Abstract
Background
Rest-activity rhythm (RAR) is a modifiable behavioral factor associated with affect and cognition. Identifying RAR characteristics associated with affect and cognition among stroke survivors provides insight into preventing poststroke affective and cognitive impairment.
Objective
To examine the associations of RAR characteristics with affect and cognition among community-dwelling stroke survivors.
Methods
Forty participants with mild stroke (mean age = 52.8; 42.5% female; 55% White) reported their affect and cognitive complaints using ecological momentary assessment and wore an accelerometer for 7 consecutive days and completed the National Institutes of Health Toolbox Cognition Battery. RAR characteristics were extracted using parametric and non-parametric approaches. Multivariable linear regressions were used to identify RAR characteristics associated with affect and cognition.
Results
Later onset of rest (B = 0.45; P = .008) and activity (B = 0.36; P = .041) were positively associated with depressed affect. These associations were reversed for cheerful effect (rest onset: B = −0.42; P = .017; activity onset: B = −0.39; P = .033). Cheerful affect was also positively associated with relative amplitude (ie, distinctions in activity levels between rest and activity; B = .39; P = .030). Intra-daily variability (ie, RAR fragmentation; B = 0.35; P = .042) and later onset of activity (B = .36; P = .048) were positively associated with cognitive complaints. Less erratic RAR was positively associated with fluid cognition (B = 0.29; P = .036); RAR fragmentation was positively associated with crystallized cognition (B = 0.39; P = .015).
Conclusions
We identified RAR correlates of affect and cognition among stroke survivors, highlighting the value of managing RAR and sleep in stroke rehabilitation. Future studies should test whether advancing the onset of rest and activity, promoting a regular active lifestyle, and improving rest and sleep in the nighttime protect stroke survivors from affective and cognitive impairment.
Introduction
Poststroke affective and cognitive impairments are associated with poor outcomes, such as higher mortality,1,2 lower quality of life,3,4 and a higher risk of stroke recurrence.5,6 Despite rehabilitation efforts, residual affective and cognitive impairments are consistently widespread among stroke survivors, affecting about one-third of the population.7,8 Stroke rehabilitation practice and research typically focus on waking behaviors that constitute two-thirds of stroke survivors’ lifetime. Recently, there has been growing interest in research that addresses the other one-third of life, that is, sleep. Activity and sleep are intertwined such that they follow a pattern regulated by circadian rhythms, known as rest-activity rhythm (RAR). 9 Therefore, studying waking and sleep behaviors separately may hinder us from capturing a complete picture of stroke survivors’ life and lead to missed opportunities to prevent and reduce affective and cognitive impairment.
Circadian rhythms are under endogenous control of the hypothalamic suprachiasmatic nucleus, synchronizing internal biophysiological processes (eg, sleep-wake function, metabolism, and inflammation) with the external environment through exposure to the light-dark cycle. 10 RAR is the most evident behavioral marker of circadian rhythms in humans 9 and consists of a variety of characteristics, such as when one get going in the morning (up-mesor) or settles for the night (down-mesor) and how fragmented rest-activity periods are within a day (intradaily variability). 11 RAR disruption is commonly seen in people who do shift work and who have chronic diseases, and neurodegeneration, linked to poor health including worse affective and cognitive outcomes. 12 For instance, Smagula et al 13 found that less robust RAR contributes to later increases in depressive symptoms among older men; Xiao et al 14 found that more fragmented RAR is associated with accelerated cognitive decline in older women.
Circadian rhythm disruption is both a risk factor and consequence of stroke. 15 One study 16 found that stroke patients exhibited more fragmented RAR compared to matched non-stroke controls. Preclinical studies have identified mechanisms in which circadian rhythm disruption interferes with key functions in stroke pathophysiology (eg, neurogenesis, angiogenesis, and immune response) and impairs stroke recovery.15,17,18 Given the established associations of RAR with affective and cognitive impairment in other populations, it is plausible that RAR disruption is a contributor to the high prevalence of poststroke affective and cognitive impairment. However, little is known about the associations between RAR and affective and cognitive impairment among stroke survivors due to a dearth of research. Understanding these associations has significant clinical implications because RAR is exogenously modifiable 19 and thus a potential treatment target; clinical trials in dementia patients have shown that entraining circadian RAR leads to improved mood 20 and cognitive performance. 21
To our knowledge, only 1 study 22 has examined the association of RAR with depressive symptoms among stroke survivors, and none that has examined RAR correlates of poststroke cognitive functioning. The study conducted by Stahl et al 22 found that stroke survivors with more fragmented RAR were more likely to have depressive symptoms. However, depressive symptoms were assessed using retrospective questionnaires, which require a summative recollection of past experiences and are thus subject to recall biases, particularly when episodic memory deficit is prevalent after stroke. 23 Moreover, accurate assessment of affect requires repeated measurement as it fluctuates within and across days. 24 Ecological momentary assessment (EMA), 25 which leverages mobile health technology to repeatedly sample experience (eg, affect and cognitive complaints) as people go about their lives, overcomes these limitations by providing a more ecologically valid assessment. Thus, this study aimed to leverage EMA methodology to examine the associations of RAR characteristics with affect and cognition among stroke survivors.
Methods
Study methods and results are reported following the Strengthening the Reporting of Observational Studies in Epidemiology (see Supplemental STROBE) statement for observational studies. 26
Participants
Community-dwelling stroke survivors were recruited from a stroke registry. Inclusion criteria were 18 to 65 years old, mild to moderate stroke [National Institutes of Health Stroke Scale (NIHSS) 27 ≤15], ≥3 months post-stroke, pre-morbid modified Rankin scale 28 ≤2, owning a smartphone, and adequate communication capability to make responses to yes/no questions on the phone (to screen for aphasia). 29 Exclusion criteria were previous neurologic/psychiatric disorder, severe apraxia (Apraxia Screen of Test for Upper-Limb Apraxia 30 <6), neglect (Star Cancellation Test 31 ≤44), vision impairment (Lighthouse Near Visual Acuity Test 32 ≤20/100 corrected vision), and hearing impairment (sound repetition screens 33 >4). We excluded users of wheeled mobility devices because of our use of a thigh-worn accelerometer. The Institutional Review Board approved the study and all participants provided written informed consent.
Study Procedure
Participants visited the laboratory to complete screening and were instructed to install the Participation in Everyday Life Survey App 34 on their smartphones for EMA and were fitted with an accelerometer (activPAL) on the anterior, unaffected thigh. Participants were told to wear the accelerometer during all waking and sleeping hours and document any non-wear periods. For the next 7 days, participants were prompted to complete 8 EMA surveys, randomly distributed at 2-hour intervals from 8 AM to 10 PM. This sampling schedule allows a comprehensive capture of daily activities while meeting the recommendations for monitoring free-living physical activity in adults.35,36 After the 7-day monitoring period, participants revisited the laboratory to complete assessments including the NIH Toolbox Cognition Battery 37 and return study equipment.
Measures
Accelerometry: Rest-Activity Rhythms Characteristics
Participants wore an activPAL (PAL Technologies, Ltd) monitor on their unaffected thigh for 7 consecutive days. The activPAL is a small and lightweight tri-axial accelerometer with a dynamic range of ±2 gravitational units and a sampling frequency of 20 Hz. Activity data in 15-second epoch were exported and analyzed. Only participants with adequate data were analyzed, defined as at least 3 valid consecutive days of recording. 38 A valid day was defined as not missing more than a 4-hour period of data. Among a panel of RAR measures, we prioritized selecting RAR measures with more readily practical implications for rehabilitative practice. For instance, we computed activity during the most active 10 hours (M10) and the least active 5 hours (L5) over mesor (ie, the 24-hour average of the activity level) to assess activity because they provide more specific information about activity concerning the period of the day. This helps with clinical implications by informing appropriate strategies based on time and context.
We implemented the non-parametric approach 39 using nparACT package and the parametric approach 40 using RAR package in R to extract RAR characteristics: with the non-parametric approach, we calculated intra-daily variability (fragmentation of 24-hour rhythm), inter-daily stability (rhythm stability across days), the time occurrence and corresponding activity during the most active 10 hours (M10) and the least active 5 hours (L5), and relative amplitude (normalized difference between M10 activity and L5 activity, reflecting rhythm strength); with the parametric approach, we fitted sigmoidally transformed cosine curves representing a 24-hour period to the accelerometer data and estimated up-mesor (the time when activity passes up through the mean activity level of the fitted curve when the individual “gets going” in the morning), down-mesor (the time when activity passes down through the mean activity level of the fitted curve when the individual “settles down” for the night), and pseudo-F statistic (goodness of fit of the raw activity data to the fitted curve, reflecting rhythm robustness). Table 1 details description of the RAR characteristics. Figure 1 visually presents these parameters from 2 participants with more versus less robust RAR for illustrative purposes.
Descriptions Rest-Activity Rhythm (RAR) Characteristics and Their Descriptive Statistics of the Study Sample.
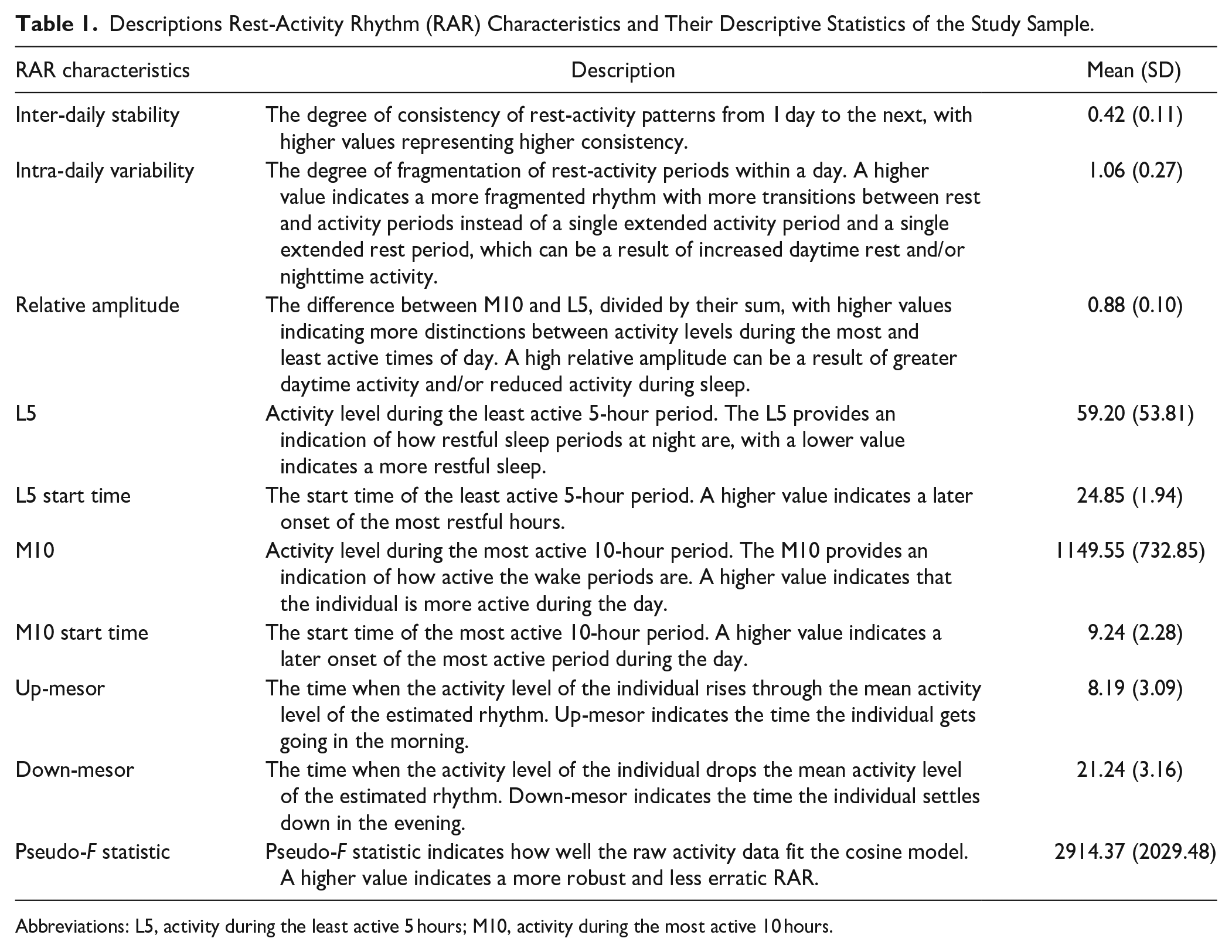
Abbreviations: L5, activity during the least active 5 hours; M10, activity during the most active 10 hours.
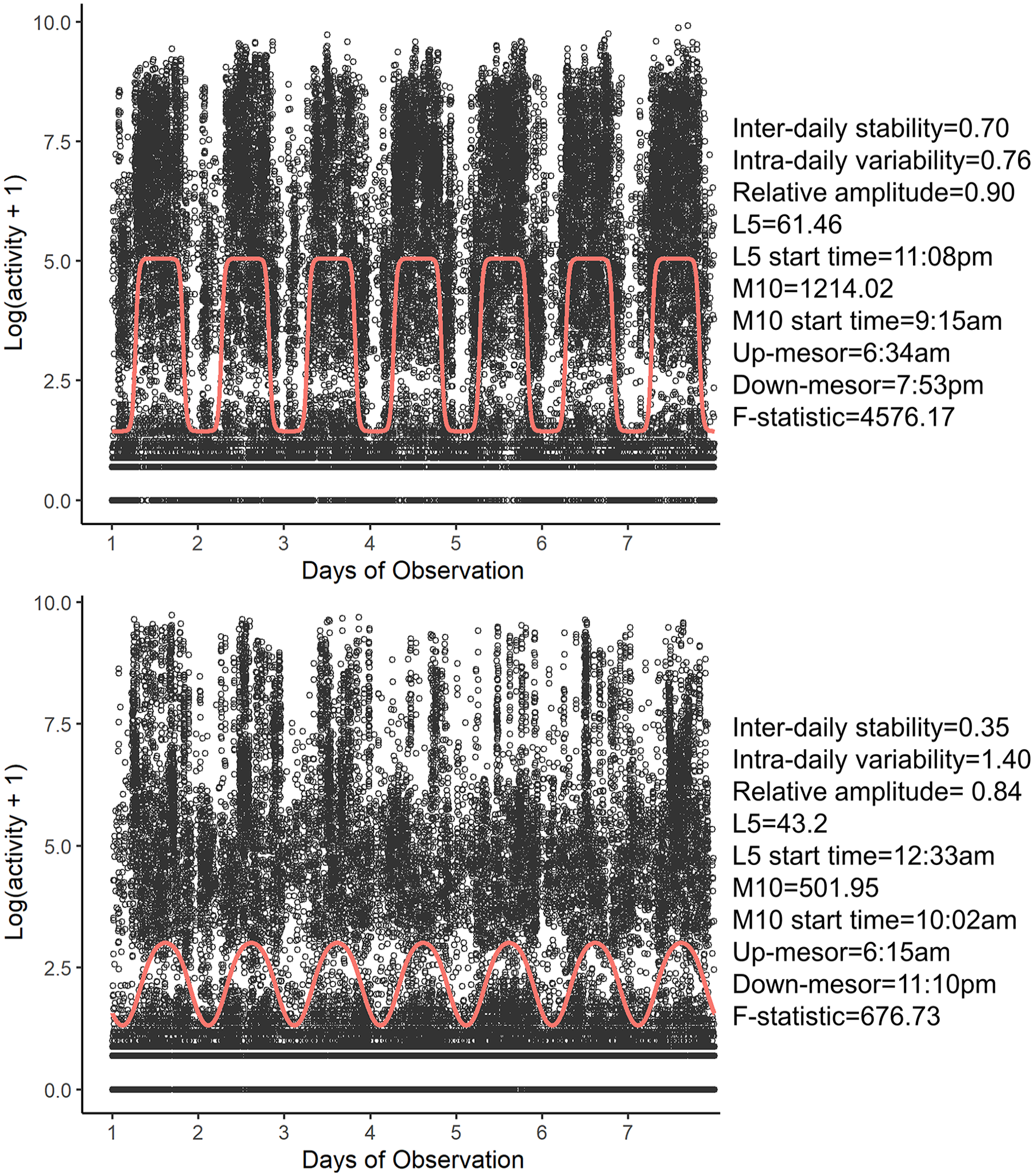
Accelerometer data and modeled rest-activity rhythm (RAR) characteristics from 2 participants. Dots represent 15-second epoch accelerometer data; line represents fitted cosine curve denoting RAR. Top: a subject with a more robust RAR (higher Pseudo-F statistic) a with defined rest and activity pattern; bottom: a subject with a less robust RAR (lower Pseudo-F statistic) with a less defined rest and activity pattern.
EMA: Affect and Subjective Cognitive Complaints
Each EMA survey contains 16 brief questions, assessing daily activity, motivation, and symptoms, that takes an average of a minute to complete. For the purpose of this study, we selected 2 questions assessing depressed and cheerful affect. Depressed and cheerful affect during the day were measured using single-item EMA measures: depressed affect (“Right now, I feel depressed”) and cheerful affect (“Right now, I feel cheerful”). We adopted the items by selecting and modifying questions with close literal meanings to depressed and cheerful affect in the Patient Health Question-9 41 and the neurology quality of life (Neuro-QoL) positive affect and well-being. 42 “Right now” was added to align the question with the EMA’s in-the-moment context. An item assessing cognitive complaints (“Right now, my thinking is slow”) was adopted from the Neuro-QoL cognition function. 43 All items were rated on a 5-point scale from 1 (not at all) to 5 (very much) and were found to have good validity with standardized measures in the stroke population. 44 Mean scores across the monitoring period were aggregated for each individual.
NIH Toolbox Cognition Battery: Objective Cognitive Functioning
The NIH Toolbox Cognition Battery 37 is a computerized assessment that measures fluid and crystallized cognition. Crystallized cognition is assessed by the Picture Vocabulary Test (receptive vocabulary) and the Oral Reading Recognition Test (verbal knowledge). Fluid cognition is assessed by the Picture Sequence Memory Test (episodic memory), the Pattern Comparison Processing Speed Test (processing speed), the List Sorting Working Memory Test (working memory), the Flanker Inhibitory Control and Attention Test (inhibitory control), and the Dimensional Change Card Sort Test (cognitive flexibility). The details of each test and the validity of the NIH Toolbox Cognition Battery in people with neurological conditions are reported elsewhere. 45 Standard scores with a normative mean of 100 (SD = 15) were generated; higher scores reflect better cognitive functioning.
Statistical Analyses
Separate multivariable linear regression models were developed to examine associations of each RAR characteristic with affective and cognitive outcomes. Due to a small sample size, we limited the number of covariates in the model to preserve power. All models were adjusted for major covariates, including age, sex, and race, that are consistent with prior studies examining RARs. Since all participants were of mild stroke severity (NIHSS ≤5) at the time of study, we did not include stroke severity as an additional covariate. All tests were 2-tailed using an alpha level of .05.
Results
Forty stroke survivors with mild stroke (mean NIHSS at the time of study = 0.70) were enrolled. As shown in Table 2, the average age was 52.8 years old and the average years of education was 14.7 years. The mean pre-morbid modified Rankin score was 0.1. The majority were male (57.5%, n = 23), White (55%, n = 22), and ischemic stroke (70.0%, n = 42). On average, participants completed 93.6% of the scheduled EMA surveys. The mean score of EMA-measured depressed affect, cheerful affect, and cognitive complaints were 1.27, 3.15, and 1.39, respectively. The mean standard score of fluid cognition and crystallized cognition were 91.08 and 106.98, respectively. All participants provided adequate accelerometer data and complete NIH Toolbox data. Thus, all participants were included in the analyses.
Participant Characteristics.

Abbreviations: EMA, ecological momentary assessment; mRS, modified Rankin scale; NIH, National Institute of Health; NIHSS, National Institutes of Health Stroke Scale;
As shown in Table 3, a later start of L5 (standardized β = .45; P = .008) and M10 (standardized β = .36; P = .041) were associated with higher levels of EMA-measured depressed affect. On the contrary, a later start of L5 (standardized β = −.42; P = .017) and M10 (standardized β = −.39; P = .033) and higher up-mesor values (ie, a later “get going” time; standardized β = −.40; P = .018) were associated with lower levels of EMA-measured cheerful affect. Additionally, higher relative amplitude (rhythm strength) was associated with higher levels of EMA-measured cheerful affect (standardized β = .39; P = .030).
Associations Between Rest-activity Rhythm Characteristics and Affect.
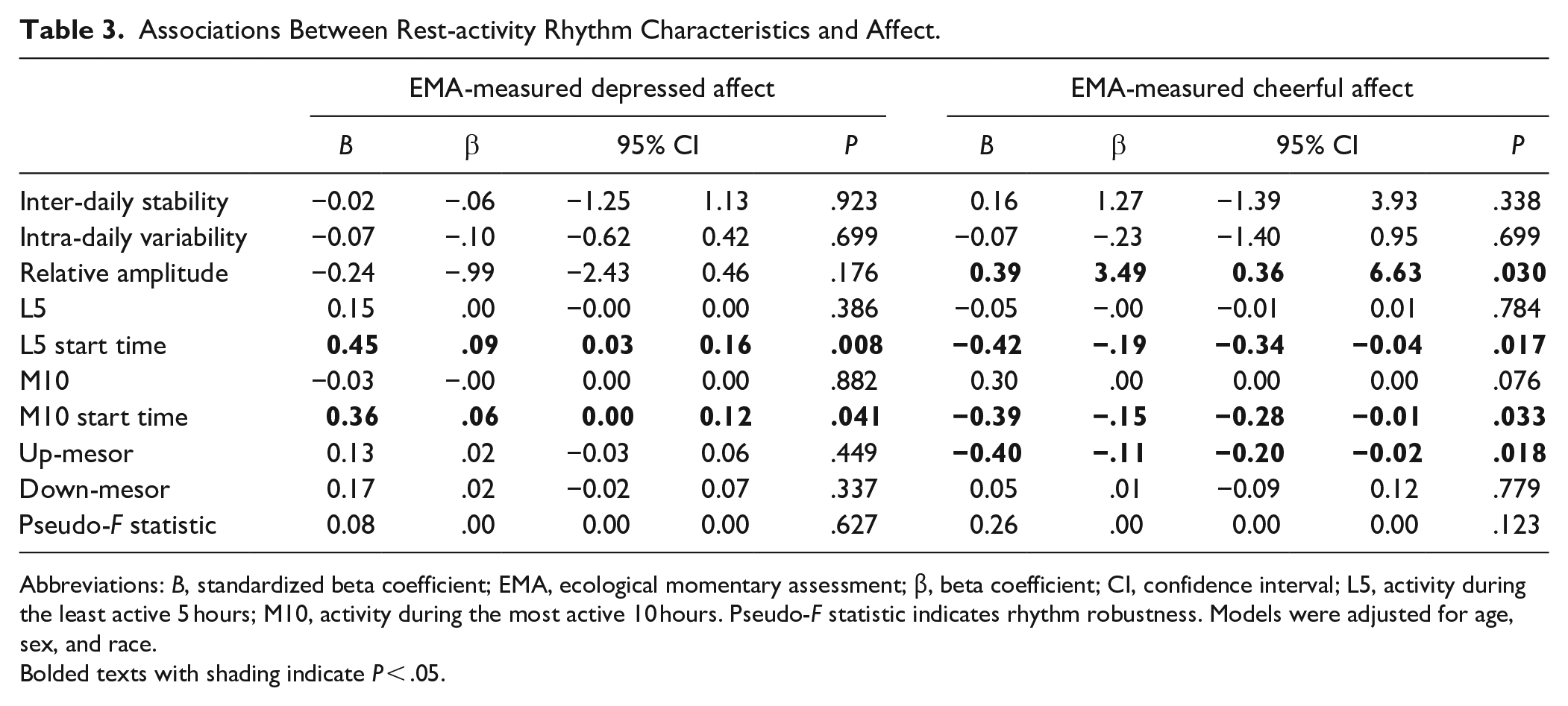
Abbreviations: B, standardized beta coefficient; EMA, ecological momentary assessment; β, beta coefficient; CI, confidence interval; L5, activity during the least active 5 hours; M10, activity during the most active 10 hours. Pseudo-F statistic indicates rhythm robustness. Models were adjusted for age, sex, and race.
Bolded texts with shading indicate P < .05.
As shown in Table 4, higher intra-daily variability (standardized β = .35; P = .042) and a later M10 start time (standardized β = .36; P = .048) were associated with higher levels of EMA-measured cognitive complaints. Pseudo F-statistics (rhythm robustness) was positively associated with fluid cognition, and intra-daily variability of rhythm was positively associated with crystallized cognition.
Associations Between Rest-activity Rhythm Characteristics and Cognition.
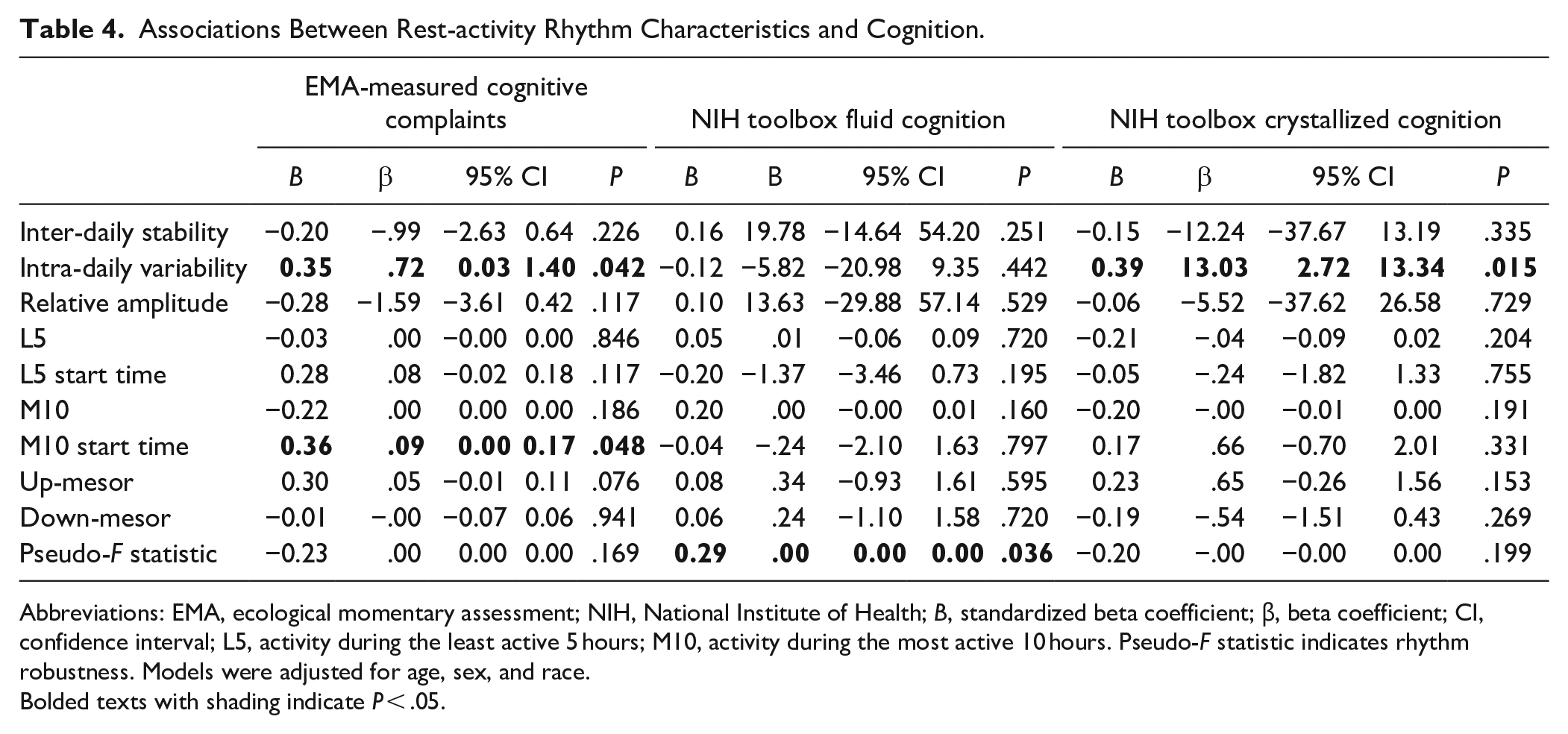
Abbreviations: EMA, ecological momentary assessment; NIH, National Institute of Health; B, standardized beta coefficient; β, beta coefficient; CI, confidence interval; L5, activity during the least active 5 hours; M10, activity during the most active 10 hours. Pseudo-F statistic indicates rhythm robustness. Models were adjusted for age, sex, and race.
Bolded texts with shading indicate P < .05.
Discussion
This study adds to the growing literature about poststroke RAR by identifying that RAR timing and rhythmicity were associated with affect and cognition among stroke survivors. In addition, RAR fragmentation was associated with greater cognitive complaints but better crystallized cognition performance. Our findings provide support for expanding the focus of stroke rehabilitation beyond waking hours and suggest the potential of RAR as a treatment target to improve stroke outcomes such as affect and cognition.
Consistent with prior literature, 46 we found that a later timing of RAR, as indicated by later L5 (least active 5 hours), M10 (most active 10 hours), and up-mesor (get going time), was positively associated with better affect among stroke survivors. These observations are in line with the literature that eveningness (later onset of rhythms) is linked with poorer affective wellbeing. 47 Eveningness can be an indication of a desynchrony in the circadian process and environmental demands or a mismatch between biological and social times, which leads to cortical secretion disruption and mood distress.48,49 Moreover, since the majority of community activities (eg, work, volunteering, and religious services) happen in the daytime rather than later in the day, starting the day later might denote having less opportunity to engage in community, which is associated with greater depressive symptoms. 50 In addition, a later onset of sleep-wake activities might be due to a lack of planning and opportunities for daily activity participation. As a result, there may be less intention and motivation to get out of bed and start the day. Several other factors, such as medication schedule, can also affect the onset of sleep-wake activities. As some of these factors are modifiable, opportunities exist for future research to identify and modify key factors to enhance RAR. The finding that relative amplitude was positively associated with cheerful affect is consistent with evidence showing that RAR is dampened in patients with affective disorders. 51 Having a low relative amplitude indicates fewer distinctions between activity levels during the most and least active times of day, that is, dampened rhythm. This can be a result of reduced daytime physical activity and/or less restful nighttime sleep, both of which can lead to reduced affective well-being. 52
Similar to the findings about affect, a later onset of M10 was associated with adverse cognitive outcomes, indicated by worse cognitive complaints. Additionally, pseudo-F statistic, that is, robustness or regularity of rhythms, was positively associated with fluid cognition. These findings replicate prior work of Xiao et al 14 who found that later activity timing and less robust rhythmicity were associated with a higher risk for mild cognitive impairment and dementia. A plausible explanation by which RAR relates to cognition is its regulation of arousal. As a behavioral marker of circadian rhythm, a less robust/more erratic RAR may be associated with reduced brain arousal or vigilance through less robust time-of-day signaling, 53 thereby impairing cognitive performance during the day. Moreover, neuroimaging studies found that RAR characteristics, such as fragmentation and amplitude, are correlates of altered white matter integrity. 54 As white matter is responsible for cerebral connectivity, it is not surprising to observe a positive association between RAR disruption and cognition. However, the causality and the underlying mechanism of the relationship remains to be clarified as a study has found that white matter lesions caused by cerebral small vessel disease are related to disturbed RARs. 55 Considering that stroke is known to cause white matter injury, 56 it is possible RAR disruption and cognitive impairment are downstream consequences of stroke.
It is interesting to note that higher intra-daily variability, that is, 24-hour rhythm fragmentation, was associated with better crystallized cognition. High intra-daily variability means that there are more frequent shorter periods of rest and activity rather than one extended daytime active period and one extended nighttime rest period. Crystallized cognition is positively correlated with educational attainment 57 and hence it is more likely that the person will work more mentally demanding jobs that are relatively sedentary. Therefore, if the daytime activity level is as low as nighttime rest, it is more likely for the person to generate activity spikes from bouts of physical activity incurred during the daytime, such as working out in the gym or walking to attend meetings in a different location, get lunch in the restaurant, or to the parking garage. However, the underlying mechanism for the positive association between intra-daily variability and crystallized cognition is unclear and our hypothesis remains to be tested in future studies.
Implications for Clinical Practice
Our findings have several important implications. First, the significant associations of RAR characteristics with affective and cognitive outcomes among stroke survivors, and the fact that RAR is exogenously modifiable, provides initial support that RAR disruption can be targeted in stroke interventions. The significant RAR correlates identified in the present study may indicate that they are more influential than others to explain the outcomes, and hence potential targets for interventions. Specifically, the associations of timing and rhythmicity with affective and cognitive outcomes suggest that promoting earlier and a more stable rest-activity routine might help prevent or mitigate affective and cognitive impairments. For example, bright light therapy is a well-established circadian intervention that has demonstrated success in strengthening the circadian rhythm to improve rhythm stability, timing, and robustness. 58 It has been shown to reduce cognitive decline 58 and prevent affective disorders. 59 Establishing a structured daily routine and wake-sleep schedule can help to regulate the times for daytime activities and sleep. Standardized instruments, such as the Activity Card Sort, 60 can be used to identify and schedule personally meaningful daily activities that motivate the person to start their day earlier. Promoting sustained physical activity engagement during the daytime and reducing sleep disturbances at night are ways to strengthen RAR rhythmicity. National programs, such as the Home- and Community-Based Services (HSBS) and the Program of All-Inclusive Care for the Elderly (PACE), should be recognized by clinicians as resources to improve RAR by promoting daily activity participation in the home and community. In addition to informing targets for intervention studies, studies such as clinical trials and larger scale longitudinal studies with longer observation period can also be conducted to further explore the causal relationships between these RAR characteristics with affective and cognitive outcomes. Moreover, the fact that RAR can be easily monitored objectively and is highly reproducible makes it a potential safety endpoint in stroke management and a marker to help predict clinical outcomes in stroke survivors. Finally, as RAR intervention requires effort to address daytime activities as well as nighttime sleep behaviors, there is also a need for more sleep research to better understand sleep disturbances after stroke and ways to improve the quality and maintenance of sleep after stroke.
Study Limitations
The findings should be interpreted in light of several limitations. First, the generalizability of our findings is limited by the exclusion of people with more severe stroke and locomotor impairments (ie, wheeled device users). Future studies should test the replicability of the findings by including more severe stroke samples and using wrist-worn accelerometers so that wheeled device users can be included. Second, our objective assessment of cognition was conducted in the laboratory. Future studies should consider using mobile cognitive assessment 61 to assess real-world cognitive performance. Third, we did not treat RAR disruption. Future intervention efforts are needed to confirm causality. Moreover, it will be meaningful to clarify the dose-dependent relationship of the significant RAR characteristics with affective and cognitive outcomes. For instance, identifying how much earlier the onset of activity and rest will lead to clinically meaningful improvements in affective and cognitive outcomes. Similarly, cut-offs for defining impairment of different RAR characteristics should be investigated. Fourth, the small sample size precluded controlling for more potential confounding covariates in the analyses. Fifth, we did not consider the use of anti-depressants in this study. Some classes of anti-depressants, such as serotonin and norepinephrine reuptake inhibitors and norepinephrine reuptake inhibitor, have activating effects which can disturb sleep quality and lead to insomnia. 62 Given the high prevalence of poststroke depression, future stroke studies should consider antidepressant use when analyzing RAR data. Sixth, we did not collect information regarding the receipt of therapy, which could confound the study findings if participants were undergoing therapies for their affect and cognitive functioning. Seventh, we did not have a healthy control group for comparison because this was a secondary data analysis. Future studies should consider including a control group for identifying patterns of association unique to stroke survivors. This helps with tailoring and adapting interventions for people with stroke. Eighth, given the sample size of this study, we did not further investigate the role of major covariates (eg, age, sex, and race) in the associations examined in this study. Future research with larger sample size can be designed to examine the role of these factors with statistical procedures like sub-group analyses to provide new insights. Ninth, we did not analyze some other RAR measures, such as amplitude and acrophase, that might convey information of different perspectives.
Conclusions
We identified timing and rhythmicity-related RAR measures as correlates of cognitive and affective outcomes in stroke survivors. This finding suggests that RAR may be a viable focus for stroke rehabilitation, broadening the spectrum of available strategies for preventing and managing poststroke affective and cognitive impairments. For example, optimizing daily routines with personally meaningful activities and improving sleep hygiene could advance RAR by promoting engagement with activities in the morning and facilitating more efficient sleep at night. However, clinical trials are needed to determine whether improving these RAR measures will prevent or mitigate affective and cognitive impairments among stroke survivors.
Supplemental Material
sj-doc-1-nnr-10.1177_15459683241230027 – Supplemental material for Associations of Circadian Rest-Activity Rhythms With Affect and Cognition in Community-Dwelling Stroke Survivors: An Ambulatory Assessment Study
Supplemental material, sj-doc-1-nnr-10.1177_15459683241230027 for Associations of Circadian Rest-Activity Rhythms With Affect and Cognition in Community-Dwelling Stroke Survivors: An Ambulatory Assessment Study by Stephen C. L. Lau, Lisa Tabor Connor and Elizabeth R. Skidmore in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Stephen C. L. Lau: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Software; Validation; Visualization; Writing—original draft; Writing—review & editing. Lisa Connor: Conceptualization; Funding acquisition; Investigation; Methodology; Resources; Supervision; Writing—review & editing. Elizabeth Skidmore: Conceptualization; Funding acquisition; Investigation; Methodology; Resources; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
