Abstract
Background
Recovery of quiet standing balance early poststroke has been poorly investigated using repeated measurements.
Objective
To investigate (1) the time course of steady-state balance in terms of postural stability and inter-limb symmetry, and (2) longitudinal associations with lower limb motor recovery in the first 3 months poststroke.
Methods
Forty-eight hemiparetic subjects (age: 58.9 ± 16.1 years) were evaluated at weeks 3, 5, 8, and 12 poststroke. Motor impairments concerned the Fugl-Meyer assessment (FM-LE) and Motricity Index total score (MI-LE) or ankle item separately (MI-ankle). Postural stability during quiet 2-legged stance was calculated as the net center-of-pressure area (COPArea) and direction-dependent velocities (COPVel-ML and COPVel-AP). Dynamic control asymmetry (DCA) and weight-bearing asymmetry (WBA) estimated inter-limb symmetries in balance control and loading. Linear mixed models determined (1) time-dependent change and (2) the between- and within-subject associations between motor impairments and postural stability or inter-limb symmetry.
Results
Time-dependent improvements were significant for FM-LE, MI-LE, MI-ankle, COPArea, COPVel-ML, and COPVel-AP, and tended to plateau by week 8. DCA and WBA did not exhibit significant change. Between-subject analyses yielded significant regression coefficients for FM-LE, MI-LE, and MI-ankle scores with COPArea, COPVel-ML, and COPVel-AP up until week 8, and with WBA until week 12. Within-subject regression coefficients of motor recovery with change in COPArea, COPVel-ML, COPVel-AP, DCA, or WBA were generally non-significant.
Conclusions
Postural stability improved significantly in the first 8 weeks poststroke, independent of lower limb motor recovery at the most affected side within subjects. Our findings suggest that subjects preferred to compensate with their less affected side, making metrics reflecting inter-limb asymmetries in balance invariant for change early poststroke.
Introduction
Regaining steady-state balance during quiet standing is mainly achieved within the first 3 months poststroke1,2 and is a prerequisite for accomplishing independent gait and most activities of daily life.2-4 Despite its clinical importance, a limited number of observational studies have investigated how lower limb motor recovery associates longitudinally with steady-state balance improvements within this time window.
A few longitudinal studies5-9 have suggested that lower limb motor recovery follows a proportional and predictable time course in the first 3 to 6 months poststroke. This includes clinical improvements in synergistic independent motor control,5,7-9 as measured with the Fugl-Meyer lower extremity motor score (FM-LE), and strength,6,7 as measured for example with the Motricity Index (MI-LE). These findings corroborate observations of the upper limb5,7,10 as significant improvements occur in most patients up until week 55,6 to 8 7 poststroke, and a small proportion (10-15%) fail to show any motor recovery. 8
At the same time, steady-state balance control remains deficient after independent stance is regained, with stroke patients exhibiting greater postural sway of the net center-of-pressure (COP) than healthy controls and loading more body weight on the less affected leg.11-13 More recent posturographic studies14-16 examined the individual-limb COP trajectories to show that this weight-bearing asymmetry (WBA) is further characterized by an asymmetric exertion of stabilizing ankle torques. This so-called dynamic control asymmetry (DCA) reflects the most affected leg’s contribution to balance control in the sagittal plane, relative to the less affected side.14,16 It has been suggested that the DCA is associated with impairment severity, 17 although Roelofs et al 16 have recently shown that even patients with (almost) complete FM-LE recovery may still exhibit significant balance control asymmetries favoring the less affected leg. How this relationship develops within subjects over the first weeks after stroke is currently unclear.
To investigate the quality of movement regarding steady-state balance poststroke, the literature15,17 recommends complementing conventional instability measures, such as the net COP sway area (COPArea)12,13,17 and velocities in frontal (COPVel-ML) and sagittal planes (COPVel-AP),14,16 with metrics that reflect asymmetries, such as DCA and WBA. These metrics may yield different, yet complementary information about how an improved postural stability is achieved in patients with hemiparesis, by distinguishing “normalization” of inter-limb symmetry from persistent compensatory stabilization through the less affected leg, in reference to a control population of healthy adults. So far, very few attempts have been made to implement such metrics in stroke recovery studies11,18,19 and an earlier study by de Haart et al 14 investigated recovery using repeated measurements at arbitrary time-points, often beyond the period in which the recovery of muscle synergies and strength plateaus. According to this knowledge gap, the overall aim of the present observational study was to prospectively investigate the time course of quiet standing balance in terms of posture stabilization and recovery from inter-limb asymmetries early after stroke onset. Subsequently, we aimed to relate these fine-grained task performance changes to motor recovery at the level of the entire lower limb (ie, FM-LE and MI-LE) and ankle separately (by using the dorsiflexion item of the Motricity Index [MI-ankle]), considering that steady-state balance is mainly controlled through ankle torques. 20 The following research questions (RQs) were addressed:
RQ1: What is the time course of muscle synergies (ie, FM-LE) and strength (ie, MI-LE and MI-ankle) in the most affected leg within the first 3 months poststroke?
RQ2: What is the time course of postural stability (ie, COPArea, COPVel-ML, and COPVel-AP) and inter-limb symmetry (ie, DCA and WBA) during quiet stance within the first 3 months poststroke?
RQ3: How is the severity of motor impairments (ie, FM-LE, MI-LE, and MI-ankle) associated with postural instability (ie, COPArea, COPVel-ML, and COPVel-AP) and inter-limb asymmetry (ie, DCA and WBA) during quiet stance between subjects within the first 3 months poststroke?
RQ4: How are improvements in motor impairments (ie, FM-LE, MI-LE, and MI-ankle) associated with decrease in postural instability (ie, COPArea, COPVel-ML, and COPVel-AP) and inter-limb asymmetry (ie, DCA and WBA) during quiet stance within subjects over the first 3 months poststroke?
In line with recovery models of the paretic upper limb, 10 we hypothesized for RQ1 that significant time-dependent change in FM-LE, MI-LE, and MI-ankle would occur within the first 8 weeks poststroke. For RQ2, we hypothesized that steady-state balance would parallel motor recovery and follow the same pattern as previously described for upper limb motor performance.21,22 Recovery of steady-state balance is here defined as posture stabilization reflected by decreases in COPArea, COPVel-ML, and COPVel-AP. Concomitant reductions in asymmetries in DCA and WBA in the direction of norm values in age-matched healthy controls are seen as an indicator of an improved quality of movement. For RQ3, we assumed that patients with lower FM-LE, MI-LE, and MI-ankle scores would exhibit greater postural instability (ie, COPArea, COPVel-ML, and COPVel-AP) and asymmetries in DCA and WBA, with an increased involvement of the less affected leg. Lastly, we hypothesized concerning RQ4 that the within-subject associations between recovery of impairments and steady-state balance would be time-dependent. That is, rising FM-LE, MI-LE, and MI-ankle scores would associate with reductions in postural instability (ie, COPArea, COPVel-ML, and COPVel-AP) and asymmetries in DCA and WBA mainly within the first 8 weeks poststroke.
Methods
The present longitudinal study is part of the larger TARGEt research project. TARGEt is an acronym for
Participants
Patients admitted to one of the 3 cooperating hospitals and 2 rehabilitation facilities (Antwerp region, BE) after an acute ischemic or hemorrhagic stroke were screened for participation between December 2018 and December 2021. Screening and recruitment were performed by the study coordinator (JS) together with the medical doctors and physiotherapists employed at the stroke units and rehabilitation facilities. All participants met the following inclusion criteria: (1) having experienced a first-ever hemispheric stroke confirmed by CT and/or MRI scan; (2) having been included within the first 3 weeks after stroke; (3) having reduced leg strength, defined as >0 points on item 6a/b of the NIHSS (ie, at least “drift within 5 seconds”) within 72 hours poststroke and an MI-LE score <91 (ie, at least “movement against resistance but weaker” for 1 item) at inclusion; (4) age between 18 and 90 years; (5) premorbid independence in daily life activities (ie, modified Rankin Scale score of 0-1); (6) no severe orthopedic condition of the lower limbs and trunk or another neurological illness present before stroke; (7) no severe cognitive or communicative deficit that may interfere with understanding instructions and study procedures; and (8) providing written informed consent. These criteria were chosen to recruit a cohort of initially hemiplegic patients with some residual motor impairment who require inpatient rehabilitation care.
Additionally, we recruited age- and sex-matched adult subjects without reported history of neurological and/or orthopedic conditions to obtain healthy reference values of inter-limb symmetry while standing.
Procedures
In line with recommendations from the Stroke Recovery and Rehabilitation Roundtable (SRRR),25,26 serial measurements were scheduled for each participant at weeks 3, 5, 8, and 12 poststroke. At each time-point, clinical scales were complemented by posturographic measurements of steady-state balance. Two trained assessors (EE and JS) were available to administer clinical scales during face-to-face sessions, while the same observer conducted all serial measurements of individual participants. Posturography was performed by a single assessor (JS) who was trained in operating the measuring instruments. The same measurements were performed once in healthy controls for comparison.
Clinical Measurements
During intake, subjects’ sex, age, stroke type (ie, ischemic or hemorrhagic) and most affected body side (ie, left or right) were recorded. Serial follow-up measurements included, first, the “standing unsupported” item of the Berg Balance Scale (BBS-s) to determine if subjects were eligible for posturography. Second, impairments in synergistic depended motor control and strength were evaluated at the most affected side using the FM-LE 5 and MI-LE, 27 respectively. Synergy was defined as a pathological pattern of muscle co-activation occurring with voluntary movement, referring to the clinical phenomenon of “abnormal muscle synergies”.28,29 The FM-LE (0-34) is valid and highly reliable, 30 and we used a standardization method developed by See et al. 31 The MI-LE (0-99) was administered by asking subjects to produce a maximum voluntary hip flexion, knee extension, and ankle dorsiflexion against resistance. The MI-LE is valid and reliable. 27 We treated the MI-ankle as a separate outcome variable.
Posturographic Measurements
The current study investigated steady-state balance defined by Shumway-Cook and Woollacott 32 as “the ability to control the body’s center-of-mass (COM) relative to the base of support in fairly predictable conditions and non-changing environments.” Accordingly, subjects were instructed to stand quietly on both legs for 40 seconds with their arms alongside their trunk and their eyes fixated on a non-moving visual target. The bare feet were positioned side-by-side in a standardized way (8.4 cm heel-to-heel distance and 9° toe-out angle) and subjects were asked to stand still without further instructions regarding weight-bearing. Measurements started as soon as patients could stand (ie, BBS-s >0) and, if tolerated, 3 trials were performed with seated resting breaks in between. The first 10 seconds were removed from each trial.
We used either 2 laboratory-grade force plates (Type OR6-7 Biomechanics Force Platform, AMTI, MA, USA) or a portable plantar pressure plate (0.5 m Footscan pressure plate 3D, RS Scan/Materialize, BE) to record ground reaction forces in- or outside the lab environment. The collected raw force data was converted to the net and individual-limb COP trajectories (Supplemental Appendix B, force data acquisition and COP calculations) which were low-pass filtered with a 10 Hz second-order Butterworth filter. Comparability of the 2 instruments for measuring COP was assessed in advance in healthy controls during vision-deprived stance, yielding high consistency according to Pearson correlation coefficients, yet significant mean differences (Supplemental Appendix A, comparability analyses). To account for these systematic differences, serial within-subject measures were always performed with the same type of plate, while between-subject variations explained by the choice of measurement instrument were corrected by entering INSTRUMENT as an additional covariate in the final analyses (Supplemental Appendix A, correction method).
To align the individual-limb COP with the anatomical ankle position, the coordinate system was rotated. As subjects may experience difficulties with maintaining the standardized position, the actual feet orientation was determined trial-by-trial with an optoelectronic device (Vicon Motion Systems Ltd, UK) during force plate measurements, or by the plantar pressure distribution (Footscan, RS Scan/Materialize, BE). The AP axis was defined by a line drawn between the head of the second metatarsal bone and the heel, and the ML axis perpendicular to it.
Performance Measures of Steady-State Balance
To quantify postural stability, we first calculated the COPArea by fitting an ellipse in mm2 that encloses about 85% of the entire signal, using principal component analysis. 33 This metric served as a general stability index by estimating the total amount of postural sway. Second, the root mean square of the AP- and ML-COP velocities (COPVel-ML, COPVel-AP; in mm/s) served as estimates of the global balance control efficacy in specific sway directions.14,16
Quality of movement was operationally defined by comparing stroke subjects’ task performance directly with that of healthy controls. 34 That means, the better they were able to achieve postural stability with equal contributions by both limbs, the higher their movement quality. 23 To estimate how the stabilizing mechanism of ankle torques in each leg contributed to balance control, we calculated the DCA in percentage as a symmetry index of the individual-limb COPVel-AP.14,16 It is restricted to the sagittal plane, since ankle torques are less relevant to frontal plane balance. 20 A score of 0% estimates symmetry. Positive and negative values reflect greater contribution of the less and most affected leg, respectively. WBA was calculated by dividing the average vertical force component (ie, FZ) below the most affected leg by the total FZ (ie, body weight), to establish a subject’s preferred stance. A value of 50% was distracted from WBA, such that 0% means symmetry comparable to DCA. All posturographic metrics were averaged over 3 (or at least 2) successive trials per session to maximize reliability of study outcomes. 35
Statistical Analyses
To investigate time courses (RQ1 and and RQ2), we first plotted individual time-series of the outcome variables FM-LE, MI-LE, MI-ankle, COPArea, COPVel-ML, COPVel-AP, DCA, and WBA, to observe trends in recovery. Next, for each outcome variable, a multivariable linear mixed model was applied, treating the main fixed effect, that of TIME (week 3, week 5, week 8, and week 12), as a categorical predictor variable reflecting progress of time after stroke onset. AGE (years), SEX (female, male), AFFECTED SIDE (left, right), and INSTRUMENT (force plates, pressure plate) were included as covariates. A random intercept per subject was added to account for dependency between repeated measurements. Post-hoc analyses involved Tukey’s HSD multiple comparison method, yielding regression coefficients (β) for time-dependent change over the entire period (ie, weeks 3–12) and across each epoch (ie, weeks 3−5, weeks 5−8, and weeks 8−12). DCA and WBA values were statistically compared between stroke and healthy subjects at each time-point using the non-parametric Steel’s test for multiple pair-wise comparisons, with the healthy values treated as control. The significance level was set at <.05.
RQ3 was addressed using linear mixed models, with COPArea, COPVel-ML, COPVel-AP, DCA, or WBA as the dependent variable, and either FM-LE or MI-LE as the independent variable. TIME, AGE, SEX, AFFECTED SIDE, and INSTRUMENT were added as covariates with a subject-specific intercept. Sub-analyses included 4 separate models at weeks 3, 5, 8, and 12. For RQ4, the within-subject associations were calculated using the same model architecture but using change scores (ie, Δ) with sub-analyses across the 3 different epochs. Regarding RQ3 and RQ4, the final regression coefficient (β) predicts change in COPArea, COPVel-ML, COPVel-AP, DCA, or WBA for a 1-unit increase in either FM-LE or MI-LE. Multiple testing was accounted for by using Bonferroni-corrected probability values (ie, P < .05/n).
All models were fitted using JMP Pro (version 16). Histograms and Q–Q plots of residuals were inspected to check model assumptions.
Results
Figure 1 shows a flow chart of the recruitment of subjects and serial measurements. Approximately 250 first-ever stroke survivors were screened during the recruitment period, of which 66 patients were enrolled for this cohort study. Forty-eight of these subjects participated in at least 2 posturographic measurements and were subsequently included in the analyses. Table 1 shows their main baseline characteristics at 3 weeks poststroke. As shown, the included subjects had a mean (SD) age of 58.9 (16.1) years, 19 were female, 36 had suffered an ischemic stroke, and 25 had left-sided impairments. Ten healthy control subjects were additionally included with a similar mean age of 46.9 (14.1) years and sex ratio (40% female).
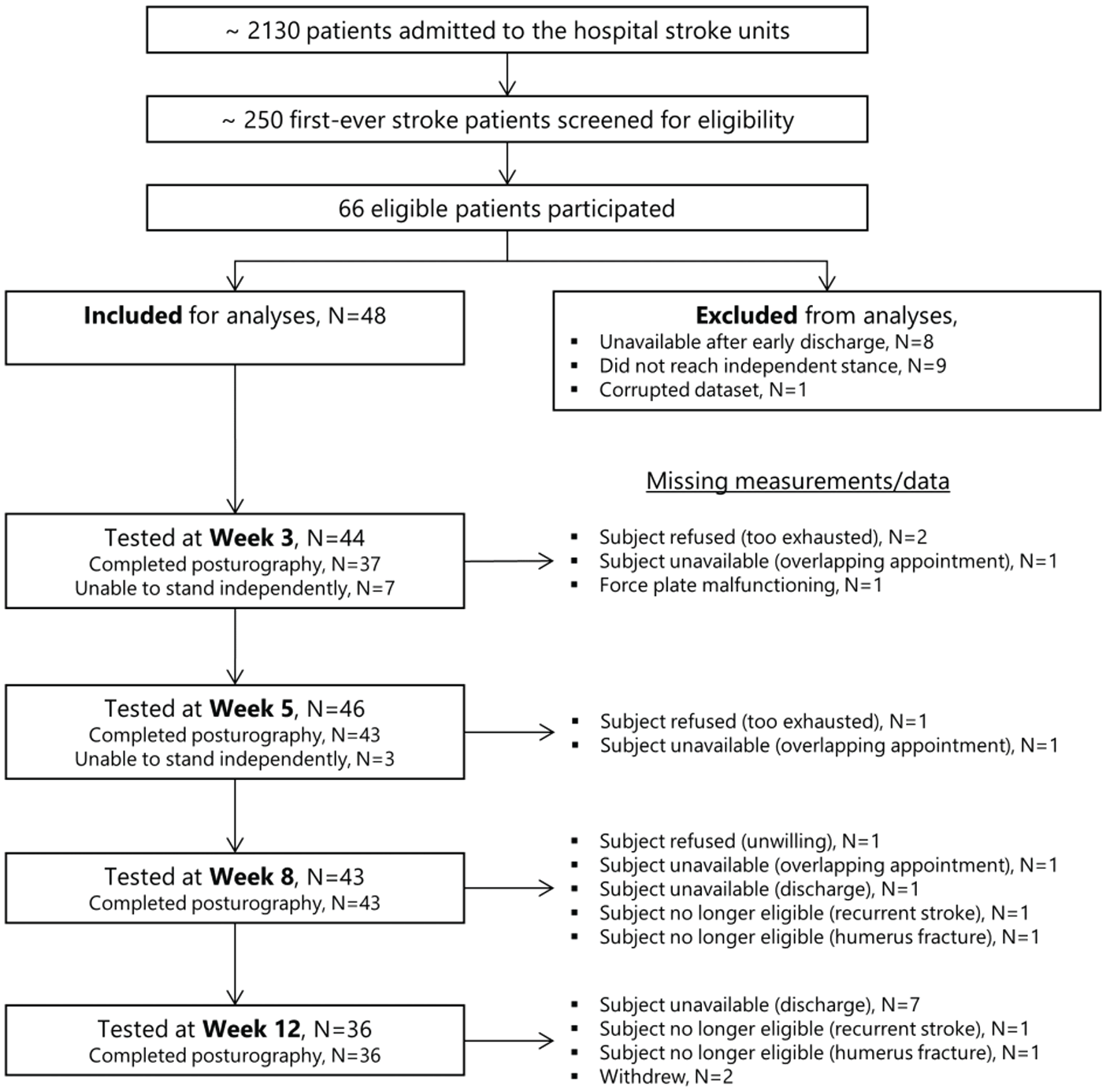
Flowchart of screening, inclusion, and follow-up.
Subject Characteristics at Baseline (ie, 3 Weeks Poststroke).

Abbreviations: FM-LE, Fugl-Meyer lower extremity motor score; MI-LE, Motricity Index lower extremity score; BBS-s, standing unsupported item of the Berg Balance Scale; COP, center-of-pressure; COPArea, area of the net COP; COPVel-ML, rms velocity of the net COP in the frontal plane; COPVel-AP, rms velocity of the total COP in the sagittal plane; DCA, dynamic control asymmetry; WBA, weight-bearing asymmetry.
Values are means ± SD if marked (*), otherwise counts are shown. Demographics and stroke information was collected from all included subjects (N = 48) at enrollment. Clinical characteristics were obtained in 44 subjects that could be tested at week 3, of which 37 could stand independently to perform the standardized balance task. Their posturographic baseline characteristics are shown as well (N = 37).
As summarized in Figure 1, 4 measurements were missed at week 3. Out of the 44 subjects that could be tested, 37 were able to stand independently and participated in the posturographic measurement. At week 5, 2 measurements were missing and 3 subjects had too poor balance to perform the posturographic task. At week 8 and 12, 5 and 12 measurements were missed, respectively. The main reason was unavailability after hospital discharge. As a result, 24 participants could be tested at all 4 occasions. Fifteen and 9 subjects participated in 3 and 2 serial measurements, respectively. The mean time after stroke onset (SD, range) and the number of participants whose data was available at each time-point were as follows: 24.9 (1.8, 22-28) days and N = 37 for week 3; 38.6 (2.1, 35-42) days and N = 43 for week 5; 59.2 (2.2, 55-63) days and N = 43 for week 8; and 88.2 (3.7, 84-103) days and N = 36 for week 12.
Effects of Time on Recovery of Lower Limb Muscle Synergies and Strength
Panel A of Figure 2 depicts individual and mean time-dependent change in FM-LE, MI-LE, and MI-ankle. TIME was a significant factor (P < .001) affecting recovery of FM-LE (β = 3.84, 95% CI [2.58; 5.11], P < .001), MI-LE (β = 12.37, 95% CI [7.77; 16.97], P < .001), and MI-ankle (β = 4.99, 95% CI [2.92; 7.05], P < .001) from week 3 to 12. As further shown in Table 2, significant time-dependent change was found between weeks 3 and 5 for FM-LE (β = 1.66, 95% CI [0.50; 2.82], P = .002), MI-LE (β = 5.63, 95% CI [1.43; 9.84], P = .004), and MI-ankle (β = 2.83, 95% CI [0.92; 4.71], P < .001). A significant increase was also seen for FM-LE between weeks 5 and 8 (β = 1.49, 95% CI [0.36; 2.61], P = .004), whereas a non-significant change was found in MI-LE and MI-ankle scores (P > .05, Table 2). TIME was not a significant factor from week 8 onwards.
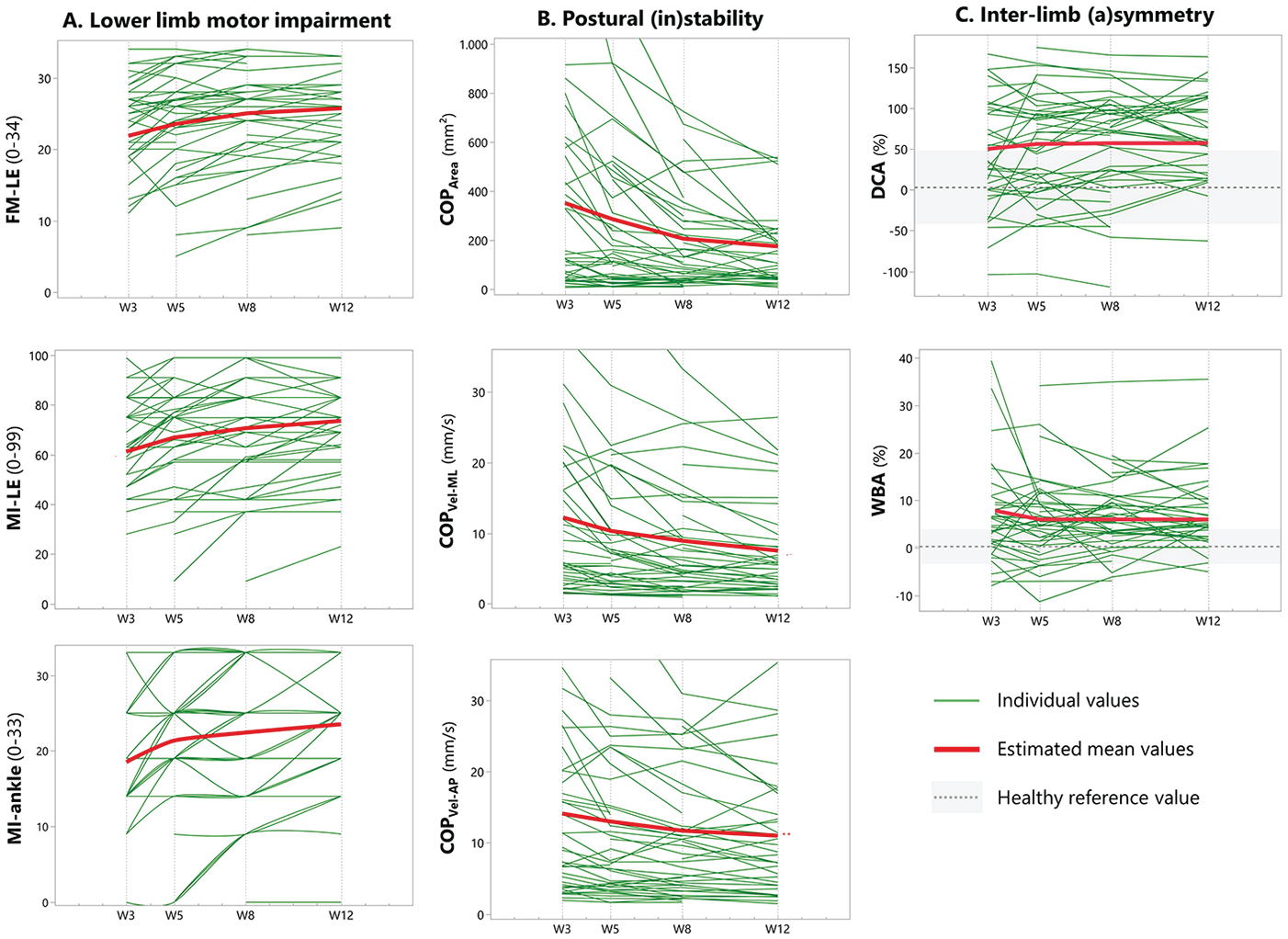
(A-C) Time-courses of muscle synergies and strength, and metrics reflecting steady-state balance during quiet stance between weeks 3 and 12 poststroke.
Effects of Time on Recovery of Muscle Synergies and Strength, and Metrics Reflecting Steady-state Balance During Quiet Stance Within the First 12 weeks Poststroke.
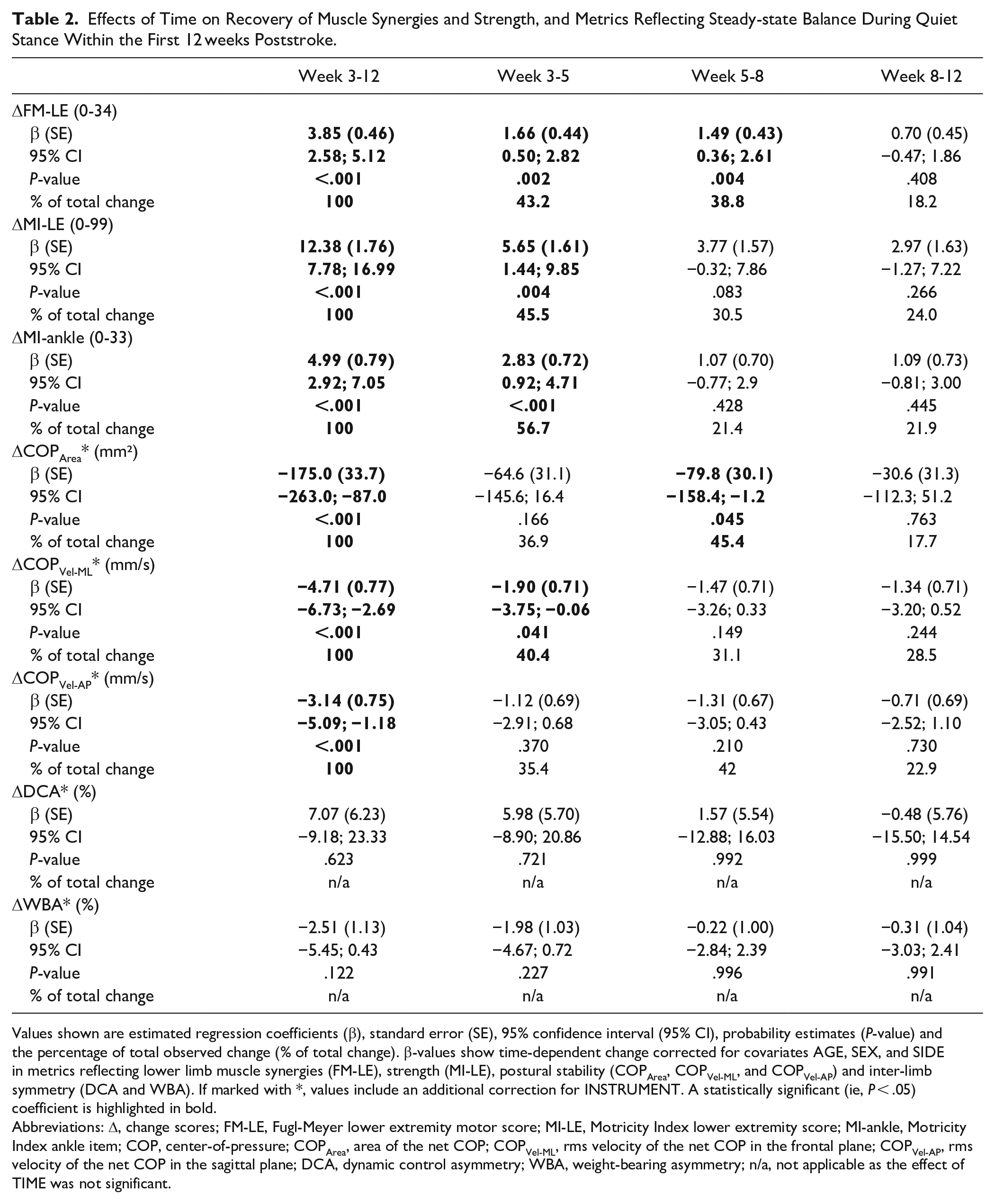
Values shown are estimated regression coefficients (β), standard error (SE), 95% confidence interval (95% CI), probability estimates (P-value) and the percentage of total observed change (% of total change). β-values show time-dependent change corrected for covariates AGE, SEX, and SIDE in metrics reflecting lower limb muscle synergies (FM-LE), strength (MI-LE), postural stability (COPArea, COPVel-ML, and COPVel-AP) and inter-limb symmetry (DCA and WBA). If marked with *, values include an additional correction for INSTRUMENT. A statistically significant (ie, P < .05) coefficient is highlighted in bold.
Abbreviations: Δ, change scores; FM-LE, Fugl-Meyer lower extremity motor score; MI-LE, Motricity Index lower extremity score; MI-ankle, Motricity Index ankle item; COP, center-of-pressure; COPArea, area of the net COP; COPVel-ML, rms velocity of the net COP in the frontal plane; COPVel-AP, rms velocity of the net COP in the sagittal plane; DCA, dynamic control asymmetry; WBA, weight-bearing asymmetry; n/a, not applicable as the effect of TIME was not significant.
Effects of Time on Recovery of Steady-State Balance during Quiet Stance
Panels B and C of Figure 2 show individual and mean time-dependent change in postural stability and symmetry metrics, respectively. As shown in Table 2, TIME was a significant factor for improvements from week 3 to 12 in COPArea (β = −175.0, 95% CI [−263.0; −87.0], P < .001), COPVel-ML (β = −4.71, 95% CI [−6.73; −2.69], P < .001), and COPVel-AP (β = −3.14, 95% CI [−5.09; −1.18], P < .001), after correction for INSTRUMENT as the only significant covariate for change in COPArea (β = 134.3, 95% CI [77.4;191.3], P < .001), COPVel-ML (β = 4.86, 95% CI [2.90; 6.83], P < .001), and COPVel-AP (β = 6.28, 95% CI [4.40; 8.16], P < .001). Further sub-analyses yielded significant reductions in COPArea between weeks 5 and 8 (β = −79.8, 95% CI [−158.4; −1.2], P = .045) and in COPVel-ML between weeks 3 and 5 (β = −1.90, 95% CI [−3.75; −0.06], P = .041).
No significant time-dependent change was found for DCA and WBA. Comparison with mean symmetry values in healthy subjects (DCA: 16.3%, SD = 31.8; WBA: −1.1%, SD = 3.5) showed significant differences in WBA at week 3 (difference = 7.7%, standard error [SE] = 3.0, P = .001), week 5 (difference = 7.2%, SE = 2.9, P = .005), week 8 (difference = 7.5%, SE = 2.9, P = .009), and week 12 (difference = 8.3%, SE = 3.0, P = .008). Differences in DCA were statistically significant at week 8 (difference = 42.5%, SE = 20.8, P = .029) and week 12 (difference = 51.2%, SE = 21.2, P = .012). Figure 3 depicts sway profiles measured at each time-point in a single subject.
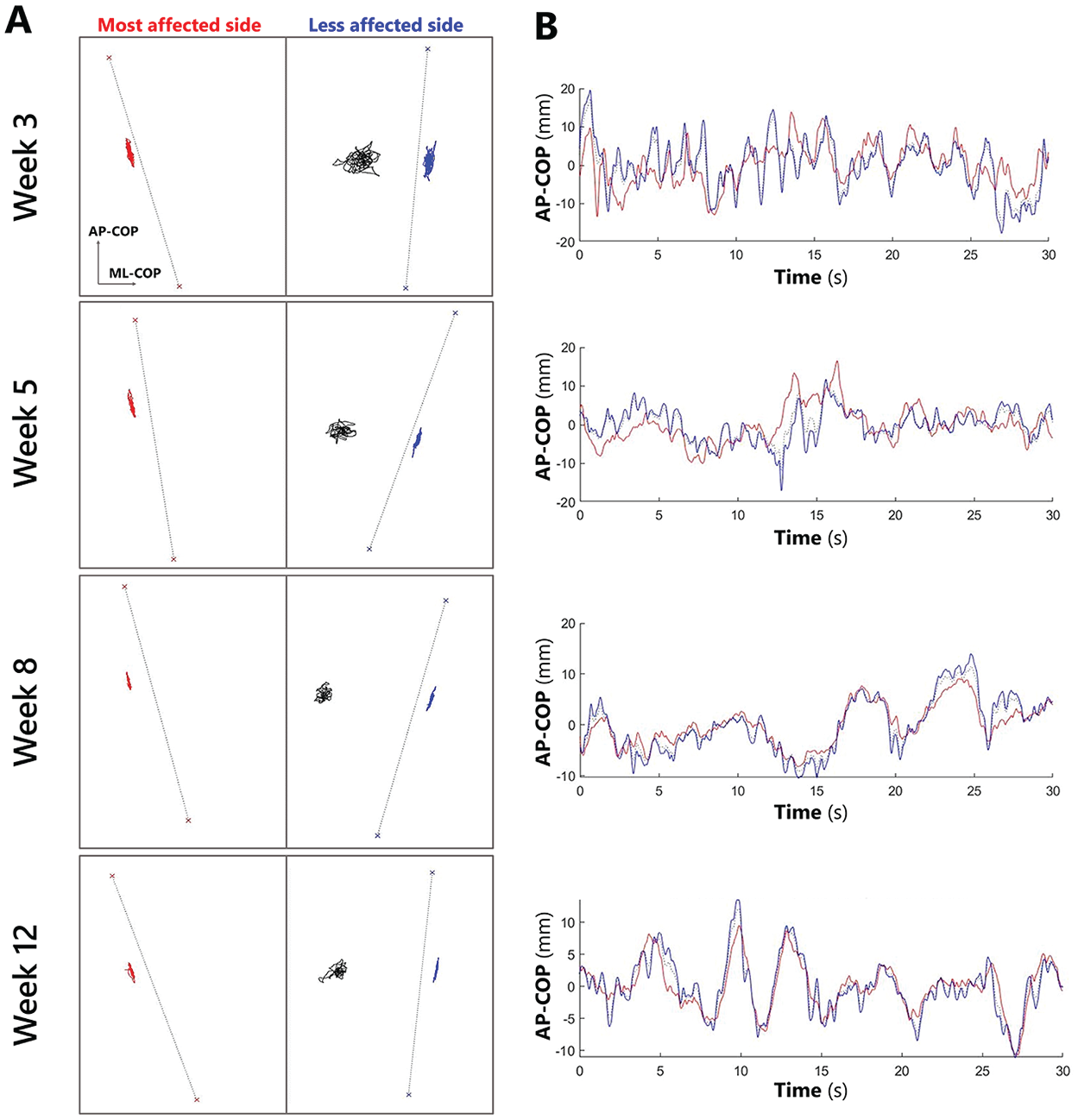
(A, B) Center-of-pressure (COP) sway profiles of an individual subject standing quietly, recorded at week 3, 5, 8, and 12 poststroke. The figures on the left (A) show the 2-dimensional individual-limb COP sway for the 2 sides separately (less affected side: blue/right; most affected side: red/left) and the net COP for both sides combined (black/mid). Note the decreasing net COP sway area (week 3: 318 mm²; week 5: 166 mm²; week 8: 127 mm²; week 12: 139 mm²), indicating gradual posture stabilization despite a persistent weight-bearing asymmetry reflected by the net COP deviation toward the less affected side. The figures on the right (B) show the corresponding time series of the anteroposterior COP displacement (AP-COP) in mm. Note the larger and faster movement on the less affected side (blue line) relative to the most affected side (red line). Accordingly, asymmetries in DCA were found (week 3: 27.3%; week 5: 32.5%; week 8: 47.8%; week 12: 44.7%) indicating a persistently greater balance contribution of the less affected leg.
Between-Subject Associations of Lower Limb Impairment Severity with Steady-State Balance
Table 3 shows the between-subjects analyses applied cross-sectionally at weeks 3, 5, 8, and 12 for either FM-LE, MI-LE, or MI-ankle with COPArea, COPVel-ML, COPVel-AP, DCA, or WBA. Scatterplots of these associations with their linear regression lines are provided in the supplement (Supplemental Figure 3, Supplemental Appendix C). The main effects of FM-LE, MI-LE, or MI-ankle were significant for COPArea, COPVel-ML, and COPVel-AP, as well as for WBA (P < .001). Additional significant covariates were INSTRUMENT (P < .001) for the associations with COPArea, COPVel-ML and COPVel-AP as the dependent variables; TIME (P < .05) for COPArea and COPVel-ML; and AFFECTED SIDE (P < .05) for COPArea (Table 3). Between-subject analyses with DCA yielded non-significant results.
Between-Subject Associations of Lower Limb Motor Impairment Severity With Steady-State Balance During Quiet Stance at week 3, 5, 8, and 12 Poststroke.
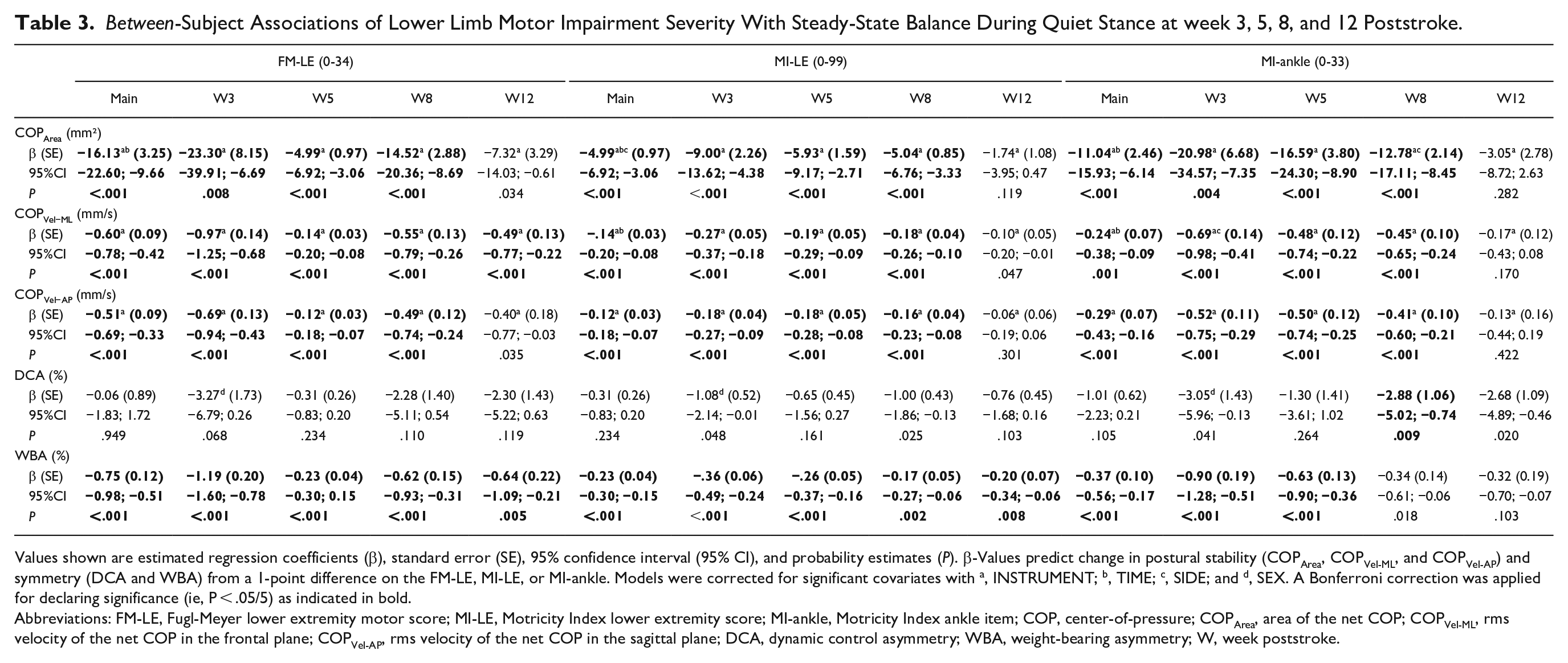
Values shown are estimated regression coefficients (β), standard error (SE), 95% confidence interval (95% CI), and probability estimates (P). β-Values predict change in postural stability (COPArea, COPVel-ML, and COPVel-AP) and symmetry (DCA and WBA) from a 1-point difference on the FM-LE, MI-LE, or MI-ankle. Models were corrected for significant covariates with a, INSTRUMENT; b, TIME; c, SIDE; and d, SEX. A Bonferroni correction was applied for declaring significance (ie, P < .05/5) as indicated in bold.
Abbreviations: FM-LE, Fugl-Meyer lower extremity motor score; MI-LE, Motricity Index lower extremity score; MI-ankle, Motricity Index ankle item; COP, center-of-pressure; COPArea, area of the net COP; COPVel-ML, rms velocity of the net COP in the frontal plane; COPVel-AP, rms velocity of the net COP in the sagittal plane; DCA, dynamic control asymmetry; WBA, weight-bearing asymmetry; W, week poststroke.
Sub-analyses concerning FM-LE, MI-LE, and MI-ankle scores yielded significant regression coefficients up until week 8 for COPArea, COPVel-ML, COPVel-AP, and WBA (P < .01, see Table 3 for more detail). At week 12, FM-LE remained a significant predictor of COPVel-ML (β = −0.49, 95% CI [−0.77; −0.22], P < .001) and WBA (β = −0.64, 95% CI [−1.09; −0.21], P = .005). Additionally, a single significant coefficient was identified for MI-LE scores at week 12 concerning WBA (β = −0.20, 95% CI [−0.34; −0.06], P = .008).
Within-Subject Associations of Lower Limb Motor Recovery with Change in Steady-State Balance
Regression coefficients between ΔFM-LE, ΔMI-LE, or ΔMI-ankle on the one hand, and ΔCOPArea, ΔCOPVel-ML, ΔCOPVel-AP, ΔDCA, or ΔWBA on the other were estimated for weeks 3 to 5, weeks 5 to 8 and weeks 8 to 12, using 36, 38, and 35 individual change scores, respectively. Scatterplots with their linear regression lines are provided in the supplement (Supplemental Figure 4, Supplemental Appendix C). As shown in Table 4, the main effects of ΔFM-LE, ΔMI-LE, and ΔMI-ankle were not significant for any dependent variable. Sub-analyses across the 3 epochs yielded a single significant regression coefficient for ΔMI-LE with ΔCOPVel-ML between weeks 8 and 12 (β = −.12, 95% CI [−0.21; −0.04], P = .007).
Within-Subject Associations of Lower Limb Motor Recovery and Change in Steady-State Balance During Quiet Stance Within the First 12 Weeks Poststroke.
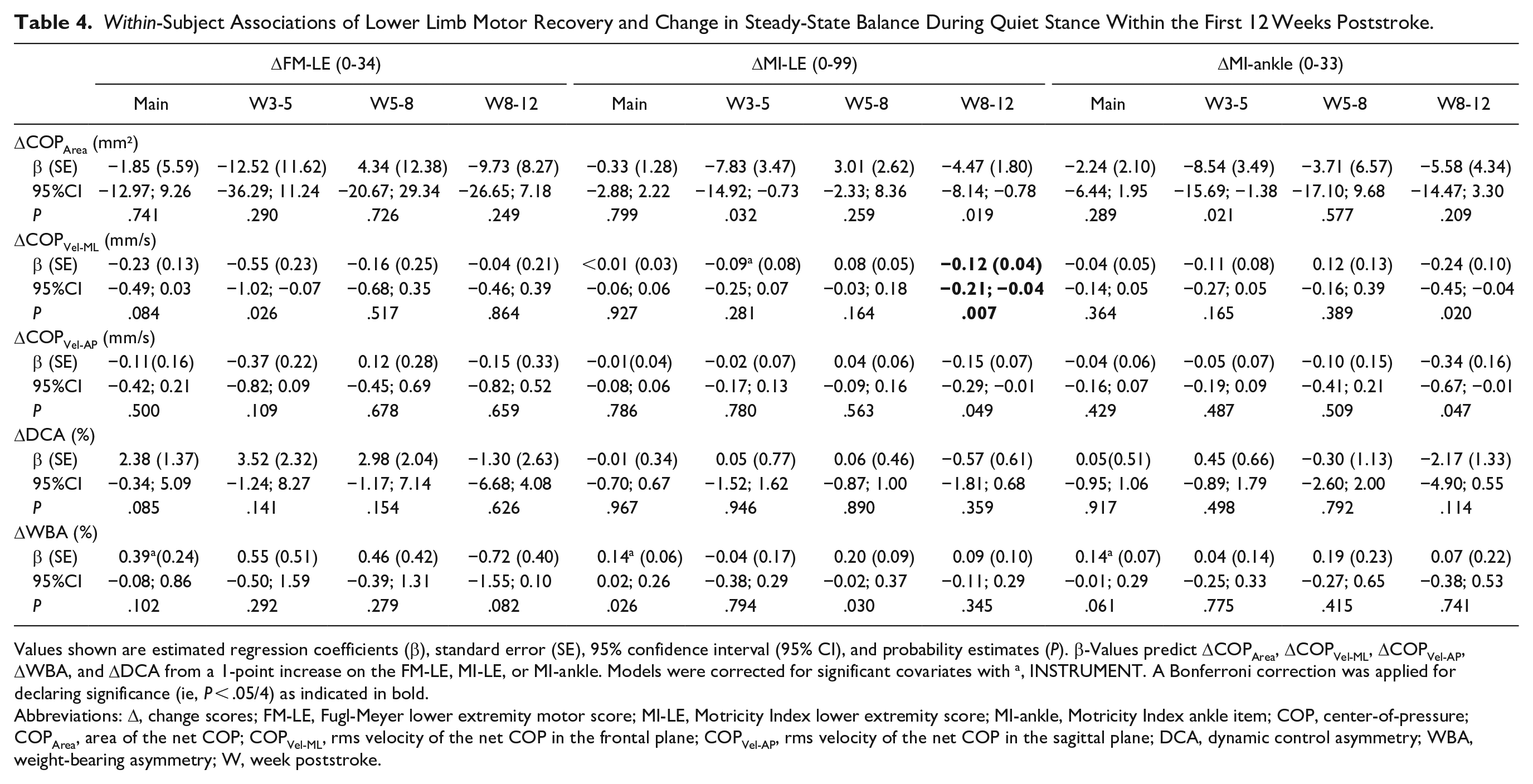
Values shown are estimated regression coefficients (β), standard error (SE), 95% confidence interval (95% CI), and probability estimates (P). β-Values predict ΔCOPArea, ΔCOPVel-ML, ΔCOPVel-AP, ΔWBA, and ΔDCA from a 1-point increase on the FM-LE, MI-LE, or MI-ankle. Models were corrected for significant covariates with a, INSTRUMENT. A Bonferroni correction was applied for declaring significance (ie, P < .05/4) as indicated in bold.
Abbreviations: Δ, change scores; FM-LE, Fugl-Meyer lower extremity motor score; MI-LE, Motricity Index lower extremity score; MI-ankle, Motricity Index ankle item; COP, center-of-pressure; COPArea, area of the net COP; COPVel-ML, rms velocity of the net COP in the frontal plane; COPVel-AP, rms velocity of the net COP in the sagittal plane; DCA, dynamic control asymmetry; WBA, weight-bearing asymmetry; W, week poststroke.
Discussion
The present prospective cohort study involving 48 subjects investigated the time course of steady-state balance during quiet stance in relation to lower limb motor recovery within the first 3 months poststroke. Controlling a high-positioned COM above a small base of support while standing is an easily standardized, yet skilled motor task requiring continuous postural corrections by the lower limbs. Unlike other prospective recovery studies in this field,11,14,17-19 we were interested in how clinically assessed impairments in muscle synergies (ie, FM-LE) and strength (ie, MI-LE and MI-ankle) of the most affected limb are associated with postural stability (ie, COPArea, COPVel-ML, and COPVel-AP) and asymmetric limb contributions to balance (ie, DCA and WBA) during quiet 2-legged stance. We therefore performed serial measurements in the same subjects and at fixed times poststroke.25,26 Our main findings are summarized below.
A restricted time window of recovery concerning motor impairments and postural stability that occurs within the first 8 weeks poststroke (Table 2).
Stroke subjects differ significantly from healthy controls with respect to inter-limb asymmetry in DCA and WBA.
Lack of recovery from asymmetries in DCA and WBA in the first 3 months poststroke, despite significant motor improvements in the most affected leg (Table 2).
Significant between-subject associations between motor impairment severity and postural instability (ie, COPArea, COPVel-ML, and COPVel-AP) as well as a preferred asymmetric stance (ie, WBA) within the first 3 months poststroke (Table 3).
Lack of significant between-subject associations of motor impairment severity with DCA (Table 3).
An overall lack of significant within-subject associations between improved muscle synergies and strength of the lower limb and recovery in postural stability and symmetry (Table 4).
In agreement with our first hypothesis, the contribution of the progress of time as a reflection of spontaneous neurobiological recovery, 7 was most pronounced for FM-LE, MI-LE, and MI-ankle between weeks 3 and 5 poststroke. Approximately half of the total observed change occurred within this relatively short epoch (FM-LE: 43.2%, MI-LE: 45.5%, and MI-ankle: 56.7%, Table 2). Recovery rapidly leveled off thereafter, which is in agreement with previous studies.5-7 In the literature, this restricted time window has also been described for the paretic upper limb5,10 as well as for other neurological impairments including visuospatial inattention 36 and aphasia, 37 suggesting spontaneous neurological restitution within the first 5 to 8 weeks poststroke.
Confirming our second hypothesis, the present study shows that progress of time is also an independent factor contributing to improved postural stability. Significant reductions in COPVel-ML and COPArea were most prominent within the first 8 weeks poststroke, responsible for about 75% of the total observed change (Table 2). Although COPVel-AP was not statistically significant within a specific epoch, it displayed a similar pattern of change in the first 12 weeks poststroke (Figure 2B). As such, steady-state balance became increasingly efficient, as reflected by a general COP sway reduction, in approximately the same time window as that seen for lower limb motor recovery.
A shared period of significant recovery has also been found in kinematic studies investigating the quality of upper limb motor performance relative to the Fugl-Meyer arm motor score.21,22 In contrast, the present study showed that DCA and WBA were, on average, invariant for change over time (Figure 2C). The persistent asymmetry of approximately 45% to 60% greater contribution of the less affected limb in terms of DCA approaches values reported in chronic patients. 16 Moreover, an unchanged asymmetric weight-bearing (about 40% of body weight placed on the most affected leg), despite significant COP sway reductions over time, agrees with other longitudinal studies starting their measurements within the first 3 months poststroke.14,18,19,38 Obviously, subjects preferred to keep and control their balance predominantly with their less affected side to achieve posture stabilization while standing. Figure 3 illustrates persistent asymmetries in a typically behaving subject.
In agreement with our third hypothesis, relatively strong between-subject associations were found, such that a preferred asymmetric stance appears strongly dependent on the lower limb impairment severity. It was previously shown in healthy subjects that a gradually loaded leg is increasingly involved in balance control.15,39,40 Thus, persistent loading of the less affected leg may indicate an attempt to even increase the contribution of this leg’s stabilizing ankle torques while standing. Our subsequent finding of a significant time-dependent association of impairment severity with postural instability up until week 8 poststroke (Table 3), furthers point toward an optimization of this compensatory strategy after independent stance is regained. Interestingly, impairment severity was not significantly associated with the DCA when comparing between subjects. This dissociation was already shown in the chronic phase poststroke 16 and may involve significant reliance on compensatory stabilization with the less affected leg even in mildly affected subjects (Supplemental Figure 3, Supplemental Appendix C).
As shown in Table 4, a dissociation between impairment scales and DCA was also found within subjects over time. This mismatch between motor improvements of the paretic leg on the one hand, and persistent inter-limb asymmetries on the other is a novel finding, as earlier longitudinal studies14,18,19,38 lacked measurements of change within the window of spontaneous neurobiological recovery. This finding may further explain our subsequent finding that FM-LE, MI-LE, and MI-ankle recovery neither explained within-subject postural stability improvements (ie, fourth hypothesis), despite a shared recovery time window at the group level. Seemingly, recovery of the most affected leg did not significantly contribute to an improved steady-state balance from 3 weeks poststroke onwards, complementing our finding of persistent asymmetries favoring the less affected side. Our results corroborate findings from a series of longitudinal electromyography (EMG) studies led by Jayne Garland, showing that balance reactions with the most affected leg in anticipation of rapid arm movements hardly “normalize” in the first 3 months poststroke,19,38,41 even after a mild stroke. 19 Instead, significant anticipatory change was consistently observed on the less affected side.19,38,41 The same studies19,38 found an asymmetric control during quiet stance, similar to the present findings, suggesting that this compensatory behavior generalizes across postural tasks.
It should be noted, however, that the present recovery study does not give an answer to why patients preferred compensatory strategies despite significant motor improvements at the most affected side. Obviously, steady-state balance is a multifactorial skill. Besides motor impairments, postural deficits in stroke patients standing upright have also been linked to impaired integration of afferent somatosensory and vestibular information,42,43 a resultant visual dependency17,44 and misperception of verticality,45,46 as well as reduced balance confidence to prevent falls. 47 To disentangle the relative importance of sensory impairments, cognition, and mood, we should have measured these factors as well in a longitudinal way. Alternatively, one may assume that observed intra-limb improvements in FM-LE and MI-LE (Table 2) were too small and incomplete for introducing restitution of inter-limb symmetry. As a result, relying on their less affected side may have been perceived as more efficient by patients. Similar to our findings, Roelofs et al 16 showed that ”well-recovered” patients in terms of FM-LE may show a significant balance control asymmetry, suggesting that DCA is a more responsive marker of remaining motor deficits than traditional clinical scales.
In summary, our findings suggest that stroke subjects recover their quiet standing balance mainly in the first 8 weeks poststroke by optimizing, rather than “normalizing” compensatory strategies involving the less affected limb. The independency of steady-state balance improvements and motor recovery of the most affected limb further suggests that only instrumented performance measures reflecting inter-limb asymmetry, such as DCA, are suitable to address the quality of movement in order to improve our understanding of balance recovery mechanisms poststroke.
Limitations
Several limitations of the present study should be considered. First, our sample size is limited and larger epidemiological studies incorporating serial instrumented performance measures are needed to generalize our findings. Second, since we started our assessments at 3 weeks poststroke, we may have missed some early changes in motor performance. Despite this, the study was successful in collecting data serially within subjects early poststroke by applying a postural task with relatively low functional demands. A third limitation is that our results are restricted to quiet 2-legged standing, which obviously allows compensation strategies. This may have prevent us from measuring the extend of “true” neurological recovery in the most affected leg for controlling balance. Fourth, as emphasized, our analyses are restricted to motor impairments in terms of FM-LE and MI-LE. Consequently, we did not investigate recovery in other potentially relevant impairments, such as muscle tone, 48 sensation, 42 or visuospatial perception. 49 Additionally, the FM-LE and MI-LE assess distal motor control through movement range and strength in ankle dorsiflexion, whereas quiet standing balance is mainly controlled by plantarflexor activity that resists forwards toppling due to gravity. 50 This narrow emphasis of clinical scales on foot elevation alone, may have prevented us from finding significant associations. Fifth, we used 2 measuring instruments to allow data acquisition in various settings. Since we used the same instrument within subjects and added the covariate INSTRUMENT systematically to our final analyses, we believe that the use of 2 different platform types did not affect our conclusions. Nevertheless, more research is needed for the development and validation of portable instruments to enable even larger multicenter studies with longer follow-ups beyond hospitalization. Lastly, we did not monitor treatment content and are unable to decide whether our findings were influenced by, for example, therapy dose or focus.
Future Directions
An unaddressed key question arising from the current study is: “Why do clinical improvements in muscle synergies and strength of the most affected leg hardly generalize to an improved quality of steady-state balance?” Addressing this question requires future studies with serial measurements of sensory and cognitive perception deficits as well as patients’ mood (eg, by using standardized questionnaires of balance confidence 51 ). In addition, future studies with serial EMG measurements are needed to show if the actual changes in intra-limb coordination of the paretic leg make a beneficial contribution to posture stabilization or, alternatively, should be seen as “noise” that needs to be suppressed while standing. Unravelling a potential mismatch between the preferred postural strategy and subjects’ capacity to normalize their quality of movement by an increased balance contribution by the most affected leg is important to address another unsolved question: “Are therapies aiming to restore symmetry, such as the Bobath approach 52 or visual feedback training, 53 counterproductive if we aim at posture stabilization and avoiding falls?” This question is important as falls remain a major health care problem at all stages of the disease. 54
To drive the field forward, it is important to reach agreement on a shared language and the metrics applied to assess qualitative aspects of movement. The SRRR mobility task force—a group of experts in the field of balance and gait research—currently gathers intending to build consensus on how future trials should address recovery. This will include standardized recommendations on taxonomy, timing, and choice of assessments as well as the metrics used to measure the quality of quiet standing balance and mobility performance within the first 6 months poststroke.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231186983 – Supplemental material for Recovery of Quiet Standing Balance and Lower Limb Motor Impairment Early Poststroke: How Are They Related?
Supplemental material, sj-docx-1-nnr-10.1177_15459683231186983 for Recovery of Quiet Standing Balance and Lower Limb Motor Impairment Early Poststroke: How Are They Related? by Jonas Schröder, Wim Saeys, Elissa Embrechts, Ann Hallemans, Laetitia Yperzeele, Steven Truijen and Gert Kwakkel in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors wish to thank Patrick De Bock (University of Antwerp) for assisting posturographic measurements in the course of this study, and Erik Fransen (StatUa, University of Antwerp) for statistical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary investigator of this study (JS) received a doctoral (PhD) grant for strategic basic research from the Research Foundation—Flanders (FWO application no.: 1S64819N) to perform the research yielding the results reported here.
Data Availability
The datasets supporting the results of this article are available from the corresponding author upon reasonable request.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
