Abstract
Background
The key neural pathological characteristics of autism spectrum disorder (ASD) include abnormal synaptic plasticity of the medial prefrontal cortex (mPFC). Exercise therapy is widely used to rehabilitate children with ASD, but its neurobiological mechanism is unclear.
Methods
To clarify whether the structural and molecular plasticity of synapses in the mPFC are related to improvement in ASD behavioral deficits after continuous exercise rehabilitation training, we applied phosphoproteomic, behavioral, morphological, and molecular biological methods to investigate the impact of exercise on the phosphoprotein expression profile and synaptic structure of the mPFC in valproic acid (VPA)-induced ASD rats.
Results
Exercise training differentially regulated the density, morphology, and ultrastructure of synapses in mPFC subregions in the VPA-induced ASD rats. In total, 1031 phosphopeptides were upregulated and 782 phosphopeptides were downregulated in the mPFC in the ASD group. After exercise training, 323 phosphopeptides were upregulated, and 1098 phosphopeptides were downregulated in the ASDE group. Interestingly, 101 upregulated and 33 downregulated phosphoproteins in the ASD group were reversed after exercise training, and these phosphoproteins were mostly involved in synapses. Consistent with the phosphoproteomics data, the total and phosphorylated levels of the proteins MARK1 and MYH10 were upregulated in the ASD group and reversed after exercise training.
Conclusions
The differential structural plasticity of synapses in mPFC subregions may be the basic neural architecture of ASD behavioral abnormalities. The phosphoproteins involved in mPFC synapses, such as MARK1 and MYH10, may play important roles in the exercise rehabilitation effect on ASD-induced behavioral deficits and synaptic structural plasticity, which requires further investigation.
Keywords
Background
Autism spectrum disorder (ASD) is a serious, early-onset neurodevelopmental disorder with several symptoms, typically including deficits in social interaction and communication and repetitive behavior, which are often accompanied by attention deficits, hyperactivity, anxiety, depression, or other mental disorders.1 -3 The prevalence rate of ASD among children is up to 1/36 in the USA and 1% in China and is continuing to increase.1,4 Due to the lack of effective drugs and therapies to cure ASD, exercise therapy is widely used in the rehabilitation of children with ASD and can improve social deficits and repetitive behavior to a certain extent.5 -7 However, the neurobiological mechanism by which exercise training alleviates the core symptoms of ASD is unclear.
The medial prefrontal cortex (mPFC), including the prelimbic cortex (PrL) and infralimbic cortex (IL), plays an important role in cognitive memory, emotional regulation, motivation, and sociability by integrating information from the sensory cortex, motor cortex, and many subcortical areas.8 -10 Numerous studies have shown that ASD-induced social disorders, repetitive behavior, and emotional and cognitive abnormalities are closely related to the neural number and morphology, protein expression, and phosphorylation level in the mPFC.11,12 It has been shown that the abnormal proliferation of pyramidal neurons in the mPFC area may induce the overgrowth of brain tissue in ASD patients.12,13 Interestingly, the density of prefrontal dendritic spines in ASD patients at birth is similar to that in healthy people; however, during childhood, the density of dendritic spines in ASD patients is abnormally increased, suggesting that the synaptic structure undergoes abnormal remodeling. 14 Many high-risk mutation genes in ASD patients, such as NRXN1, SHANK, NLGNs, MYH10, and SYNGAP1, are involved in the coding of synapse-related proteins that, affect the formation and maturation of synapses and the strength and efficiency of synaptic connections.15 -18 In addition, exercise affects the structural and functional plasticity of synapses and alleviates the symptoms of ASD.5,6,19,20 Given the critical role of prefrontal molecular and synaptic plasticity in the development of ASD, thoroughly studying the effects of exercise training on the proteins involved and synaptic plasticity in the mPFC of ASD is vital.
Phosphorylation is the most common and important method of protein posttranslational modification and regulates almost all life activity processes, such as cell proliferation, development, differentiation, and signal transduction. Currently, as mass spectrometry-based phosphoproteomics can both detect the phosphorylated peptides and sites and further identify the phosphorylated proteins, it is becoming a powerful tool in systems biological mechanism research of disease pathogenesis and treatment.21,22 In addition, many diseases, including ASD, have been demonstrated to be caused by abnormal protein phosphorylation.23,24 Therefore, we applied phosphoproteomics along with behavioral, morphological, and molecular biological methods to investigate the neurobiological mechanism by which exercise training alleviates core symptoms in valproic acid (VPA)-induced ASD rats.
In this study, we found that the differential synaptic structures in subregions of the mPFC may be the basic neural architecture of behavioral abnormalities in ASD. In addition, the phosphoproteins associated with synapses in the mPFC, such as microtubule affinity-regulating kinase 1 (MARK1) and myosin heavy chain 10 (MYH10), may play important roles in exercise training in the rehabilitation of ASD-induced behavioral deficits and abnormal synaptic structures, which requires further investigation.
Methods
For the detailed methods, please see the Supplemental material. Briefly, young male VPA-induced ASD rats and control rats underwent a period of wheel running training and swimming training. A series of behavioral experiments were performed, namely, the social behavior test, open field (OF) test, repetitive behavior test, Y-maze test, and novel object recognition (NOR) test, to evaluate the effect of exercise training on the behavioral deficits of ASD rats. Then, specimens were prepared for transmission electron microscopy (TEM) to observe the density, morphology, and ultrastructure of synapses. A phosphoproteomic analysis was carried out based on liquid chromatography–mass spectrometry (LC-MS/MS) to detect the differential phosphorylated peptide and sites and further identify the phosphoproteins. Phos-tagged and conventional SDS-PAGE Western blot analyses were conducted to detect the levels of phosphorylated and total MARK1, MYH10, Rho GTPase activating protein 44 (ARHGAP44), and neurabin-1 proteins.
Results
Effect of Exercise Training on the Behaviors of VPA-Induced ASD Rats
Numerous studies have shown that the offspring of rats exposed to VPA during pregnancy have the core symptoms of ASD, including social disability and repetitive behavior.25 -27 Therefore, we performed an intraperitoneal injection of 300 mg/kg VPA once a day on the 10th and 12th days of pregnancy, collected the offspring as ASD model rats and then recorded the physiological indexes and behavioral performance. 28 Compared with the NS rats, the body weight of the ASD rats was decrease, and the pain threshold was increased. There was no difference between the NS rats and ASD rats in the eye opening test scores (Supplemental Figure 1a-c). The social behavior test showed that compared with the NS rats, the ASD rats spent less time in the chamber containing an unfamiliar rat (stranger 1) and had less sniffing and head contact with stranger 1. When the 3-chamber apparatus contained stranger 1 and another unfamiliar rat (stranger 2) simultaneously, the ASD rats spent less time in the chamber that contained stranger 2 and had sniffing and head contact with stranger 2 (Supplemental Figure 1d-k). The repetitive behavior observations showed that the ASD rats spent more time grooming than the NS rats when placed in an OF (Supplemental Figure 1l, m). The OF test showed that the groups did not significantly differ in the total distance traveled. However, compared with the NS rats, the ASD rats exhibited significantly decreased travel in the central area (Supplemental Figure 1n-q). The Y-maze test showed that there were no significant differences between the groups in the ratio of time spent in the novel arm (Supplemental Figure 1r, s). The NOR test showed that the ASD rats had a lower desire to explore the novel object than the NS rats (Supplemental Figure 1t, u). To evaluate the effect of exercise training on the behavioral deficits of ASD, 28-day-old male NS rats and ASD rats were randomly divided into exercise groups (NSE and ASDE) and sedentary groups (NS and ASD). The exercise groups were trained with wheel running combined with swimming for 4 weeks after 1 week of adaptation, and the sedentary groups were raised in their home cages during the same period. After the exercise training, the ASD rats exhibited a significant improvement in sociability, social novelty preference (Figure 1A-H) and short-term working memory (Figure 1Q and R) and alleviation of repetitive (Figure 1I and J) and anxiety (Figure 1L-N) behavior. There was no significant difference in locomotor activity (Figure 1K) or short-term working memory (Figure 1O and P) among the groups.
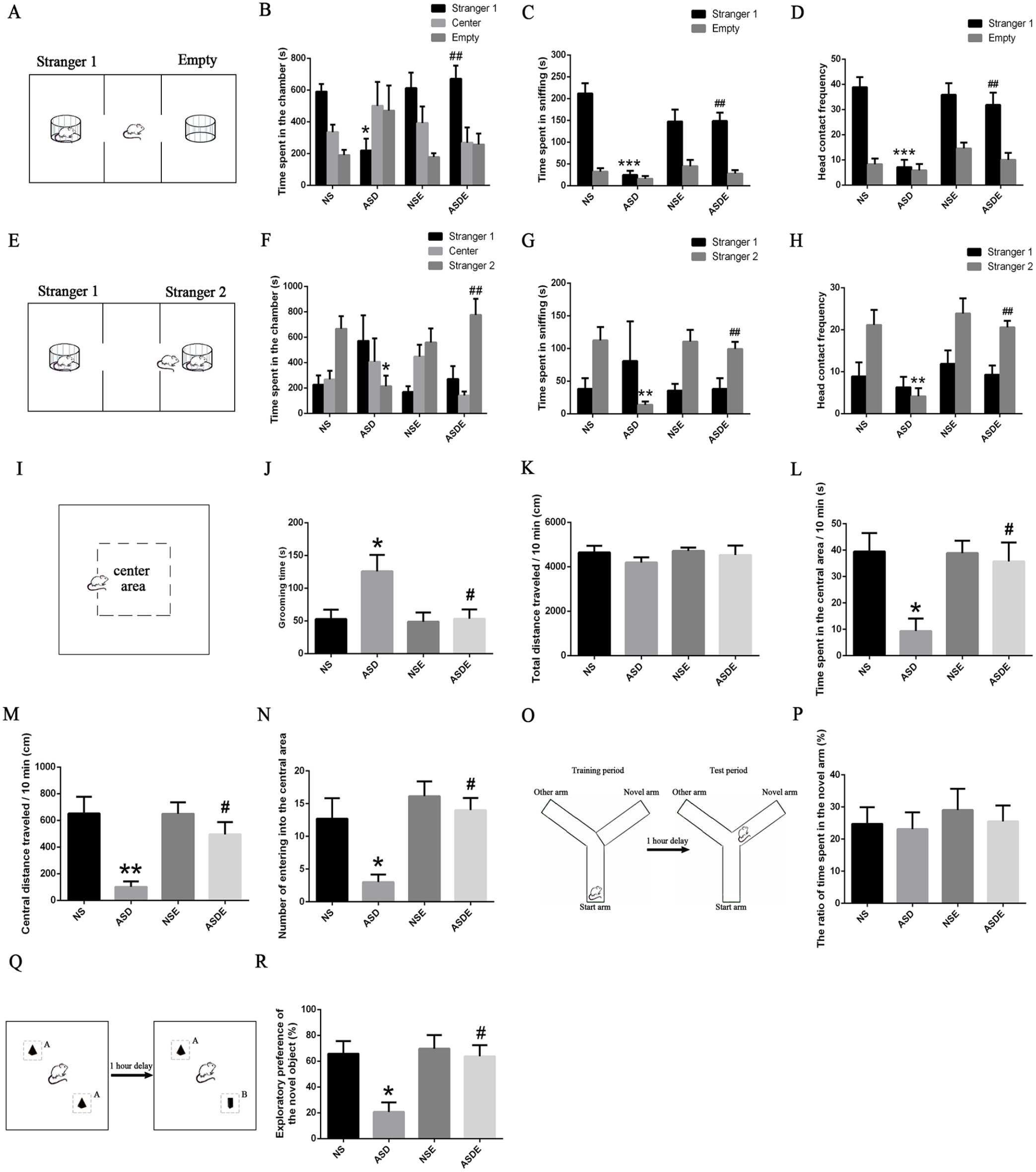
Effect of exercise training on the behaviors of VPA-induced ASD rats (n = 7 rats/group). Exercise increased the sociability (A-D) and social novelty preference (E-H) of VPA-induced ASD rats. (I, J) Exercise alleviated the repetitive behavior of VPA-induced ASD rats. (K) The locomotor activity of the rats in each group did not significantly differ. (L-N) Exercise alleviated the anxiety behavior of VPA-induced ASD rats. (O, P) The short-term working memory of rats in each group did not significantly differ. (Q, R) Exercise enhanced the short-term recognition memory of novel objects in VPA-induced ASD rats.
Effect of Exercise Training on the Synaptic Structure in the mPFC in VPA-Induced ASD Rats
Structural disorganization in the mPFC, such as excess neurons, abnormally increased dendritic spines, dysregulation of layer formation, and layer-specific neuronal differentiation, is the most obvious pathological feature of ASD children.12 -14 The synapse is the key connection between neurons, and its structural basis is the spine. Many high-risk ASD mutation genes are involved in the coding of synapse-related proteins,15 -17 suggesting that there may be abnormal changes in the synaptic architecture. Interestingly, both clinical and laboratory studies revealed that exercise can improve the abnormal behavior and synaptic plasticity of ASD.5,6,19,20 Therefore, to determine whether exercise training can alter the synaptic structure in the mPFC in VPA-induced ASD rats, we collected the PrL and IL subregions from the NS, NSE, ASD, and ASDE groups to observe the density, morphology, and ultrastructure of synapses using TEM. The classic morphology of frown synapses, flat synapses, and smile synapses is shown in Figure 2A. The synapse density in the PrL was significantly increased, but it was decreased in the IL in the ASD group and was reversed in both areas after exercise training (Figure 2B and I). In addition, the morphology of synapses was differentially changed in the PrL and IL. Flat synapses and frown synapses, but not smile synapses, were significantly increased in the ASD group, and only frown synapses were significantly decreased in the PrL after exercise training (Figure 2C-E). In the ASD group, only smile synapses were significantly decreased, which was reversed after the exercise training in the IL (Figure 2J-L). In addition, we observed synaptic ultrastructures containing the length of the active zone, the thickness of the postsynaptic density (PSD) and the width of the synaptic cleft. We found that the width of the synaptic cleft was significantly decreased in the ASD group and was reversed after exercise in both the PrL and IL (Figure 2F and M). In contrast, the length of the active zone was significantly increased in the ASD group and was reversed after exercise in the IL but not the PrL (Figure 2G and N). The thickness of the PSD was significantly increased in the ASD group and was reversed after exercise in the PrL but not the IL (Figure 2H and O).
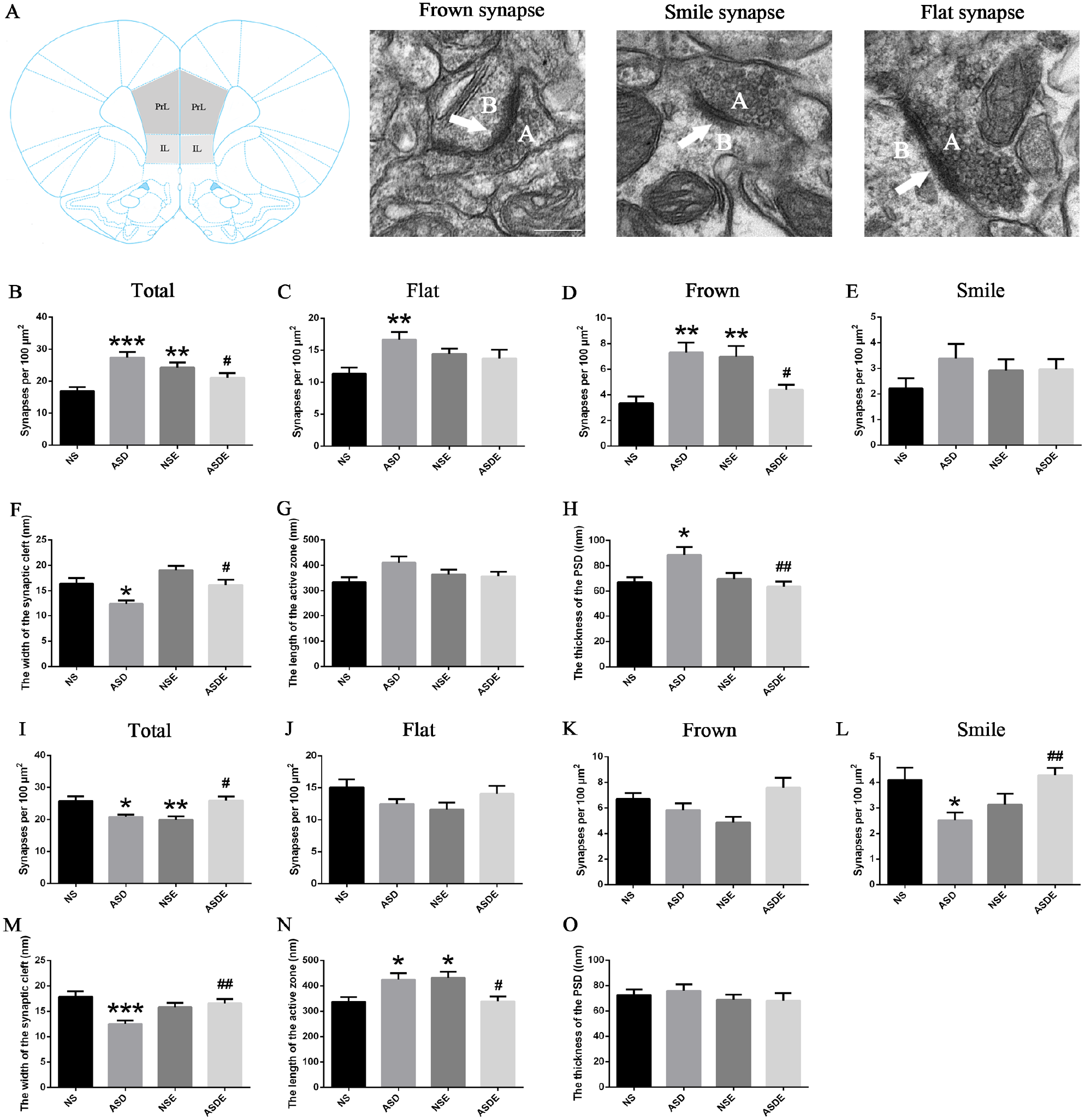
Effect of exercise training on the synaptic structure in the mPFC in VPA-induced ASD rats (n = 3 rats/group). (A) The anatomical position of the PrL and IL and the representative morphology of frown, flat, and smile synapses. “A” represents presynaptic, and “B” represents postsynaptic. (B) The total synapse density was increased in the ASD and NSE groups and decreased in the ASDE group in the PrL. (C) The flat synapse density was increased in the PrL in the ASD group. (D) The frown synapse density was increased in the ASD and NSE groups and decreased in the ASDE group in the PrL. (E) There was no significant difference in the smile synapse density in the PrL among the groups. (F) The width of the synaptic cleft was decreased in the ASD group and increased in the ASDE group in the PrL. (G) None of the groups showed a significant difference in the length of the active zone in the PrL. (H) The thickness of the PSD was increased in the ASD group and decreased in the ASDE group in the PrL. (I) The total synapse density was decreased in the ASD and NSE groups and increased in the ASDE group in the IL. (J, K) No significant difference in the flat and frown synapse density in the IL was found among the groups. (L) The smile synapse density was decreased in the ASD group and increased in the ASDE group in the IL. (M) The width of the synaptic cleft was decreased in the ASD group and increased in the ASDE group in the IL. (N) The length of the active zone was increased in the ASD and NSE groups and decreased in the ASDE group in the IL. (O) None of the groups exhibited significant differences in the thickness of the PSD in the IL.
Effect of Exercise Training on Phosphoproteins Expressed in the mPFC of VPA-Induced ASD Rats
Exercise alleviated the behavioral deficits and regulated the synaptic structural plasticity in the mPFC in ASD rats, but the possible neurobiological mechanism is still poorly understood. Because many diseases, including ASD, have been demonstrated to be caused by the abnormal phosphorylation of proteins,23,24 it may be meaningful to understand the effect of exercise training on ASD by thoroughly studying the protein phosphorylation profile in the mPFC of VPA-induced ASD rats, which is a key brain area related to sociability. Therefore, quantitative phosphoproteomics using IBT labeling combined with LC-MS/MS was utilized. In total, 15019 phosphopeptides were identified, and 9921 phosphorylation sites were located on 3242 phosphorylated proteins (data not shown). In addition, we further quantified the phosphopeptides and determined the differentially phosphorylated peptides among the NS, ASD, and ASDE groups by setting the fold change threshold as >1.2 or ≤0.83 and the statistical P ≤ .05. Compared with the NS group, 1031 phosphopeptides were upregulated and 782 phosphopeptides were downregulated in the ASD group. After the training program, 323 phosphopeptides were upregulated and 1098 phosphopeptides were downregulated in the ASDE group compared with the ASD group (Figure 3A-D). We considered that the mechanism by which exercise training alleviated the behavioral deficits of VPA-induced ASD rats may involve the significant differential phosphopeptides that were both upregulated (or downregulated) in the ASD group and downregulated (or upregulated) in the ASDE group. Therefore, a hierarchical cluster analysis (HCA) of the 138 differential phosphopeptides of interest was performed. We found that 34 phosphopeptides were both downregulated in the ASD group and upregulated in the ASDE group, corresponding to 33 phosphoproteins, including TRA2B (S99, S26), ARHGAP44 (S788, S781), and neurabin-1 (S160; Figure 3E and Supplemental Table 1). In addition, 104 phosphopeptides were both upregulated in the ASD group and downregulated in the ASDE group, corresponding to 101 phosphoproteins, including NEFH (S808, S616, S754, and S774), CDK5R2 (T3, S6, and S8), MAP1A (S322, S894, S898, and S2228), CACNA1A (S2220), MYH10 (S1952 and S1145), MARK1 (S611), and SYNGAP1 (S1099, S94, and S1106; Figure 3F and Supplemental Table 2). Notably, the phosphorylation levels of T169 (downregulated) and T11/S15 (upregulated) of DNAJC5, S3497/S3504 (downregulated) and S2016 (upregulated) of BSN and S127 (downregulated) and T143 (upregulated) of MARCKS changed differentially in the ASD group. After exercise, the phosphorylation levels of the corresponding sites of these 3 proteins were reversed.
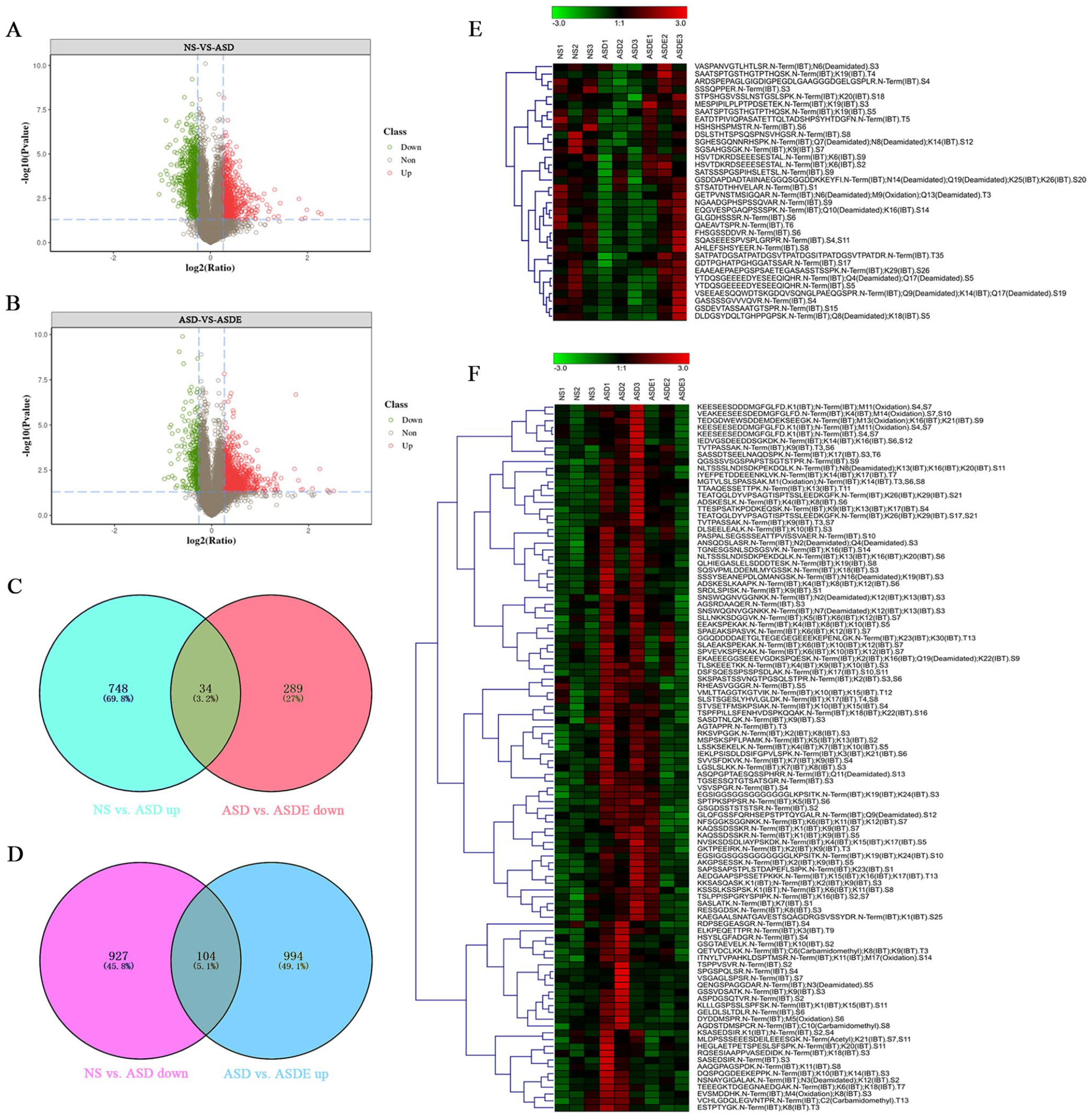
Effect of exercise training on differentially phosphorylated peptides expressed in the mPFC of VPA-induced ASD rats (n = 3 rats/group). Differentially phosphorylated peptides were defined as follows: ratio >1.2 or ≤0.83 and P ≤ .05. (A) Volcano plots of the expression of phosphorylated peptides between the NS and ASD groups. The green plots indicate downregulated phosphopeptides, red plots indicate upregulated phosphopeptides, and the grey plots indicate phosphopeptides that did not significantly differ. (B) Volcano plots of the expression of phosphorylated peptides between the ASD and ASDE groups. (C, D) Compared with the NS group, there were 782 downregulated phosphopeptides and 1031 upregulated phosphopeptides in the ASD group. Compared with the ASD group, there were 323 upregulated phosphopeptides and 1098 downregulated phosphopeptides in the ASD group. In addition, 34 phosphopeptides were downregulated in the ASD group but upregulated in the ASDE group, and 104 phosphopeptides were upregulated in the ASD group but downregulated in the ASDE group. (E) Hierarchical clustering diagram of differentially phosphorylated peptides meeting the criteria of downregulation in the ASD group (compared with the NS group) and upregulaton in the ASDE group (compared with the ASD group). Each row represents a phosphopeptide, and the colors represent the phosphorylation levels of the peptides, demonstrating that the higher the phosphorylation level, the deeper the red color; the lower the phosphorylation level, the deeper the green color. (F) Hierarchical clustering diagram of differentially phosphorylated peptides meeting the criteria of upregulation in the ASD group (compared with the NS group) and downregulation in the ASDE group (compared with the ASD group).
Effect of Exercise Training on the Function of Differential Phosphoproteins Expressed in the mPFC of VPA-Induced ASD Rats
To further study the neurobiological mechanism by which exercise training alleviates the core symptoms of VPA-induced ASD rats, GO enrichment, COG annotation, and KEGG pathway enrichment analyses were performed to examine the 134 differential phosphoproteins that were upregulated (or downregulated) in the ASD group and downregulated (or upregulated) in the ASDE group. The GO enrichment analysis showed that the significantly enriched phosphoproteins that were both upregulated in the ASD group and downregulated in the ASDE group were mainly synaptic components (such as synapse part, neuron part, synapse, postsynaptic density, and postsynaptic specialization). Their major molecular functions were binding (such as calmodulin binding, alkali metal ion binding, microtubule binding, cytoskeletal protein binding, and ADP binding) and structural molecule activity (such as structural constituents of ribosomes, the presynaptic active zone, and synapses). Their major biological processes were nervous system development and differentiation (such as neurogenesis, generation of neurons, neuron projection development, and dendrite development) and response to stimulus (such as response to thyroid stimulating hormone and cellular response to thyroid stimulating hormone and glycoprotein; Figure 4A and C). The COG annotation showed that most of these 101 phosphoproteins were involved in cellular processes and signaling, such as signal transduction mechanisms and the cytoskeleton (Figure 4E). The KEGG pathway enrichment analysis showed that most phosphoproteins were enriched in pathways related to human diseases and organismal systems, and there were 18 significantly enriched pathways, such as those related to ribosomes, long-term potentiation and glutamatergic synapses (Figure 5A and B). Interaction networks of pathways of interest are shown in Figure 5C. The significantly enriched phosphoproteins that were both downregulated in the ASD group and upregulated in the ASDE group were mainly synaptic components (such as dendrite, postsynaptic density, postsynaptic specialization, synapses, and asymmetric synapses). Their major molecular functions were binding (such as calmodulin, protein, and GTPase binding), and their major biological processes were cellular and developmental processes (such as aging, mitotic cytokinesis, synapse organization, and the establishment of protein localization to the plasma membrane) and responses to stimuli (such as corticosterone and mineralocorticoids; Figure 4B and D). The COG annotation showed that most of these 33 phosphoproteins were also involved in cellular processes and signaling, such as signal transduction mechanisms and the cytoskeleton (Figure 4F). The KEGG pathway enrichment analysis showed that most phosphoproteins were also enriched in pathways of human diseases and organismal systems, and there were 2 significantly enriched pathways, namely, those associated with endocytosis and Parkinson’s disease (Figure 5D and E). Interaction networks of the pathways of interest are shown in Figure 5F. In addition, the results of the GO enrichment, COG annotation, and KEGG pathway enrichment analyses of the differential phosphoproteins between the NS and ASD group and between the ASD and ASDE group are shown in Supplemental Figures 2 and 3 and Supplemental Tables 3 and 4.
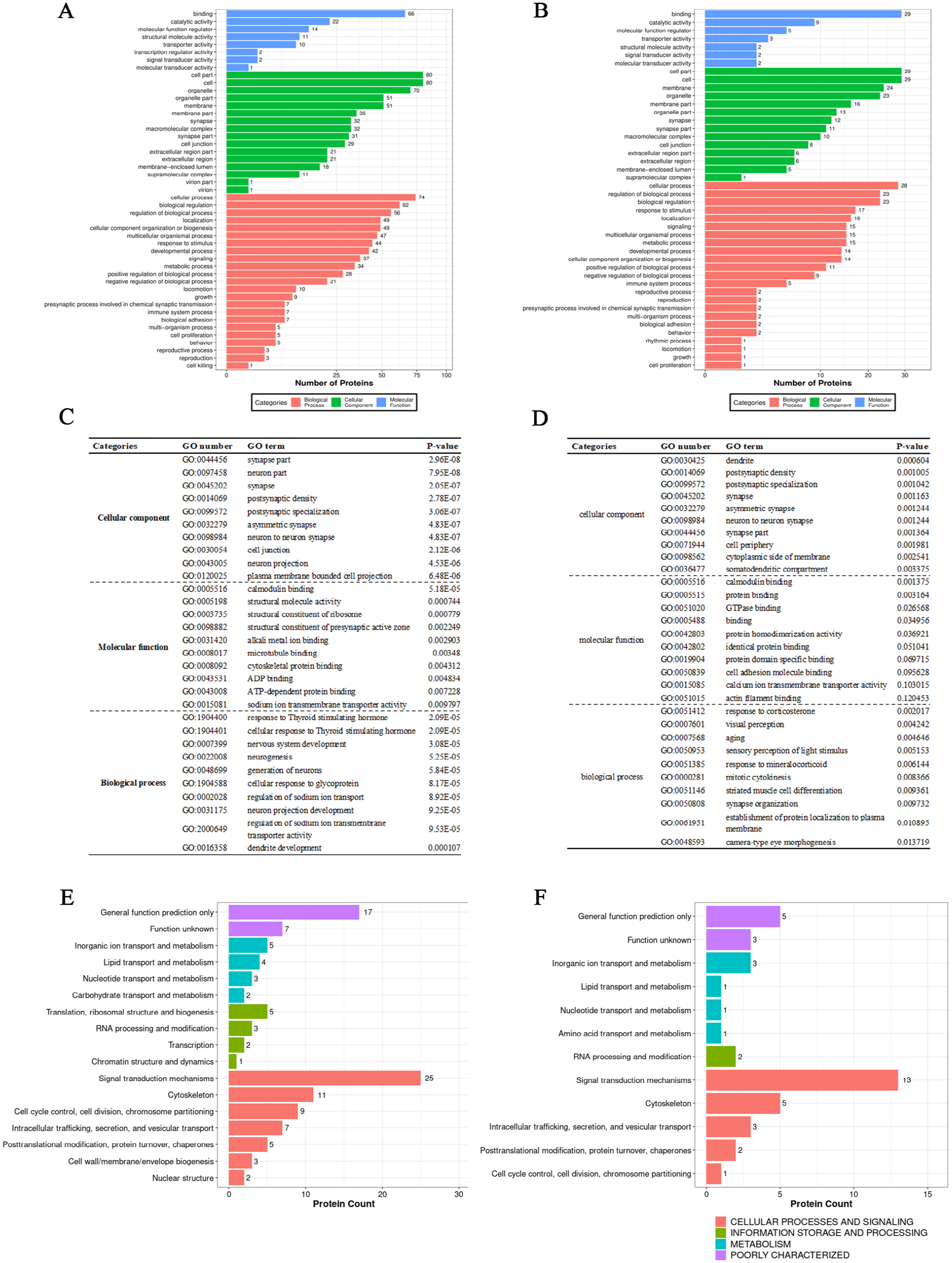
Effect of exercise training on GO and COG annotation of differential phosphoproteins of interest expressed in the mPFC of VPA-induced ASD rats (n = 3 rats/group). (A) GO annotation of the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. (B) GO annotation of the differential phosphoproteins that were downregulated in the ASD group but upregulated in the ASDE group. (C) The top 10 significantly enriched GO terms of the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. (D) The top 10 significantly enriched GO terms of the differential phosphoproteins that were downregulated in the ASD group but upregulated in the ASDE group. (E) COG annotation of the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. (F) COG annotation of the differential phosphoproteins that were downregulated in the ASD group but upregulated in the ASDE group.
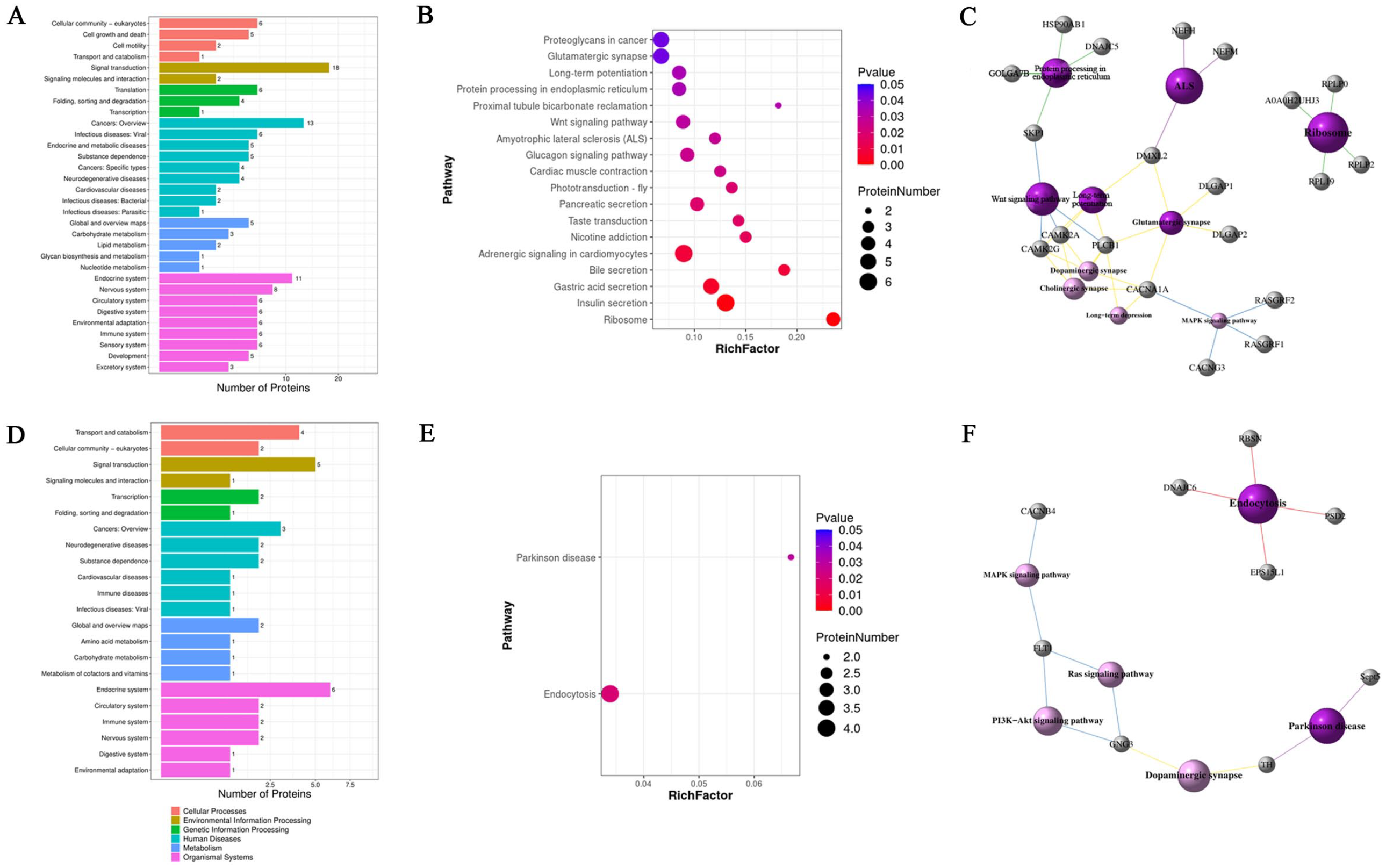
Effect of exercise training on the KEGG enrichment pathway of the differential phosphoproteins of interest expressed in the mPFC of VPA-induced ASD rats (n = 3 rats/group). (A) Overview of the KEGG enrichment pathway of the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. (B) The significantly enriched KEGG pathway terms of the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. The size of the circle indicates the number of phosphoproteins enriched in a pathway, and the color indicates the significance of the pathway. (C) Pathway interaction networks of differential phosphoproteins of interest that were upregulated in the ASD group but downregulated in the ASDE group. The dark purple sphere represents a significantly enriched pathway, the light purple sphere represents a nonsignificantly enriched pathway, and the gray sphere represents a differentially phosphorylated protein. Different colored lines represent different classifications of pathways; red represents cellular processes, blue represents environmental information processing, green represents genetic information processing, purple represents human diseases, and yellow represents organismal systems. (D) Overview of the KEGG enrichment pathways of the differential phosphoproteins that were downregulated in the ASD group but upregulated in the ASDE group. (E) The significantly enriched KEGG pathway terms of the differential phosphoproteins that were downregulated in the ASD group but upregulated in the ASDE group. (F) Pathway interaction networks of differential phosphoproteins of interest that were downregulated in the ASD group but upregulated in the ASDE group.
Effect of Exercise Training on the Phosphorylation Motifs of the Differential Phosphoproteins Expressed in the mPFC of VPA-Induced ASD Rats
To gain deeper insight into the upstream pathways, a motif analysis with the Motif-X algorithm was performed using MoMo software between the −7 and +7 positions of the serine (S), threonine (T), and tyrosine (Y) phosphorylation sites in the differentially phosphorylated proteins in the NS, ASD, and ASDE groups. Seven motifs (the top 3 motifs of significance, including SPxx, SxxxSP, and SPxxxxR) were discovered in the upregulated S-site phosphorylated proteins, and 7 motifs (the top 3 motifs of significance, including SPxx, SPxK, and SPxxxK) were discovered in the downregulated S-site phosphorylated proteins in the NS versus ASD group. Fourteen motifs (the top 3 motifs of significance, including SPxx, SPxxxxS, and RxxS) were discovered in the upregulated S-site phosphorylated proteins, and 3 motifs (including SPxx, SxxxSP, and RxxS) were discovered in the downregulated S-site phosphorylated proteins in the ASD vs. ASDE group (Figure 6A). One TPxx motif was detected among the upregulated and downregulated T-site phosphorylated proteins in the NS versus ASD and ASD versus ASDE groups (Figure 6B). No motif was detected among the upregulated and downregulated Y-site phosphorylated proteins in the NS versus ASD and ASD versus ASDE groups because there were few Y-site phosphorylated proteins. Among these detected motifs, the SPxx and TPxx motifs are known to be WW domain binding motifs and are frequently involved in gene regulatory proteins. The SPxx motif is phosphorylated by mitogen-activated protein kinases (MAPKs), extracellular regulated protein kinases (ERKs), and cyclin-dependent protein kinases (CDKs).29 -31 The SxxxSP motif is phosphorylated by glycogen synthase kinase-3 (GSK-3), 32 and the SPxK and SPxR motifs are phosphorylated by CDKs and growth-associated histone H1 kinase.33,34 The SxE motif is phosphorylated by casein kinase Fam20C, 35 the RxxS motif is phosphorylated by protein kinase A (PKA) and ribosomal protein S6 kinase II,36,37 the RxxSxP motif is phosphorylated by testicular protein kinase 1 (TESK1), 38 the SxxE and SDxE motifs are phosphorylated by casein kinase II,39,40 and the SDxx motif is phosphorylated by tank-binding kinase 1 (TBK1). 41 In addition to the above known protein kinase motifs, there were 12 novel motifs, namely, SPxA, SPxxxK, SPxxxxS, SPxxxxR, SPxxxP, SPxxxxK, DxSP, SPTxx, RxxSxS, SPxG, SExE, and SLxx. Interestingly, these unknown motifs were mostly detected in the downregulated phosphorylated proteins in the NS versus ASD group and the upregulated phosphorylated proteins in the ASD versus ASDE group, indicating that some uncharacterized kinase activity may play a role in the development of ASD and the intervention effect of exercise on ASD. To thoroughly explore the upstream pathways of the potential targets of ASD exercise intervention, we performed a motif analysis of the 134 differential phosphoproteins expressed in the VPA-induced ASD rats after exercise. One SPxx motif was detected among the S-site phosphoproteins, and no motif was detected among the T-site or Y-site phosphoproteins, which were upregulated in the ASD group but downregulated in the ASDE group. There were 29 phosphopeptide sequences containing the SPxx motif, and their corresponding phosphoproteins were involved in synapses, except for DCLK1 (S307), SRRM1 (S429), SLC9A1 (S697), and LOC317456 (S131; Figure 6C). In addition, no motif was detected among the S-site, T-site, or Y-site phosphoproteins, which were downregulated in the ASD group but upregulated in the ASDE group.
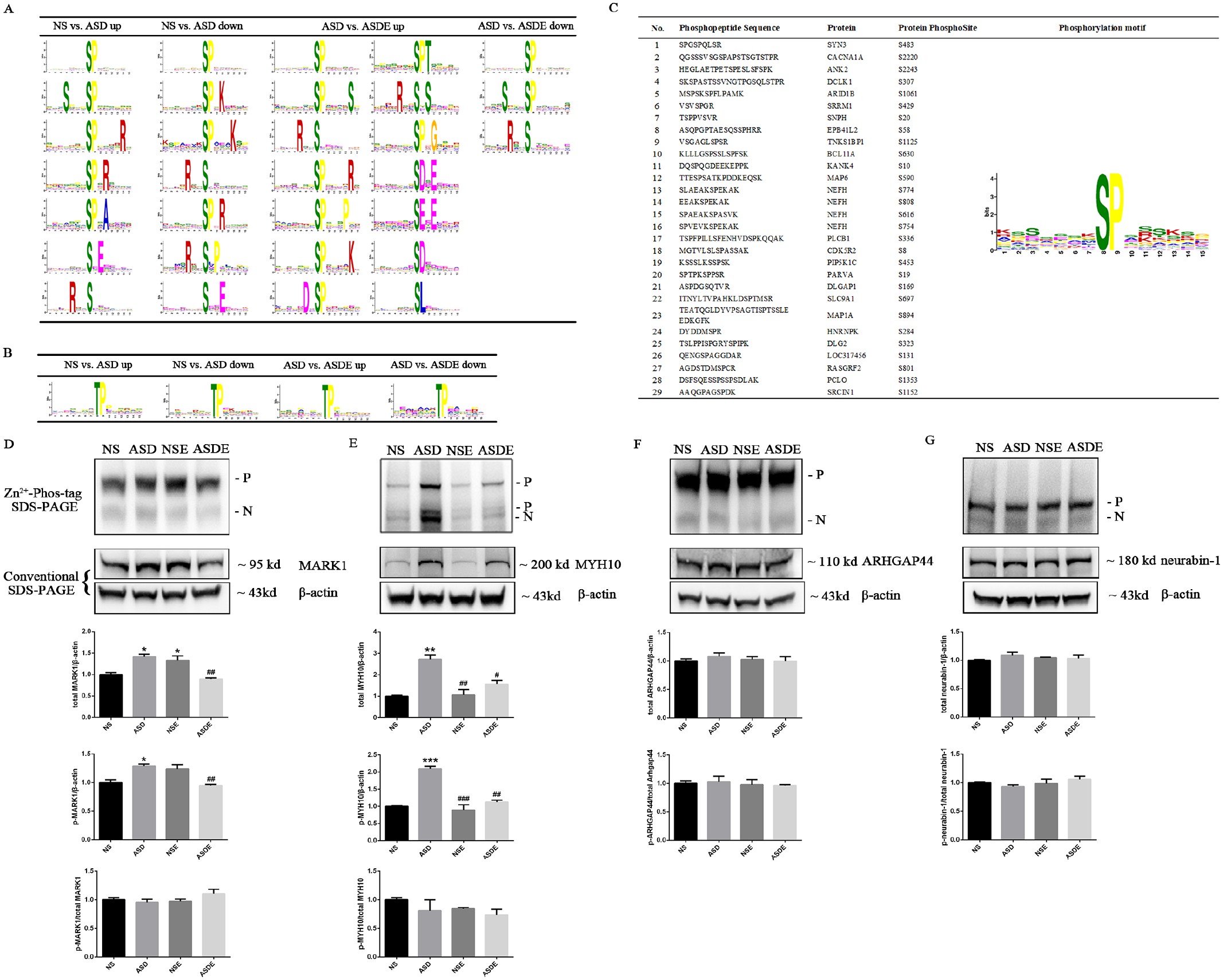
Effect of exercise training on the phosphosite motifs of differential phosphoproteins expressed in the mPFC of VPA-induced ASD rats and the expression of total protein and phosphoproteins of MARK1, MYH10, ARHGAP44, and neurabin-1 (n = 3 rats/group). (A) Logo-like representations of significantly enriched S-site motifs identified from differential phosphoproteins in the NS, ASD, and ASDE groups. The height of the residues represents the frequency of their occurrence at the respective positions. (B) Logo-like representations of significantly enriched T-site motifs identified from differential phosphoproteins in the NS, ASD, and ASDE groups. (C) The significantly enriched S-site motif identified from the differential phosphoproteins that were upregulated in the ASD group but downregulated in the ASDE group. (D and E) The total protein and phosphoproteins of MARK1 and MYH10 expressed in the ASD group were significantly increased and then decreased after the exercise training. (F and G) There was no significant difference in the expression of the total protein and phosphoproteins of ARHGAP44 and neurabin-1 among the groups.
Effect of Exercise Training on the Expression of MARK1, MYH10, ARHGAP44, and Neurabin-1 in the mPFC of VPA-Induced ASD Rats
Our phosphoproteomics results showed that the phosphoproteins MARK1 and MYH10 were upregulated and ARHGAP44 and neurabin-1 were downregulated in the mPFC of ASD rats and reversed after exercise training (Supplemental Tables 1 and 2). As these phosphoproteins are related to ASD and synapse plasticity,18,42 -46 we utilized conventional and Phos-tagged SDS-PAGE Western blot analyses to detect the total and phosphorylated levels according to previous studies.47,48 We found that in line with the phosphoproteomics results, the total protein and phosphoproteins of MARK1 and MYH10 expressed in the ASD group were significantly increased and then decreased after the exercise training (Figure 6D and E). However, there was no significant difference in the expression of the total protein and phosphoproteins of Arhgap44 and neurabin-1 among the groups (Figure 6F and G).
Discussion
In this study, we first established an exercise intervention protocol in young ASD rats and showed that 4 weeks of wheel running and swimming in 28-day-old VPA-induced ASD rats effectively improved their social deficits and short-term novel object cognition and alleviated repetitive behavior and anxiety. In addition, we observed that the density, morphology, and ultrastructure of synapses in the PrL and IL subregions of the mPFC changed differentially after the exercise intervention. Furthermore, exercise training induced extensive changes in the phosphorylation level of proteins in the mPFC in the ASD rats, and the key differential phosphoproteins such as MARK1 and MYH10 were mainly related to synapse plasticity.
Children under the age of 6 years with ASD should undergo rehabilitation training, such as educational and behavioral interventions, to obtain a favorable prognosis as the strongest developmental plasticity occurs during this period. 49 Exercise, such as jogging, swimming, and participating in ball games and martial arts, can effectively improve deficiencies in children’s social communication and stereotyped repetitive behavior.5 -7 To explore the neurobiological mechanism by which exercise training alleviates behavioral deficits in ASD children, we first established an exercise intervention protocol in VPA-induced young male ASD rats. Maternal exposure to VPA during pregnancy is a classic rodent model of ASD in which offspring mimic typical behavioral abnormalities in ASD children, and the degree of neuropathological changes depends on the dose and duration of VPA.25,26 In addition, in line with a clinical study that showed that boys have a 3 to 4 times higher risk of ASD than girls, male VPA-induced autism model rats have more obvious ASD-like behavioral phenotypes than females.2,27 In our study, we found that the eye-opening time of ASD offspring rats did not significantly differ from that of NS offspring. However, the ASD offspring rats exhibited weight loss, hypoalgesia, deficits in social communication and communication ability, repetitive behavior, anxiety, and cognitive memory impairment, which successfully simulated the typical behavioral deficits of ASD children.1,2 Following the combined intervention of multiple exercise schemes in the clinical treatment of ASD children,5 -7 we used wheel running combined with swimming to exercise young (4 weeks) VPA-induced ASD rats. We found that this exercise scheme effectively reduced the social disorder and repetitive behavior of the ASD rats and improved anxiety and NOR disorder, which laid a foundation for us to further study the neurobiological mechanism of ASD-related behavioral deficit rehabilitation through exercise training.
Synapses are the key connections between neurons. The pyramidal neurons in the PrL and IL receive functional inputs from limbic and cortical regions and play different roles in signal processing. Although PrL and IL allow actions to be controlled proactively, they play opposing roles in controlling premature responses. Compared with PrL neurons, IL neurons exhibit a higher frequency of local field potentials.8,10 In addition, the dysfunctional connectivity between the mPFC and other brain regions in ASD indicates that synapses have undergone pathological changes. 50 In accordance with these studies, we determined the differential regulation of synaptic density in the PrL and IL of ASD rats, which may result from the different functions of these 2 subregions. After the exercise training, the abnormally increased total synaptic density in the PrL and the abnormally decreased total synaptic density in the IL of the ASD rats were corrected. It has been reported that synapse function is closely related to both synaptic density and synaptic ultrastructure, such as the width of the synaptic cleft, the length of the active zone and the PSD thickness.51,52 We found that exercise reversed the ASD-induced decrease in the width of the synaptic cleft and the increased thickness of the PSD in the PrL, indicating that exercise synergistically regulated the synaptic density and ultrastructure to protect against abnormally enhanced neuronal signal transmission in the PrL that was ASD-induced. However, exercise reversed the ASD-induced decrease in the width of the synaptic cleft and the increase in the length of the active zone in the IL, which is inconsistent with the change in synaptic density, indicating the necessity of electrophysiological studies to determine the effects of ASD- and exercise-induced synaptic structural plasticity on synapse function.
According to their morphology, synapses can be divided into smile synapses, frown synapses, and flat synapses. The shape of a smile synapse is caused by the presynaptic density being pushed into the postsynaptic element, while the frown synapse is caused by the opposite effect. The flat synapse shape occurs because the presynaptic density is parallel to the postsynaptic element. 53 In this study, we found that the increased density of frown synapses in the PrL and the decreased density of smile synapses in the IL of the ASD rats were also reversed by exercise. Different synapse shapes may reveal the location of postsynaptic terminals. Synapses on dendritic shafts or spine heads may be flat, synapses on soma or spine heads may have a frown shape, and synapses on spine necks may have a smile shape. The shape of synapses is also affected by the addition of presynaptic dense elements, synaptic cytoskeletal activation, transmitters, etc. 53 Although it is difficult to determine how exercise differentially regulates the synaptic shape in the 2 subregions in ASD rats, synaptic plasticity signaling must be involved.
It has been reported that the core symptoms of ASD are linked to abnormalities in brain structure and function in the mPFC, such as an increased volume of the prefrontal lobe, weaker activity of mirror neurons, and increased density of dendritic spines. 54 An increasing number of ASD-associated genes, such as NRXN1, SHANK, NLGN, MYH10, and SYNGAP1, are related to synaptic maturation and function.15 -18 These studies imply that abnormal synaptic structure and function are important pathological features of ASD. Because de novo likely gene-disrupting (LGD) mutations are easier to explain than missense mutations, studies usually limit their focus to LGD, which mostly causes the downregulation and loss of function of target proteins. However, it is difficult to pinpoint the specific contribution of each pathway associated with this mutant protein due to the confounding effects of other dysfunctional pathways in the same model. 55 As the abnormal phosphorylation of proteins also plays an important role in the development of ASD,23,24 in-depth studies could enhance our understanding of the signaling pathways related to ASD and identify possible therapeutic targets. However, to the best of our knowledge, no study has focused on the mPFC in ASD to determine the potential abnormal phosphoproteins involved in synapses, and knowledge regarding the changes in phosphoprotein expression in ASD after exercise training is limited. Using quantitative phosphoproteomics, we found that exercise can reverse some of the abnormal protein phosphorylation in the mPFC of VPA-induced ASD rats, which is closely related to synapse plasticity. These results reflected the changes in synaptic structure observed by electron microscopy at the molecular level. Notably, our study provides potential targets relevant to the study of exercise-induced neural mechanisms that improve ASD behavioral deficits. For example, neurofilament heavy polypeptide (NEFH) can cause an abnormal neuronal cytoskeleton and has been found to be abnormally expressed in humans with ASD. 56 MAP1A, a new exome-wide significant gene related to the risk of ASD, binds along the microtubule lattice and acts as the dominant protein in the formation and development of axons and dendrites. 57 MAP6 regulates microtubule stabilization and synaptic plasticity. It has been proposed that reduced MAP6 may impair oligodendrocyte myelination and synaptic function in neurons to mediate ASD-associated behaviors. However, a systematic investigation of how MAP1A is involved in mediating ASD-associated behaviors is lacking. 17 Interestingly, MARK1 regulates microtubule dynamics and stability by phosphorylating MAPs. 42 MARK1 was reported to be overexpressed in the prefrontal cortex in postmortem brain tissues from ASD patients. 46 MYH10 is strongly associated with ASD and is required for the formation and maturation of dendritic spines and excitatory synaptic transmission.18,45 Voltage-dependent P/Q-type calcium channel subunit α (CACNA1A) is predominantly expressed in the frontal cortex and plays key roles in the balance of excitatory/inhibitory inputs. Mutants of CACNA1A impair synaptic transmission and cognizance and have been found in ASD patients of different ethnicities.58,59 Neurabin-1 is an F-actin-binding protein that recruits protein phosphatase 1 (PP1) to synapses to promote AMPAR dephosphorylation during long-term depression (LTD), weakening the synaptic strength. 44 ARHGAP44, also known as Rich2, is a new Shank3 partner, and the Shank3-Rich2 interaction regulates spine morphology and synaptic plasticity. 43 In accordance with these studies, our results suggest that the differential phosphoproteins that contain the SPxx motif, such as NEFH, MAP1A, MAP6, and CACNA1A, are involved in the pathogenesis of ASD and that the signaling pathways of MAPKs, ERKs, or CDKs may be involved in the mechanism by which exercise alleviates the behavioral deficits and abnormal synaptic structure of ASD. In addition, we preliminarily verified that the phosphorylation of MARK1 and MYH10, but not ARHGAP44 or neurabin-1, is consistent with that of phosphoproteomics. The reason for the failure to verify the phosphorylation of ARHGAP44 and neurabin-1 by phos-tagged SDS-PAGE Western blot may be that the expression of other phosphopeptides overwhelmed the detection of target phosphopeptides, making the significantly differential levels of the phosphopeptides inconsistent with the total phosphoprotein levels. Further investigations are needed, such as constructing simulated phosphorylation and nonphosphorylation mutants of target phosphopeptides, to clarify the expression and function of specific phosphorylated sites and their possible upstream and downstream signals.
Our study provides a rat model for studying the mechanism of the exercise rehabilitation of ASD-induced behavioral deficits. We found that exercise training differentially changed the synaptic density, morphology and ultrastructure in the PrL and IL in ASD rats. In addition, we comprehensively analyzed the changes in phosphorylated proteomics in the mPFC, which may provide potential targets and signaling pathways for further studies investigating the neural mechanism by which exercise alleviates core ASD symptoms. In summary, our results suggest that exercise may change the synaptic density, morphology, and ultrastructure in the mPFC by reversing various abnormally phosphorylated proteins induced by ASD, including synaptic-related proteins, such as MARK1 and MYH10, and eventually improves behavioral deficits.
Supplemental Material
sj-doc-2-nnr-10.1177_15459683231152814 – Supplemental material for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid
Supplemental material, sj-doc-2-nnr-10.1177_15459683231152814 for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid by Genghong Tu, PhD, Youli Guo, PhD, Ruoshi Xiao, MD, Lianying Tang, MD, Min Hu, PhD and Bagen Liao, PhD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231152814 – Supplemental material for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid
Supplemental material, sj-docx-1-nnr-10.1177_15459683231152814 for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid by Genghong Tu, PhD, Youli Guo, PhD, Ruoshi Xiao, MD, Lianying Tang, MD, Min Hu, PhD and Bagen Liao, PhD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-3-nnr-10.1177_15459683231152814 – Supplemental material for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid
Supplemental material, sj-tif-3-nnr-10.1177_15459683231152814 for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid by Genghong Tu, PhD, Youli Guo, PhD, Ruoshi Xiao, MD, Lianying Tang, MD, Min Hu, PhD and Bagen Liao, PhD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-4-nnr-10.1177_15459683231152814 – Supplemental material for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid
Supplemental material, sj-tif-4-nnr-10.1177_15459683231152814 for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid by Genghong Tu, PhD, Youli Guo, PhD, Ruoshi Xiao, MD, Lianying Tang, MD, Min Hu, PhD and Bagen Liao, PhD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-5-nnr-10.1177_15459683231152814 – Supplemental material for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid
Supplemental material, sj-tif-5-nnr-10.1177_15459683231152814 for Effects of Exercise Training on the Phosphoproteomics of the Medial Prefrontal Cortex in Rats With Autism Spectrum Disorder Induced by Valproic Acid by Genghong Tu, PhD, Youli Guo, PhD, Ruoshi Xiao, MD, Lianying Tang, MD, Min Hu, PhD and Bagen Liao, PhD in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
LB, HM, and TG designed the study and revised the manuscript. TG and GY wrote the manuscript. TG, GY, XR, and TL performed the experiments and analyzed the data. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (32000837 to G Tu), the University Scientific Research Project of Guangdong Provincial Department of Education (2019KQNCX065 and 2020KTSCX067 to G Tu), and the Guangdong Provincial Key Laboratory of Physical Activity and Health Promotion (2021B1212040014 to M Hu).
Ethics Approval and Consent to Participate
All procedures performed in this study were in accordance with the ethical standards of the Institutional Review Board of the Guangzhou Sport University, Guangzhou, Guangdong, China.
Availability of Data and Materials
The datasets for this study are available from the corresponding author upon reasonable request.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
