Abstract
Background. Geographical location, socioeconomic status, and logistics surrounding transportation impede access of poststroke individuals to comprehensive rehabilitative services. Robotic therapy may enhance telerehabilitation by delivering consistent and state-of-the art therapy while allowing remote monitoring and adjusting therapy for underserved populations. The Hand Mentor Pro (HMP) was incorporated within a home exercise program (HEP) to improve upper-extremity (UE) functional capabilities poststroke. Objective. To determine the efficacy of a home-based telemonitored robotic-assisted therapy as part of a HEP compared with a dose-matched HEP-only intervention among individuals less than 6 months poststroke and characterized as underserved. Methods. In this prospective, single-blinded, multisite, randomized controlled trial, 99 hemiparetic participants with limited access to UE rehabilitation were randomized to either (1) the experimental group, which received combined HEP and HMP for 3 h/d ×5 days ×8 weeks, or (2) the control group, which received HEP only at an identical dosage. Weekly communication between the supervising therapist and participant promoted compliance and progression of the HEP and HMP prescription. The Action Research Arm Test and Wolf Motor Function Test along with the Fugl-Meyer Assessment (UE) were primary and secondary outcome measures, respectively, undertaken before and after the interventions. Results. Both groups demonstrated improvement across all UE outcomes. Conclusions. Robotic + HEP and HEP only were both effectively delivered remotely. There was no difference between groups in change in motor function over time. Additional research is necessary to determine the appropriate dosage of HMP and HEP.
Introduction
There are almost 6.8 million stroke survivors in the United States, with 795 000 new occurrences each year 1 of whom nearly 65% experience significant or permanent disability.2,3 By 2030, 3.88% of the US population >18 years of age is projected to have had a stroke, a 20.5% increase in prevalence from 2012. 4 The management of stroke survivors costs the US health care system more than $68.9 billion annually 5 and is projected to increase to $240.67 billion by 2030. 4 Only 10% recover completely, and many require further rehabilitation. 6 One important aspect of improving the lives of these survivors is by seeking opportunities to reduce the magnitude of their residual impairments.
Access to poststroke rehabilitation is a significant barrier for many individuals because of being underinsured, having difficulty with transportation, dependence on caregivers, lower socioeconomic status, and/or the lack of stroke rehabilitation programs and providers in their geographic area.7,8 Residents of minority communities demonstrate lower socioeconomic status, greater barriers to health care access, and greater risks for and burden of disease compared with the general populations living in the same metropolitan and micropolitan statistical area, county, or state.9,10 Nearly half of Americans live more than an hour away from a primary stroke center. 11 Rural areas may be especially underserved, with a lack of facilities to treat stroke survivors and/or limited therapy resources.12,13 Therefore, novel approaches to improve access to rehabilitation such as telerehabilitation (TR) would be meaningful in minimizing disability, and ultimately, reducing the economic burden.
Telerehabilitation is defined as the provision of rehabilitation services at a distance using information and communication technologies.14 -18 It continues to grow as a service delivery alternative to traditional rehabilitation. Evidence exists demonstrating the value and effectiveness of TR stroke programs.19,20 A systematic review of TR interventions showed improved stroke survivors’ and caregivers’ health. 21 High levels of satisfaction and acceptance of home-based TR interventions by both health professionals and users has also been demonstrated.21,22 Some key policy issues that have an impact on the use of home-based TR include the following: cost, reimbursement, privacy and informed consent, fraud, liability, licensure, and systems security. 15
Robotics is an innovative approach to rehabilitation that can be integrated within a TR service delivery model.23 -25 Robotic neurorehabilitation has the potential to have a greater impact on impairment because of ease of deployment, applicability across a wide range of motor impairments, high measurement reliability, and the capacity to potentially deliver the optimal dose and intensity of training protocols that are patient specific. 26 Exercising the hemiparetic hand and wrist is essential in all stages of a stroke rehabilitation program, 27 and robot-mediated rehabilitation can be delivered in every phase of rehabilitation. 28 To date, most studies of robotic TR have been case reports and small studies that have not systematically addressed the efficacy of using a robotic device in a home environment.29,30 However, Piron et al 22 showed greater satisfaction and arm improvement using a home-based TR virtual reality training than a comparable group receiving this training in a hospital environment.
Kinetic Muscles Inc’s, Hand Mentor is an upper-extremity (UE) robotic device originally developed under SBIR R43 HD41805. Results from previous clinical trials supported the utility of the Hand Mentor in home use, 31 ease of use, and the effectiveness of a therapist providing expert feedback. The Hand Mentor Pro (HMP) has added benefits of “store-and-forward” communication and Web-based monitoring. Because medically underserved or rural communities have a need for home-based health services for older adults, 32 they could benefit from a robotic TR program. This study is the first to systematically assess the efficacy and feasibility of a robotic TR intervention compared with a home exercise program (HEP) for the UE of survivors within 6 months poststroke, with limited access to rehabilitation services. We hypothesized that the HEP intervention, when enhanced with robotic-assisted therapy, would be feasible and result in significantly better outcomes in UE motor function.
Methods
Design Overview
The protocol and design for this prospective, multisite, single-blind, randomized controlled clinical trial have been described in our previous publication. 33 Prior to the intervention, all participants signed an informed consent approved by the institutional review board of the Emory University (Atlanta, GA) or Cleveland Clinic (Cleveland, OH).
Participants
A total of 556 potential participants from the Atlanta, GA, and Cleveland, OH, geographic areas were screened. The following inclusion criteria were adopted: (1) a unilateral ischemic or hemorrhagic stroke within the previous 6 months confirmed by neuroimaging; (2) persistent hemiparesis with some UE voluntary movement, as indicated by a score of 11 to 55 on the Fugl-Meyer Assessment (FMA) 34 ; (3) ineligibility to receive any further upper-extremity therapy; and (4) preserved cognitive function (Short Portable Mental Status Questionnaire). 35
Exclusion criteria were the following: (1) inability to provide informed consent; (2) not independent before the stroke (determined by score >1 on the Modified Rankin Scale 36 ); (3) hemispatial neglect, as determined by >3 errors on the Star Cancellation Test 37 ; (4) sensory loss ≥2 on the sensory item of the NIHSS (National Institutes of Health Stroke Scale); (5) hypertonic affected UE as indicated by a score ≥3 on the Modified Ashworth Scale 38 ; (6) antispasticity injection in hemiparetic UE since onset of the stroke; (7) presence of upper-extremity pain or uncorrected vision problems; (8) unmanaged psychiatric issues; and (9) terminally ill with an anticipated survival of less than 1 year.
To ensure uniformity in participant selection procedures, both study sites used standardized forms and an inclusion/exclusion criteria checklist.
Randomization
An adaptive, stratified, computer-driven randomization procedure was used for group assignment to balance critical participant characteristics 39 and minimize imbalance between groups across gender, premorbid handedness, age (<62 or ≥62 years of age), and level of impairment (≤33 or >33 on FMA). Figure 1 presents a consort diagram for this study.
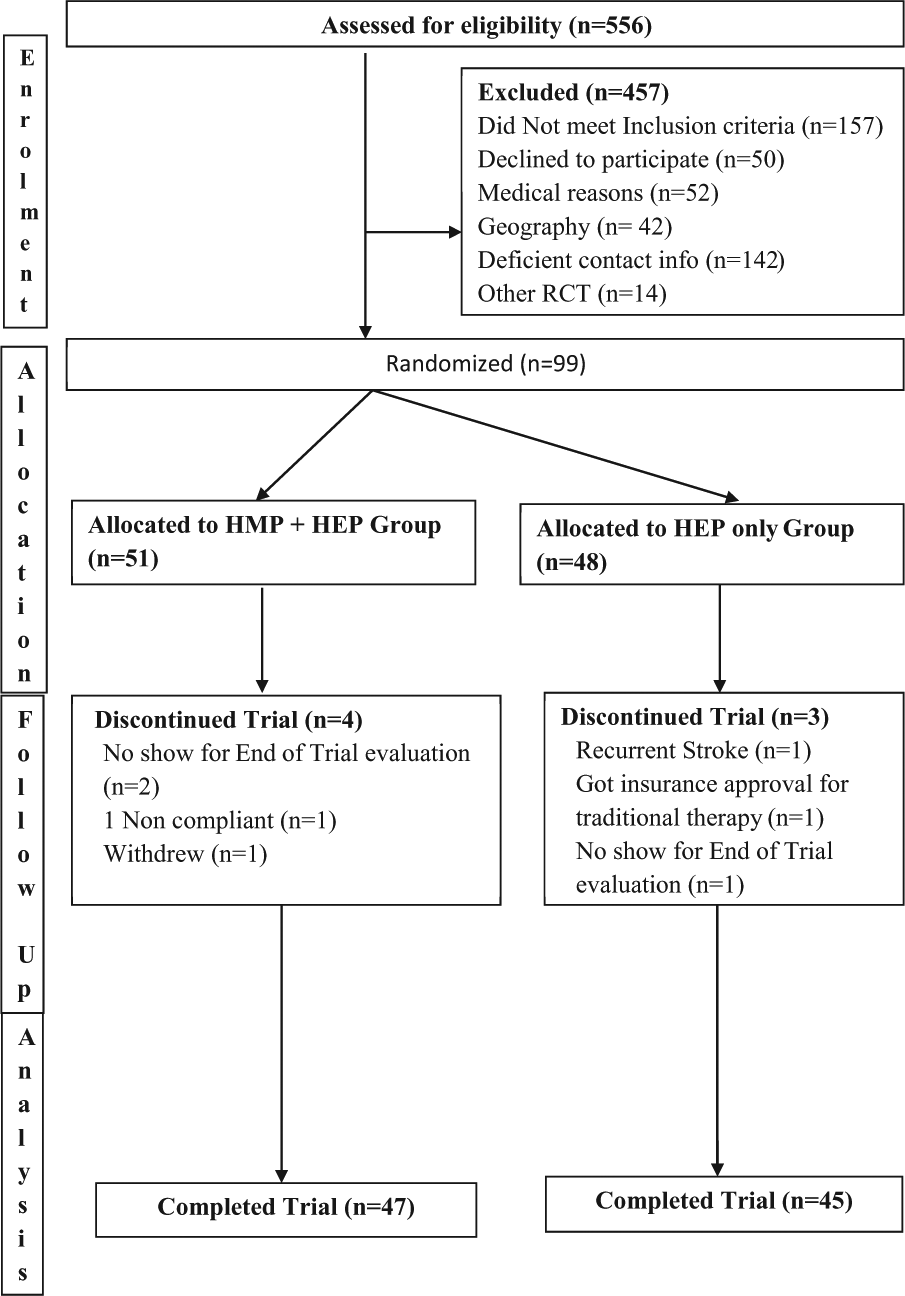
Consort diagram for the HAPPI trial.
Sample Size
Relying on data reported in the VECTORS study,40,41 we anticipated an effect size of 0.50, or a decrease of 20% in median time required to accomplish the Wolf Motor Function Test (WMFT) tasks. This effect size or greater would yield >90% statistical power to detect an interaction (Time × Group), if 40 individuals in each of the 2 groups completed the trial. Assuming a 15% drop-out rate, we planned to enroll 48 patients per group.
Interventions
A detailed description of each intervention can be found in our protocol article. 33 Study therapists at each site were trained for robotic device use and HEP interventions. The interventions were controlled for frequency (3 hours, 5 times per week) and duration (8-12 weeks); participants were asked to complete 120 hours in an 8-week period. 42 The intervention for the control group (CG) consisted of an HEP that included preparatory UE exercises of self-range of motion, weight-bearing activities, active assisted exercises with a cane, shoulder exercises, elbow/forearm exercises, wrist/hand exercises, and task-based activities. Each exercise was presented pictographically and clearly specified technique and dosage, consistent with current clinical practice standards. Participants were asked to complete 2 hours of these and 1 hour of functional activities that incorporated the movements learned during these exercises, each day, 5 d/wk for 8 weeks.
The experimental group (EG) received HEP and HMP training modules. The HMP uses a pneumatic artificial muscle to facilitate movement about the wrist and fingers while providing visual biofeedback about the quality and quantity of wrist movements utilizing video games with a touch screen to facilitate user interface. A customized prescription of different programs—spasticity reduction and basic motor and advanced motor control, per participant’s motor capabilities using the HMP (Figure 2)—was formulated and set. This group was asked to perform 2 hours of training on the robotic device and 1 hour of the HEP. 42 Thus, EG participants performed functional activity within the 1 hour of HEP to incorporate movements used during the robotic training into relevant tasks. The HEP used by both groups addressed motor control of the whole hemiplegic UE. The CG received only the HEP, whereas the EG was provided with the HMP and HEP; however, the EG participants did less formal wrist and finger exercises because these were covered by the HMP. The types of activities prescribed to both groups represented a similar ratio of functional activities.
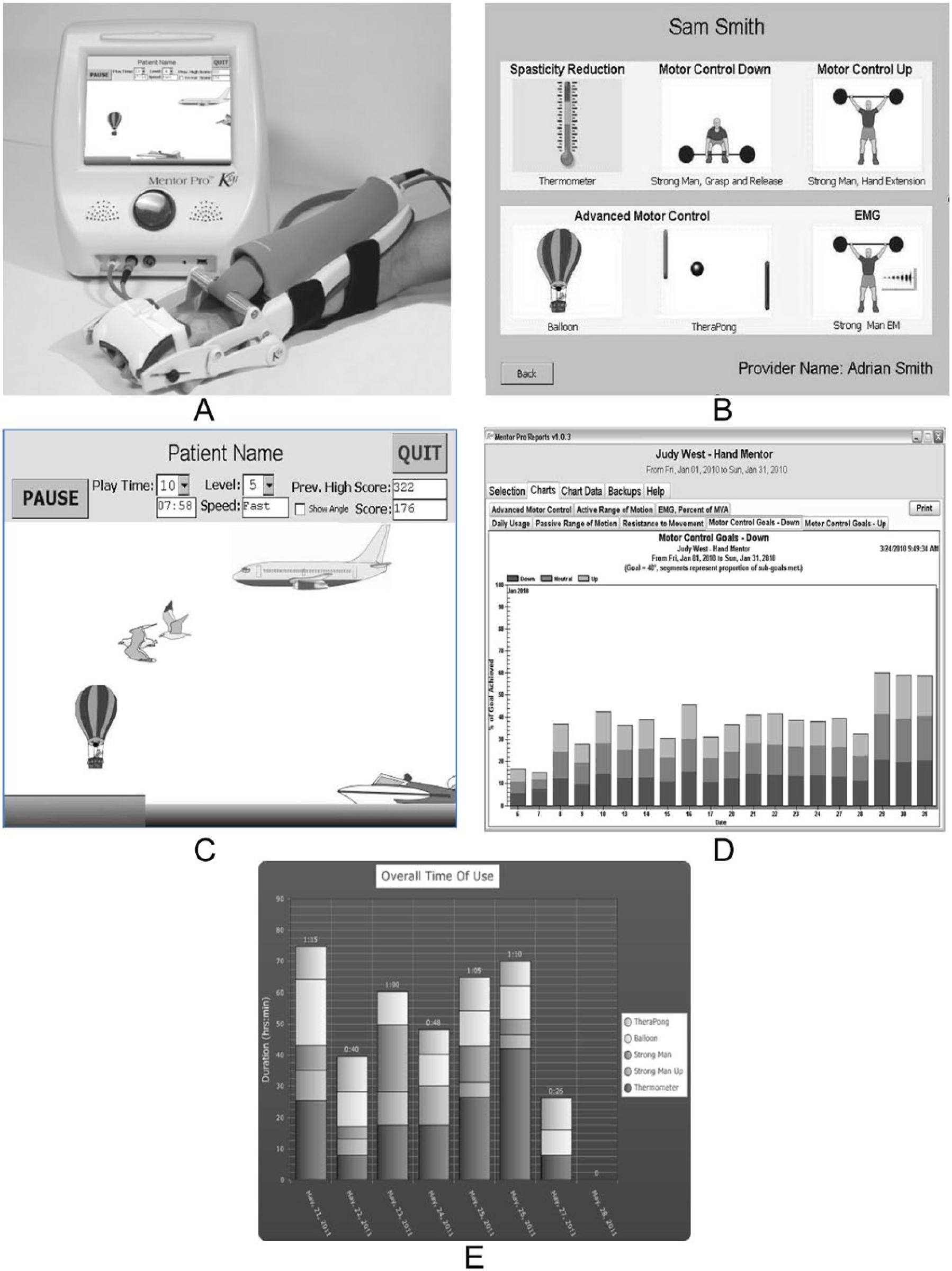
Hand Mentor Pro showing (A) the system and air muscle assembly; (B) program/training options; (C) the balloon game to increase controlled active range of motion; (D) summary performance table to show relative increases or decreases in movement compared with training instruction; (E) summary daily game activity chart.a
In summary, both groups represented a similar ratio of functional activities, with the main difference being that the CG participants completed approximately 2 hours of traditional impairment-based exercises and 1 hour of functional activities, whereas the EG participants completed 2 hours of robotic-based exercises and 1 hour of functional-based activities. The clinical rationale for prescribing functional-based activities to both groups was to incorporate newly acquired movement into functional daily tasks. Finally, both the CG and EG participants were taught their respective HEP, instructed in appropriate TR and monitoring, and matched for frequency/duration of their interventions.
Telerehabilitation and Monitoring
A home visit from the study therapist for training in the respective interventions was scheduled within 2 weeks of randomization. The EG participants were taught how to use the HMP. All this information was contained in a binder along with a signed behavioral contract to facilitate compliance, a frequently asked questions sheet, a daily diary used to record participation in the HEP and robotic use, a troubleshooting guide, and a schedule for weekly monitoring. The study therapist reviewed objective data from the secured Web site to monitor duration of use of device, modules completed, assistance required, and wrist flexion/extension angles achieved for the HMP users. Data from the HMP were transmitted via landline dial up, cellular Internet, or high-speed router connection to the Mentor Home Web site. In cases of failed data transmission, data were stored on the device and transmitted electronically during subsequent uploads. No loss of HMP data was experienced during the trial.
The study therapists made weekly contacts via telephone calls or e-mail with all participants who answered a questionnaire and reported diary information. The HEP was advanced by modifying or adding exercises, encouraging participants to incorporate the more-involved UE into functional activities and activities of daily living and providing solutions to difficulties identified. For the EG, in addition to the HEP, HMP module settings were modified to appropriate stages and levels, and repetitions/time were either increased, decreased, or kept the same as needed. Data regarding occurrences of adverse events were also gathered. There was no prescreening regarding participant or caregiver past experience in electronic information transmission, and participants were considered for enrollment regardless of their technological skills or abilities.
Measures
Participants were assessed before randomization (T1) and after completion of the intervention (T2) by occupational/physical therapists who were trained in the use of standardized assessment protocols and blinded to participant group assignment. 42 The primary outcome was the total change in score, from T1 to T2, of the affected UE on the Action Research Arm Test (ARAT). The ARAT consists of 19 tasks, which are categorized into 4 domains (grasp, grip, pinch, and gross movements). 43 Quality of movement is scored on a 4-point scale (0-3), with a score of 3 indicating normal performance of the task within 5 s and a score of 0 indicating the inability to perform any part of the task within 60 s. The ARAT is a valid and reliable tool for UE deficits following stroke.44 -46 The minimal clinically important difference is 12 points if the dominant UE is affected and 17 points if the nondominant UE is affected. 47 For the ARAT, the interrater minimal detectable change is 13.1, and test-retest minimal detectable change is 3.5. 48
The secondary outcome was the change in score, from T1 to T2, of the WMFT and the FMA. The WMFT consists of 15 timed tasks and 2 strength tasks. Tasks begin with isolated shoulder movements and progress to fine motor tasks of the hand. Patients are encouraged to perform each timed task as quickly as they can. Shorter times reflect better performance. Timed movements are also graded with a functional ability scale for quality of movement. The WMFT has been validated for use with acute to chronic stroke patients, and its clinometric properties have been published.49,50 The FMA is an impairment-based measure consisting of 33 movements, with higher scores indicating increased ability of the patient to move out of synergistic patterns toward more isolated movements. Movement quality of the affected UE is compared with that of the nonaffected UE on a 0 to 2 ordinal scale, with 0 indicating no movement at all, 1 indicating partial movement of the affected extremity, and 2 indicating movement equivalent to the nonaffected UEs. The FMA is a reliable and valid tool for measuring UE impairment following stroke.34,51
Study Analysis
Data were double-entered into a customized MS Access database and exported to where a statistical software package checked and validated both sets of data for accuracy. For purposes of this intent-to-treat analysis, we assumed that data were missing at random, and all data collected from all participants were included in all analyses. Internal consistency of scales was estimated using Cronbach’s α. Baseline demographic and clinical characteristics of the 2 groups were compared using independent-samples t tests for continuous variables and χ2 or Fisher’s exact tests for categorical variables. Summary data are reported as mean ± standard deviation or count (percentage).
Changes in functional outcome scores from T1 to T2 were analyzed using a mixed-model approach, with random effects for participant scores. For purposes of this intent-to-treat analysis, we assumed that data were missing at random. The mixed-analysis approach was used, with no ad hoc imputation.40,41 The estimate of primary interest was the time (T1, T2) × intervention (HEP, HEP + HMP) interaction. Outcome scores, except the FMA, were adjusted for participants’ age at enrollment, time between stroke and enrollment, baseline Center for Epidemiologic Studies–Depression Scale (CES-D) score, and baseline FMA score. Analyses of the Fugl-Meyer scores were adjusted for participants’ age at enrollment, time between stroke and enrollment, and the baseline CES-D score. A P value of .05 was used as the criterion for statistical significance, and no adjustments were made for multiplicity. Analyses were performed using SPSS version 22 (IBM Corp, Armonk, NY). 41
Results
Between June 2010 and June 2013, 99 participants were assigned to the experimental (n = 51) or control (n = 48) groups with 7 dropouts (Figure 1). All functional outcome scales and subscales showed adequate reliability during both measurement periods (α met or exceeded .80). Demographic and clinical characteristics did not differ significantly at baseline for the 2 intervention groups (Table 1).
Participant Demographics.
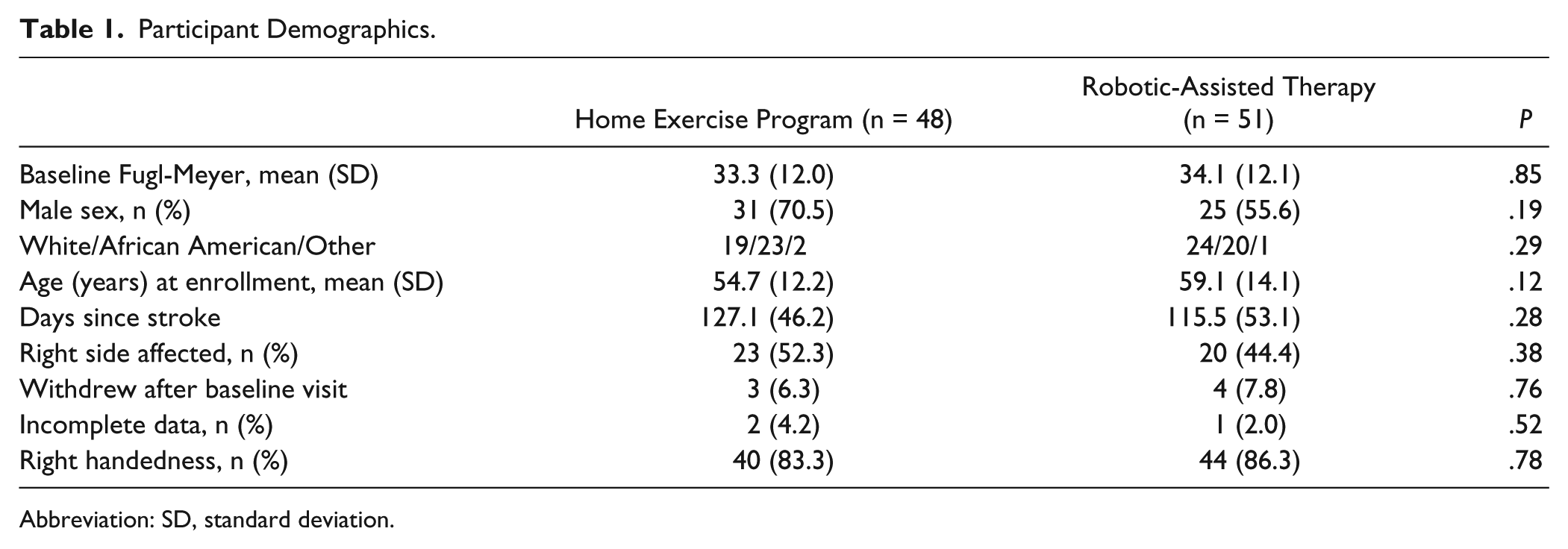
Abbreviation: SD, standard deviation.
The 7 participants who withdrew after their baseline visit did not differ significantly from those who completed the trial on any of the functional outcome measures at T1 (data not shown). In addition to those lost to follow-up, 2 participants in the CG (4.2%) and 1 in the EG (2.0%) failed to complete all functional outcome measures at both visits.
Total time spent engaging in the therapeutic interventions was calculated using self-reported time spent performing prescribed interventions for participants in the CG (8369 ± 3373 minutes, range = 2790-24325 minutes) and self-reported time spent performing prescribed activities/exercises + device-recorded HMP use for those in the EG (8052 ± 4042 minutes, range = 928-21195 minutes, P = .68). Participants in the EG used the device for 2172 ± 1388 minutes (range = 12-5153 minutes).
Means and 95% confidence intervals for functional scale scores for participants in the 2 groups at T1 and T2 are provided in Table 2. T1 scores did not differ across groups for any functional scale. Preliminary analyses were conducted to assess the potential effect of site (Cleveland Clinic, Emory) on change in each of the outcome scores. Because none of these interactions was significant (data not shown), data were collapsed across study locations. Although total time spent within each intervention did not differ across groups, we determined whether it moderated or mediated the impact of the interventions on outcome scores. Analyses conducted testing both moderational and mediational effects did not alter conclusions that may be derived from the results provided in Table 2 (data not shown).
Means (95% Confidence Intervals) for Key Variables.
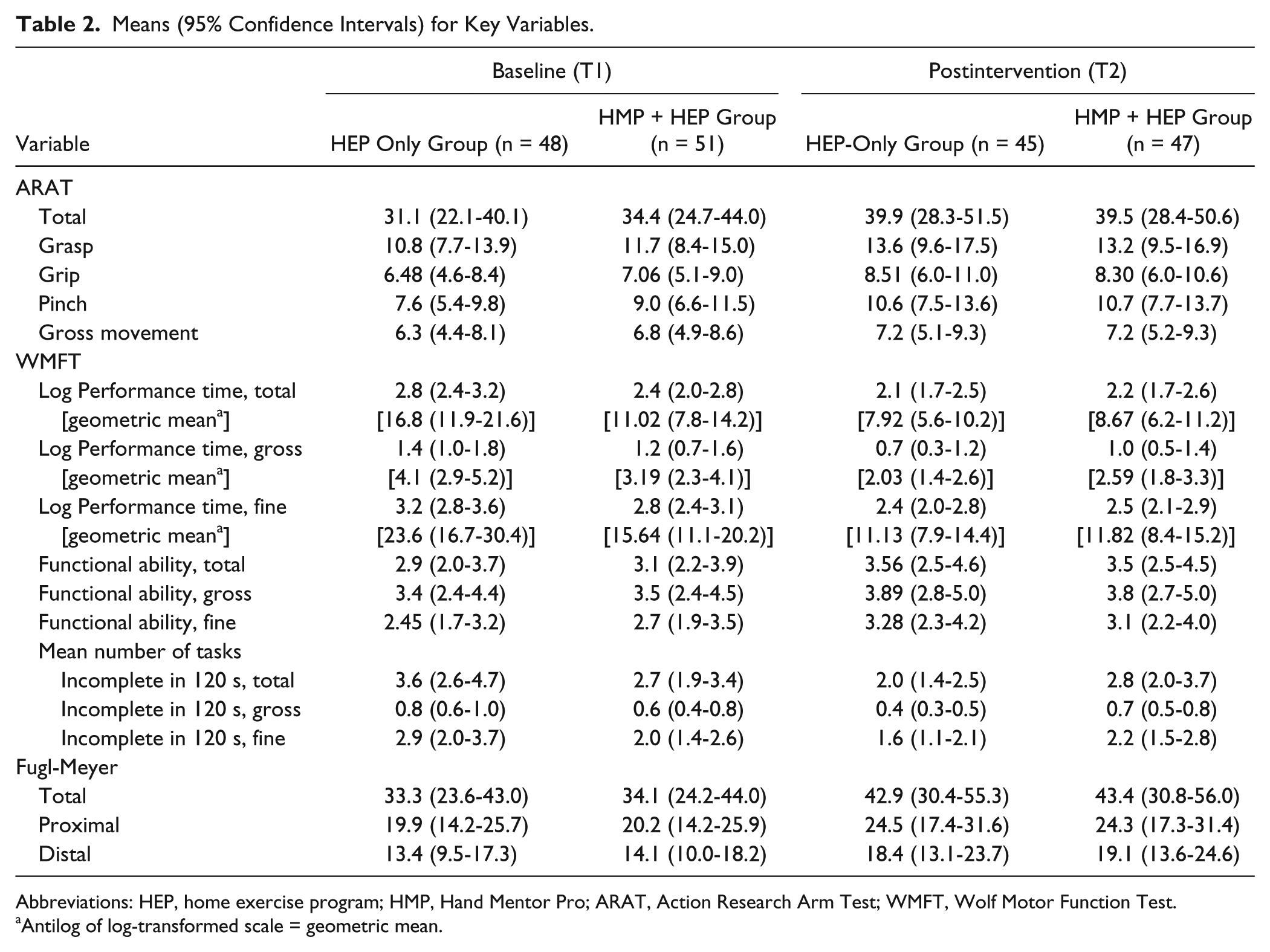
Abbreviations: HEP, home exercise program; HMP, Hand Mentor Pro; ARAT, Action Research Arm Test; WMFT, Wolf Motor Function Test.
Antilog of log-transformed scale = geometric mean.
The P values and 95% confidence intervals for outcome measures (Table 3) display the ARAT scores and WMFT tasks by total and subcomponent scores. Both groups improved significantly, and between-group differences (last column) were observed for WMFT performance time on total and fine motor tasks, and number of total and fine motor tasks not completed. Each of these between-group differences favored the CG. The only one for which significant within-group changes did not occur (for EG) was for reacquisition of task completion within 2 minutes involving distal (digital) movements.
Outcome Measures. a
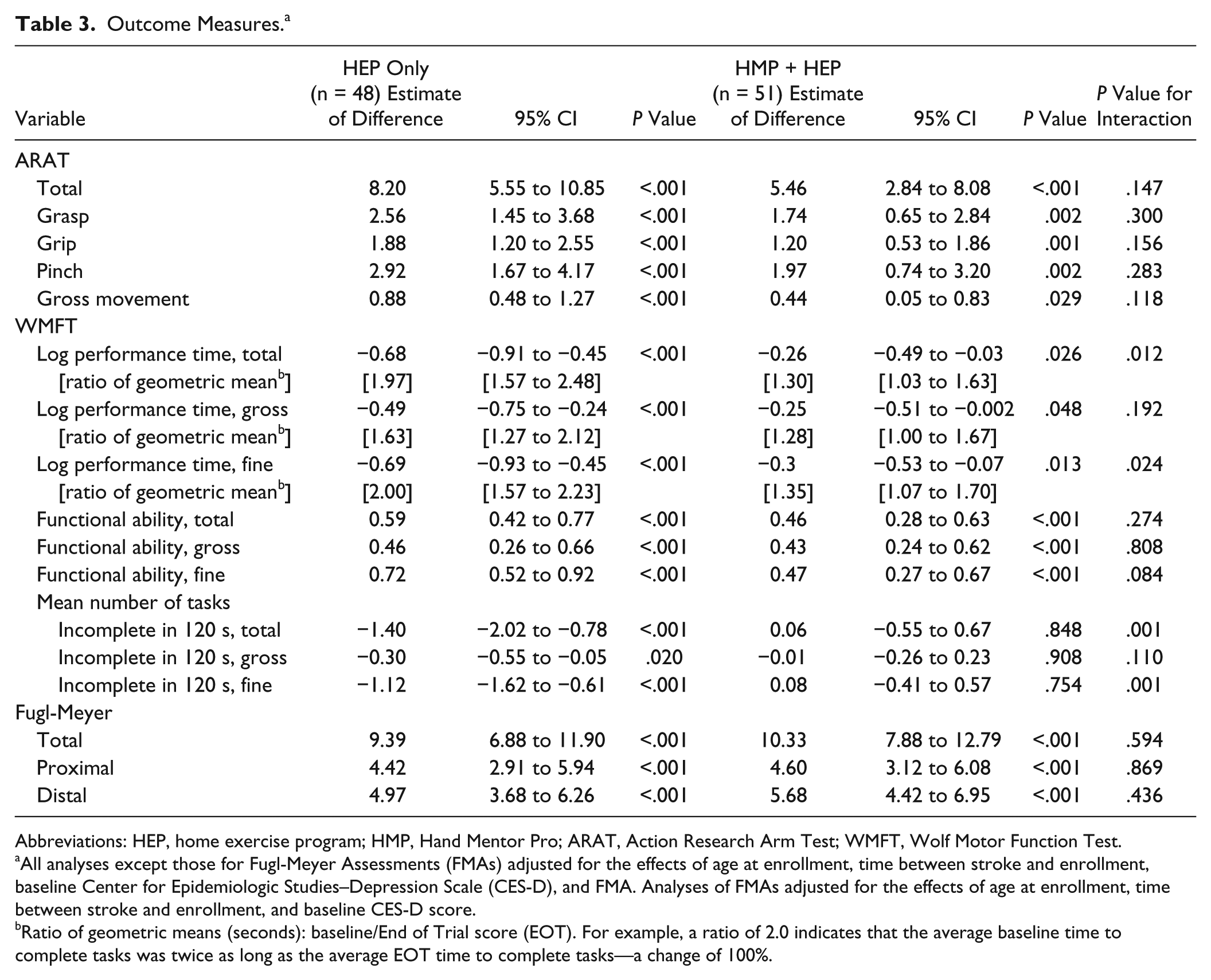
Abbreviations: HEP, home exercise program; HMP, Hand Mentor Pro; ARAT, Action Research Arm Test; WMFT, Wolf Motor Function Test.
All analyses except those for Fugl-Meyer Assessments (FMAs) adjusted for the effects of age at enrollment, time between stroke and enrollment, baseline Center for Epidemiologic Studies–Depression Scale (CES-D), and FMA. Analyses of FMAs adjusted for the effects of age at enrollment, time between stroke and enrollment, and baseline CES-D score.
Ratio of geometric means (seconds): baseline/End of Trial score (EOT). For example, a ratio of 2.0 indicates that the average baseline time to complete tasks was twice as long as the average EOT time to complete tasks—a change of 100%.
Discussion
The HAAPI trial presents one of the first large-scale systematic studies of a robot-mediated neurorehabilitation program for patients with UE impairments poststroke, with lack of access and financial barriers in a home-based telehealth delivery model. Significant within-group improvements were noted for both groups. These findings indicate that a robotic TR program can be successful in producing significant improvements in motor outcomes for subacute stroke survivors, especially in the home environment. The results also demonstrate that HEP + robotic TR can be equally effective as an individualized HEP. Therefore, this home-based robotic intervention may be a valuable alternative for rural or underserved stroke populations with limited access to traditional therapy as a result of financial, caregiver, or transportation constraints or lack of rehab facilities.
Our results also underscore the prospects for other socioeconomic groups to succeed with this and other robotic-TR applications and that this work can be undertaken among comparable populations in different cities (Atlanta and Cleveland) with comparable results. In this trial, there was a relatively low drop-out rate (eg, <10%) across both groups. Retention of participants across both groups suggests that when a HEP or a technology-enhanced HEP intervention is augmented by weekly, albeit short, <15 minutes on average, interactions with a physical/occupational therapist, patients will remain engaged in their treatment plan. A potential advantage to using a robotic device along with a HEP is the automatic transmission of usage and performance data gathered with the robotic device. This objective information can rapidly inform the therapist about the patient’s level of function, amount of practice, and areas of success. Information about device use and physical function was provided to make patient-specific adjustments in HMP protocols and HEP tasks; the provision of actionable information about which both the patient and therapist are aware may facilitate therapist-patient communication and take a potentially isolating therapeutic approach and transition it to one in which the provider and patient are actively engaged. Hence, we believe that the combination of technology and clinical practice has significant potential to facilitate the recovery of upper-extremity motor function in patients with stroke.
We saw no differences in compliance by site; there were no pain or discomfort issues to distinguish participants; and most seemed to enjoy the experiences. In part, compliance appeared to be related to the individuals’ intrinsic motivation to regain movement and function. Some participants found value in the prescribed program and adhered to the intervention and dosage diligently, whereas others did not. Additionally, family and life role responsibilities such as taking care of children or going to work occasionally interfered with compliance. There may have been some additional factors that represented generational gaps in comprehending computers and information/technology transmission that we thought were solved but might not have been in our absence. Again, many of these issues are discussed, and overcoming the obstacles will undoubtedly enhance the feasibility.
Additionally, this study demonstrates that more severely affected participants poststroke (as evidenced by low FM scores) can feasibly use and actively participate in prolonged, repetitive task practice with little oversight by family or therapists. Finally, the HMP robotic system was used by both young and old participants, which demonstrates that the technology is applicable across a range of ages.
There are limits to this study. First, because participants were less than 6 months poststroke, spontaneous recovery might have contributed to significant motor gains. Second, the HEP-only group exceeded our expectations in terms of gains in motor performance. Typically, lack of motivation and musculoskeletal limitations have been cited as 2 major barriers limiting adherence to a HEP for patients poststroke, 52 whereas telephone coaching can improve compliance. 53 This study made a concerted effort to engage participants in both groups with weekly phone calls to help participants problem-solve through any issues, such as pain or difficulty with exercise, and encouraged use of the affected limb in daily activities. Chiang et al 54 have identified family support and exercise design as key factors for HEP compliance. Responses to informal inquiries suggest that there was high caregiver support and participation in this study. HEPs were also individually designed and progressively adapted throughout the study. These combined efforts may have had a favorable impact on both groups, producing motor gains that might not have been present if the participants were asked to complete the HEP independently. Our design did not allow us to delineate the relative level of caregiver participation or enthusiasm for so doing. However, we did observe occasional expressed frustration on the part of caregivers if HMP participants did not readily comprehend the computer interface.
Third, although the robotic intervention was delivered remotely, therapist contact time was still involved. TR services involved screening candidates for study eligibility, an in-home assessment, training and education with the HEP and/or HMP, and ongoing monitoring during the length of the study. On average, therapists spent 13.0 ± 9.1 min/wk with the HMP + HEP group participants. Thus, although it is a viable option to deliver rehabilitation services, TR does not eliminate the therapist-client relationship. We did not formally examine the cost-effectiveness of delivering interventions for the HMP + HEP group versus the HEP-only group, but one can use our existing data (13 min/wk for HMP instruction) as a basis for assessing cost to benefit for home-based robotic use among poststroke survivors, including those living in remote or underserved locations. In this context, a 2013 Cochrane Collaboration Review of TR services for stroke concluded that no existing studies have proven or even adequately assessed the cost-effectiveness of TR compared with traditional therapy services. 18 Equipment costs, training on the equipment, and remote monitoring are a few of the factors typically associated with TR. 18 Future studies involving TR interventions should assess the costs of delivery. 18 In addition, clinicians or organizations choosing to engage in TR will need to consider ways in which they can effectively, efficiently, and productively deliver TR measures in our dynamically changing health care system. Another variable that must be considered in the utilization of robotics for neurorehabilitation is the potential to use these systems for the tracking of motor function over time after the patient has been discharged from inpatient rehabilitation. Robotic systems can provide clinically useful biomechanical measures (eg, range of motion, measure of spasticity) through the use of a relatively simple sensor package. These objective outcomes, over time, could guide subsequent outpatient therapy and delivery of care into remote areas or underserved populations.
Finally, while we demonstrated that the HEP + HMP intervention can be successfully used in the home environments of underserved stroke survivors, it did not yield superior outcomes as hypothesized. In fact, between-group differences showed greater improvements on several aspects of the WMFT and number of distally based tasks reacquired (secondary outcomes) favoring the HEP group (Table 3). Although floor and ceiling effects of both the ARAT and WMFT for patients who are severely or mildly impaired have been reported,48,55,56 we cannot attribute the between-group differences to this possibility because the mean difference between the 2 groups at baseline was well within measurement error. However, these between-group differences might be related to the extremely varied use time of the robotic device (12-5153 minutes) and the fact that the device, while recording resistance to extension movement generated within wrist and finger extensors, does not retrain explicitly for total or fractionated finger movements, thus limiting the extensiveness of the functional retraining. This possibility is feasible and speaks to the importance of incorporating independent finger movement training/gaming within hand robotic, home-based interfaces. Future studies involving home use of robotics and telecommunication of data should have design features that take into account comprehension and ease of use on the part of both the consumer and caregiver, greater specification of the program to meet user needs, and a clearer assessment of frequency of direct contact to maximize compliance.
Conclusion
This study uniquely incorporated a TR component to the robotic-assisted therapy in the home and was novel because it combines HEP and robotics, which may serve as a comparable alternative to the traditional therapeutic relationship. With more poststroke therapy directed toward the home, the need for such an alternative exists. TR plus robotics was found to be feasible and has the potential to be safe. The device is portable and has a wireless and Web-based capability of transmitting data from a home to a secured base station. As a result, the TR component may be a practical and valuable approach to delivering poststroke care when limited resources, manpower shortages, long distances, or compromised patient mobility restrict or limit access to other treatment locations; however, a more detailed selection of users will be required before this approach could become better than a home-based exercise program.
Footnotes
Acknowledgements
The author appreciates the contributions offered by Susan Murphy, Marsha Bidgood, PT, and Cindy Clark, OTR/L, in scheduling and evaluating participants.
Authors’ Note
Dr Wolf was Chairman of the Scientific Advisory Board for Kinetic Muscles Inc, during this study and was previously a paid consultant for Kinetic Muscles Inc. Sharon Buchanan was a paid consultant for Kinetic Muscles Inc. No other authors have any financial relationship with Kinetic Muscles Inc.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by NIH grant RC3NS070646 from the National Institute of Neurological Disorders and Stroke. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Neurological Disorders and Stroke or the National Institutes of Health.
