Abstract
Background
Anodal transcranial direct current stimulation (AtDCS), a neuromodulatory technique, has been applied to treat traumatic brain injury (TBI) in patients and was reported to promote functional improvement. We evaluated the effect of contralesional AtDCS on axonal sprouting of the intact corticospinal tract (CST) and the underlying mechanism in a TBI mouse model to provide more preclinical evidence for the use of AtDCS to treat TBI.
Methods
TBI was induced in mice by a contusion device. Then, the mice were subjected to contralesional AtDCS 5 days per week followed by a 2-day interval for 7 weeks. After AtDCS, motor function was evaluated by the irregular ladder walking, narrow beam walking, and open field tests. CST sprouting was assessed by anterograde and retrograde labeling of corticospinal neurons (CSNs), and the effect of AtDCS was further validated by pharmacogenetic inhibition of axonal sprouting using clozapine-N-oxide (CNO).
Results
TBI resulted in damage to the ipsilesional cortex, while the contralesional CST remained intact. AtDCS improved the skilled motor functions of the impaired hindlimb in TBI mice by promoting CST axon sprouting, specifically from the intact hemicord to the denervated hemicord. Furthermore, electrical stimulation of CSNs significantly increased the excitability of neurons and thus activated the mechanistic target of rapamycin (mTOR) pathway.
Conclusions
Contralesional AtDCS improved skilled motor following TBI, partly by promoting axonal sprouting through increased neuronal activity and thus activation of the mTOR pathway.
Keywords
Introduction
With the highest incidence of all common neurological disorders, traumatic brain injury (TBI) results in a wide range of challenges, including cognitive, psychological, sensory, and motor impairments, and represents a substantial public health burden.1,2 Approximately 30% of TBI survivors have indicated experiencing motor deficits, such as limb weakness, difficulties with walking, balance issues, and muscle spasticity, that significantly impede their ability to engage in everyday activities or participate fully in life.3,4 Despite the great advances in emergency surgery and neurocritical care management, many TBI patients remain moderately to severely disabled at 5 years and are rehospitalized at up to 10 years postinjury. 5 Therefore, according to the current understanding, TBI should be treated as a chronic disease.5,6 TBI interrupts the corticospinal tract (CST), which descends from the ipsilesional motor cortex to the contralesional spinal cord; this results in motor impairments that can be partially abrogated by reestablishing connections between the brain and spinal cord.7,8 Synaptic plasticity and axonal sprouting in the CST are modulated by the excitability of neurons during neuronal development.9,10 In the adult brain, CST axonal sprouting can be promoted by the activation of corticospinal neurons (CSNs). 11 Recent animal experiments have revealed that the regrowth of ipsilesional descending fibers from the unaffected hemisphere to denervated motor neurons plays a significant role in the restoration of motor function.12,13 The possible underlying mechanisms include the promotion of CST axonal sprouting in the unaffected hemisphere across the midline into the denervate (affected) area of the spinal cord. 14 However, due to the limited potential for axonal outgrowth in adults, neurological recovery is difficult to achieve. 15
Neuromodulatory techniques have become of great interest in recent years as potential rehabilitative therapies after injury because of their capacity to alter neuronal activity and neural circuits in targeted brain regions. 16 A recent systematic review indicated that neuromodulation can reverse TBI-induced deficits in coordination, balance, and locomotor activity and ameliorate the impairment of memory, learning, and impulsivity. 16 Anodal transcranial direct current stimulation (AtDCS), as a noninvasive and safe technique, modulates the excitability of neurons through stimulation of brain areas with an anode electrode through the skull. 17 Recent studies have indicated that noninvasive brain stimulation can alleviate cognitive deficits and verbal retrieval deficits.18,19 However, whether AtDCS could improve motor function after TBI remains unclear. 20
The vicariation model and the inter-hemispheric inhibition model are 2 theoretical frameworks that have been proposed to explain the mechanisms underlying recovery after brain injury, particularly in the recovery of motor functions. 21 The vicariation model refers to a pattern of reorganization in which activity in the unaffected hemisphere compensates for the functions lost in damaged areas. The inter-hemispheric inhibition model assumes a mutual, balanced inhibition between the hemispheres, which would be disrupted once 1 hemisphere was damaged, leading to the affected hemisphere suffered additional damage by increased inhibition from the unaffected hemisphere. Pino et al suggested that the 2 models of recovery were oversimplified and thus combined them into a new model—the bimodal balance recovery model to establish a connection between interhemispheric equilibrium and functional recuperation, which was determined by the structural reserve remaining after the injury: the vicariation model was more important for predicting recovery in patients with little structural reserve, while the inter-hemispheric inhibition model was more suitable for minor damage with high structural reserve. Based on the bimodal balance recovery model, Pino et al 21 suggested that the application of cathodal tDCS over the unaffected motor cortex or AtDCS over the affected motor cortex was beneficial for improving motor function when the structural reserve remained high after the injury. Conversely, when the structural reserve was low, stimulation of the unaffected hemisphere exhibited a more important effect; hence, AtDCS over the unaffected hemisphere might be beneficial. 20 Therefore, in the present study, we employed a severe TBI mouse model to evaluate the effect of contralesional AtDCS on axonal sprouting in the unaffected CST and the underlying mechanism to provide more preclinical evidence for the use of AtDCS to treat TBI.
Materials and Methods
Experimental Animals
A total of 121 wild-type C57/BL6 mice (male, 8-10 weeks, 20-25 g) were obtained from the Experimental Animal Center of Army Medical University. All animal procedures were reviewed and approved by the Laboratory Animal Welfare and Ethics Committee of Army Medical University (No. AMUWEC20232691) and performed according to the Guide for the Care and Use of Laboratory Animals. These experiments are reported in compliance with the Animal Research: Reporting In Vivo Experiments guidelines, version 2.0. The mice were housed in a temperature-controlled room under specific-pathogen-free conditions on a standard 12-hour light/dark cycle and provided ad libitum access to food and water. Randomization was carried out by using odd and even numbers.
Establishment of a TBI Mouse Model
TBI was induced by a contusion device (68093, RWD) as previously described. 22 Briefly, the mice were anesthetized with 2% isoflurane (R510-22-10, RWD) and placed in a stereotaxic frame (68077, RWD). After making a midline incision over the skull surface, an ~3-mm-diameter left parietal craniotomy centered −0.7 mm anteroposterior (AP) and −1.5 mm mediolateral (ML) to bregma was made without injuring the underlying dura. The tip of a footplate (~2.5 mm in diameter) was placed under the dura at a depth of 2.5 mm. An 18 g rod was freely dropped from a height of 10 cm and guided to hit the footplate with a pipe. Sham mice underwent left parietal craniotomy without injury. After craniotomy, the bone flap was replaced and sealed using dental cement (3303L, Vitremer, 3M). The mice were kept on a heating pad to maintain body temperature.
Anodal Transcranial Direct Current Stimulation
Electrodes were surgically implanted in all mice prior to stimulation according to a previously described protocol.23-25 In brief, a circular copper electrode with a diameter of 2.5 mm and a length of approximately 5 mm, which was covered with a poly(vinyl) chloride membrane (ZCBV-2.5, PIGEON ELECTRIC WIRE & CABLE), was affixed to the skull at −0.7 mm AP and 1.5 mm ML from bregma using dental cement (3303L, Vitremer, 3M). An electrocardiogram electrode (2238, Red Dot, 3M), which was used as the cathode electrode, was adhered to the abdomen of the mice. At 1 week after surgery, AtDCS with a 150 μA current intensity was performed for 30 minutes/day. Mice received AtDCS for 5 days per week followed by a 2-day interval for 7 weeks. During AtDCS, the mice could freely move in individual cages.
Retrograde Labeling of CSNs in the Sensorimotor Cortex
To selectively label CSNs projecting to the lumbar spinal cord, we utilized a virus-based intersectional labeling method (Figure 1A). Briefly, the mice were anesthetized with 2% isoflurane and placed in a stereotaxic apparatus equipped with a heated pad for spinal cord injection. Following a skin incision, blunt dissection, and laminectomy, AAV2/Retro-Syn-GcaMP6s (diluted to 5.12 × 1012 copies/ml, BrainCase, Wuhan, China) was delivered bilaterally into the lumbar spinal cord (ML: ±0.3 mm; dorsoventral, DV: 0.5 mm, L1-L5; 5 sites in total; 100 nl/site) with a glass micropipette using a nanoliter injector (Nanoject II, Drummond Scientific). Injection was performed 2 weeks prior to TBI.
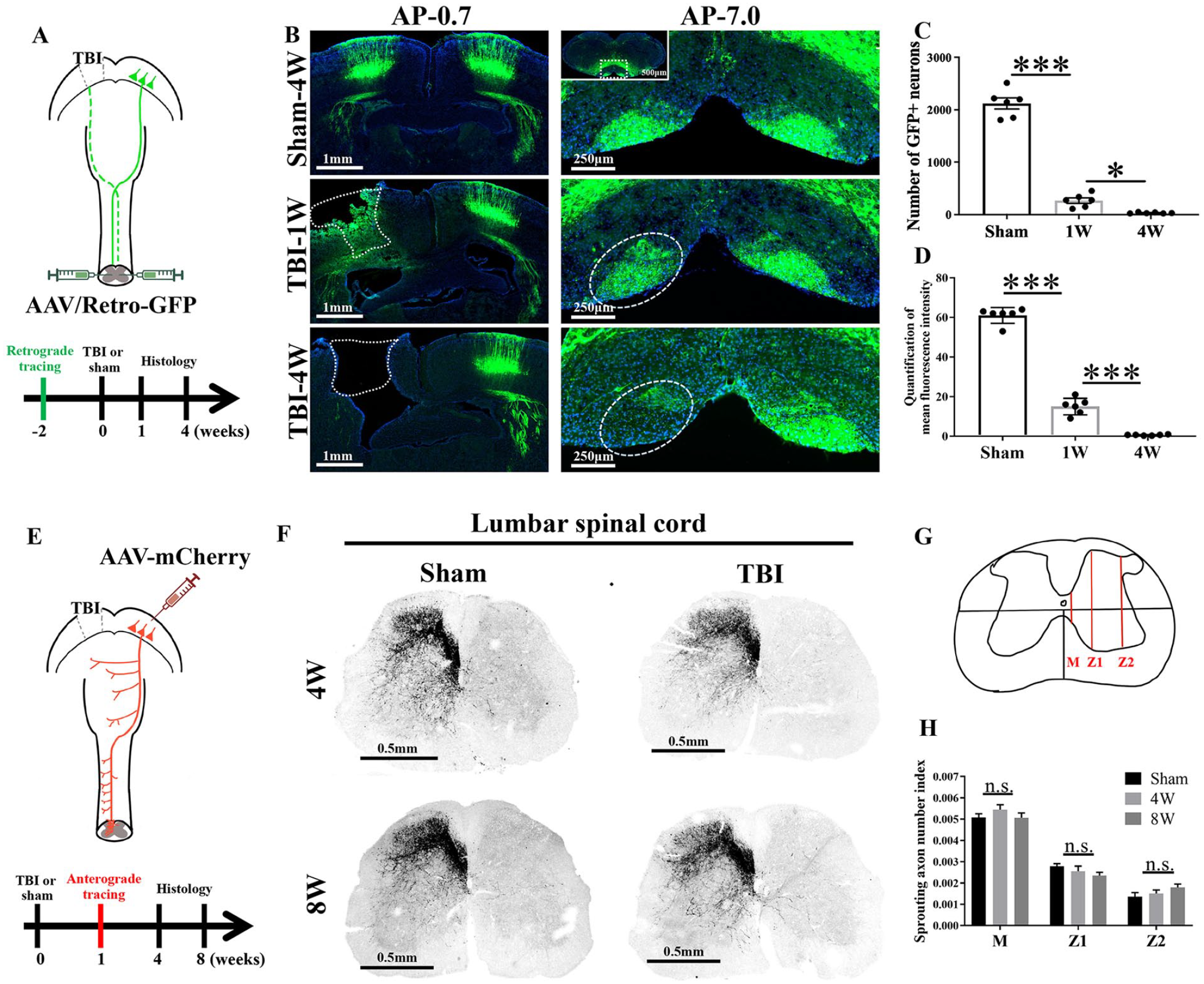
TBI to the region of the sensorimotor cortex corresponding to the hindlimb induced loss of CSNs without significant sprouting of the CST axons on the contralesional side. (A) Schematic diagram and timeline of the experimental procedure for retrograde tracing. The bilateral cortex was labeled by injecting AAV/Retro-GFP into the lumbar spinal cord. The mice received bilateral injection of the virus into the spinal cord at −2 weeks (W) and were subjected to unilateral TBI to the region corresponding to hindlimb motor function (the left side) at 0 W, and then histological analysis was performed. (B) Representative images of several sections of the brain (−0.7 and −7.0 mm AP). Left, the injured area is marked with the white dotted line (scale bar = 1 mm); right: the area in which CSNs were lost is marked with the white dotted line (scale bar = 250 μm). (C) The average number of GFP-positive neurons in the injured cortex in each group. n = 6. ***P < .001, *P < .05. (D) The mean fluorescence intensity in CST axons in sections of the brain stem in each group. n = 6. ***P < .001. (E) Schematic diagram and timeline of the experimental procedure for anterograde tracing. The unilateral cortex was labeled by injecting AAV-mCherry into the sensorimotor cortex. The mice were subjected to unilateral TBI to the region corresponding to hindlimb motor function (the left side) at 0 W, the virus was injected into sensorimotor cortex at 1 W, and then histological analysis was performed. (F) Representative images of lumbar spinal cord sections from each group. Scale bar = 0.5 mm. (G) The schematic diagram shows the midline of the spinal cord section. M, midline; Z1 and Z2, lateral regions. (H) The sprouting axon number in regions of the spinal cord in each group. n = 5. n.s., no statistical significance.
To visualize the reestablishment of CSN connections following AtDCS (Figure 4A), we employed a protocol based on that reported in a previous study. 8 After performing a skin incision, blunt dissection, and laminectomy on the targeted lumbar spinal cord section, we injected AAV2/Retro-CAG-H2B-GFP (diluted to 5.91 × 1012 copies/ml, BrainVTA, Wuhan, China) into the left (unaffected) side of the lumbar spinal cord (ML: −0.6 mm, DV: 0.5 mm, L1-L5; 5 sites in total; 50 nl/site). Additionally, AAV2/Retro-CAG-H2B-RFP (diluted to 5.61 × 1012 copies/ml, BrainVTA, Wuhan, China) was injected into the right (affected) side of the lumbar spinal cord (ML: 0.6 mm, DV: 0.5 mm, L1-L5; 5 sites in total; 50 nl/site) after TBI.
CSNs were retrogradely labeled by injecting AAV2/Retro-Syn-GcaMP6s into the left side of the lumbar spinal cord (ML: −0.6 mm, DV: 0.5 mm, L1-L5; 5 sites in total; 100 nl/site) 2 week before TBI. After injection, the glass pipette was left in place at the injection site for a duration of 10 minutes before being retracted. Then, the expression of pS6 in CSNs in the region of the sensorimotor cortex corresponding to the unaffected hindlimb was observed by immunofluorescence (Figure 5A).
Anterograde Labeling of the CST in the Lumbar Spinal Cord
A virus-based intersectional labeling method was also employed to anterogradely trace sprouting fibers (Figure 1E). Following a scalp incision, a craniotomy (centered at −0.7 mm AP and 1.5 mm ML from bregma, 0.5 mm × 0.5 mm) was made on the unaffected cortex using a microdrill. Following craniotomy, AAV2/9-hSyn-hChR2(H134R)-RFP (diluted to 5.03 × 1012 copies/ml, BrainCase, Wuhan, China) was injected into the unaffected cortex (AP: −0.7 mm, ML: 1.5 mm, 300 nl) with a glass micropipette via a nanoliter injector (Nanoject II, Drummond Scientific) 7 days after TBI. The glass pipette was left in place for 10 minutes before being retracted. After injection, the mice were maintained for 4 weeks before being sacrificed for further analysis.
Pharmacogenetic Inhibition of CSN Axon Sprouting
To achieve selective expression of hM4Di from the affected lumbar segment to the unaffected cortex (Figure 6AD), AAV2-syn-FLEX-hM4Di (diluted to 5.67 × 1012 copies/ml, BrainVTA, Wuhan, China) was injected into the unaffected cortex (AP: −0.7 mm, ML: 1.5 mm, 300 nl) following craniotomy. Simultaneously, AAV2/Retro-hsyn-Cre (diluted to 5.12 × 1012 copies/ml, BrainVTA, Wuhan, China) was injected into the affected lumbar segment (ML: 0.6 mm, DV: 0.5 mm, L1-L5; 5 sites in total; 50 nl/site) at 8 weeks after TBI. Four weeks after the expression of hM4Di, clozapine-N-oxide (CNO, SML2304, Sigma) or saline was administered (1 mg/mg, i.p.). Subsequently, the animals were evaluated using the irregular ladder walking and narrow beam walking tests to reassess their skilled motor function.
Behavioral Tests
All behavioral data were quantified and analyzed in a blinded manner. Gross motor function was assessed by the total distance traveled and average speed in the open field test, while skilled motor function was assessed by the successful step rate of the affected hindlimb in the narrow beam walking and irregular ladder walking tests.
Irregular Ladder Walking Test
Performance in the irregular ladder walking test is dependent on cortical function during the recovery period. 26 The apparatus used for the horizontal ladder walking test was 100 cm long, 19 cm high, and 10 cm wide as previously described. 27 Metal rungs (3 mm diameter) were inserted into the clear side walls to create a floor with a minimum distance of 1 cm between the rungs; the spacing of the rungs was variable and changed periodically. Before TBI, the animals were trained to walk on a ladder with rungs spaced regularly (1 cm between each rung) and to anticipate the location of the rungs 2 weeks before TBI. An irregular rung pattern (30 randomly spaced rungs; maximum distance of 3 cm between the rungs) was used for each trial, and the pattern was changed between trials to prevent the animals from learning the pattern. The success rate of affected hindlimb placement over 100 steps was analyzed.
Narrow Beam Walking Test
The beam walking test was used to assess fine motor coordination by examining the ability of the animal to remain upright and walk on a narrow beam. The narrow beam was approximately 0.6 cm wide and 120 cm in length and elevated 60 cm above soft bedding material as previously described. 27 All mice were trained to walk on the beam 2 weeks before TBI, and only mice whose paws slipped from the horizontal surface of the beam (foot faults) fewer than 10 times per 100 steps were used for the experiments. The number of affected hindlimb foot faults over 100 steps was counted and analyzed.
Open Field Test
The open field test was performed to evaluate the gross motor performance of mice during the recovery period after TBI. The mice were placed in a 50 cm × 50 cm × 50 cm chamber for 5 minutes and video recorded. The mice were placed in separate chambers, and the chambers were cleaned between each trial. The total distance traveled and average velocity were recorded and analyzed.
Immunohistochemistry
The mice were euthanized using 1% pentobarbital sodium (100 mg/kg body weight, i.p.). Then, they were transcardially perfused with cold saline and 4% paraformaldehyde. The brain and spinal cord were extracted, fixed, dehydrated and sliced in the transverse plane at a thickness of 30 μm using a cryostat. The primary antibodies included rabbit red fluorescent protein antibody (anti-RFP, 1:200, ab62341, Abcam), rabbit green fluorescent protein antibody (anti-GFP, 1:200, ab290, Abcam), rabbit anti-phosphorylation of S6 ribosomal protein (anti-pS6, 1:1000, 4857S, Cell Signaling Technology), and rabbit anti-cellular oncogene fos (anti-c-Fos, 1:1000, 2250S, Cell Signaling Technology) antibodies. The secondary antibodies used were Alexa Fluor 488-conjugated chicken anti-rabbit immunoglobulin G (IgG) (1:500, A21441, Thermo) and CFL 555-conjugated mouse anti-rabbit IgG (1:200, sc-516249, Santa Cruz). To stain the nuclei, 4′,6-diamidino-2-phenylindole (DAPI, 1:500, 236276, Roche) was utilized. The same imaging parameters (laser power and pinhole size) were maintained for all experiments. Images were acquired using a Zeiss LSM 880 confocal microscope.
Neuron Counting and Quantification
Each image of the target brainstem region (AP = −7 mm) containing stained neurons was binarized by an appropriate threshold. The number of CSNs in each cortical section was calculated using ImageJ software. Then, the number was summed to determine the total number of neurons expressing each marker. The percentage of RFP-positive neurons was determined using the following formula: the number of RFP-positive neurons divided by the number of GFP-positive neurons in arbitrary units (a.u.). The fluorescence intensities of GFP, c-Fos, and pS6 in the CST in target brain regions were quantified in a blinded manner using the following formula: mean green fluorescence intensity—mean background fluorescence intensity in arbitrary units (a.u.). We quantified the average fluorescence intensity in 5 sections along the rostral-caudal axis for each animal.
Axon Counting and Quantification
Images of lumbar spinal cord sections were binarized by an appropriate threshold to preserve the real signal as much as possible. Then, fibers crossing the spinal cord midline were counted at ×10 magnification, and branching of these fibers was evaluated at 2 defined distances from the midline in the gray matter using a virtual grid (Z1 and Z2) comprising lines separated by 200 μm as described in a previous study. 28 Only fibers crossing the 3 lines were counted on each section. The number of fibers was normalized to the number of CST fibers in the medulla, and 5 coronal sections of the lumbar spinal cord per animal were assessed in a blinded manner.
Statistical Analysis
Statistical analysis was performed using Prism 9.0 software. The data are expressed as the mean ± SD. Comparisons between 2 groups were analyzed using 2-tailed Student’s t tests. Comparisons among 3 groups or multiple time points were analyzed using 1-way ANOVA (1 factor) followed by Tukey’s post hoc test. A P value <.05 was considered to indicate statistical significance.
Results
TBI Caused Damage to CSNs But Had No Effect on the Sprouting of Intact Corticospinal Axons During Spontaneous Recovery
Retrograde labeling of CSNs was performed 2 weeks before TBI, and histology was performed in the first and fourth weeks after TBI and fourth week after the sham operation (Figure 1A). We observed a progressive reduction in the number of CSNs in the injured area after TBI, while the number of CSNs in the contralesional CST was not affected (Figure 1B). The number of GFP-positive neurons in the affected cortex and the mean fluorescence intensity of GFP in the affected CST in the brainstem (AP: −7 mm) were decreased significantly (Figure 1C and D). Sprouting of the unaffected corticospinal axons across the midline to innervate the denervated (affected) spinal cord has been proven to be an important mechanism of functional recovery after CNS injury. 29 In this study, an anterograde tracer was injected into the unaffected cortex, and axonal sprouting in the lumbar spinal cord was observed and assessed in the fourth and eighth weeks; the results are shown in Figure 1E. Representative images of sections of the lumbar spinal cord from each group are shown in Figure 1F. The lumbar spinal cord was divided into 3 regions (Figure 1G) in the transverse plane as previously described 28 to analyze the axonal sprouting index. We found no significant difference in the axonal sprouting index between the sham and TBI groups regardless of region and time (Figure 1H, sham vs 4 weeks vs 8 weeks, P > .05).
AtDCS Improved Skilled Motor Functions After TBI
We next examined whether AtDCS can improve motor function after TBI. AtDCS of CSNs on the unaffected cortex was performed 5 days per week followed by a 2-day interval for 7 weeks (Figure 2A). Skilled motor functions, as assessed by the successful step rate of the affected hindlimb in the narrow beam walking (Figure 2B) and irregular ladder walking tests (Figure 2C), were significantly compromised after TBI (sham vs TBI, P < .001). AtDCS significantly improved performance in the narrow beam walking and irregular ladder walking tests from week 4 and week 6, respectively (P < .05). These results suggested that AtDCS improved skilled motor functions after TBI. However, gross motor functions, as assessed by the total distance traveled (Figure 2E) and average speed (Figure 2F) in the open field test, were not significantly different between the TBI and AtDCS groups (TBI vs TBI + AtDCS, P > .05).
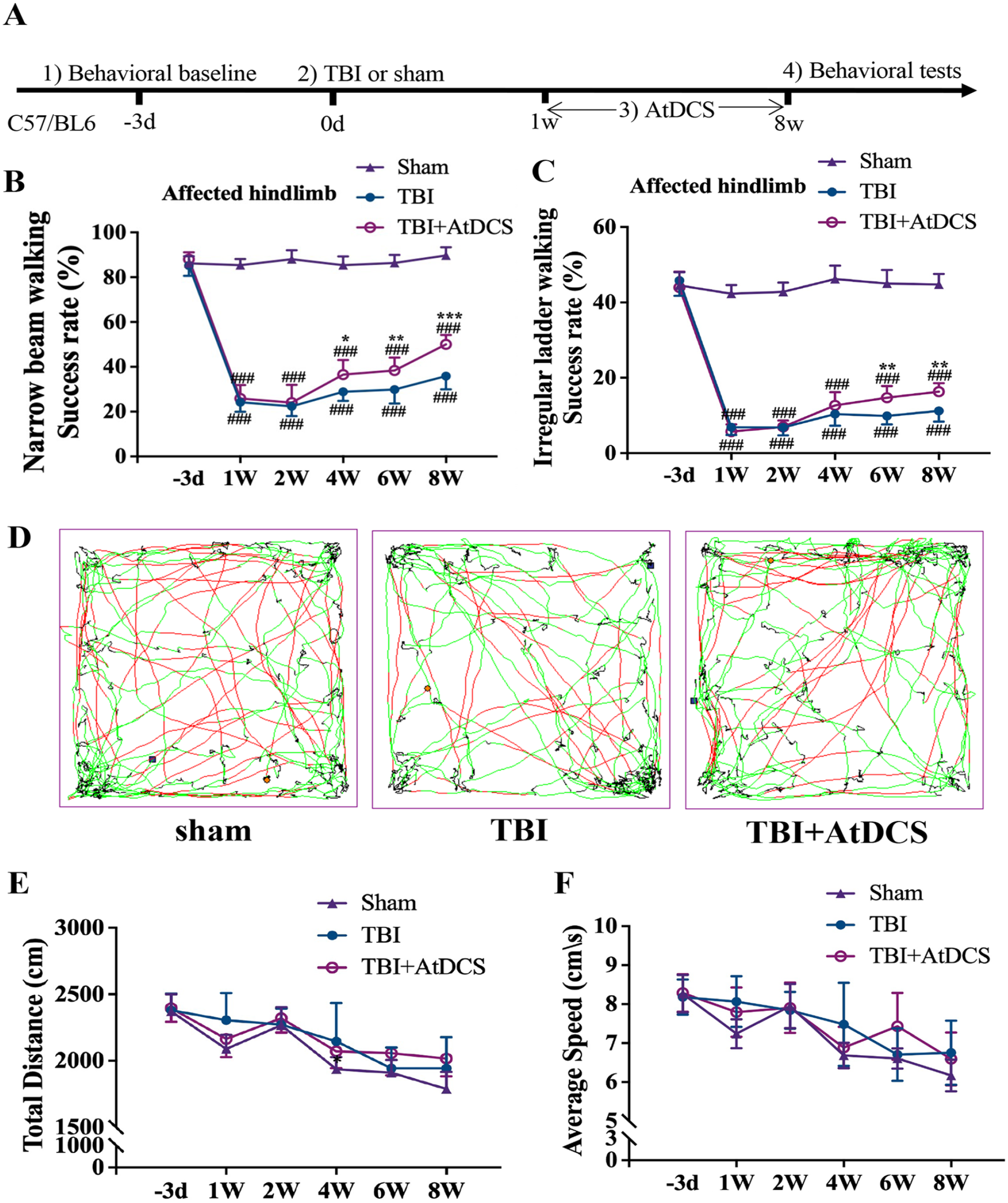
Effect of AtDCS on skilled and gross motor functions. (A) Timeline of the motor function tests after AtDCS. (B-C) Function of the affected hindlimb in the narrow beam walking (B) and irregular ladder walking tests (C). n = 6. #, versus the sham group; ### P < .001; *, versus the TBI group; *P < .05; **P < .01; *** P < .001; n.s., no statistical significance. (D) Representative trajectory of mice in the open field test. The red trajectory indicates a velocity < 25 cm/s; the green trajectory indicates a velocity <25 but >7 cm/s; and the black trajectory indicates a velocity <7 cm/s. (E) Total distance traveled by the different groups in the open field test. n = 6. (F) Mean speed of each group in the open field test. n = 6. n.s., no statistical significance.
AtDCS Promoted Axonal Sprouting and Circuit Rewiring in the Lumbar Spinal Cord After TBI
We next investigated the effect of AtDCS on CSNs after TBI using retrograde and anterograde tracing (Figures 3A and 4A). We observed no obvious sprouting of CSN axons in the denervated (affected) spinal cord in the TBI and sham groups (Figure 3B and C). Interestingly, AtDCS induced a moderate number of unaffected CSN axons to cross the midline and innervate the denervated (affected) hemicord; the number of CNS axons crossing the midline was significantly higher in the AtDCS group than in the TBI group (Figure 3B, P < .001).
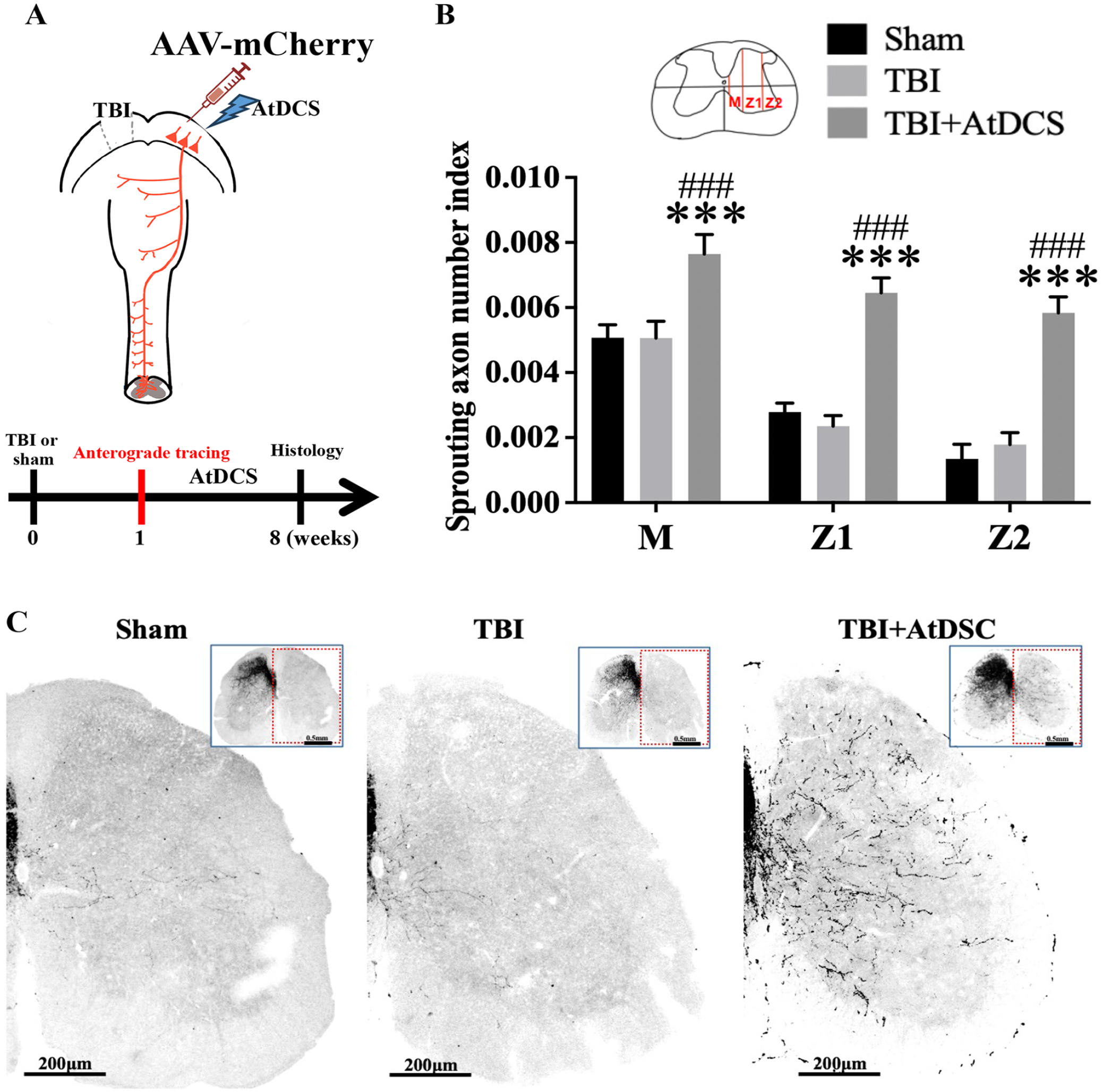
AtDCS promoted the axonal sprouting of CSNs in the lumbar spinal cord after TBI to the region corresponding to hindlimb motor function. (A) Schematic drawing and timeline of the experimental procedure. The mice were subjected to unilateral TBI of the region corresponding to hindlimb motor function at 0 Week (W), neurons in the sensorimotor cortex were labeled by injection of AAV-mCherry virus and the mice were administered AtDCS at 1 W, and histological analysis was performed at 8 W. (B) The sprouting axon number in different regions of the spinal cord in each group. The schematic diagram in the upper panel shows how the spinal cord was divided. n = 5. *, versus the TBI group; ***P < .001; #, versus the sham group; ###P < .001. (C) Representative images of lumbar spinal cord sections in each group. Scale bar = 200 μm.
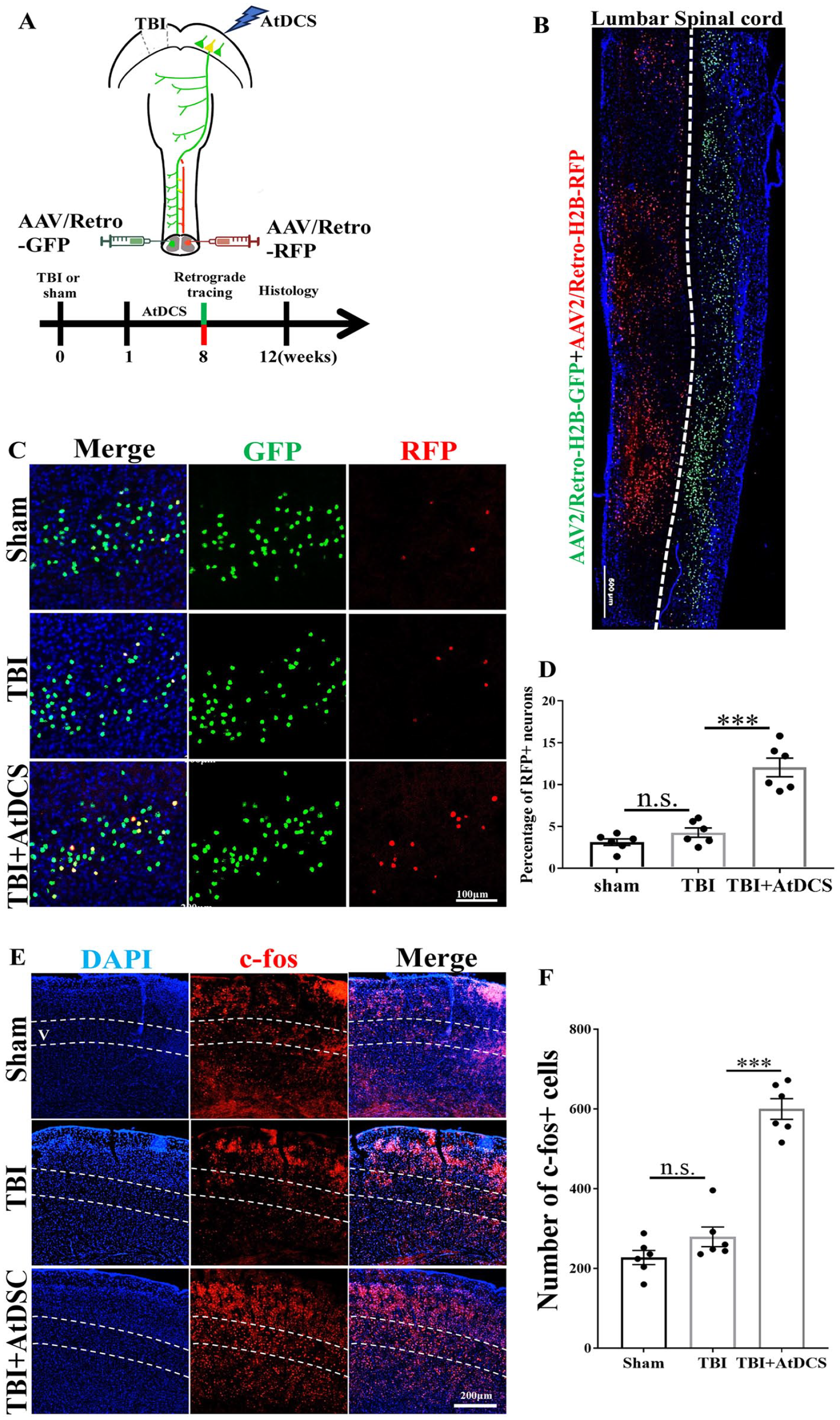
AtDCS promoted the formation of connections between the injured lumbar spinal cord and cortex. (A) Schematic diagram and timeline of the experimental procedure. The mice were subjected to unilateral TBI at 0 week (W), AtDCS was applied from 1 to 8 W, lumbar spinal cord neurons were labeled by injection of AAV/Retro-GFP (left) and AAV/Retro-RFP (right) at 8 W after AtDCS, and histological analysis was performed at 11 W. (B) Representative images of horizontal sections of the lumbar spinal cord showing virus expression. The nuclei of CSNs on the left side of the lumbar spinal cord were labeled with GFP, and those on the right side were labeled with RFP. Scale bar = 500 μm. (C) Representative images of RFP-positive nuclei in the region of the sensorimotor cortex corresponding to the hindlimb in each group after retrograde tracing. Scale bar = 100 μm. (D) Percentage of RFP-positive neurons in each group. n = 6. *, versus the TBI group; ***P < .001; n.s., no statistical significance. (E) Representative images of c-Fos-positive nuclei in the sensorimotor cortex in each group. The dashed line indicates layer V. Scale bar = 200 μm. (F) Quantification of the number of c-Fos-positive cells in layer V in each group. n = 6. *, versus the TBI group; ***P < .001; n.s., no statistical significance.
Two viruses for retrograde labeling (encoding GFP or RFP) were injected individually into the 2 sides of the lumbar spinal cord (Figure 4A and B). Representative images of RFP- and GFP-positive nuclei in the sensorimotor cortex of each group after retrograde tracer injection are shown in Figure 4C. The percentage of RFP- and GFP-positive neurons in the sensorimotor cortex was increased after AtDCS (Figure 4D, TBI vs TBI + AtDCS, P < .001), suggesting that more unaffected CSNs formed new connections with the affected lumbar spinal cord.
AtDCS Increased the Expression of c-Fos and the Activation of the mTOR Pathway
We then examined the activity of c-Fos, a marker of activated neurons, to assess the effectiveness of CSN stimulation. 30 C-Fos-positive cells (activated neurons) were preferentially located in layer V, which CSNs innervate (Figure 4E). After AtDCS, the number of c-Fos-positive cells in the sensorimotor cortex (AP: 0.7 mm) was increased (Figure 4F, TBI vs TBI + AtDCS, P < .001).
Because previous studies showed that the phosphoinositide 3-kinase/mechanistic target of rapamycin (mTOR) pathway plays an important role in axonal outgrowth after injury,31,32 we further explored whether AtDCS activates mTOR in CSNs. An antibody against the phosphorylated form (Ser235/236) of ribosome protein S6 (pS6), a reliable marker for estimating mTOR activity, 33 was used. We first injected rAAV-Syn-GFP into the unaffected lumbar spinal cord (L1-L5) of animals in the different groups before assessing pS6 activity in retrogradely labeled CSNs (GFP-positive) in the region of the sensorimotor cortex corresponding to the hindlimb, and the results are shown in Figure 5B. The percentage of pS6-positive CSNs in the AtDCS group was higher than that in the spontaneous recovery group (Figure 5C, TBI vs TBI + AtDCS, P < 0.01); these cells might be responsible for promoting regenerating axons to cross the midline of the spinal cord.
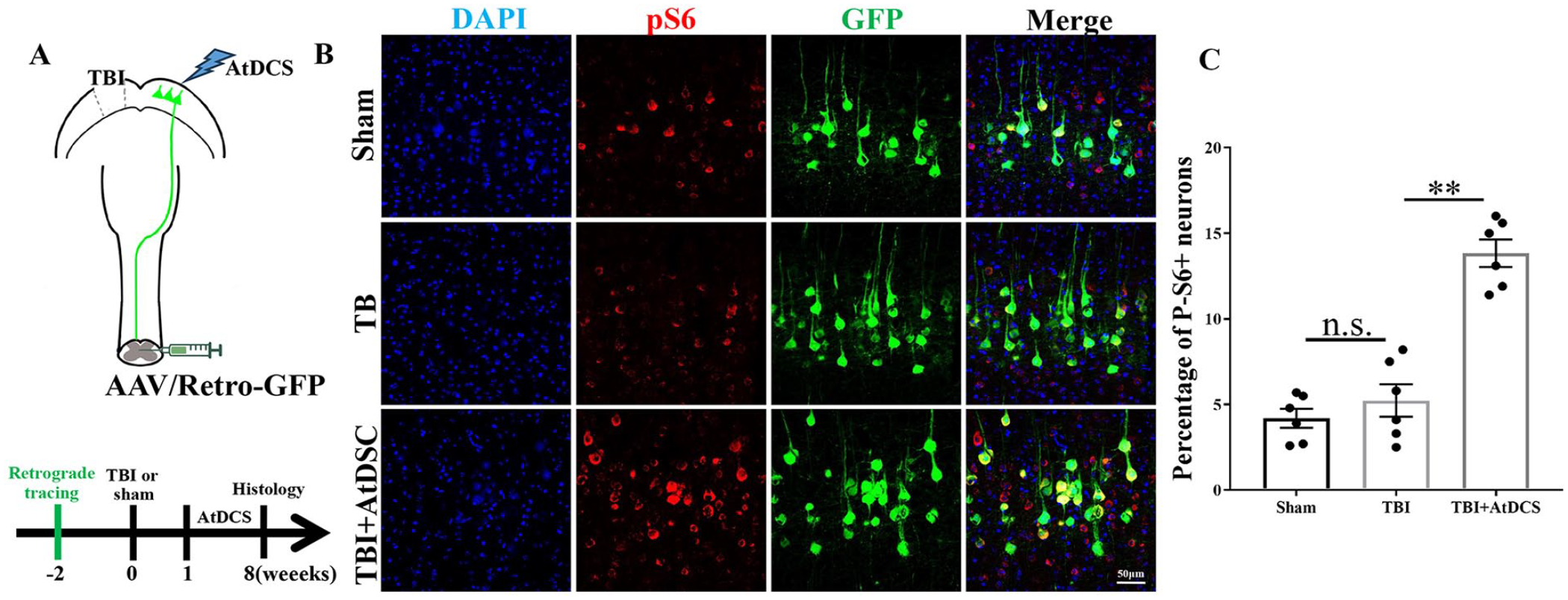
AtDCS increased the expression of pS6 in CSNs. (A) Schematic diagram and timeline of the experimental procedures. The mice were injected with a retrograde tracer at −2 week (W), they were subjected to TBI to the region corresponding to hindlimb motor function at 0 W, AtDCS of the sensorimotor cortex was performed from 1 to 8 W, and histological analysis was performed at 8 W after AtDCS. (B) Representative immunofluorescence images of CSNs (GFP) and phosphorylated S6 (pS6, RFP) in the region of the sensorimotor cortex corresponding to the contralesional hindlimb. Scale bar: 50 μm. (C) Percentage of pS6-positive neurons in each group. n = 6.
Suppression of Sprouting Axon Loss Restored Skilled Motor Function After AtDCS
We next assessed whether sprouting axons crossing the midline are vital for the enhancement of skilled motor function after AtDCS. We injected AAV/Retro-Syn-Cre into the left side of the lumbar spinal cord (L1-L5) and the inhibitory DREADD virus (AAV-Syn-Flex-hM4Di) into the right side of the cortex at 1 week after craniotomy, simultaneously (Figure 6A). The hM4Di receptor-based chemogenetic designer receptors exclusively activated by designer drugs (DREADD) system has been widely used to suppress neuronal activity. 34 We conducted experiments to verify the feasibility of this method, and the results are shown in Figure 6B. We found that pre-infecting unaffected axons with hM4Di-DREADD did not affect behavior following intraperitoneal injection of saline. However, after intraperitoneal injection of CNO, motor function associated with these axons was temporarily suppressed, and this inhibitory effect was abolished as CNO was metabolized and removed from the body (Figure 6C). Therefore, we performed the same experiment with a TBI model (Figure 6D,E). CNO temporarily suppressed the ability of AtDCS to improve behavioral functions after TBI (Figure 6F, TBI vs TBI + AtDCS, P > .05), indicating that AtDCS promoted compensatory axonal sprouting across the midline, which led to the binding of hM4Di-DREADD to these axons. Thus, sprouting axons of CSNs that crossed the midline were vital for the recovery of skilled hindlimb movements.
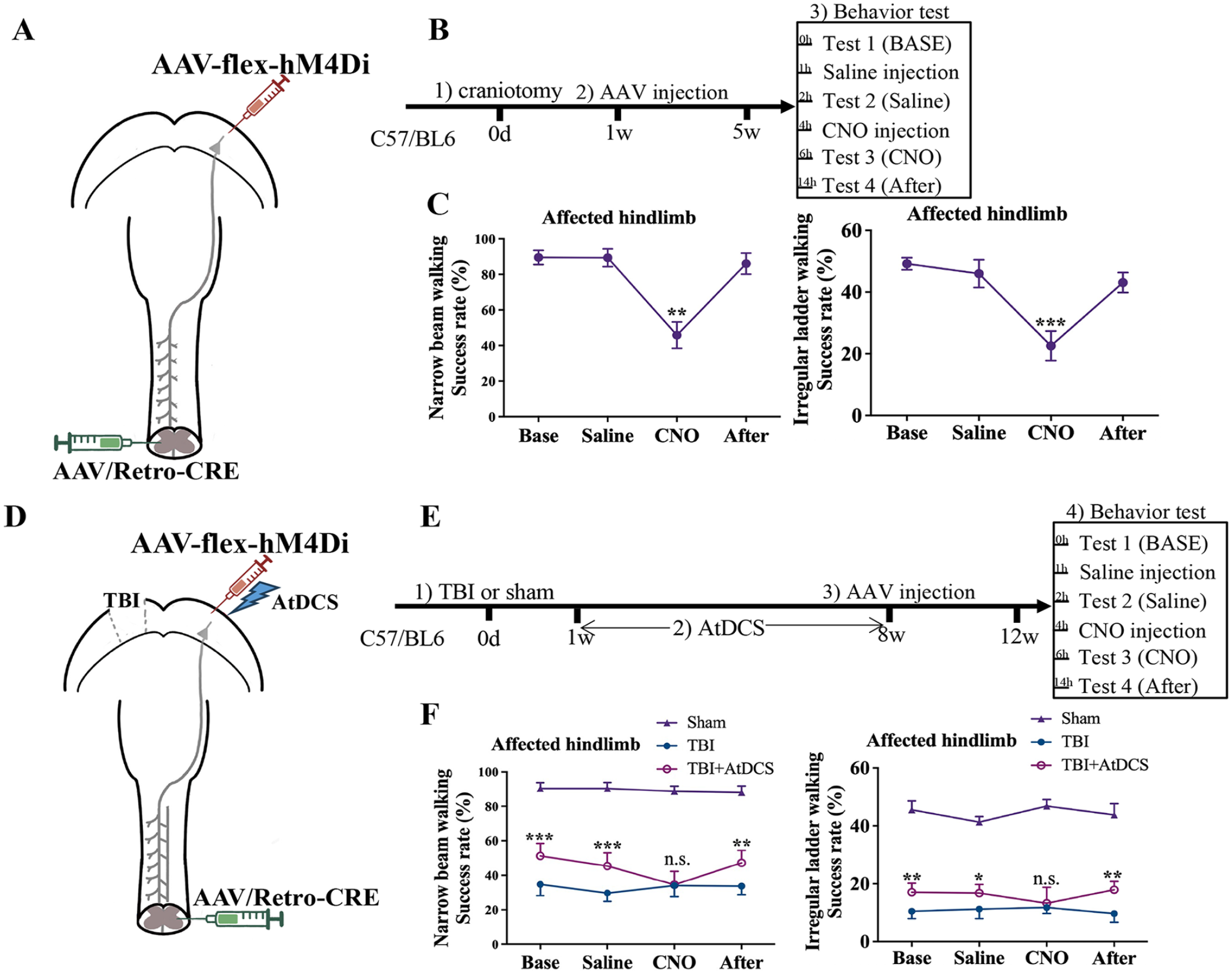
Suppression of spouting axon ablation restored skilled motor function. (A) Schematic diagram of the experimental procedures. (B) Timeline of the behavioral studies. (C) Effect of suppressing unaffected CSN activity on performance in the narrow beam walking and irregular ladder walking tests. n = 6. *, versus the BASE groups; **P < .01; ***P < .001. (D) Schematic diagram of the experimental procedures. (E) Timeline of the behavioral studies. (F) Effect of suppressing the activity of sprouting CSN axon on performance in the narrow beam walking and irregular ladder walking tests.
Discussion
In this study, we utilized a mouse model to evaluate the efficacy of AtDCS in treating TBI and explored the underlying mechanism. We found that AtDCS of the unaffected side improved the skilled motor function of the affected hindlimb after TBI. Through AAV-based anterograde/retrograde labeling, we found that AtDCS of the unaffected cortex promoted axonal sprouting of spared CSNs, contributing to the formation of new neural circuits. As previously reported, neuronal activity is associated with the mTOR signaling pathway, which is crucial for CST axonal outgrowth and synapse formation. Therefore, we speculated that AtDCS promotes the activation of the mTOR signaling pathway (as indicated by increased expression of the downstream protein pS6) by stimulating neuronal activity (as indicated by increased expression of the neuronal activity marker c-Fos), thereby promoting CST axonal outgrowth and synapse formation and ultimately leading to improved motor function after TBI.
Globally, more than 50 million individuals suffer from TBI annually, and over 3 million survivors are left with lifelong disabilities in the USA.35-37 However, effective treatment methods are limited, and the therapeutic effects of these treatment methods are satisfactory in a minority of patients. 38 Restoring nervous system function following TBI is a challenge; therefore, appropriate rehabilitation approaches need to be explored. Reversibly altering the function of the central, peripheral, or autonomic nervous system via neuromodulation through electrical, chemical, or mechanical means is widely considered a potential strategy for rehabilitation after CNS injury.39-41 AtDCS has been employed as a noninvasive neuromodulatory technique in both clinical and preclinical studies on TBI. 16 Preclinical studies have demonstrated the potential of AtDCS to promote motor function recovery and increase cortical neuron excitability in animals and athletes following TBI.42,43 However, the mechanism by which AtDCS contributes to rehabilitation after TBI is still unclear. In stroke patients, stimulation of the supplementary motor area (SMA) using AtDCS has been observed to improve gait stability by increasing the excitability of CST axons, suggesting that AtDCS of specific functional regions may increase the plasticity of specific neuronal synapses, which is critical for improving motor function. 44 Our previous study also indicated that repetitive magnetic stimulation can promote neural stem cell proliferation and differentiation to promote recovery after intracerebral hemorrhage. 45 Another study indicated that AtDCS of the unaffected primary motor cortex (responsible for controlling the dominant hand) can improve motor function in older adults, suggesting that ipsilesional control may serve as a potential mechanism for promoting motor function recovery in patients. 46 Researchers elicited latent potential in the unaffected hemisphere and established a novel sensory feedback pathway for the paralyzed hand by manipulating the peripheral nerve through cervical nerve relocation in a preclinical study on animals. 8 In our previous study, AtDCS also exhibited potential in alleviating neuropathic pain after spinal cord injury in preclinical models. 47 Therefore, we hypothesized that AtDCS may promote spared CST axon sprouting and thus lead to better outcomes.
We studied the impact of AtDCS on neurons in the region of the sensorimotor cortex corresponding to the hindlimb and their projections within the CST in TBI model mice. We observed that the model mice exhibited specific behavioral deficits, particularly impairment of the skilled motor function of the hindlimb, while the integrity of the contralesional CST and CSNs were preserved. These distinct characteristics of our model rendered it an optimal tool for investigating the consequences of spared CST axons after TBI. 28 Moreover, anodal AtDCS of apparently unrelated spared CSNs on the unaffected side improved hindlimb skilled motor function; this finding is consistent with the phenomenon of “ipsilesional control” observed in prior studies of central nervous system injuries.8,46,48 Retrograde and anterograde labeling revealed that AtDCS promoted neural connections on the affected side; this may explain the observed improvement of skilled motor function following stimulation. The role of the mTOR pathway, a critical regulator of neuronal axon regeneration, in repairing central nervous system injuries has been extensively studied. 49 Electrical stimulation has been reported to activate the mTOR pathway in the cortex, promoting axonal regeneration of undamaged neurons.50,51 Additionally, chronic electrical stimulation of the motor cortex can increase the number of spinal cord neurons expressing c-Fos and rapidly upregulate mTOR in the corticospinal system. 11 Inhibition of the mTOR pathway abolishes the stimulation-dependent sprouting of axons, underscoring the pivotal role of mTOR signaling in the beneficial effect of electrical neuromodulation.11,52 In this study, we observed a substantial increase in the number of c-Fos-positive neurons in the unaffected cortex, accompanied by activation of the mTOR pathway, which is consistent with previous findings.
After central nervous system injury, such as TBI, there are alterations in plasticity, excitability, and connectivity, often accompanied by synaptic dysfunction.21,53,54 Noninvasive brain stimulation has been reported to alter synaptic efficacy in glutamatergic and gamma-aminobutyric acid (GABA) circuits, which are essential for motor learning. 55 AtDCS could deliver direct constant electrical currents to the cortex to modulate neuronal excitability and cause subsequent changes including increased GABA-mediated inhibition and neuroplastic alterations. 20 Therefore, neuromodulation by AtDCS exhibits promising prospects for motor recovery applications. In our study, we found that contralesional AtDCS improved motor skills following TBI, partly by promoting axonal sprouting through activation of the mTOR pathway. In fact, electrical stimulation of the cortex could also activate the Jak/Stat pathway in the CST. Interestingly, Stat3 improved functional recovery by promoting the formation of CST presynaptic sites/synapses, not axonal sprouting. 11 At present, the mechanism of promoting motor recovery after AtDCS is not fully understood, and further exploration is needed.
Our study has some limitations that need to be addressed. First, in order to eliminate the interference of sex hormones and other underlying diseases on outcomes of TBI mice,56,57 as well as to simulate real-world situations regarding the rising burden of TBI from increases in road traffic incidents predominantly affecting young individuals,1,58 we only selected young, healthy male mice. The selection of homogeneous animals cannot fully reflect the overall manifestation of this condition. Second, it is essential to investigate the optimal stimulation parameters, such as the ideal treatment window and dosage, to improve the efficacy of AtDCS. Third, further research is needed to examine the specific mechanisms underlying the observed effects of AtDCS. Multiple mechanisms must be involved in the reorganization of ipsilesional motor pathways after injury, and these mechanisms need to be further elucidated. Additionally, contralesional AtDCS potential might impact on reticular/bulbar targets, which could offer bilateral projections for recovery in severe cases. 59 The potential role of bulbar-mediated projections in recovery is also worth further exploration. Forthly, we did not conduct a comparison with ipsilesional AtDCS to examine if its impact on the denervated affected spinal cord was diminished, thus questioning its equally value for motor function. 60
Although meta-analyses of clinical stroke trials have revealed some implications of using tDCS in TBI for motor recovery,61,62 the efficacy of tDCS remains inconsistent, and there are relatively few studies on tDCS in motor recovery after TBI. Our study provides evidence for the potential of tDCS to promote motor recovery in a TBI model associated with more diffuse network disruptions. In clinical practice, there are challenges to applying this technique due to individual variability in electrical currents, responses, and optimal stimulation targets. 20 Personalized tDCS might be the future direction for tDCS development for motor recovery after TBI. With the development of computational modeling and software-based signal processing techniques, we believe that individualized tDCS approaches, which could provide optimized stimulation parameters and thus maximize patient recovery, will be possible.
In conclusion, the ability of AtDCS of the contralesional side to improve skilled motor functions following TBI was partly attributed to the promotion of axonal sprouting through increased neuronal activity and thus activation of the mTOR pathway.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Beike Chen: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing.
Qiang Tan: Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing.
Hongyan Zhang: Data curation; Investigation; Methodology.
Weihua Chu: Data curation; Formal analysis; Investigation; Methodology.
Huizhong Wen: Investigation; Methodology; Resources.
Xuelong Tian: Investigation; Methodology; Resources.
Yang Yang: Conceptualization; Data curation; Investigation; Methodology.
Weina Li Data curation; Formal analysis; Investigation; Methodology.
Wenyan Li Data curation; Formal analysis; Investigation; Methodology.
Yujie Chen Conceptualization; Funding acquisition; Resources; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Hua Feng Conceptualization; Funding acquisition; Resources; Supervision; Validation; Visualization.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82371399 to Yang Yang, 82371333 to Yujie Chen), Chongqing Medical Scientific Research Project (Joint Project of Chongqing Health Commission and Science & Technology Bureau, No. 2023GGXM003 to Hua Feng), and State Key Laboratory of Trauma, Burn and Combined Injury (SKLYQ202002 to Yujie Chen)
Ethical Approval
All experiments are reported in compliance with the Animal Research: Reporting in vivo Experiments guidelines. The experimental protocols were approved by the Laboratory Animal Welfare and Ethics Committee of Third Military Medical University (AMUWEC20232691) and performed according to the Guide for the Care and Use of Laboratory Animals.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
