Abstract
Background
To date, no study has longitudinally assessed the dose–response association between handgrip strength and incidence of Parkinson’s Disease (PD).
Objectives
to investigate the longitudinal association between handgrip strength and the development of PD within a representative European population of older adults.
Methods
Individuals aged 50 years and older from 27 European countries and Israel participated. We retrieved data from the Survey of Health, Ageing and Retirement in Europe waves 1, 2, 4, 5, 6, 7, and 8. Handgrip strength was measured using a hand dynamometer and participants reported whether they had a medical PD diagnosis. Time-varying exposure and covariates were modeled using both Cox regression and restricted cubic splines.
Results
A total of 71 702 participants (mean age 65.2 years) were followed over a median period of 5.0 years. Among them, 314 participants developed PD. In the fully adjusted model, we observed a higher risk (hazard ratio [HR]: 2.50; 95% CI:1.92-3.32) of PD for participants with lower handgrip strength (third 1) and a lower risk of PD for participants in the second third (HR: 1.41; 95% CI: 1.06-1.87). In dose–response analyses, men showed lower risk of PD from 27 kg (HR:0.94; 95% CI: 0.91-0.97) to 59 kg (HR:0.10; 95% CI: 0.04-0.22), whereas women showed significant reductions from 24 kg (HR:0.68; 95% CI: 0.46-0.99) to 38 kg (HR:0.44; 95% CI: 0.22-0.88).
Conclusions
Handgrip strength ought to be incorporated as one of the measures in the prognostic toolbox for the screening of older adults who are possibly at risk of developing PD.
Introduction
The etiology of Parkinson’s disease (PD) is multifactorial, including both genetic and environmental factors.1,2 However, to date, only few prognostic factors have been identified. Since PD is a progressive movement disorder, it can be hypothesized that muscle strength could be an important prognostic factor. In this regard, handgrip strength is known as a simple, objective, and effective way to measure muscular strength, 3 and could be a useful tool to assess muscular strength in patients at risk or with PD, as with other chronic conditions. 4 In addition, since loss of muscular strength is associated with reduced physical functioning,5-7 and therefore a lower quality of life, early detection of those with lower muscular strength could also help to implement effective training programs. 8
Previous studies have investigated muscular strength in patients with PD. For example, Corcos et al 9 showed that PD patients exhibit muscular weakness, even in the absence of medication. Similarly, a cross-sectional study by Roberts et al 10 revealed an association between increased PD severity lower handgrip strength. Moreover, a recent meta-analysis showed that muscular strength and power are impaired in PD patients. 11 Nonetheless, these studies were conducted with relatively small sample sizes and have primarily focused on individuals who already developed PD. As the disease is known to impair motor function, the observation of decreased muscle strength in individuals who have already manifested the condition is not unexpected. Gustafsson et al 12 showed lower handgrip strength in late adolescence in men diagnosed with PD 30 years later. However, it is worth noting that this study exclusively involved a cohort of young men from Sweden.
At the physiological level, motor symptoms in PD patients arise due to loss of dopaminergic neurons in the substantia nigra.2,13 Over the years, researchers and clinicians have developed several treatments to alleviate these motor symptoms, however there is currently no curable treatment.1,2,13 Since most patients have already lost a significant amount of nigrostriatal neurons at the time of diagnosis, 13 early identification of PD is crucial to offer treatment options that may slow down or potentially reverse the disease progression. Additionally, establishing effective prognostic measures to identify individuals at risk of developing PD is of importance. Grip strength could potentially serve as a useful marker indicating the risk of developing PD and inform focussed interventions.
Importantly, to date, no study has longitudinally assessed the association between handgrip strength and PD among older adults. Moreover, there is little knowledge regarding the dose–response association between handgrip strength and the onset of PD, a factor that could significantly contribute the design of personalized preventive measures. Therefore, the main objective of this study was to investigate the longitudinal and dose–response association between the levels of handgrip strength and the risk of developing PD within a substantial and representative European population of older adults.
Methods
Study Design and Population
The present study included data from waves 1, 2, 4, 5, 6, 7, and 8 from the Survey of Health, Ageing and Retirement in Europe (SHARE).14,15 Wave 3 was excluded from our analysis due to the absence of handgrip strength assessment. Representativeness of SHARE waves is achieved by using a multi-stage stratified sampling design in which countries are divided into different strata according to their geographical area. Municipalities or zip codes within these strata are primary sampling units. Data collection was conducted through home computer-assisted personal interviews from February 2004 to January 2021. SHARE data were collected using ex-ante harmonised interviews. New respondents are added in each wave to compensate for attrition bias from loss to follow-up. Our study exclusively included participants aged 50 years or older, with no prior diagnosis of PD at study entry.
Individuals with less than 2 waves of follow-ups or any missing values in any study variable were removed from the analyses. From an initial sample of 209 357 participants, exclusions were made for duplicates, participants aged younger than 50 years, those outside the study’s date range, those with less than 2 follow-ups, those with a Parkinson’s diagnosis at the study entry, and those with missing values. These exclusions resulted in an analytical sample of 71 702 participants, as illustrated in Supplemental Figure S1 in the flowchart. Complete-case analyses were conducted. More information on the study cohort profile is shown in Supplemental Material 1. This study received the approval of the Ethics Committee of Research in Humans of the University of Valencia (registered code 1510464) and was reported according to Strengthening the Reporting of Observational Studies in Epidemiology. All participants provided written informed consent.
Handgrip Strength (Exposure)
Qualified interviewers conducted 2 measurements of handgrip strength for each hand employing a handheld dynamometer (Smedley, S Dynamometer, TTM, Tokyo, 100 kg). Consistent with the SHARE protocol, participants received instructions to position their elbow at a 90° angle flexion, either in a standing or seated posture, maintaining a neutral wrist position, with the upper arm vertically aligned against the trunk. Interviewers provided standardized verbal encouragement to participants, instructing them to exert maximal effort in squeezing the dynamometer for a few seconds. Handgrip strength was considered as the maximum value of either hand.
PD (Outcome)
Participants were followed throughout the study period to determine whether they were diagnosed with PD. This was determined through the following question that participants responded in each SHARE wave14,15: “Has a doctor ever told you that you had/currently have any of the conditions on this card? With this we mean that a doctor has told you that you have this condition, and that you are either currently being treated for or bothered by this condition.” The referred card included the “Parkinson’s disease” option among other 18 options related to other chronic conditions. Possible answers comprised “Yes”, “No”, “Refuse,” or “Don’t know.” Participants selecting these 2 last options were removed from the analyses.
Covariates
Based on a literature review on the topic, age, sex, country of residence at the time of the interview, education, body mass index, smoking habit, alcohol consumption, physical inactivity, fruits and vegetables consumption, and number of chronic diseases were identified as potential confounders.1,10,16 More details on the aforementioned covariates have been provided elsewhere. 15
Statistical Analyses
All statistical analyses were conducted using Stata version 16.1 (StataCorp, Texas, USA). Using Cox regression, we conducted complete-case analysis to estimate the hazard ratios (HRs) for PD, using time-in-study measured in months as the timescale. We examined the proportional hazards assumption by testing interactions with log(time) and found no evidence of assumption violation. Furthermore, we conducted a Wald test to examine interactions between absolute categorized handgrip strength with both age and sex and observed no significant interaction, thus results are presented without either age or sex stratifications for the main analyses. Nevertheless, we observed a significant interaction between continuous handgrip strength and sex, thus we opted for conducting stratified dose–response analyses. Two time-variant models were tested: a model including age at the time of the interview and sex as confounders (Model A) and a fully adjusted model (Model B) that included covariates of Model A plus country, education, body mass index, smoking habit, alcohol consumption, physical inactivity, fruit and vegetable consumption, and number of chronic conditions as confounders. For a more comprehensive presentation of the results, we stratified handgrip strength into thirds based on sex-specific tertiles and visualized the outcomes through forest plots.
Additionally, we assessed the dose–response associations of handgrip strength, modeled as a continuous exposure, and the occurrence of PD using restricted cubic splines to allow for potential non-linearity; we trimmed observations less than 5% and greater than 95% of the distribution and pre-specified knots placed at the 5th, 50th, and 95th percentiles of the exposure distribution. Deviation from linearity was checked with a Wald test assessing the null hypothesis that the coefficient of the third spline was equal to zero. We assumed linearity for values below the 5th percentile and for values above the 95th percentile. Results are reported as HRs with 95% confidence intervals (CIs) and levels of significance were set at P < .05.
Results
In total, 71 702 participants were followed-up during a median of 5.0 years (Interquartile range 3.2-7.5; 383 443 person/year), in which 314 were diagnosed with PD. At study entry, average age was 65.2 years of whom 55.2% were women. Table 1 presents the characteristics of participants at study entry.
Characteristics of Participants at Study Entry.
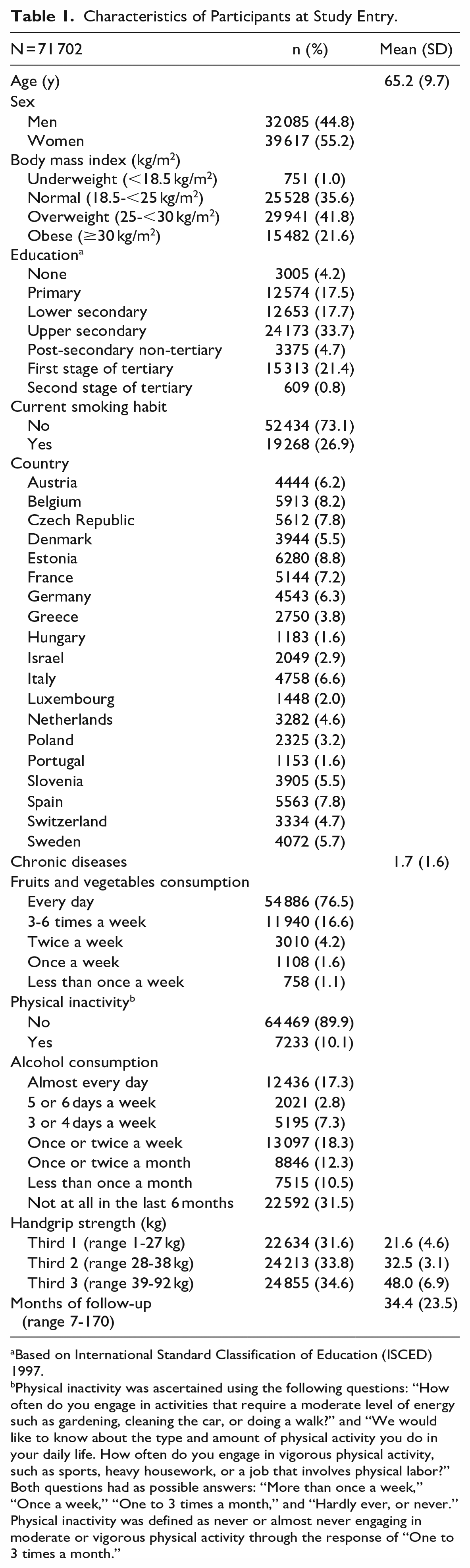
Based on International Standard Classification of Education (ISCED) 1997.
Physical inactivity was ascertained using the following questions: “How often do you engage in activities that require a moderate level of energy such as gardening, cleaning the car, or doing a walk?” and “We would like to know about the type and amount of physical activity you do in your daily life. How often do you engage in vigorous physical activity, such as sports, heavy housework, or a job that involves physical labor?” Both questions had as possible answers: “More than once a week,” “Once a week,” “One to 3 times a month,” and “Hardly ever, or never.” Physical inactivity was defined as never or almost never engaging in moderate or vigorous physical activity through the response of “One to 3 times a month.”
For model A, both the first third and the second third of participants showed a higher risk of developing PD, with HR of 2.81 (95% CI: 2.14-3.69) and 1.39 (95% CI: 1.04-1.85), respectively (Figure 1). Subsequently, in the fully adjusted Model B, as depicted in Figure 2, participants with lower handgrip strength exhibited a higher risk of developing PD (HR: 2.50; 95% CI: 1.92-3.32), whereas those in the second third displayed a significantly reduced risk (HR: 1.41; 95% CI: 1.06-1.87).
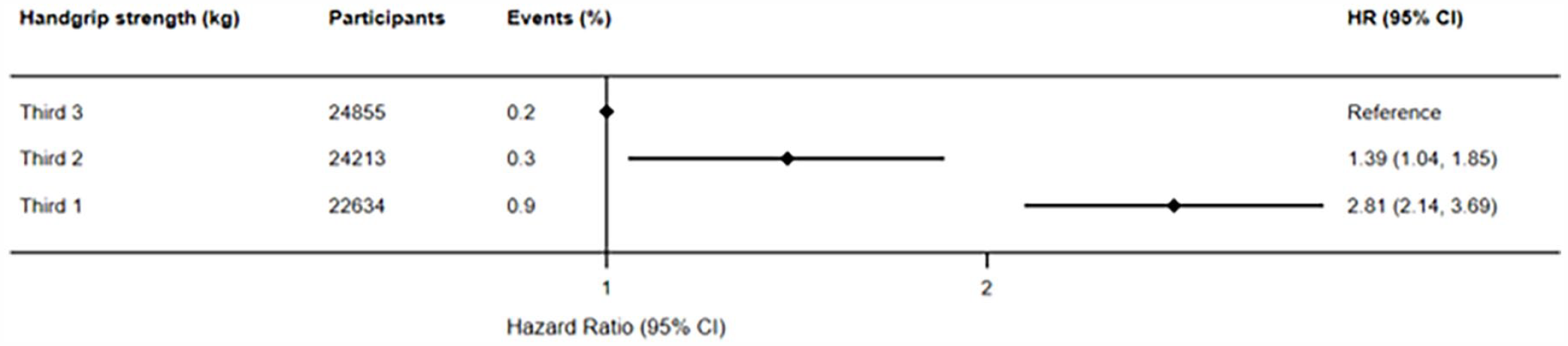
Prospective associations between handgrip strength (kg) and Parkinson’s disease (crude model).
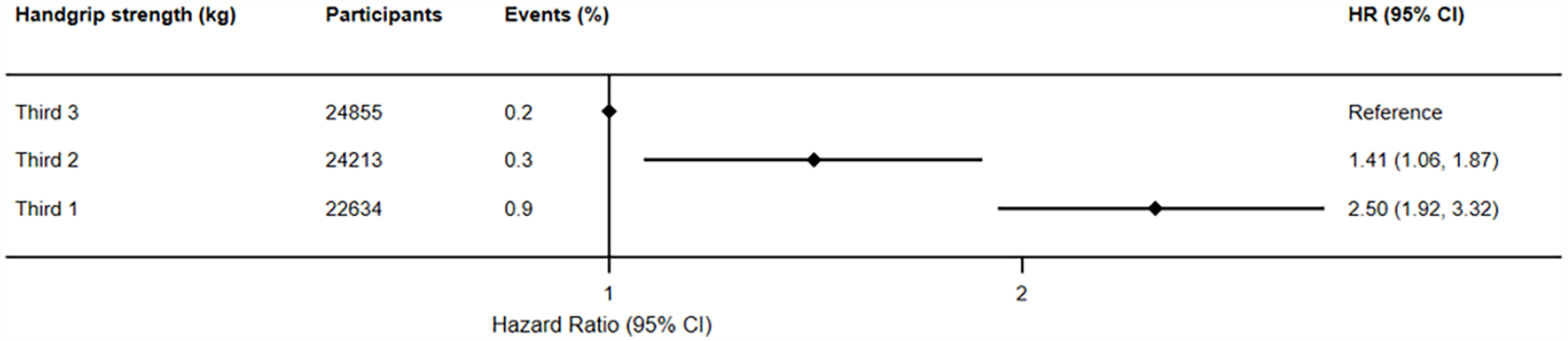
Prospective associations between handgrip strength (kg) and Parkinson’s disease (adjusted model).
Regarding dose–response analyses, both male and female participants demonstrated a close-to-linear inverse association between handgrip strength and developing PD. Specifically, men showed significant reductions for PD across a range of handgrip strengths, ranging from 27 kg (HR:0.94; 95% CI: 0.91-0.97) to 59 kg (HR:0.10; 95% CI: 0.04-0.22; Figure 3), while women similarly displayed significant risk reductions for PD within the range of handgrip strengths from 24 kg (HR: 0.68; 95% CI: 0.46-0.99) to 38 kg (HR: 0.44; 95% CI: 0.22-0.88; Figure 4).
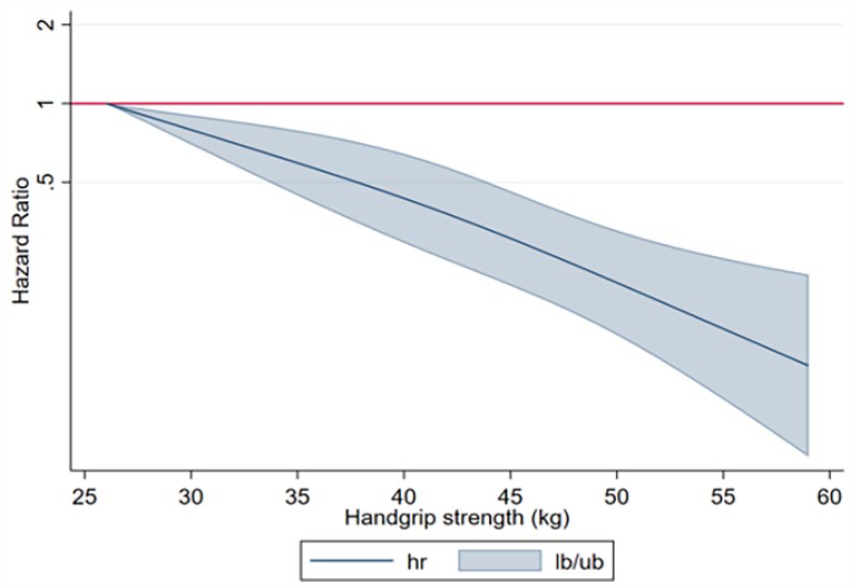
Dose–response association (adjusted hazard ratios and associated 95% confidence interval band) between handgrip strength (kg) and Parkinson’s disease in men aged 50 years or over. Adjusted for Model B (age, country, education, body mass index, smoking habit, alcohol consumption, physical inactivity, fruits and vegetables consumption, and number of chronic conditions.).
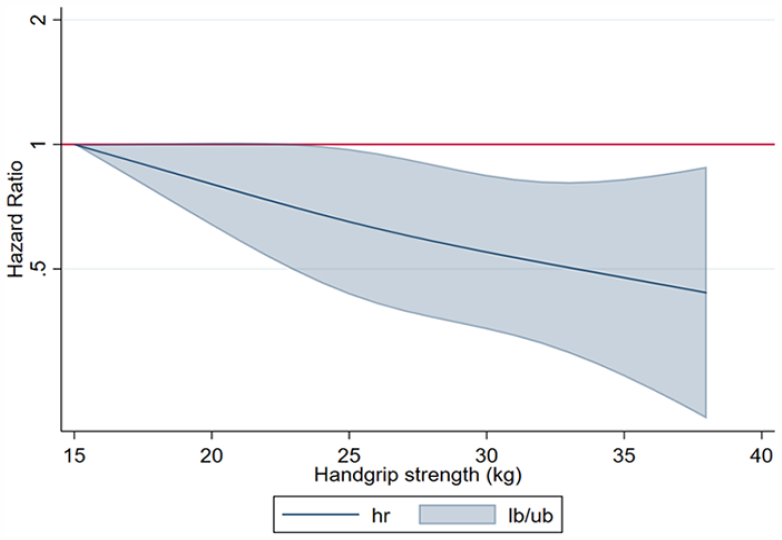
Dose–response association (adjusted hazard ratios and associated 95% confidence interval band) between handgrip strength (kg) and Parkinson’s disease in women aged 50 years or over. Adjusted for Model B (age, country, education, body mass index, smoking habit, alcohol consumption, physical inactivity, fruits and vegetables consumption, and number of chronic conditions.).
Discussion
This large-scale longitudinal study aimed to investigate the association between handgrip strength and the development of PD. Our findings indicate a higher risk of PD for participants with lower handgrip strength and found a close-to-linear inverse association between handgrip strength and PD in the dose response analysis.
Specifically, we identified an association between having lower handgrip strength and developing PD later in life. This association was observed in a close-to linear inverse association in both men and women within specific handgrip strength ranges. Our observation aligns with the research conducted by Gustafsson et al, 12 which similarly reported an association between lower handgrip strength and the onset of PD in a cohort of younger Swedish males (ie, aged 18 years and over). It is noteworthy, however, that the aforementioned study reported a lower risk of PD than observed in our study, which may be attributed to different features of the study population such as age, sex, and different lifestyle behaviors. Moreover, it is essential to consider that muscle loss and the concomitant onset of motor symptoms typically commence in later stages of life, 2 which could significantly contribute to the differences observed in the estimations. Notably, no prior longitudinal studies have been conducted to assess the association of handgrip strength and the risk of developing PD. However, our findings are in agreement with a previous cross-sectional study reporting that increased severity of PD is associated with lower handgrip strength. 10 In addition, a recent study found that muscular strength and power are impaired in PD patients. 11
A novel and relevant finding in our study is the dose–response association, showing that men with handgrip strengths ranging from 27 to 59 kg, and women with handgrip strength ranging from 24 to 38 kg, have a lower risk of developing PD later in life. These cut points align with other cut points found for handgrip strength and reduction of all-cause, cancer and cardiovascular mortality in older adults, 17 as well as for handgrip strength and the risk of depression. 4 This alignment suggests that the underlying protective mechanism of handgrip strength may extend to various diseases, implying broader implications. It can also be speculated that a low level of muscle strength might serve as an early indicator of disease progression, potentially preceding clinical diagnosis by a medical practitioner.
To better understand the potential underlying mechanism between handgrip strength and the pathophysiology of PD, it is essential to explore relevant mechanistic aspects. For instance, a study by Pineda-Pardo et al 18 demonstrated that in early PD, striatal dopaminergic denervation follows a somatotopic pattern, beginning with the upper limb representation in the putamen. The putamen is known for its significant role in motor control and coordination. 19 The putamen projects cortically to specific areas in the brain, forming a complex network involved in regulating motor functions. Changes in this somatotopic pattern observed in the putamen have been shown to correlate with the evolution of motor features in PD patients, reflecting the progressive nature of the disease. 18 It can therefore be speculated that higher levels of muscle strength, particularly in the upper limbs, might exert a protective effect on the nigrostriatal pathway, potentially mitigating the neurodegenerative processes associated with PD. Enhanced muscle strength may provide increased support and compensation for the early loss of dopaminergic neurons, contributing to the preservation of motor functions.
Our finding also displays an absence of effect beyond specific thresholds of handgrip strength, indicating the absence of further reduction in HR for developing PD for individuals with handgrip strength exceeding 59 kg for men and 38 kg for women. The range of cut-off points in between which a possible preventive effect can be observed is lower for women compared with men. One possible explanation may be related to the influence of oestrogens, which have been associated with higher physiological striatal dopamine levels and therefore a higher age at onset for symptoms of PD and milder motor deterioration in women. 20
On the other hand, a possible mechanism explaining this dose–response association could be that during the prodromal or premotor symptoms phase of the disease, loss of nigrostriatal dopaminergic neurons lead to an increased inhibition of the thalamus and therefore a decreased excitation of the motor cortex, and thus causing muscle loss.10,21 This process could already be present in such an amount that loss of muscle strength is present before the start of motor-symptoms, like tremor or rigidity.13,21 Besides, it has been observed that asymmetrical muscle weakness is present in patients with PD and that it is not specifically related to tremor or rigidity, 22 suggesting that loss of muscle strength could be an early detectable sign of PD.
Strengths and Limitations
The main strength of the present study is the use of a large and representative cohort of men and women with objective repeated measures of objectively assessed handgrip strength and relevant covariates. Furthermore, we adjusted for potential time-varying confounding factors, reducing the probability of confounding bias. Nonetheless, this study also includes some limitations. First, due to the high number of participants with missing values, or with less than 2 waves of follow-ups, selection bias may potentially exist. Importantly, because the outcome variable is self-reported, a certain degree of misclassification bias and recall bias could have affected the data. Another limitation of our study is that the assessors were not masked. In addition, although handgrip peak force is usually achieved before 2 to 3 seconds,23,24 it is plausible that handgrip measurements were too short for those patients with bradykinesia. Finally, a time-varying confounding bias might influence our estimations.
Conclusion
Our results suggest that handgrip strength may serve as an effective prognostic measure to complement screening of older adults at risk of developing PD. It is worth considering that handgrip strength could potentially represent a preventive factor for PD, particularly within a specific sex-specific range. Additionally, early identification of individuals at risk of PD holds significant importance for implementing appropriate treatments aimed at potentially slowing down or reversing the progression of the disease. This could involve tailored strength training programs. Further research is needed to explore the potential mechanisms of tailored resistance programs and slowing down disease progression of PD.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231207359 – Supplemental material for Is Handgrip Strength Associated With Parkinson’s Disease? Longitudinal Study of 71 702 Older Adults
Supplemental material, sj-docx-1-nnr-10.1177_15459683231207359 for Is Handgrip Strength Associated With Parkinson’s Disease? Longitudinal Study of 71 702 Older Adults by Rochelle Mey, Joaquín Calatayud, José Casaña, Rodrigo Núñez-Cortés, Luis Suso-Martí, Lars Louis Andersen, José Francisco López-Gil and Rubén López-Bueno in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
This paper uses data from SHARE Waves 1, 2, 4, 5, 6, and 7 (DOIs: 10.6103/SHARE.w1.710, 10.6103/SHARE.w2.710, 10.6103/SHARE.w4.710, 10.6103/SHARE.w5.710, 10.6103/SHARE.w6.710, 10.6103/SHARE.w7.711, 10.6103/SHARE.w8.0, see Börsch-Supan (2013) for methodological details. The SHARE data collection has been funded by the European Commission through FP5 (QLK6-CT-2001-00360), FP6 (SHARE-I3: RII-CT-2006-062193, COMPARE: CIT5-CT-2005-028857, SHARELIFE: CIT4-CT-2006-028812), FP7 (SHARE-PREP: GA No. 211909, SHARE-LEAP: GA No. 227822, SHARE M4: GA No. 261982, DASISH: GA No. 283646), and Horizon 2020 (SHARE-DEV3: GA No. 676536, SHARE-COHESION: GA No. 870628, SERISS: GA No. 654221, SSHOC: GA No. 823782), and by DG Employment, Social Affairs & Inclusion. Additional funding from the German Ministry of Education and Research, the Max Planck Society for the Advancement of Science, the U.S. National Institute on Aging (U01_AG09740-13S2, P01_AG005842, P01_AG08291, P30_AG12815, R21_AG025169, Y1-AG-4553-01, IAG_BSR06-11, OGHA_04-064, and HHSN271201300071C) and from various national funding sources is gratefully acknowledged (see ![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RLB is supported by the European Union—Next Generation EU.
Data Availability Statement
This paper uses publicly available data from SHARE Waves 1, 2, 4, 5, 6, and 7 (DOIs: 10.6103/SHARE.w1.710, 10.6103/SHARE.w2.710, 10.6103/SHARE.w4.710, 10.6103/SHARE.w5.710, 10.6103/SHARE.w6.710, 10.6103/SHARE.w7.711, and 10.6103/SHARE.w8.0, see Börsch-Supan (2013) for methodological details.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
