Abstract
Background.
Altered dynamic functional connectivity has been associated with motor impairments in the acute phase post-stroke. Its association with somatosensory impairments in the early sub-acute phase remains unexplored.
Objective.
To investigate altered dynamic functional connectivity associated with somatosensory impairments in the early sub-acute phase post-stroke.
Methods.
We collected resting state magnetic resonance imaging and clinical somatosensory function of the upper limb of 20 subacute stroke patients and 16 healthy controls (HC). A sliding-window approach was used to identify 3 connectivity states based on the estimated dynamic functional connectivity of sensorimotor related networks. Network components were subdivided into 3 domains: cortical and subcortical sensorimotor, as well as cognitive control network. Between-group differences were investigated using independent t-tests and Mann–Whitney-U tests. Analyzes were performed with correction for age, head motion and time post-stroke and corrected for multiple comparisons.
Results.
Stroke patients spent significantly less time in a weakly connected network state (state 3; dwell time: pstate3 = 0.003, meanstroke = 53.02, SDstroke = 53.13; meanHC = 118.92, SDHC = 72.84), and stayed shorter but more time intervals in a highly connected intra-domain network state (state 1; fraction time: pstate 1 < 0.001, meanstroke = 0.46, SDstroke = 0.26; meanHC = 0.26, SDHC = 0.21) compared to HC. After 8 weeks of therapy, improvements in wrist proprioception were moderately associated with decreases in dwell and fraction times toward a more normalized pattern.
Conclusion.
Changes in temporal properties of large-scale network interactions are present in the early rehabilitation phase post-stroke and could indicate enhanced neural plasticity. These findings could augment the understanding of cerebral reorganization after loss of neural tissue specialized in somatosensory functions.
Introduction
The most common cause of multifaceted long-term disability is stroke.1,2 Only a small portion of stroke survivors achieve full recovery and up to 75% of them require some assistance for activities of daily living during their further lives. 3 Sensorimotor impairments of the upper limb are among the most frequently observed impairments. 4 In the last decade, there has been an increasing interest in the effect of acute somatosensory impairments on motor recovery after stroke.5 -7 Additionally, knowledge and insights into the structural and functional connectivity of the brain, and how this brain organization links to impairment severity and recovery are growing.8,9 Nevertheless, a consensus paper of stroke experts concluded that finding biomarkers for somatosensory function and recovery is research priority. 10 Moreover, they suggest resting state functional magnetic resonance imaging (fMRI) for the assessment of functional connectivity to be a promising technique.10,11 Up until now, resting state functional connectivity was investigated mostly by averaging the signal over the time of the entire scan sessions, typically lasting several minutes, losing information on potential temporal connectivity changes within these minutes. Recently, time-varying (dynamic) functional connectivity analysis has been proven to be a robust technique to evaluate fluctuations in moment-to-moment connectivity strengths, increasing the temporal resolution from minutes to seconds.12 -14 Typically, dynamic connectivity states are defined to identify similar characteristics of connectivity reoccurring over time. One exemplary connectivity state described in the literature exhibits strong connectivity within the motor area of each hemisphere and low connectivity with other regions or the other hemisphere and was called “the highly intra-domain connected state.”15 -18 Other reported connectivity states are: (1) a combined inter- and intra-domain connected state and (2) a weakly connected state consisting of respectively (a) high connectivity between and within a specific area such as the motor regions and (b) low connectivity between and within other (non-motor) areas.15 -18 The strength of dynamic functional connectivity is the assumption that changes in connectivity can occur within a scanning time of several minutes. Hence, subjects can enter a different connectivity state every few seconds. Interestingly, differences between patients and healthy controls (HC) are found in several neurological diseases for their flexibility to switch between connectivity states and time spent in a specific connectivity state. Hence, it has been suggested to be a powerful tool to investigate the effects of diseases such as Parkinson’s disease, 19 Huntington’s disease 20 and other neurological diseases.16,21 -23 In case of stroke, a few papers have used this technique and have indicated the robustness within the stroke population.17,18,24 -26 Further, different connectivity states were found to be particularly relevant for HC and stroke patients with mild and severe motor impairments. For instance, more moderately affected patients were found to spend more time in a more integrated state, while more severely affected patient had a higher likelihood to transition into a state with high segregation. 24 Altogether, these findings suggest that underlying cerebral mechanisms may depend on severity of impairment. 24
However, the differences in dynamic functional connectivity or times spend in specific connectivity states have not been investigated yet for somatosensory impairments in the upper limb.
Therefore, we aim to investigate the underlying neural correlates of somatosensory impairments post-stroke, from a dynamic functional connectivity perspective, allowing the delineation of altered pattern of functional connectivity. We hypothesize that, in line with the prior findings for motor impairments, 24 stroke patients will show an altered pattern consisting of more time spent into the highly connected intra-domain connectivity and low inter-domain connectivity compared to HC. Altogether, we will focus only on brain regions related to somatosensory function as described by Allen et al. 12 These regions were located in 3 domains: cortical, subcortical sensorimotor, and cognitive control domain.
Methods
Participants
We recruited 28 stroke patients from 3 different rehabilitation centers in Flanders, Belgium: UZ Leuven (Pellenberg), Jessa hospitals (Herk-de-Stad), and Heilig Hart Hospital (Leuven). Patients were included when they encountered their first stroke within 8 weeks of symptom onset; experienced sensorimotor impairment in the upper limb defined as <52 out of 57 on Action Research Arm Test (ARAT) 27 and score within the impairment range (0 to −100) of the standardized somatosensory deficit (SSD) composite score scale 28 ; were 18 years or older and had sufficient cooperation. Exclusion criteria were (1) other neurological or musculoskeletal disorders affecting the upper limb, (2) severe cognitive or communication deficits, (3) contraindications to perform magnetic resonance imaging (MRI), and (4) no informed consent.
Nineteen handedness- and age-matched HC were recruited from the community with exclusion criteria: history of neurological disorders, presence of somatosensory, or motor impairments in the upper limb and contra-indications for MRI. HC subjects that showed unknown brain lesions on their MRI scan were also excluded from further analysis. Three HC and 8 stroke patients were excluded from further analysis due to excessive in-scanner head motion (see paragraph “controlling for head motion” in Supplemental Appendix 2 for detailed information). Accordingly, final analyzes were conducted with a total of 20 stroke patients and 16 HC.
Data collection methods are described extensively elsewhere. 29 A summary of the methodology applicable to the subset of data used in this paper will be provided below. Research Ethics Committee of UZ/KU Leuven has given approval (s60278) and the trial was registered prospectively at clinicaltrials.gov (NCT03236376). Participants gave written informed consent before inclusion.
Note that the full data set has previously been analyzed with respect to the effects of sensorimotor therapy on resting-state fMRI alterations. 30 However, the scope of the previous and current studies differ markedly (effect of therapy longitudinally on static FC vs cross-sectional differences in dynamic FC between stroke patients and HC). All presented results and associated research questions are novel.
Clinical Assessment of Upper Limb Function
Hand sensorimotor deficits were quantified using the (i) Perceptual Threshold of Touch (PTT) for light touch perception, 31 Texture Discrimination Test (TDT) for tactile discrimination, 32 Wrist Position Sense Test (WPST) for wrist proprioception, 33 and functional Tactile Object Recognition Test (fTORT) for stereognosis. 34 All assessments are reliable and valid in the stroke population and norm values are available in the literature. The stroke patients were clinically assessed and underwent a MRI scan, at baseline (T1), after 4 weeks of additional upper limb therapy (additional to their usual rehabilitation program; T2) and after 4 weeks of follow-up (T3; see study design in Supplemental Appendix 1). In this manuscript, only baseline MRI scans (T1) were used for further analysis. HC were clinically assessed once, directly after the MRI scan. All subjects were assessed with the same clinical assessments and scanned in the same scanner at UZ Leuven hospital, Belgium using the same MRI protocol.
MRI Acquisition and Preprocessing
Acquisition of resting-state functional MRI data was performed on the same 3.0 Tesla Philips MR scanner (Best, The Netherlands) for all subjects (stroke and HC, on average 41 days after the stroke for patients) consisting of anatomical T1 and T2-weighted fluid-attenuated inversion recovery images (FLAIR) and a 7 minutes resting-state fMRI scan. During the resting-state scan, participants were instructed to lie still in the scanner with the eyes fixated on a white cross and to not think of anything in particular. To check for quality after the scanning procedure, patients were asked about their scanning. If patients reported general difficulties or even falling asleep and in case the investigator observed gross movements during the scanning process, it was noted down and taken into account during preprocessing. Detailed information concerning scanning parameters and preprocessing of rs-fMRI data can be found in Supplemental Appendix 2.
Preprocessing was performed toward an elderly brain MNI template 35 using the clinical toolbox for SPM and consisted of co-registration, realignment, normalization taking into account the lesions 36 using enantiomorphic transformation, scrubbing, and 8 mm smoothing. An overlaymap of the lesions can be found in Supplemental Appendix 3.
Intrinsic Connectivity Networks
Estimation of network components based on predefined network components of HC (from Allen et al) 12 was used for spatially constrained ICA analysis.37,38 The advantage of a spatially constrained ICA approach based on healthy subjects is the possibility to avoid bias induced by stroke lesions. Notwithstanding, the network components are adaptive to each individual network’s characteristics. Furthermore, these ICA components are freely available for downloads (https://trendscenter.org/data/) which increases the potential for replication or adoption of our approach by other research teams. Fourteen network components related to sensorimotor function were selected for this analysis and subdivided into 3 domains based on the networks reported by Allen et al 12 : cognitive control network, cortical, and subcortical sensorimotor network. An overview of the categorization of each component can be found in Table 1. Note that the resting state scan does not contain the cerebellum of each participant due to a limited field of view. Hence, cerebellar components were not included.
Components of the Cognitive Control, Cortical Sensorimotor, and Subcortical Sensorimotor Network Domains.

Based on components of Allen et al. 12
After back reconstruction of spatial distributions and corresponding time-courses of all components, we performed a quality check to assess spatial (in)accuracies and low ratio or low-to-high frequency spectra.
Subsequently, we employed a sliding window approach, as implemented in the GIFT toolbox (http://trendscenter.org/software/gift),12,39 -41 to estimate the dynamic functional network connectivity (dFNC) between each component pair based on the l1—regularized precision matrix. Age, mean FD, and time post-stroke were included as nuisance regressors to the analysis.
Next, k-mean clustering was used to create distinct, so-called “dynamic connectivity states.” Each of these states represents a recurrent pattern of connectivity across and within subjects. This cluster analysis was performed twice. In an initial run, we determined the optimal number of clusters based on the elbow criterion. Afterwards, we employed this optimal number for a more definite connectivity state construction. Each time window of each patient was then assigned to one of these connectivity states. Note that there is no guarantee that each subject will visit each connectivity state.
Finally, connectivity state characteristics such as dwell time (time a subject spent in a state without switching to another expressed in number of windows), fraction time (the percentage of the total time a subject spent in a given state expressed in percentage relative to total scanning time), and a number of transitions (how often a subject switches between states) were extracted for each subject. 15 These individual connectivity values were averaged per state for further analysis. Note that, in order to compare actual average time parameters (ie, dwell time and fraction time) of each state between both groups with a more accurate estimate of the actual dwell and fraction time in each state for each group, we have chosen to treat time windows that not enter a state as missing data for that specific state. However, we are aware of the possible alternative and to increase the transparency of our work, we have added the other approach in the secondary analysis below.
Statistical Analysis
Descriptive analysis was performed for patient characteristics. Additionally, independent, two-sided t-tests and Mann–Whitney U tests were used to investigate between-group differences after checking for normality. Due to ordinal clinical data, spearman rho correlations were calculated for all clinical assessments (PTT, TDT, WPST, and fTORT) with connectivity state characteristics such as dwell and fraction time (for each state separately). Note that correlation analyzes were performed for stroke subjects only. Correction for multiple comparisons was applied using the Bonferroni correction, statistical significance was defined as P < .0008. All statistical analyzes were performed using SPSS version 28.
Secondary Analysis
In a secondary analysis, the same analyzes were performed as described before, but with individual connectivity data averaged per state with missing data set to 0. Hence, if a subject did not enter a state, all linked values (ie, dwell time, fraction time, and dynamic connectivity) were set to zero. Results of this secondary analysis can be found in Supplemental Appendix 4.
Results
Participant Characteristics
We included 20 stroke patients and 16 age- and handedness-matched HC in the analysis with a mean age of 64.86 years old. About 57.1% of the stroke patients and 37.5% of the HC were male. Four stroke patients and 1 HC were left-handed. Stroke patients were on average 41.43 days post-stroke and suffered mostly from ischemic stroke, in the right hemisphere (ischemic stroke and right hemisphere both in 71.4%). Participant characteristics can be found in Table 2.
Participant Characteristics.
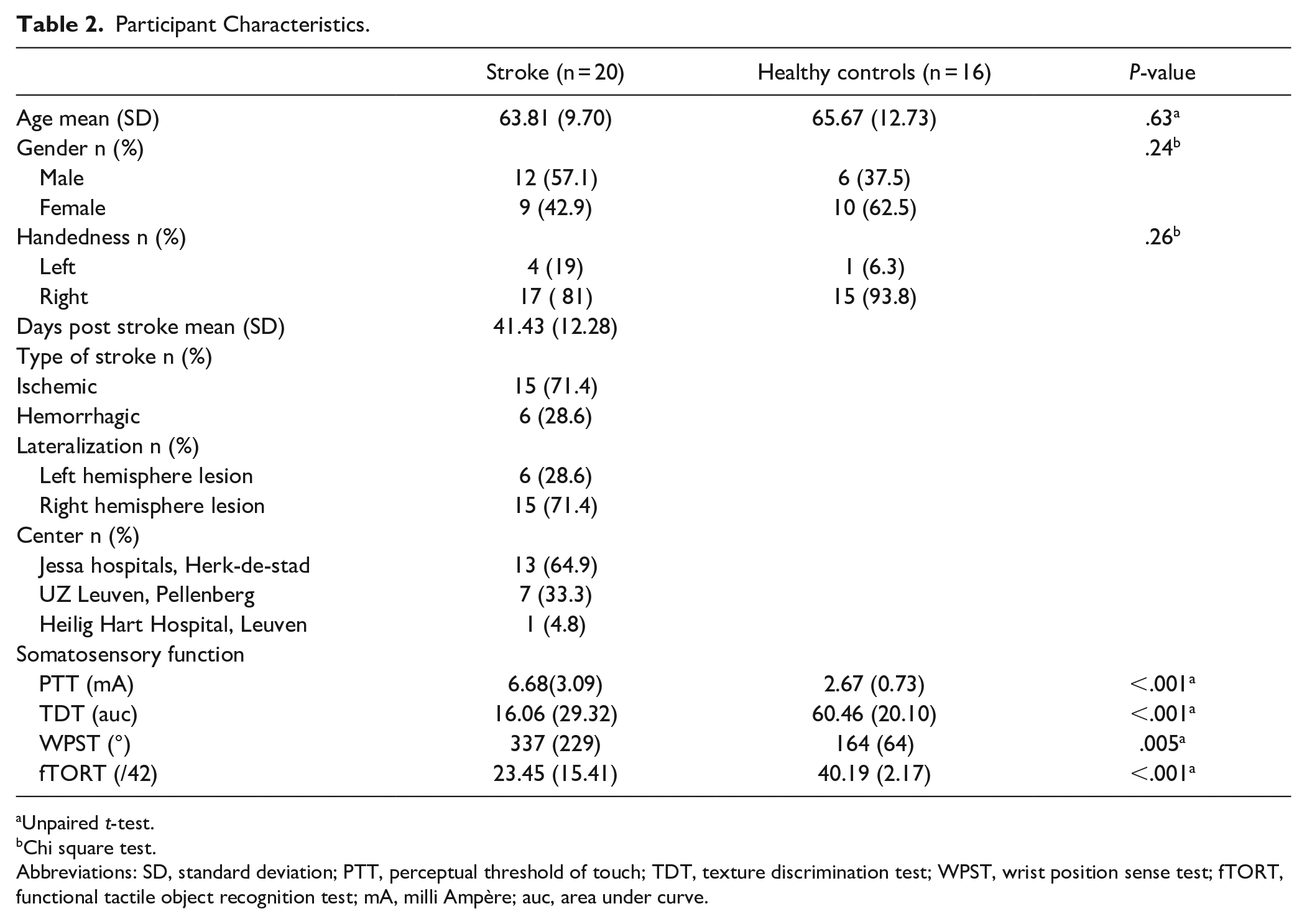
Unpaired t-test.
Chi square test.
Abbreviations: SD, standard deviation; PTT, perceptual threshold of touch; TDT, texture discrimination test; WPST, wrist position sense test; fTORT, functional tactile object recognition test; mA, milli Ampère; auc, area under curve.
Dynamic Functional Network Connectivity (dFNC) States
Three states of dFNC were identified showing reoccurring quasi-stable connectivity patterns over time and across subjects and within and across the 3 predefined network domains (cognitive control network, cortical, and subcortical sensorimotor network). Visualization of these 3 states, separately for patients and HC, can be found in Figure 1. The first state, our so-called “regionally densely connected state,” was characterized by high connectivity within the cortical and subcortical sensorimotor domains in combination with largely negative correlations between these domains. The domain of cognitive control networks featured neither pronounced positive nor negative connectivity except for the precentral + supplementary motor area (SMA) component of the cognitive control network, which showed high positive connectivity with the cortical sensorimotor network. A second state consisted of a combination of strong positive intra-component connectivity within the sensorimotor component and slightly positive connectivity between the sensorimotor and cognitive control component with exception of the same precentral + SMA component of the cognitive control network showing again higher connectivity with the cortical sensorimotor network. Third, the weakly connected state consists of low to no connectivity between network domains (sensorimotor, cognitive control, or subcortical) as well as weak intra-domain connectivity with main connectivity between the superior parietal lobe and paracentral lobe. The majority of stroke patients entered all 3 states (nstate1 = 20, nstate2 = 14, and nstate3 = 15), in contrast to HC of which the majority only entered the third, weakly connected state (nstate1 = 5, nstate2 = 6, and nstate3 = 15). Missing data can be calculated based on the number of subjects that had entered each state. Hence stroke patients had no missing data for state 1, missing data for 6 subjects for state 2 and 5 subjects for state 3; for HC missing data was largest in state 1 and 2, 11 and 10 subjects respectively; only 1 HC subject did not entered state 3.
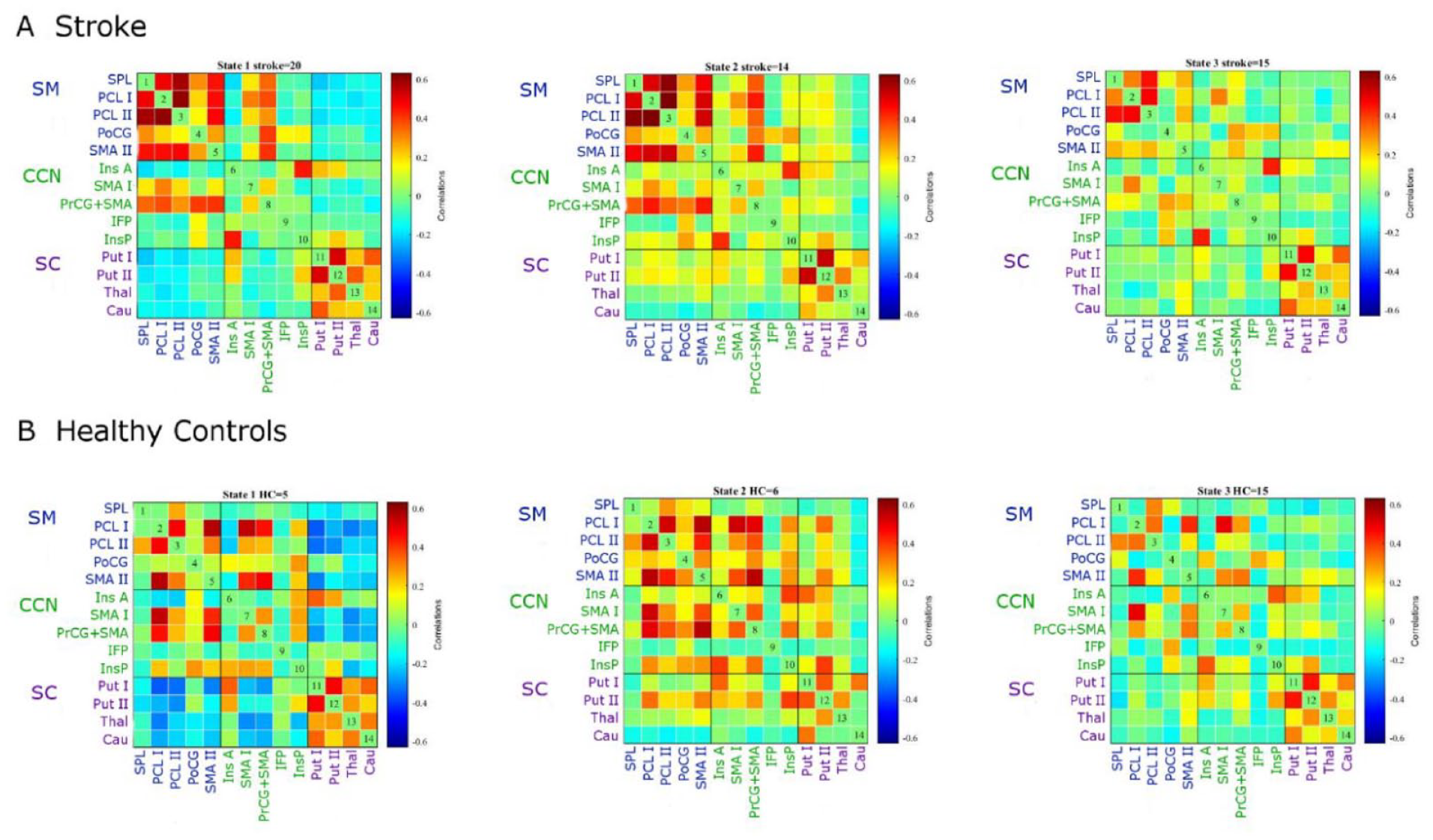
Dynamic functional network connectivity states. (overview of abbreviations of components can be found in Table 1; SM: cortical sensorimotor components, CCN: cognitive control network components; SC: subcortical sensorimotor components) state 1: highly positive intra-domain and largely negative inter-domain connectivity, state 2: combined positive inter-and intra-domain connectivity, state 3: overall weakly connected connectivity. The majority of stroke patients entered all 3 states (nstate1 = 20, nstate2 = 14, and nstate3 = 15), in contrast to HC of which the majority only entered the third, weakly connected state (nstate1 = 5, nstate2 = 6, and nstate3 = 15).
Differences in dFNC Between Strokes and HC
Figure 2 shows between-group differences for each component pair in each connectivity state after FDR-correction for multiple comparisons. Strong differences were found for connections between the superior parietal lobe and all further components of the cortical sensorimotor domain. These differences showed higher functional connectivity in stroke patients compared to HC. Remarkably, the connection between the superior parietal lobe and paracentral lobe was significantly increased in stroke patients in all 3 states.
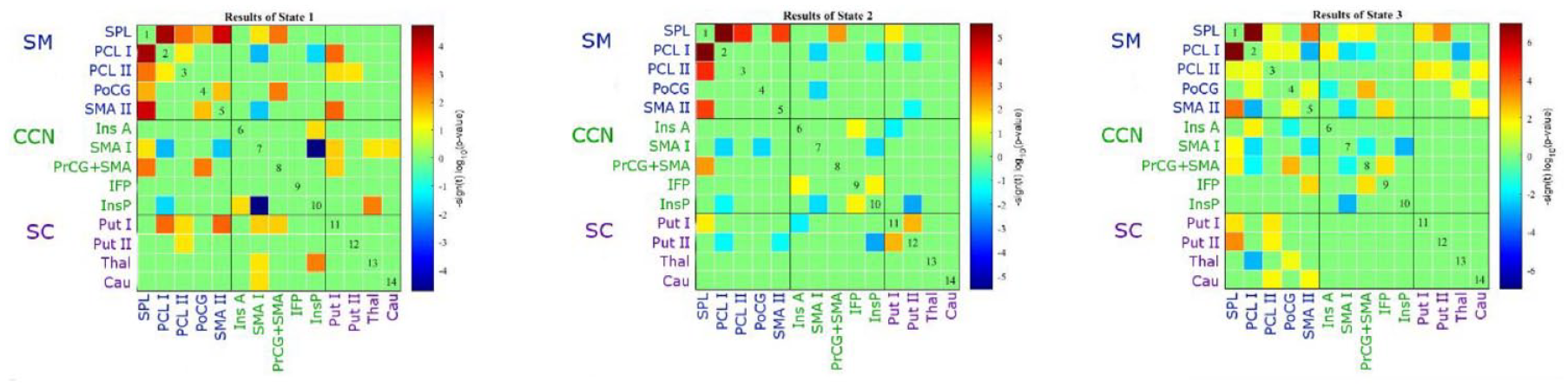
Connectivity matrix of between-group difference for each component pair in each connection state. Stronger differences are shown in favor of the stroke group (red) or healthy control (HC) (blue). SM: sensorimotor network, CCN: cognitive control network, SC: subcortical network; state 1: highly positive intra-domain and largely negative inter-domain connectivity, state 2: combined positive inter-and intra-domain connectivity, state 3: overall weakly connected connectivity. An overview of abbreviations of components can be found in Table 1.
Further, differences in dynamic measures, such as dwell and fraction times, between HC and stroke patients were assessed. Independent t-tests revealed significant differences in temporal features between both groups in states 1 and 3 (see Figure 3). Stroke patients stayed longer in state 1 in contrast to HC who spent most time in state 3. Fraction times of stroke patients were significantly higher for state 1 (fraction timestate1 < 0.001, meanstroke = 0.46, SDstroke = 0.26; meanHC = 0.26, and SDHC = 0.21) and a trend toward lower for state 3 (fraction timestate3 = 0.03; meanstroke = 0.43, SDstroke = 0.30; meanHC = 0.80, SDHC = 0.24). Additionally, dwell times of stroke patients were significantly lower for state 3 (dwell timestate3 = 0.003, meanstroke = 53.02, SDstroke = 53.13; meanHC = 118.92, SDHC = 72.84). No between-group differences were found for a number of transitions. However, stroke patients tended to switch more often between states (not reaching significance) compared to HC who stayed longer in 1 state (P < .03, meanstroke = 2.95, SDstroke = 1.63; meanHC = 1.63, and SDHC = 1.89).
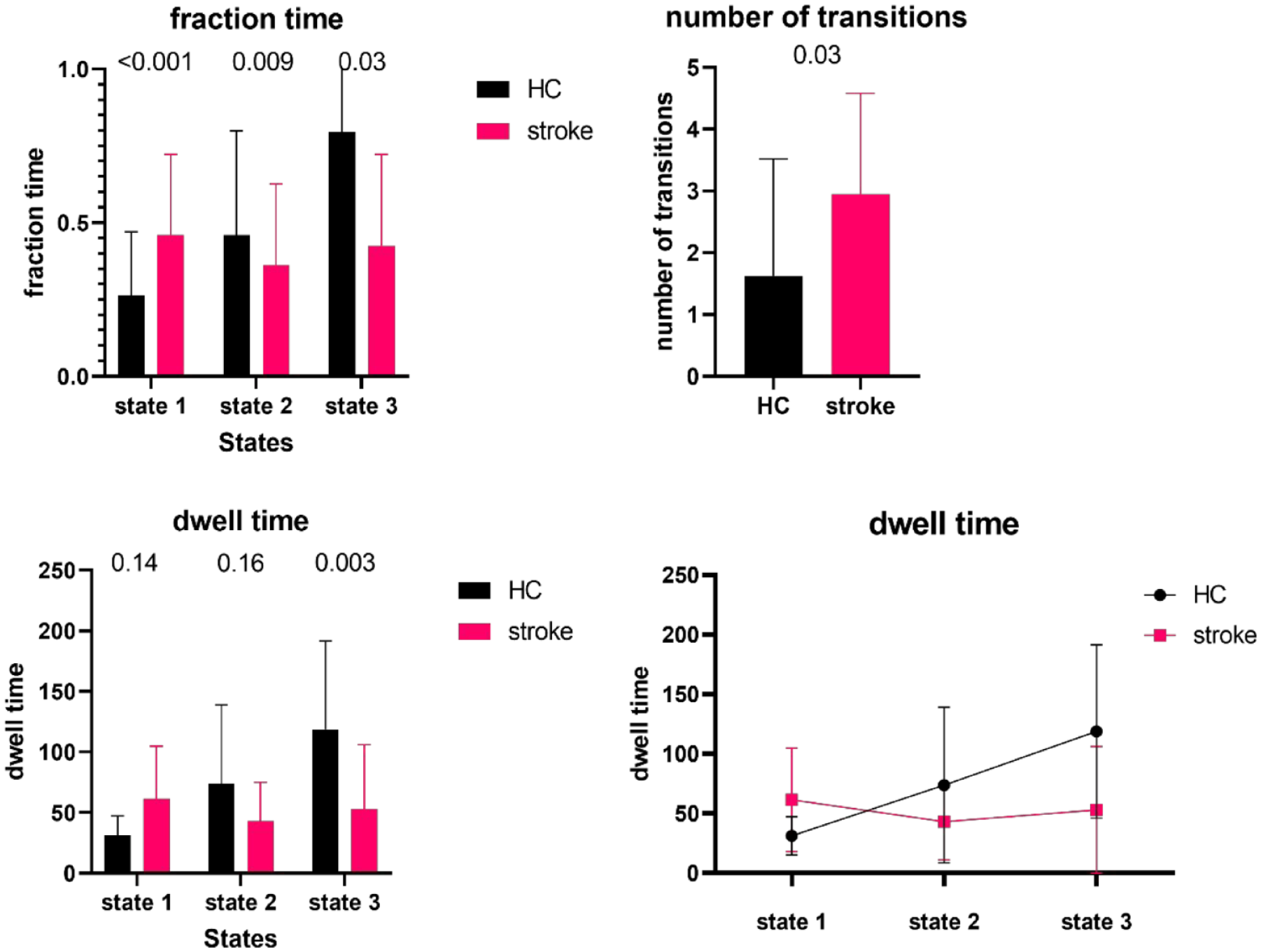
Between-group differences in dynamic functional connectivity measures. Differences in fraction time (expressed in percentage relative to total scanning time), dwell time (expressed in number of time windows), and number of transitions between stroke patients and healthy controls (HC) for each state were investigated using independent t-tests. state 1: highly positive intra-domain and largely negative inter-domain connectivity, state 2: combined positive inter-and intra-domain connectivity, state 3: overall weakly connected connectivity. After correction for multiple comparison, significant P-values were set on P < .008.
Brain–Behavioral Relationships
To gain insights into whether these significant group differences were also clinically interpretable, non-parametric spearman rho correlations were used to investigate the association between changes in sensorimotor outcome and dynamic functional connectivity parameters (fraction time, dwell time, and number of transitions) in the stroke population. Hence, results are focussed on scans from T1 in relation to behavioral changes from T1 to T2 or T3. After Bonferroni correction for multiple comparisons, the level of significance was set to P < .008. Significant correlations were found for improvements from baseline to follow-up (8 weeks) in Perceptual Threshold of Touch (PTT) with longer dwell timesstate1 (rho = −.80, P < .001). Additionally, significant correlations only without correction for multiple comparisons were found and reported as trends toward significant correlations. Both trends toward higher fraction (rho = −.56; P = .03) and dwell times (rho = −.51, P = .04) in state 2 were associated with improvements in wrist position sense test (WPST) after 8 weeks. Further, trends in 4-week improvements on stereognosis (fTORT; rho = .47, P = .04) and wrist proprioception (WPST; rho = .66, P = .003) were associated with longer dwell timesstate2 and shorter fraction timesstate1, respectively. An overview of all correlations can be found in Table 3. Detailed information about clinical improvements over time can be found in our previous work. 42
Brain–Behavioral Associations.
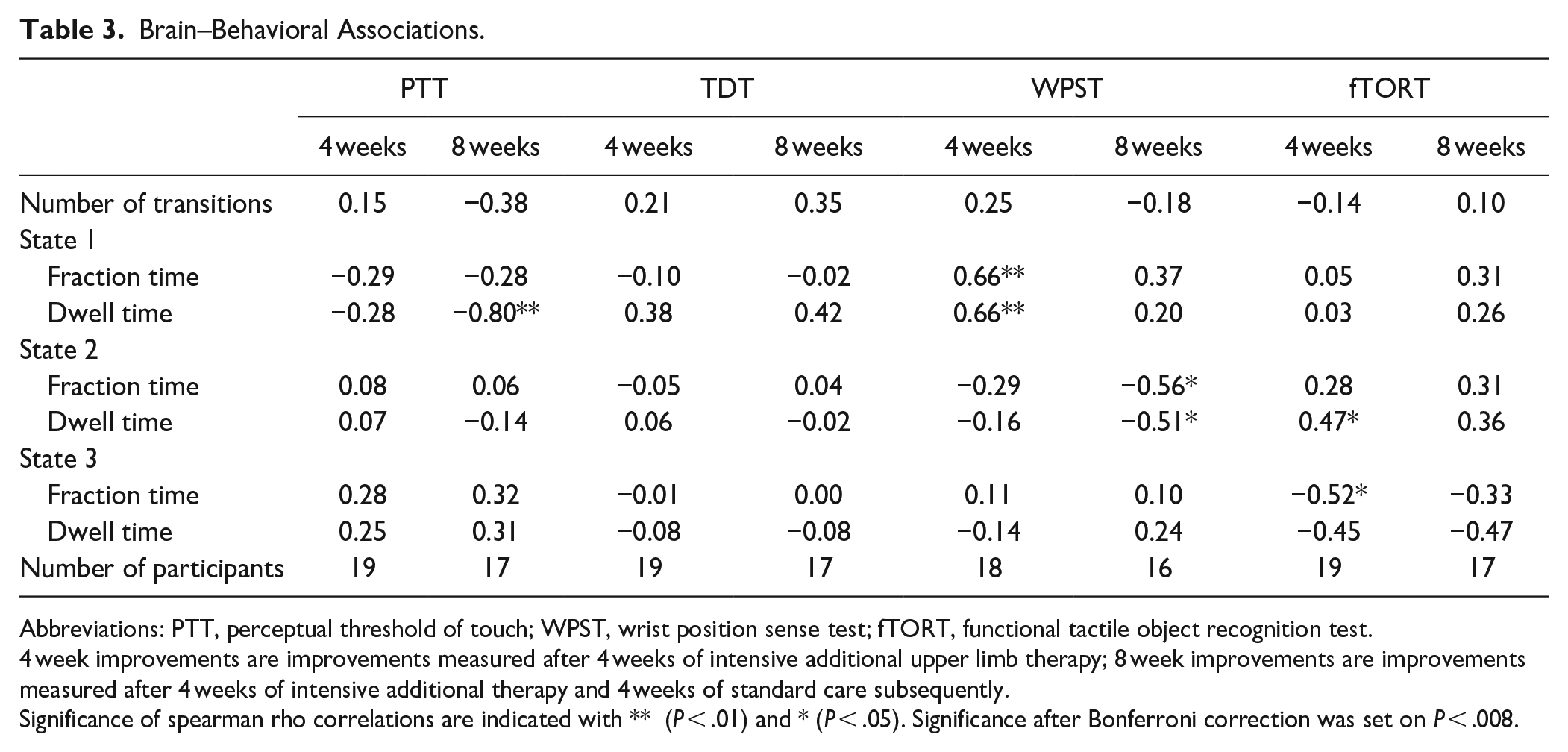
Abbreviations: PTT, perceptual threshold of touch; WPST, wrist position sense test; fTORT, functional tactile object recognition test.
4 week improvements are improvements measured after 4 weeks of intensive additional upper limb therapy; 8 week improvements are improvements measured after 4 weeks of intensive additional therapy and 4 weeks of standard care subsequently.
Significance of spearman rho correlations are indicated with ** (P < .01) and * (P < .05). Significance after Bonferroni correction was set on P < .008.
Discussion
We investigated differences in dynamic functional connectivity features between stroke patients with sensorimotor impairments of the upper limb and HC. Evaluating the dynamic connectivity for 3 specific domains: the cortical motor, subcortical motor, and cognitive controls domain which are based on the categorization by Allen et al 12 (see Table 1 for overview of consistence of every domain), we found 3 dynamic connectivity states to be the most prominent ones. They consisted of (1) a connectivity state 1 with highly positive intra-domain and largely negative inter-domain connectivity, (2) a connectivity state 2 with combined positive inter- and intra-domain connectivity, and (3) an overall weakly connected connectivity state 3. Significant differences were observed in the time spend for each state between stroke patients and HC. Stroke patients showed more time spend in state 1 with highly positive intra-domain connectivity whereas HC spend most time in state 3 with overall weak connectivity. Hence, stroke patients spent more time in a state consisting of strong connections within the defined domain, for example, cortical motor domain, but showed weak connectivity with other domains. In contrast to HC who showed mostly weak connectivity within and between all domains. Additionally, stroke patients showed a non-significant slightly higher amount of transitions between states. Finally, somatosensory improvements over time were associated with dynamic characteristics of state 2.
We have identified 3 states of dynamic functional connectivity, which were in line with states described in previous literature,15 -18,43,44 supporting the validity of the findings. Similar to Bonkhoff et al 15 investigating motor function, we found differences in state preference between stroke patients, who spent most time in the highly intra-domain connected state (= state 1), and HC, who preferred state 3 (weakly connected state). The pronounced higher time values for state 3, consisting of a more frequently identified weakly connected state, and less identified high connected states 1 and 2 in HC, is in line with previous literature, indicating the consistency of dynamic connectivity characteristics.12,16,18,19,45, 46 Comparably with our results concerning state preferences of the stroke population, Bonkhoff et al 15 described a preference for a state similar to our state 1 and for a state resembling our state 3 in severely and moderately motor impaired stroke patients respectively. Additionally, better recovery of motor function was found to be associated with shorter times spent in a functionally integrated state (cfr our state 1). 26 Similarly, in cognitive stroke patients, longer mean dwell times for state 1 are reported by Yue et al. 44 In contrast, Wang et al 17 described the opposite. However, differences in stroke patient characteristics are apparent. Wang et al investigated pontine stroke within the chronic phase (>6 months post stroke) and Bonkhoff et al acute phase (<16 days post stroke) and cortical and subcortical stroke whereas we have investigated cortical (majority MCA) stroke in the subacute phase. Additionally, differences in brain areas assessed were present. Wang et al have assessed whole-brain connectivity, which could overshadow specific sensorimotor-related differences between groups. 17 Another paper investigating stroke patients in a post stroke period comparable to ours (1 month post stroke) and moderate to severe motor impairments, found a decreased global efficiency in dynamic functional connectivity states. This is in line with our findings of decreased inter-domain connectivity. 43 Previous literature already has shown differences in dynamic functional connectivity of different phases post stroke 18 or lesion location 47 which is consistent with our differences in findings. The highly intra-domain connected state was previously reported to be associated with enhanced levels of brain plasticity 48 and could indicate the recovery process of the brain in the subacute phase post-stroke.
Second, we have identified associations between somatosensory improvements and dynamic functional connectivity characteristics. Eight-week improvements for proprioception (WPST) and light touch perception are moderately associated with higher fraction and longer dwell times in state 1 (PTT) and state 2 (WPST). Additionally, 4-week improvements on stereognosis (fTORT) were associated with longer dwell times in state 2 and lower fraction time in state 3. Further 4-week improvements on WPST were found to be moderately associated with a decrease in fraction and dwell time of state 1. The latter correlations are particularly interesting since they suggest that clinical recovery of wrist proprioception is also noticeable in connectivity characteristics adapting toward the pattern of HC both in state 1 and state 2. Similarly, previous studies reported associations between recovery of motor function and dynamic functional connectivity.18,24,26. More specifically, Bonkhoff et al 26 investigated the potential of dynamic functional connectivity measures for the prediction of motor outcomes. They concluded that motor recovery in the first 6 months was best predicted by the dwell times of connectivity states combined with the initial impairment. Changes in dynamic functional connectivity between the acute and subacute phase, more specifically increases in temporal variability in ipsilesional M1, were found to be associated with recovery of motor function by Hu et al. 18 On the other hand, Wang et al 17 could not find correlations with motor function in chronic pontine stroke patients. The latter emphasizes the potential difference in the effect of lesion location and phase post-stroke on brain connectivity changes.
Looking more in detail at the specific regions showing strong connections in the different states, we found that SPL is a prominent region. In all 3 states, SPL shows stronger connectivity with the paracentral lobe in stroke patients compared to HC indicating the important role of sensorimotor function. Previous literature has already indicated the important role of SPL for sensory integration of different modalities as well as for upper limb coordination. 49 Hence, increased dFNC could indicate the compensatory role of the SPL for sensorimotor function in stroke patients. Similar to our hypothesis, Rehme et al 49 hypothesized that the increased SPL activity they found during task fMRI could be a compensatory mechanism for controlling the paretic arm. Contrary, decreased dFNC between the posterior insular cortex and SMA was also found in all 3 states but was strongest in the first state which can indicate the important role of the posterior insular cortex for sensorimotor integration. The insular cortex was previously described to play a complex but important role in sensory processing and integration with motor function.50,51 We could hypothesize that the posterior insular cortex has a unique function within this integration, which cannot be compensated by other neighboring areas. This could explain the association between decreased dFNC and somatosensory impairments that all included stroke patients encountered.
An important remark for interpretation of the results is our choice of handling subjects not entering a specific state. While there are good reasons to choose either one of the approaches (setting the values to zero or handle them as missing data), we have privileged the missing data approach above the setting to zero approach as it may give a more accurate estimate of the actual dwell and fraction time in each state for each group. When the dwell or fraction times for a specific state of a patient are set on zero, this could substantially affect the average dwell or fraction time of this state (eg, subjects could differ in their likelihood to enter a state, but have comparable dwell and fraction times when they do). Our aim was to compare actual average time parameters of each state between both groups (HC and stroke patients). Therefore, we have chosen to treat time windows that not enter a state as missing data. However, we are aware of the possible alternative. To increase the transparency of our work, we have added the other approach as secondary analysis.
Further, some limitations should be recognized. First, the sample size is rather small hampering to find significant results for the brain–behavior correlations. However, our sample size is comparable with other recent dFNC studies in stroke.15,17,25,26 Second, heterogeneity of the lesion size and locations makes it more difficult to draw conclusions. We have employed enantiomorphic normalization 52 and hence took stroke lesions into account when performing spatial normalization, ensuring a high quality of these normalizations. Moreover, parcelation of the ICA network components was based on HC to minimize the effect of lesions. Future research needs to be done to further differentiate between dynamic connectivity patterns associated with patient recovery profiles to be able to predict long-term outcome after stroke and to identify responders and non-responders of therapy based on dynamic functional connectivity patterns. Last, scrubbing was performed during pre-processing, which influences the temporal position of the frame. However, when scrubbing was too extensive (mean FD >0.5 and/or number of scrubbed frames >125 out of 250), these subjects were excluded from further analysis. Based on Laumann et al, 53 mild scrubbing does not hamper the dynamic analysis significantly. Additionally, the results of our HC are in line with connectivity states and dynamic characteristics of previous literature.12,19 45,46
Conclusions
In this study, we investigated alterations in patterns of dynamic functional connectivity in subacute stroke patients associated with somatosensory impairments in the upper limb. We found that stroke patients spent more time in the highly intra-domain connected state and showed a higher number of transitions between states suggesting potential enhanced brain plasticity in this early subacute phase post-stroke to compensate for the loss of function.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231179172 – Supplemental material for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683231179172 for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke by Nele De Bruyn, Anna K. Bonkhoff, Leen Saenen, Liselot Thijs, Bea Essers, Kaat Alaerts, Prof and Geert Verheyden, Prof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683231179172 – Supplemental material for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke
Supplemental material, sj-docx-2-nnr-10.1177_15459683231179172 for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke by Nele De Bruyn, Anna K. Bonkhoff, Leen Saenen, Liselot Thijs, Bea Essers, Kaat Alaerts, Prof and Geert Verheyden, Prof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683231179172 – Supplemental material for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke
Supplemental material, sj-docx-3-nnr-10.1177_15459683231179172 for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke by Nele De Bruyn, Anna K. Bonkhoff, Leen Saenen, Liselot Thijs, Bea Essers, Kaat Alaerts, Prof and Geert Verheyden, Prof in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-4-nnr-10.1177_15459683231179172 – Supplemental material for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke
Supplemental material, sj-tif-4-nnr-10.1177_15459683231179172 for Altered Dynamic Resting State Functional Connectivity Associated With Somatosensory Impairments in the Upper Limb in the Early Sub-Acute Phase Post-Stroke by Nele De Bruyn, Anna K. Bonkhoff, Leen Saenen, Liselot Thijs, Bea Essers, Kaat Alaerts, Prof and Geert Verheyden, Prof in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to acknowledge all physiotherapists, occupational therapists, and nursing staff of the neurorehabilitation wards of UZ Leuven, campus Pellenberg, Jessa Hospitals Herk-de-Stad, and Heilig Hart Hospital Leuven who helped with the practical side of patient recruitment. Further, MSc students David Bryon, Gert Vangilbergen, Silke Francken, Lien Francken, Jana De Smeyter, and Justine Cools are acknowledged for their practical support during MRI scanning sessions.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Flanders Research Fund (FWO; grant numbers 1189819N and 1519719N) and Post-Doctoral fellowship of KU Leuven (PDM/20/187). A.K.B. is supported by an MGH ECOR Fund for Medical Discovery (FMD) Clinical Research Fellowship Award.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
