Abstract
Background
Over activity of the rectus femoris is often cited as a main cause for stiff knee gait (SKG). Botulinum toxin (BoNT) can be used to reduce this over activity. Inconsistent results for the effect of BoNT injections were found in literature which can possibly be explained by the study design as these were uncontrolled or non-randomized studies.
Objective
To conduct a randomized controlled trial (RCT) to investigate the effect of botulinum toxin type A (BoNT-A) injections in the rectus femoris on gait kinematics and functional outcome in adult stroke patients.
Methods
Twenty-six participants were included in this triple-blind cross-over RCT. The intervention consisted of an injection with BoNT-A. Placebo is an injection with saline. Besides knee and hip kinematics, functional outcomes were measured.
Results
Comparison of the effect of BoNT-A injection to placebo injection showed a significant increase in peak knee flexion and knee range of motion of 6.7° and 4.8° respectively. There was no difference in hip kinematics. In functional outcomes, only the 6 Minute Walking Test showed a significant increase of 18.3 m.
Conclusions
BoNT-A injections in the rectus femoris is a valuable treatment option for stroke patients walking with a SKG to improve knee kinematics. To study the effect on functional outcome more research is necessary with different functional outcome measures that can capture the effect in kinematics. It is important to use kinematic measurements to demonstrate effects in quality of movement that are not captured by commonly used functional outcome measurements post stroke.
Introduction
Stiff knee gait (SKG) is characterized by a diminished knee flexion during the swing phase of gait and frequently affects patients with an upper motor neuron lesion (eg, stroke or traumatic brain injury). The lack of knee flexion can lead to foot clearance problems, increase the risk of tripping and falling and restrict the patients in their daily live activities.1,2 Several mechanisms that can cause SKG are described in literature. Some authors attribute SKG to overactivity of the vastii muscles of the quadriceps,3,4 others to weak hip flexors, 5 altered foot-ankle mechanisms (lack of push off), 5 overactivity of the rectus femoris,6-10 or decreased knee flexion velocity at toe-off in pre-swing. 11 The discussion about etiology is still ongoing, but overactivity of the rectus femoris is assumed to be the overarching cause.
Several treatment options for SKG are used in clinical care, mainly aimed at influencing the abnormal activity or force production of the rectus femoris. These include injections with botulinum toxin (BoNT) in the rectus femoris12,13 and/or a rectus femoris transfer.14,15
While the efficacy of BoNT injections to reduce abnormal activity or (spasticity) has been reported in literature, 16 inconsistent results for BoNT injections were found in the effect on kinematics and functional outcomes in stroke patients walking with a SKG. Robertson et al 17 and Hutin et al 18 found increases in peak knee flexion of 8° and 9°, respectively. In contrast, Stoquart et al 12 and Caty et al 19 found an increase of only 5°. In addition, inconsistent results were found in walking speed. Hutin et al, 18 Lampiere et al, 20 and Bernuz et al 21 reported a significant increase in self-selected walking speed calculated from the 10 meter walking test (10 MWT), while Tok et al 22 and Robertson et al 17 found no differences. Variable outcomes were also found on the 6 minutes walking test (6 MWT). Robertson et al 17 and Bernuz et al 21 measured no significant improvement in covered distance during the 6 MWT while Tok et al 22 found a significant improvement. Finally, energy expenditure decreased significantly in the studies of Tok et al 22 and Caty et al 19 while Stoquart et al 12 found no significant results for energy expenditure.
The variability of results reported in these studies can possibly be explained by the study design. Since all studies reported in literature so far are uncontrolled or nonrandomized studies, a potential placebo effect was not considered which could have biased the results. Only the study by Tok et al 22 used a placebo group, but this study lacks randomization and did not use a double-blind design which may cause additional bias in the results.
Furthermore, it is unclear if the effect of BoNT injection of the rectus femoris for reducing SKG also leads to significant improvements in functional outcomes, such as walking speed and walking distance, energy cost as well as patients self-reported quality of gait. Therefore, the primary objective was to evaluate the effect of botulinum toxin type A (BoNT-A) injection on knee and hip kinematics during swing. The secondary objective was to investigate if the effect of BoNT-A injection to reduce SKG also leads to improvements in functional outcomes and patients self-reported quality of gait.
To rule out bias by confounding factors such as blinding in the current trial, we performed a randomized triple blind placebo-controlled cross-over trial to examine the efficacy of BoNT type A injection into the rectus femoris on SKG in a group of stroke patients.
Based on the literature, we expect an increase in knee kinematics as well as improved scores on functional outcome measures.
Methods
Participants
Participants were recruited from 2013 to 2019 Roessingh, Center for Rehabilitation (Enschede, the Netherlands). Inclusion criteria were stroke survivors (at least 6 months post stroke), age over 18 years, activity of the rectus femoris in second part of initial swing and/or mid swing. This was determined by measuring muscle activity using surface electromyography (sEMG) of the rectus femoris and vastus lateralis simultaneously to differentiate between true activity and cross-talk.8,9 Subjects were able to walk independently with or without walking aids and had diminished peak knee flexion in swing (<50°) 1 as established by video observation and willingness to provide written informed consent.
Exclusion criteria were: Chemodenervation of the rectus femoris prior to the study, co-activation of the vastii in initial- or midswing, length of the rectus femoris <65° (measured with the slow Duncan Ely test), presence of a reduced joint range of motion impeding walking or other neurological problems. During participation in the study, participants were allowed to continue regular treatment. However, treatments focused on improvement of knee flexion were not allowed.
Study Design
This study was conducted as a triple-blind randomized controlled trial (RCT) with a cross-over design. The participants, the physician performing the BoNT-A and placebo injections and the researchers involved in post injection measurements were all blinded for group allocation. The study was approved by the local medical ethical research committee (MERC Enschede). The study was registered in the Netherlands Trial Register (NTR) with the unique identifier: NTR2169. Clinical trial registration: https://trialsearch.who.int/Trial2.aspx?TrialID=NTR2169.
A computerized randomization into 2 groups was done in blocks of 4 participants to ensure similar group size. Group 1 first received a BoNT-A injection followed by a wash out period of 5 months, which was followed by the placebo injection; group 2 first received the placebo injection followed by a wash out period of 5 months which was followed by the BoNT-A injection. A wash-out period was implemented to exclude a carry-over effect of the BoNT-A injections. 23 An overview of the study design is presented in Figure 1.
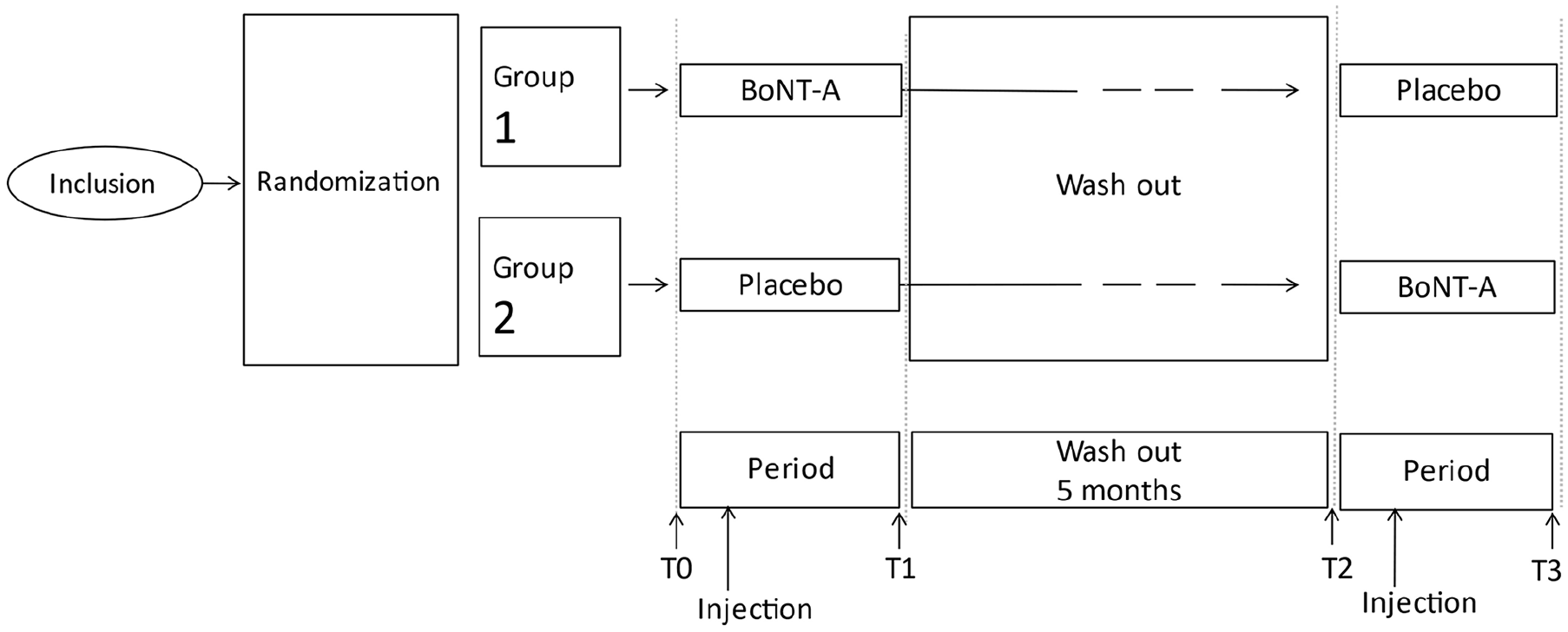
Study design RCT.
Study Protocol
Data were collected before and after the BoNT-A and placebo injection. This resulted in 4 measurement moments, T0, T1, T2, and T3 as can be seen in Figure 1. Pre injection measurements (T0 and T2) were done 1 week before the injection, for optimal peak effect of the BoNT-A injection, post injection measurements (T1 and T3) were done 4 to 6 weeks after the injection. Data collection consisted of 3D kinematics, determination of energy expenditure during walking and the execution of functional outcome measures and assessment of the perceived level of impairment. The data of one measurement moment (eg, T0) were collected in 1 day. The randomization key was released when all data were fully analyzed.
Study Drug Intervention
Interventions consisted of injecting BoNT-A or a placebo into the rectus femoris.
A total amount of 200U BoNT-A (Botulinum Toxin Type A, Allergan) was injected in the rectus femoris, at 6 points (6 injections of 33U) according to the method of Stoquart et al. 12 For the placebo a saline (NaCl 0.9%) solution was injected using the same procedure as the BoNT-A injection. All injections were performed by one experienced physician (A.N.) with the aid of intramuscular electrical stimulation (0-5 mA) for optimal needle placement of the injection.
Gait Analysis
To determine 3D kinematics, an infrared opto-electronic 3D-motion analysis system (VICON MX + 8 MX13 cameras, frame rate 100 Hz; Vicon Motion Systems, Oxford, UK) was used with a standard marker placement (lower-limb Plug-in-Gait model VICON Oxford UK 24 ). Initial contact was detected as the first point at which weight is detected on the force plate (20 N). Toe-off was detected as the first point at which the ground reaction force was not detected. During gait analysis participants walked on a 10-m walkway at least 20 times with sufficient rest periods. For data analysis, a minimum of 10 strides were analyzed. Outcome measures were calculated for each gait cycle and averaged the outcomes measures afterward. Participants walked at their natural comfortable speed and used the same walking aids, orthoses, and shoes during all data collection.
Kinematic Outcome Measures
Kinematics of knee and hip are calculated with an inhouse MATLAB based software. Peak knee flexion and peak hip flexion were defined as the maximum amount of flexion in swing. The range of motion of knee and hip was calculated as the difference between maximum extension in stance and maximum flexion in swing. Knee flexion angular velocity at toe off was calculated with a 5 points calculation.
Energy Expenditure
After the gait analysis session, energy expenditure was measured during the 6 MWT using the Cosmed K4 b2 system (Cosmed, Rome, IT). Participants completed the 6 MWT at a self-selected, comfortable speed on a circular track of 75 m. Patients were notified of the elapsed time every minute and were allowed to rest in case they were not able to walk any further. Patients were not allowed to talk during the 6 MWT. Assistive devices and necessary orthoses were allowed during walking but remained identical during the entire study.
To determine steady state during the 6 MWT, a moving window (of 2 minutes) was shifted over the measurement from 2 minutes after the start of the 6 MWT.25,26 This was done for 5 time blocks: 2-4 minutes; 2.5-4.5 minutes; 3-5 minutes; 3.5-5.5 minutes; and 4-6 minutes.
To check for steady state, O2 rate and Respiratory Exchange Ratio (RER; ration between VCO2 and VO2) were averaged over these time blocks and were checked whether they stayed within the borders of variance of 20%. 27 Furthermore, the respiratory exchange RER was checked whether it was below 1 as this is the threshold between aerobic and anaerobic activity. For every individual patient, the 5 time blocks were checked for the steady state conditions and RER score. The time block for which the stated conditions were met for most patients was used in the analysis. The energy cost was computed by dividing O2 rate by the walking speed (m/s) which was computed by dividing the covered distance on the 6 MWT by 360 seconds.
Functional Outcome Measures
Each participant completed the 6 MWT, Timed Up and Go Test, 10 m walk test, the Motricity Index, and the Rivermead Mobility Index. The Medical Research Council (MRC) score was used to assess the strength of the hip flexors and knee extensors. The Duncan-Ely Test was assessed to obtain information about the spasticity of the rectus femoris. The Visual Analogue Scale (VAS) (range 0-10) 28 and a Borg CR10 score (range 0-10) 29 were used to determine the subjective value of the experienced SKG during walking. A lower value of the VAS and Borg means less experienced SKG during walking. Finally, the Stroke Impact Scale (SIS) 30 was used to determine the level of participation of the subjects.
Gait analysis, functional tests, energy expenditure, and the analysis of the complete data set, were carried out by the same person (MT).
Statistical Analysis
Due to the design of the study as a randomized controlled blind cross-over trial, carry-over and period effects for each variable must be excluded before treatment effects can be analyzed. Testing for carry-over effects was done using an unpaired t-test for the sum of the variables at T0 and T2. Testing for period effects was done by an unpaired t-test for the difference between BoNT-A and placebo injection at T1 and T3.
Before statistical tests were chosen, the normal distribution of the data was checked. This was done using the Shapiro–Wilk test and a visual inspection of histograms.
In case data were normally distributed, we used the paired or independent samples t-test. For variables that were not normally distributed, a Wilcoxon Signed Rank test or Mann–Whitney U test was used.
For statistical analysis we use IBM SPSS statistics 19.0 for Windows (IBM Inc. Chicago, IL, USA).
Power Analysis
A power analysis showed a sample size of 23 patients to achieve 91% power to detect a mean differences of 5 with an estimated standard deviation of 7.3 (2-sided paired z-test, α = .05). Inclusion of 26 patients allowing a 10% dropout.
Results
In all, 26 patients were included in the study. One patient dropped out due to family problems. Data from this participant was not included in the analysis. All remaining 25 participants completed the study. No side-effects of the BoNT-A injections were reported. In contrast, 2 patients reported a small side-effect after receiving placebo. One patient reported headache for a short period on the day of the injection. The other person reported headache 1 day after the injection. Participant characteristics are shown in Table 1.
Participants Characteristics.
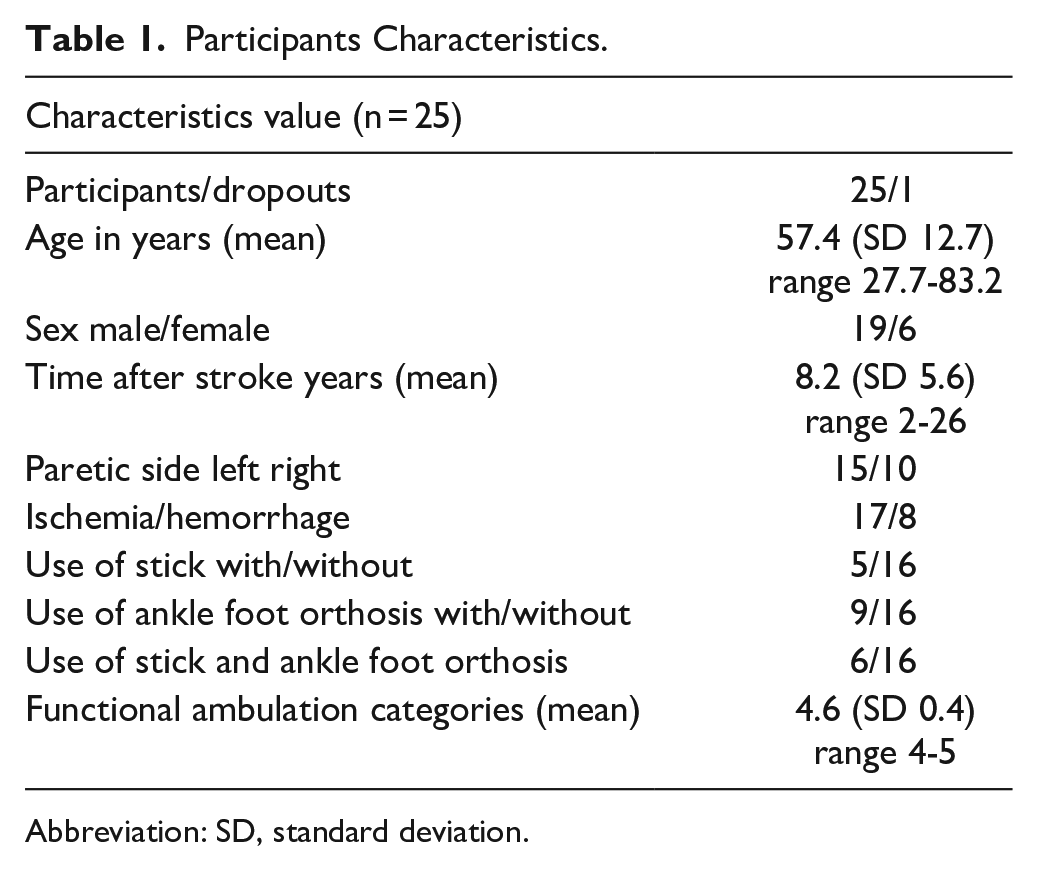
Abbreviation: SD, standard deviation.
Statistical Check for Carry Over Effect or Period Effect
The Motricity index Total score lower extremity was the only test that showed a carry-over effect. Therefore, we only used data from T0 and T1 in the statistical analysis. There was no significant difference (independent t-test) for the Motricity Total score.
For all other variables, all 4 data points were used and checked with the paired t-test.
Kinematics
All results are displayed in Table 2 (affected side). The between group analysis, comparison of the effect of the BoNT-A injection to the effect of the placebo injection, showed a significant difference of 6.7° in peak knee flexion in favor of the BoNT-A injection. Knee range of motion and knee angular flexion velocity at toe off showed a significant difference of 4.8° and 24.9°/s, respectively. The Cohen’s d effect size for peak knee flexion is 0.63 (medium to large effect). For Knee range of motion and knee angular velocity at toe-off is the Cohen’s d effect size respectively 0.41 and 0.31 (medium effect).
Kinematic Outcomes.
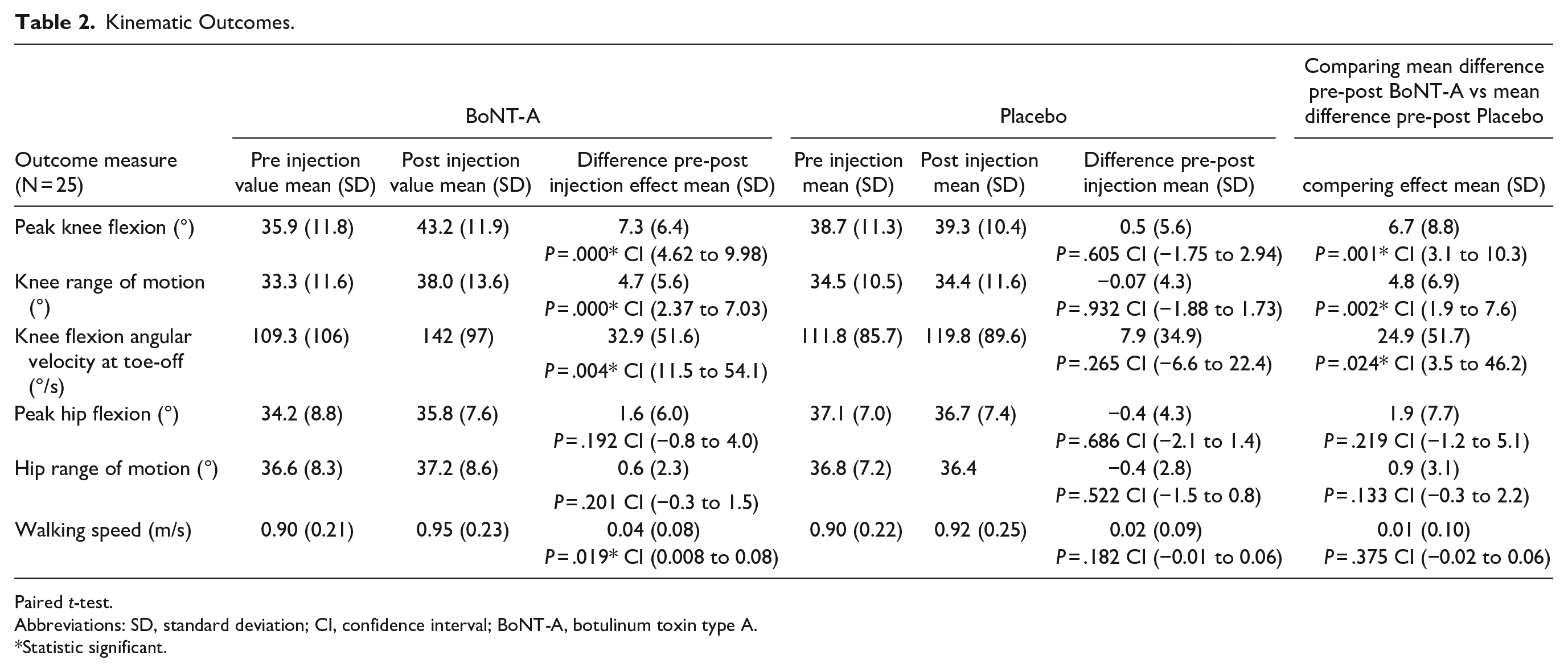
Paired t-test.
Abbreviations: SD, standard deviation; CI, confidence interval; BoNT-A, botulinum toxin type A.
Statistic significant.
No significant differences were found for the peak hip flexion and hip range of motion and walking speed.
Functional Outcome Measurements
All results are displayed in Table 3. Between group analysis showed a significant difference of 18.3 m on the 6 MWT and a significant decrease of 0.7 points on spasticity score of the Duncan Ely test in favor of the BoNT-A injection.
Functional Outcomes.
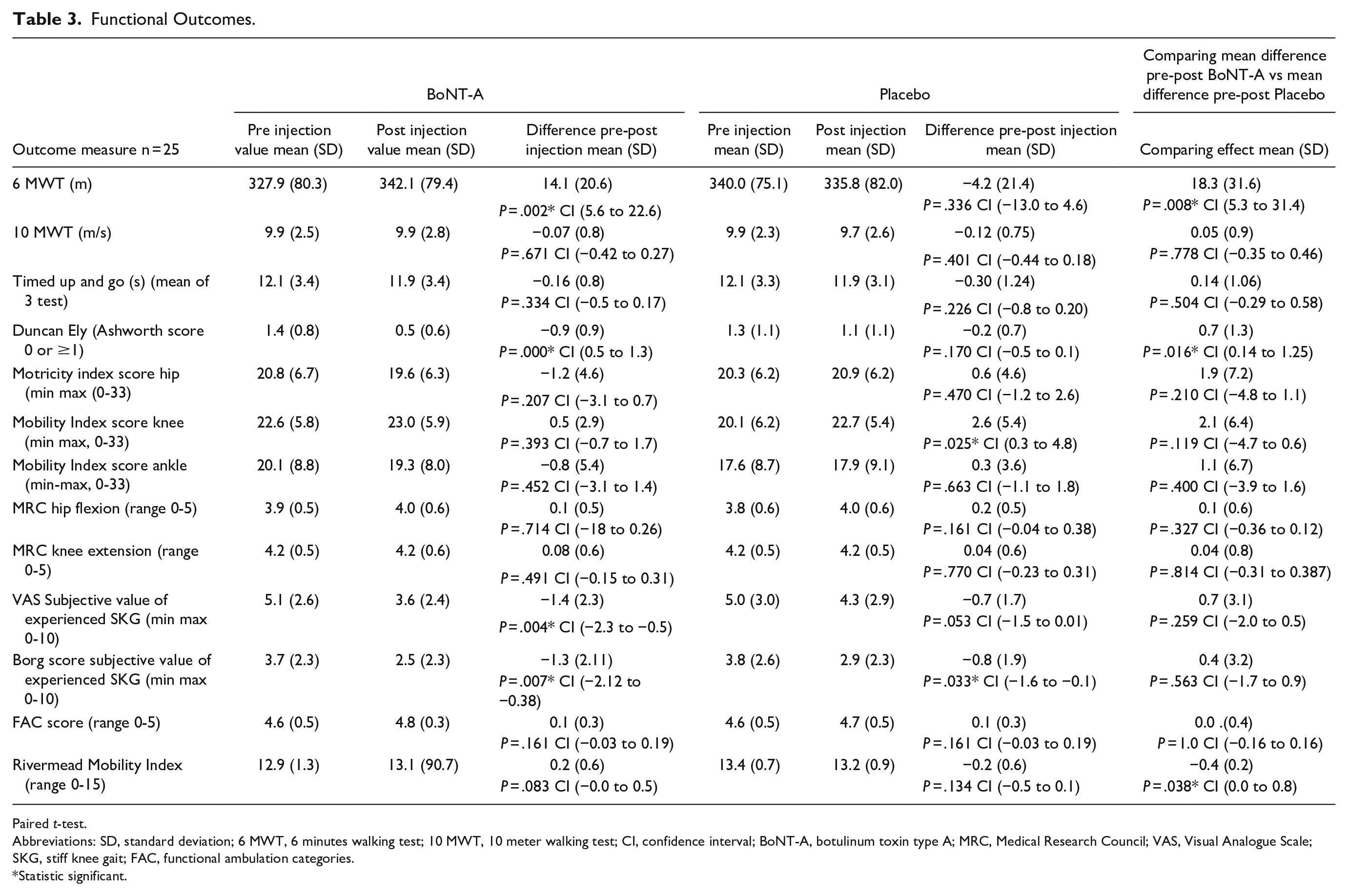
Paired t-test.
Abbreviations: SD, standard deviation; 6 MWT, 6 minutes walking test; 10 MWT, 10 meter walking test; CI, confidence interval; BoNT-A, botulinum toxin type A; MRC, Medical Research Council; VAS, Visual Analogue Scale; SKG, stiff knee gait; FAC, functional ambulation categories.
Statistic significant.
No significant differences were demonstrated in the other functional outcomes (see Tables 3 and 4).
Stroke Impact Scale.
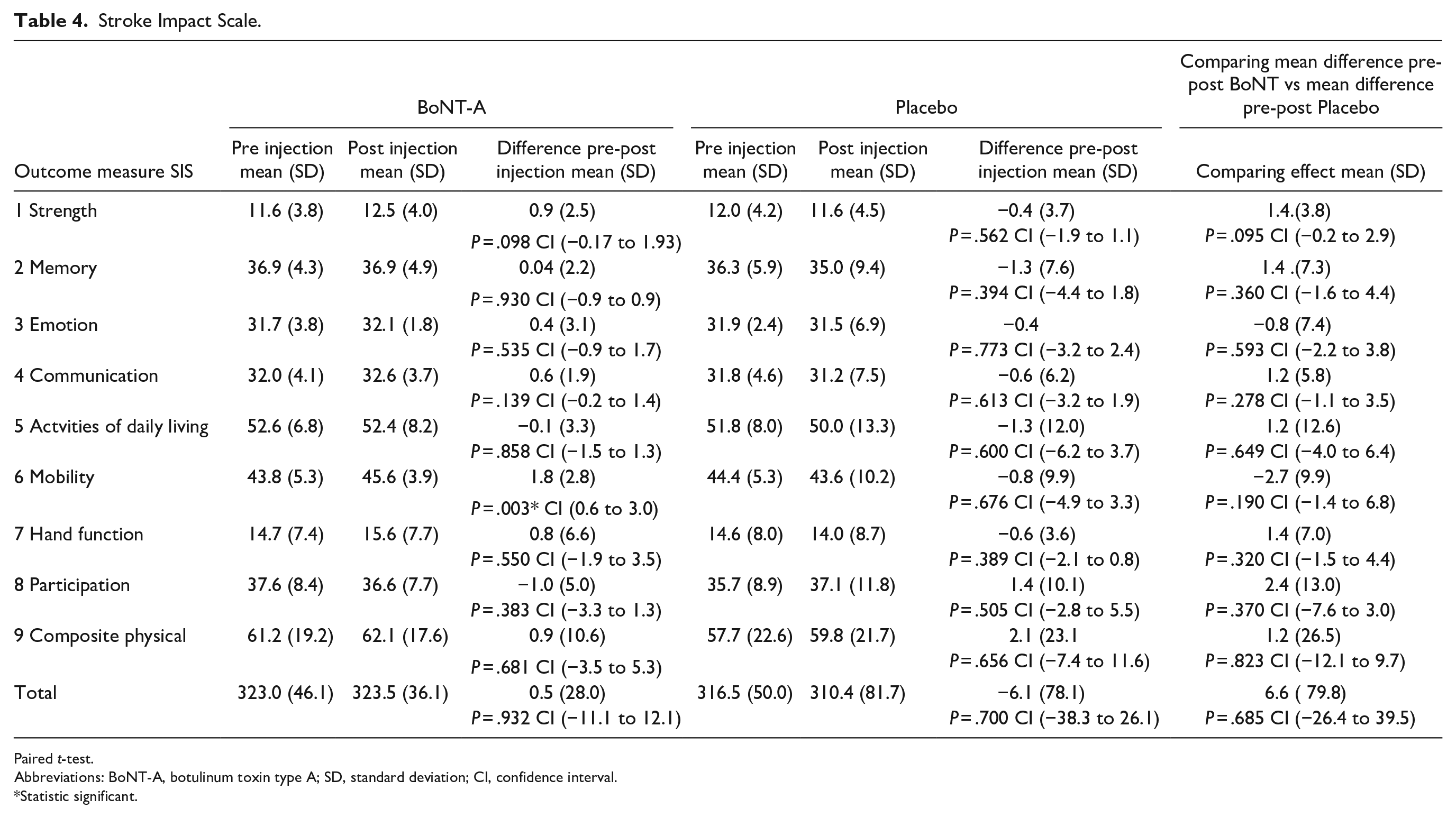
Paired t-test.
Abbreviations: BoNT-A, botulinum toxin type A; SD, standard deviation; CI, confidence interval.
Statistic significant.
Energy Expenditure
The results of the energy expenditure are displayed in Table 5. The energy expenditure measurements only started after the METC approval of an amendment for measurements with the Cosmed device during the 6 MWT. Consequently, only the last 16 included patients were measured with Cosmed. Two patients didn’t reach the steady state and the RER value <1 with the Cosmed measurement. The results of these patients were not included in the results. Comparison of the effect of the BoNT-A injection to that of the placebo injection did not show a significant difference.
Energy Expenditure.
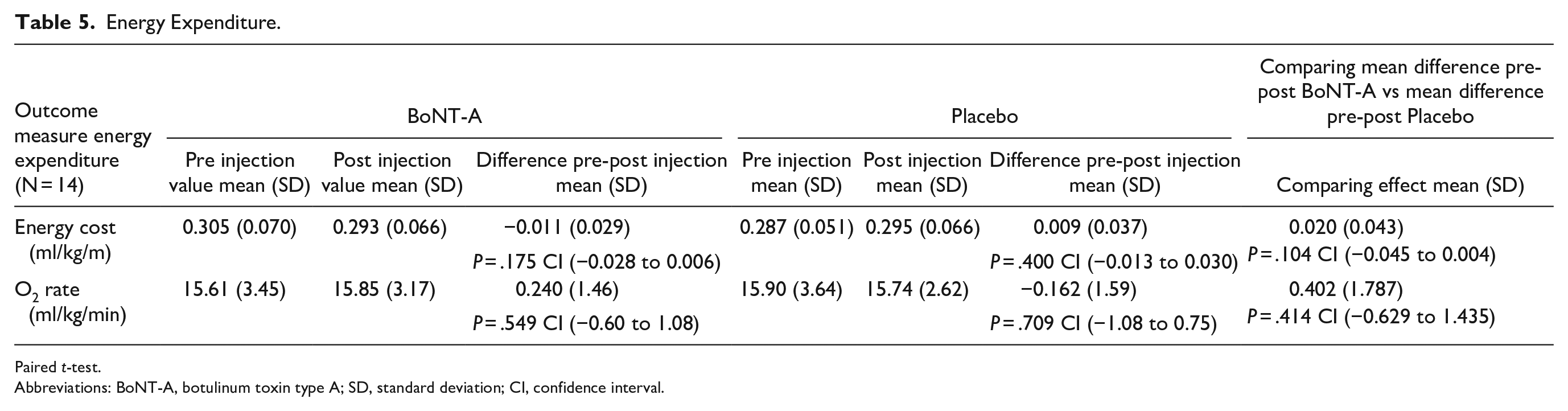
Paired t-test.
Abbreviations: BoNT-A, botulinum toxin type A; SD, standard deviation; CI, confidence interval.
Discussion
In this RCT we investigated the effect of BoNT-A injections in the rectus femoris of stroke patients walking with a SKG. The results of this RCT confirms findings on knee kinematics found in uncontrolled studies. We found in this RCT a significant difference in peak knee flexion (6.7°) which is in line with the 7.37° increase that was found in the systematic review by Tenniglo et al 13 that pooled the data of stroke patients in uncontrolled studies. The reported significant difference in knee range of motion of 4.8° is in line with the significant increase of 6° that was reported by Stoquart et al 12 and the significant increase of 6.83° reported by Tok et al. 22
The significant difference in knee angular velocity at toe off 24.9°/s however is less than the 53.01°/s found in the systematic review. 13 The reason for this difference in outcome remains unclear. Possible differences in the determination of knee angular velocity may have played a part.
Our result showed that there is no decrease in hip kinematics which is in accordance with other studies.17,18 This is an important clinical outcome, because the rectus femoris, being a biarticular muscle, not only stretches the knee but also flexes the hip.9,31 BoNT decreases the function of the rectus femoris, meaning that it decreases knee extension and possibly also hip flexion. This in turn, can increase foot clearance problems which can be a reason for caution in BoNT treatment. Our study, however, does not confirm this risk as results show no adverse effects in hip flexion during walking.
Against our hypothesis, we found that reduction in SKG did not lead to significant improvements in walking speed, energy cost nor scores on the different questionnaires of self-report such as SIS, Rivermead Mobility Index, VAS score, and Borg sore. Also, measurements of selectivity or strength with the Motricity Index or MRC score did not change. Obviously, effects of BoNT-A to reduce SKG does not lead to significant changes in walking speed, energy cost, strength, or patient self-reported outcomes.
We found only a significant difference in walking distance on the 6 MWT, in line with a few uncontrolled studies18,20-22 in this field. However, it should be noted, that found significant difference in walking distance was only 18.3 m and with that not clinically meaningful from the perspective of the patient. For achieving improvements beyond the minimal clinical difference (MCD) on the 6 MWT, Tang et al 32 found a MCD of 34.4 m on the 6 MWT for patients after stroke. Fulk et al 33 found a MCD varying between 44 and 74 m (classified for different walking speeds) on the 6 MWT in post-stroke.
The question is why found improvements in kinematics reflecting SKG and reductions in muscle tone 34 do not generalize to significant improvements in functional outcomes such as walking speed, energy cost, and self-reported outcome. One explanation might be that the effects are too small to impact these functional outcomes. Alternatively, one may also argue the lack of responsiveness of most used functional outcome measures because they reflect different underlying constructs, acknowledging that quality of movement is not captured by these functional outcomes and self-reported outcome.
The current study emphasizes the importance of using kinematic metrics such as peak knee flexion, knee range of motion, next to functional outcomes, in recovery trials to capture these changes. This latter finding is in line with the recommendations seen in the upper extremity in the second Stroke and Recovery and Rehabilitation Round table task force (SRRR) 35 and support the need for global SRRR consensus on balance and mobility outcomes including biomechanics.
The inclusion of rectus femoris overactivity is based on electromyography (EMG) measurement and not, as in some studies,12,19,36 based on the Duncan Ely test. In a recent study 37 we showed that the Duncan Ely test has no predictive value for determining abnormal activity of the rectus femoris during gait in patients after stroke. We therefore recommend not to use the Duncan Ely test to predict abnormal rectus femoris activity during swing, and instead use EMG. That the Duncan Ely test has no predictive value for determining abnormal activity of the rectus femoris may be a reason for a weaker effect on knee kinematics19,36 in the studies that based their inclusion on the Duncan Ely test instead of EMG.17,18
Furthermore, regarding patient inclusion, it is unclear whether there is a relation between specific patient characteristics and treatment effect. This is of importance because we saw that the treatment effect varied from little effect to a good effect. Typical examples of these different treatment effects are graphically displayed in Figure 2. One may hypothesize that individuals with higher levels of selectivity, for instance, respond differently to a BoNT injection when compared to individuals with lower levels of selectivity. Whether relations between patient characteristics and treatment effect exist, and more importantly which patient characteristics are of influence, could be a topic for future research.
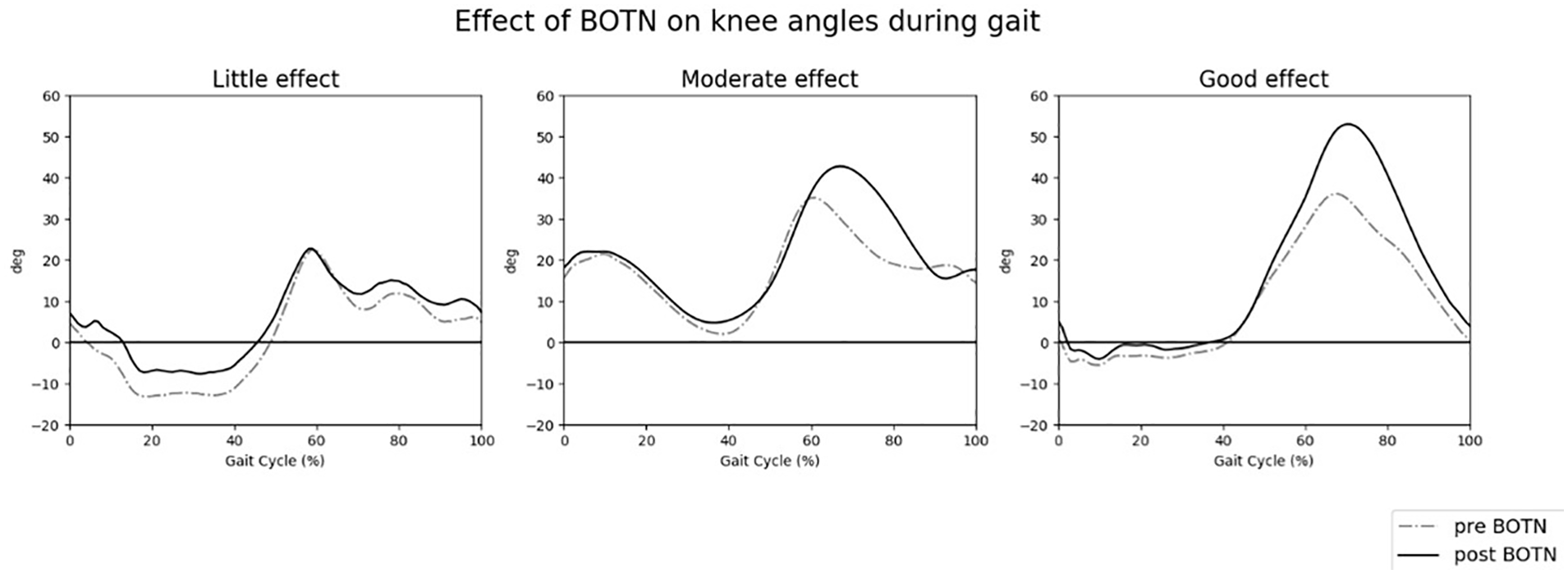
Treatment effect varied from little effect to a good effect.
Strengths and Limitations
The major strengths of the study are the fact that this trial was designed as a triple-blinded cross-over RCT and that we based our inclusion criteria on sEMG which led to an objective and verifiable inclusion of participants with abnormal activity of the rectus femoris.
Another strength of the study was that we chose a primary outcome measure that reflects normalization of the gait pattern which is in line with the anticipated effect of BoNT.
A limitation of the study are the used functional outcome measures as they may not be sensitive or specific enough to measure difference on functional measure abilities after BoNT in the rectus femoris. Furthermore, we did not study the effect of BoNT-A injection in the rectus femoris on functioning in everyday life but focused on performance in a lab environment instead.
Finally, we did not measure the effect of BoNT-A of the rectus femoris on foot clearance problems like stumbling, tripping, or falling in the current trial. Reducing foot clearance problems is an often made remarkable comment of patients after the injection. Reason for the reduced foot clearance problems can be that the increase in knee kinematics normally give an increase in foot/ankle lift from the ground. This can reduce the functional foot clearance problems like stumbling or tripping. However, with the current outcome measures we were not able to assess foot clearance. Patients also mentioned improvements of functional abilities like gait initiation, getting in and out of the car, obstacle avoiding, automatically walk and turning. Unfortunately, all these items are not covered by the functional outcomes of mobility or questionnaires that are available for use in stroke survivors. It is important to use kinematic measurements to demonstrate effects in quality of movement that are not captured by commonly used functional outcome measurements post stroke.
Conclusions
This triple blind RCT showed that knee kinematics significantly improve after BoNT-A injection in the rectus femoris. For functional outcomes, the distance walked over the 6 MWT was significantly increased. Other functional outcomes didn’t improve. BoNT-A injections in the rectus femoris is a valuable treatment option for stroke patients walking with a SKG to improve knee kinematics. To study the effect on functional outcome more research is necessary with different functional outcome measures that can capture the effect in kinematics. This study emphasizes that it is important to use kinematic measurements to demonstrate effects in quality of movement that, otherwise, are not captured by commonly used functional outcome measurements post stroke.
Supplemental Material
sj-doc-1-nnr-10.1177_15459683231189712 – Supplemental material for The Effect of Botulinum Toxin Type A Injection in the Rectus Femoris in Stroke Patients Walking With a Stiff Knee Gait: A Randomized Controlled Trial
Supplemental material, sj-doc-1-nnr-10.1177_15459683231189712 for The Effect of Botulinum Toxin Type A Injection in the Rectus Femoris in Stroke Patients Walking With a Stiff Knee Gait: A Randomized Controlled Trial by Martin J. B. Tenniglo, Anand V. Nene, Johan S. Rietman, Jaap H. Buurke and Erik C. Prinsen in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank Professor J. van der Palen, PhD, for assisting with the statistical analysis and L. Schaake, BSc for his assistance during the data analysis.
Authors’ Note
All authors have read and approved the submitted original research article. No part of this work has been published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
