Abstract
Background
The Gait Deviation Index for Spinal Cord Injury (SCI-GDI) was recently proposed as a dimensionless multivariate kinematic measure based on 21 gait features derived from 3-dimensional kinematic data which quantifies gait impairment of adult population with incomplete spinal cord injury (iSCI) relative to the normative gait of a healthy group. Nevertheless, no validity studies of the SCI-GDI have been published to date.
Objective
To assess the construct validity of the SCI-GDI in adult population following iSCI. Methods. SCI-GDI data were obtained from a sample of 50 healthy volunteers and 35 adults with iSCI. iSCI group was also assessed with the following measures: 10-Meter Walk Test (10MWT) at both self-selected (SS) and maximum speeds, Timed Up and Go Test (TUGT), SS and maximum levels of the Walking Index for Spinal Cord Injury (WISCI) II, mobility items of the Spinal Cord Independence Measure III (SCIM IIIIOMob), Lower Extremity Motor Score (LEMS), and Modified Ashworth Scale (MAS). Spearman’s correlation coefficient was used to investigate the relationship with the SCI-GDI.
Results
SCI-GDI shows strong correlation with the 10MWT (r ≥ −.716) and good correlation with LEMS (r = .638), TUGT (r = −.582), SS WISCI II levels (r = .521), and SCIM IIIIOMob (r = .501). No significant correlations were found with maximum WISCI II levels and MAS (P > .006).
Conclusions
Construct validity of the SCI-GDI was demonstrated with the 10MWT, TUGT, LEMS, SCIM IIIIOMob, and SS WISCI II levels for independently walking adults with iSCI. Future work will include assessing the psychometric characteristics with a more heterogeneous sample, also considering the pediatric population.
Keywords
Introduction
Gait impairment depends on the extent and severity of a spinal cord injury (SCI), 1 with a direct consequence in the loss of muscle strength, the presence of spasticity if the injury is the result from an upper motor neuron lesion, 2 and sensory and balance impairments.3,4 Regaining independent ambulation has been demonstrated to be a functional outcome indicator of the quality of life 5 and a high priority for recovery among individuals following incomplete SCI (iSCI). 6 Periodic gait measurements are used to evaluate the response to different therapeutical approaches to assess walking changes over time, and to discriminate between normal and altered gait. 7
The International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) published by the American Spinal Injury Association (ASIA) defines concepts as the neurological level of injury (NLI), its severity by the ASIA Impairment Scale (AIS), and the Lower Extremity Motor Score (LEMS), which quantifies the strength of lower extremity (LE) muscles. 1 Nevertheless, AIS or LEMS were never intended to be used as measures of preserved functions related to activities or independence. 8 Therefore, it is highly recommended to combine the ISNCSCI assessments with walking ability outcome measures to assess gait and balance in SCI.
Observational gait assessment methods allow to evaluate walking ability and the degree of deviation from a normal gait pattern. 9 However, the subjectivity of these measurements has been criticized and the need for other techniques which might objectively quantify changes in gait has been pointed out. 10 Three-dimensional gait analysis (3DGA) is recommended to be used as the gold standard for determining gait impairments and to guide rehabilitation interventions in order to improve walking function of people with traumatic and non-traumatic iSCI. 11 Nevertheless, the large amount of data for every joint and plane does not provide a complete picture of the altered gait pattern. 12 Since most of these kinematic data are interrelated, dimension reduction algorithms allow to combine and reduce the amount of data while still capturing the essence 13 of the kinematics, allowing for a more straightforward quantification of how much individual gait deviates from a normal pattern.
However, it is necessary to explore the psychometric properties of a measure to consider its suitability for clinical use.14,15 The COnsensus-based Standards for the selection of health Measurement INstruments 16 establishes a taxonomy of properties within 3 quality domains that should be evaluated for any measurement instrument: validity, reliability, and responsiveness. Validity is defined as the degree to which an instrument measures the construct it intends to measure, and it contains the following properties: content, construct, and criterion validity. 17 In a rehabilitation context, a measuring instrument is considered useful when it is consistent with respect to its relationship to the scores of other measurement instruments. 18 Since there is no ‘gold standard’ among the walking ability outcome measures in SCI, the construct validity of a tool must be assessed.
The SCI-GDI (Gait Deviation Index for SCI) is a dimensionless metric derived from a dimensionality reduction algorithm applied to multivariate kinematic measures represented as a single score for the gait deviation of an individual with iSCI from a normative reference group. SCI-GDI is based on 21 gait features which reconstruct gait data with high fidelity and account for most of the variance of the iSCI dataset derived from 9 kinematic curves obtained by 3DGA from a sample of adult population with iSCI. 19 It has shown to be more sensitive to gait impairment than the original GDI, based on 15 gait features from a sample of children with cerebral palsy. 20 GDI turned out not to be an appropriate metric to represent the deviation of gait pattern in adult population with iSCI from a normal gait profile when it is compared with the levels of walking impairment described by the Walking Index for Spinal Cord Injury (WISCI) II. 21 SCI-GDI has better discriminative properties of more WISCI II levels than the original GDI when applied to adults with iSCI. 19 These results would point to the SCI-GDI as a metric capable of capturing the kinematic variability of gait in the adult population with iSCI beyond the external aids needed for walking. It could potentially be used in clinical settings to detect gait kinematic changes as a primary outcome measure following rehabilitation interventions aimed at improving functional gait after iSCI. Nevertheless, construct validity of the SCI-GDI has not yet been studied and its correlation with other commonly used walking ability outcome measures in the adult iSCI population is unknown.
A previous study published by Sinovas-Alonso et al 22 concluded that spatiotemporal walking and balance measures, such as the 10-Meter Walk Test (10MWT) and the Timed Up and Go Test (TUGT), and categorical measures such as the WISCI II made the most representative set of measures to cover the whole functional spectrum of walking ability in SCI. However, WISCI II levels are described for a fixed walking distance of 10 m, which does not reflect activities of daily living (ADLs). The mobility items of the Spinal Cord Independence Measure III (SCIM IIIIOMob) 23 fill this gap by assessing the assistance levels of individuals with SCI at different walking distances and in other more realistic contexts such as climbing and descending stairs. Furthermore, since the population with SCI may also present spasticity, which manifests as intermittent or sustained involuntary activation of muscles 24 disturbing gait control, 2 its measurement should also be considered, as assessed internationally with the Modified Ashworth Scale (MAS). 25
Therefore, studies analyzing the construct validity of the SCI-GDI with other valid and commonly used walking ability and clinical measures are needed. The aim of this study was to assess the construct validity of the SCI-GDI with spatiotemporal walking and balance measures, categorical, and clinical measures in the adult population with iSCI. We hypothesize that SCI-GDI values would be correlated positively with clinical and categorical walking ability outcome measures such as LEMS, MAS, WISCI II, and SCIM IIIIOMob, but negatively with spatiotemporal walking and balance measures like 10MWT and TUGT.
Methods
Study Design
An observational cross-sectional study composed of 2 samples, individuals with SCI and healthy volunteers (HV), was conducted at the Biomechanics and Technical Aids Unit of the National Hospital for Paraplegics of Toledo (Spain). STrengthening the Reporting of OBservational studies in Epidemiology guidelines were followed to standardize the reporting of this work (Figure 1). According to the Helsinki Declaration as revised in 2013, all participants were informed of the purpose and procedures of the study, as well as their right to withdraw from the study. Individuals gave written informed consent to participate in this study and to allow publication of images. The study protocol was reviewed and approved by the Local Ethics Committee of University Hospital Complex of Toledo, Spain (Ref. No. 795; 27/10/2021).
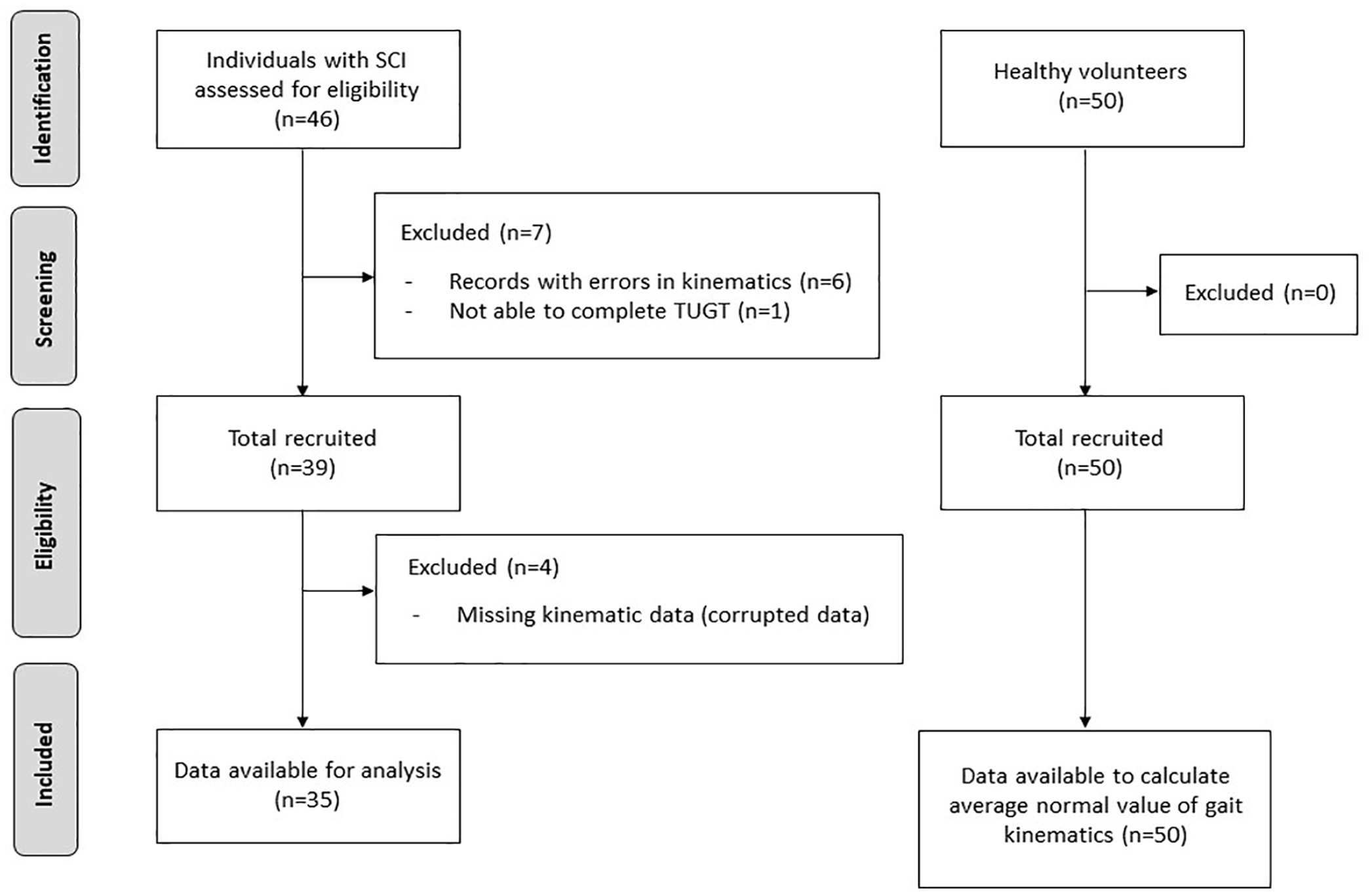
STROBE flow chart.
Participants
A retrospective research of the hospital database and a prospective recruitment of participants in the study were conducted by a clinician. Data of individuals with SCI were gathered from May 2021 to July 2022. Patients included met the following inclusion criteria: (i) individuals aged 16 years or over; (ii) having suffered a traumatic or non-traumatic SCI, regardless of time since injury and NLI; (iii) iSCI classified as C, D, or E by the AIS; (iv) with the ability of walking 10 m independently with any type of external assistance needed (orthoses, crutches, or canes); and (vi) capacity to be informed and give written consent to participate in the study. Exclusion criteria included: (i) suffering from orthopedic, rheumatic, or other neurological conditions that affected gait but were unrelated to SCI; (ii) following early stages of gait recovery requiring support on parallel bars or external physical assistance to walk 10 m safely; and (iii) psychiatric or cognitive conditions that may interfere with the completion of the study protocol.
Additionally, data of HV previously recruited at the hospital in April 2021 were gathered. Based on their medical history, they were excluded from the study if they had musculoskeletal or neurological conditions that impaired gait.
The age cutoff for individuals in both groups was set at 16 years old in accordance with the adult demographic profile that was taken into account when developing the SCI-GDI in the initial study. 19
Experimental Protocol
Several walking ability outcome measures widely used in SCI were selected to analyze the construct validity of the SCI-GDI (primary outcome measures): WISCI II, SCIM IIIIOMob, 10MWT, and TUGT. Secondary outcome measures included LEMS and MAS. One clinician administrated all assessments to the sample of individuals with iSCI on the same day and in the same order. Firstly, demographic characteristics (age and sex) and clinical data (time since injury, etiology, NLI, and AIS) were gathered for each individual. Then, subjects were assessed with categorical walking outcome measures (WISCI II and SCIM IIIIOMob), and TUGT and 10MWT were administrated. Next, 3D kinematic gait data were obtained and, finally, iSCI individuals were examined using clinical measures, that is, LEMS and MAS.
Demographic information and 3D kinematic gait data were also collected on HV by the same clinician.
– Walking ability and clinical outcome measures
The WISCI II assesses physical limitation for walking secondary to impairment after SCI. It is composed of 21 levels which are ordered from the most to least severe impairment, integrating the use of ambulatory assistive devices (AADs), orthoses, and the physical assistance needed to complete a 10-m walking distance. 26 WISCI levels differ from self-selected (SS) WISCI, defined as the condition in which the individual walks in the community or the household, and maximum WISCI, which is related to the highest level at which a person can safely walk 10 m. 27 Both, SS and maximum WISCI II levels, were gathered for each individual with iSCI. WISCI II is a valid,28,29 reliable,30,31 and responsive outcome measure to assess walking ability in people with SCI. 32
The SCIM III is a global disability scale developed for individuals with SCI to measure independence to perform ADLs. 23 Mobility items (SCIM IIIIOMob) are classified in 3 groups to assess walking ability: mobility indoors, mobility for moderate distances (10-100 m), and mobility outdoors (more than 100 m). All of them are scored out of 8: from the requirement of total assistance (scored 0) to the independence of AADs, orthoses, and personal supervision (scored 8). Stair management was also considered in the study, scored from 0 (individual unable to ascend or descend stairs) to 3 (individual able to ascend and descend at least 3 steps without any support or supervision). Therefore, a total score of 27 is assigned to the least impaired individuals with SCI. Mobility items of a previous version, SCIM IIIOMob, have shown to be a valid and responsive outcome measure to assess ambulatory function in population with SCI. 33
The TUGT measures the time needed to stand up from a chair, walk 3 m, turn around a cone, and sit back on the chair, all at a maximum and safe speed, with or without AADs. 34 The TUGT is strongly correlated with balance and the risk of falls, 35 and it has shown to be a valid and reliable outcome measure in people with SCI.36,37
The 10MWT reflects walking speed measuring the time invested in walking 10 m with physical assistance, orthoses, or any AADs required. The 10MWT was conducted with a dynamic start to allow 2-m acceleration before a timed 10-m distance and 2-m deceleration once reaching the end of 10-m pathway.5,38 Two conditions of the 10MWT were performed, firstly at a preferred walking speed (SS 10 MWT) and then at maximum speed (maximum 10 MWT). 34 The 10 MWT is a reliable,36–38 valid,36,37 and responsive 32 outcome measure to assess walking ability in individuals with SCI.
The average of 3 trials of each condition for the 10MWT and the TUGT was taken into account to enable iSCI individuals to become familiar with the tests.34,37 During the tasks, they were allowed to use their own AADs and orthoses if necessary.
The LEMS quantifies LE muscle strength on a rating scale from 0 to 50, with a maximum of 5 for each of 5 key muscles in each LE (hip flexors, knee extensors, ankle dorsiflexors, long toe extensors, and ankle plantar flexors). 1
The MAS 39 is a numerical scale that grades spasticity from 0 to 4, with 0 being no resistance and 4 being a limb rigid in flexion or extension, where the category 1+ has been added to the original scale 40 to increase its sensitivity. MAS has shown adequate reliability to assess LE spasticity in patients with SCI. 41 Testing was performed bilaterally for the rectus femoris—considering it is a biarticular muscle: hip flexor and knee extensor-, knee flexors, hip adductors, and ankle plantar flexors. These muscle groups were selected based on the most frequent location of LE spasticity in the population with SCI.
– Tridimensional gait analysis
Both groups, HV and individuals with iSCI, underwent 3DGA, whose kinematic data were obtained with the Codamotion photogrammetry system (Charnwood Dynamics, Ltd, UK). Anthropometric measures were taken and 22 active markers were placed on each LE. Markers were positioned following the protocol described in previous studies19,21 (Figure 2). To collect markers trajectories, 3 scanners were placed on the sides of a 10-m walkway with 2 Kistler force platforms embedded. Individuals were instructed to walk naturally at their SS speed with the minimal external assistance required (maximum WISCI II). Data were recorded at a sample frequency of 200 Hz. About 3 to 5 complete valid gait cycles were collected and time-normalized for each individual, in which each foot steps on a force platform. Each stride was considered as an independent register. A total of 350 and 446 strides were collected for the group with iSCI and the HV group, respectively. Records were processed using the software for data analysis ODIN v.2.02 (Codamotion Ltd., UK) to calculate 9 kinematic curves of a single stride of the right and left LE: (i) pelvic orientation and hip angles in sagittal, frontal and transversal planes, (ii) knee flexion and extension, (iii) ankle dorsiflexion and plantar flexion, and (iv) foot progression angle.
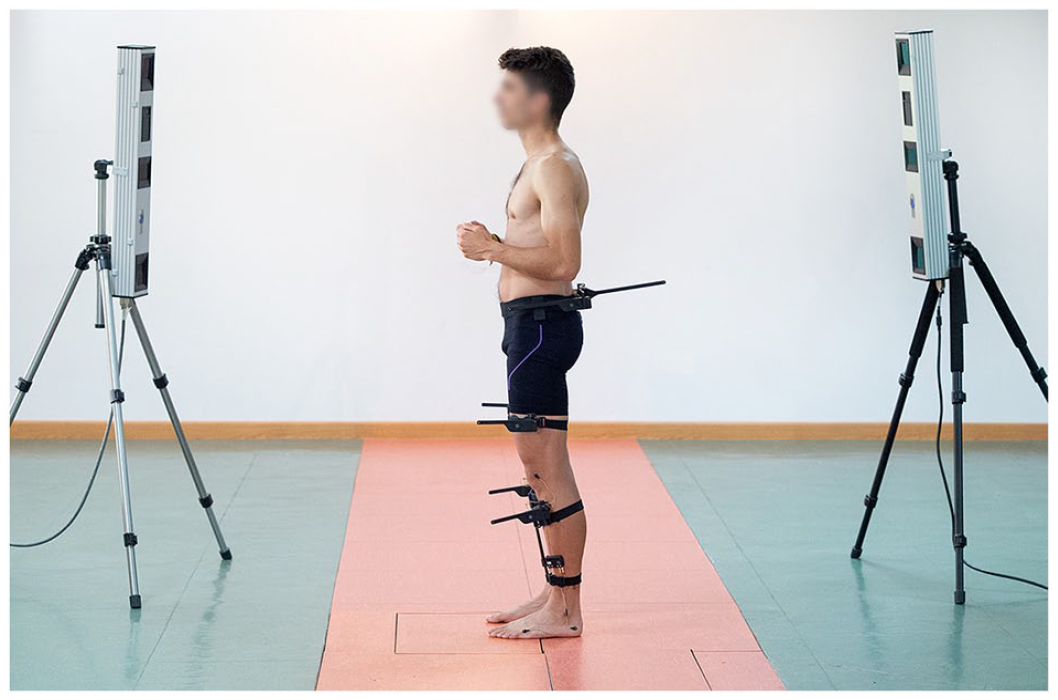
Placement of markers on an individual from HV group: sacrum (2 lateral markers), anterior superior iliac spines, posterior superior iliac spines, lateral surface of the thigh (anterior and posterior femur markers), lateral femoral condyles, lateral surface of the leg (anterior and posterior tibia markers), lateral malleolus, calcaneus (posterior lateral heel), and fifth metatarsal head.
Statistical Analysis
The SCI-GDI was calculated using the HV and iSCI dataset collected in the study following the procedure shown in Herrera-Valenzuela et al. 19
Statistical analysis was performed using the commercial IMB SPSS v22 software package (IMB Corporation, Armonk, NY, USA). Descriptive statistics was applied to explain demographic and clinical data, expressed as mean and standard deviation. Shapiro–Wilk test was used to check normality of walking ability and clinical measures of individuals with iSCI. Except for SCI-GDI values, variables analyzed were not normally distributed. Therefore, Spearman’s correlation coefficient was used to investigate the relationship between the SCI-GDI and WISCI II (SS and maximum levels), SCIM IIIIOMob, 10MWT (SS and maximum speeds), TUGT, LEMS, and MAS. MAS was previously categorized and transformed into a quantitative variable, assigning it a score range from 0 to 5, and calculated for both LE. Magnitude of correlations was defined as poor if correlation coefficients <.30, moderate if between .3 and.5, good if between .50 and .70, and strong if values >.70. 42 Simple linear regression analysis was applied to give a general idea of the extent to which variability in SCI-GDI values is predictable from outcome measures administered to individuals with iSCI. A Bonferroni correction was performed to adjust for multiple testing. With 8 comparisons, a P value of α = .05/8 (.006) was considered statistically significant.
Results
Data from 35 adults with iSCI and 50 HV who matched the inclusion criteria were collected (Table 1). Clinical characteristics of individuals with iSCI are shown in Table 2.
Demographics of Individuals Recruited for the Study.

Abbreviations: HV, healthy volunteers; iSCI, incomplete spinal cord injury; M, male; F, female; SD, standard deviation; BMI, body mass index.
Parameters reported as mean (SD).
Clinical Characteristics of Individuals With iSCI.
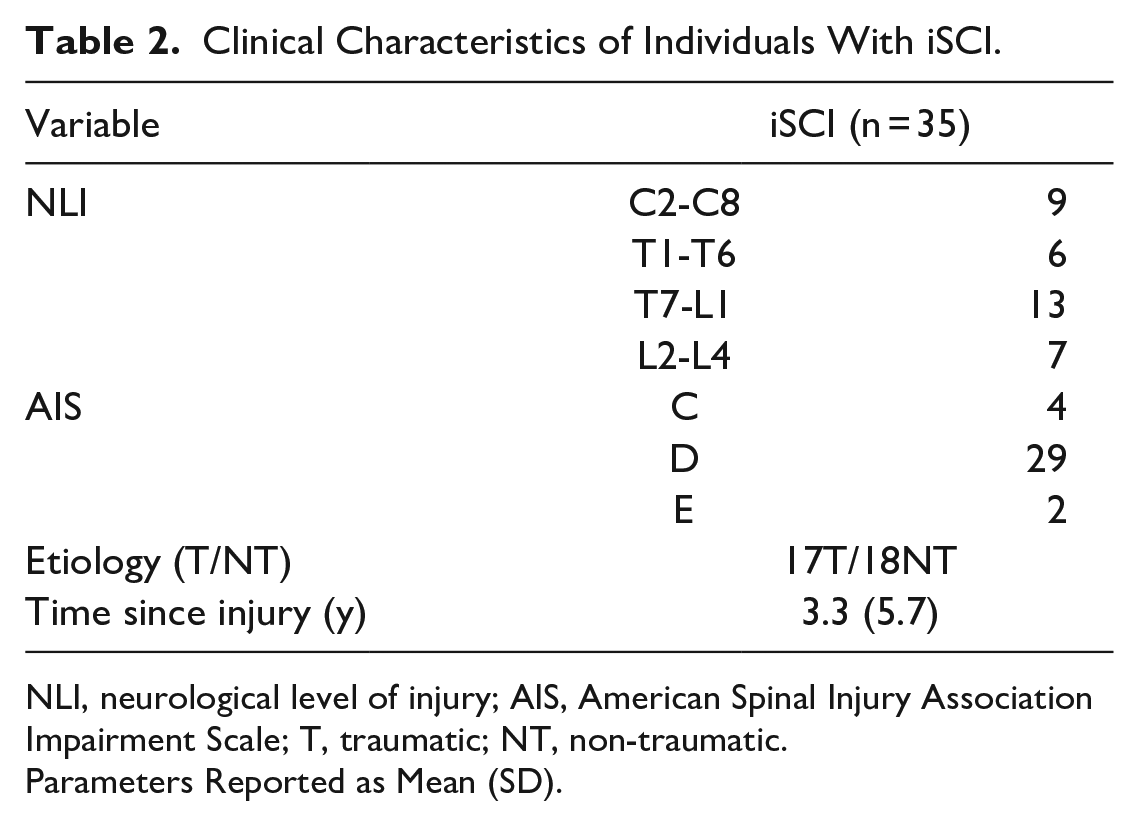
NLI, neurological level of injury; AIS, American Spinal Injury Association Impairment Scale; T, traumatic; NT, non-traumatic.
Parameters Reported as Mean (SD).
Mean values and standard deviations of the outcome measures administered to individuals with iSCI, Spearman’s correlations between them and the SCI-GDI, and variance are presented in Table 3. Figures 3A to H illustrate the correlation between SCI-GDI and the outcome measures.
Spearman’s Correlations Between the Outcome Measures Administered and the SCI-GDI in Individuals With iSCI.
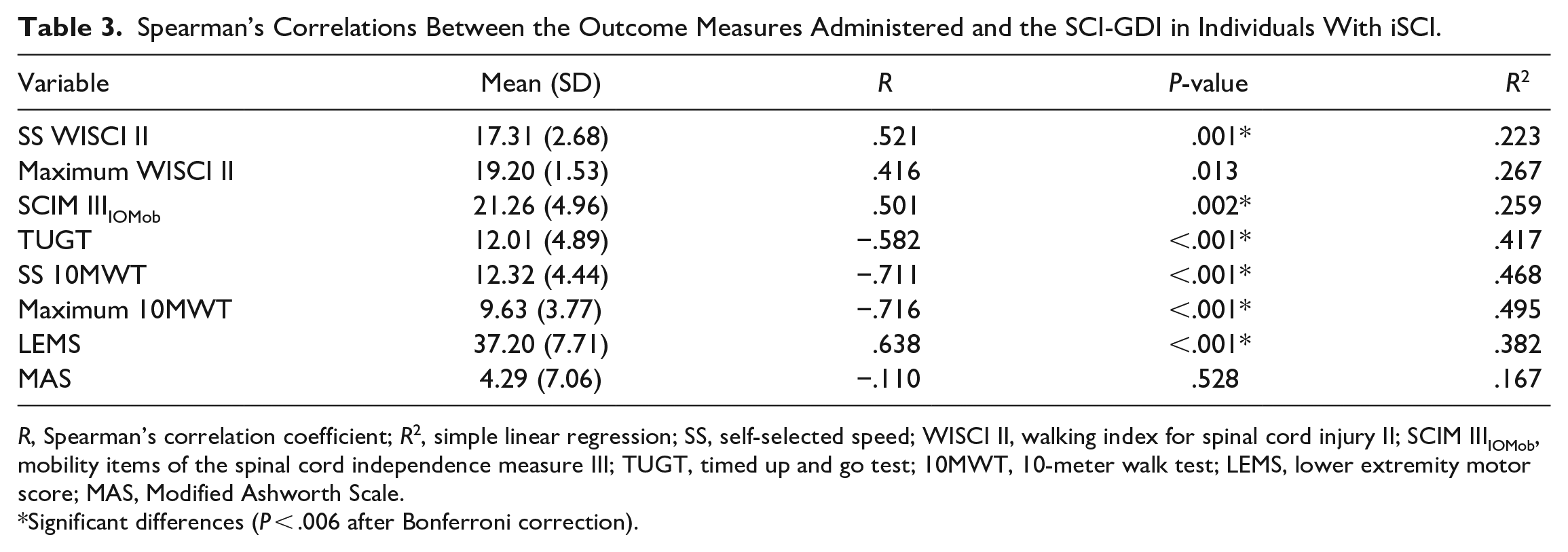
R, Spearman’s correlation coefficient; R2, simple linear regression; SS, self-selected speed; WISCI II, walking index for spinal cord injury II; SCIM IIIIOMob, mobility items of the spinal cord independence measure III; TUGT, timed up and go test; 10MWT, 10-meter walk test; LEMS, lower extremity motor score; MAS, Modified Ashworth Scale.
Significant differences (P < .006 after Bonferroni correction).
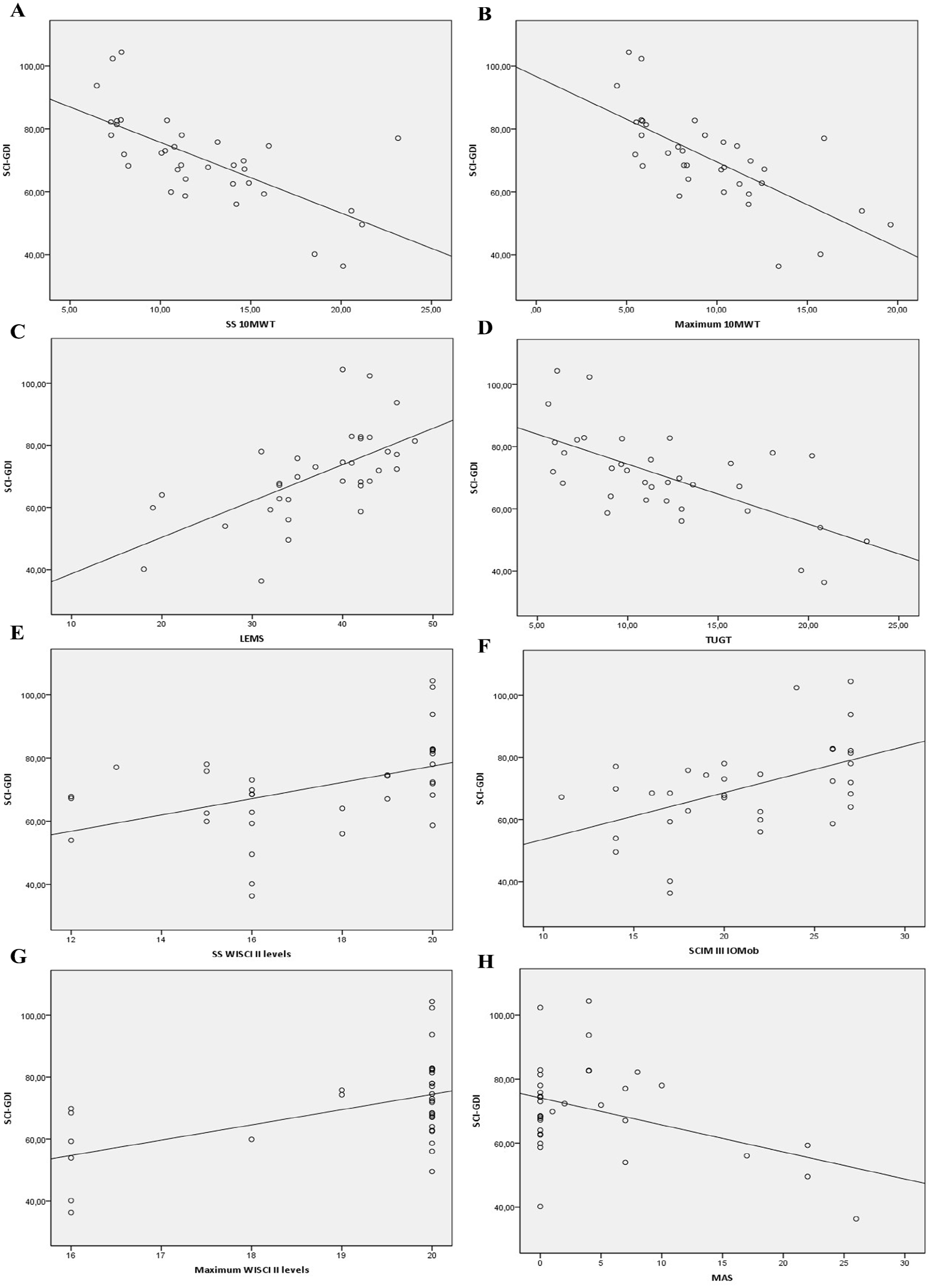
Correlation between SCI-GDI data and (A) SS 10MWT, self-selected (SS) speed for 10-meter walk test. (B) Maximum 10MWT, maximum speed for 10MWT. (C) LEMS, lower extremity motor score. (D) TUGT, timed up and go test. (E) SS WISCI II levels, SS walking index for spinal cord injury II levels. (F) SCIM IIIIOMob, mobility items of the spinal cord independence measure III. (G) Maximum WISCI II levels. (H) MAS, Modified Ashworth Scale.
The SCI-GDI showed strong correlation with the SS 10MWT and maximum 10MWT (r = −.711 and −.716, Figures 3A and B, respectively), and good correlation with the LEMS (r = .638, Figure 3C), TUGT (r = −.582, Figure 3D), SS WISCI II levels (r = .521, Figure 3E), and SCIM IIIIOMob (r = .501, Figure 3F). No significant correlations were found with the maximum WISCI II levels (P = .013, Figure 3G) and MAS (P = .528, Figure 3H).
The predictive capacity of the SCI-GDI to explain the variability of the data collected from the walking ability and clinical outcome measures administered to individuals with iSCI was higher for the maximum 10MWT and SS 10MWT (49.5% and 46.8%, respectively), followed by the TUGT (41.7%), and the LEMS (38.2%). From 26.7% to 22.3% of the variability of the SCI-GDI was explained by maximum WISCI II levels, SCIM IIIIOMob, and SS WISCI II levels, in descending order. The variability of the SCI-GDI was only explained by 16.7 % for the data of MAS.
Discussion
The aim of this study was to explore the construct validity of the SCI-GDI in adult population following iSCI. The findings of the study indicate that the SCI-GDI presents: (i) strong correlation with the SS 10MWT and maximum 10MWT; (ii) good correlation with the LEMS, TUGT, SS WISCI II levels, and SCIM IIIIOMob; and (iii) no significant correlation with the maximum WISCI II levels and MAS. To our best knowledge, construct validity studies of the SCI-GDI in individuals with iSCI have not been published to date.
Results of the construct validity of the SCI-GDI are congruent with the nature of the outcome measures to which it correlates. The 10MWT, in both SS and maximum speeds, showed a strong correlation with the GDI-SCI (Figures 3A and B). This makes sense, as less altered gait kinematics allows for a higher preferred walking speed and thus also a maximum walking speed. Gait speed has classically been considered an indirect indicator of overall gait quality. 43 However, the variability of the SCI-GDI is slightly better encompassed by the maximum 10MWT than the SS 10MWT. This may be explained by the fact that walking at a “natural” preferred speed for an individual with a gait impairment in an “artificial” context such as a gait laboratory under the gaze of an evaluator is hardly consistent. Nevertheless, a maximum gait speed is more easily reproducible and consistent for an individual.
Good correlations were found between the SCI-GDI and the LEMS, TUGT, SS WISCI II levels, and SCIM IIIIOMob with decreasing correlation coefficients, respectively. The highest correlation coefficients belong to the LEMS and TUGT (r = .638 and −.582). Concerning the LEMS, a positive correlation justifies that better gait kinematics is associated with greater LE strength (Figure 3C). Previous studies have already pointed to LEMS as a predictor of ambulatory outcomes.44–46 With respect to TUGT, being a speed-related balance test, a negative correlation is explained by the fact that individuals with less altered gait kinematics spend less time completing the required task (Figure 3D). Moreover, TUGT includes several walking and balance skills (standing up, walking, turning, and sitting) related to overall community walking ability, 34 in which gait kinematics plays a relevant role. Regarding the predictive capability of LEMS and TUGT to explain SCI-GDI data variability, TUGT was superior to LEMS. This could be explained by the fact that 2 individuals with iSCI showed SCI-GDI values similar to a normal gait pattern (close to and above 100) but a LEMS with a score below the maximum, which is 50 (Figure 3C). The latter could be justified given the difficulty for the clinician to differentiate muscle strength grading 4 and 5 during the physical examination of the individual (moderate and full resistance expected from an otherwise unimpaired person, respectively, in a muscle specific position for an active movement). 1 Good correlations were also found between the SCI-GDI and SS WISCI II levels, and SCIM IIIIOMob. However, lower Spearman’s correlation coefficients were obtained (r = .521 and .501, respectively). These results are congruent with the categorical nature of the measures. WISCI II considers the degree of impairment of individuals with SCI according to the use of AADs, orthoses, and the physical assistance needed to walk a 10-m distance. As shown in Figure 3E, SS WISCI II levels 16 and 20 present a wide spectrum of SCI-GDI values. This means that the preferred gait of an individual with 2 crutches (SS WISCI II level 16) manifested different gait patterns. These results support the criticisms made by other authors regarding the WISCI II, as it does not incorporate spatiotemporal parameters which could better categorize walking ability.29,32,47 A ceiling effect is observed in SS WISCI II concerning the variability of SCI-GDI values and therefore gait impairment in population with SCI, what has already been described in the literature.48,49 As for the SCIM III, although it is not a specific scale for ambulation, the mobility items showed a good correlation with the SCI-GDI (Figure 3F). Like the WISCI II, the SCIM IIIIOMob assess individual’s walking needs concerning the use of AADs, orthoses, and the need for personal supervision, but in 3 different mobility contexts: indoors, moderate distances ranging from 10 to 100 m, and >100 m outdoors. A ceiling effect is also observed in SCIM IIIIOMob.
Finally, no significant correlations were found between the SCI-GDI and the maximum WISCI II levels (P = .013, Figure 3G), and MAS (P = .528, Figure 3H). Concerning the maximum WISCI II levels, unlike SS WISCI II, they correspond to the most demanding condition that individuals with iSCI were able to tolerate to walk 10m during 3DGA in terms of required AADs and orthoses. Only maximum WISCI II levels 16, 18, 19, and 20 were recorded to describe the variability of gait kinematics in relation to SCI-GDI data. About 26 of the 35 individuals were able to walk without any AAD, orthosis, or physical assistance (maximum WISCI II level = 20), but showed a wide range of SCI-GDI values that did not always approach a normal gait pattern, as these individuals preferably used some ADD or orthoses in their daily life. Thus, a ceiling effect is also observed in WISCI II for the maximal condition. Besides, maximum WISCI II level 16 was the most demanding condition for 6 of the 35 individuals in the sample with iSCI, but their SCI-GDI values were spread out. In relation to MAS, the absence of significant correlation could be explained by the type of construct assessed, that is, spasticity. Even though spasticity may be helpful in individuals with iSCI who present LE weakness by enabling them to walk with less assistance, it may also limit gait kinematics. We hypothesize that the latter might justify the relationship found between spasticity and gait kinematics in the sample with iSCI. Nevertheless, the score is only classified into 6 grades according to the increase in muscle tone and the resistance in the range of motion (ROM), which is insufficient to describe the wide variability of gait kinematics in population with iSCI: only 16.7 % of the variability of SCI-GDI was explained by the MAS. Previous studies have already shown inconsistent results in terms of inter-rater reliability and test–retest reliability of MAS in SCI, limiting its validity.25,41,50–53 Although the measurements were taken in a standardized manner by the same clinician, different factors already reported in previous studies hindered the assessment of spasticity in the iSCI population, such as the difficulty to distinguish spasticity from passive viscoelastic changes, the presence of clonus, and ROM limitation. 50 Despite the fact that the MAS is the most widely used tool to assess the effectiveness of rehabilitation interventions for the treatment and management of spasticity in the SCI population, 54 other tools like the Spinal Cord Assessment Tool for Spastic reflexes, 55 which has been shown to be a valid measure of 3 different types of spastic motor behaviors in SCI, could be considered an alternative to the MAS for assessing the construct validity of the SCI-GDI.
Overall, the results of this study showed construct validity of the SCI-GDI for an independently walking adult population with iSCI. The same procedure should be carried out with broader levels of gait impairment to assess the discriminative validity of the SCI-GDI, including pediatric population.
Authors encourage the use of the SCI-GDI in clinical settings with the aim of detecting gait kinematic changes following rehabilitation interventions in iSCI adult population, such as treatment of spasticity, provision and fitting of orthoses, or assessment of different gait therapy approaches. Since walking ability is a complex set of functional tasks involving not only gait kinematics, but also spatiotemporal aspects of gait and balance, the authors recommend performing gait assessment by the combined use of SCI-GDI with other outcome measures whose strong and good correlation with SCI-GDI has been demonstrated in this study, thus covering the whole functional spectrum of walking ability in iSCI. Eventually, future work should aim to propose a new metric that considers SCI-GDI results together with other walking and balance outcome measures as a whole set.
Study Limitations
Our study presents some noteworthy limitations. The main one is the variability of gait functionality of the iSCI population sample, affected by the health situation associated with the COVID-19 pandemic. Further studies with a more heterogeneous sample are needed to analyze the psychometric characteristics of the SCI-GDI. Secondly, the construct validity of the SCI-GDI has only been tested using a subset of the most representative collection of measures to account for the whole functional spectrum of walking ability in SCI. This is due to the limited time available to perform both the outcome measures and the 3DGA prior to the onset of fatigue in individuals with iSCI. Nevertheless, correlation of the SCI-GDI with other valid walking and balance outcome measures would be of interest, providing complementary data to those presented in this work. Lastly, most of the iSCI subjects included in the study were classified as C or D by the AIS, meaning that in many cases they had sensory and proprioception impairments that directly influenced gait functionality. Furthermore, individuals with iSCI who were able to walk independently without the need for any external assistance were allowed to participate in the study. Therefore, both the use of AADs and orthoses, ankle-foot orthoses in all the cases, also had an impact on the modification of gait kinematics and on the TUGT and 10MWT results. Nevertheless, the safety of each of the participants during the performance of the test was considered as the top priority of the study.
Conclusions
This study demonstrates that the SCI-GDI correlates with the most representative set of walking ability outcome measures (10MWT, TUGT, LEMS, SCIM IIIIOMob, and SS WISCI II levels) in adults who walk independently after iSCI, indicating construct validity to assess gait impairment in this population. Future work will include assessing the psychometric characteristics with a more heterogeneous sample, also considering the pediatric population.
Footnotes
Acknowledgements
We acknowledge all participants (hospital staff, students, and patients) who accepted voluntarily to be part of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by MCIN/AEI/10.13039/501100011033 and by “ERDF A way of making Europe” [RTI2018-097290-B-C31, PID2021-124111OB-C33].
