for comprehensive semi-quantitative and qualitative evaluation. These features included global cortical atrophy,13,14 medial temporal lobe atrophy, 15 posterior trophy,16,17 basal ganglia atrophy, thalamic atrophy, brainstem atrophy, and cerebellar atrophy. The presence of leukoaraiosis, 18 ventricular enlargement, 19 basal ganglia abnormalities, thalamic abnormalities, brainstem lesions, and signal intensity ratio (SIR) of each lesion were also considered. Subdural effusion/hemorrhage and midline structural displacement were also assessed. The extraction process of the structural information of the region of interest (ROI) and the evaluation of semi-quantitative csMRI images are detailed in the Methods section, Figures S2 to S6, and Table S1 in the Supplemental Materials. All evaluations were conducted in consensus by 2 pediatric neuroradiologists with 8 and 10 years of experience, respectively. Although the 1-year post-injury outcomes of pediatric pDoC were known, the readers were blinded to the outcomes during the review. In cases of disagreement, the 2 neuroradiologists repeated the double-blind reading with a third senior neuroradiologist until a consensus was reached.
The Mann–Whitney U-test and Least Absolute Shrinkage and Selection Operator (LASSO) regression were employed to analyze the training set for feature screening. Supplemental Figure S7 illustrates the detailed feature screening process. The selected feature were employed to establish ML models using support vector machine (SVM), random forest (RF), and logistic regression (LR), which are suitable for small sample sizes. The models were trained and validated using a fivefold cross-validation technique on the training set, ensuring model robustness and hyperparameters determination using the grid search method. The test sets were exclusively utilized to assess the generalizability of the models, encompassing metrics such as the area under the curve (AUC), accuracy (ACC), sensitivity (SEN), and specificity (SPE) based on receiver operating characteristic (ROC) curve analysis. Importantly, the performance on the training set served only as a reference, whereas the test set remained entirely independent of any training processes to accurately assess generalizability. Figure 1 shows a detailed pipeline of the ML process.
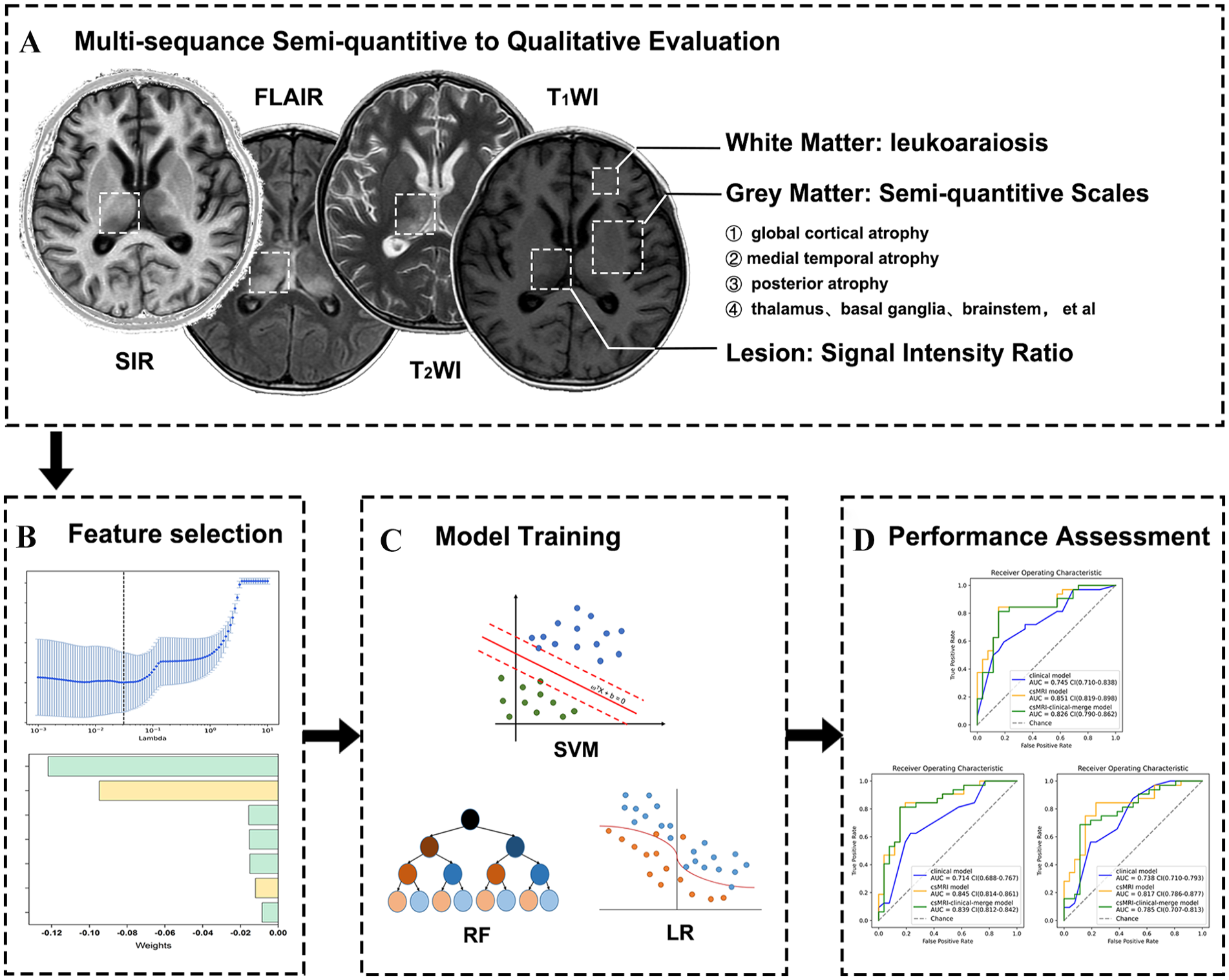
The workflow for establishing the csMRI model. (A) A comprehensive assessment was conducted using a semi-quantitative to qualitative analysis of visual evaluation of the gray matter, white matter, and lesion morphology and signal intensity on cranial csMRI images obtained within 4 to 12 weeks following the injury. (B) Features associated with the recovery of consciousness 1 year after injury were screened using LASSO regression in the training set, with the LASSO coefficient calculated for each feature. (C) All model was established using fivefold cross-validation by SVM, RF, and LR. (D) The generalization ability of these models was validated in the test set.
Procedure of Clinical Model Establishment
The clinical prediction model was established using the demographic and clinical characteristics of all participants (Table 1), including sex, age, age of onset (≥2 or <2 years, considering the relationship between age and white matter myelination), initial status (UWS/VS or MCS), the CRS-R total score, and the CRS-R subset score. The clinical characteristics were selected for evaluation based on their partial mention in previous studies.20-23 The total CRS-R score and the score on every subscale were extracted from the patient’s electronic medical records on the day of the MR examination. If the CRS-R assessment was not performed on the day of the MR examination, it was complimentarily assessed by 2 rehabilitation physicians with 5 to 10 years of experience based on the course records of the patient on the day of the MRI. The readers were blinded to review outcomes. In cases of disagreement, a third senior rehabilitation specialist performed a double-blind review.
Demographic and Clinical Characteristics.
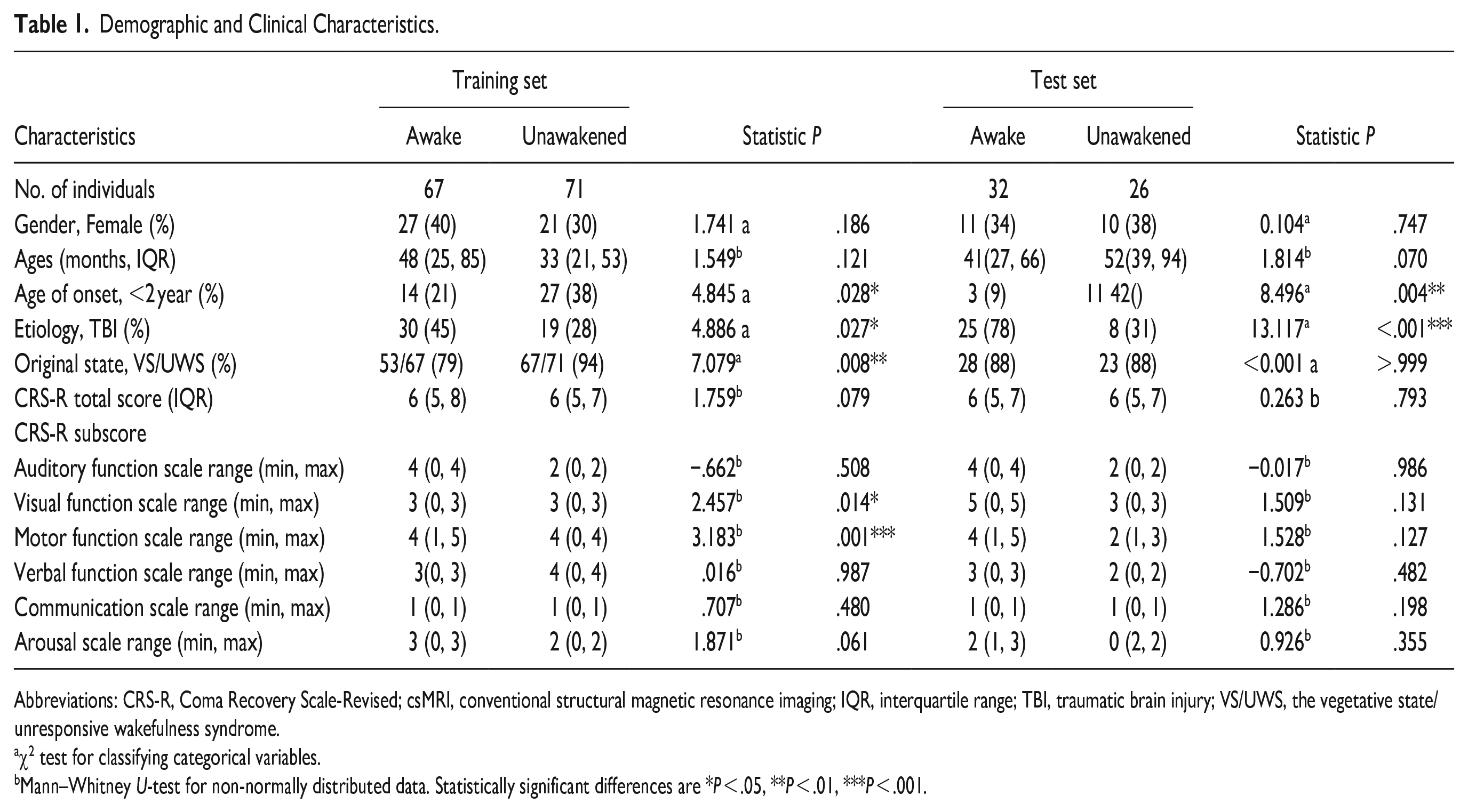
Abbreviations: CRS-R, Coma Recovery Scale-Revised; csMRI, conventional structural magnetic resonance imaging; IQR, interquartile range; TBI, traumatic brain injury; VS/UWS, the vegetative state/unresponsive wakefulness syndrome.
χ2 test for classifying categorical variables.
Mann–Whitney U-test for non-normally distributed data. Statistically significant differences are *P < .05, **P < .01, ***P < .001.
The same approach used to construct the csMRI model was utilized to establish the clinical model based on the demographics and clinical features in the training set. The generalizability of the clinical model was validated using the test set.
Procedure of csMRI-Clinical-Merge Model Establishment
The merged model was constructed by integrating the features of the aforementioned csMRI and clinical prediction models.
Statistical Analysis
Data comparisons between FPG and PPG in both the training and test sets were conducted using Python (version 3.7.12, www.python.org/). The Mann–Whitney U-test was performed to compare distributed continuous variables between the FPG and PPG, while categorical variables were analyzed using the χ2 test. The AUC of the ROC curve was used to evaluate the model performance, with 95% confidence intervals (CI) calculated. Additionally, the ACC, SEN, and SPE were computed. The AUCs of the different models were compared using the paired DeLong test facilitated by the MedCalc software (version 19.07). Statistical significance levels were denoted as follows: *P < .05, **P < .01, and ***P < .001.
Results
Demographics and Clinical Characteristics
Table 1 summarized the demographic and clinical characteristics of the study participants. No significant differences in sex, age, CRS-R total score, and CRS-R sub-scores of auditory function, verbal function, communication, and arousal were observed between the FPG and PPG, either in the training set or test sets (both P > .05). However, significant differences were observed in age at onset, etiology, initial state, and CRS-R subscale scores for visual and motor functions between the 2 groups (all P < .05), except for the initial state and visual and motor function scores in the test set (p > 0.05).
csMRI and Clinical Features
Based on the relative importance ranking, 7 features related to the recovery of consciousness were identified in the csMRI model, including bilateral basal ganglia atrophy/lesions, bilateral thalamic atrophy, diffuse cortical atrophy, left posterior (including the posterior cingulate cortex and precuneus) atrophy/lesions, and left medial temporal lobe atrophy. In the clinical model, 4 features were found to be associated with the recovery of consciousness, including motor and visual subscales of the CRS-R, age of onset, and etiology. The csMRI-clinical-merge model combined the 11 features mentioned above.
Performance of ML Model
Figure 2 shows a detailed comparison of ROCs of models by SVM, LR, and RF at both the training and test sets. Except for the clinical model established using SVM, RF, and LR, which achieved AUCs of 0.775, 0.756, and 0.708 in the training set and 0.745, 0.714, and 0.738 in the test set, respectively. All other models attained AUC values exceeding 0.8.
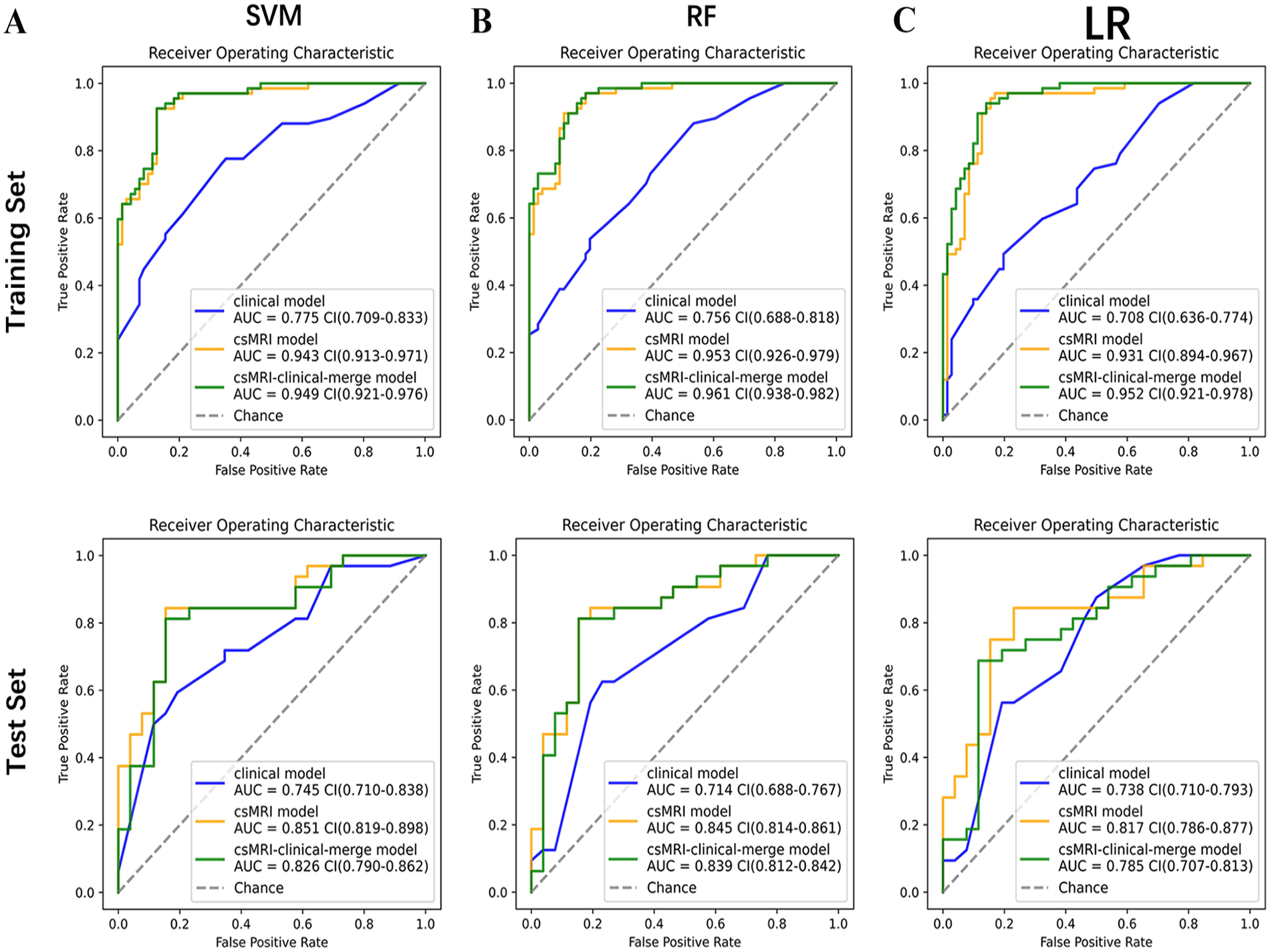
Comparison of ROCs of models by SVM, LR, and RF at both the training and test sets. (A-C) The ROCs of models by SVM, RF, and LR on the training set. (A) The ROCs of models by SVM. (B) The ROCs of models by RF. (C) The ROCs of models by LR. (D-F) The ROCs of models by SVM, RF, and LR on the test set. (D) the ROCs of models by SVM. (E) the ROCs of models by RF. (F) the ROCs of models by LR.
Table 2 summarizes the performances of all the models. The 3 models developed using SVM demonstrated relatively robust performance, with better AUC, ACC, SEN, and SPE than those developed using RF and LR. Among all models, the csMRI model developed with SVM exhibited superior robustness and performance, achieving the highest values, with an AUC, ACC, SEN, and SPE of 0.851, 0.845, 0.844, and 0.846, respectively.
Indicators of 3 Models.
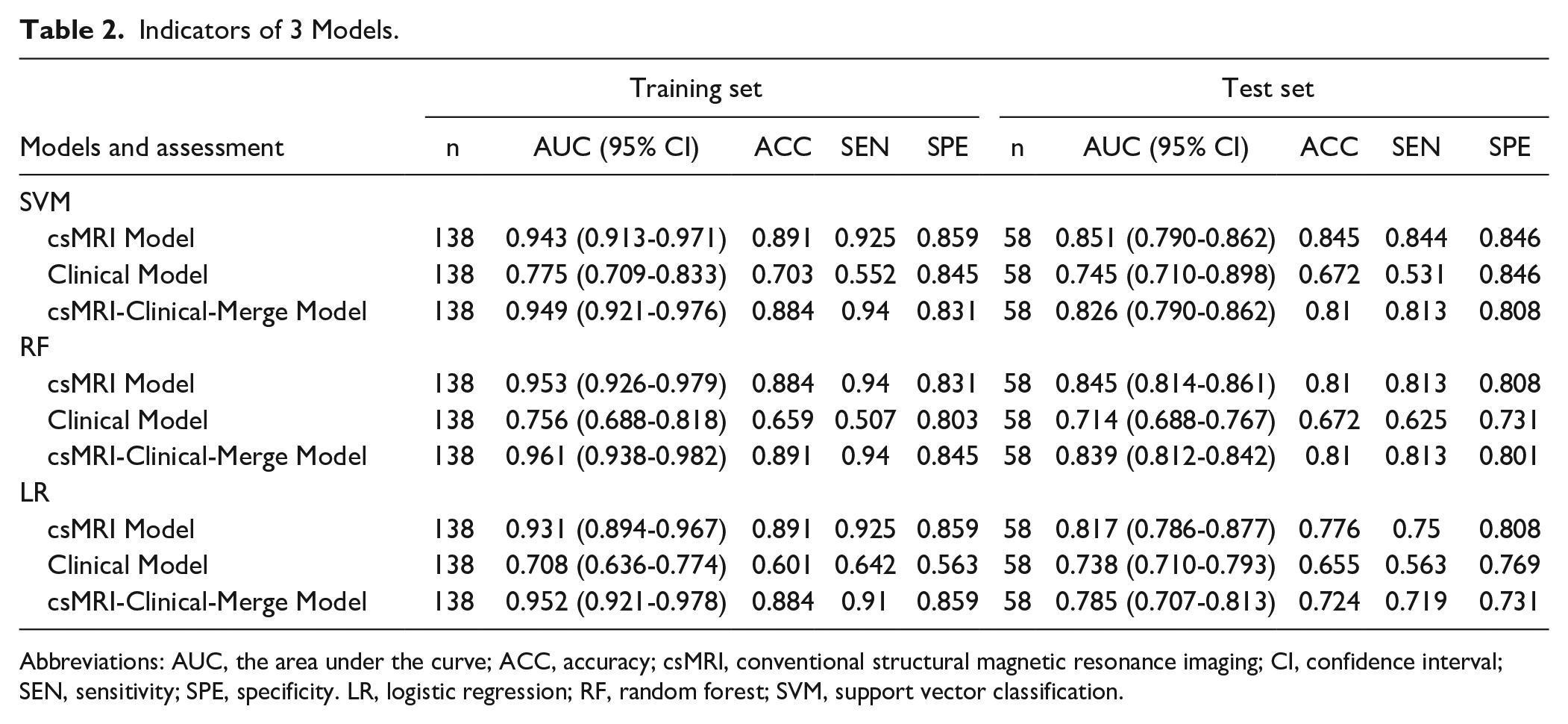
Abbreviations: AUC, the area under the curve; ACC, accuracy; csMRI, conventional structural magnetic resonance imaging; CI, confidence interval; SEN, sensitivity; SPE, specificity. LR, logistic regression; RF, random forest; SVM, support vector classification.
For the statistical comparison of model performance in predicting the prognosis of pediatric pDoC 1 year after the onset of brain injury, the DeLong test revealed no statistically significant difference in the AUCs between the csMRI model developed using SVM, RF, or LR, as well as the clinical and csMRI-clinical-merge models (all P > .05). Based on a comprehensive comparison, we selected the csMRI model developed using the SVM as the optimal choice for predicting the prognosis of pediatric pDoC.
To visually compare the predictive performances of all models, a confusion matrix and the 4 representative MRI cases were employed. (Figures 3 and 4, respectively).
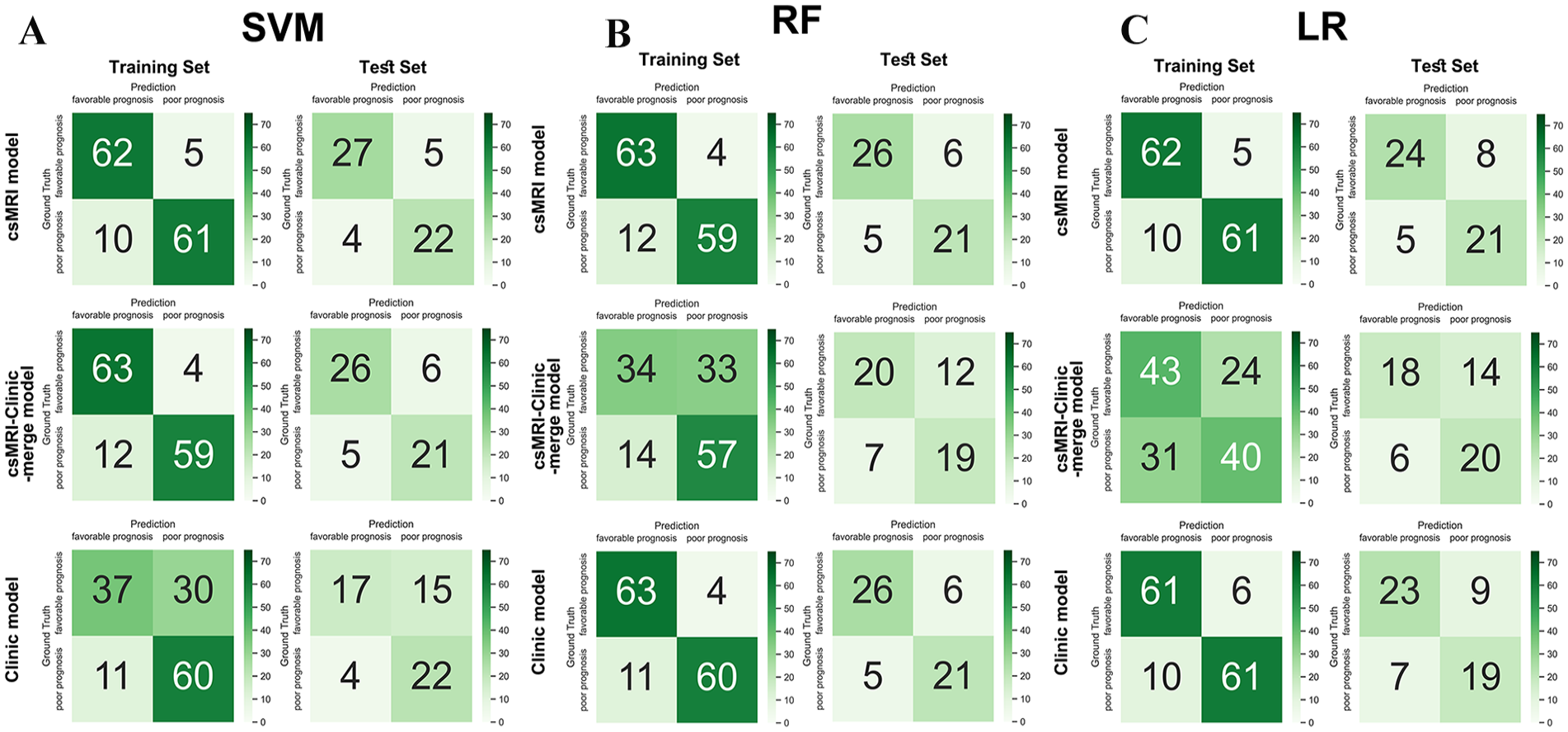
Confusion matrixes of all models on the training set and the test set. (A) Graph of the confusion matrixes of the 3 models by SVM. (B) Graph of the confusion matrixes of the 3 models by RF. (C) Graph of the confusion matrixes of the 3 models by LR.
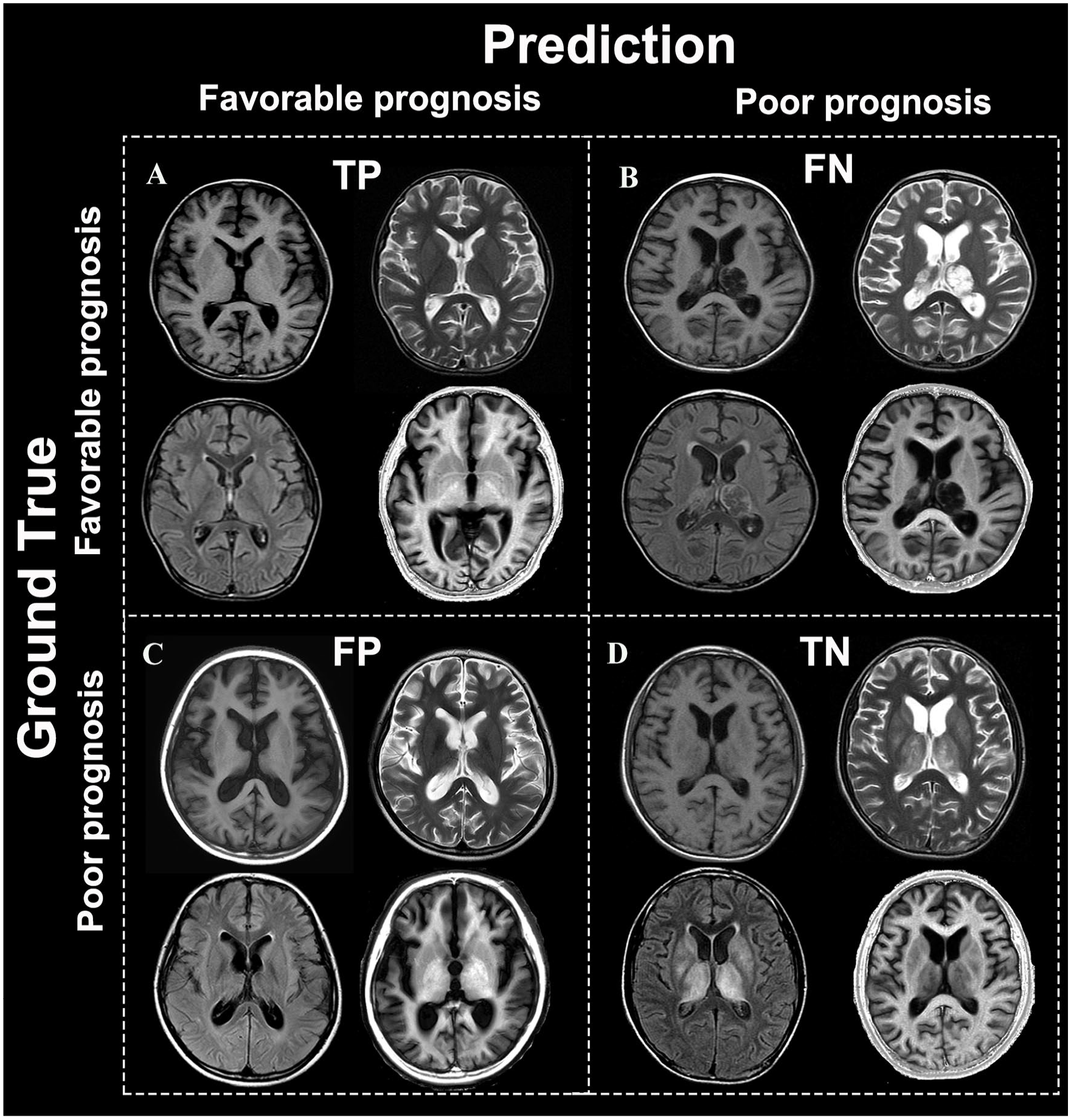
Representative MRI cases in the test set. Representative MRI images of the test set. The 4 typical MRI cases were diagnosed using the csMRI model, with the 1-year post-injury outcomes being positive predictions, resulting in (A) true positive, (B) false negative, (C) false positive, and (D) true negative.
Discussion
To our knowledge, this study is the first to use ML based on csMRI data and demographic characteristics to develop models for predicting recovery of consciousness in children with pDoC at 1-year post-brain injury. Instead of employing more advanced imaging techniques such as advanced structural and functional MRI, we used csMRI as the SVM classification data due to its broader clinical applicability across all MRI scanners. This investigation focused specifically on children <18 years old, contrasting with previous prognostic studies on pDoC that primarily involved adult cohorts. Additionally, the contribution of each neuroimaging feature to prognosis classification was meticulously retained and elucidated, providing clinicians with a valuable tool for individual-level prognosis prediction in children with pDoC; This method is generally more acceptable to professionals and families than relying solely on uncertain and unexplained predictions.
Among the neuroimaging characteristics in our study, bilateral basal ganglia atrophy/lesion, bilateral thalamus atrophy, diffuse cortical atrophy, left posterior atrophy/lesions, and left medial temporal lobe atrophy were the top 7 contributors to the prognostic prediction of the state of consciousness 1 year after brain injury.
The thalamus plays a crucial role in relaying sensory and motor signals and is involved in the regulation of consciousness, sleep, and alertness, while the basal ganglia acts as a relay station for extrapyramidal systems. Information is transmitted to the cerebral cortex through the thalamus while impulses are also conveyed downwards via it. Previous studies have shown that the basal ganglia and thalamus play important roles in the development and maintenance of consciousness,24-27 plausibly explaining why bilateral damage to these areas served as a robust predictor of consciousness recovery in our model.
The cerebral cortex, characterized by an intricate network of neurons, is the most advanced neural center in humans. This brain region is widely acknowledged to be the fundamental neurobiological basis of human consciousness. The extent of diffuse cortical atrophy strongly correlated with the recovery of consciousness in individuals with pDoC, consistent with previous findings. 9 Our study also found a strong correlation between damage to the left posterior regions and the left medial temporal lobe and the recovery of consciousness. The posterior cingulate cortex (PCC) and its adjacent precuneus (PCU) are the important areas in the posterior regions, playing a predominant role in encoding autobiographical memories and facilitating self-referential cognitive processes as pivotal constituents of the default mode network. 28 The hippocampus is an important component of the medial temporal lobe. The study conducted by Van den Heuvel and Sporns 29 proposed that the hippocampus and PCC/PCU are part of “brain hubs,” which are recognized as regions exhibiting a higher degree of connectivity compared to other brain areas, playing a crucial role in facilitating global information integration across different network modules. Although these results are derived from functional changes, structure often underlies function, and abnormalities in structural organization can partially reflect abnormalities in functional processes. Notably, the posterior atrophy/lesion and the medial temporal lobe atrophy associated with consciousness recovery in our study tended to involve the left side, aligning with previous research findings.28,30,31 Injury to the left side has a greater predictive value for the outcome compared to the bilateral or the right side, which requires further investigation. We speculate that this phenomenon may be attributed to the predominance of the left hemispheres in humans, which is responsible for highly demanding tasks such as language and fine motor coordination. 32 However, the specific mechanism requires further study.
In this study, we made the first reported attempt to employ SIR values to assess the extent of myelin damage in individuals with pDoC. The results did not show a statistically significant association between SIR values and the recovery of consciousness. This could be attributed to the wide age range of pediatric patients with pDoc in our cohort, posing challenges in accurately evaluating lesion severity based on the SIR values obtained from a single time MRI examination. In future studies, it would be beneficial to monitor the changes in SIR values through individual longitudinal follow-ups to elucidate the extent of myelin damage or repair. Based on these findings, further studies should be conducted to investigate potential correlations between pediatric pDoC recovery and fluctuations in SIR values.
Currently, the prevailing consensus suggests that etiology, age of onset, and clinical evolution play significant roles in guiding the prognosis of individuals with pDoC. In our clinical model, children with pDoC due to TBI exhibited a more FP than those with non-TBI.1,22 The superior prognosis observed in children compared to adults following brain injury is widely acknowledged and potentially attributed to the enhanced neuroplasticity inherent in the developing brain.33,34 The prognostic significance of age of onset in pediatric pDoC remains unclear. However, we observed a more favorable prognosis among children with brain injury who experienced an age of onset ≥2 years compared to those with an age of onset <2 years in our pediatric cohort. We speculate that the process of white matter myelination in children is usually finalized by approximately 2 years of age, and myelinated brain tissue exhibits greater resilience against injury than non-myelinated or demyelinating brain tissue, which requires further research to confirm.
Currently, the CRS-R 35 remains widely acknowledged as the most reliable and validated clinical tool for diagnosing and prognosticating pDOC.1,35,36 However, it has not been validated in pediatric cohorts. Our findings suggested that the CRS-R total score may not be a reliable indicator for assessing the prognosis of children with pDoC, which is inconsistent with the results of previous studies conducted on adults with pDoC.37,38 Interestingly, we observed that the visual and motor subscales of the CRS-R emerged as predictors for prognostic evaluation in pediatric pDoC, which aligns with previous findings.39-41 This may be attributed to the fact that visual and motor skills precede language skills in children’s developmental milestones.
Based on the aforementioned 7 features in csMRI and 4 clinical features, csMRI, clinical, and csMRI-clinical-merge models using SVM, RF, and LR were successfully developed to differentiate pDoC prognosis. While there were no statistically significant differences in the AUCs across all models, the csMRI model developed with SVM exhibited the best robustness and performance, with higher ACC, SEN, and SPE. This model’s performance was comparable to that of the csMRI-clinical-merge model, indicating that incorporating clinical features did not significantly enhance the predictive capability of the combined model. This result may be attributed to the relatively small sample size, considerable variability in clinical features, and their impact on the classification results in pediatric pDoC. Therefore, it is reasonable to assert that the csMRI model holds substantial potential as a decision-making tool for predicting the prognosis of pediatric pDoC.
This study had certain limitations. First, we used only a single time MRI point to assess the relationship between brain injury and recovery of consciousness, without conducting longitudinal follow-ups at multiple time points to observe the connection between changes in brain injury and recovery of consciousness. Second, this study primarily focuses on the prognostic classification of children with pDoC based on neuroimaging data and incorporated limited clinical features. Future investigations should aim to improve model performance by integrating additional clinical features, electroencephalograms and advanced structural and functional MRI attributes. Finally, this study was based on a small-scale single-center cohort. Future research should expand the sample size across multiple centers to validate the model’s generalizability.
Conclusions
In summary, we pioneered the use csMRI and clinical data to develop ML models for predicting 1-year outcomes in pediatric patients with pDoC. Our findings indicate that the SVM-based csMRI model outperformed the clinical model and performed similarly to the csMRI-clinical-merge model. Additionally, other ML models, including RF and LR models, demonstrated robustness in their predictions. These results underscore the potential of csMRI to accurately predict the long-term prognosis of pediatric pDoC and provide clinicians with valuable prognostic information that can assist in formulating targeted rehabilitation plans tailored to the needs of patients with pDoC.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241287187 – Supplemental material for Predicting Long-Term Outcome of Prolonged Disorder of Consciousness in Children Through Machine Learning Based on Conventional Structural Magnetic Resonance Imaging
Supplemental material, sj-docx-1-nnr-10.1177_15459683241287187 for Predicting Long-Term Outcome of Prolonged Disorder of Consciousness in Children Through Machine Learning Based on Conventional Structural Magnetic Resonance Imaging by Helin Zheng, Shuang Ding, Ningning Chen, Zhongxin Huang, Lu Tian, Hao Li, Longlun Wang, Tingsong Li and Jinhua Cai in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Helin Zheng: Conceptualization; Data curation; Formal analysis; Resources; Writing—original draft. Shuang Ding: Data curation; Methodology; Software. Ningning Chen: Data curation; Investigation. Zhongxin Huang: Data curation; Resources. Lu Tian: Data curation; Investigation. Hao Li: Data curation; Investigation. Longlun Wang: Data curation; Investigation; Methodology. Tingsong Li: Conceptualization; Resources; Writing—review & editing. Jinhua Cai: Conceptualization; Resources; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
