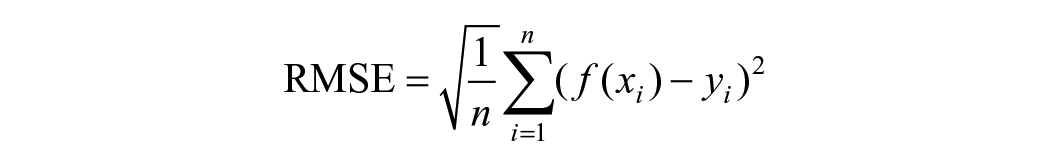Abstract
Background
Stroke can impair manual dexterity, leading to loss of independence following incomplete recovery. Enhancing our understanding of dexterity impairment may improve neurorehabilitation.
Objectives
The study aimed to measure dexterity components in acute stroke patients with and without hand motor deficits, compare them to those of healthy controls (HC), and to explore the neural substrates involved in specific components of dexterity.
Methods
We used the Dextrain Manipulandum to quantify fine finger force control, finger selection accuracy, coactivation, and reaction time (RT). Dexterity was evaluated twice (2 days apart) in 74 patients and 14 HC. Voxel-Lesion-Symptom-Mapping (VLSM) was used to analyze the relationship between tissue damage and dexterity. Results. Due to severe paresis or fatigue, 24 patients could not perform these tasks. In 50 patients (included 4.6 ± 3.3 days post-stroke), finger force control improved (P < .001), as it did in HC (P = .03) who performed better than patients on both evaluations. Accuracy of finger selection did not improve significantly in any group, but the HC performed better on both evaluations. Unexpectedly, coactivation was better in patients than in HC at D3 (P = .03). There were no between-group differences in RT. VLSM showed that damage to the superior temporal gyrus (STG) impaired finger force control while damage to the posterior limb of the internal capsule (PLIC) impaired finger selectivity.
Conclusions
Acute stroke affecting the STG or PLIC impaired selective components of dexterity. Patients with mild to moderate impairment showed better finger force control and accuracy selection within 48 hours, suggesting the feasibility of detecting early dexterity improvements.
Introduction
Dexterity refers to the ability to perform efficient hand and relatively independent finger movements (RIFM) to grasp and skillfully manipulate objects to interact with our ever-changing environment.1,2 Motor impairment of the upper limb (UL), including dexterity impairment,3,4 is a frequent consequence of stroke and has been widely acknowledged as a major cause of disability among adults.3,5 Patients who recover some finger extension and shoulder abduction within 72 hours post-stoke may potentially expect full UL recovery 6 months later. 6 Most recovery of strength and finger individuation occurs within the first 3 months post-stroke; afterward, the recovery of finger strength and individuation are closely correlated up to a strength threshold of approximately 60% of full recovery. 7
Despite the fact that functional recovery occurs during the first post-stroke weeks and months in most patients, 8 55% to 75% of patients retain significant UL impairments 3 to 6 months later. 9 In the chronic phase (>6 months post-stroke), dexterity remains impaired in 62% of patients, 4 which can lead to reduced independence. 10 Hand impairments cause limitations in activities of daily living (ADLs) and decrease the quality of life (QoL).9,11,12 Indeed, the ability to grasp and manipulate objects with precision is necessary for autonomy in most ADLs such as turning a key, eating, dressing up, using a computer, and writing.11,13,14 Hence, recovering manual dexterity after a stroke is crucial for regaining QoL and independence.11,15 Skillfully manipulating objects requires both exquisite motor control, including precise coordination between the fingers, and fine somatosensory processing. Through haptic exploration, we gather and integrate information about the objects we use (their size, weight, and shape), 16 which further refines how skilled we become at manipulating them. The somatosensory and motor systems must be closely coupled to sustain dexterity.16,17
After a stroke, the severity of hand function impairments depends on the location and extent of the lesion. 18 The primary motor cortex (M1) and corticospinal tract (CST) are essential neural structures for the control of RIFMs.16,19 The recovery of selective finger movements correlates with the integrity of the CST.18,20 Additionally, a distributed network involving parietofrontal cortical areas and subcortical structures is involved in the control of dexterity.21,22 Parietal and thalamic lesions can cause somatosensory deficits, leading to loss of tactile sensitivity and dexterity impairments. 23 Damage to the premotor areas can result in difficulties in executing skilled hand movements, as these areas are crucial for planning and coordinating hand movements. 24 Cerebellar strokes can induce dexterity deficits because the cerebellum is involved in movement coordination and timing. 25 Strokes affecting the basal ganglia can also lead to dexterity deficits, especially in tasks requiring precise finger movements.26,27 Finally, cognitive deficits, involving either a network of prefrontal and temporoparietal associative areas, may also impair dexterity.16,18
Because the hand motor repertoire is vast and complex, different components of dexterity can be impaired simultaneously or independently by a stroke. For example, impairment of whole-hand grasping is primarily related to a persistent inability to extend the fingers, notably the thumb and index.16,28 and beyond a certain threshold, strength improvement does not translate into further finger individuation. 7 Dexterity is not easily summarized by a single measure and should rather be explored through its multiple components.13,16,18,20 Recent robots and dexterity devices allow the simultaneous assessment of several dexterity components such as velocity, force control (ie, the capacity to control precisely finger force), 29 finger individuation (ie, the capacity to move the fingers independently of each other), 19 and reaction time (RT). 30 Enhancing our comprehension of these dexterity components and their recovery after stroke could contribute to enhance neurorehabilitation. 13 We aimed to quantify whether and how key components of manual dexterity were affected during the acute/early subacute phase of stroke (acute: post-stroke days 1-7, subacute: between 8 days and 3 months) 31 compared to healthy controls (HC).
Our first goal was to quantify dexterity with a new device in acute stroke patients, and to determine whether changes could be observed after 2 days, in the absence of a specific dexterity training. Our second goal was to compare patients and HC to help interpreting potential changes in dexterity. Our third goal was to analyze magnetic resonance imaging (MRI) data with Voxel-Lesion-Symptom-Mapping (VLSM) to investigate the neural substrates involved in dexterity impairments caused by acute stroke.
Methods
Subjects
This study (ClinicalTrials.gov, NCT04171856) was approved by the local ethics committee. All patients and HC provided written informed consent. Patients admitted to the Stroke Unit of the CHU UCL Namur (Godinne site) were screened for inclusion. The inclusion criteria were as follows: (1) first, unique acute ischemic or hemorrhagic stroke demonstrated by brain imaging; (2) being between 24 hours and 21 days since stroke onset; and (3) being aged 18 to 90 years. Exclusion criteria were (1) multifocal stroke, (2) severe aphasia or cognitive impairment (evaluated according to the NIH Stroke Scale [NIHSS] 32 ), (3) psychiatric disorder, (4) medical instability, and (5) contraindication to MRI (Supplemental Materials). For HC, the inclusion criteria were as follows: (1) 18 to 90 years old, and (2) no neurological condition, and the exclusion criteria were (3) drug/alcohol abuse or (4) psychiatric conditions.
Clinical Assessment
Based on the International Classification of Functioning, Disability, and Health, 33 several clinical scales were used to evaluate motor and cognitive functions. Motor impairment of the UL was assessed with the Fugl-Meyer Upper Extremity (FMA-UE, motor part), 34 global cognitive impairment with the Montreal Cognitive Assessment (MoCA), 35 the bimanual activity limitation of ADL with the Arm Motor Ability Test (AMAT), 36 and the stroke severity at admission with the NIHSS. 32
Study Design and Data Acquisition
This study is an ancillary study of a larger research project investigating motor skill learning during acute stroke (Supplemental Figure 1). Over 3 consecutive days, the patient and HC trained with the REAplan® robot (Axinesis, Wavre, Belgium) on a task emphasizing shoulder and elbow movements, not requiring hand or finger dexterity. The patients benefited from physical therapy for 30 minute/day, typically walking, restoring strength in the limbs, and grasping/releasing objects. We used the Dextrain Manipulandum (The Dextrain® Company, Igny, France https://fr.dextrain.com), a tool designed to assess and train manual dexterity through visuomotor tasks 13 The Dextrain Manipulandum, composed of 5 force sensors connected to pistons, allows simultaneous quantification and recording of the forces applied by each finger (Figure 1A). On the first day after inclusion (D1), several dexterity components were quantified in the contralesional UL or ipsilesional UL in the case of a cerebellar stroke. Each participant was seated in front of a monitor, with their forearm resting on the table and shoulders in a relaxed, slightly flexed, position. They could hold the Dextrain Manipulandum with their preferred grasp as long as the 5 fingertips remained on the pistons. To ensure that contact was maintained with the pistons during finger extension, small flat magnets were taped on each fingertip. The same assessment was repeated on day 3 (D3). HC used their non-dominant hands for the assessment.
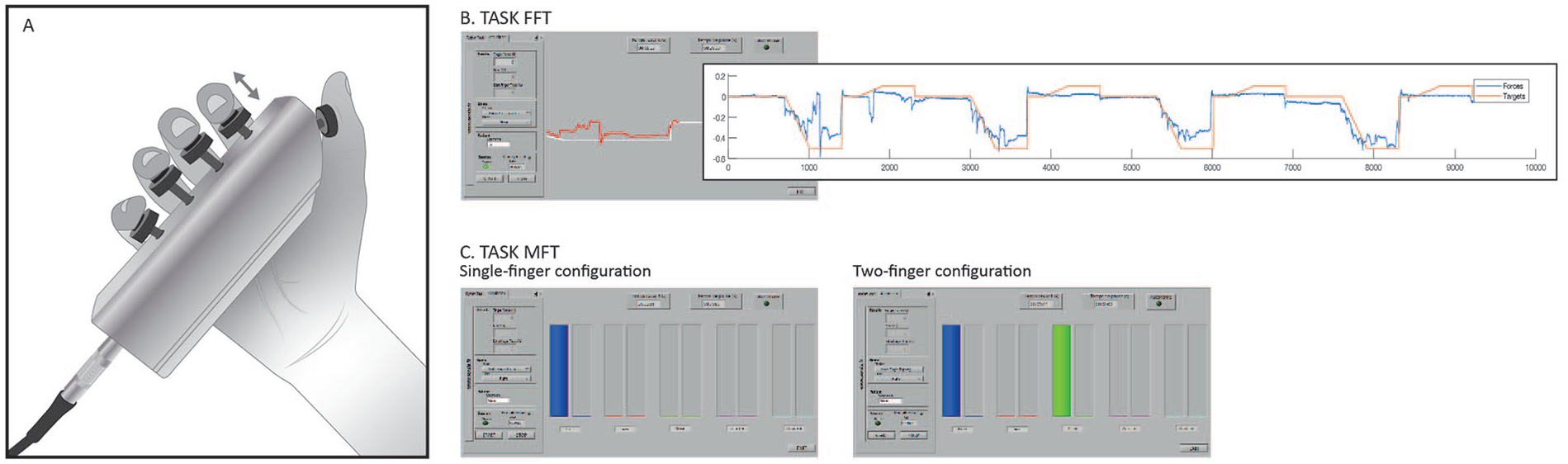
Dexterity tasks. (A) The Dextrain Manipulandum is composed of 5 force sensors connected to pistons that simultaneously allow quantifying and recording the forces applied by each finger. To ensure maintaining contact with the pistons during finger extension, small flat magnets were taped on each fingertip. (B) Finger Force Tracking task (FFT). Subjects controlled a red cursor line which provided real-time feedback about the flexion/extension of the index finger. They were instructed to follow the target, a white line, as closely as possible by pressing (slight flexion) and releasing (slight extension) the piston. (C) Multi-Finger Tapping task (MFT), single-finger (left) and two-fingers (right) press configurations. When the target(s) column(s) was (were) highlighted, subjects were instructed to press the appropriate piston(s) as soon as possible and to release it immediately after. Five columns adjacent to the target columns provided online feedback about the force exerted on each piston.
Finger Force Tracking
First, the subjects performed a finger force tracking task (FFT), which quantifies fine finger force control.13,20 They controlled the red cursor line, providing real-time feedback about the flexion/extension of the index finger. They were instructed to follow the target, a white line scrolling from right to left, as closely as possible by pressing (slight flexion) and releasing (slight extension) the piston (Figure 1B). The trial started with a baseline phase of 7 seconds at 0 N (ideally a perfect balance between index flexion/extension), then the white line moved down over 3 seconds (ramp phase) to the target flexion force of 0.5 N, remained steady during 4 seconds (hold-flexion phase), then the white line moved up over 2 seconds to the target extension force of 0.1 N where it remained steady during 4 seconds (hold-extension phase), and returned over 7 seconds (release phase) to baseline (0 N). This cycle was repeated 4 times (task duration 1 minutes and 33 seconds).
Multi Finger Tapping
The second task was the Multi-Finger Tapping Task (MFT), with single-finger and two-finger tap configurations (Figure 1C).13,20 Five pairs of 2 adjacent columns were displayed on the computer screen. The left column, in each pair, indicated which target piston to tap by switching from uncolored to colored for a duration of 1.5 seconds. The height of this column was fixed at 1 N. The subjects were instructed to tap the appropriate piston when the target column was highlighted as soon as possible and to release it immediately after (no hold). The gauge in the adjacent column provided real-time feedback of the finger force flexion, but there was no need to match the target column’s height. After 2.5 seconds, the next target appeared. First, subjects performed 20 trials in a pseudo-randomized order with a single-finger configuration: 4 trials for each of the 5 fingers, with a rest of 3 seconds after 4 trials. After a short break, subjects performed the next 20 trials with the two-finger configuration, requiring simultaneous tap of the thumb against 1 of the 4 other fingers in turn (4 × 4) or simultaneous index and middle finger tap (1 × 4). Overall, all subjects performed 40 trials (task duration: 1 minutes and 29 seconds).
Data Analysis
Dextrain Outcomes
Custom MATLAB routines (MATLAB® and Statistics Toolbox Release 2019a, The MathWorks Inc., Natick, MA, USA) were used to compute the following outcomes. 13
For FFT, we separately computed the root mean square error (RMSE) for each subject, that is, the continuous difference between the target force and the applied force over 4 cycles (Figure 1B).13,20 The smaller the RMSE, the more accurate the control of the index finger force. Individual RMSE values were averaged (mean ± SD) by group.
For MFT, we computed press error (inverse of finger selection accuracy), coactivation, and RT. 20 First, if a finger applied pressure >0.5 N before the target finger, this was counted as an error. For each target, we compared the specific force peaks associated with each tap. This methodology enabled to quantify the timing, spatial position, and magnitude of force for each tap. The errors were summed across 20 trials for each configuration. For the single-finger configuration, there were 4 potential errors/trials (min = 0, max = 80), and for the two-finger configuration, there were 3 potential errors/trials (min = 0, max = 60).
Coactivation was defined as when the subject first pressed (>0.5 N) the correct piston(s), but when unwanted presses (>0.5 N) were recorded in 1 or several extra fingers. The extra finger presses were summed across 20 trials for each configuration, giving 4 potential coactivations/trials for the single-finger configuration (min = 0, max = 80) and 3 potential coactivations/trials for the two-finger configuration (min = 0, max = 60).
The RT (ms) was calculated only when the correct finger was pressed first. For the two-finger configuration, the 2 correct RTs were averaged by trial. The individual errors, coactivations, and RT were averaged (mean ± SD) by group.
Magnetic Resonance Imaging and Voxel-Based Lesion-Symptom Mapping
MRI data were acquired during the first post-stroke week (Supplemental Methods). Using MRIcron® (version 1.40, NeuroDebian), acute strokes (Diffusion weighted imaging (DWI) positive areas) were manually drawn as volume of interest (VOI) in the native space under the supervision of a neuroradiologist. The lesion map was then normalized according to the DWI volume in the native space to the standard Montreal Neurological Institute stereotactic space (MNI-152) with SPM12®. Because motor control and dexterity impairment do not occur are the same way by damage to the right or left hemisphere. 37 VLSM was performed with unflipped VOIs. To increase the statistical power, the right-sided VOIs were flipped to the left with MRIcron®. In addition, overlay maps were created to compare the topography of strokes in patients able to perform the task versus those unable to perform the task (Supplemental Figure 2).
Statistical Analysis
Statistical analyses of the behavioral data were performed using RStudio Version 1.3.1093© 2009 to 2020. Seven continuous outcomes were analyzed: the logarithm of the RMSE in the FFT, the quantity of errors, coactivations, and RTs in both the single-finger and two-finger configurations of the MFT.
For each outcome, a linear mixed effect model was fit to the data with Time, Group, and the Time x Group interactions as fixed effects and a random intercept for each subject. The models were fitted by the lmer function from the lme4 package and contrasts were calculated through the emmean function to the emmeans package. The following contrasts were computed:
(1) Differences between HC and patients on each day.
(2) D1 to D3 progression for each group to analyze dexterity change.
(3) For the VLSM, a non-parametric rank-order Brunner-Menzel analysis, was conducted to calculate lesion-behavior relationships using the non-parametric mapping toolbox in MRIcron®. Only voxels (expressed in mm3) commonly affected in at least 10% of the patients were considered. The behavioral data tested for lesion-behavior relationships were the D1 and D3 to D1 delta for RMSE from FFT and for the errors and coactivation from MFT. The resulting maps were corrected for multiple comparisons at P < .05, using permutation thresholding (3000 permutations). However, a t uncorrected at P < .001 was considered if the multiple comparison at P < .05 showed no clusters. Finally, we obtained the precise region of interest for impaired behavioral performance according to the MNI coordinates in MRIcron®. 38 These clusters were identified using the Atlas of the Human Brain. 39
Correlations between the clinical scales (MoCA, the FMA-UE, AMAT, and NIHSS) and results at D1 and delta (D3-D1) on the FFT and MFT were computed with Pearson correlations. Estimates from the model and Pearson correlations were accompanied by a 95% confidence interval.
Results
Between February 2021 and February 2023, 453 patients were screened and 379 were excluded (Supplemental Figure 3). Of the 74 patients included, 50 were able to perform the tasks on the Dextrain Manipulandum tasks. These patients had the following characteristics: 4.6 ± 3.3 days post-stroke (mean ± SD), mild UL impairment (FMA-UE: 61.4 ± 6, range: 38-66), and mild cognitive impairment (MoCA: 22.5 ± 5.1, range: 9-30; Supplemental Tables 1 and 2). The 24 remaining patients participated in the study at 7.3 ± 5.4 days post-stroke and were unable to perform the tasks due to severe hand impairment (N = 13) or fatigue (N = 11). Overall, they had moderate to severe UL impairment (FMA-UE: 33.4 ± 27.3, range: 0-66) and cognitive impairment (MoCA: 17.9 ± 7.6, range: 3-30). Additionally, 14 HC aged 67.4 ± 8.2 years were included in the study (see Supplemental Table 3). However, 2 HC were excluded from analysis due to missing data (flow diagram-modified, Supplemental Figure 3). There were no withdrawals or adverse events.
Finger Force Tracking
At D1 and D3, HC achieved a better RMSE than the patients (P = .01 for both timepoints, Table 1, Figure 2A). From D1 to D3, the log RMSE significantly decreased, that is improved, in the HC (from −1.5 ± 0.6 to −1.8 ± 0.4, P = .03, Supplemental Table 4). Likewise, the log RMSE significantly decreased in patients, improving from −0.9 ± 0.7 to −1.3 ± 0.7 (P < .001). The between-group difference in RMSE change was not statistically significant (P = .83).
Summary of Results.
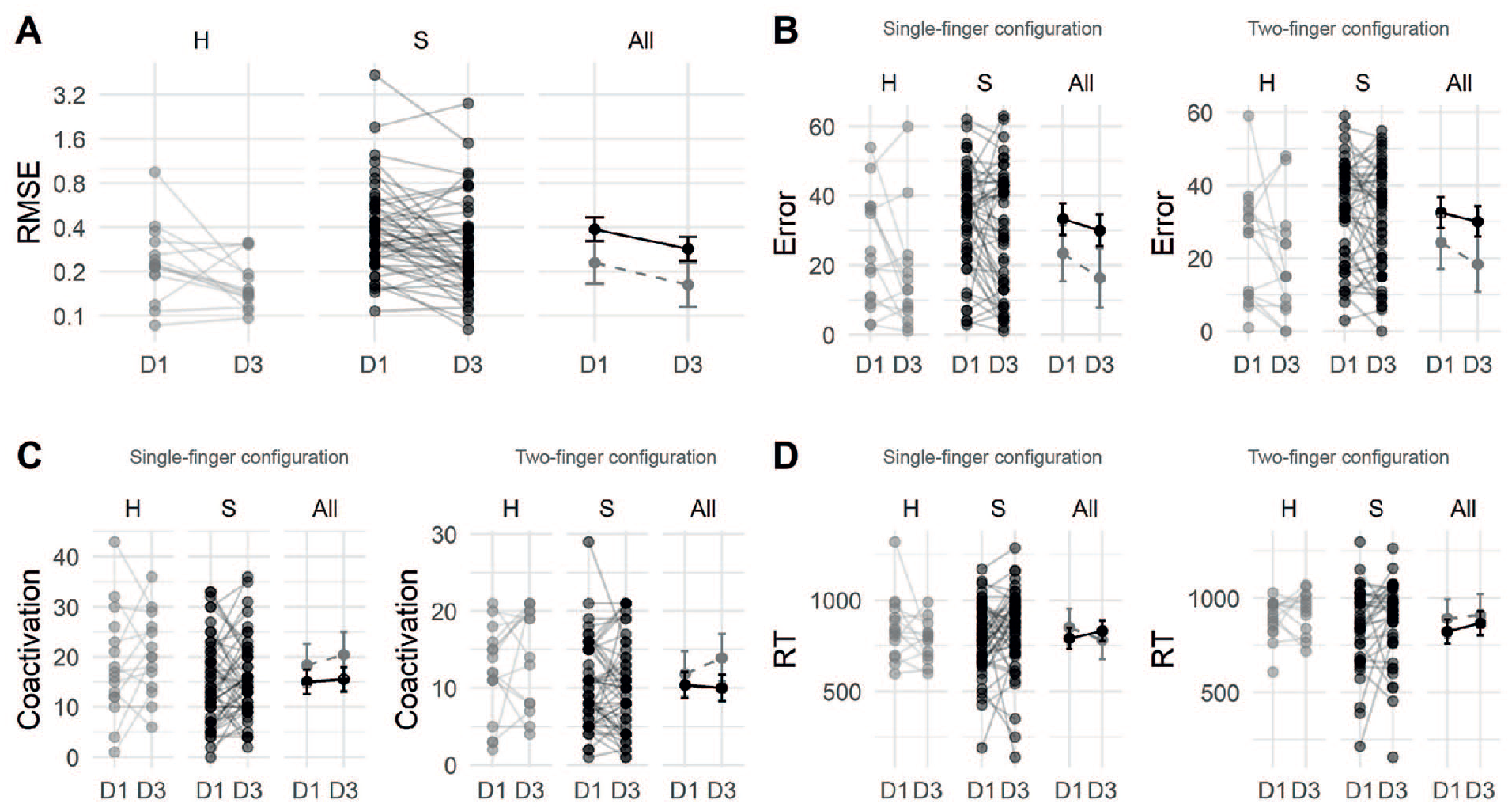
Main results. (A) FFT task. Root mean square error (RMSE), the continuous difference between the target force and the force applied by the index finger over 4 cycles on D1 and D3, in Newton (N). The smaller the raw RMSE, the more accurate the control of the index finger force. From D1 to D3, the fine force control of the index finger improved in healthy controls (HC) and stroke patients (S; here: raw data, 1 line per subject). All: group mean ± SD (gray: HC, black: patients). (B) Number of errors on the MFT task. For the single-finger configuration, the errors were summed across 20 trials (min = 0, max = 80). HC made less errors than the patients on D1 and D2. For the two-finger configuration, the errors were also summed across 20 trials (min = 0, max = 60). (C) Coactivation level (sum of the unwanted presses) on the MFT task in single- and two-finger configuration, same configuration as for the error measure. Unexpectedly, coactivation level increased in both groups the between D1 and D3 in single-finger configuration. For the two-finger configuration, there was a significant difference at D3 between groups in favor of the patients. (D) The reaction time (RT, in ms) was calculated only when the correct finger was pressed first on the MFT task. For the two-finger configuration, the 2 correct RTs were averaged by trial. For the single-finger configuration, RT tended to decrease in HC (not significant, NS) and tended to increase in patients (also NS). In the two-finger configuration, RT tended to increase in both groups (NS).
Multi Finger Tapping
When comparing the single-finger configuration on D1, HC exhibited significantly fewer errors (23.4 ± 16) than did patients (33.3 ± 14.8; P = .04, Table 1, Figure 2B). However, there was no statistical difference between HC and patients regarding the level of coactivation (P = .18, Figure 2C) or RT (P = .30). On D3, HC continued to make fewer errors (16.9 ± 16.2) compared to the patients (30 ± 17.7, P = .01). Again, there was no significant between-group difference at D3 for the level of coactivation (P = .06) and RT (P = .41).
From D1 to D3, the number of errors showed a non-significant decrease in HC (P = .08, Supplemental Table 4) and patients (P = .13). There was no between-group difference in error progression (3.8 ± 4.5, P = .40, Table 1). Between D1 and D3, the coactivation level did not significantly change in HC (P = .34) and patients (P = .69), and there was no significant between-group difference (P = .51). From D1 to D3, the RT did not significantly change in HC (P = .25) or patients (P = .20), and there was no significant between-group difference (P = .10), although there was a decreasing trend in the HC group and an increasing trend in the patient group (Figure 2D).
In the two-finger configuration, on D1, HC tended to make fewer errors (24.4 ± 15.2) than the patients (32.5 ± 13.8, P = .06, Table 1, Figure 2C). Neither the level of coactivation (P = .40) nor the RT (P = .26) was significantly different between HC and patients. On D3, HC made significantly fewer errors (19 ± 15.3) than the patients (30.1 ± 14.9, P = .01). Unexpectedly, for coactivation, the HC group exhibited a higher occurrence of unwanted finger presses (14.3 ± 6.6) compared to the patients (10 ± 5.9, P = .03, Figure 2C). There was no significant between-group difference in RT (P = .49).
From D1 to D3, there was no significant decrease in the number of errors in the HC group (P = .09) or patients (P = .20, Supplemental Table 4). There were no significant between-group differences in progression (P = .37, Table 1).
Coactivation did not significantly change in HC (P = .17) or patients (P = .64); there was no significant between-group difference in progression (P = .15). Finally, the RT did not significantly change in the HC (P = .74) and patients (P = .21), and there was no significant between-group difference in RT evolution (P = .71).
Correlation With Clinical Scales
There was no significant correlation between clinical scales and the FFT or MFT outcomes on D1 nor on D3 to D1 evolution (all P > .07).
Voxel-Based Lesion-Symptom Mapping
In the unflipped-VOI VLSM analyses, no voxels survived the thresholds corrected for multiple comparisons. The following analyses were thus reported at an uncorrected threshold of P < .001, with all VOIs flipped to the left side.
For the FFT task on D1, an acute stroke involving the superior temporal gyrus (STG) was associated with a poorer RMSE (cluster size: 128 mm3, MNI coordinates: 50, −42, and 25, P < .001 uncorrected; Figure 3A).
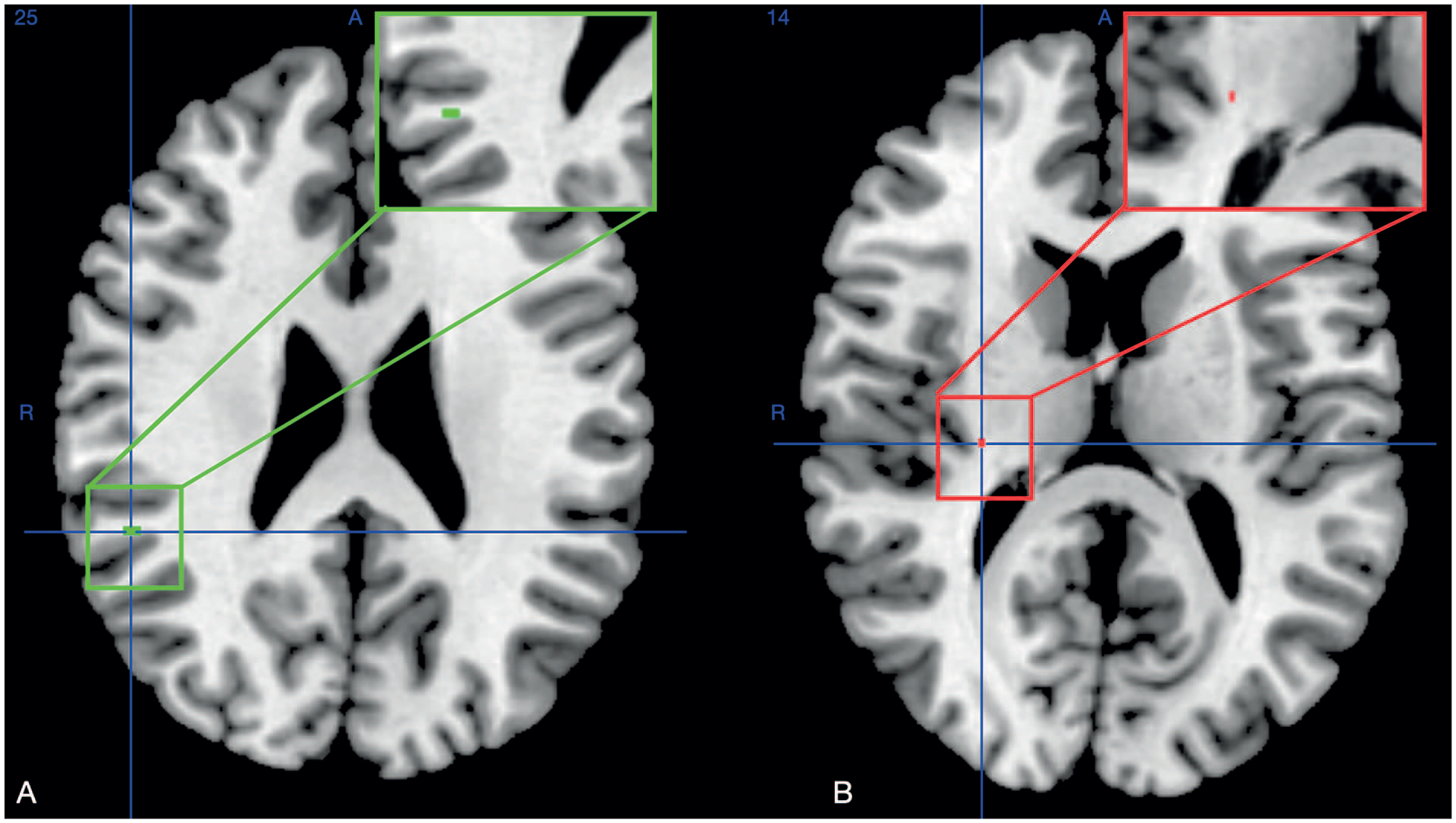
Voxel-based lesion-symptom mapping (VLSM) for the acute stroke patients able to perform the dexterity tasks (N = 48, motion artefacts in 2 patients). All the resulting maps are reported at the uncorrected threshold of P < .001, with right-side flipped VOIs. (A) For the FFT task performed on D1, acute stroke involving the Brodmann area (BA) 21 in the superior temporal gyrus (STG) correlated with poorer RMSE. (B) For the MFT two-finger configuration task on D1, more errors were committed by the patients with a stroke involving the posterior limb of the internal capsule (PLIC) at the border of the putamen.
For the single-finger configuration MFT on D1, there was no significant lesion-behavior relationship. For the two-finger configuration MFT, more errors were committed when the stroke involved the posterior limb of the internal capsule (PLIC) at the border of the putamen (cluster size: 96 mm3, MNI coordinates: 27, −22, and 14, P < .001 uncorrected; Figure 3B). There was no significant correlation between stroke mapping and FFT or MFT evolution.
Discussion
The Dextrain manipulandum enabled the quantification of key parameters of manual dexterity in both HC and acute stroke patients with moderate to mild impairments. Specific components of finger dexterity of the contralesional hand were slightly impaired in patients. After 2 days without specific dexterity training, both groups improved fine finger force control, with the HC group demonstrating greater improvement. The HC also displayed more accurate finger selection in single-finger configuration compared to the patients at D1 and D3. The RT did not change in both groups. Acute stroke involving the STG impaired fine finger force control, while involvement of the PLIC decreased finger selection accuracy.
Finger Force Tracking
The 50 patients recruited at the (sub)acute phase of stroke were able to achieve the complex FFT task with their contralesional hand or ipsilesional hand in case of a cerebellar stroke (N = 3). HC of the same age group had better index finger force control with their non-dominant hand on both D1 and D3 and were able to significantly improve it on D3. Similarly, finger force control improved in 50 patients who had no or mild UL motor impairment (FMA-UE score: 61.4 ± 6) and received standard stroke unit physiotherapy (30 minute/day). Hence, even in the presence of acute dexterity impairments and the lack of specific dexterity training, it was possible to detect improvements in finger force control after 48 hours with the Dextrain Manipulandum in the setting of a stroke unit, and in HC. Exposure to the FFT was short (~1.5 minute) on D1 and D3. In HC, finger force control improved from D1 to D3, likely highlighting their ability to learn new motor skills (here: the performance of the FFT) through small amounts of practice and repetition.40-42 Finger force control also improved in acute stroke patients despite the absence of specific training, they might thus also have achieved motor skill learning, even with these short exposures. Alternatively, they might also have benefited of a small amount (2 days) of spontaneous recovery, which could support the theory of the “enhanced plasticity window” during the (sub)acute stroke phase.3,43 Interestingly, there was no significant correlation between the clinical scales (NIHSS, FMA-UE, AMAT, and MoCA) and finger force control. Currently, it is not clear whether this impaired finger force control would have fully recovered over time, either spontaneously or following neurorehabilitation, or whether it would have gone unnoticed owing to the limitations of the clinical assessments. Further investigations are needed to determine the possible long-term persistence and consequences of this impairment.
VLSM analyses showed that precise finger force control was significantly more impaired when acute stroke involved the STG. Interestingly, the VLSM voxels were located close to the white matter. The main white matter pathway associated with the STG is the arcuate fasciculus (AF), which connects the frontal, parietal, and temporal lobes.44,45 In the right hemisphere, the AF plays a key role in visuospatial processing and some aspects of language processing. 46 At first look, it is surprising that neither the M1 nor the subcortical CST, which are crucial neural substrates of dexterity, 16 were revealed to be critical for finger force control by these analyses. This might be partly because, in this cohort with a high FMA-UE score (61.4 ± 6), 13 patients had no clinically detectable UL motor deficit (FMA-UE = 66). Alternatively, the performance of such a complex FFT task, requiring precise finger force control as well as spatial and temporal integration of the online visual feedback, requires an intact higher-order integrative network upstream of the motor execution system. 47 The STG is positioned where the “what” and “where” streams of cortical visual processing intersect and receives input from both streams, which suggests that the STG is involved in the integration of information from multiple sensory modalities.48-50 Furthermore, the STG is involved in hand movement, grasping observation, ideation of simple finger movement,51,52 and hand movement coordination in response to visual stimuli. 53 Thus, an acute stroke upstream of M1 and the CST might induce subtle dexterity impairments that were not detected by the clinical scales we used, but that would potentially have been identified with specific dexterity assessments (eg, 9 Hole Peg Test and pinch dynamometer).
One-third (N = 24) of the patients (N = 74) were unable to perform the dexterity tasks because their hand was too paretic (N = 13) or because they were overly fatigued (N = 11). Compared to the 50 remaining patients (t-tests), they had more severe UL impairment (FMA-UE: 33.4 ± 27.3, P < .001), but similar cognitive impairment (MoCA: 17.9 ± 7.6, P = .12), and were recruited for the study later (post-stroke days: 7.3 ± 5.4 vs 4.6 ± 3.3, P = .005). It is likely that with a longer follow-up period, some patients would have recovered at least some finger force control, especially those who were overly fatigued. However, even at 6 months post-stroke, many patients still struggled with impaired dynamic precision in grip control. 10
Multi Finger Tapping
In both tested configurations, the MFT requires rapid target identification, fast and accurate activation of the appropriate finger(s), and selective activation (ie, with the lowest possible coactivation of unwanted fingers). In terms of accuracy, patients were impaired compared to the HC group at several time points, but there was no between-group difference in the lack of significant progression from D1 to D3. The accuracy of finger selection on D1 was more impaired in patients whose stroke involved the PLIC, at the border of the putamen (MNI coordinates: 27, −22, and 14), well in line with the role of the CST in dexterity and RIFM.16,18,26,54,55 The PLIC is a prominent white matter tract that contains ascending (sensory) and descending (motor) projections. Thalamocortical fibers transmit information from the thalamus to the primary somatosensory cortex through the PLIC. 56 Stroke in the PLIC can impair proprioception (MNI coordinates: 27, −21, and 24) and light touch (27, −19, and 14), and stroke of sensory fibers in the superior thalamocortical radiations and parietal operculum is associated with exteroceptive and proprioceptive deficits in the UL/hand. 57 Thus, acute stroke to the thalamocortical projections in the PLIC could have contributed to the dexterity impairment in the current study. It is worth noting that, in human brain imaging and monkey tracing studies, the CST bundles originating from different cortical areas as well as thalamocortical projections overlap substantially in the PLIC.58-60
The RT, quantifying the speed for exclusively pressing the correct finger(s), was not significantly different between the HC group and patients. However, from D1 to D3, in the single-finger configuration, the RT tended to decrease (improve) in HC and to increase in patients, although neither change was significant. From D1 to D3, the RT remained stable in both groups and tend to increase in two-finger configuration, which could reflect the difficulty of the task. Prolonged MFT training is required to significantly improve RT.
The level of coactivation (ie, pressing with unwanted fingers) was significantly better in the patients than HC, exclusively on D3 and in the two-finger configuration. No other significant differences were observed. These observations are counterintuitive because 1 hallmark of post-stroke dexterity impairment is a loss of finger selectivity, leading to increased coactivation.13,55 It is possible that the dexterity impairment was too slight in these 50 patients to demonstrate worse performance than in HC. As for RT, this may be due to a lack of extensive training, which might reveal a progressive between-group difference.
Study Limitations
This study had several limitations. First, the 50 patients able to use the Dextrain Manipulandum had mostly mild or absent UL deficits, which limited the group’s dexterity impairments compared to the HC group (ceiling effect). Since the patients were evaluated during the acute stroke phase, we intentionally kept the experimental session duration around 40 minutes, which resulted in a lack of time to perform other clinical dexterity assessments. This practical constraint may have contributed to the absence of correlations, highlighting the need for future research to incorporate specific clinical scales for a more comprehensive evaluation. Next, the FFT and MFT tasks are challenging even for the HC, which may explain why one-third of patients were unable to achieve it. Thus, their data could thus not be included in the behavioral and VLSM analyses. However, as can be appreciated in Supplementary Figure 2, the topographic maps of the acute stroke lesions were very similar and grossly centered on the PLIC/putamen in both the 48 patients who performed the dexterity tasks (row B) and the 24 who could not (row C). We are thus confident that if these 24 patients would have been able to minimally perform the dexterity tasks and included in the VLSM analysis, it would have reinforced rather than modified the main results of the current VLSM analysis. Assessing the patients longitudinally (eg, at 1, 3, and 6 months post-stroke) would provide finer details about the evolution of dexterity outcomes. Finally, another potential limitation is the relatively short measurement sessions and the small number of trials conducted, which could reduce the statistical power of the findings.
Conclusions
Different components of manual dexterity were quantified in patients with acute stroke. Compared to HC, the patients exhibited impaired finger force control and finger selection accuracy. Finger force control improved in both patients and HC, but HC consistently outperformed patients. VLSM revealed that acute stroke to the STG and PLIC was associated with impairments in different dexterity components. These findings highlight the importance of quantifying specific dexterity impairments, particularly in patients with moderate to mild strokes; however, this might not be applicable for patients with severe strokes.
Supplemental Material
sj-docx-4-nnr-10.1177_15459683241230029 – Supplemental material for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates
Supplemental material, sj-docx-4-nnr-10.1177_15459683241230029 for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates by Eloïse Gerardin, Maxime Regnier, Laurence Dricot, Julien Lambert, Coralie van Ravestyn, Béatrice De Coene, Benoît Bihin, Påvel Lindberg and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
sj-jpeg-3-nnr-10.1177_15459683241230029 – Supplemental material for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates
Supplemental material, sj-jpeg-3-nnr-10.1177_15459683241230029 for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates by Eloïse Gerardin, Maxime Regnier, Laurence Dricot, Julien Lambert, Coralie van Ravestyn, Béatrice De Coene, Benoît Bihin, Påvel Lindberg and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
sj-jpg-1-nnr-10.1177_15459683241230029 – Supplemental material for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates
Supplemental material, sj-jpg-1-nnr-10.1177_15459683241230029 for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates by Eloïse Gerardin, Maxime Regnier, Laurence Dricot, Julien Lambert, Coralie van Ravestyn, Béatrice De Coene, Benoît Bihin, Påvel Lindberg and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
sj-jpg-2-nnr-10.1177_15459683241230029 – Supplemental material for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates
Supplemental material, sj-jpg-2-nnr-10.1177_15459683241230029 for Dexterity in the Acute Phase of Stroke: Impairments and Neural Substrates by Eloïse Gerardin, Maxime Regnier, Laurence Dricot, Julien Lambert, Coralie van Ravestyn, Béatrice De Coene, Benoît Bihin, Påvel Lindberg and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We are grateful to the patients with stroke patients and HCs who participated in the study. We thank Maxime Teremetz for providing help and support with the Dextrain manipulandum and for assisting with software development.
Author Contributions
Eloïse Gerardin: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Software; Validation; Visualization; Writing—original draft; Writing—review & editing. Maxime Regnier: Formal analysis; Supervision. Laurence Dricot: Software; Supervision; Validation; Visualization. Julien Lambert: Formal analysis; Software; Supervision; Validation. Coralie van Ravestyn: Investigation. Béatrice De Coene: Software; Supervision. Benoît Bihin: Formal analysis; Methodology; Software; Supervision; Validation; Visualization; Writing—review & editing. Pavel Lindberg: Software; Supervision; Validation; Writing—review & editing. Yves Vandermeeren: Conceptualization; Formal analysis; Funding acquisition; Methodology; Project administration; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PGL has patented the method for measurement of manual dexterity (EP3860459A1) and is a founding member of start-up company Dextrain but receives no financial compensation for advisory role. YV is a member of the scientific committee of the Dextrain company but receives no financial compensation for advisory role.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of YV is supported by the following grants: Fonds de la Recherche Scientifique—FNRS 1.R.506.16, 1.R.506.18, 1.R.506.20F and 1S00722F, Fonds Spécial de Recherche (FSR) grant from the UCLouvain, and Fondation Mont-Godinne grants (2019-2023). The work of EG is supported by the PDR-FNRS grant T.0239.19. The work of CvR is supported by a FSR grant from the UCLouvain (2022-2023), and Fondation Mont-Godinne grant (2022-2023).
Ethical Approval and Consent to Participate
After providing written informed consent and obtained from each participant, all procedures were approved by the hospital CHU UCL Godinne (No: B039201421432) and complied with the Declaration of Helsinki. Clinical Trial number: NCT04171856.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.