Abstract
Introduction
Stroke is one of the leading causes of mortality and is a major cause of handicap worldwide. 1 Spasticity, with an occurrence after stroke of between 18% and 38%, may interfere with the execution of daily activities, social participation, and quality of life. 2 In particular, spastic equinovarus of the foot (SEF) is a common pattern of neurological impairment that limits the walking ability of these patients. This deformity is reported in 18% of stroke patients. 3
Although contraction of the tibialis anterior muscle may sometimes be responsible for the varus deformity, SEF is primarily caused by overactivity of the calf muscles (triceps surae, tibialis posterior, flexor hallucis longus muscles), which is often associated with weakness of the antagonist muscles and complicated by musculo-tendinous shortening. Treatments for this condition are multimodal, including medical and surgical strategies in association with neurorehabilitation and orthoses. 4 Diffuse spasticity is usually treated by oral medications and intrathecal baclofen therapy, whereas focal spasticity is treated by chemical (alcohol, phenol, botulinum toxin injections) or surgical (neurotomy) denervation of the overactive muscles. Orthopedic surgery (tendon lengthening and/or transfer) is indicated in the case of fixed muscle contracture and/or to structurally rebalance the alignment of muscles.
Botulinum toxin (BTX) injections induce a selective blockade of the neuromuscular junction, ensuring a reduction in muscular activity. The effectiveness of BTX as an SEF treatment has been demonstrated in randomized, double-blind, placebo-controlled studies.5-7 However, this expensive treatment has a reversible effect that necessitates repeated injections every 4 to 6 months. 8 Tibial nerve neurotomy (TNN) is a neurosurgical procedure consisting of a partial and selective section of the motor nerve branches innervating the spastic muscles. For a moderate cost, it can permanently reduce spasticity. This long-lasting effect is the main advantage of TNN, which is supported by several longitudinal studies. 9
The effectiveness of TNN highly depends on the determination of the spastic muscles involved in the SEF deformity. Diagnostic nerve blocks with anesthetics should be systematically performed before surgery to assist the decision. Using this technique, Decq et al suggested that spasticity of the soleus muscle was predominantly involved in most SEF deformities. 10 Conversely, because of their bi-articular and fusiform properties, gastrocnemius muscles are suspected to have a higher risk of contracture after denervation. 11 This suggests that they should be spared by the procedure in most cases, although it remains a matter of debate.
Recently, a systematic review aimed to evaluate the efficacy of TNN as a treatment of choice for adult patients presenting with SEF. 9 The 11 selected studies, including a total number of 314 patients, suggested that TNN was a safe and effective procedure. However, these studies all involved nonrandomized, uncontrolled case-series studies, designated as level III or level II-3 according to the Preventive Services Task Force. The etiology of SEF was variable in most articles (stroke, traumatic brain injury, spinal cord injury, multiple sclerosis, etc), and the assessment tools used were generally qualitative, subjective, and not validated.
The International Classification of Functioning, Disability and Health (ICF) describes how a disease can affect the body, individual, and social perspectives. This conceptual framework is recommended as a background for reports in clinical research because it provides health professionals with a common language for the description of human functioning. 12 It is divided into 3 domains: body function and structure, activity, and participation. Previous studies focused on the body function and structure domain, whereas activity, participation, and quality of life have poorly been assessed.
The objective of the present study was to demonstrate that TNN was at least as effective as BTX injections in adult chronic stroke patients presenting with SEF, by comparing both treatments in a single-blind, randomized, controlled trial (RCT) based on the evaluation of the 3 ICF domains. Body functions and structures were evaluated by an objective measurement of muscle stiffness, a clinical examination, and an instrumented gait analysis, whereas activity, participation, and quality of life were assessed through validated questionnaires.
Methods
Patient Selection
Between July 2008 and January 2011, stroke patients presenting with SEF were screened in the rehabilitation department of 2 Belgian university hospitals. Before inclusion, candidates for denervation underwent a selective diagnostic nerve block with anesthetics of the motor nerve branch to the soleus muscle. 13 A disposable needle for conduction anesthesia was introduced in the popliteal fossa according to bony landmarks (10 mm above the upper extremity of the fibula and 17 mm lateral to the middle of the popliteal fossa), and precise location of the nerve was determined using electrical stimulations and H-reflex monitoring. A 1-mL dose of 2% Lidocaine was injected when a movement of ankle plantar flexion was seen with a stimulus of 0.01 ms duration and 4 mA intensity. The procedure was occasionally completed by an additional block of the motor nerve branch to the tibialis posterior muscle (anatomic landmarks: 45 mm above the upper extremity of the fibula and 17 mm lateral to the middle of the popliteal fossa), when the ankle movements during gait had not sufficiently been improved after block of the motor branch to the soleus muscle.
This block was systematically performed by the same physician (TD) to determine which spastic muscles were implicated in the SEF.
The inclusion criteria were spastic hemiparesis secondary to stroke (delay ≥ 6 months), SEF due to overactivity of the calf muscles (soleus, tibialis posterior, flexor hallucis longus muscles), improvement of foot positioning during gait after selective motor nerve branches block, a lack of associated contracture (passive ankle dorsal flexion ≥ 0°), and ability to walk on a treadmill (walking speed ≥ 0.14 m s−1) for a sufficient time (~2 minutes) to allow for a gait analysis. The exclusion criteria were a previous history of local surgery, phenol denervation or BTX injection in the past 6 months, and any other pathology that could interfere with gait. Sixteen stroke patients were finally enrolled in the present study. The experimental protocol was approved by the local ethics committee, and each participant signed an informed written consent. The trial was registered at ClinicalTrials.gov, Number NCT00825097.
Randomization and Masking
The patients were randomized (TD) into 2 homogeneous groups (ratio 1:1) by constrained balance randomization (minimization method described by Pocock and Simon 14 ) using 5 criteria: age (<40 years, 40-60 years, >60 years), global neurological impairments as assessed by the Stroke Impairment Assessment Set 15 (<45, 45-60, >60), hemiparetic side, sex, and delay after stroke (<1 year, 1-2 years, 2-5 years, >5 years). Table 1 shows the clinical descriptions of both groups, which were similar at inclusion.
Clinical Description of Both Groups of Patients at Inclusion Time (T0) a .
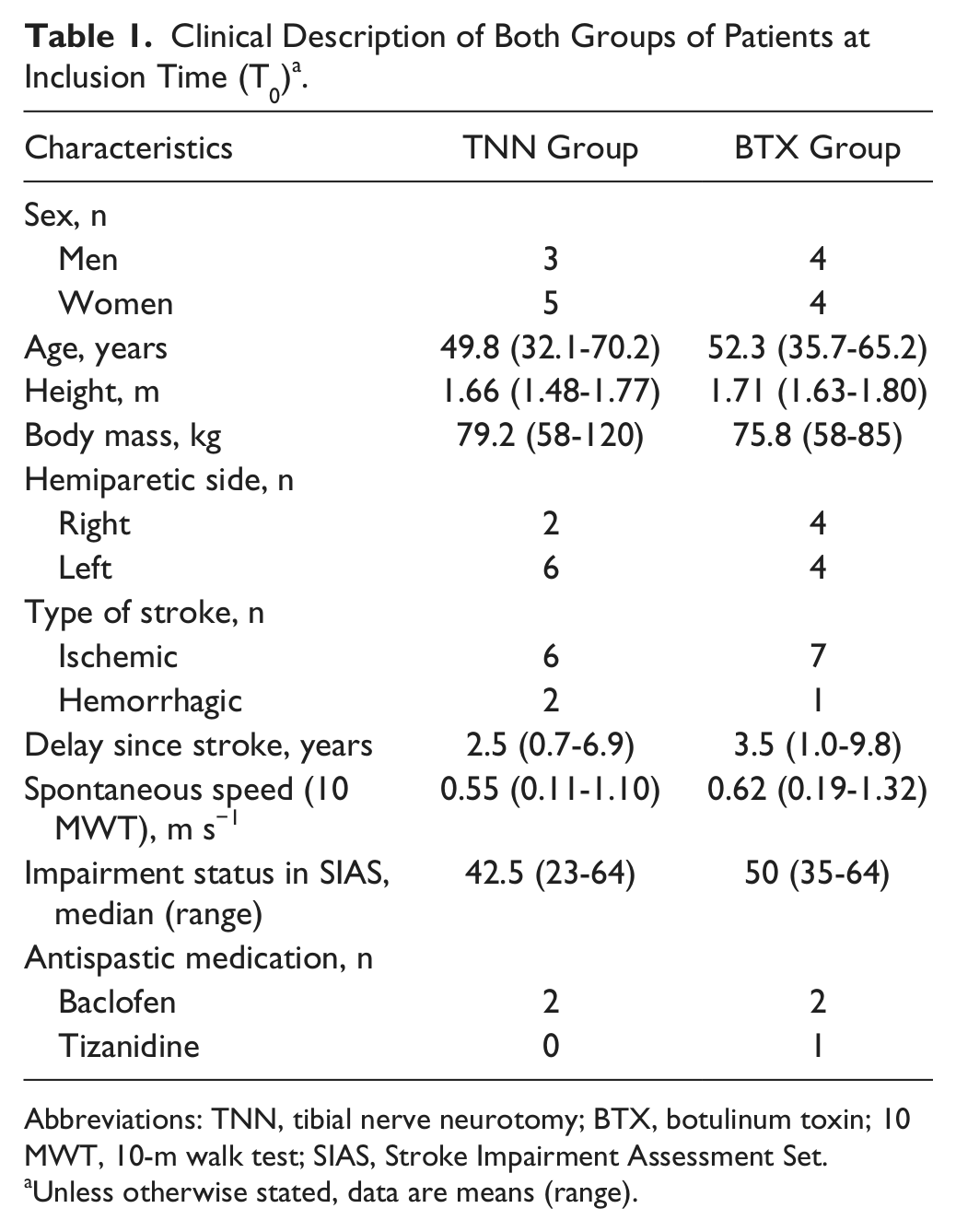
Abbreviations: TNN, tibial nerve neurotomy; BTX, botulinum toxin; 10 MWT, 10-m walk test; SIAS, Stroke Impairment Assessment Set.
Unless otherwise stated, data are means (range).
Treatment
The spastic muscles to treat were identified prior to randomization, according to the result of the motor nerve block. Eight patients (BTX group) were treated by the same physician (TD) with botulinum toxin A (Botox; Allergan, Irvine, CA). The toxin was injected under electrical stimulation guidance using a 100 UI/2 mL dilution. The soleus (200 UI) was injected in all patients, and the tibialis posterior (125 UI) and flexor hallucis longus (75 UI) muscles were treated in 4 patients. Eight other patients were treated with selective tibial neurotomy (TNN group) performed by the same neurosurgeon (TG) according to the method previously described by Deltombe and Gustin. 16 The surgery involved soleus nerve in all cases (mean section ~ 2/3) and the tibialis posterior (mean section ~ 1/2) and flexor hallucis longus (100% section) nerves in 5 cases.
Assessment Following the ICF Framework
The patients were evaluated by the same blinded assessor (BB) before the treatment (T0) and 2 months (T1) and 6 months (T2) after the treatment. A bandage was applied on the popliteal fossa of each subject to make the presence of a scar invisible.
Body Function and Structure
The primary outcome was the quantitative measurement of ankle stiffness, similar to the method described by Lehmann et al. 17 The resistance of the ankle joint to passive sinusoidal displacements of 5° was measured at 10 different oscillation frequencies (3-12 Hz). The torque and displacement signals were decomposed by a Fourier analysis into elastic and viscous stiffness components. The total viscoelastic stiffness was computed from the total path length (L-path) formed over the range of 10 oscillation frequencies, by the apex of the 10 vectors on a viscous versus elastic stiffness plot. This method was developed for quantitatively assessing muscle tone and was demonstrated to be sensitive in objectively evaluating the antispastic effect of a treatment. 18
The clinical examination included spasticity assessment of the soleus muscle (with the knee flexed) and of the triceps surae muscle (with the knee extended) using the Tardieu Scale at fast stretching speed (0-4) 19 and the Modified Ashworth Scale (MAS; 0-4), 20 evaluation of the triceps surae and tibialis anterior muscle strength with the Medical Research Council (MRC; 0-5), 21 and measurement of the passive range of motion (PROM) of the ankle. Global neurological impairments were assessed using the Stroke Impairment Assessment Set (SIAS). 15
The spontaneous walking speed with the usual walking aids was measured using the 10-m walk test (10 MWT).
22
Gait was also assessed using a 3-dimensional analysis, following the protocol described in more detail elsewhere.23,24 Segmental kinematics were recorded by 8 infrared cameras (200 Hz) with the Eliclinic system (BTS, Milan, Italy), while the patients were walking at comfortable speed on a treadmill (Mercury LTMed, HPCosmos, Nussdorf, Germany). The ground reaction forces (GRF) were synchronously recorded (200 Hz) using 4 strain gauges located under the corners of the treadmill (Pharos System Inc, Rochester, NY), and the net joint moments in the sagittal plane were computed from the GRF, kinematic, and anthropometric data. Only data for the ankle and knee joints were analyzed in the present study (see Figure 1). The maximal knee extension during the stance phase (
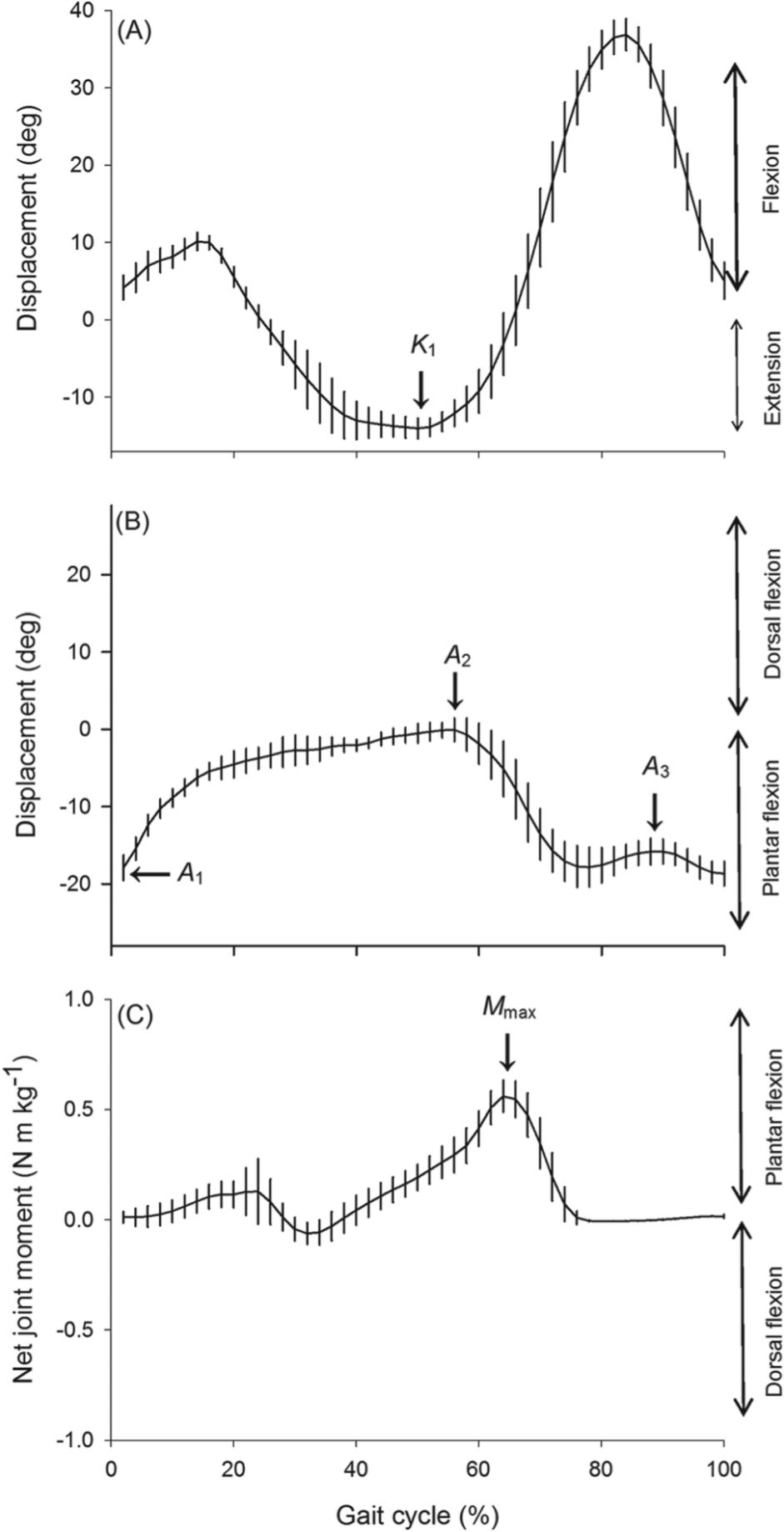
Typical traces of knee kinematics (A), ankle kinematics (B), and ankle kinetics (C) in the sagittal plane in one patient walking at 0.61 m s−1 at inclusion time (T0).
Activity
The walking ability was assessed using 2 standard ordinal scales: the Functional Walking category (FWC; 0-6) 25 and the Functional Ambulation categories (FAC; 0-5). 26 A linear and unidimensional scale validated by Rasch analysis (the 13-item ABILOCO questionnaire) was also used to assess the locomotion ability in stroke patients. 27
Participation and Quality of Life
Satisfaction in participation was assessed using the SATISPART-Stroke, another linear and unidimensional questionnaire validated by Rasch analysis. 28 The general quality of life was evaluated with norm-based scoring (NBS) of the 36-item Short-Form Health Survey. 29
Questionnaires were not presented to patients showing a major cognitive dysfunction as observed with the Mini-Mental State Examination (MMSE ≤ 24), 30 which concerned 1 patient in the TNN group and 3 patients in the BTX group.
Statistics
A 2-way repeated-measures analysis of variance (ANOVA) was computed to study the effect of time and treatment on parametric data. For nonparametric data, a repeated-measures ANOVA on ranks (Friedman) was used to study the effect of time on each treatment group, and a rank sum test (Mann–Whitney) was computed to study the influence of treatment after subtracting the baseline value from those recorded at the subsequent assessments (T1 − T0; T2 − T0). A post hoc test with Bonferroni correction completed the analysis to determine which groups were significantly different. The analysis was performed on an intention to treat basis, and all patients completed the entire follow-up. Sigmastat 3.5 software was used for all statistical analyses.
Results
It was required that medication and physical therapy remained unchanged throughout the follow-up. Five patients were taking antispastic medication (Table 1), and all but 2 (1 patient in each group) received regular physiotherapy.
Body Function and Structure
Ankle Stiffness
Total viscoelastic stiffness of the ankle significantly decreased with time (
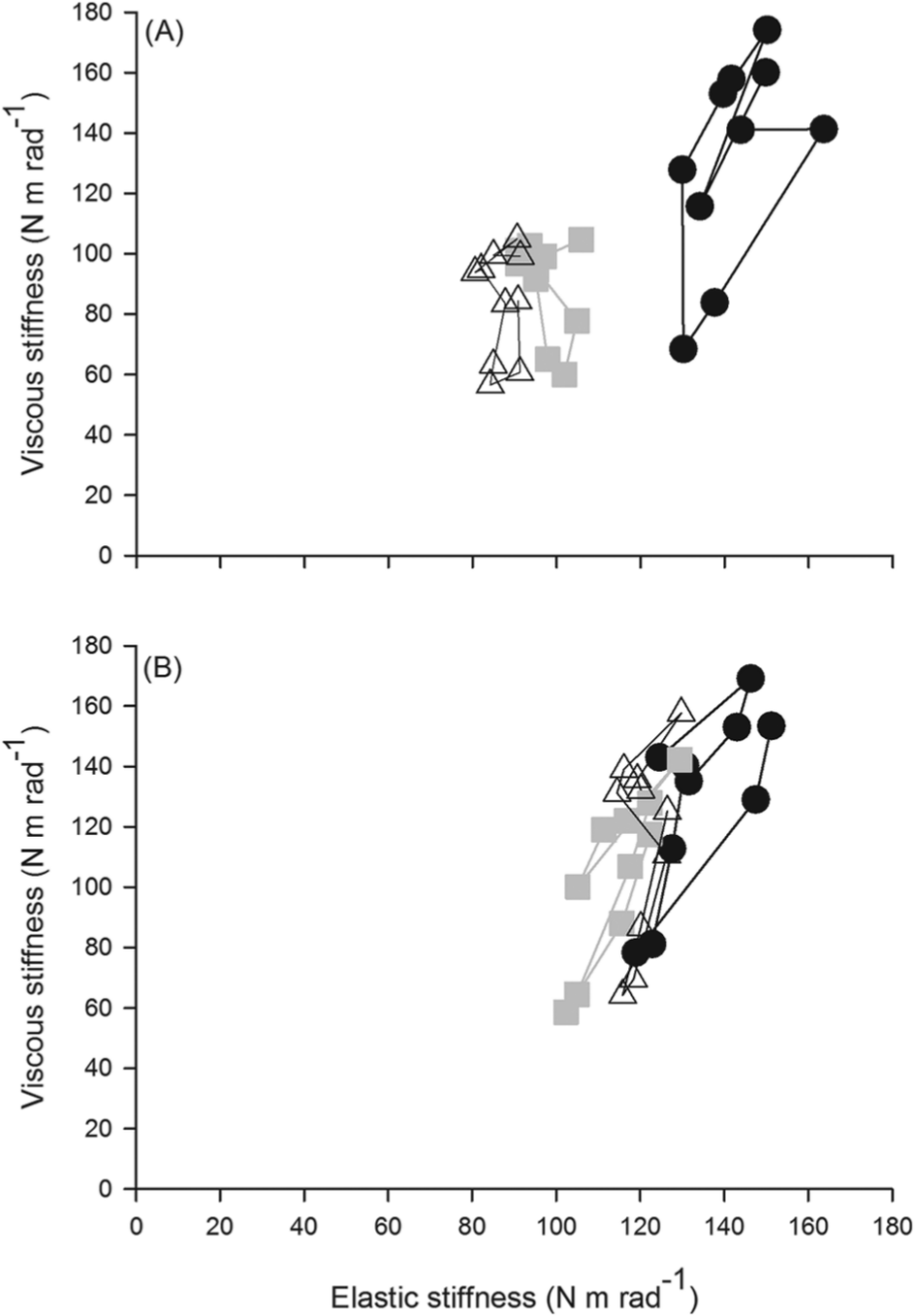
Phase diagrams of the mean viscous stiffness (N m rad−1) versus the mean elastic stiffness (N m rad−1) for the TNN (A) and BTX (B) groups.
Neurological Impairment
The results of the clinical examinations are presented in Table 2. Both treatments induced a decrease in muscle tone based on the MAS and the Tardieu Scale, and this improvement was greater after TNN than after BTX over both the short term (T1 − T0) and the long term (T2 − T0). TNN also induced an improvement in the SIAS that was significant at T1 and T2, whereas BTX had no effect. The PROM and the strength of the triceps surae and tibialis anterior muscles remained unchanged in both groups.
Clinical Assessment of Both Groups of Patients Before Treatment (T0) and 2 Months (T1) and 6 Months (T2) After Treatment a .
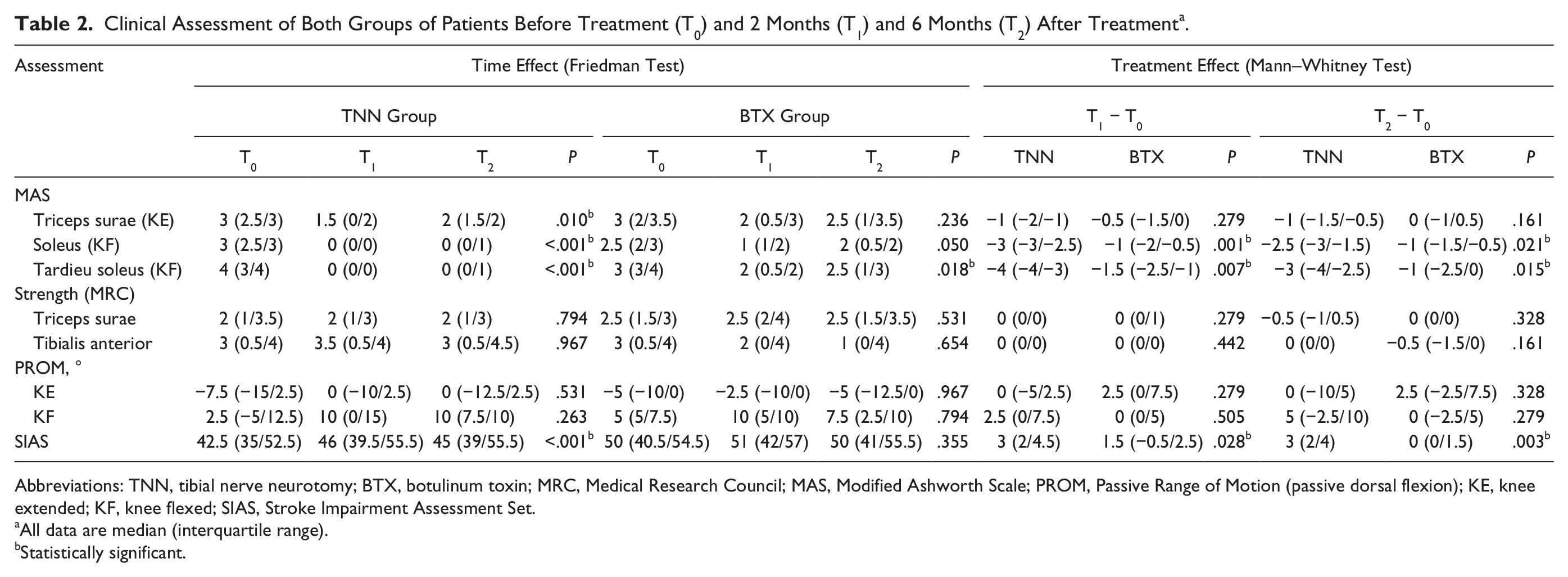
Abbreviations: TNN, tibial nerve neurotomy; BTX, botulinum toxin; MRC, Medical Research Council; MAS, Modified Ashworth Scale; PROM, Passive Range of Motion (passive dorsal flexion); KE, knee extended; KF, knee flexed; SIAS, Stroke Impairment Assessment Set.
All data are median (interquartile range).
Statistically significant.
Gait Analysis
The outcomes of the gait analysis are listed in Table 3. Both treatments induced a progressive improvement in the ankle kinematics with time, and the results were not different between TNN and BTX.
Gait Analysis of Both Groups of Patients Before Treatment (T0) and 2 Months (T1) and 6 Months (T2) After Treatment a .
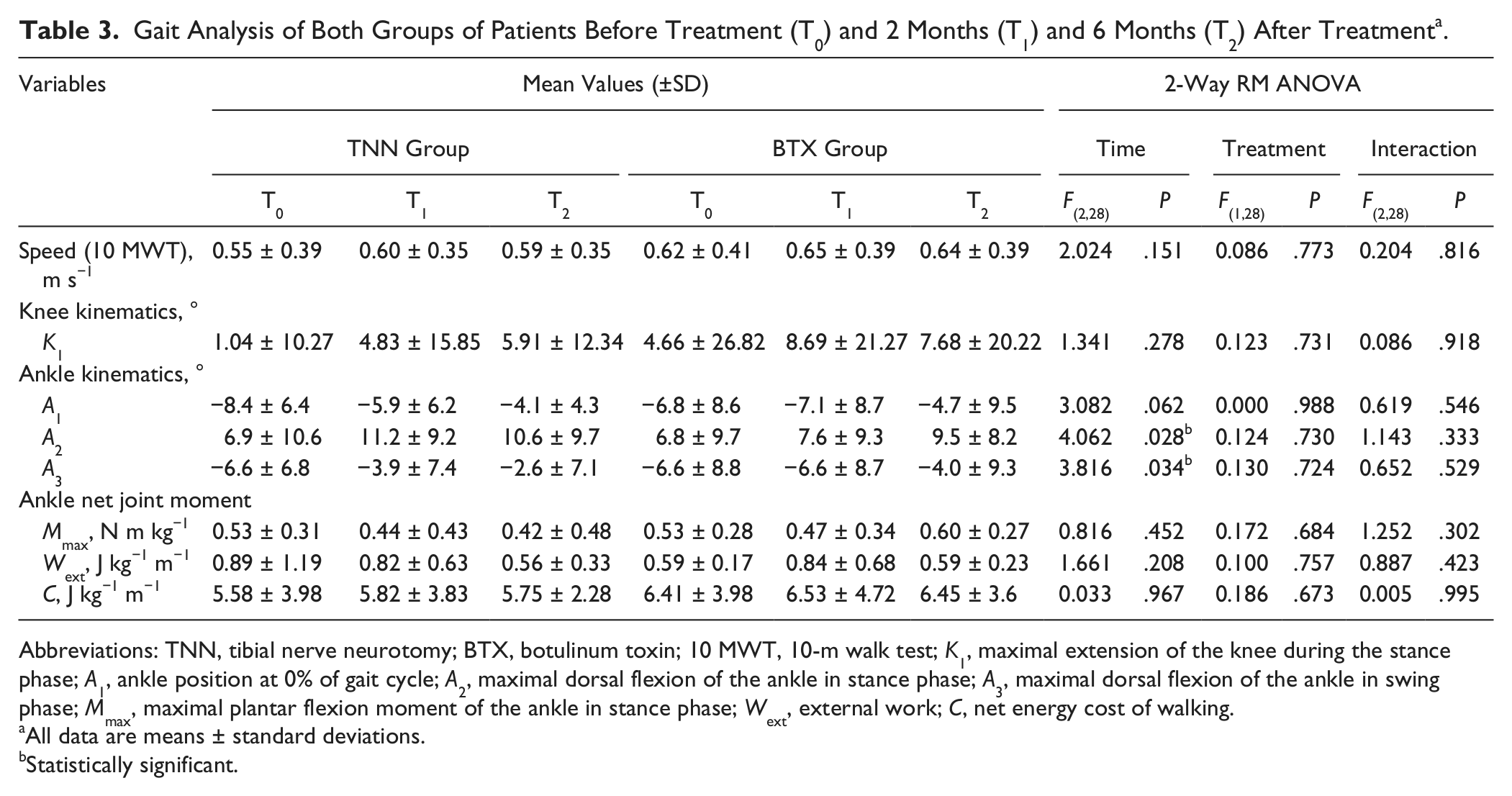
Abbreviations: TNN, tibial nerve neurotomy; BTX, botulinum toxin; 10 MWT, 10-m walk test;
All data are means ± standard deviations.
Statistically significant.
Activity, Participation, and Quality of Life
The results of the FWC (median: 5) and FAC (median: 4) did not change during the follow-up. As presented in Table 4, the treatments had no effect on the activity (ABILOCO) or on the participation satisfaction (SATISPART-Stroke). Similarly, the quality of life (SF-36) remained unchanged at the different evaluations in both groups of patients.
Activity, Participation, and Quality of Life in Both Groups of Patients Before Treatment (T0) and 2 Months (T1) and 6 Months (T2) After Treatment a .
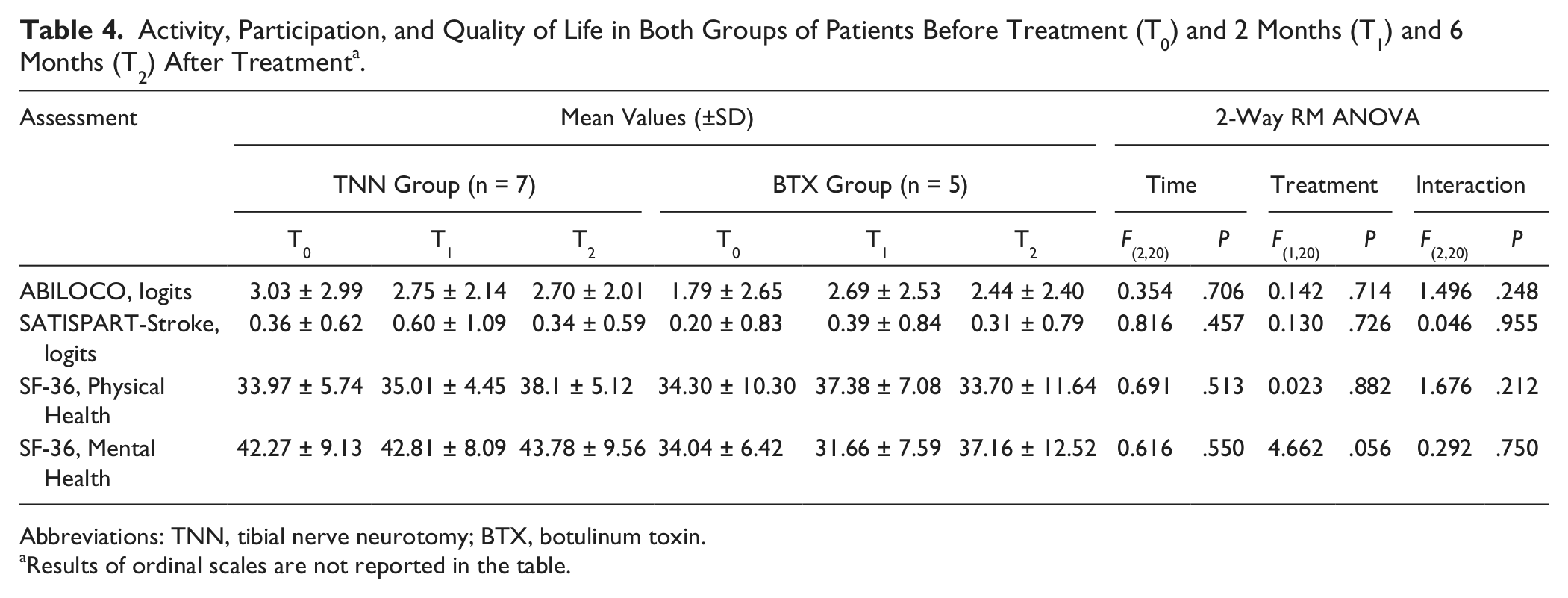
Abbreviations: TNN, tibial nerve neurotomy; BTX, botulinum toxin.
Results of ordinal scales are not reported in the table.
Adverse Events
Early postoperative complications were observed in 3 patients in the TNN group, including 1 patient with a healing delay, 1 patient with a transient calf edema, and 1 patient with a discrete but permanent hypoesthesia without neuropathic pain around the scar. No complications were observed in the BTX group.
Discussion
The present study is the first single-blind RCT to assess the effects of TNN as a treatment for SEF, and it used quantitative, validated, and objective assessment tools based on the 3 ICF domains. The results confirm with evidence that TNN is an effective treatment to reduce the impairments in stroke patients presenting with SEF, which was already suggested in previous case-series studies. 9
This study shows that TNN and BTX induce a decrease in spasticity, with results similar to previous publications.5-7,9,31 In the BTX group, the decrease is significant with respect to the Tardieu scale and close to statistical significance when measured by the MAS. In the TNN group, spasticity is almost abolished in the soleus muscle using both scales. This study is the first to demonstrate the superiority of TNN compared with BTX to reduce muscle tone, which was suspected in a previous crossover study. 31 These findings are confirmed by the measurement of the L-path, which was chosen as the primary outcome to objectively assess muscle tone.
The present study shows that the effects of TNN and BTX are similar on most gait variables. Because the duration of the BTX effect was reported to be variable in the treatment of spasticity, ranging from a few weeks to several months, 8 BTX injections could be used as a long-lasting block to perform prior to TNN, to predict the functional improvement expectable with the surgery. Conversely, this result also suggests that this surgical procedure could be a first-line treatment for properly selected patients.
Due to an unavoidable section of efferent motor fibers, muscle weakening was described as a transient adverse event of TNN.
9
In the present study, the strength of the triceps surae muscle was not altered by any treatment, although this muscle was already weak in most patients at inclusion (Table 1).
The instrumented gait analysis demonstrated that angular displacements of the ankle in the sagittal plane progressively and significantly improved after TNN. This result was similar after BTX, which was considered the reference treatment in the present study. Using gold-standard methods of assessment, our results confirm the results suggested in previous studies.
9
Conversely,
Optimizing the execution of daily activities and involvement in life situations is a major objective for the rehabilitation of patients presenting with chronic disorders. Activity, satisfaction in participation, and quality of life are latent variables that are difficult to measure accurately. Due to our inclusion criteria, the walking ability of the participants recruited in the present study is not strictly representative of the global stroke population. The results would likely be different for patients presenting with a lower level of autonomy at baseline. Moreover, many factors may interfere with the achievement of daily activities in hemiparetic patients. The disability may be more related to negative signs of the upper motor neuron syndrome (paresis, loss of dexterity) than to positive signs (spasticity, abnormal postures). TNN is a focal procedure, only aiming to reduce overactivity of the dorsal leg muscles. Moreover, the relatively short duration of the study may have been insufficient for the patients to increase their level of activity regarding their improved gait pattern. The concomitant use of rehabilitation was allowed, but the content and frequency of the sessions remained unchanged throughout the present study. An intensive rehabilitation program focused on specific activities could provide a synergic effect with TNN and result in the long-term expansion of the range of patients’ abilities.
The number of subjects included in each group could be considered a limit of the present study. This was particularly true regarding the results of the questionnaires, for which the number of participants was further reduced. However, the study size did not prevent statistical significance from being reached for several parameters.
BTX is a reversible treatment of SEF that must regularly be readministered to the patients. Moreover, the frequency of injections and the doses are restricted in each patient to avoid adverse events. TNN is described as an alternative treatment, whose efficacy in inducing long-term relief of dorsal leg muscles spasticity is supported by several nonrandomized and uncontrolled studies. 9 In the present study, the follow-up was limited to 6 months. This delay corresponds to the classical duration of BTX action, and the long-term evaluation of TNN efficacy was beyond the scope of the study. Further studies should be conducted on a larger population to demonstrate the long-term effectiveness of TNN, since it constitutes the main advantage of one medical intervention over the other. Such studies should combine TNN with a designed rehabilitation program to assess the long-term efficacy of this treatment on the 3 ICF domains.
The present protocol could also be improved by the systematic evaluation of pain, by the use of a multisegmental foot and ankle motion analysis that would allow a better assessment of varus deformity, and by the evaluation of specific goal attainment determined by the patient prior to the treatment. The exact percentage of motor nerve section should be determined with precision in the future to improve the surgical procedure, and the necessity of treating gastrocnemius muscles should also be investigated to obtain better results in some patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Benjamin Bollens is a Clinicien-chercheur doctorant of the Belgian National Fund For Scientific Research (FNRS). This work was also supported by grants from the FNRS and the Horlait-Dapsens Fund.
