Abstract
Background
Reaching is a fundamental motor skill often impaired in cerebral palsy (CP). Studies on manual function, intervention, and underlying brain mechanisms largely focus on unilateral CP. This first electroencephalography (EEG) evaluation of reaching exclusively in bilateral CP aims to quantify and relate brain activation patterns to bimanual deficits in this population.
Methods
A total of 15 children with bilateral CP (13.4 ± 2.9 years) and 13 with typical development (TD: 14.3 ± 2.4 years) performed 45 reaches per hand while recording motion capture and EEG data. The Box and Blocks test was administered bilaterally. Cortical sources were identified using independent component analysis and clustered using k-means. Alpha (8-12 Hz) and beta (13-30 Hz) band event-related desynchronization (ERD) values were compared across groups and hands within clusters, between dominant and non-dominant sensorimotor clusters, and related to reach kinematics and the Box and Block test.
Results
The group with CP demonstrated bimanual motor deficits with slower reaches, lower Box and Blocks scores, and stronger hand preference than in TD. Beta ERD, representing motor execution, was notably higher in the dominant sensorimotor cluster in CP compared to TD. Both groups demonstrated more contralateral than ipsilateral activity in both hands and clusters, with CP showing a less lateralized (more bilateral) alpha response. Higher brain activation was generally related to better function.
Conclusion
Bimanual deficits in bilateral CP and related EEG differences warrant more clinical and research attention particularly earlier in life when greater potential for neural and functional recovery exists.
Introduction
Reaching is an important developmental milestone and fundamental motor skill. Individuals with cerebral palsy (CP), the most common childhood-onset motor disorder, 1 often demonstrate poor motor control, 2 which can negatively impact reaching ability and everyday functioning.3,4
The type and severity of functional deficits in CP are dependent on the extent and location of the damage to the developing brain. 5 CP is generally categorized as unilateral or bilateral, depending on which brain hemispheres are affected. 6 CP can also be classified by functional abilities using the Gross Motor Function Classification System (GMFCS) 7 and Manual Ability Classification System (MACS), 8 which describe levels of mobility and functional arm and hand use, respectively. Both have 5 levels, with Level I indicating the highest function. In unilateral CP, upper limb involvement is often the primary intervention focus, compared to bilateral CP where mobility is often of greatest concern. This difference is evident in the CP literature where the preponderance of studies on upper limb assessment and intervention contain only or predominantly children with unilateral CP. In a recent review on upper limb motor planning in CP, only 3.5% of individuals in included studies had bilateral CP. 9 Similarly, in another review on the evaluation and characterization of manual function, 23% of participants had bilateral CP. 10 However, the disproportionate focus on upper limb function in unilateral CP does not signify that manual function is not impaired in bilateral CP. Makki et al 11 evaluated upper limb function in 100 children, 26 with unilateral and 74 with bilateral CP. They reported that 83% had hand control difficulties and that ABILIHAND logit scores were lower for bilateral compared to unilateral CP, indicating that upper limb disability may be as great or even greater in bilateral CP.
One of the few studies specifically comparing reaching with the dominant hand across CP subtypes and controls showed significantly reduced quality of reaching kinematics in bilateral compared to unilateral CP. Both CP groups performed worse than controls with slower reach velocity, greater curvature, and more movement units per reach. 6 Non-dominant hand function and the neural correlates of the presumably bimanual deficits in bilateral CP remain important knowledge gaps.
Functional neuroimaging studies in CP during motor tasks, particularly functional magnetic resonance imaging (fMRI), also show a unilateral CP bias, with none focusing exclusively on bilateral CP. This is perhaps in part because children with unilateral CP tend to be in GMFCS levels I to II and are less likely to have involuntary movements or heightened startle responses that limit the acquisition of high-quality images. Advances in mobile neuroimaging technologies such as electroencephalography (EEG) and functional near infrared spectroscopy (fNIRS) and processing methods have made the study of cortical processes involved in motor planning and execution more feasible for those with greater neurological involvement. The most common cause of bilateral CP is intraventricular hemorrhage after preterm birth, which primarily affects central sensorimotor regions (lower limbs) and may extend laterally to affect upper limbs as well. The process of brain development and organization with bilateral injury is uniquely different from unilateral injury, because there is no relatively intact hemisphere to serve as a scaffold. 12 Therefore, it is imperative to conduct imaging studies exclusively on bilateral CP. Whereas children with unilateral CP may experience limited function in 1 arm and hand, in bilateral CP both may be impaired, affecting unimanual and bilateral abilities and presenting unique rehabilitation challenges.
Motor-related EEG research primarily focuses on the sensorimotor or mu rhythm, which is comprised of alpha (8-13 Hz)13,14 and beta (15-30 Hz)15,16 frequency bands that fluctuate in power during a motor task such as reach-to-grasp. 17 These fluctuations, termed event-related synchronization (ERS) and desynchronization (ERD), represent movement-related changes in activity in sensorimotor brain regions. 18 While the brain response is most robust during voluntary movement, similar modulations are also seen with motor imagery, somatosensory stimulation (eg, vibratory or electrical stimulation), and action observation. 17 In healthy individuals, alpha and beta ERD occurs during movement planning and continues throughout execution. 18 Movement related activity also occurs in other frequency ranges, including slow cortical potentials that signal movement intention or low gamma ERS time-locked to movement onset and are presumed to be related to the timing of a movement sequence. 19
Démas et al 17 summarized existing motor-related EEG literature in CP, including what is known about brain oscillatory activity in those without brain injury. They reported that most EEG studies in CP are on adolescents, and except for 1 case study in a 4-year-old, all participants have been older than 5. All studies were on unilateral CP or a mixed cohort, with none focused exclusively on bilateral CP. Lee et al 20 published a case report including 2 participants with bilateral CP and 2 with unilateral CP and describing active brain areas in EEG compared to fMRI, but did not present ERD data which evaluates the magnitude of activation. Only 1 study has quantified ERD during an upper limb task in unilateral CP compared to controls, 21 finding decreased alpha band ERD over the contralateral (injured) sensorimotor regions when using the affected limb in unilateral CP compared to controls. Other studies mentioned in that review reported EEG findings without ERD data or controls (eg, Lee et al, 20 Rigoldi et al, 22 and Weinstein et al 23 ). In 2 EEG studies published after the Demas et al review, we identified alpha and beta ERD during treadmill walking in children with and without unilateral CP, showing similar trends across sensorimotor hemispheres as Inuggi et al 21 found for reaching.24,25
The data reported in this study are a subset from a larger study on electrocortical activation in motor-related brain regions combined with motor performance data during 2 specific unimanual tasks in bilateral CP and an age-matched cohort with typical development (TD): (1) reaching for blocks from a standard rest position, which is the focus here; and (2) grasp and transport of sets of blocks across the midline to a specific target. Results from the transport task revealed slower transport times and less direct movement trajectories in both hands in the cohort with bilateral CP compared to controls. Greater ERD was seen in the dominant motor cluster with the more affected hand in CP compared to the less affected hand and both hands in controls. 26 Generally, higher EEG activation when using the more affected hand was related to poorer motor performance as measured by task speed and directedness of the movement, and scores on the Box and Blocks Test (BBT).
Specifically, in this study, EEG data were recorded over the entire cortical surface during reaching with each hand in bilateral CP compared to an age-matched cohort with TD. Reaching performance using motion capture data and the BBT, a well-validated clinical test of unimanual dexterity, 27 were also measured to quantify the degree of impairment in our participants with CP and for use as correlates with the EEG outcomes, to relate motor behavior to brain activation patterns. Aims were to: (1) compare EEG outcomes, specifically alpha and beta ERD, during reaching across groups and hands; and (2) relate EEG findings to manual abilities in bilateral CP, to potentially inform the development of more effective rehabilitation paradigms in this subtype. A major advantage of EEG is that we can evaluate brain activation during functional motor tasks such as reaching and walking, but this requires simultaneous quantification of brain activation and motor performance to better understand mechanisms underlying abnormal control. We hypothesized that children with bilateral CP would show poorer upper limb function with both hands and greater alpha and beta band ERD compared to controls and that increased ERD would correspond to greater reaching impairments.
Methods
Participants
Thirteen children with TD (14.3 ± 2.4 years) and 15 with bilateral CP (13.4 ± 2.9 years) participated, with no significant age difference between groups (P = .40). Ten were born preterm with periventricular leukomalacia (PVL). The remaining 5 were full term with spastic (n = 2) or dystonic/dyskinetic CP (n = 3) of unknown etiology (Table 1). Inclusion criteria were diagnosis of bilateral CP or neurotypical development, 5 to 18 years, and ability to reach and grasp objects with both hands (MACS level I-III in CP). All refrained from caffeine for 24 hours prior, as it can affect brain activation. Children with uncontrolled seizures were excluded. The protocol was approved by the National Institutes of Health Institutional Review Board with informed consent and assent obtained for all participants.
Demographics for Participants With Cerebral Palsy (CP) and Typical Development (TD).
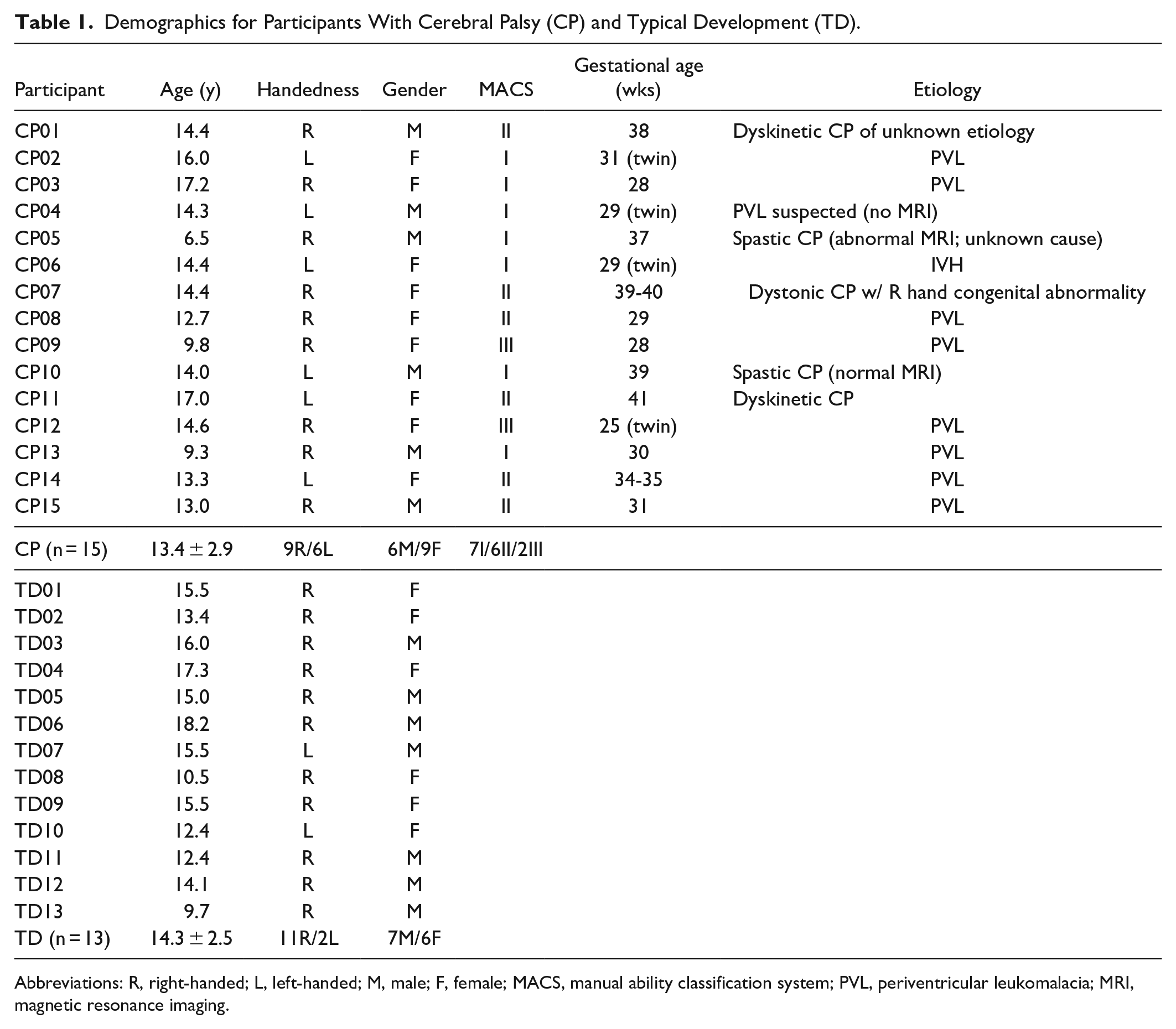
Abbreviations: R, right-handed; L, left-handed; M, male; F, female; MACS, manual ability classification system; PVL, periventricular leukomalacia; MRI, magnetic resonance imaging.
Procedures
Box and Blocks Test
Prior to EEG data set up and collection, all performed the BBT with each hand by transferring as many blocks as possible out of a total of 150 from 1 compartment to another in 60 seconds with 1 hand. 27 Specifically, the compartment containing the blocks at the start was always on the side of the hand being tested, and the task was to transfer blocks laterally 1 at a time and as fast as possible to the empty compartment in front of the opposite hand. The BBT measures unimanual dexterity and has been validated in CP and other clinical populations, with normative data available for right and left hands and in males and females starting at age 3 years. 28
Experimental Task
Reflective markers were placed on both hands, and the EEG cap and electrodes were placed on the participant’s head while seated in front of a table with 3 blocks arranged in 3 squares on the same side of the table as the reaching hand (Figure 1). Placing the designated hand palm down on the center of the table defined the start and rest position. First, a 2-minute rest trial was collected. Reaching trials started with a 3 second rest. On the verbal cue to “Go,” participants reached for the closest of 3 blocks. Then, they were to transfer it and subsequently each of the other 2 blocks to the corresponding empty squares on the opposite side of the table. Data from the transfer portion were analyzed in a separate study. This study only examined the period from the cue to the grasp of the first cube. Each performed 45 reaches with each hand. Digital video was recorded to evaluate performance, including difficulties reaching for or grasping blocks.

Experimental set-up showing a participant after receiving the cue to reach for and grasp the closest block on the same side of the table as the reaching hand.
Kinematic Methods
A 12-camera motion capture system (Vicon Motion Systems Ltd, Denver, CO, USA) recorded kinematic data via reflective markers placed bilaterally on the dorsal midpoints of the wrists, second and fifth metacarpals, and fingernails of the thumb and index fingers. A simple kinematic model was constructed in Visual 3D software (C-Motion, Inc., Germantown, MD, USA) in order to track task time and hand trajectory. Reach start was defined as when hand velocity first exceeded 0.05 m/s following rest. Reach end was defined as when hand velocity reached its minimum (block contact). Reach time was calculated as the time from reach start to end. Path length was the 3D distance the hand traversed during the reach.
EEG Methods and Data Analysis
EEG data were collected using a 64-channel Brain Products EEG array (Brain Vision, Morrisville, NC, USA). Electrodes were configured according to the 10 to 20 international system (Easy Cap, Germany), and impedance was verified as below 20 kHz before data collection.
EEGLAB open-source software 29 was utilized for EEG data processing, and the processing stream we employed is detailed in an earlier publication. 26 Briefly the steps included removal of bad channels, attenuation of power line noise using the EEGLAB Cleanline function, 30 creation of a merged file comprising all rest and experimental conditions, visually scrolling the merged file and removal of noisy periods, down-sampling to 250 Hz, application of Artifact Subspace Reconstruction (ASR), 31 re-referencing to a common-average, and 1 Hz high-pass filtering.
Adaptive Mixture Independent Component Analysis 32 parsed mixed scalp signals into maximally temporally independent component (IC) signals. We applied the Independent Component Analysis (ICA) weights computed from processed data to the individual datasets for each condition and hand without ASR applied.
EEGLAB DIPFIT algorithm was used to generate a best-fit equivalent current dipole for each IC. 33 Before clustering, we reversed the dipole locations of left-hand dominant participants, to align data by dominant and non-dominant instead of left and right hemispheres. Dipoles with residual variance >20% or topographical sparseness >5 were removed. 24 ICLabel, a machine-learning algorithm that generates a percent confidence that an IC is of cortical, ocular, muscular, or artifactual origin was applied. 34 Scalp topographies, power spectra, and dipole locations were evaluated together with ICLabel results, and ICs deemed of non-cortical origin were removed. Remaining IC time courses were epoched from 0.5 seconds before to 1.28 seconds after reach initiation. Epoch end time was calculated to encompass 95% of reaches in the slowest condition (non-dominant hand in CP). Rest data were subdivided into epochs of the same length for use as a baseline measure.
We then applied k-means to generate cortical clusters of in-terest so that on average there was 1 IC per subject per cluster.
We calculated a time-frequency spectrogram for each IC and condition with time-warping to account for varying reach durations, using the median reach latency per group (0.625 seconds for CP; 0.475 seconds TD). We computed the difference in the power spectrum between the trials and rests. EEG outcome measures included task-related mean ERD in alpha (8-13 Hz) and beta (15-30 Hz) frequency bands, described previously for quantifying event-related sensorimotor cortical activity. 17
Statistical Analysis
EEG and behavioral measures (path length, reach time, and BBT) were analyzed in IBM SPSS Statistical Software v29 using repeated measures general linear models (GLM) with hand as the within-group factor and group as the between-group factor. GLM analysis was performed for each EEG cluster for alpha and beta ERD values. Results with P < .05 were retained. A Shapiro-Wilks normality test was run on data with significant main effects or interactions, to determine appropriate post-hoc tests with non-parametric tests used for non-normal data. Otherwise, independent or paired-samples t-tests were utilized as indicated.
To elucidate hemispheric differences between bilateral CP and TD groups, a GLM was run to compare EEG data from the sensorimotor regions.
To relate task-related alpha and beta ERD values to the corresponding behavioral data, we performed Pearson correlation procedures (P < .05) separately by group.
Results
Behavioral Results
Main effects for group and hand were found for path length, reach time, and BBT, and a significant hand by group interaction was found for kinematic measures. (Table 2 shows means by group and hand with p-values for group comparisons). The group with CP had longer path lengths with the non-dominant hand (0.35 ± 0.04 meters) compared to TD (0.29 ± 0.02 meters), with no significant group differences in the dominant hand (CP: 0.31 ± 0.4 meters; TD: 0.29 ± 0.03 meters). Reach times were greater for both hands in CP (dominant: 0.65 ± 0.17 seconds; non-dominant: 0.87 ± 0.32 seconds) compared to TD (0.50 ± 0.08 seconds for both hands). The hand by group interactions for kinematic measures were explained by the CP group demonstrating poorer performance with the non-dominant compared to the dominant hand, while the TD group had no significant differences between hands.
Mean Behavioral Outcomes for the Groups With Typical Development (TD) and Cerebral Palsy (CP) With P < .05 Considered Significantly Different.
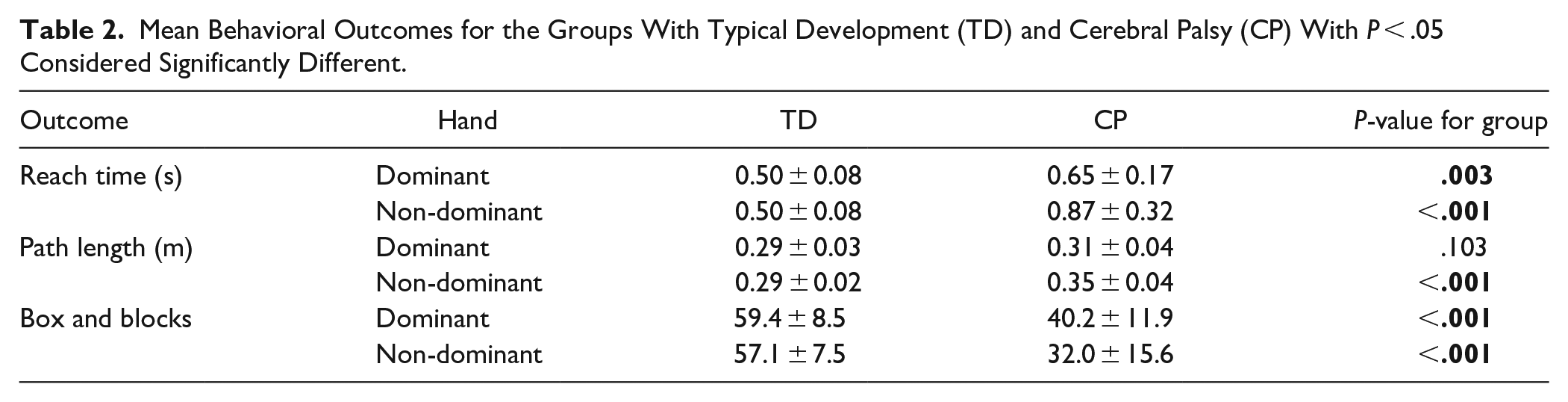
The group with CP also demonstrated significantly lower BBT scores (dominant: 40.2 ± 11.9; non-dominant: 32.0 ± 15.6), ie, transferred fewer blocks, than TD (dominant: 59.4 ± 8.5; non-dominant: 57.1 ± 7.5) for both hands and greater asymmetry in performance across hands than seen in the group with TD.
EEG Results
Nine clusters were identified using k-means: non-dominant and dominant sensorimotor, non-dominant and dominant posterior parietal, non-dominant and dominant prefrontal, premotor, secondary sensorimotor, and superior parietal. Figure 2 shows topographical power maps for each cluster and the participants and ICs included in each. Supplemental Table 1 summarizes GLM results for clusters with significant findings.
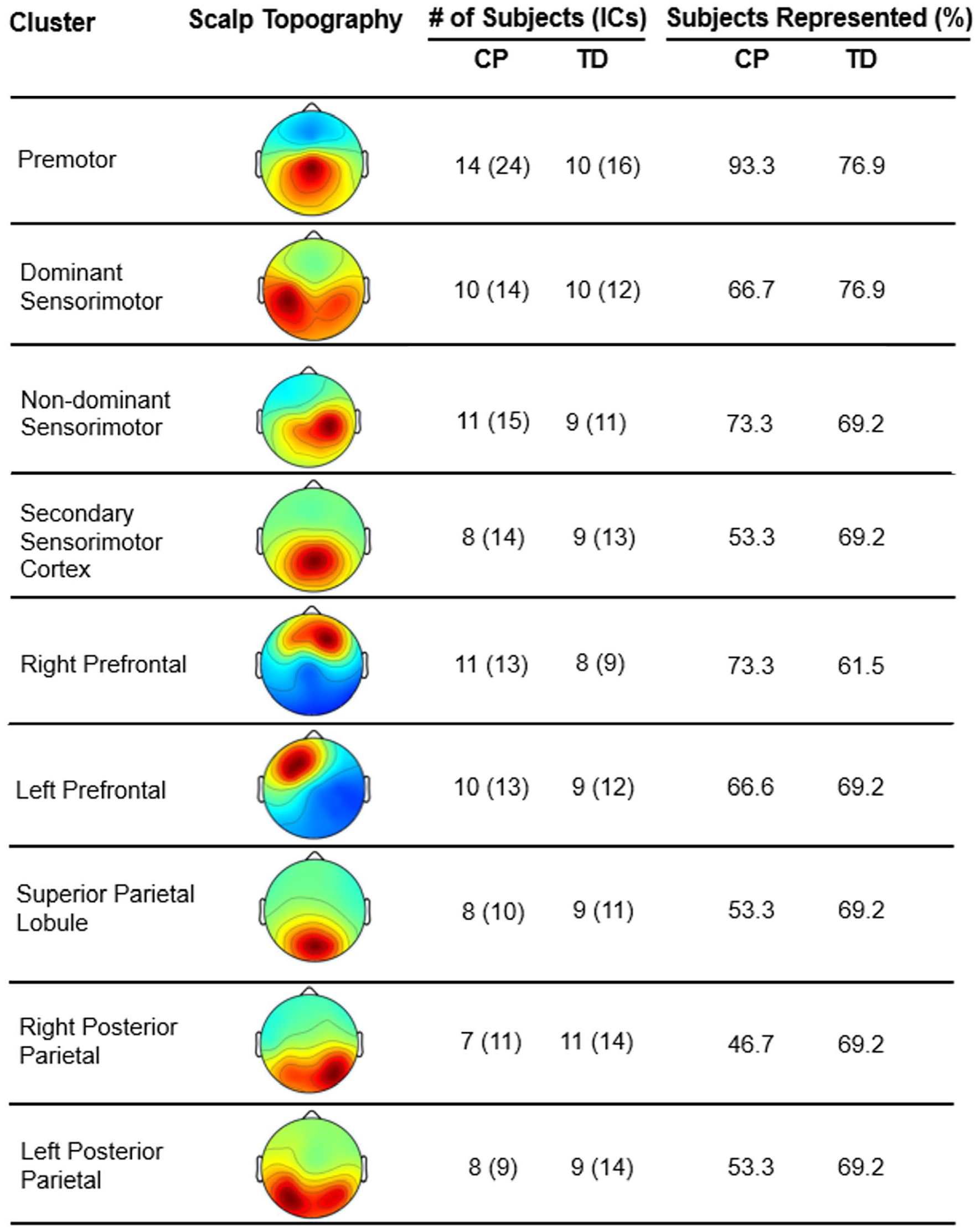
Clusters identified through k-means, including their scalp topography, with the number and % of participants per group who had independent components (ICs) in each cluster. Topographical maps do not reflect the artificially-flipped dipoles to account for left or right handedness. TD represents the group of typically-developing children and CP represents the group with cerebral palsy.
Group Effects Within Clusters
No significant main effects for group were found in the alpha band for any cluster. A main effect for group was found in the beta band only for the dominant sensorimotor cluster (P = .01). This reflected increased beta ERD in both dominant (CP: −1.51 ± 1.07 dB, TD: −0.61 ± 0.63 dB, P = .02) and non-dominant hands (CP: −1.52 ± 0.60 dB, TD: −0.62 ± −0.76 dB, P = .004) in CP compared to TD (Figure 3, top left).
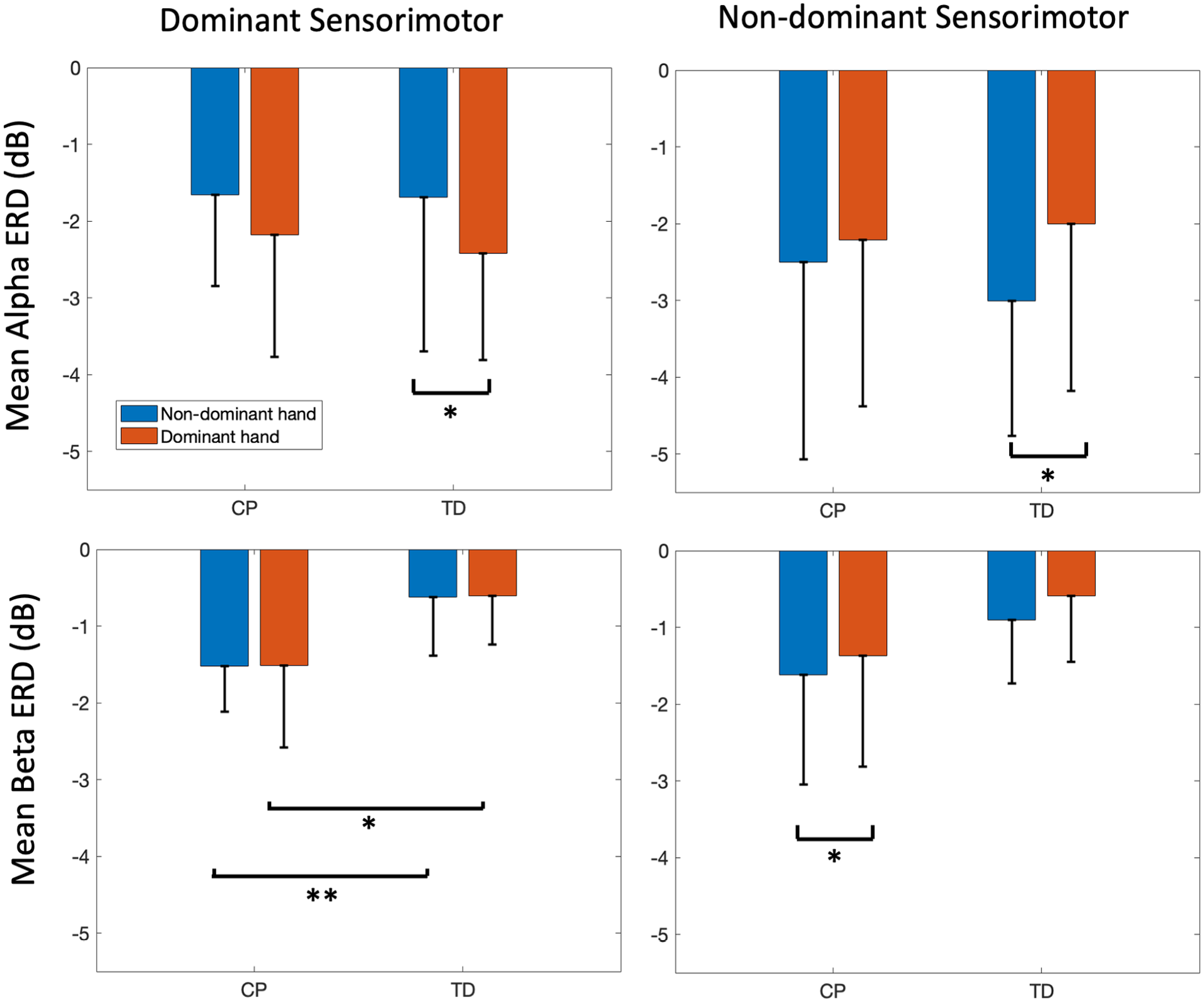
Bar graphs showing hand main effects for mean alpha (top) and beta (bottom) event-related desynchronization (ERD) in the dominant (left) and non-dominant (right) sensorimotor areas for each hand of the groups with typical development (TD) and with cerebral palsy (CP). Error bars represent the mean ± 1 standard deviation.
Hand Effects Within Clusters
Significant main effects for hand were found only in dominant and non-dominant sensorimotor clusters. In the dominant sensorimotor cluster, hand effects were found only for alpha ERD in TD (P = .03), reflecting greater activity when using the dominant (−1.66 ± 1.19 dB) compared to the non-dominant hand (−2.18 ± 1.59 dB, P = .03, Figure 3, bottom left). In the non-dominant cluster, hand effects were found for both alpha (P = .03) and beta (P = .03), with both groups demonstrating more contralateral than ipsilateral activity (Figure 3 bottom and top right, respectively). In the non-dominant cluster beta band, post hoc tests revealed significantly greater mean beta ERD in the CP non-dominant (−1.62 ± 1.42 dB) compared to the dominant hand (−1.37 ± −1.44 dB, P = .03). There was no significant hand by group interaction.
ERD Comparison Between Dominant and Non-Dominant Sensorimotor Clusters
Supplemental Figure 1 depicts the event-related spectral perturbation plots by sensorimotor hemisphere, hand, and group. Supplemental Table 2 lists mean ERD values for alpha and beta bands in these clusters by hand and group. A significant main effect for group was found in the beta band (P = .04), with CP showing overall greater mean activity with hemispheres combined. Post hoc tests revealed significantly greater activity for the dominant hand in the dominant hemisphere in CP compared to that same hand in TD, with all other data in CP showing a similar but non-significant pattern (Figure 4). There were no significant main effects for hand or hemisphere or interactions for beta ERD.
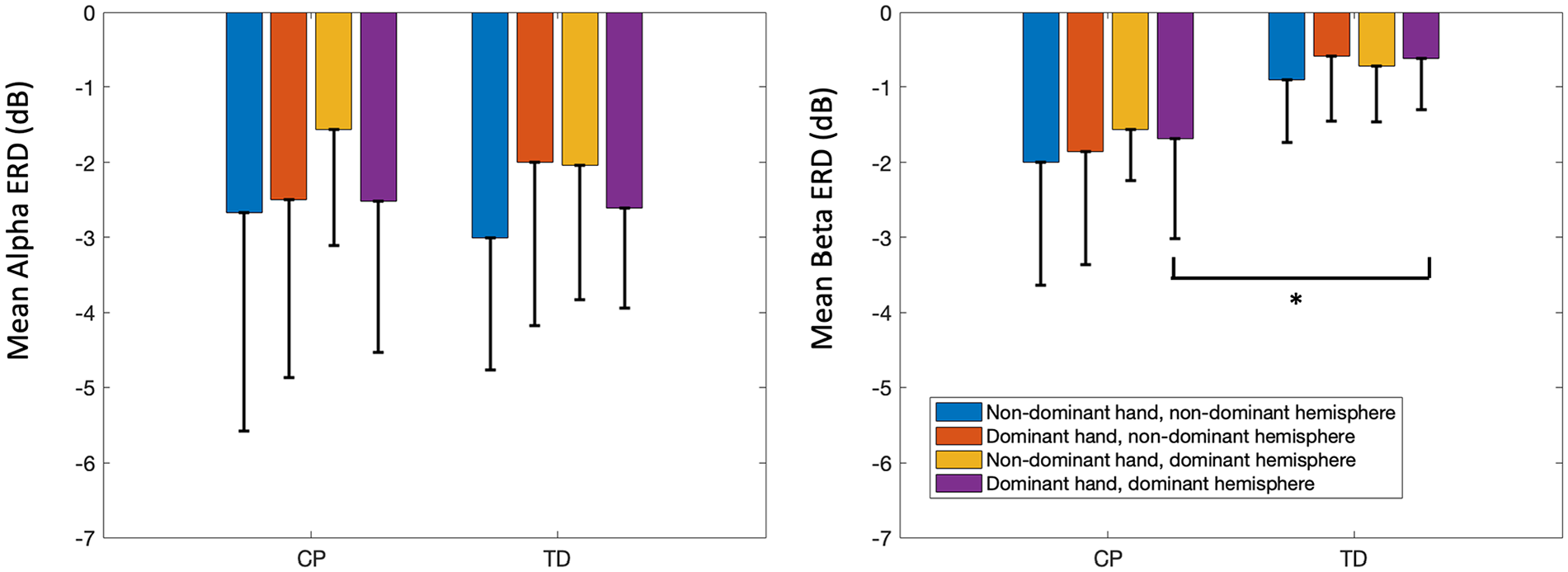
Bar graphs showing mean alpha (left) and beta (right) event-related desynchronization (ERD) across groups with typical development (TD) and with cerebral palsy (CP) to demonstrate the hand*hemispheres interaction in the sensorimotor areas. Error bars represent the mean ± 1 standard deviation.
In the alpha band, there was only a significant interaction between hemispheres and hands (P = .03), explained by lower activation in the dominant and non-dominant hemispheres when using the non-dominant hand along with higher activation in the non-dominant hemisphere when using the dominant hand in CP relative to corresponding values in TD, even though the overall amount of ERD across groups did not differ.
Correlations of EEG and Behavioral Measures
Table 3 shows correlations between alpha and beta mean ERD values and behavioral measures (path length, reach time, and BBT scores) for each group. Two clusters in TD, the dominant sensorimotor (dominant hand) and right pre-frontal (both hands), showed significant inverse correlations between the Box and Blocks score and mean beta ERD, with greater ERD associated with better skill.
Pearson r Values for All Significant Correlations (P < .05) Between Motor and EEG Outcomes by Cluster, Hand, and Frequency, Within the Groups With Cerebral Palsy (CP) and Typical Development (TD).
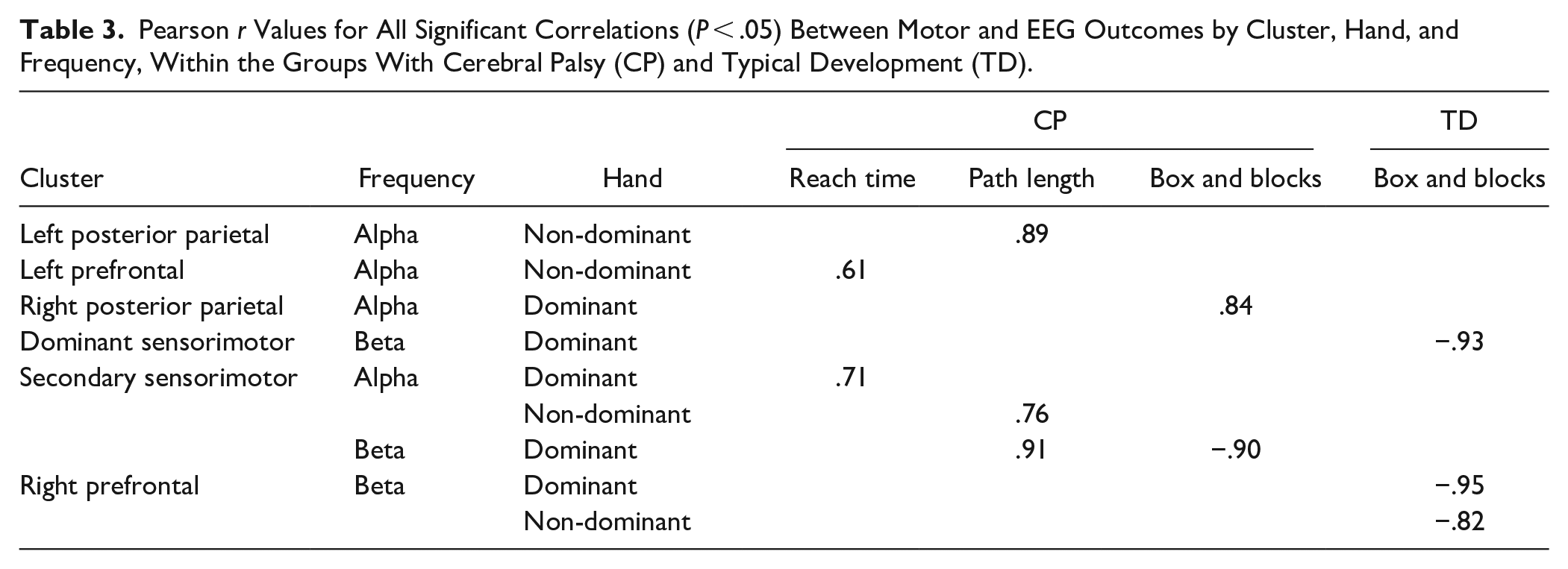
Four clusters in CP showed significant correlations between EEG and behavioral measures. In the secondary sensorimotor cluster, dominant hand Box and Blocks score was inversely correlated with the mean beta ERD, again with higher ERD associated with better performance. There was an opposite result for the dominant hand in the right posterior parietal region, where higher mean alpha ERD was associated with a lower (worse) Box and Blocks score.
All significant kinematic and EEG correlations in the left posterior parietal (non-dominant hand), left pre-frontal (non-dominant hand), and secondary sensorimotor clusters (both hands) in CP showed the same pattern across hands and frequency bands, with higher mean ERD associated with shorter (better) reach times and path lengths.
Discussion
This study is believed to be the first to quantify and relate spatiotemporal aspects and cortical activity in bilateral CP during reaching compared to controls and to employ EEG source localization techniques instead of using specific electrodes to represent different brain regions (ie, C3 and C4).21,22
As hypothesized, the group with bilateral CP had significant bimanual deficits that were relatively greater on 1 side, in contrast to TD who had similar kinematic results across hands. This supports the few other studies which evaluated motor impairments6,11 in bilateral CP. The BBT results even more clearly demonstrated the degree of impairment in bilateral CP, with a mean group difference of 19 and 25 (nearly 30% and 50%) fewer blocks transferred with the dominant and non-dominant hands, respectively, compared to controls. A recent study determined that the minimal clinically important difference in unilateral CP was 7 blocks, which was far exceeded by both hands in this study. 35 This cohort with CP also demonstrated slightly more asymmetry than TD in scores across hands, also clinically significant but still probably far more subtle than asymmetries across hands in unilateral CP. A prominent hand preference is an important red flag in early development that may be strongly indicative of unilateral brain or peripheral injury (eg, neonatal stroke or brachial plexus palsy, respectively). In contrast, less asymmetric bilateral deficits may be less apparent and therefore less likely to be addressed. Results from this and other studies suggest that greater attention be directed to evaluating and addressing upper limb function in infants with bilateral CP to leverage the greater potential for neuroplasticity.
Interest in utilizing EEG for studying motor-related brain activation has been increasing in recent years. The high temporal resolution and portability of EEG make it ideal for studies on neural mechanisms underlying normal and impaired motor skills, as compared to fMRI or MEG which lack portability 36 and only allow for the study of a limited number of movements performed in non-naturalistic settings. Weinstein et al 23 compared fMRI, transcranial magnetic stimulation, and EEG for evaluation of brain activation during upper extremity tasks in unilateral CP and concluded that EEG was the most feasible and reliable measure. MEG shares the exquisite temporal resolution of EEG but has better spatial resolution than EEG, which is challenged by volume conduction of electrical signals recorded at the scalp. However, advances in EEG source localization and artifact rejection techniques have enabled better spatial resolution of cortical signals during movement,30,37,38 thus improving its utility for functional neuroimaging.
In the current EEG analysis, each identified cluster included at least 50% of participants per group, except for the right posterior parietal cluster in CP, which included 48.7% of the entire sample. This contrasted with our findings in unilateral CP during walking where only 38% of participants with CP were represented in the non-dominant sensorimotor cluster compared to 91% of TD. 24 This difference across CP subtypes is presumably due to either more extensive damage to the non-dominant hemisphere or greater representation of the non-dominant limb in the uninjured hemisphere in unilateral CP.
Significant group differences in EEG activation were only seen in the dominant sensorimotor cluster, with greater beta ERD in each hand in CP compared to the corresponding hand in TD. The sensorimotor beta band reflects activity during motor execution, and greater activity in CP was coincident with bimanual kinematic differences. Finding a difference only in the dominant sensorimotor hemisphere suggests greater reliance on this hemisphere during movement in bilateral CP compared to controls. Deiber et al 39 demonstrated that unilateral finger tapping in healthy adults generated bilateral alpha ERD, indicating that some amount of bilateral activation is normal, but we propose here that an excessive amount is pathological. Our EEG results here are similar to results from our earlier fNIRS study during hand squeezing in TD, unilateral CP, and bilateral CP. The laterality index for bilateral CP when using the non-dominant hand showed relatively more activity in the contralateral versus ipsilateral hemisphere, but the mean value was almost exactly between that of TD (more strongly contralateral) and unilateral CP (slightly ipsilateral). 40 These results combined suggest that children with bilateral CP are not as lateralized as those with TD when using their more affected side. The reasons may be that the brain injury itself is asymmetric and/or the child develops an early preference for the more functional of 2 impaired limbs, leading to a more asymmetrical pattern of brain organization.
Main effects for hand were only seen in dominant and non-dominant sensorimotor clusters, largely showing similar patterns across groups. The alpha band showed an interesting hands by hemispheres interaction. Most activity was seen in the hemisphere contralateral to the moving hand, with less in the ipsilateral hemisphere. This was as expected in TD and also the case in bilateral CP. However, while ipsilateral activity was symmetric across hands in TD, in CP the dominant hand had relatively greater and the non-dominant hand relatively less ipsilateral activity, indicating differential developmental organization patterns across sides.
The lack of statistical differences in other clusters suggests that cognitive and sensory integration aspects for this task were likely minimal and/or did not present a significantly greater challenge to those with bilateral CP at the group level. However, given the range of upper limb impairment in this cohort, some relationships between function and EEG activity in other clusters were uncovered. Kinematic measures in both hands and dominant hand Box and Block scores correlated with mean ERD values in CP, with higher ERD associated with better performance in all cases except for the right posterior parietal cluster, where higher mean ERD was related to a worse dominant hand Box and Blocks score. The right posterior parietal region is involved in sensorimotor integration, which can be more challenging in CP, thus requiring more cortical effort. TD also had significant correlations between the Box and Blocks score, with higher ERD linked to a better score. Links between motor performance and related power changes have been reported in divergent directions in both typically developing and clinical cohorts. Inuggi et al 21 showed that in unilateral CP lower alpha ERD magnitude in the non-dominant hemisphere correlated with poorer function, similar to most results here. In contrast, in the MEG knee extension force matching study in bilateral CP, 41 greater ERD was associated with poorer function, and in our treadmill walking EEG study, greater activation in unilateral CP was needed for similar gait performance in TD. 24 Higher brain activation has been linked to greater motor proficiency in adults and infants with TD. 42 Conflicting results across studies suggest that the direction of correlation between activation and function may shift depending on the task and brain region involved as well as participant factors (eg, age and diagnosis).
Limitations in this study are similar to other studies in CP with respect to group heterogeneity. Even participants with the same CP subtype (ie, bilateral CP) were not all born preterm with diffuse white matter injury. Thus, one cannot assume that the disruptions or differences in cortical activation patterns from controls are similar across etiologies. Our sample was too small to examine whether different etiologies have their own unique patterns, but this is an important next step. Since Weinstein et al 23 propose that each child with CP has a unique response in how their brain organizes when facing the challenge of development with a brain injury, research designs that seek to understand this individual variability to a greater extent, rather than obscure it in mean group analyses, are also warranted. 43
Conclusions
Even on this simple yet fundamental motor task, children with bilateral CP exhibited bilateral deficits with notable asymmetry across hands, warranting far greater clinical and research attention on hand function in bilateral CP. An overreliance on the dominant hemisphere when using the non-dominant hand was uncovered that we hypothesize may be more amenable to training in bilateral than unilateral CP since most individuals have diffuse white matter injury and generally exhibit more activation in the hemisphere contralateral to the more affected hand.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231195044 – Supplemental material for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching
Supplemental material, sj-docx-1-nnr-10.1177_15459683231195044 for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching by Connor Phillips, Julia Kline, Christopher J. Stanley, Thomas C. Bulea and Diane L. Damiano in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683231195044 – Supplemental material for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching
Supplemental material, sj-docx-2-nnr-10.1177_15459683231195044 for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching by Connor Phillips, Julia Kline, Christopher J. Stanley, Thomas C. Bulea and Diane L. Damiano in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tiff-3-nnr-10.1177_15459683231195044 – Supplemental material for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching
Supplemental material, sj-tiff-3-nnr-10.1177_15459683231195044 for Children With Bilateral Cerebral Palsy Exhibit Bimanual Asymmetric Motor Deficits and EEG Evidence of Dominant Sensorimotor Hemisphere Overreliance During Reaching by Connor Phillips, Julia Kline, Christopher J. Stanley, Thomas C. Bulea and Diane L. Damiano in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was funded by the Intramural Research Program of the National Institutes of Health Clinical Center (Protocol # 13-CC-0110, PI: Damiano)
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
