Abstract
Introduction
Higher cortical activity has been observed in people with multiple sclerosis (pwMS) during walking and dual-tasking. However, further studies in overground walking and considering pre-frontal cortex (PFC) sub-areas are necessary.
Objectives
To investigate PFC activity during a cognitive-motor dual-task (DT) and its single component tasks, in combination with behavioral outcomes in pwMS.
Methods
Fifteen pwMS (EDSS 3.5 [2-5.5], 42 ± 11 years) and 16 healthy controls (HC, 45.2 ± 13.2 years) performed 3 conditions: single motor-walking (SWT), single cognitive – subtracting sevens (SCT), and a DT. Meters walked and the number of correct answers were obtained from which, respectively, the motor (mDTC) and cognitive (cDTC) DT costs were calculated. A functional Near-Infrared Spectroscopy covering the frontopolar and dorsolateral PFC (DLPFC) areas was used to concentration of relative oxyhemoglobin (ΔHbO2) and deoxyhemoglobin (ΔHHb) in the PFC. A repeated 2-way ANOVA (group × conditions) was used to compare ΔHbO2/ΔHHb and behavioral outcomes.
Results
PwMS walked shorter distances (
Conclusion
Higher frontopolar activity in pwMS compared to HC in the SWT suggests reduced gait automaticity. Furthermore, it seems that only HC increased neural activity in the frontopolar in the SCT and DT, which might suggest a limit of cognitive resources to respond to DT in pwMS.
Introduction
Multiple sclerosis (MS) is a chronic, auto-immune, inflammatory, and degenerative disease of the central nervous system that manifests with a variety of symptoms, among which are motor impairments, such as walking, 1 and cognitive disorders.2,3 Difficulties in walking alongside impairments in cognitive functions may result in a higher risk of falling.4,5 During community ambulation, people with MS (pwMS) walk slower and with larger gait variability than healthy controls (HC). 6 Daily walking often accompanies cognitive demands (e.g., talking while walking), demanding increased executive functioning and attention. 7 This simultaneous performance of 2 tasks is called dual tasking. During a dual task (DT), decrement in performance may occur in one or both tasks compared to the performance during a single task. The interaction between cognitive and motor tasks is called cognitive-motor interference (CMI). It can be measured using the DT cost (DTC) (i.e., the percentage change in performance during the DT relative to the single task). 8
PwMS usually report increased difficulties with motor-motor and cognitive-motor dual-tasking in daily life, for example, during balance and walking activities.9-11 Although research regarding dual tasking during walking has increased, there are still inconclusive results about the magnitude of the CMI in pwMS compared to healthy controls (HC).9,12 Studies did not consistently demonstrate a higher DTC in pwMS than in HC, 13 albeit performance declines in absolute values are present.
At the brain level previous research has shown that pwMS often presented higher neural activity in motor (planning) areas with poorer motor and cognitive performance than HC during single walking or in situations that mimic daily life and require a higher cognitive load (i.e., DT or crossing an obstacle).10,14-19 Previous studies have investigated brain activation, specifically the pre-frontal cortex (PFC), using the dual-tasking paradigm while walking on a self-paced treadmill.16-19 Three studies investigated the dual-tasking paradigm during overground walking, measuring either the PFC15,20 or the premotor cortex (PMC) and the supplementary motor area (SMA). 14 For example, Saleh et al, 14 investigated the role of the PMC and SMA during a single cognitive task (SCT), a single walking task (SWT), and a DT. The DT condition required increased PMC activation compared to the SWT, only for the HC. In addition, higher cortical activity was observed in the PMC and in the SMA during the DT condition, compared to the SCT, for both groups. The PFC, specifically, the Brodmann areas 10 (frontopolar), 9, and 46 (dorsolateral pre-frontal cortex-DLPFC), plays an important role in executive function, human walking, balance, and dual-tasking.7,21,22 Studies have investigated the PFC activity under DT conditions in several populations (e.g., older adults, young people, MS, Parkinson’s disease).23,24 In general, neurodegenerative disorders and older adults have shown increased PFC activation during DT, suggesting the need for more attentional resources. For pwMS, previous studies investigating PFC under DT and in self-paced treadmill walking found that pwMS increased PFC activation in order to respond to DT demand with poorer walking and cognitive performance.16-19 On the other hand, in overground condition, the results are mixed among the studies. While Kupchenko et al, 15 observed no increase in PFC activity from SWT to DT, Holtzer et al, 20 observed an increase in the DT condition compared to single task. Importantly, the majority of the studies did not look to sub-areas of the PFC (i.e., frontopolar—responsible for strategic planning, decision-making, and integration of sensory input, cognitive and motor processes; and DLPFC—responsible for executive functions, working memory, attention, and gait adaptation in complex conditions) during single walking,25,26 single cognitive and dual-task, did not use short-separation channels (increasing false positive results chance) and did not exclude the initial phase of the task (which refers to adaptation to the task or gait initiation process). 27 Also, DTC cost, for behavioral data, was only mentioned in 2 previous studies.14,19 Therefore, further investigations are necessary to fully understand the role of the PFC (frontopolar and DLPFC) activity in a DT overground walking condition.
Walking is normally controlled by 2 supraspinal locomotor pathways (automatic and executive networks), and it involves the interaction of various brain structures. 28 The loss of walking automaticity leads to the use of more executive function control,7,10,29 perhaps causing competition for neural resources between walking and cognitive processes during a DT (capacity sharing model—Leone, Patti, and Feys 8 ). Additionally, one may argue that the compensation-related utilization of neural circuits hypothesis (CRUNCH) 30 can also explain the CMI in neurological populations such as MS. 24 The CRUNCH hypothesis states that in the aging brain increased neural activity and recruitment of other brain regions might lead to similar performance (e.g., gait or cognitive performance). However, as the complexity of the task increases, a ceiling in neural activation can be reached, consequently affecting performance. Therefore, considering that pwMS have a lower neural reserve, 31 one might expect insufficient capacity to recruit neural resources and poorer task performance during DT compared to their healthy counterparts. 30
The current study aims to examine the frontopolar and DLPFC activation and behavioral performance during 3 different conditions (SWT, SCT, and DT) in pwMS and HC. Our hypotheses, based on the capacity sharing model and the CRUNCH hypothesis, are the following: (i) higher frontopolar and DLPFC activation will be found in PwMS compared to HC during SWT, SCT, and DT, (ii) additional recruitment of the cortical frontopolar and the DLPFC areas to respond to the DT compared to the ST conditions (SWT and SCT) will be found only in the HC group, and (iii) lower performance during walking and cognitive tasks in PwMS compared to HC due to the MS-neurodegeneration.
Method
Participants
PwMS and HC were recruited via flyers, social media, and the National MS Center Melsbroek in Belgium. Inclusion criteria were between 18 and 65 years old, minimal cognitive functioning as measured with the Montreal Cognitive Assessment test of ≥26, and able to walk consecutively for 6 minutes according to the participants’ own estimate. Specifically, for pwMS, additional inclusion criteria were being diagnosed with MS according to the McDonald criteria, 32 no relapse within the past 30 days, and a score on the Expanded Disability Status Scale (EDSS) of ≥2.0 and ≤5.5. Exclusion criteria were other medical conditions interfering with mobility, other neurological diagnoses, or major hearing or visual problems. The leading (UHasselt) and local (National MS Center Melsbroek) Ethical Committee approved the study, and all participants gave their consent by signing an informed consent (ClinicalTrials identifier NCT04538872).
Experimental Procedure
The fNIRS and related gait and DT performance data presented in the present study were collected during an fNIRS task embedded within a study on motor learning (results published in Veldkamp et al, 33 ). Therefore, the current study is a secondary aim of the previous study reported elsewhere 33 and no power analysis was performed a priori for such purpose. In short, this study was a randomized controlled pilot trial to examine motor sequence learning of a goal-directed stepping task involving dynamic balance in pwMS and HC. The study consisted of 3 test moments, namely (1) baseline testing, (2) a learning session, and (3) a delayed retention session 24 hours after the learning session. On the last test moment, the tasks with the fNIRS measures reported in this study were additionally performed. In the present study, data from the baseline (descriptive and clinical measures) and from the fNIRS task are used. All measurements took place at 2 different sites in Belgium: at the REVAL research center of the Faculty of Rehabilitation Sciences at the University of Hasselt in Diepenbeek (healthy controls group) or at the National Multiple Sclerosis Centre—Melsbroek (pwMS group).
Descriptive and clinical measures were taken. Demographic measures included age, sex, height, weight, and level and years of education. Additionally, for pwMS, type of MS, EDSS, date of diagnosis, and MS medication were noted.
Six tests of cognitive functioning were obtained. The Montreal Cognitive Assessment (MOCA) was used for cognitive screening. The Brief Visuospatial Memory Test (BVMT-R) and the 10/36 Spatial Recall Test (SPART) were administered to assess visual learning and visuospatial learning. The Symbol Digit Modalities Test (SDMT) and the Paced Auditory Serial Addition Test (PASAT)−3 seconds were conducted to assess information processing speed and concentration. Auditory and visual working memory was assessed with the auditory digit span backwards and the visually digit span forward (Corsi) tests, respectively.
To assess motor functioning, participants performed the 6-minute walking test (6MWT), the Timed-Up and Go (TUG), the Timed 25-foot walk (T25FW), the Four-Square Stepping Task (FSST), the Berg Balance Scale (BBS), the Timed Tandem Walk 3 meters (TTW-3), and the Motricity Index.
Lastly, patient-reported outcomes were the Movement Specific Reinvestment Scale (MSRS) to assess someone’s disposition for conscious control of movement, the dual-task questionnaire (DTQ), the Multiple Sclerosis Walking Scale-12 (MSWS-12), the modified Fatigue Impact Scale (MFIS), the MS neuropsychological screening questionnaire (MSNQ), and the activities-specific balance confidence scale (ABC).
Dual-Task Paradigm & Behavioral Measures
The participants randomly performed 3 conditions in a block design, repeated 5 times each, resulting in 15 trials. See Figure 1 for a representation of the block design. Each trial of a specific condition lasted 25 seconds, with 10 to 25 seconds of rest between the trials, where the participant stood still in an upright position. The 3 conditions were (i) a single walking task (SWT) consisting of walking at a comfortable pace back and forth a 30-meter hallway; (ii) a single cognitive task (SCT) consisting of subtracting sevens aloud from a given starting number while standing still, and (iii) a cognitive-motor DT consisting of subtracting sevens while walking at a comfortable pace. This cognitive task was chosen based on previous studies that found a larger DTC when using a subtraction task compared to other cognitive tasks and on previous literature of neurological conditions as stroke and Parkinson’s disease.34-38 Each of the 5 SCT trials had a different starting number (i.e., 152, 165, 174, 186, or 198), of which the order was randomized between participants. The same 5 starting numbers were used for the DT trials, but participants never received the same starting number consecutively. The last 5 seconds of the rest period consisted of instructions about the subsequent trial given via an audio file through a speaker. Participants were asked to start walking when hearing a ‘beep’ sound for the SWT. For the SCT and DT trials, the task should be initiated when hearing the starting number of the subtracting sevens. A “beep” at the end of each trial indicated the end of that trial and the beginning of the rest period. Distance walked (i.e., meters) during the SWT and DT were measured. The average distance walked in the SWT and DT over the 5 respective trials was used as the outcome measure. The answers given during the single and dual cognitive subtasks were noted. The outcome measure used was the single task and dual task mean number of correct subtractions over the 5 respective trials. In addition, the DTC was calculated for both walking (mDTC) and cognitive (cDTC) conditions (DTC = ((DT performance − single condition performance)/single condition performance)*100). A negative value represents worse performance on the DT compared to the single tasks.
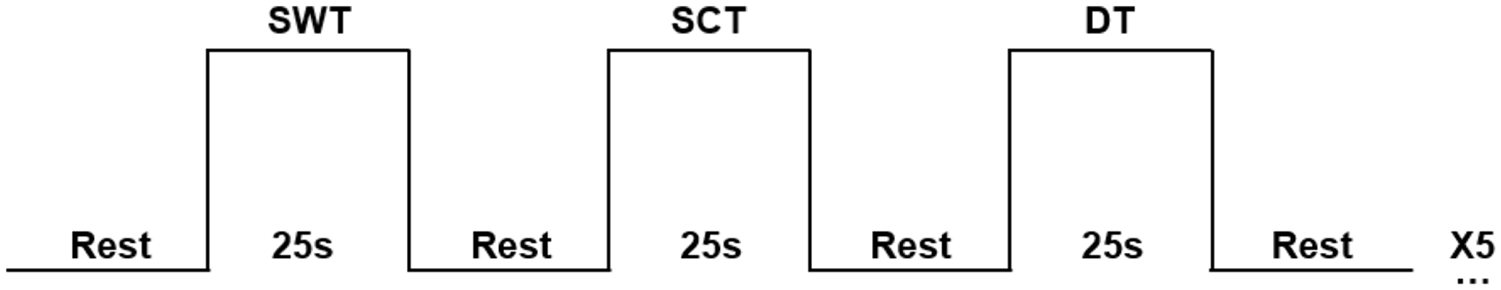
An example of the block design used in the study. Conditions of SWT (single walking task), SCT (single cognitive task), and DT (dual task) were randomized over 5 repeated trials.
Three wearable inertial sensors (Opal, v1, APDM Inc., Portland Oregon), acquiring data at 128 Hz, were positioned at both feet and in the lumbar region to quantify spatial-temporal gait parameters. Data were transmitted wireless to the data collection computer, allowing participants to move freely during the walking tasks. In addition, the Mobility Lab software was used for data acquisition. It provided the following gait variables: cadence, stride length, step duration, percentage in double support, and percentage in swing and stance phase. Gait variability was also calculated as the standard deviation from the whole trial for step duration and swing and stance phase variables.
Functional Near-Infrared Spectroscopy (fNIRS)
The fNIRS acquisition followed the guidelines for fNIRS acquisition during gait. A portable fNIRS (NIRSport2, NIRx Medical Technologies, Germany), with data collection frequency set at 10.2 Hz and wavelength ranging between 760 and 850 nm, was used for brain hemodynamics measurement. The system consisted of 8 detectors and 8 sources, with 1 detector used as a short-channel and with a minimum source-detector distance of 30 to 40 mm. The fNIRS cap was placed over the frontopolar (Broadman 10) and DLPFC (Broadman 9 and 46) using the fNIRS Optodes Location Decider (FOLD), with the 10-10 International system electrode placement used as reference. To minimize interferences of external light, an additional cap was worn over the fNIRS cap on which the optodes were attached. fNIRS data was collected through Aurora Software (NIRx Medical Technologies, Germany). PsychoPy was used to synchronize fNIRS with the execution of the task conditions.
fNIRS data processing was performed in a custom-made script using functions presented on open source toolbox Homer2 39 for MATLAB (V. R2022a) and followed the recent guidelines recommendations for posture and gait research on fNIRS. 27 The following steps were performed to obtain the relative oxy-hemoglobin (HbO2) and deoxy-hemoglobin (HHb) concentration to baseline periods: (i) Conversion of raw data (light intensity) to optical density (Homer2 function: hmrIntensity2OD); (ii) Correction for motion artefact using a wavelet filter correction 40 (Homer2 function: hmrMotionCorrectWavelet); (iii) Conversion from optical density to hemoglobin (Hb) concentration applying the modified Beer-Lambert law (Homer2 function: hmrOD2Conc), 41 using a constant differential path length factor of 6; (iv) Visual inspection for long and short channels remotion: channels not presenting physiological response (i.e., heartbeat), or with higher amplitude signal, not representing fNIRS characteristics response, were removed from the final analysis; (v) Application of a 2nd order Butterworth filter (high-pass [0.01 Hz], low-pass [0.09 Hz] and band stop [0.06-0.14 Hz]) to remove instrumented-related artefacts, vascular endothelial regulation, respiration, heart rate, and Mayer waves 27 ; (vi) Filtering of long-channels using the nearest short-separation channel using a 3rd order polynomial regression 42 ; (vii) Grouping of the channels in the respective correspondents areas (i.e., frontopolar- Broadman 10; and dorsolateral pre-frontal cortex—Broadman 9 and 46); (viii) Next, every 5 trials of the 3 conditions were divided into a baseline period (7.5 seconds before the beginning of the audio instruction) and task (5-25 seconds after the command of “start”) (see Figure 1); and lastly (ix) the relative HbO2 (ΔHbO2) and HHb (ΔHHb) concentrations were then obtained by subtracting the average value of the baseline from the average value of the task (Figure 2). 43
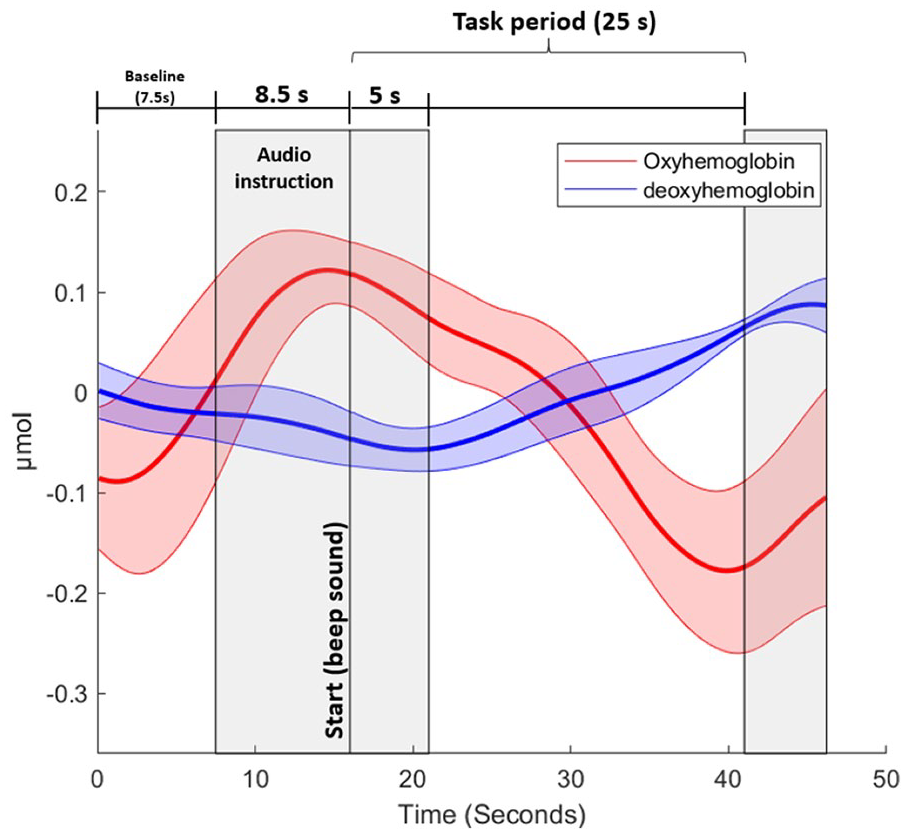
Representation of the periods for fNIRS analysis of 1 healthy control participant. The gray areas were not included in the analysis. The thick line and shadow areas represent the average and standard deviation for oxy-hemoglobin and deoxy-hemoglobin, respectively.
Statistical Analysis
The software SPSS (V. 28.01) was used for statistical analysis. Graphs were made on MATLAB® (V. 2022a) and RStudio (V. 2023.03.0+386). Shapiro-Wilk checked data normality. Independent t-tests and signed Wilcoxon rank tests were used for group comparisons on anthropometric, cognitive, motor, and patient report outcomes. Our statistical analysis was performed as follows:
(i) Two-way repeated measures ANOVA was used to compare groups (pwMS × HC) and conditions (SWT × DT) for the distance walked and gait parameters obtained with the APDM;
(ii) Two-way repeated measures ANOVA was used to compare groups (pwMS vs HC) and conditions (SCT vs DT) for the correct answers;
(iii) The cDTC and mDTC were compared between groups through a Mann-Whitney test;
(iv) fNIRS data were analyzed by 2 repeated measures 2-way ANOVA’s, 1 for each cortical region, with group (pwMS vs HC) and condition (SWT vs SCT vs DT) as factors;
v) Correlation between the relative HbO2 and HHb with behavioral data (meters walked, correct answer, and gait parameters) were performed by the Spearman rank correlation test. The coefficients were interpreted as follows: negligible (0-0.1), weak (0.1-0.39), moderate (0.4-0.69), strong (0.7-0.89), and very strong (0.9-1).
In case of violation in data sphericity, the Greenhouse-Geiser correction was applied in the ANOVA analyses. Bonferroni post-hoc corrections were applied when necessary. The partial eta-square (η2—small > 0.01, moderate > 0.06, or large > 0.14) 44 was used as effect size. The level of significance was set at 0.05.
Results
Descriptive analysis and group comparisons of anthropometric, cognition, mobility, and patient-reported outcomes can be found in Table 1. In general, pwMS presented higher symptoms of fatigue (MFIS physical, mental, psychosocial, and total), and worse performance in all mobility measurements and patients reported outcomes compared with HC (
Average ± SD for Normal Distributed Data and Median [Range] for Non-normal Distributed Data, From Anthropometric, Cognition, Mobility, and Patient-Reported Outcomes.
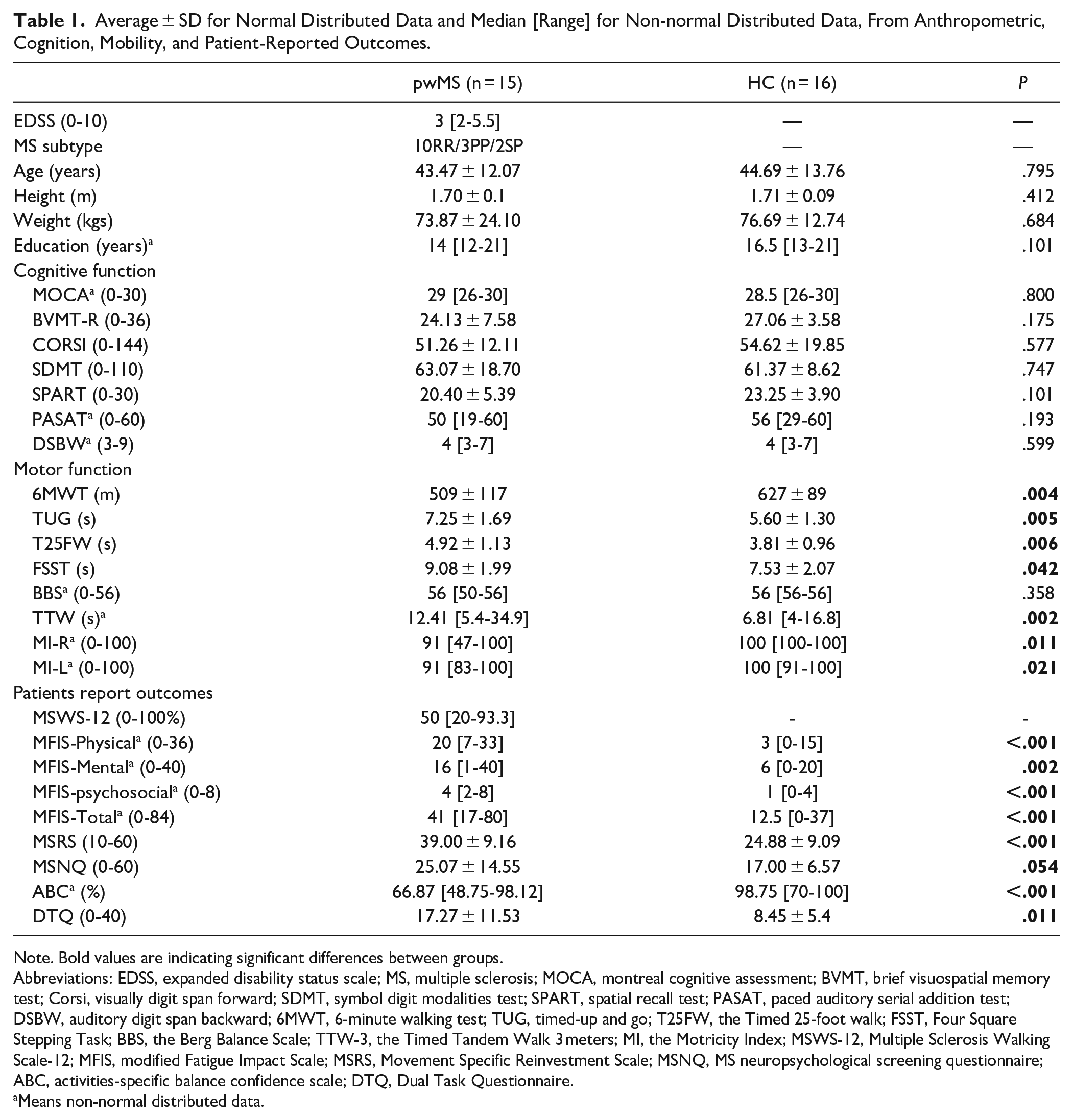
Note. Bold values are indicating significant differences between groups.
Abbreviations: EDSS, expanded disability status scale; MS, multiple sclerosis; MOCA, montreal cognitive assessment; BVMT, brief visuospatial memory test; Corsi, visually digit span forward; SDMT, symbol digit modalities test; SPART, spatial recall test; PASAT, paced auditory serial addition test; DSBW, auditory digit span backward; 6MWT, 6-minute walking test; TUG, timed-up and go; T25FW, the Timed 25-foot walk; FSST, Four Square Stepping Task; BBS, the Berg Balance Scale; TTW-3, the Timed Tandem Walk 3 meters; MI, the Motricity Index; MSWS-12, Multiple Sclerosis Walking Scale-12; MFIS, modified Fatigue Impact Scale; MSRS, Movement Specific Reinvestment Scale; MSNQ, MS neuropsychological screening questionnaire; ABC, activities-specific balance confidence scale; DTQ, Dual Task Questionnaire.
Means non-normal distributed data.
Performance on Single Walking, Single Cognitive and Dual-Task
Behavior and APDM
No statistical significance was observed for the group*condition interaction for the behavior data (distance walked and correct answers). In general, the main effect of group showed that pwMS walked a shorter distance [F1,29 = 11.54,
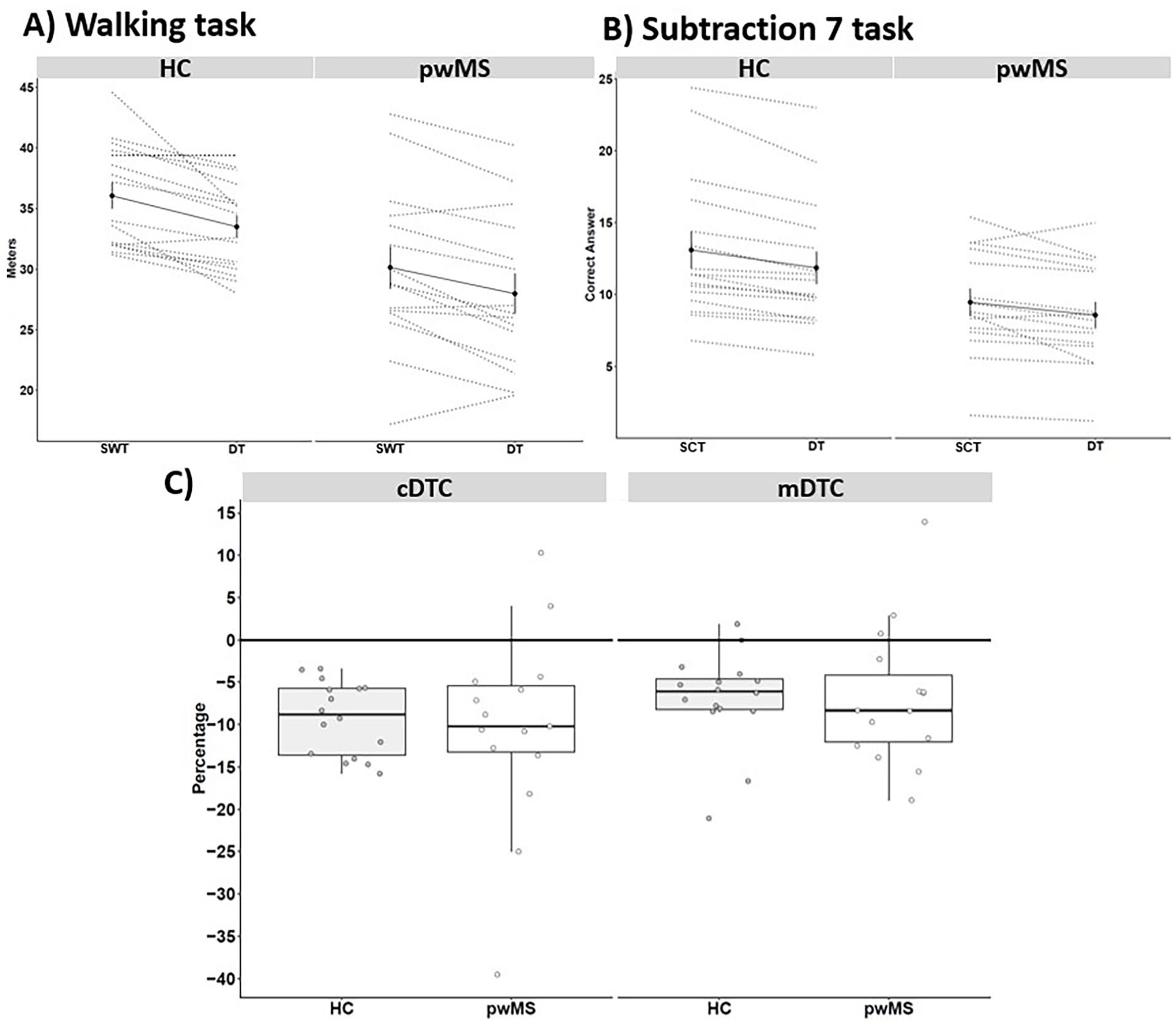
Means and standard error (straight line) and individual data (dotted line), separate by group, for (A) meters walked in the single walking-task and dual-task (DT), (B) correct answers in the simple task-cognitive and DT, and (C) the motor (mDTC) and cognitive (cDTC) DT cost.
Additional gait parameters are presented in Supplemental Table S1. A group*condition interaction showed an increase in step duration variability for HCs in DT condition. In general, pwMS walked slower with shorter stride length, higher double support, and stance phase compared to HC group. The main effect of condition showed a worsening in all gait parameters in the DT compared with the SWT (
Functional Near-Infrared Spectroscopy (fNIRS)
fNIRS data is presented in Figure 4, and the fNIRS data over time can be found in Supplementary Material (Figure S1 and S2).
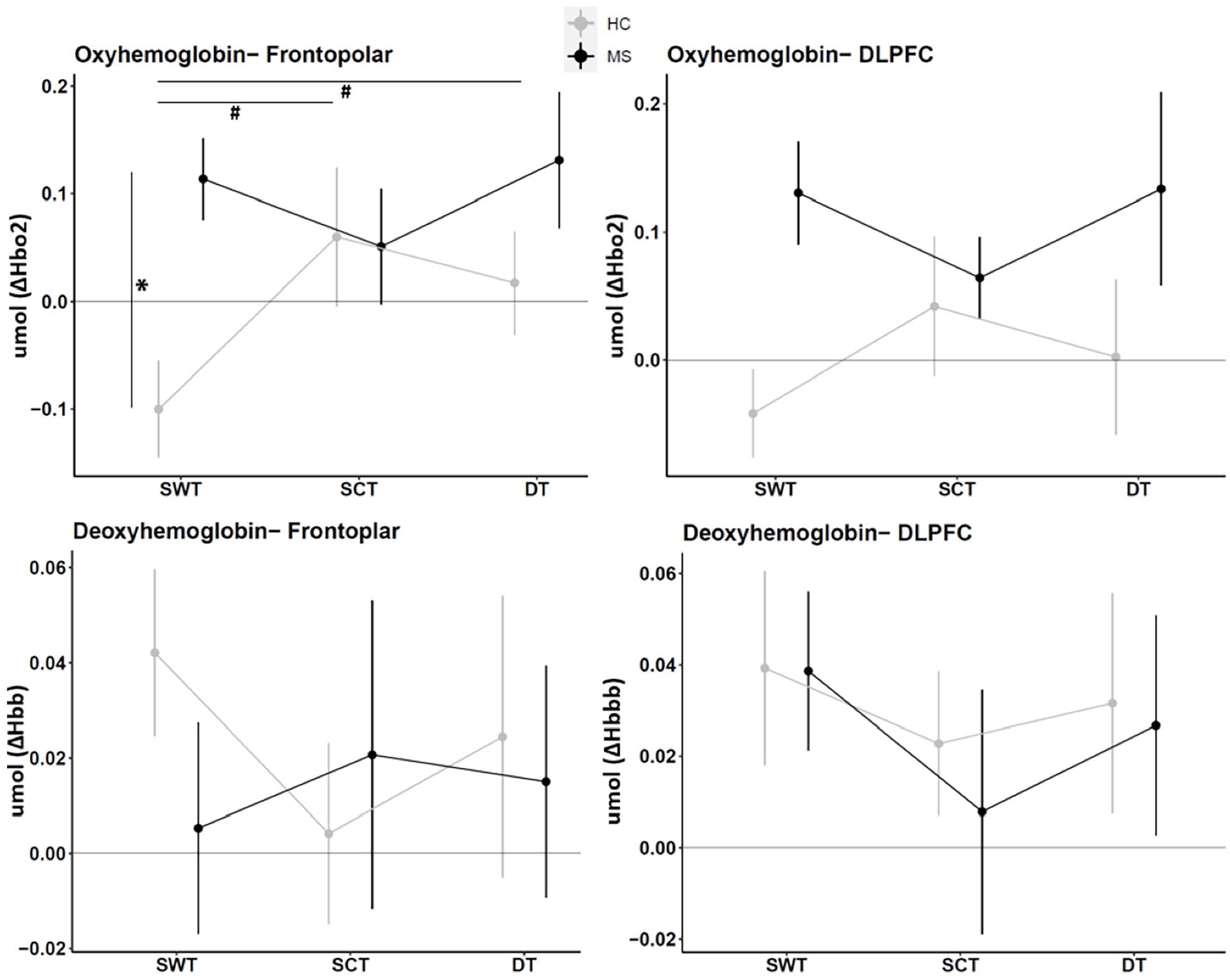
Relative means and standard error of oxy-hemoglobin (ΔHbO2) and deoxy-hemoglobin (ΔHHb) per region of interest (frontopolar—Broadman area 10 and Dorsolateral pre-frontal cortex (DLPFC)—Broadman area 9 and 46), group (people with MS and healthy controls—HC) and condition (Single task-walking, single task-cognition and dual-task (DT)).
For the frontopolar area, a group*condition interaction for the HbO2 was observed [F2,58 = 4.24,
For the DLPFC, no group*condition interaction effect was observed for HbO2 [F2,58 = 1.604,
Correlation Analysis
Spearman rank correlation coefficient between fNIRS data with behavioral data is presented in Figure 5. For pwMS, frontopolar HbO2 was moderately-positively correlated to step duration variability (single walking and dual-task conditions) and stance phase variability (dual-task condition) (
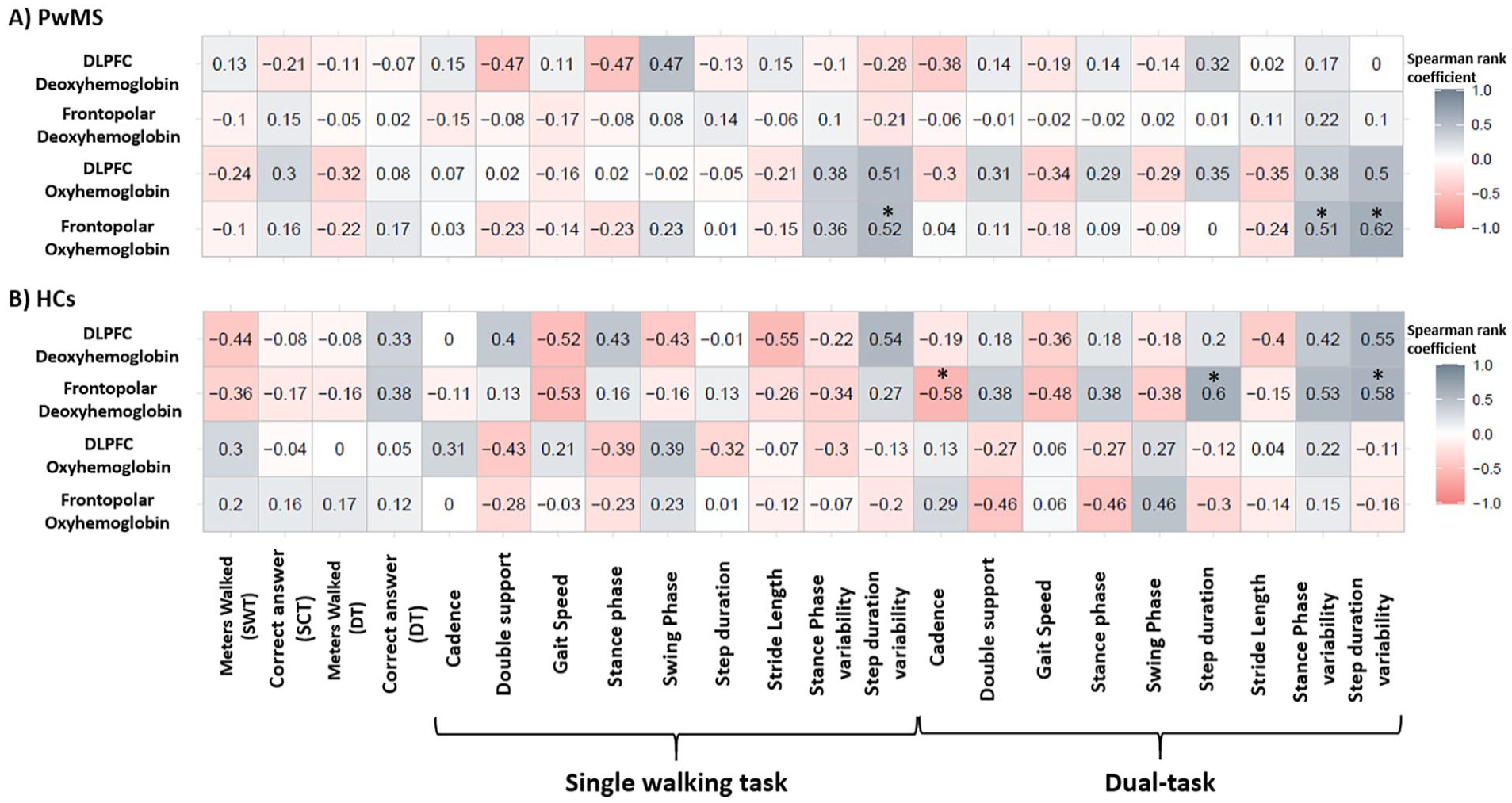
Spearman rank correlation coefficient between pre-frontal cortex (Frontopolar and dorsolateral pre-frontal cortex-DLPFC) and behavioral data and gait parameters in the single walking task, single cognitive task, and dual-task.
Discussion
The present study investigated the role of the PFC by means of examining hemodynamics in the frontopolar and DLPFC regions in 15 pwMS and 16 HC during single walking and cognitive tasks and during dual tasking. Confirming partially our first hypothesis, pwMS presented higher frontopolar activation relative to HC during single walking conditions but not in the single or dual-tasking. In general, pwMS also presented higher DLPFC activation compared to HC. However, only HC exhibited increased cortical activation in frontopolar areas as the SCT and DT compared to SWT supporting our second hypothesis. The mDTC and cDTC were similar for pwMS and HC. However, pwMS walked less distance, presented worse gait parameters, and had less correct answers during single and dual-task conditions, which goes in line with our third hypothesis. Noteworthy, our results and explanations have to be carefully interpreted given our small sample size.
During single walking, our results suggest that pwMS exhibited higher frontopolar activation compared to HC in combination with lower gait speed, and shorter stride length. This result seems to align with previous findings regarding hemodynamics measured using fNIRS17-19 and electroencephalography techniques 10 concomitantly with spatial-temporal gait parameters. 10 Walking control involves 2 pathways: (i) automatic: primary motor cortex, spinal cord, and cerebellum are involved and (ii) executive control: cortical regions (SMA, PFC, PMC) and basal ganglia have an essential role in planning, balance, walking speed modulation, and sensorimotor integration during walking.29,45,46 Damage due to neurological diseases such as MS can shift the walking control from more automatic to more executive control. 7 In our study, there is an indication that pwMS might present an overactivation of the frontopolar cortex during walking, concomitantly to impairments in rhythm, and pace gait domains, supporting the loss in gait automaticity theory.
Furthermore, the current results of our study suggest that pwMS could not recruit additional neural resources to perform more complex tasks, such as dual-tasking during overground walking. This result is contradictory with previous literature on DT in self-paced treadmill walking and older pwMS and in neurological diseases.18,19,23 However, goes in line with Saleh et al, 14 and Kupchenko et al, 15 in pwMS and with Maidan et al, 47 in Parkinson’s disease that used the same DT paradigm as the current study. This can be explained by the fact that pwMS already presented higher use of cognitive resources in the single task (i.e., walking) compared to HC, limiting the ability to recruit additional cognitive resources to perform the DT. 24 In accordance with the capacity sharing model, the brain capacity for task performance is limited, and CMI occurs when this capacity is exceeded during DT performance. Consequently, the performance on one or both tasks will decline.48,49 Nevertheless, the cDTC and mDTC were similar between groups, thus HC presented a similar relative decrease in performance as pwMS during dual tasking compared to single walking, despite increased activation. One possible explanation emerges from the compensatory theory (or CRUNCH hypothesis). 30 It might be that pwMS recruit other brain regions to overcompensate the inefficiency of the frontopolar and DLPFC to respond to the DT, as previously reported in elderly. 23 Unfortunately, we were not able to measure other brain regions given our equipment. Another possible explanation is that the current study was part of a retention session of a study reported elsewhere 33 in which DT were practiced, this might have affected the DT performance of both groups by a learning effect. However, these assumptions are speculative and need further evidence.
The correlation analysis suggested that pwMS presenting higher frontopolar activity also presented higher gait variability in both single walking and DT. Mirelman et al, 50 found similar results during complex walking in healthy older adults but not in young adults and confirmed later by Nobrega-Sousa et al. 51 One plausible explanation is the gait automaticity theory where a less automatic gait (i.e., higher PFC activity) would produce more noise (i.e., the gait is more variable). 7 To our knowledge, no previous studies have investigated PFC correlation with gait variability specifically in pwMS. In contrast, in HC lower cortical activity (by means of HHb) was associated with higher step duration variability. Noteworthy, previous studies reported that HHb is less reliable 52 and presented more noisy compared to HbO2. 53 Therefore, taken this into account and the small sample size for the correlation analysis (for both HCs and pwMS findings), the results should be carefully interpreted.
Noteworthy, our results have clinical implications. For example, pwMS with higher PFC activity in combination with gait impairment in a single walking task might require a rehabilitation program to recover gait automaticity. 24 In addition, pwMS might benefit from complementary therapy (e.g., transcranial direct current stimulation) to improve gait and cortical activity capacity to perform walking and more complex walking tasks. 54
There are methodological considerations. First, the number of participants in our study is relatively small and may help to explain the lack of group*condition interaction for the DLPFC region or the detection of increase from single task to DT in pwMS. In addition, our MS sample was composed of mild-to-moderate pwMS limiting generalization. Future studies can recruit large cohorts covering different levels of disability to understand how disability level can impact neural activity changes during single and DT conditions. Second, previous studies have shown high reliability for the mDTC but low reliability for the cDTC. 55 We can, however, not exclude that findings could be different to some extent when using another cognitive distractor than subtracting sevens. 20 Third, our MS cohort was assessed in a delayed retention session after a session of motor sequence learning of a goal-directed stepping task, 33 with baseline assessment involving walking and the same DT paradigms. As such, a learning effect in performing the conditions may have occurred, 55 which could help to explain the lack of difference in the cDTC and mDTC. Fourth, the data presented here were collected in 2 centers, which might impact the fNIRS data given the potential sensibility of external factors. However, the test conditions between the sites were similar (same researcher, equipment, conditions, and quiet environment). However, as mainly healthy controls were tested at the university department and persons with MS at a clinical center, we cannot exclude, albeit minimal, interference in the fNIRS-generated data.
Conclusion
PwMS presented increased frontopolar cortical activity related to HC during the walking condition, indicating reduced automaticity of walking. HC recruited more cortical resources to respond to the cognitive task and DT conditions. Conversely, pwMS maintained similar cortical activity in the cognitive task and DT conditions, suggesting a limited capacity to recruit more neural resources to respond to more complex tasks. For gait and cognitive performance, in general, pwMS showed worse performance in the single walking and single cognitive tasks and DT compared to HC. However, DTC for motor and cognitive aspects were similar between groups.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241279066 – Supplemental material for Hemodynamics of the Frontopolar and Dorsolateral Pre-Frontal Cortex in People with Multiple Sclerosis During Walking, Cognitive Subtraction, and Cognitive-Motor Dual-Task
Supplemental material, sj-docx-1-nnr-10.1177_15459683241279066 for Hemodynamics of the Frontopolar and Dorsolateral Pre-Frontal Cortex in People with Multiple Sclerosis During Walking, Cognitive Subtraction, and Cognitive-Motor Dual-Task by Felipe Balistieri Santinelli, Renee Veldkamp, Rodrigo Vitório, Daphne Kos, Maxine Vos, Ruth Nijssen, John DeLuca, Cintia Ramari and Peter Feys in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all the participants of the study for collaborating with the advance in the knowledge and the research foundations for their financial support.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R.V. received BOF funding from Hasselt University (#BOF17DOC08). F.B.S received funding from Research Foundation—Flanders (#11O5823N and #K228322N). Charcot Foundation funded the acquisition of the functional Near Infrared Spectroscopy System.
Supplementary material for this article is available on the
