Abstract
Background
An artificial intelligence (AI)-integrated electromyography (EMG)-driven robot hand was devised for upper extremity (UE) rehabilitation. This robot detects patients’ intentions to perform finger extension and flexion based on the EMG activities of 3 forearm muscles.
Objective
This study aimed to assess the effect of this robot in patients with chronic stroke.
Methods
This was a single-blinded, randomized, controlled trial with a 4-week follow-up period. Twenty patients were assigned to the active (n = 11) and control (n = 9) groups. Patients in the active group received 40 minutes of active finger training with this robot twice a week for 4 weeks. Patients in the control group received passive finger training with the same robot. The Fugl-Meyer assessment of UE motor function (FMA), motor activity log-14 amount of use score (MAL-14 AOU), modified Ashworth scale (MAS), H reflex, and reciprocal inhibition were assessed before, post, and post-4 weeks (post-4w) of intervention.
Results
FMA was significantly improved at both post (P = .011) and post-4w (P = .021) in the active group. The control group did not show significant improvement in FMA at the post. MAL-14 AOU was improved at the post in the active group (P = .03). In the active group, there were significant improvements in wrist MAS at post (P = .024) and post-4w (P = .026).
Conclusions
The AI-integrated EMG-driven robot improved UE motor function and spasticity, which persisted for 4 weeks. This robot hand might be useful for UE rehabilitation of patients with stroke.
Introduction
Stroke is a leading cause of disability in adults in many countries. Each year, approximately 7 95 000 people experience a new or recurrent stroke in the United States. 1 The prevalence of stroke is expected to increase as the population ages, and nearly 4% of the United States population is projected to have had a stroke by 2030. 2 Hemiparesis of the upper extremity (UE) is a common problem in stroke patients. More than two-thirds of people have UE paresis in the acute phase which persists in half of the population affected by this disease to 6 months after stroke.3,4 It is thought difficult to achieve a marked improvement in motor function for more than a few months after onset.5,6 UE hemiparesis has a significant impact on the performance of activities of daily living (ADL). It is particularly important to acquire a grip-and-release and pinch-and-release to use the paretic hand in ADL. 7
There are many rehabilitation treatments to improve UE motor function, such as occupational therapy, constraint-induced movement therapy, neuromuscular electrical stimulation (NMES), functional electrical stimulation, non-invasive brain stimulation (NIBS), robotics, and brain–computer interface (BCI).8,9 There are, however, few ways to improve hand function in patients with severe hemiparesis. In recent years, many types of UE rehabilitation robots have been developed. One systematic review suggested that robotic devices improved arm function, but not hand function. 10
Rehabilitation robots can be divided into active, passive, and active-assisted robots. 11 Active-assisted robots support patients’ movements by detecting the intention of actions by force sensors or surface electromyography (EMG). Some EMG-driven robots can detect the intent of UE movement by EMG activity. Active-assisted robots assist the patients’ voluntary movements. Voluntary contraction combined with peripheral afferent inputs has been shown to increase corticospinal excitability. 12 Assisted voluntary movements are expected to induce more neuroplasticity than passive movements. 13 For UE robotic rehabilitation after stroke, it has been suggested that active-assisted devices are more beneficial than passive devices. 14 However, few studies have compared the therapeutic effects of passive and active-assisted robots for post-stroke UE hemiparesis. There were some EMG-driven NMES and robotic devices.15,16 These EMG-driven, active-assisted devices require detecting the joint movements or EMGs of the target muscles. The EMG-driven robots detect motor intent when an EMG signal above a predetermined threshold is obtained in a particular muscle. 17 Therefore, they are often not indicated for patients with severe hemiparesis or severe spasticity, because they may not correctly determine the movements’ intent.
Recently, an artificial intelligence (AI)-integrated EMG-driven robot hand for UE rehabilitation has been developed by MELTIN MMI (Tokyo, Japan). It is an exoskeletal robotic hand that assists the patient’s finger movement by determining movement intention from the EMG patterns of the forearm muscles. By determining the movement intention from the muscle activity of the entire forearm, rather than the activity of specific flexor or extensor muscles, this robot can discriminate the cross-talk of antagonist and agonist EMG activities and exclude the effect of spasticity. Therefore, this robot can be applied to patients with severe hemiplegia and spasticity.
Using the newly developed AI-integrated EMG-driven robot hand, the effects on hand function of the active-assisted robot and the passive robot were compared in patients with moderate to severe UE hemiparesis.
Methods
Study Design and Participants
This single-blinded, randomized trial had a parallel design that conformed with CONSORT 2010 (Supplemental Table S1). Participants were recruited from the outpatient clinic of the Department of Rehabilitation Medicine of Juntendo University Hospital. The inclusion criteria of this study were as follows: (a) first-time unilateral supratentorial stroke; (b) time from stroke onset longer than 60 days; (c) age 20 to 80 years; (d) patients who could not move their paretic fingers individually (stroke impairment assessment set (SIAS) 12 finger function test score ≤2); (e) patients who could raise their paretic hand to the height of their nipple (SIAS knee mouth test score ≥2); 18 (f) no severe proprioceptive deficit in the affected UE; (g) modified Ashworth scale (MAS) 19 of the paretic finger 2 or less; and (h) in participants treated with botulinum toxin, more than 2 months passed since injection.
The exclusion criteria were as follows: (a) severe cardiac disease; (b) uncontrolled hypertension; (c) acute illness and fever; (d) recent medical history of pulmonary embolism, acute cor pulmonale, or severe pulmonary hypertension; (e) severe liver failure or renal failure; (f) orthopedic complication preventing exercise; (g) severe cognitive impairment or mental illness; and (h) another metabolic disease.
Participants were divided into the AI-integrated EMG-driven robot hand (active) group and the passive robot (control) group. The intervention was assigned to individuals randomly through a randomly permuted block technique with a block size of 6. The intervention used in this study was performed according to a randomized list prepared by an individual who was not part of the study. All participants provided written, informed consent according to a protocol approved by the local institutional review board. The study was performed according to the Declaration of Helsinki, was approved by the Juntendo University Hospital Ethics Committee (J20-001), and was registered in the Japan Registry of Clinical Trials (jRCT, registration number: jRCTs032200045, URL: https://jrct.niph.go.jp/).
Intervention
AI-Integrated EMG-Driven Robot Hand (active) Group
Figure 1 shows the newly developed, AI-integrated EMG-driven robot hand. Patients wore the wrist splint, which stabilized the wrist and had an attachment to the robot hand. The hand parts of the robotics were made of plastic and weighed 500 g. The length of each finger part was adjustable for each finger length and adjusted to each finger joint (metacarpophalangeal (MP) joint, proximal interphalangeal (PIP) joint, and distal interphalangeal (PIP) joint). Three pairs of surface electrodes (30 mm × 50 mm) were placed on the paretic flexor digitorum superficialis (FDS), flexor digitorum profundus (FDP), and extensor digitorum communis (EDC). These EMG electrodes can detect EMG amplitude as low as 20 nv.
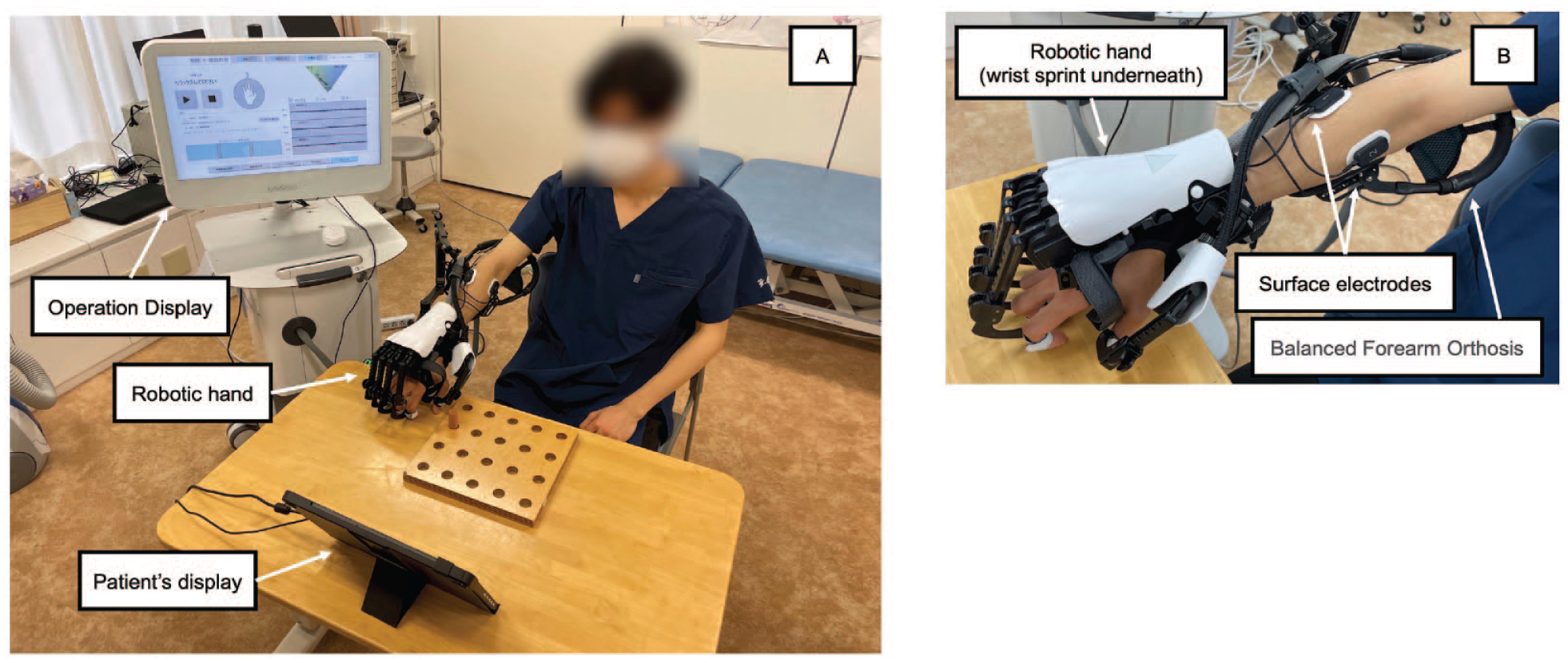
AI-integrated EMG-driven robot. (A) Overview of the robot: the main unit with operation display, patient’s display, and robotic hand. (B) The enlarged view around the robotic hand. The length of each finger part is adjustable for each finger length. Each finger part is connected to the main unit via wires, and its operation is controlled. Three pairs of surface electrodes are placed on the paretic FDS, FDP, and EDC. A balanced forearm orthosis is used to support the patient’s forearm.
The AI-integrated EMG-driven robot hand flexes and extends the paretic fingers according to 3 EMG (FDS, FDP, and EDC) activities. In the active group, the AI-integrated EMG-driven robot hand was used for UE rehabilitation twice a week, 40 minutes per training session, for a total of 8 sessions. Before each training session, EMG activity patterns of the patient’s finger movements, such as relaxing, finger extension, and finger flexion, were calibrated. AI identified a patient’s intention of finger movement based on the EMG activity patterns of the 3 muscles. Then, a robotic hand supported the patient’s finger movement. The patients were trained in grip-and-release and pinch-and-release actions under the guidance of the physical therapist. Using the AI-integrated EMG-driven robot hand and arm support, patients were asked to pick up and put down the peg and take the cue ball and move it to the designated location.
Passive Robot (Control) Group
The control group performed passive finger exercises provided by the same robot as the active group. In the control group, the robot passively flexed and extended its fingers. Robot hand movement was not triggered by the patient’s EMG activities. The timing of finger flexion and extension was operated by the physical therapist. The control group received the same training as the active group.
Outcome Measures
Clinical Assessments
UE motor function, spasticity, amount of use of the paretic hand, and electrophysiological assessments were assessed before, immediately after (post), and 4 weeks after intervention (post-4w). As a primary outcome, UE motor function was assessed with the Fugl-Meyer Assessment UE motor score (FMA). 20 The FMA consists of 4 categories: (A) shoulder/elbow/forearm (FMA-A); (B) wrist (FMA-B); (C) hand (FMA-C); and (D) coordination/speed (FMA-D). The FMA includes 33 items, and the maximum score is 66. Spasticity was measured with the MAS of the fingers, wrist, and elbow. 19 The amount of use scale of the Motor activity log-14 scale (MAL-14 AOU) was used to assess the amount of use of the paretic UE in ADL. 21 All clinical assessments were scored by a masked examiner, who did not know which patients were assigned to which group.
Electrophysiological Assessment
The H reflex and reciprocal inhibition (RI) were studied in the paretic forearm. 22 With the patients seated and relaxed, H reflexes were elicited from the paretic flexor carpi radialis (FCR) by submaximal electrical stimulation of the median nerve at the antecubital fossa with a 1-ms square-wave constant current. The reflex responses were measured as the peak-to-peak amplitude of the H reflex recorded by a bipolar disc electrode placed over the FCR muscle. RI was assessed using an FCR H reflex conditioning-test paradigm. Ten conditioned and 10 test H reflexes were averaged at each time point. The test FCR H reflex amplitude was maintained at 15% to 20% of the maximal M wave amplitude (M max) for each block trial. Conditioning stimulation to the radial nerve was delivered at the spiral groove. The stimulus intensity of the conditioning stimulation was 1.0 motor threshold (MT). The MT was defined as a 100-μV response of extensor carpi radialis. The conditioning-test stimulus interval was set at 0 and 20 ms.
Data Analysis
To calculate the mean value of the MAS score, score 1+ was transformed to 2, and scores 2 and 3 were transformed to 3 and 4. The results are expressed as mean–standard deviation. Statistical analysis was performed primarily to determine the efficacy of this robot in chronic stroke patients, and data analysis was performed on an intention-to-treat basis. The Mann–Whitney U test was used to compare nonparametric and parametric data between the 2 groups, and the χ2 test was used to compare nominal data.
Normality was assessed with the Shapiro–Wilk test. When normality was confirmed, 2-way analysis of covariance (ANCOVA), using the baseline measure as the covariate, with the factors group (active and control) and time (before, post, and post-4w assessment), was performed to investigate whether the AI-integrated EMG-driven robot can improve UE motor function (FMA and MAS), daily activity (MAL-14 AOU), and electrophysical assessments (H/Mmax, RI0ms, and RI20ms) in stroke patients. The post hoc paired t-test and Wilcoxon signed-rank test were used to compare outcome measures relative to the before value. Effects were considered significant if P < .05. Statistical analyses were performed using IBM SPSS version 28.0.1 for Windows (IBM Corp., Armonk, NY, USA).
The sample size was determined based on the difference in the change in FMA scores due to each intervention between the active and control groups. In a previous study on UE function in patients with hemiplegia in chronic stroke, 23 the difference in change in the FMA scores between the active and control groups was about 2.5 ± 2 points; thus, if the clinically significant difference in improvement between these groups is about 2.5 points, the same results should be obtained. Based on a 2-sample Student’s t-test with a significance level of .05 and a power of 0.8, a sample size of 12 subjects in each group (24 subjects in total) was selected. To account for some loss to follow-up, the sample size was increased from 12 to 15 per group.
Results
Participants were recruited from the outpatient clinic of the Department of Rehabilitation Medicine at Juntendo University hospital from June 2020 to February 2022. A total of 20 participants were recruited and randomly allocated to the active (n = 11) and control (n = 9) groups. The flowchart of the experimental design is shown in Figure 2. One patient in the control group was not assessed due to an infectious disease at the post-4w assessment. Thirty participants were needed based on the sample size calculation, but it was difficult to recruit the required number of stroke patients from the outpatient clinics due to the declaration of a state of emergency related to SARS-CoV-2. No adverse events related to the intervention were seen in both the active and control groups.
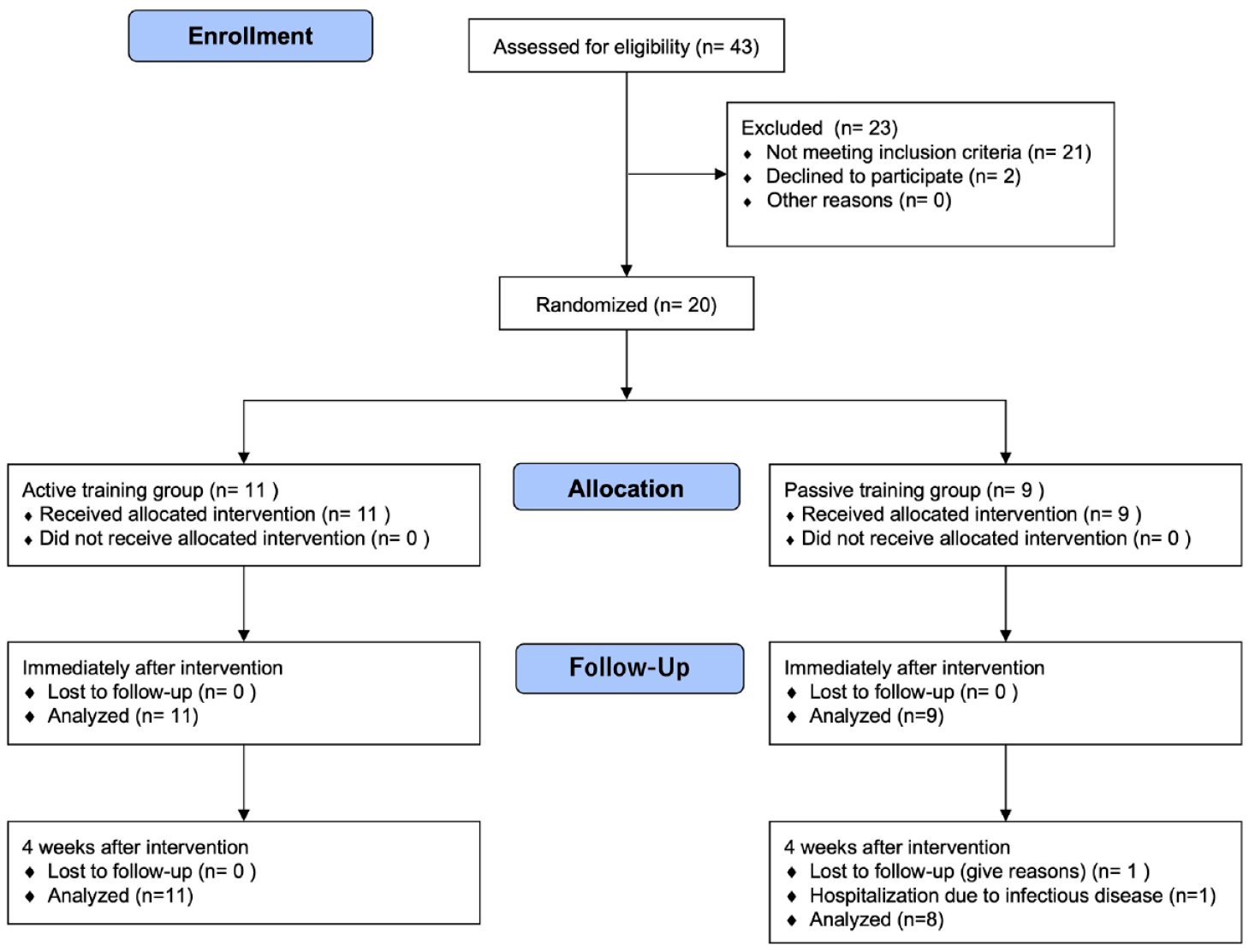
CONSORT 2010 flow diagram.
Demographic data at clinical assessment and electrophysiological data at baseline are shown in Table 1. There were no significant differences in baseline values between the active and control groups.
Demographics of Study Participants.
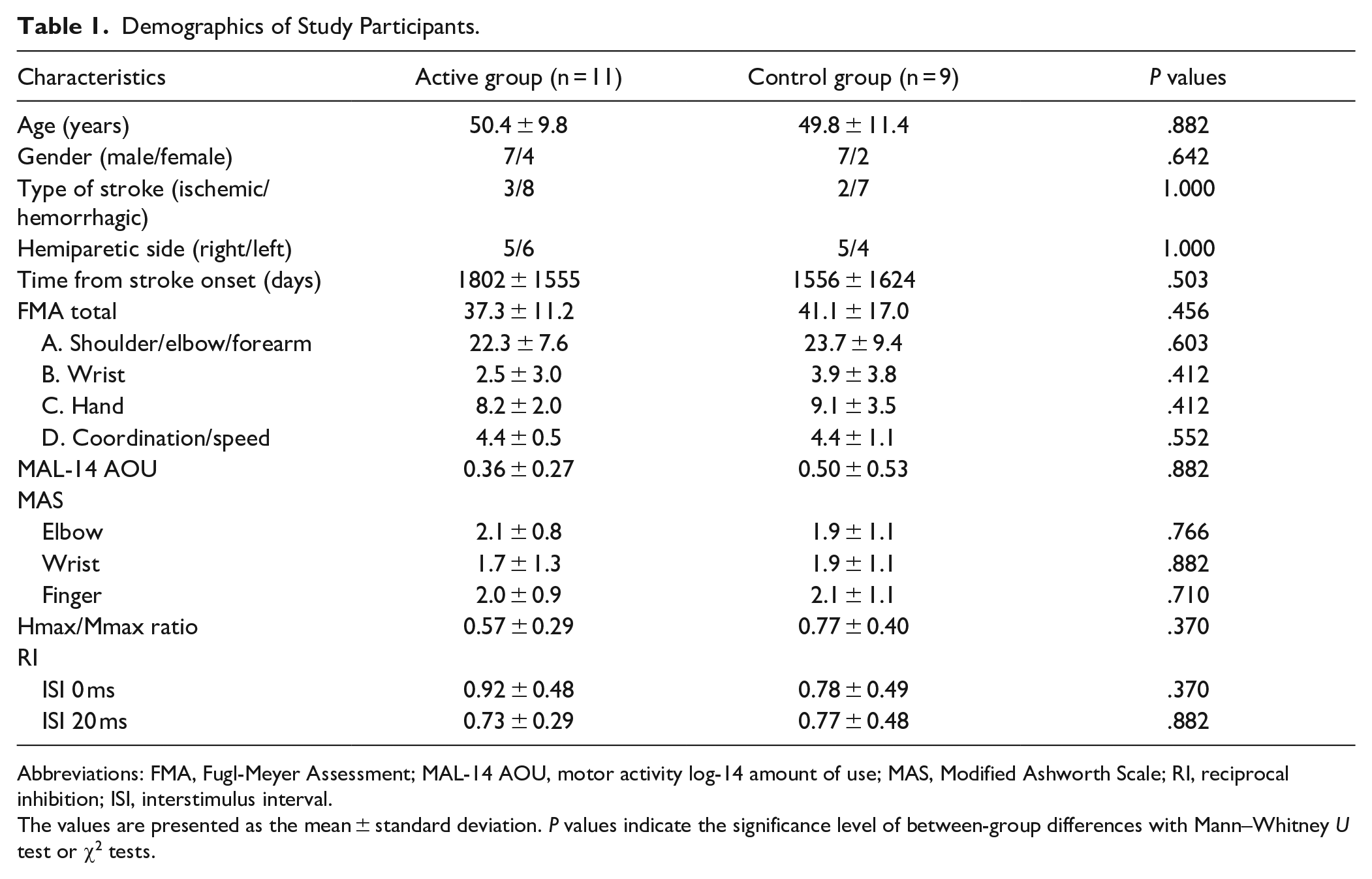
Abbreviations: FMA, Fugl-Meyer Assessment; MAL-14 AOU, motor activity log-14 amount of use; MAS, Modified Ashworth Scale; RI, reciprocal inhibition; ISI, interstimulus interval.
The values are presented as the mean ± standard deviation. P values indicate the significance level of between-group differences with Mann–Whitney U test or χ2 tests.
Table 2 shows the FMA scores before, post, and post-4w assessment. The significant main effect was observed only for the factor of the evaluation time point in the FMA scores by 2-way ANCOVA (P < .001, η2 = .946).
Before, Post, and Post-4w Scores of Outcome Measures in the Active and Control Groups.
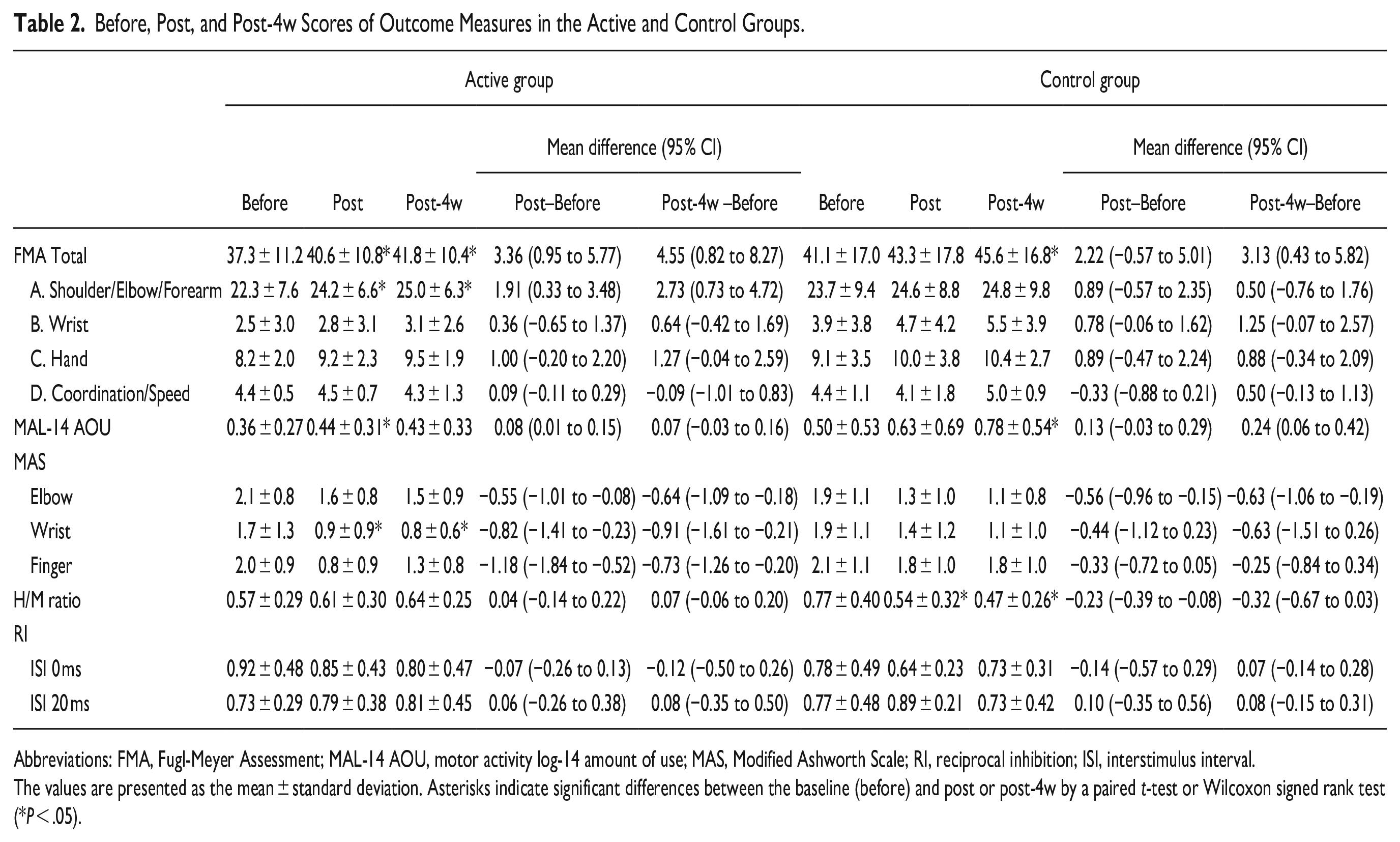
Abbreviations: FMA, Fugl-Meyer Assessment; MAL-14 AOU, motor activity log-14 amount of use; MAS, Modified Ashworth Scale; RI, reciprocal inhibition; ISI, interstimulus interval.
The values are presented as the mean ± standard deviation. Asterisks indicate significant differences between the baseline (before) and post or post-4w by a paired t-test or Wilcoxon signed rank test (*P < .05).
For the active group, FMA scores at post and post-4w were significantly improved compared to before (P = .011 and .021, respectively). In the control group, we could not find significant improvement of FMA scores between before and post. The mean difference (95% CI) of FMA score between before and post was 3.36 [0.95 to 5.77] in the active group and 2.22 [−0.57 to 5.01] in the control group. In the control group, we found a significant improvement of FMA score between before and post-4w (P = .029).
To test the recovery of the upper limb following the intervention in detail, the FMA sub-score was analyzed by a paired t-test. The analysis showed the FMA-A (shoulder/elbow/forearm) was significantly improved at post and post-4w (P = .022, .010, respectively), and FMA-C (hand) was marginally improved (P = .09, .06, respectively) in the active group compared to before. These results show that AI-Integrated EMG-driven robots can improve not only hand motor function, but also whole UE motor function.
The findings of secondary outcome measures are also shown in Table 2. The significant main effect of the time factor was observed in the MAL-14 AOU and the MAS of the wrist on 2-way ANCOVA (MAL; P = .019, η2 = .824, MAS; P = .003, η2 = .956). The interaction of time and group was only found in the MAL-14 AOU (P = .017, η2 = .412). For the MAL-14 AOU, in the active group, the score was significantly improved at post compared to before (P = .03), whereas the MAL score was significantly improved at post-4w in the control group (P = .02). The MAS score of the wrist was significantly improved at post and post-4w in the active group compared to before (P = .01, .02, respectively). The MAS score of the wrist was not changed in the control group.
The H/M ratio and RI were directly compared by the Wilcoxon signed-rank test due to their non-normal distributions. The H/M ratio was significantly decreased at post and post-4w in the control group compared to before (both P = .01). No changes in RI0ms and RI20ms were observed in the active and control groups.
Discussion
Robot-assisted UE training is thought to be effective and is used in neurorehabilitation of patients with stroke. 24 Various EMG-driven robots have been developed. It has been hypothesized that active-assisted, EMG-driven, robotic training is effective in improving motor function. 25 Few studies compared active-assisted hand robots and passive hand robots in chronic stroke patients. Hwang et al 26 studied the effect of 4 weeks of active-assisted robot hand training and 2 weeks of passive robot hand training followed by 2 weeks of active-assisted robot hand training. They showed that the effect of the active-assisted robot in improving UE motor function was dose-dependent. In the present study, the therapeutic effects of active hand training with the newly developed AI-integrated EMG-driven robot hand were compared with those of passive robot hand training. There were significant improvements in UE motor function, amount of use, and spasticity with the AI-integrated EMG-driven robot hand. The active training with an AI-integrated EMG-driven robot improved UE motor function better than passive robotic training. Passive robotic training improved post-4w FMA and MAL-14 AOU.
Active motor training with afferent stimulation (electrical stimulation and passive movement) induces Hebbian plasticity with the convergence of descending volleys from the motor cortex and ascending volleys from afferent nerves. Voluntary contraction combined with electrical afferent stimulation increases motor cortex excitability 12 and modulates spinal reciprocal inhibition. 27 For motor learning and improvement of hand function, active-assisted training may be practical and increase the amount of use of the paretic hand in patients with chronic stroke. 15
In the active training with assisted robotic training, activation of the motor cortex was combined with afferent stimulation of finger movement. Therefore, descending volleys from the motor cortex and ascending afferent activation were convergent and induced the Hebbian effect of the cortico-spinal pathway to the paretic fingers. For motor learning and improvement of hand function, active-assisted training may be practical and increase the amount of use of the paretic hand in chronic stroke. 15
The passive robotic hand training reduced the H/M ratio in the paretic forearm. The H/M ratio showed alfa motor neuron excitability. Passive stretching exercise is one of the most common therapeutic techniques for spasticity. 28 During passive robotic training, participants moved their arms to the target object, and the robotic hand performed their grip and release of the target object passively. Participants moved only their arms. They did not need to make an effort to move their fingers. That inhibits the increase of alfa motor neuron excitability during training. This result suggests that the robotic finger stretch exercise decreased the excitability of alfa motor neurons in the control group and may have contributed to improvement in FMA at the post-4w assessment.
Active-assisted robots need the patients’ intention to move through the torque sensors, position sensors, and EMG. 29 Torque sensors and position sensors require some degree of voluntary movement to trigger. In contrast to torque sensors and position sensors, EMG sensors can detect patients’ voluntary muscle activation in real-time. EMG-triggered robots, therefore, can be applied to a broader range of patients than torque sensor or position sensor-triggered robots.
EMG-driven robots should detect the target muscle EMG activities to trigger. However, it is difficult for patients with moderate to severe hemiparesis or spasticity to activate selective muscle activation.
BCIs were used for UE motor training in such patients. 13 Shindo et al 30 applied the BCI to patients with severe hemiparetic stroke, which detected the patients’ intention to extend their fingers by using event-related desynchronization (ERD) of the motor cortex and extended their paretic fingers with robotic devices. This BCI training improved finger motor function and increased motor cortex excitability in patients with severe hemiparetic stroke.
To detect patients’ intentions, BCIs detect brain signals with electroencephalography (EEG), magnetoencephalography (MEG), and near infra-red spectroscopy (NIRS). 13 Problems in using BCI in clinical practice are stable recording of brain signals and feasibility. EEG-based BCI is more feasible than BCIs with MEG or NIRS. It is, however, difficult to record ERD or other motor cortex activities of the affected hemisphere in patients with stroke in clinical settings. 31
It is easier to set the EMG electrodes on the affected UE than EEG electrodes on the scalp. Therefore, for the clinical setting, EMG is more useful than EEG. It is, however, difficult to detect EMG activities in severely paretic muscles. In particular, detecting the finger extensor muscles is difficult in patients with severe hemiparesis. That is why usual EMG-driven robotic devices are challenging to use for patients with severe hemiparesis.
The newly developed, AI-integrated EMG-driven robot hand can detect the patients’ intention of finger extension and flexion with the EMG patterns of 3 muscles in patients whose surface EMG activities were difficult to detect. Even in severe hemiparetic patients, it is hypothesized that motor cortex activities differ with finger extension and finger flexion. Different cortical activities result in different descending signals to the finger flexor muscles and extensor muscles. Therefore, EMG activity patterns of FDS, FDP, and EDC must differ between patients’ intentions to perform finger flexion and to perform finger extension. According to this hypothesis, the newly developed AI identified the patients’ intention of finger movement based on the EMG activity patterns of 3 muscles even in flaccid or spastic patients.
In our study, we applied 8 training sessions in both the active and the control groups. Eight training sessions are a lower number of sessions to compare other robotic training studies. 32 Japanese healthcare system allows 1 or 2 rehabilitation sessions per week in patients with chronic stroke. Most of the chronic patients cannot receive rehabilitation therapy if it has passed over 6 months from their stroke onset. Therefore, we apply a small number of training sessions and a short period of training.
Several limitations of this study should be mentioned. First, the sample size was small. Because of the COVID-19 pandemic, we could not recruit a pre-determined sample size. The real mean difference between before and post was 3.36 in the active group and 2.22 in the control group. According to these values, we re-calculated the sample size with alpha .05 and power 80%. We found a sample size of 20 was sufficient. Therefore, the results of this study were considered valid.
Second, the interventions were time-matched and not intensity-matched, allowing for variability in the number of repetitions delivered per participant. Third, blinding therapists and patients was not possible. Future studies could include clinical studies with more subjects, studies on patients in different phases, such as acute, and studies of intervention duration and frequency. Furthermore, the relationship between the patient’s movement intention from the present EMG activities and brain activities and the observation of changes in brain activity after this treatment are future issues.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231166939 – Supplemental material for New Artificial Intelligence-Integrated Electromyography-Driven Robot Hand for Upper Extremity Rehabilitation of Patients With Stroke: A Randomized, Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683231166939 for New Artificial Intelligence-Integrated Electromyography-Driven Robot Hand for Upper Extremity Rehabilitation of Patients With Stroke: A Randomized, Controlled Trial by Yuhei Murakami, Kaoru Honaga, Hidemi Kono, Koshiro Haruyama, Tomofumi Yamaguchi, Mami Tani, Reina Isayama, Tomokazu Takakura, Akira Tanuma, Kozo Hatori, Futoshi Wada and Toshiyuki Fujiwara in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The corresponding author, Toshiyuki Fujiwara, had received payments for his lectures from Sumitomo Pharma and MELTIN MMI. The other author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by MELTIN MMI, and the robotic machine was provided by MELTIN MMI.
Supplementary material
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
