Abstract
Background. Prospective memory (PM) is essential for productive and independent living and necessary for compliance with prescribed health behaviors. Parkinson disease (PD) can cause PM deficits that are associated with activity limitations and reduced quality of life. Forming implementation intentions (IIs) is an encoding strategy that may improve PM in this population. Objective. To determine the effect of IIs on PM performance in PD. Methods. This was a laboratory-based randomized controlled trial. Participants with mild to moderate PD without dementia (n = 62) performed a computerized PM test (Virtual Week) under standard instructions. One week later they were randomly allocated to perform it again while using either IIs or a rehearsal (RR) encoding strategy. Results. PM performance was better with the use of both strategies relative to standard instructions. This effect was larger for tasks with event-based compared with time-based cues. In addition, IIs resulted in a larger effect than RR for the nonrepeated tasks. Conclusions. Strategies that support full encoding of PM cues and actions can improve PM performance among people with PD, particularly for tasks with cues that are readily available in the environment. IIs may be more effective than RR for nonrepeated tasks, but this finding warrants verification. Future work should address transfer of strategy use from the laboratory to everyday life. Targeted strategies to manage PM impairment could improve function and quality of life and significantly affect clinical care for people with PD.
Introduction
Cognitive impairment is a well-established feature of Parkinson disease (PD) without dementia and is associated with activity limitations, reduced quality of life, and restricted participation.1-6 Prospective memory (PM) has received increasing attention in PD research over the past decade because it is a highly functionally, clinically, and theoretically relevant aspect of cognition.7,8 PM is the ability to remember to execute delayed intentions at the appropriate moment in the future. In time-based PM tasks, a certain time or the passage of a specified amount of time serves as the cue that signals the appropriate moment for execution. In event-based PM tasks, the occurrence of an event serves as the cue that signals the appropriate moment for execution. Examples of everyday time-based PM tasks include remembering to attend a meeting at 3:00
Successful PM performance depends on the ability to formulate and plan an intention (intention formation), retain its contents in long-term memory over a delay while performing other unrelated tasks (intention retention), recognize when the appropriate moment occurs for it to be carried out and retrieve its details from memory (intention retrieval), and finally, execute it (intention execution). 12 This multiphase process requires the integration of episodic memory processes and executive or attentional control processes, such as planning, working memory, and cognitive flexibility, 12 all of which can be impaired in PD.13-16
PM impairment in PD is thought to stem from deficits in intention formation and intention retrieval. 12 Whereas retention of well-formed intentions and execution of intentions once they are retrieved are fairly intact in PD, encoding, planning, and/or retrieval of intentions can be impaired, particularly under conditions of high executive control demand.9,10,17-19 This impairment is attributed to frontostriatal circuitry dysfunction caused by dopamine depletion in the prefrontal cortex and basal ganglia. 12 For example, Kliegel et al 19 found that PD participants formed less-elaborate plans for accomplishing a complex intention compared with healthy older adults and, subsequently, were less likely to initiate the intention at the appropriate moment. In another study, PD participants had poorer PM for intentions that required self-initiated encoding at intention formation relative to those for which encoding was externally guided. 17 In terms of intention retrieval, PM tasks with cues that are not integral to performing the ongoing activity (eg, time-based tasks) and require strategic monitoring of the environment are impaired in PD.10,17,18,20,21 In contrast, PM tasks with cues that are integrated into the processing of the ongoing activity (eg, some event-based tasks) and can be processed relatively automatically are not impaired in PD.17,22 However, although intention retrieval in PD may be supported by features that facilitate automatic processing of PM cues, deficient intention formation can preclude this benefit. 17
These findings indicate that suboptimal intention formation is a key barrier to successful PM performance in PD and suggest that a PM intervention for PD should focus on improving intention formation, one aspect of which is encoding. Indeed, evidence from retrospective and PM studies implies that although people with PD do not self-initiate effective encoding strategies, they can make use of externally guided encoding to improve their performance.17,23-25 Thus, a cognitive rehabilitation approach that teaches specific PM encoding strategies may improve PM in PD. The formation of implementation intentions (IIs) 26 is a method of encoding and planning intentions that was originally designed to facilitate goal attainment and has since been applied to PM. The strategy involves specifying and stating aloud the circumstances under which one will carry out an intention (“When X, I will do Y”; eg, “When I eat dinner, I will take my medication”) and visualizing oneself encountering those circumstances and executing the intention. By forcing elaborate and specific encoding, IIs are thought to heighten the accessibility of PM cues and strengthen the association between PM cues and their intended actions, thereby facilitating more automatic cue detection and intention retrieval.26-30 Of relevance to PD, IIs provide an explicit structure for good associative encoding of intentions that may compensate for the PD-related deficit in internally generated intention formation strategies. This then should reduce the need for controlled intention retrieval processes (which are impaired in PD) by fostering reliance on more automatic retrieval processes (which are spared in PD).10,12,30
There is evidence for the beneficial effect of IIs on PM performance in healthy older adults,
31
stroke,
32
multiple sclerosis,
33
and very mild Alzheimer’s disease.
34
To our knowledge, this strategy has not been tested in PD. The purpose of this study was to investigate the effect of IIs on PM performance in PD. We used Virtual Week (a computerized board game that mimics everyday life PM tasks) to assess PM because it is reliable and sensitive in PD and, importantly, for present purposes, allows the analysis of different PM task types (repeated, nonrepeated) and cues (event, time).17,35,36 Repeated tasks are those that occur multiple times throughout the game (eg, take antibiotics each day at breakfast), whereas nonrepeated tasks occur only once (eg, get a haircut at 1
We hypothesized that an instructed encoding strategy (IIs, RR) would be associated with greater gains in event-based compared with time-based PM performance. We reasoned that strategic encoding of the PM cue and the intended action would be of less value for the time-based tasks, for which detection of the PM cue presumably requires strategic monitoring. That is, we would not expect strategic encoding of intentions to obviate the need for strategic monitoring in time-based tasks; thus, impaired monitoring in PD would still interfere with time-based PM task performance.
In addition, we anticipated that IIs would be particularly beneficial relative to RR for the nonrepeated PM tasks. Repeated tasks are reinstructed on each virtual day and, thus, receive multiple encodings. In contrast, the nonrepeated tasks are presented for encoding only once and amid other PM tasks. Here, the encoding challenges are high, and thus, the advantage of a mnemonically superior strategy (IIs) should be especially important.
Methods
This study was approved by the university’s human research protection office, and all participants gave written informed consent.
Participants
Participants were community-dwelling volunteers with PD recruited from the university’s movement disorders center. Inclusion criteria were the following: at least 50 years of age, diagnosed with idiopathic PD, 37 and classified as Hoehn and Yahr stage I to III. 38 Exclusion criteria included suspected dementia (determined by physician or caregiver report or Mini Mental Status Exam score <27), 39 medications that interfere with cognitive function (eg, anticholinergics, tricyclic/tetracyclic antidepressants), change in medication over the course of the study, other neurological disorders, history of brain surgery, significant psychiatric conditions, or any other features that would interfere with study participation (eg, non–English speaking).
Design
This was a randomized controlled trial (NCT01469741). Participants performed a computerized PM test on enrollment (Virtual Week). One week later, they returned to the laboratory and were randomly assigned to an encoding strategy group—IIs or RR—stratified by sex and age (±62 years). Participants were then taught their respective encoding strategy and used it while performing a parallel version of Virtual Week.
Assessment
Assessment was conducted at the university while participants were on their regular antiparkinsonian medications. Participants’ testing sessions were scheduled for the same time of day to control for potential dosage timing effects within subject. During the baseline testing session, participants provided demographic information and completed the Montreal Cognitive Assessment 40 to assess global cognition and the Beck Depression Inventory II 41 to assess depressive symptoms. Clinical characteristics related to PD (eg, Unified Parkinson’s Disease Rating Scale Motor Scale score from within 3 months of testing, 42 Hoehn and Yahr stage, disease duration, medications) were accessed through clinical records.
Primary Outcome Measure: Virtual Week
A computerized version of the board game Virtual Week was used to measure PM35,36,43 (see the appendix). At each testing session, participants first completed a practice day during which detailed automated messages and the experimenter explained the game. Then, they completed 3 test days (Monday, Tuesday, Wednesday). Two equivalent versions of the test days were counterbalanced across testing session to reduce practice and order effects. Participants played the game on a desktop computer, using the mouse to interact with the software. They moved their token around the board on the screen by clicking a die in the middle of the board and clicking the corresponding square of the board. One circuit around the board represented 1 day (7:00
Each day had 8 embedded PM tasks: 4 repeated and 4 nonrepeated tasks. The repeated tasks were health-related tasks that were repeated every day, and the nonrepeated tasks were different each day. In this version of the game, the repeated tasks did not receive enhanced encoding at the onset of the game (as in Foster et al
17
) but instead were administered at the beginning of each day similar to the nonrepeated tasks. Half of the repeated and nonrepeated tasks each day were cued by Event Cards (event based), and half were cued by the virtual time of day displayed on the clock in the middle of the board (time based). Thus, the event-based tasks had cues that were integrated into the ongoing activity of playing the game, whereas the time-based task had cues that required monitoring for information that was not integrated into playing the game. The repeated event-based tasks were, “Take antibiotics at breakfast and dinner,” and the repeated time-based tasks were, “Take asthma medication at 11
Intervention
For the first testing session, all participants completed Virtual Week under standard instructions. For the second testing session 1 week later, the practice day incorporated encoding strategy training and practice. Participants in the II group were told that each time they encountered a PM task, they should create a “When X, I will do Y” statement, repeat the statement out loud 3 times, and close their eyes and visualize themselves performing the task at the appropriate moment within the context of the game. Those in the RR group were told to repeat the administered PM tasks out loud 3 times but were given no specific instructions on how to do so. During the test days, automated messages (and, if necessary, the experimenter) reminded participants to use their strategy when PM tasks were administered (see the appendix). In addition, for the second session, the game was programmed to display the PM tasks on the screen for at least 30 s before allowing participants to continue. These features ensured that participants used the strategy they were taught and controlled for time spent on the PM tasks across conditions.
Sample Size Determination
In a pilot study, 12 PD participants completed Virtual Week under standard instructions during one testing session and then returned to the laboratory 1 to 3 weeks later to complete it a second time either while using IIs (n = 6) or under standard instructions (n = 6). There was a large between-group effect in favor of the II group at the second testing session (II M = 0.75, control M = 0.50, pooled SD = 0.28; d = 0.89). A sample size of 20 participants per condition was estimated to detect such an effect with α = .05 and 80% power. Because our pilot study did not use an active control condition, we increased our target sample size for the current study to 30 participants per group and recruited 68 to account for potential attrition. Of relevance to the current results, there was no difference in Virtual Week performance between testing sessions for the control group (ie, no apparent practice or learning effect). Furthermore, a test-retest study of Virtual Week with standard instructions in older adults that used the same counter-balanced parallel versions as the current study also showed no practice or learning effect. 44
Analysis
Data were stored and managed using REDCap electronic data capture tools 45 and analyzed with IBM SPSS Statistics 22. Descriptive statistics were calculated for all variables, and independent samples t-tests and χ2 tests were used for group comparisons of demographic and clinical characteristics. To determine the effect of strategy use on PM performance, proportions of correct PM responses were submitted to a 2 × 2 × 2 × 2 mixed ANOVA with the between-group variable encoding strategy group (II, RR) and within-group variables PM task (repeated, nonrepeated), PM cue (event, time), and time of assessment (T0, T1). Interactions were followed up with ANOVA and pairwise comparisons. All statistical tests were 2-tailed. An α level of P < .05 was considered significant. Effect sizes were estimated using partial ηp2 and Cohen’s d.
Results
Participant Characteristics
A total of 62 participants (n = 31 per group) had usable data for this study (Figure 1). The II and RR groups were equivalent on all demographic and clinical characteristics (Table 1). Antiparkinsonian medication regimens included levodopa-carbidopa only (16 II, 18 RR); levodopa-carbidopa with a dopamine agonist, COMT inhibitor, or both (11 II, 11 RR); dopamine agonist only (1 II, 0 RR); MAO inhibitor only (1 II, 0 RR); and no antiparkinsonian medications (2 II, 2 RR) and did not differ between groups; χ2 = 2.84, P = .83.
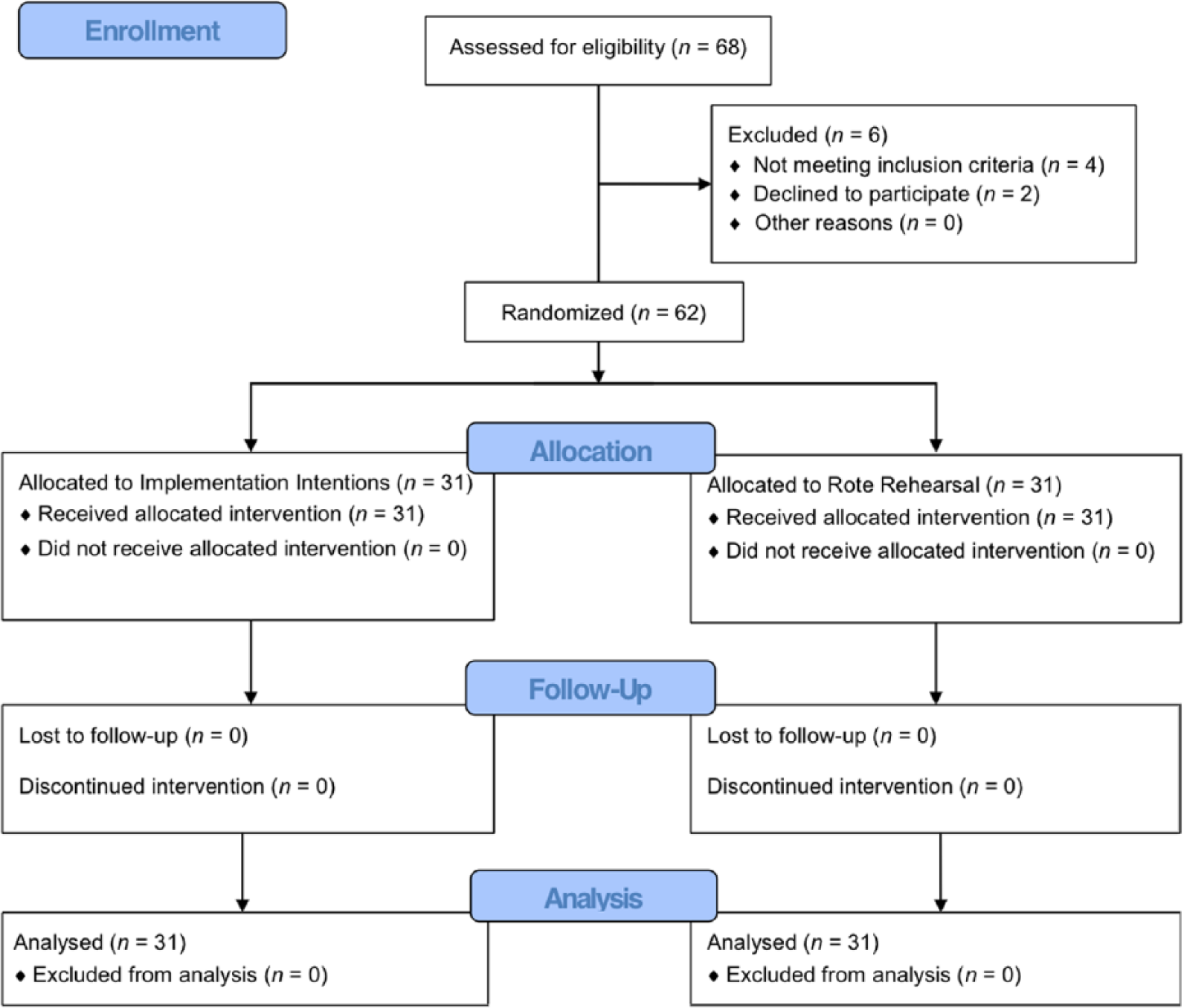
CONSORT flow diagram illustrating participant recruitment, randomization, and attrition.
Demographic and Clinical Characteristics of the Sample (n = 62). a
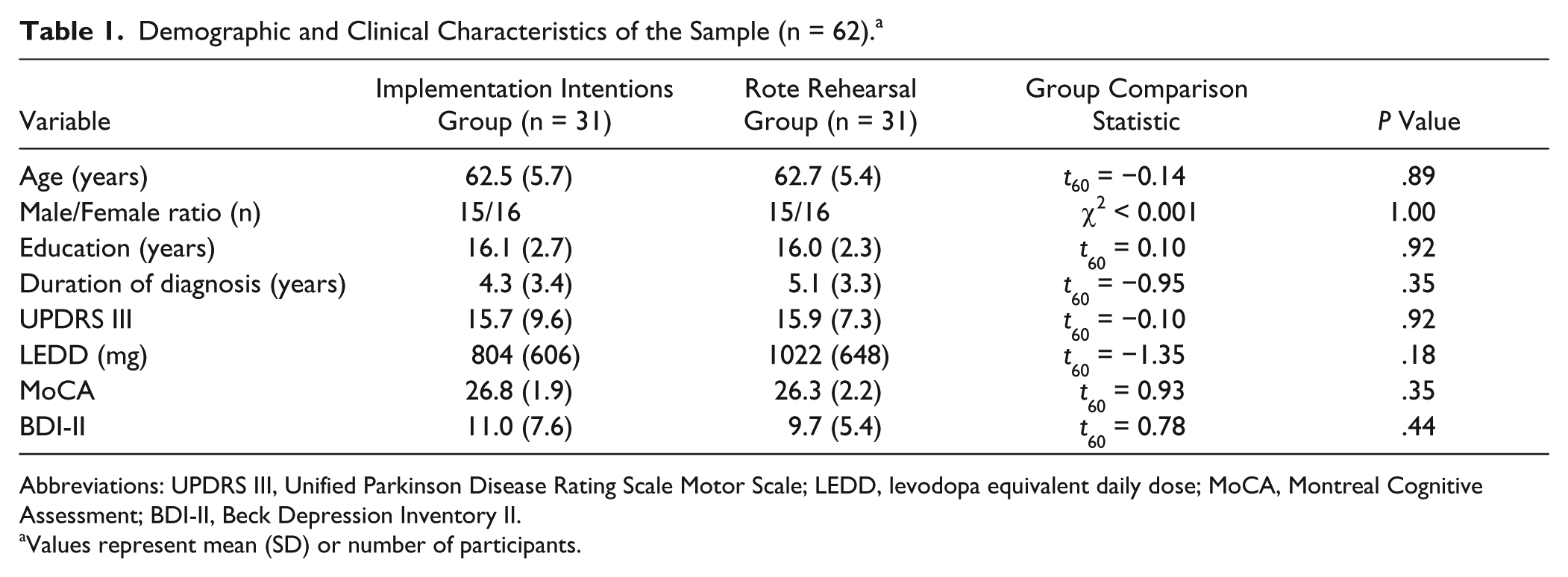
Abbreviations: UPDRS III, Unified Parkinson Disease Rating Scale Motor Scale; LEDD, levodopa equivalent daily dose; MoCA, Montreal Cognitive Assessment; BDI-II, Beck Depression Inventory II.
Values represent mean (SD) or number of participants.
Effect of Encoding Strategy on PM Performance
Proportions of correct PM responses are presented in Table 2, and the initial ANOVA results are in Table 3. Overall, performance was better for repeated tasks, event cues, and at T1 (with strategy use) compared with nonrepeated tasks, time cues, and at T0 (baseline, without strategy use), respectively; Fs ≥ 23.54, Ps < .001, ηp2 ≥ 0.28. There was an interaction between PM cue and time of assessment—F(1, 60) = 3.96, P = .05, ηp2 = 0.06, such that event-based tasks showed a larger improvement at T1 than time-based tasks. There was a 3-way interaction between PM task, group, and time of assessment: F(1, 60) = 7.55, P = .008, ηp2 = 0.11. Group did not interact with any other variable.
Group Mean (SD) Proportion of Correct Prospective Memory Responses for Each Task Type Without and With Strategy Use. a
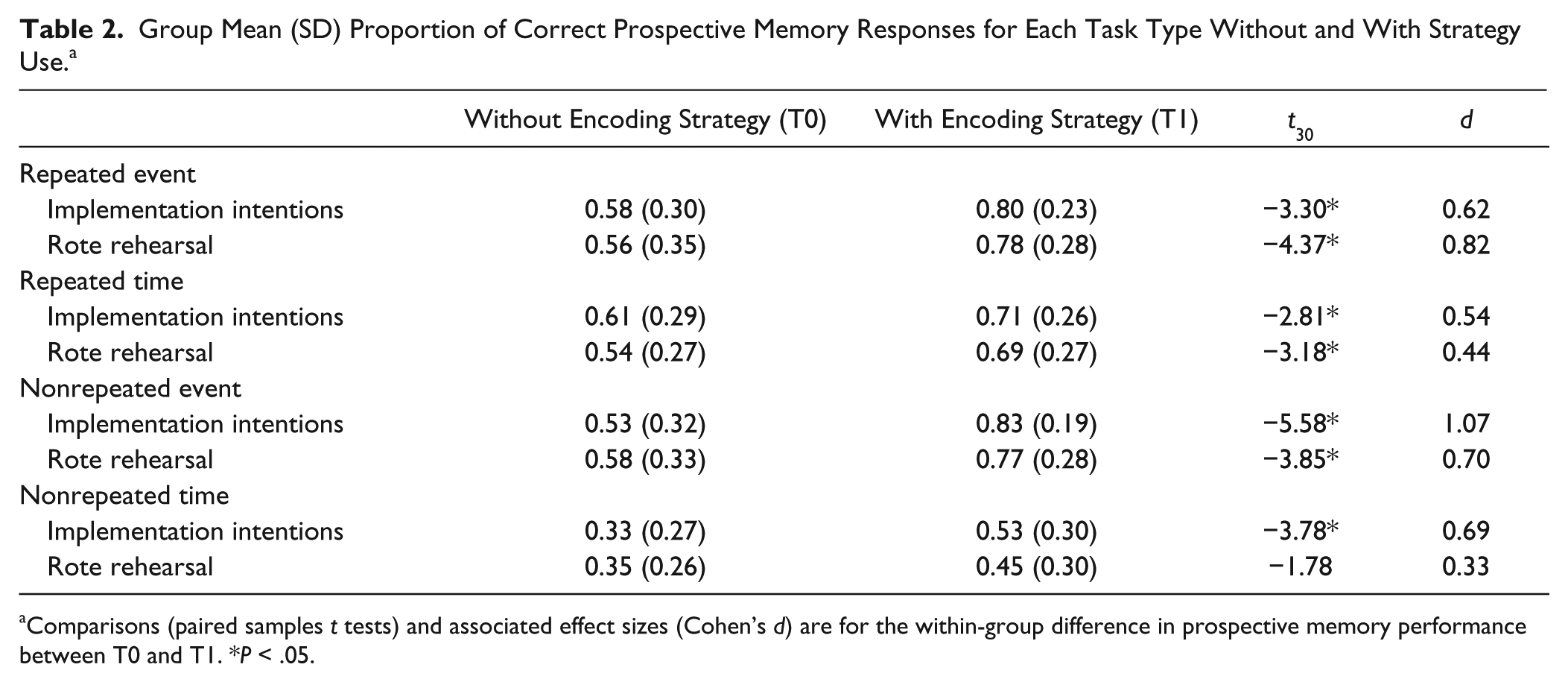
Comparisons (paired samples t tests) and associated effect sizes (Cohen’s d) are for the within-group difference in prospective memory performance between T0 and T1. *P < .05.
Results From the 2 × 2 × 2 × 2 Mixed ANOVA. a
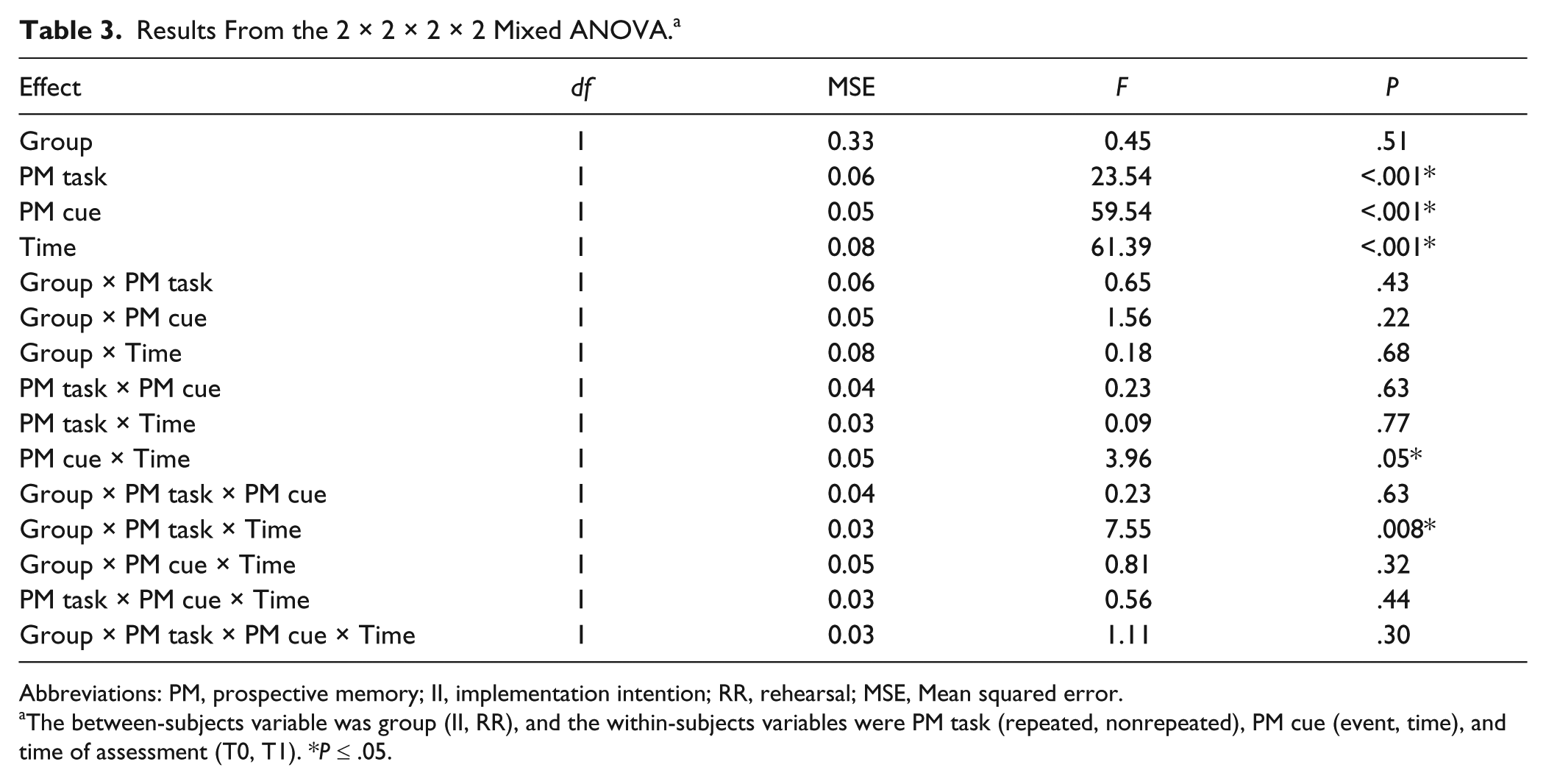
Abbreviations: PM, prospective memory; II, implementation intention; RR, rehearsal; MSE, Mean squared error.
The between-subjects variable was group (II, RR), and the within-subjects variables were PM task (repeated, nonrepeated), PM cue (event, time), and time of assessment (T0, T1). *P ≤ .05.
To follow up the 3-way interaction, separate 2 (group) × 2 (time of assessment) ANOVAs were conducted for nonrepeated and repeated tasks. On nonrepeated tasks, there was an effect of time of assessment—F(1, 60) = 47.29, P < .001, ηp2 = 0.44—such that performance was better at T1. There was also a marginally significant interaction between group and time of assessment—F(1, 60) = 3.29, P = .08, ηp2 = 0.05—such that the II group had a larger improvement at T1 than the RR group (II d = 1.02, RR d = 0.59; Figure 2). On repeated tasks, there was an effect of time of assessment—F(1, 60) = 40.62, P < .001, ηp2 = 0.40—such that performance was better at T1, but there was no interaction between group and time of assessment: F(1, 60) = 1.21, P = .28, ηp2 = 0.02.
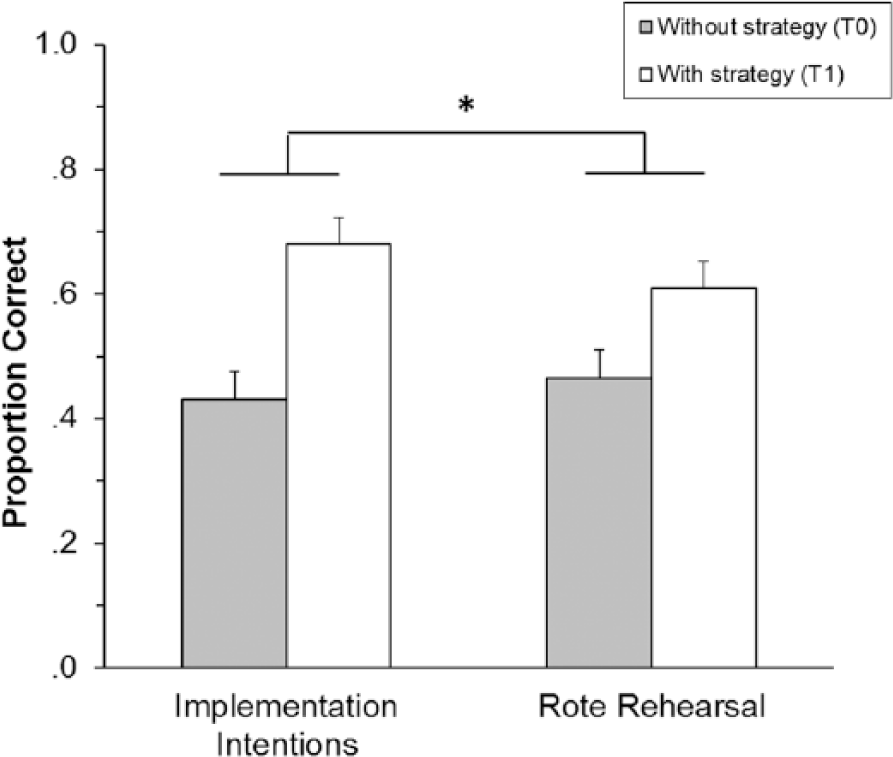
Mean proportion of correct prospective memory responses on nonrepeated tasks as a function of encoding strategy group and time of assessment. Error bars depict standard error of the mean (*Group × Time of assessment; P = .08).
Discussion
This study tested the effect of encoding strategies on PM performance in individuals with PD but without dementia. Specifically, we aimed to determine the types of PM tasks for which various encoding strategies would benefit PD individuals. We also were interested in whether a mnemonically enhanced encoding strategy (IIs) would produce greater improvements in PM performance for PD individuals than a typically less-effective encoding strategy (RR). We used the Virtual Week PM test, which includes repeated and nonrepeated PM tasks cued by events or time. PD participants were randomly assigned to perform Virtual Week under standard instructions (T0) and also while using either the implementations intentions or RR encoding strategy (T1). Both strategies improved PM performance relative to standard instructions, particularly for tasks cued by events. In addition, IIs resulted in a larger effect than RR, but only for nonrepeated tasks.
Our data are consistent with the view that poor executive control of intention formation—namely, poor self-initiated strategic encoding—is a key cognitive mechanism underlying PM impairment in PD. 12 Previous studies have suggested this by showing that people with PD naturally form less-elaborate intentions and are then less likely to initiate those intentions than their healthy peers 19 but have better PM performance when external testing conditions facilitate better encoding of intentions.17,23 This study expands on previous work to demonstrate that when people with PD use explicit encoding strategies, their PM performance improves substantially, especially for event-based PM tasks. It provides support for cognitive rehabilitation approaches that train people with PD to use PM encoding strategies.
As predicted, the encoding strategies were more effective for event-based compared with time-based tasks. The event-based tasks were cued by specific Event Cards that appeared throughout the day and that the person interacted with to play the game. In contrast, the time-based tasks required the person to periodically disengage from the game to check the clock in the middle of the board. Thus, whereas event cues were processed as a part of the ongoing activity, time cues required the deployment of strategic attentional resources (ie, monitoring the virtual time of day, which involves internally driven shifting of attention from the ongoing activity) to be processed. Our results support the notion that specification and repetition of PM intentions during encoding heightens perceptual readiness for and facilitates detection of cues encountered in the environment26,30—in this case, the event cues. However, heightened cue accessibility would not facilitate detection of time cues in the absence of strategic monitoring (or shifting) because those cues would not be encountered. Some evidence suggests that IIs increase monitoring for nonfocal PM cues
46
(cues that are not processed as a part of the ongoing activity), which may explain the improvement in time-based tasks; however, consistent with other studies, our findings indicate that this mechanism is less robust than the automatic processing facilitated by IIs for tasks with focal event cues
31
(cues that are processed as a part of the ongoing activity). Direct assessment of monitoring by recording time checks would have helped confirm this explanation and should be considered for future studies. Regarding PM intervention, these results suggest that in addition to the provision of intention formation strategies, people with PD may need support to enhance their monitoring for time cues. Alternatively, a more effective approach could be to teach them to associate intentions with externally available cues that do not require monitoring (essentially turning time-based tasks into focal event-based tasks; eg, feed the dog when you turn on the evening news rather than at 5:00
More novel was that IIs tended to produce greater gains than RR for PM tasks with challenging encoding conditions: the nonrepeated tasks, which were instructed only once and amid other PM tasks. In fact, IIs produced nonrepeated task performance in PD participants in the current study that was better than that of a healthy older adult group from a previous Virtual Week study. 17 Thus, IIs presumably compensated for PD-related difficulties with intention formation and substantially improved PM performance for these difficult tasks that arguably are often present in the lives of older adults—one-off PM tasks that are encoded along with other tasks the adult has to perform during the day.
This pattern is consistent with, though possibly not as robust as, past findings with non-PD patients that IIs are superior to RR for nonrepeated tasks, 33 purportedly because they force specification of the PM cue and intended action rather than allowing one to simply state the intention (“I will Y”), which could occur with RR. Still, this finding is suggestive rather than definitive because it was only of marginal statistical significance.
The absence of an advantage of IIs (relative to RR) for repeated tasks suggests that repeated encoding reduces encoding challenges for the PM task, so that any explicit strategy, even RR, is sufficient for PD. Alternatively, this could have stemmed from overlap in the application of the 2 strategies in the context of this particular experimental paradigm. In Virtual Week, PM task administration specifies the PM cue and intended action. Although RR participants were not explicitly trained to form “When [cue], I will [action]” statements, their RRs would have involved coverbalization of the cue and action if they were repeating the information provided to them. In this way, RR may have facilitated cue accessibility and strengthened associative encoding to a similar degree as IIs.
The neural mechanism of PD-related PM impairment has not been studied directly but is often attributed to disruption of prefrontal cortical regions responsible for the executive control of intention retrieval.10,12,18 In contrast, the hippocampal networks thought to underlie more automatic intention retrieval are relatively spared in PD.47,48 This aligns with the proposed mechanism of IIs, which is that they promote a shift from controlled to automatic processing. Specifically, they allow intention retrieval to occur in a reflexive, stimulus-driven fashion rather than require self-initiated retrieval processes.26,30 This notion is supported by a functional magnetic resonance imaging study showing that IIs shifted brain activity from a region associated with top-down control of PM processing (lateral BA 10) to one associated with bottom-up PM responding (medial BA 10). 49 However, BA 10 (the region most consistently implicated in PM studies 50 ) is not one of the regions directly disrupted by frontostriatal circuitry dysfunction in PD. Thus, the underlying neural mechanisms of PM impairment and recovery in PD are unclear and warrant further investigation.
We designed this study to examine the potential benefits of encoding strategies (IIs and RR) on PM in PD, but there are some issues that limit our conclusions. We cannot rule out the potential effect of practice; however, it is unlikely to have caused the observed pattern of improvement in PM performance. First, there is no reason that practice alone would be more beneficial for event-based compared with time-based tasks. Instead, we contend that the larger improvement on event-based tasks was a result of enhanced encoding of the PM cue and associated intention, thereby allowing environmental (event) cues and their associated actions to be more automatically detected and retrieved. Second, if practice was a major driver of improvement, then performance on repeated tasks, which were repeated within and across testing sessions, should have increased proportionately more than performance on nonrepeated tasks, but this was not the case. In addition, the limited differentiation between IIs and RR could have been a result of insufficient power. Our pilot study found a larger between-group effect of IIs compared with standard instructions and, similar to a test-retest study of Virtual Week, no practice effect with standard instructions. 44 We increased our sample size to account for the use of an active control condition, but our estimate may have been inadequate. A larger study with a no-strategy control condition would help address these limitations and substantiate our conclusions regarding the relative effects of IIs and RR.
This study is the first to evaluate a cognitive strategy training intervention for PM in PD. PM is essential for productive and independent living and necessary for compliance with prescribed health behaviors (eg, taking medications, keeping therapy appointments, performing home exercises). Targeted strategies that enable people with PD to successfully perform PM tasks could improve function and quality of life and significantly affect clinical care for this population. We have demonstrated that when people with PD use simple encoding strategies at intention formation, they can improve their performance on a variety of PM tasks in a laboratory setting and that such strategies may be most helpful for tasks with cues that are readily available for processing in the environment (event-based tasks). The specific strategy of IIs may be particularly effective for nonrepeated PM tasks, but further work is required to verify this finding. This provides a valuable starting point for research on PM strategy training in PD and cognitive rehabilitation approaches for PM impairment in PD. Additional work is required to directly inform clinical application. A next step is to understand whether—or how training should be structured so that—people with PD can independently initiate the use of intention formation strategies to support their PM performance. Future studies should also address the degree to which strategy use and effectiveness transfer to people’s real-world PM tasks.
Footnotes
Appendix
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by NIH K23HD071059 and UL1TR000448 (Washington University Institute of Clinical and Translational Sciences) and the Advanced Research Center of the Greater St Louis Chapter of the American Parkinson Disease Association.
