Abstract
Background
Concussions result in transient symptoms stemming from a cortical metabolic energy crisis. Though this metabolic energy crisis typically resolves in a month, symptoms can persist for years. The symptomatic period is associated with gait dysfunction, the cortical underpinnings of which are poorly understood. Quantifying prefrontal cortex (PFC) activity during gait may provide insight into post-concussion gait dysfunction. The purpose of this study was to explore the effects of persisting concussion symptoms on PFC activity during gait. We hypothesized that adults with persisting concussion symptoms would have greater PFC activity during gait than controls. Within the concussed group, we hypothesized that worse symptoms would relate to increased PFC activity during gait, and that increased PFC activity would relate to worse gait characteristics.
Methods
The Neurobehavior Symptom Inventory (NSI) characterized concussion symptoms. Functional near-infrared spectroscopy quantified PFC activity (relative concentration changes of oxygenated hemoglobin [HbO2]) in 14 people with a concussion and 25 controls. Gait was assessed using six inertial sensors in the concussion group.
Results
Average NSI total score was 26.4 (13.2). HbO2 was significantly higher (P = .007) for the concussed group (0.058 [0.108]) compared to the control group (−0.016 [0.057]). Within the concussion group, HbO2 correlated with NSI total symptom score (ρ = .62; P = .02), sagittal range of motion (r = .79; P = .001), and stride time variability (r = −.54; P = .046).
Conclusion
These data suggest PFC activity relates to symptom severity and some gait characteristics in people with persistent concussion symptoms. Identifying the neurophysiological underpinnings to gait deficits post-concussion expands our knowledge of motor behavior deficits in people with persistent concussion symptoms.
Keywords
Introduction
A concussion results in transient cognitive, somatic, and affective symptoms that typically resolve within a month. Unfortunately, up to 53% of concussed individuals can experience these symptoms up to a year post-concussion. 1 Concussion symptoms are linked to a metabolic energy crisis, which stems from the need to re-establish neurophysiological homeostasis at the neuronal level following a concussion. 2 Despite the typical metabolic recovery timeline being about a month, traditional neuroimaging studies (ie, magnetic resonance imaging and transcranial magnetic stimulation) identified cortical microstructural and neurophysiological differences between those with and without a concussion, differences that can persist for years after the concussion.2-7 Beyond symptomology, people with a concussion experience gait impairment, more consistently observed when concussion symptoms are present.8,9 However, the effects of the observed neurophysiological differences have on gait performance post-concussion are limited.
Concussion-related neurophysiological changes have primarily been quantified in the frontal lobe at rest or during a cognitive assessment. Results from these studies suggest there is a task-specific increased activation of the prefrontal cortex (PFC) in the concussed individual.10,11 Not only did increased PFC activation persist for up to 9 months, especially in the symptomatic group, connectivity between the PFC and parietal cortex remained impaired in the concussed group.11,12 Traditional neuroimaging modalities that identify neurophysiological effects of concussion on motor performance are limited to quantifying neurophysiology independent of the motor task performance.13,14 Traditional neuroimaging techniques are sensitive to motion artifact, so any motor tasks need to limit the amount of movement during neuroimaging procedures. Though imperfect, these neuroimaging-motor studies uncover a relationship between worse motor performance and impaired neurophysiology after concussion.5-7,13,14 While these results indicate an associative or indirect effect of concussion on motor performance, only quantifying cortical activity during motor tasks will elucidate direct effects.
Gait is adversely affected in the concussion population, the cortical underpinnings of which are poorly understood. The evidence for gait impairments is supported by decades of research that suggest subtle gait instability exists following a concussion. People with a concussion exhibit impaired gait characteristics for as long as 6 years after concussion.8,15,16 People with a concussion exhibit slower gait and decreased stability during the symptomatic phase of a concussion, as well as during the asymptomatic phase.8,17,18 A recently developed model of gait in the concussed identified domains of pace, rhythm, and variability as explaining 63.4% of the variance in gait among this population. 19 Within the pace, rhythm, and variability gait domains, the stride-based metrics and gait speed were among the strongest gait characteristics in the concussion gait model. 19 Whether individual gait characteristics (eg, gait speed, stride time, and center of mass movement) or gait domains (eg, pace and variability) are quantified, people with a concussion exhibit slower, more variable, and less stable gait than those without a concussion.8,9,17-19 The neurophysiological underpinnings for the established gait impairments in people with a concussion remain poorly understood.
A next step in understanding the cortical neurophysiological underpinnings of gait impairment in the concussed population requires mobile neuroimaging techniques, such as electroencephalography (EEG) and functional near-infrared spectroscopy (fNIRS). Like the MRI-based neuroimaging studies, the EEG and fNIRS neuroimaging literature is dominated by cognitive and simple motor tasks.20-25 Studies using mobile neuroimaging demonstrate altered cortical activity during cognitive and balance assessments in people with a concussion.26-29 Altered cortical activity measured by EEG during quiet standing related to poorer balance metrics in asymptomatic concussed individuals.30,31 Similarly, fNIRS protocols highlight altered frontal cortex activity, including increased activity in the PFC, motor cortex, and parietal cortex during gait and balance tasks.27-29,32 Specifically, adolescents with a concussion exhibit decreased PFC activity during quiet standing on firm ground, but increased PFC activity when on a foam surface. 28 The increased difficulty of standing on foam may require a concussed individual to recruit higher order cognitive processing cortices compared to the firm surface condition. 28 Further, adolescents with a concussion exhibit increased PFC activity during gait, particularly dual task gait, highlighting an increased frontal cortex burden during increasingly complex motor tasks in concussed adolescents. 32 The PFC, associated with executive function and attention, could be recruited to compensate for neurophysiologically impaired motor control regions responsible for maintaining standing posture.
The compensation-related neural circuit hypothesis (CRUNCH) posits higher order compensatory mechanisms are required for reduced motor automaticity, with evidence in older populations with motor impairment.33,34 The compensatory mechanisms of CRUNCH are required when gait automaticity deficits inhibit ability to successfully complete task within the established motor pathway. 35 Not only has PFC activity been associated with gait automaticity, but measures of stride-to-stride variability and postural control have been related to gait automaticity.36-39 Quantifying PFC activity during gait in the persistently symptomatic concussion adult population could provide insight into gait deficits observed in the persistently symptomatic.
The purpose of this study was to explore the effects of a concussion on PFC activity during gait in adults with persisting symptoms. We hypothesized that people who continue to self-report symptoms within 3 months of a concussion would have greater PFC activity during gait than people without a concussion. Within the concussed group, we hypothesized that worse symptoms would relate to increased PFC activity during gait, and that increased PFC activity would relate to gait characteristics that reflect gait automaticity.
Methods
Participants
A total of 39 people were included in this study; 14 participants with a recent concussion and 25 healthy controls. The people in the concussion group were still symptomatic and were recruited from a larger study (Award Number: W81XWH-15-1-0620, ClinicalTrials.gov identifier: NCT02748109) to participate in this study. The data for the 25 healthy controls were extracted from an existing repository from the Balance Disorders Laboratory at Oregon Health & Science University. The inclusion and exclusion criteria were the same for the larger study, 40 as there were no methodological requirements that would preclude participation based on the addition of fNIRS. The inclusion criteria were (1) having a diagnosis of concussion within 12 weeks of participation; (2) being between 18 and 60 years old; (3) having a Sport Concussion Assessment Tool version 5 symptom evaluation sub-score ≥1 for balance, dizziness nausea, headache, or vision AND a minimum total score of 15; (4) and having no or minimal cognitive impairment (≤9 on the Short Blessed Test). The exclusion criteria were (1) having other musculoskeletal, neurological, or sensory deficits that could explain dysfunction; (2) having moderate to severe substance use disorder within the past month; (3) being in severe pain during the evaluation (≥7/10 subjective rating); (4) being pregnant; (5) being unable to abstain from medications that might impair balance 24 hours before testing; (6) having contraindications to rehabilitation such as unstable C-Spine; and (7) actively participating in physical therapy for their concussion. Participants were permitted to undertake other forms of treatment for their symptoms such as massage, acupuncture, and counseling. The mechanism of injury was not restricted but was recorded. All participants were provided and signed a written consent approved by the Institutional Review Board at Oregon Health & Science University.
The fNIRS data for the 25 control participants were pulled from an existing data repository, which the participants agreed to have their data stored in for future analyses. This data repository agreement was verified with an Oregon Health & Science University IRB repository agreement signed by each control participant in this study. Controls were selected based on age and gender, to mirror the concussion group. To mirror the concussion group inclusion criteria the controls extracted were between 18 and 60 and identified as a male or female for gender. The previous investigation set inclusion criteria for the control group enrollment to (1) being between 20 and 90 years old and (2) able to walk for 2 minutes without assistance. 41 The exclusion criteria were any musculoskeletal, vestibular, visual, or other medical condition that would affect gait. 41 No gait data were available for the control participants in the data repository.
Procedures
Participants’ age and gender were recorded. In addition, participants with a recent concussion provided time from injury and mechanism of injury (eg, motor vehicle accident, sport participation, and fall).
The concussion group completed the Neurobehavior Symptom Inventory (NSI). The NSI questionnaire is a valid and reliable concussion symptom assessment. 42 The NSI consists of 22 items, each rated on a 5-point Likert scale, with a higher score indicating more severe symptoms. The maximum NSI score possible is 88. Each concussed participant was asked to rate each item based on how that symptom disturbed them over the previous 2 weeks.
All participants were instructed to walk at their self-selected pace, back and forth over a 9-m path, with 180° turns at each end. There was an initial 20 seconds quiet standing period (baseline period) at the beginning of each walk trial, followed by 80 seconds of walking (task period). The concussion group’s gait performance was recorded at 128 Hz by 6, body worn inertial measurement units (Opals, APDM, a Clario Company) placed on each foot and wrist, as well as the sternum and the lumbar region of the spine. These inertial measurement units are proven valid and reliable for characterizing gait across age and neurological populations.43,44
All participants in the concussion and control groups were fitted with a portable, 8 channel, continuous wave fNIRS system (OctaMon, Artinis Medical Systems, Netherlands) that bilaterally recorded the relative changes in oxygenated (HbO2) and deoxygenated (HHb) hemoglobin over the PFC at a sampling rate of 50 Hz. Two of the fNIRS device optodes were light detectors, while 8 optodes emitted infrared light at wavelengths of 760 and 850 nm. Two (1/hemisphere) of the 8 channels were short-separation channels (15 mm between transmitter and detector), while the remaining 6 channels were long-separation (35 mm between transmitter and detector) channels. The optodes were secured to a headband, according to the international 10 to 20 EEG system. After the headband (with optodes) was placed over the participant’s forehead, a digitizer (Patriot 3D digitizer, Polhemus, Vermont) was used to verify the position of the optodes based on the registration of anatomical references (nasion, vertex, inion, and bilateral preauricular points).
Data Processing
The processing of the fNIRS data followed accepted guidelines. 45 The data from the 3D digitizer were entered into the software package NIRS-SPM (http://www.nitrc.org/projects/nirs_spm), which was performed in MATLAB 2020a (Mathworks Inc, Massachusetts). The spatial registration routine was used to find the correspondence between the scalp location, where the fNIRS quantification was performed, and its underlying cortical surface where the source signal was located. The cortical regions quantified by fNIRS correspond to Brodmann areas 9 and 10.
The fNIRS data were processed in MATLAB 2020a, using custom-made, multi-step algorithms. First, the HOMER2 (https://homer-fnirs.org/) function was applied which ensured: (1) raw intensity data were converted into optical density, (2) artifacts (eg, movement) were removed or attenuated by wavelet filtering, (3) optical density data were converted into HbO2 and HHb concentrations, and (4) remaining artifacts were removed or attenuated by applying the correlation-based signal improvement method. Next, the data were baseline corrected by subtracting the mean signal of the 20 seconds quiet standing period from the following 80 seconds of the trial. Then, superficial hemodynamic responses were removed from the 6 long-separation channels. Briefly, the residuals were obtained by scaling the short-separation channel series to fit the long channel series in a least-squares sense over the entire trial duration. 46 A low-pass filter with a cutoff frequency of 0.14 Hz removed any remaining high-frequency noise and the 6 long-separation channels were median averaged. The outcome variable is the average median relative changes in HbO2 concentration during gait across the 6 long-separation channels, which can reduce the variability of a single channel approach.47,48 No attempt was made to lateralize PFC activity as evidence to justify a lateralized approach was limited and outside the scope of the study.
Spatiotemporal and kinematic gait characteristics were calculated from the inertial measurement units using the Mobility Lab software V2 (APDM, Inc., Oregon). The gait data represents the 80 seconds of the walking component of the trial. The following gait measures were calculated and included in analyses: gait speed (m/s), stride length (m), stride time (s), stride time variability (s), lumbar coronal range of motion (°), and lumbar sagittal range of motion (°). These variables were chosen based on our previous work which highlighted different domains of gait in the concussion population.9,19
Statistical Analyses
Data normality was tested using the Shapiro–Wilks Test of normality. Only stride length variability and lumbar sagittal motion were not normally distributed. A 1-way Analysis of Variance (ANOVA) was used to compare groups differences for relative changes in HbO2 concentration during gait. Levene’s test determined that relative changes in HbO2 concentrations did not have equal variances between groups, so Welch’s correction was used. Spearman rank correlations were used to quantify the relationship between NSI scores and relative HbO2 concentration changes, while Pearson correlations were used to determine the relationship between most gait performance and HbO2 concentrations in the concussion group. NSI scores are calculated from a Likert scale, necessitating a rank correlation approach. Cohen’s d effect sizes were also calculated for reference. Rank correlations were also used for the gait variables stride length variability and lumbar sagittal motion to account for their not normal distribution. Cohen’s d effect sizes were categorized using the subsequent range: small (0.2-0.5), moderate (0.5-0.8), and large (>0.8). 49 Alpha was set a priori at <.05. Data presented as mean(standard deviation), unless otherwise specified. All statistical analyses were completed in IBM SPSS 28.
Results
The concussed and control groups were similar in age (concussed: 34.3[11.0] years old; control: 34.4[9.7] years old; t37 = 0.06, P = .96) and gender (concussed: 9 [64%] female; control: 15 [60%] female; χ2 = 0.07, P = .79). The concussion group was 62.6 (38.6) days from their concussion, with 6 (43%) resulting from a motor vehicle accident, 4 (29%) resulting from sport participation, 1 (7%) from a fall, and 3 (21%) from accidents. The average total NSI score for the concussion group was 26.4 (13.2). The concussed group’s gait speed, stride length, stride time, stride time variability, and each of the lumbar range of motion variables are presented in Table 1.
Gait Performance.
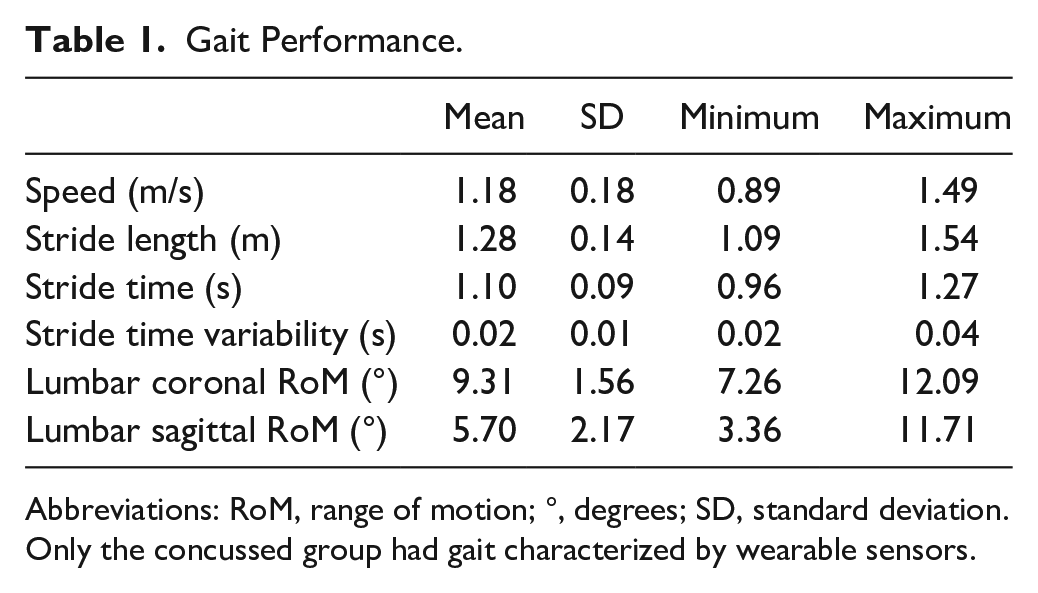
Abbreviations: RoM, range of motion; °, degrees; SD, standard deviation.
Only the concussed group had gait characterized by wearable sensors.
PFC Activity Differences Between the Concussed and Controls
The relative change in HbO2 concentration data is presented in Figure 1. The concussed group had a significantly greater increase in HbO2 during walking (Welch’s F1,17.21 = 5.78, P = .03; Cohen’s d = 0.97) compared to the control group. The relative HHb change in concentration values for the concussed group was −0.018 (0.060) and for the controls was 0.006 (0.030).
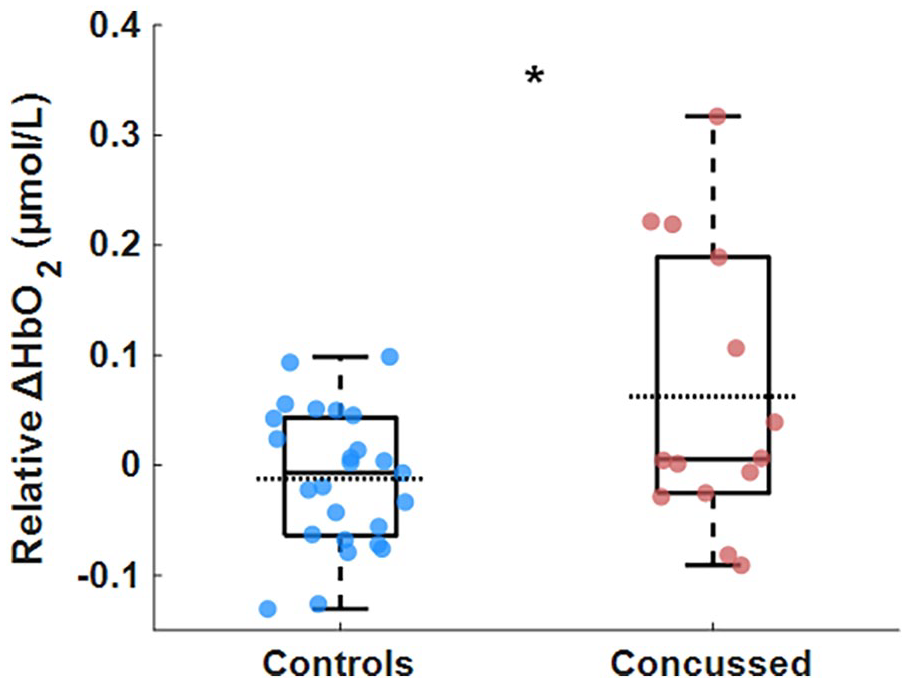
Box and scatter plot of the relative change in oxygenated hemoglobin (relative ΔHbO2) during gait.
Relationships Between PFC Activity and Both Symptom Severity and Gait Performance
The NSI total score significantly related to the relative change in HbO2 concentration during gait, such that worse symptom severity related to increased PFC activity (Figure 2). Both stride time variability and lumbar sagittal range of motion significantly correlated with the relative change in HbO2 concentration during gait, such that less variability and greater range of motion related to increased PFC activity (Figure 3). None of gait speed (Pearson’s r = .50), stride time (Pearson’s r = −.37), stride length (Pearson’s r = .47), stride length variability (Spreaman’s ρ = .05), nor lumbar coronal range of motion (Pearson’s r = .26) significantly correlated with the relative change in HbO2 concentration during gait.
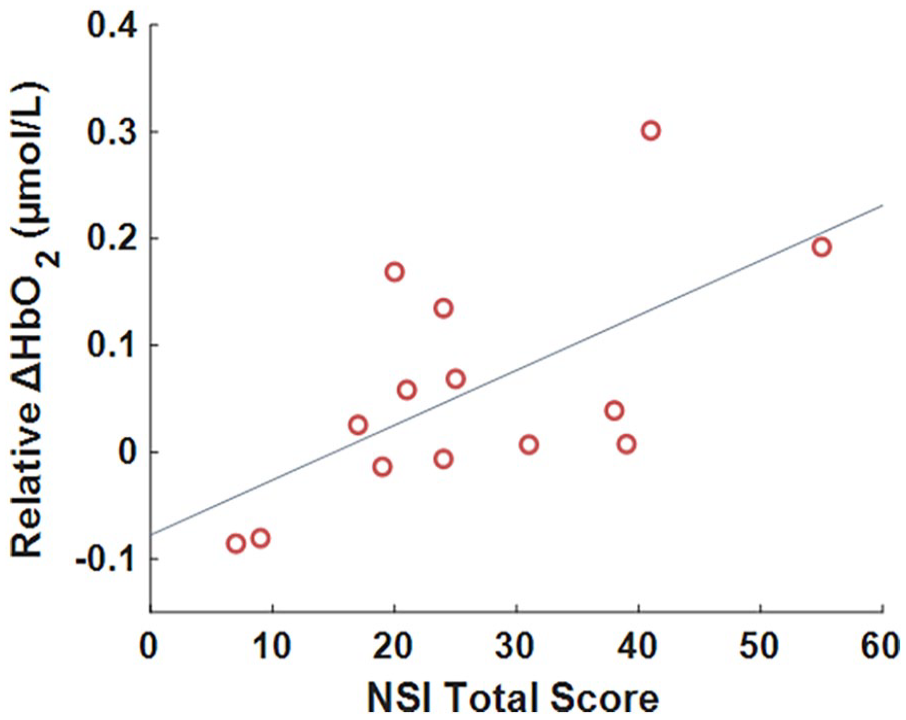
Scatter plot of the relationship between the relative change in oxygenated hemoglobin (relative ΔHbO2) during gait and self-reported symptom severity.
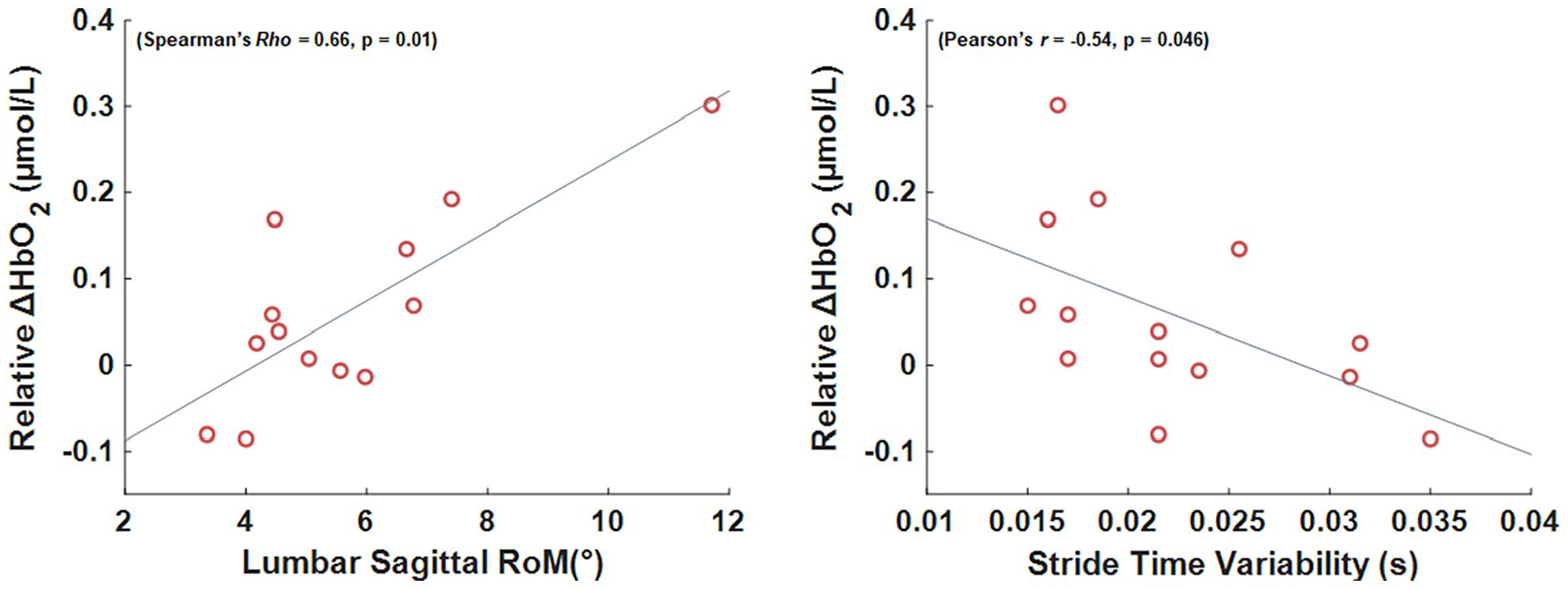
Scatter plots of the relationship between the relative change in oxygenated hemoglobin (relative ΔHbO2) during gait. Left: lumbar sagittal range of motion and right: stride time variability. Gray line represents best fit line.
Discussion
The purpose of this study was to explore PFC activity during gait in people with concussion and to begin to identify the relationship between persistent symptoms to PFC activity. We found that the concussion group exhibited increased PFC activity during gait compared to people without a concussion. Though our hypotheses were only partially support for the correlation analyses. Specifically increased PFC activity during gait significantly related to worse symptomology and gait automaticity characteristics (variability and some stability measures). These results begin to establish the neurophysiological underpinnings of gait dysfunction in adults with persistent symptoms after concussion.
Building off the concussion literature that quantified cortical activity and gross motor performance, we observed increased PFC activity during gait in people who were still symptomatic, 2 months after a concussion. Our results extend the current literature by addressing complex motor ability after a concussion in an adult population with persisting concussion symptoms. In an earlier study, not only was frontal lobe activity increased in the symptomatic concussion groups during balance tasks, as balance task complexity increased, frontal lobe activity typically increased more in the symptomatic concussion groups.28,32,50 An investigation focused on adolescent concussion observed decreased frontal lobe activation during increasingly complex balance tasks in a persistently symptomatic concussion group, though the reduced activity could be explained by differences in regions of interest within the frontal lobe. 51 Reduced frontal activation was observed in the left frontopolar cortex, while increased activity was observed in the right frontopolar cortex.50,51 The difference was explained by the study team as a difference in task-related activation, though another possibility could be that interhemispheric coherence is reduced in the symptomatic concussion population.27,51 That is, coherence between the left and right hemispheres is reduced following concussion, suggesting that left and right frontopolar cortices could exhibit different activation patterns during similar balance tasks after a concussion. A second study observed an increase PFC activity during gait in an adolescent concussion group within a month from injury, particularly during dual-task gait. 32 Reduced neural efficiency was also reported in the adolescent concussed, which could relate to compensatory network activation to complete increasingly difficult gait tasks. 32 Though, this interpretation is incomplete without additional studies quantifying a broader array of neurological networks during gait in the concussed. The combination of our results with those in the literature further suggest that frontal lobe activity is increased during complex motor tasks (eg, standing balance and gait) in people with persistent symptoms after a concussion.
Increased demand of the PFC during gait supports the growing notion that people with a concussion have reduced gait automaticity. Increased frontal lobe control was previously described by the compensation-related utilization of neural circuits hypothesis (CRUNCH), which is based on the observed increased cortical activity and impaired behavior in older adults compared to young adults. 33 Increased PFC activity during straight gait in neurologically impaired and intact older adults is posited as a possible compensatory mechanism (eg, CRUNCH model for compensation) for reduced motor automaticity, a mechanism not required by healthy controls.36,39,52,53 Our results provide neurophysiological context to the plethora of concussion literature that established dual-task gait deficits after a concussion in adults. 8 Dual-task gait, concurrently performing a secondary task while walking, paradigms are the common gait automaticity assessments in the concussion literature and elicit spatiotemporal gait differences in concussed populations. However, these approaches only provide the behavioral outcome to the neurophysiological underpinnings. Our results begin to establish the relationship between concussion neurophysiology and both clinical (eg, symptoms) and behavioral (eg, gait variability and stability) outcomes in adults with persisting concussion symptoms. Importantly, a PFC activity during gait also appears to be increased in adolescents within a month of a concussion. 32 Despite the differences between adolescents and adults sensitivity to the effects of a concussion and recovery, there appears to be a similar neurophysiological shift in the PFC that may underpin gait alterations in both populations.54,55
Worse self-reported total symptom scores and gait automaticity characteristics (ie, stride time variability and postural control) significantly related to increased PFC activity during gait in the concussion group. Linear relationships between concussion symptomology and brain activity have been previously reported, suggesting that cortical neurophysiology plays a role in self-perceived concussion symptoms. 56 Though, this prior study was structured for magnetic resonance imaging protocols, limiting the types of movement permitted in the protocol. Our results extend the scanner-based studies to gross, locomotor tasks that are not feasible in scanners.12-14,30 The significant relationships between increased PFC activity and both decreased gait variability and increased sway suggests that persistently symptomatic adults post-concussion have impaired gait automaticity. As noted, stride-to-stride measures of gait variability are related to PFC activity and gait automaticity in older adult and neurological populations.36,39 Along with stride-to-stride measures of variability, postural control also appears to relate to gait automaticity in young and older adults, particularly with increasingly difficult balance tasks.37,38 Further, a principal component analysis approach to constructing a concussion gait model identified gait variability as the strongest factor that described gait in symptomatic concussed adults, suggesting that gait variability is a prominent factor in symptomatic adults post-concussion. 19 Increased gait sagittal sway in the concussed has also been observed, particularly within the first 10 days after a concussion when symptoms would be highest. 8 The effect of concussion on sway direction (eg, sagittal vs coronal) may be related to oculomotor and stereoscopic deficits post-concussion.57,58 Though not possible given the limitations of inertial measurement units, accounting for step width would provide additional context to sway directionality differences. While only 2 of 6 gait variables significantly correlated with PFC activity, the correlation coefficients of 4 other gait variables were suggestive of possible relationships (ie, r’s of −.37 to .5). Importantly, this study was not powered on the gait variables reported in this study, so future studies could examine these relationships in a larger cohort.
Quantifying cortical activity during locomotor tasks allows for a more direct assessment of the relationship between cortical activity patterns and gait outcomes, which has been limited in concussion literature.32,59 Mobile neuroimaging appears to confirm that more severe concussion symptoms relate to increased PFC control of gait in persistently symptomatic adults, providing neurophysiological support for impaired gait automaticity post-concussion. However, future investigation should characterize the relationship between symptom scores and cortical activity in controls, as well as increasingly complex gait conditions to comprehensively define and understand the observed relationships in people with persistently symptomatic concussion.
This study has limitations that affect the interpretation of the results. First, our concussion group is small, limiting the statistical approach. Secondarily, spatiotemporal gait was not quantified in the control group, so we could not compare gait between groups. However, our previous work in a similar concussion cohort established gait differences between the symptomatic concussion population and healthy controls.9,19 Additionally, the exclusion criteria for the control group in the previous study were not identical to the exclusion criteria of the concussion group. Specifically, no attempt was made to exclude people with a cognitive impairment or substance abuse issue in the previously published control group. However, any controls with a substance abuse disorder would likely exhibit similar PFC activity to the concussion group, minimizing group differences, not driving group differences. 60 Controls pulled from the previous study were between 18 and 60 years old and could not have had a neurological condition that affected gait, so cognitive impairments are possible but unlikely. Further, we did not have information on symptoms in the control group, so we were unable to confirm a relationship in healthy people for symptoms and PFC activity during walking. The concussion group in this study exhibited similar spatiotemporal gait outcomes as our previously studied persistently symptomatic concussion cohort, despite the previous cohort being a year from injury and self-reporting a higher NSI score. 9 Finally, we only quantified PFC activity during gait, while other cortical areas could have larger roles in locomotion. Though, in establishing changes in PFC activity during gait, we mirror other fNIRS-gait literature that suggest increased PFC activity during gait is associated with a compensation for neurological dysfunction elsewhere in the brain. 33
The results of this study provide an initial view of neurophysiology during gait in adults with a persistently symptomatic concussion. The observed increased PFC activity during gait in the persistently symptomatic concussion group suggests reduced gait automaticity. Impaired motor automaticity post-concussion has been postulated but is based on behavioral evidence with limited neurophysiological evidence. Identifying the neurophysiological underpinnings to motor automaticity deficits post-concussion is only the first step in expanding our knowledge of motor behavior deficits post-concussion. Conceivably, more complex motor tasks such as running, returning to duty, or returning to sport may require even more allocation of PFC resources than the increased allocation to complete gait tasks. Critically, the results of this study have significant implications for concussions in older adults, subsequent injury risk in concussed athletes, child motor development post-concussion, 32 and concussion rehabilitation efficacy.
Footnotes
Author Contributions
Douglas N. Martini: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing. Martina Mancini: Data curation; Methodology; Resources; Writing—review & editing. Prokopios Antonellis: Data curation; Investigation; Writing—review & editing. Paul McDonnell: Formal analysis; Writing—review & editing. Rodrigo Vitorio: Methodology; Data curation; Formal analysis; Writing—review & editing. Samuel Stuart: Investigation; Methodology; Writing—review & editing. Laurie A. King: Conceptualization; Funding acquisition; Methodology; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Assistant Secretary of Defense for Health Affairs under the Award Number W81XWH-15-0620. All interpretations and opinions are those of the author and are not necessarily endorsed by the Department of Defense.
