Abstract
Background
Cognitive impairment is common in patients with traumatic brain injury (TBI). Studies that have examined the effectiveness of neurofeedback (NFB) on cognitive function following TBI have had poor study designs and small sample sizes.
Objectives
This randomized controlled trial assessed the effects of low-resolution tomography Z-score NFB (LZNFB) and theta/beta NFB on cognitive impairment, return to productive activity, and quality of life in patients with TBI.
Methods
We randomly assigned 87 patients with TBI with cognitive impairment to LZNFB, theta/beta NFB, or usual care (UC) groups. Patients in both NFB groups received weekly 60-minute treatment for 10 weeks, and those in the control group received UC and telephone interviews for 10 weeks. The primary outcome was cognitive function as measured by performance on cognitive tasks; the secondary outcomes included productive activity and quality of life based on the Community Integration Questionnaire-revised (CIQ-R) and the Quality of Life after Brain Injury (QOLIBRI), respectively, at baseline and immediately after the last intervention.
Results
The LZNFB group exhibited significantly greater improvements in immediate recall, delayed recall, recognition memory, and selective attention compared with the UC group; the theta/beta NFB group exhibited improvements in only immediate memory and selective attention (P < .05). The total CIQ-R scores of the LZNFB group after treatment were significantly improved than those of the UC group were.
Conclusion
Consecutive LZNFB achieved therapeutic effects in memory, attention, and productive activity, whereas theta/beta NFB improved memory and attention in patients with TBI.
This trial was prospectively registered at ClinicalTrial.gov (registration number: NCT03515317; https://clinicaltrials.gov/ct2/show/NCT03515317).
Introduction
Cognitive impairment is the most common and debilitating residual symptom of traumatic brain injury (TBI) regardless of severity.1,2 It substantially affects an individual’s ability to return to productive activity, 3 decreases an individual’s quality of life, 4 and has a high socioeconomic and health-care burden. 5 Deficits in memory, attention, and information processing speed and the impairment of basic cognitive functions may implicate higher level cognitive functions (eg, executive function). Strategies that target the preservation of basic cognitive functions are key to preventing or delaying posttraumatic cognitive impairment.
Cognitive rehabilitation is the most frequently prescribed treatment for managing post-TBI cognitive impairment 6 ; however, 2 meta-analyses of randomized controlled trials (RCTs) have reported conflicting findings.7,8 Pharmacotherapy (eg, methylphenidate) has been employed to accelerate cognitive recovery in patients with TBI. Nevertheless, systematic reviews have failed to demonstrate its positive effects on cognitive recovery.9,10 Alternative approaches are therefore required to minimize the risk of cognitive impairment following TBI.
Both abnormal brain network connectivity11 -13 (eg, axonal injury interfering with neural communication between brain areas) and dysregulated electroencephalography (EEG) patterns (eg, increased theta activity and decreased beta activity)14 -16 following brain damage are strongly associated with deficits in neurocognitive skills, including those related to learning and memory, attention, and information processing speed. Conventional surface neurofeedback (NFB) targets and alters dysregulated brain functioning by providing real-time feedback on EEG activity through auditory or visual cues to patients. Theta/beta NFB, considered the gold standard of NFB, trains patients to decrease activity in the theta (θ) frequency band and concomitantly increase activity in the beta (β) frequency band (ie, a θ↓β↑ protocol) to improve cognitive control deficits. 17 Beta and theta band fluctuations have been associated with memory and attention. 18
A previous study introduced the technique of low-resolution electromagnetic tomography analysis (LORETA) Z-score NFB (LZNFB); this technique involves a greater number of electrodes (19-channel scalp sensors) during treatment and achieves faster effective results than conventional surface NFB. 19 LORETA NFB facilitates training aimed at improving the stability and regulation of specific dysregulated anatomical structures in the deep brain (ie, the anterior cingulate gyrus and the dorsolateral prefrontal cortex)20 -22 that are associated with the patient’s symptoms, such as cognitive deficits. LZNFB provides real-time comparisons with a reference database of Z-scores based on age-norm means and trains participants to approach Z = 0.23,24 Studies have indicated that conventional NFB (eg, theta/beta NFB) and LZNFB benefit cognitive recovery in survivors of TBI25 -29; however, the generalizability and practicability of the techniques proposed by those studies remain unclear because of single-group assignment, pre–post study designs, and small sample sizes.26 -28 In addition, those studies have failed to determine whether both NFB protocols can increase productive activity and quality of life.
In this RCT the effects of LZNFB and theta/beta NFB on cognitive impairment, particularly memory, attention, and information processing speed, in patients with TBI in the chronic phase. We also evaluated whether both NFB modules promote the return to productive activity and a higher quality of life. We hypothesized that the patients who underwent LZNFB or theta/beta NFB treatment would experience greater improvements in memory, attention, and information processing speed, and higher productive activity and quality of life compared with the patients who received usual care (UC).
Materials and methods
Study Design
This 3-arm, parallel group, assessor-blinded RCT employed convenience sampling. Data were collected from January 2018 to October 2021. The study was approved by the Joint Institutional Review Board of Taipei Medical University (N201704027) and was conducted in accordance with the guidelines of the Consolidated Standards of Reporting Trials. 30 This trial was prospectively registered at ClinicalTrial.gov (registration number: NCT03515317).
Participants and Setting
The present RCT recruited participants from neurosurgical clinics and rehabilitation departments in 3 medical centers and 2 teaching hospitals in northern Taiwan. The interventions were conducted in a biobehavioral laboratory at a university in northern Taiwan. This study included participants who met the following criteria: (1) being between 20 and 65 years of age; (2) having a diagnosis of TBI at least 6 months (chronic phase) before enrollment and a Glasgow Coma Scale score of 15 upon enrollment; (3) having difficulties in cognitive performance (eg, deficits in attention, memory, and information processing speed) combined with impairment in daily functioning (eg, forgetting to turn off faucets), as assessed by a neurosurgeon; and (4) having the ability to complete cognitive tasks (ie, Rancho Los Amigos Scale score >9), as evaluated by a trained nurse. 31 This study excluded patients with prior diagnosis of seizures, sleep disorders, psychiatric diseases, or substance abuse disorders. Furthermore, this study excluded patients who were pregnant; in menopausal transition; or with vision, hearing, or motor function impairment that was severe enough to preclude participation in the study. In addition, this study excluded patients who received cognitive-related rehabilitation and therapy prior to the study.
This study employed G*Power 3.1 software to determine the minimum sample size required to achieve a significance level of .05, a power of 0.80, and an effect size of 0.318. 32 The effect size was estimated based on the study results of Tinius et al, 33 who reported the effects of NFB on attention (posttest means for NFB and control groups = 97.1 and 104.3, respectively). 33 Based on the assumption of a potential dropout rate of 10%, the required sample size was determined to be 96.
Randomization, Allocation Concealment, and Blinding
We employed permuted block randomization to assign eligible participants to the LZNFB, theta/beta NFB, or UC groups at a 1:1:1 ratio. A computer-generated randomization list was generated by an independent statistician, which was then maintained in sealed, opaque envelopes with consecutive numbers. The data assessors were blinded to treatment allocation until the end of the study.
Interventions
Both LZNFB and theta/beta NFB training stimuli were administered using videos selected by patients from the BrainMaster BrainAvatar software system (BrainMaster Technologies; Bedford, OH, USA). When the selected video played, EEG data were recorded by the BrainMaster Discovery 24E amplifier using a 19-lead cap in accordance with the standard approach of the international 10 to 20 system, with a linked ears reference and the midline front-parietal zone as a ground electrode. 34 After fitting the cap on the head, the impedance between individual electrodes and each ear was required to be <5 kΩ. The data were collected and stored with a bandpass filter of 0.5 to 64.0 Hz and a sampling rate of 256 Hz. We implemented ten 60-minute sessions, as recommended by previous studies26,27,33 and existing protocol. 35 The participants assigned to the 2 NFB groups received weekly 60-minute training sessions (6 predesigned animated games of 10 minutes each, with a 5- to 10-minute break between games) over a 10-week period in a comfortable and sound-attenuated room.
LZNFB training protocol
We designed the protocol with the aim of improving cognitive function by training individuals to achieve a power activity of 14 to 18 Hz (low-beta) in a 7-voxel cluster of neurons in the anterior cingulate gyrus. Given the possible combinations of metrics and electrode pairs, this study calculated 5700 EEG metrics, which were continually compared in real-time to a reference database (Applied NeuroScience BrainMaster Z-Score Dynamic Link Library, Applied Neuroscience, Largo, FL, USA) to derive Z-scores. When the patients’ EEG activity achieved normative values as defined by prespecified criteria, they received positive feedback from visual or audio cues in the video. The prespecified criteria included the following: (1) Z-scores remained within a target window of ±1.5 standard deviations of the normative mean, and (2) a certain percentage of the 5700 Z-scores was manually adjusted to fall within the target window 60% to 70% of the time. We maintained a reward rate of 60% to 70% for 24 to 36 rewards per min during each session.
Theta/beta NFB protocol
The electrodes were placed at the Fz and Cz sites with a linked ears reference (A1), and the training interfaces of 10 different games were presented on a computer screen. All games displayed the patients’ response scores, which were synchronized with auditory feedback whenever the task conditions were met to simultaneously (1) increase the beta power (13-20 Hz) and (2) inhibit the theta power (4-8 Hz) relative to the baseline values determined at the beginning of a training session.
UC: The participants allocated to the UC group received standard care according to their clinical conditions (ie, clinic visit and pharmacological therapies) and maintained their usual daily activities. The UC group received no NFB training; this was done to control for practice effects due to repeated testing and the time effect on cognitive function recovery (ie, spontaneous cognitive recovery). A trained research assistant with 5 years of nursing experience caring for patients with TBI interviewed participants in the UC group weekly by phone to monitor their compliance.
Measures
The primary outcome in the current study was cognitive function (ie, attention, memory, and information processing speed), and secondary outcomes included the ability to return to productive activity and quality of life. Potential confounding factors were assessed using a predesigned information sheet.
Primary Outcomes
The Ruff 2 and 7 test was implemented to analyze selected aspects of visual attention. The participants were instructed to cross out the digits 2 and 7 under two distractor conditions. 36 This study employed the raw scores of speed and accuracy in automatic detection and controlled search trials to detect selective attention. One study indicated the high reliability (test–retest: 0.73-0.98) and discriminant validity of the Ruff 2 and 7 test with respect to age. 37
The Rey Complex Figure Test (RCFT) was employed to assess visual memory function. The participants were instructed to copy a complex geometric figure and then reproduce the figure immediately (immediate recall) and at 30 minutes after the first test (delayed recall) without warning. The maximum score of the RCFT is 36 38 for both the immediate and delayed recall trials, with higher scores indicating better visual memory function. Studies have indicated that the RCFT has high inter- and intra-rater reliability (0.85-0.97) and validity.39,40
The Symbol Digit Modalities Test (SDMT), a brief test of information processing speed, is one of the most common cognitive tasks administered to TBI survivors. 41 The participant is instructed to write the number corresponding to the symbol indicated in the key as rapidly as possible within 90 seconds. Higher scores indicate better performance. 42 One study indicated that the SDMT had a test–retest reliability of 0.70 43 and a concurrent validity (with the Digit Symbol test) of 0.78. 44
Secondary Outcomes
The Community Integration Questionnaire-revised (CIQ-R) is an 18-item ordinal scale designed to assess the respondent’s home and family integration, social integration, and productive activity. Each item is scored on a scale of 0 to 2, with higher scores indicating a greater level of independence. Studies have established the reliability and validity of the CIQ-R.45,46 The results indicated high reliability of the Chinese version of the CIQ-R in our study population (Cronbach’s α = .91).
The Quality of Life after Brain Injury (QOLIBRI) is a disease-specific instrument that assesses quality of life after TBI. 47 It comprises 37 items, with each item scored on a 5-point Likert scale. Higher scores indicate a higher quality of life overall. The QOLIBRI has high reliability and validity. 48 The Chinese version of the QOLIBRI, which was translated in 2014, also boasts excellent reliability (internal consistency: 0.79-0.95) and construct validity. 49
Potential Confounding Factors
Data on demographic and disease characteristics were collected using a predesigned information sheet. This study assessed potential risk factors reported in relevant studies, including age, sex, loss of consciousness upon injury, initial Glasgow Coma Scale score, and lesion locations.50,51 In addition, information on body mass index, marriage, employment status, time since injury, cause of TBI, and comorbidities were collected.
Statistical Analyses
All data were analyzed using SPSS 22.0 for Windows (IBM, Armonk, NY, USA), and statistical significance was indicated at P < .05. We assessed differences among the 3 groups with respect to demographic and disease characteristics, cognitive function, return to productive activity, and quality of life using the chi-square test and one-way analysis of variance.
Our study measured multiple nonindependent observations (ie, cognitive tasks, return to productivity, and quality of life) from identical participants, requiring us to account for intraindividual correlation. Unadjusted analysis may result in standard error misestimation, thus causing inaccurate estimations of treatment effects. 52 This study implemented a generalized estimating equation (GEE) model adjusted for multiple comparisons using the Bonferroni method to account for nonindependence. GEEs can be used to assess data with missing values; they yield robust parameter estimates even if the missing values are completely random. Although GEEs and linear mixed models (LMMs) share similarities with respect to statistical estimation, GEEs are employed to explore overall average effects, whereas LMMs center on subject-specific effects. 53
We selected a GEE model to examine the intervention effects on the participants in the 2 NFB groups compared with those in the UC group over time, adjusting for potential confounding variables. The value of the quasi-likelihood under the independence model criterion was employed to select the best correlation structure. 54 First, the main and interaction effect terms for both the time and group factors were included in the GEE model for each outcome. Subsequently, we assessed the significance of the time by group interaction for each outcome. We reported estimated coefficients and corresponding 95% confidence intervals for both the main effects and the interaction effects when the results indicated significant effects of time by group interaction for certain outcomes. Each group by time interaction term reflected the difference between the change of outcome from the baseline to posttest time in the corresponding intervention group and the change of outcome from the baseline to posttest time in the UC group. Therefore, a significant interaction term indicated a significant change in outcome induced by the intervention in the experimental group compared with that in the UC group. Potential confounding variables were included as control variables if their p values were < 0.25 among the 3 groups at baseline.
All analyses were conducted according to the intention-to-treat (ITT) principle. Due to study attrition, incomplete responses to questionnaires, and missing data, this study implemented multiple imputations of missing values to ensure feasible ITT analyses. We employed post-hoc power calculations 55 using G*Power 3.1 software 32 after determining an effect size estimated from the degree of variation explained by the predictors, a 2-tailed α of .05, and the number of predictors. To test the robustness of our findings, sensitivity analyses were performed by excluding patients with severe head injury (Glasgow Coma Scale score between 3 and 8).
Results
Sample Characteristics
Although 159 participants met the inclusion criteria for this study between January 2018 and October 2021, only 87 provided consent for participation and were thus randomly assigned into the LZNFB, theta/beta NFB, and UC groups (n = 29, 31, and 27, respectively). We were unable to enroll the original estimated sample size (n = 96) due to the COVID-19 pandemic. Of the 87 individuals, 7, 8, and 2 patients in the respective groups did not participate due to scheduling conflicts or relocation (Figure 1). Thus, we completed posttreatment data analyses in accordance with the ITT principle. All patients’ pharmacological therapies (eg, surgery-related therapy) remained constant for at least 3 months prior to the study and throughout the intervention; none of the patients received physical or cognitive therapy.
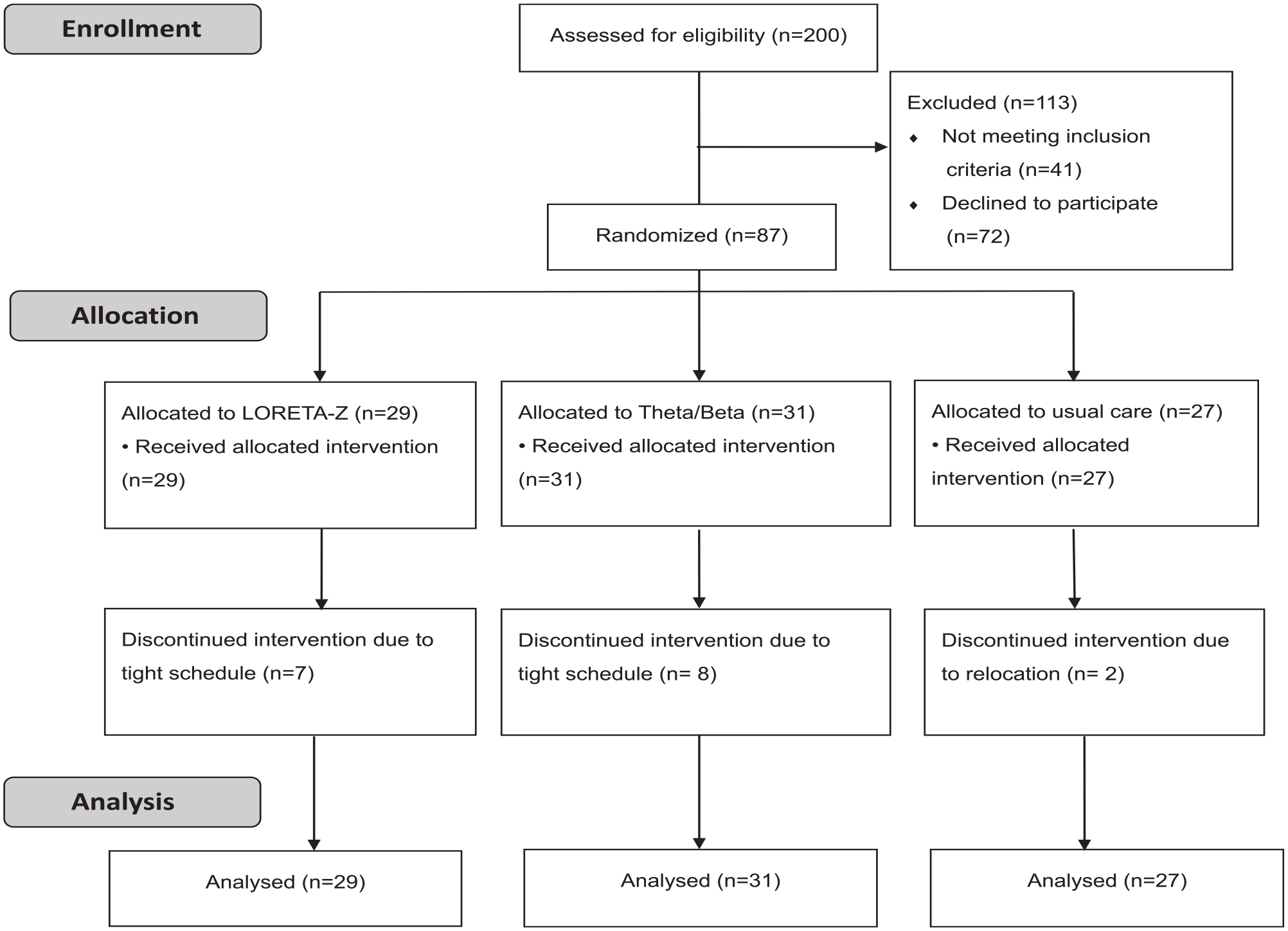
Study flow.
Table 1 presents the clinicodemographic characteristics of the 87 participants. The mean ages were 47.55, 40.52, and 42.81 years for the LZNFB, theta/beta NFB, and UC groups, respectively. The results did not indicate any significant intergroup differences in baseline clinicodemographic characteristics. Baseline age, initial loss of consciousness, and comorbidity status were included in the adjusted GEE model.
Demographic and Disease Characteristics (n = 87).
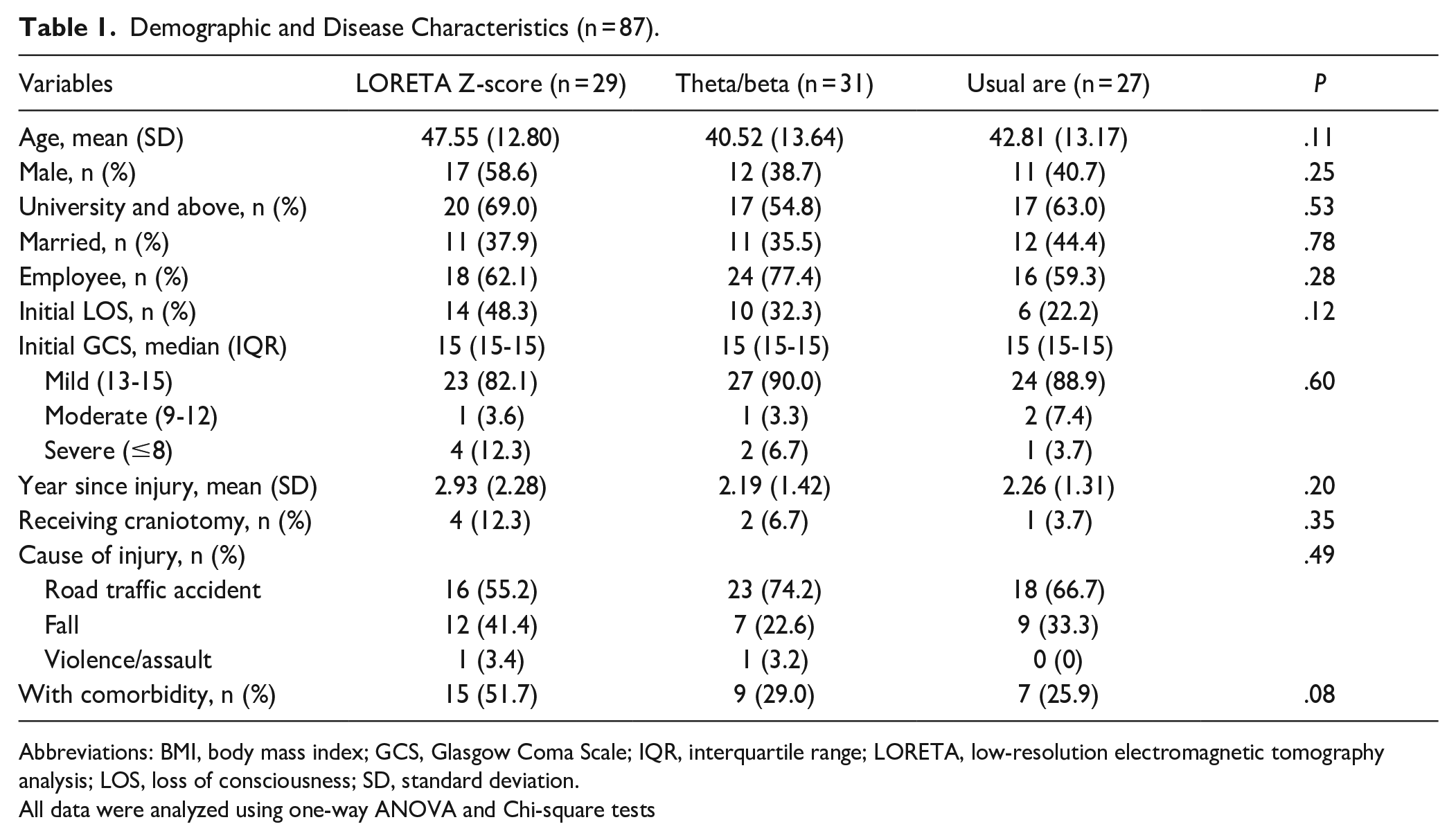
Abbreviations: BMI, body mass index; GCS, Glasgow Coma Scale; IQR, interquartile range; LORETA, low-resolution electromagnetic tomography analysis; LOS, loss of consciousness; SD, standard deviation.
All data were analyzed using one-way ANOVA and Chi-square tests
Treatment Effects on Memory, Attention, and Information Processing Speed
The results of the mean score distributions and regression coefficient estimations of the GEE model for each cognitive parameter are presented in Table 2. The LZNFB group exhibited significant improvements in RCFT scores for immediate and delayed recall and recognition memory relative to the UC group; the theta/beta NFB group exhibited a significant improvement in RCFT scores for immediate recall after treatment completion compared with those of the UC group. The results indicated significant improvements in the scores of the Ruff 2 and 7 tests for automatic detection accuracy and controlled search accuracy in the LZNFB and theta/beta NFB groups compared with those of the UC group from baseline to the 10th week. No significant difference in SDMT scores was indicated between the intervention and control groups at the 10th week.
Results of Generalized Estimating Equations in the Effects of Neurofeedback on Cognitive Parameters Over the Study Period (n = 87).
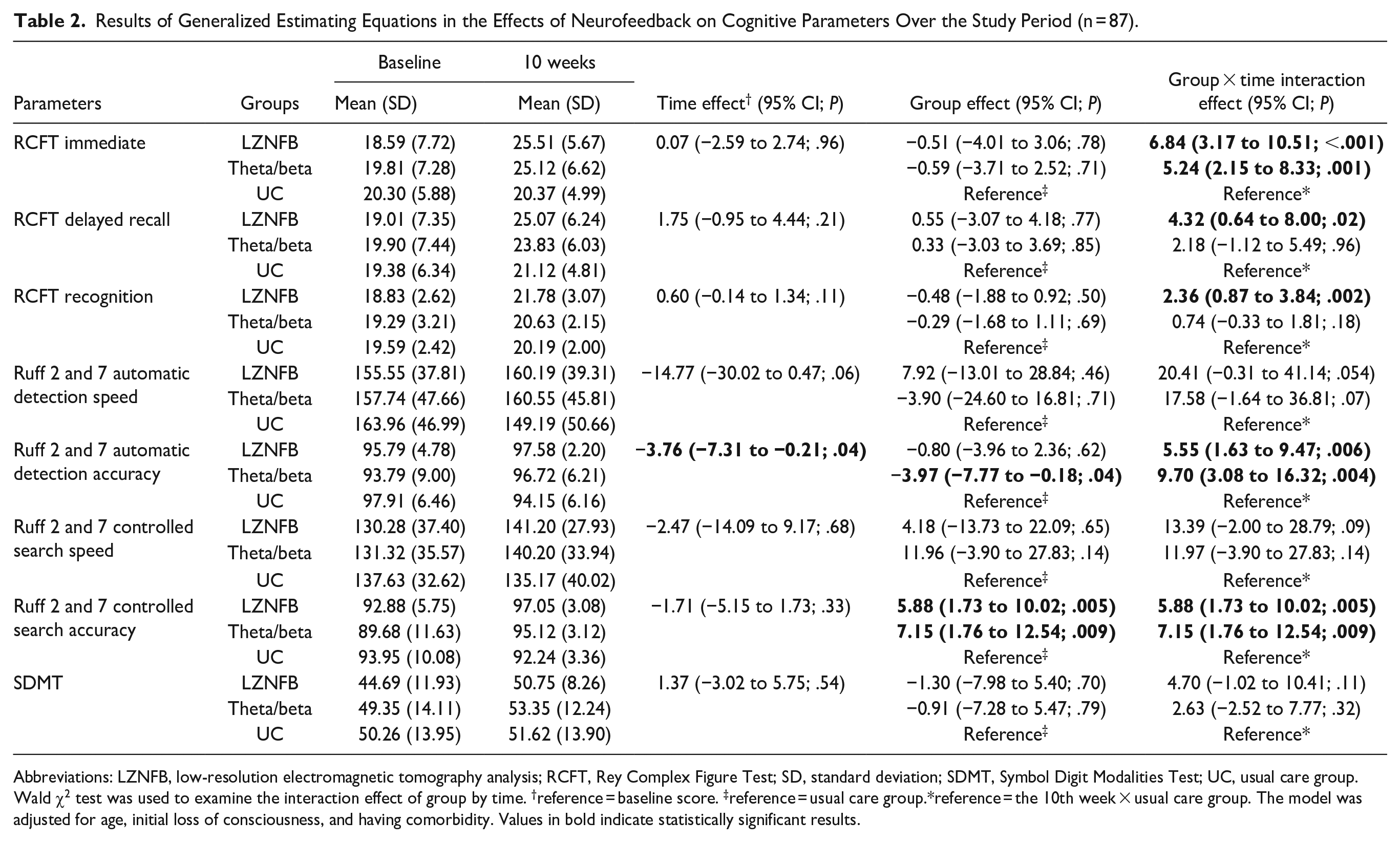
Abbreviations: LZNFB, low-resolution electromagnetic tomography analysis; RCFT, Rey Complex Figure Test; SD, standard deviation; SDMT, Symbol Digit Modalities Test; UC, usual care group.
Wald χ2 test was used to examine the interaction effect of group by time. †reference = baseline score. ‡reference = usual care group.*reference = the 10th week × usual care group. The model was adjusted for age, initial loss of consciousness, and having comorbidity. Values in bold indicate statistically significant results.
Treatment Effects on Ability to Return to Productive Activity and Quality of Life
Table 3 summarizes the treatment effects on CIQ-R and QOLIBRI total scores for the GEE model. After treatment completion, the LZNFB group had a greater improvement in total CIQ-R scores but not in total QOLIBRI scores compared with those of the UC group (Table 3).
Results of Generalized Estimating Equations in the Effects of Neurofeedback on Cognitive Parameters Over the Study Period (n = 87).
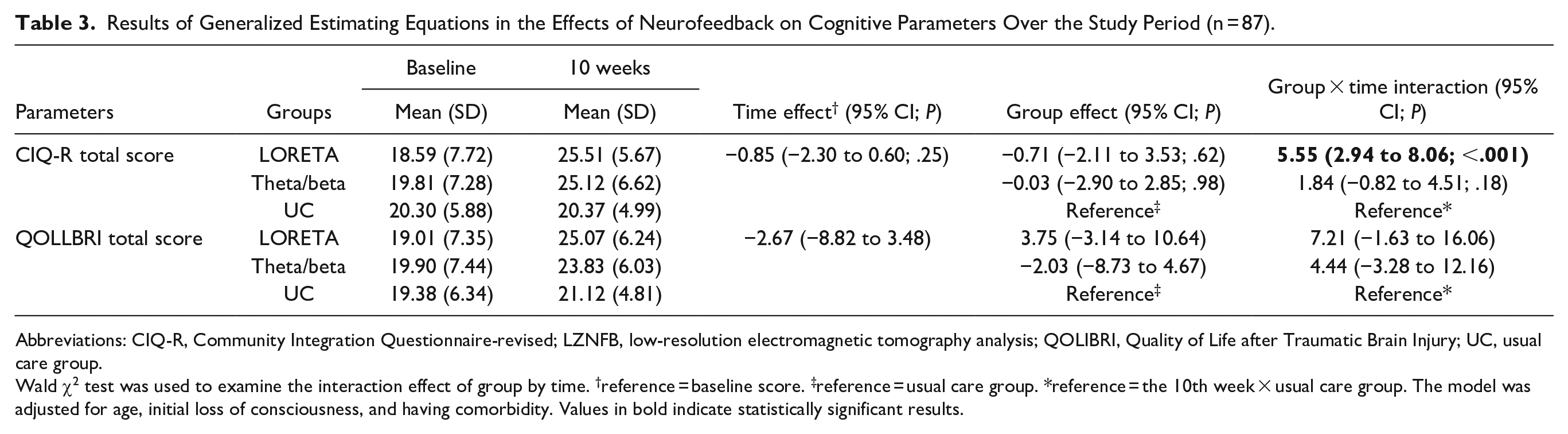
Abbreviations: CIQ-R, Community Integration Questionnaire-revised; LZNFB, low-resolution electromagnetic tomography analysis; QOLIBRI, Quality of Life after Traumatic Brain Injury; UC, usual care group.
Wald χ2 test was used to examine the interaction effect of group by time. †reference = baseline score. ‡reference = usual care group. *reference = the 10th week × usual care group. The model was adjusted for age, initial loss of consciousness, and having comorbidity. Values in bold indicate statistically significant results.
Power Analysis and Sensitivity Analysis
The post-hoc power analysis, which had a 2-tailed α of .05, a measurement number of 2, and an effect size of 0.57 derived from posttest data of attention search accuracy, yielded excellent statistical power of 0.99. In terms of primary and secondary outcomes, findings from sensitivity analyses by excluding patients with severe head injury remained similar (Supplemental Tables 1 and 2).
Discussion
The present study revealed that LZNFB improved immediate recall, delayed recall, and recognition memory; moreover, theta/beta NFB enhanced immediate memory function. TBI survivors who received either NFB treatment exhibited substantially enhanced selective attention, but not information processing speed, immediately after treatment completion compared with those who received UC. In addition, the LZNFB group exhibited a greater ability to return to productive activity than the UC group. The results jointly revealed that, compared with the UC program, the LZNFB program had beneficial effects on immediate and delayed recall, recognition memory and attention, the ability to return to productive activity, and quality of life, whereas the theta/beta NFB program merely improved immediate memory and selective attention.
Consistent with the results of a study enrolling 45 patients with TBI that employed LZNFB to enhance cognitive impairment, 27 our results indicated that LZNFB substantially improved immediate recall, delayed recall, and recognition memory and selective attention, whereas the theta/beta conventional surface protocol enhanced only immediate memory and selective attention after 10 training sessions. The advanced approach—19-channel LZNFB—may possess advantages over conventional surface NFB (eg, theta/beta or sensorimotor rhythm) 56 due to its potential ability to target the cognitive division of the anterior cingulate gyrus, resulting in improved attention and memory 57 and fewer required sessions for effective intervention. 58 These advantages may explain why patients in our study who underwent 10-session LZNFB treatment exhibited improvement in immediate recall, delayed recall, and recognition memory, whereas those who underwent 10-session theta/beta NFB treatment exhibited improvement only in immediate memory. Although this study demonstrated the beneficial effects of LZNFB, this treatment should be administered by certified professions in a neuroscience laboratory, which limits its clinical application and feasibility.
Functioning and quality of life are key indicators of the quality of a rehabilitation program. Cognitive deficits following TBI impair effective job performance due to difficulty focusing on and attending to job tasks, 59 which undermines the ability to return to productive activity and enjoy a high quality of life. 60 Our findings revealed that significant improvements in memory and attention following 10 sessions of LZNFB may further enhance the return to productive activity in patients with TBI; however, the results did not demonstrate significant effects on quality of life. Multiple factors contribute to quality of life after TBI, including age, the time since injury, prior employment status, injury location, and dependence degree,61,62 which may explain the nonsignificant effect on quality of life.
This study has several limitations. First, all enrolled participants were from hospitals in northern Taiwan, which may restrict the generalizability of the findings. Second, the study population included TBI survivors with varying severities of head injuries (approximately 90% had mild TBI, and 10% had moderate-to-severe TBI), which may have affected the internal and external validity of our findings. Third, this study did not collect data on psychological confounding variables, such as depression and anxiety, that are associated with the return to productive activity 63 and quality of life, 64 which may influence interpretations of the beneficial effects of LZNFB. Future studies should address these limitations. Fourth, this study did not investigate several covariates (eg, psychiatric and sleep disturbance symptoms) that may influence cognitive function.65,66 Therefore, these findings should be interpreted with caution. Lastly, the control group design is not structurally equivalent to that of the 2 intervention groups with respect to time and attention from the care team; therefore, this study may overestimate the therapeutic effect of NFB.
Conclusions
This RCT is the first to demonstrate the beneficial effects of LZNFB and theta/beta NFB on cognitive recovery in patients with TBI. The LZNFB group experienced significantly enhanced immediate memory recall, delayed recall memory, and selective attention compared with those of the UC group, whereas the theta/beta NFB group experienced enhanced immediate memory function and selective attention compared with those of the UC group. Moreover, TBI survivors receiving LZNFB but not theta/beta NFB exhibited a substantially enhanced ability to return to productive activity. We recommend for first-line health-care providers to integrate either LZNFB or theta/beta NFB into cognitive rehabilitation programs for patients with TBI.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231170539 – Supplemental material for Effects of Neurofeedback on Cognitive Function, Productive Activity, and Quality of Life in Patients With Traumatic Brain Injury: A Randomized Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683231170539 for Effects of Neurofeedback on Cognitive Function, Productive Activity, and Quality of Life in Patients With Traumatic Brain Injury: A Randomized Controlled Trial by Pin-Yuan Chen, I-Chang Su, Chun-Ying Shih, Yen-Chun Liu, Yu-Kai Su, Li Wei, Hui-Tzung Luh, Hui-Chuan Huang, Pei-Shan Tsai, Yen-Chun Fan and Hsiao-Yean Chiu in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Ministry of Science and Technology of Taiwan (MOST 106-2314-B-038-058-MY3 and MOST 111-2628-B-038-008).
Data Availability Statement
Data are available on request from the authors.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
