Abstract
Background:
Neuralgic amyotrophy (NA) is a common peripheral nerve disorder caused by auto-immune inflammation of nerves in the brachial plexus territory, characterized by acute pain and weakness of the shoulder muscles, followed by motor impairment. Recent work has confirmed that NA patients with residual motor dysfunction have abnormal cerebral sensorimotor representations of their affected upper extremity.
Objective:
To determine whether abnormal cerebral sensorimotor representations associated with NA can be altered by specialized, multidisciplinary outpatient rehabilitation focused on relearning motor control.
Methods:
27 NA patients with residual lateralized symptoms in the right upper extremity participated in a randomized controlled trial, comparing 17 weeks of multidisciplinary rehabilitation (n = 16) to usual care (n = 11). We used task-based functional MRI and a hand laterality judgment task, which involves motor imagery and is sensitive to altered cerebral sensorimotor representations of the upper extremity.
Results:
Change in task performance and related brain activity did not differ significantly between the multidisciplinary rehabilitation and usual care groups, whereas the multidisciplinary rehabilitation group showed significantly greater clinical improvement on the Shoulder Rating Questionnaire. Both groups, however, showed a significant improvement in task performance from baseline to follow-up, and significantly increased activity in visuomotor occipito-parietal brain areas, both specific to their affected upper extremity.
Conclusions:
Abnormal cerebral sensorimotor representations of the upper extremity after peripheral nerve damage in NA can recover toward normality. As adaptations occurred in visuomotor brain areas, multidisciplinary rehabilitation after peripheral nerve damage may be further optimized by applying visuomotor strategies. This study is registered at ClinicalTrials.gov (NCT03441347).
Keywords
Introduction
When components of the sensorimotor system are injured, motor function can be recovered through adaptations within the nervous system. 1 However, these plastic adaptations are not always beneficial to clinical outcome, and can even contribute to subsequent motor dysfunction. 1 For example, it has been suggested that functional changes in the somatosensory cortex after limb amputation may contribute to phantom limb pain, and in other instances to focal dystonia.2,3 Such maladaptive plasticity has been connected to persistent motor impairment in several central and peripheral nervous system disorders. 1 Reversal of maladaptation following injury may form a promising avenue for rehabilitation, especially in peripheral nervous system disorders where central structures are intact.
One such disorder is neuralgic amyotrophy (NA): a common (incidence of 1:1000) and disabling peripheral nerve disorder, characterized by acute autoimmune inflammation of the nerves in the brachial plexus territory, which leads to paresis of muscles innervated by damaged nerves.4,5 NA is typically asymmetric, most often affecting one upper extremity. 6 To compensate for muscle weakness, many patients develop abnormal movement patterns, most notably of the shoulder (i.e., scapular dyskinesia), which can be beneficial at first, but lead to long-term motor dysfunction and subsequent residual complaints of the affected limb, even with (partial) peripheral recovery.5,7 Recently, we have empirically confirmed our clinical suspicion5,8 that NA patients with peripheral nerve damage and residual motor dysfunction have abnormal cerebral sensorimotor representations related to their affected limb and residual symptoms. 9 The objective of the current study is to determine whether the cerebral abnormalities associated with NA can be modified by specialized, multidisciplinary outpatient rehabilitation,5,10 as compared to usual care. Whereas usual care commonly involves conventional physical therapy focused on regaining strength and endurance,5,7 multidisciplinary rehabilitation combines occupational and physical therapy, and focusses on improving self-management strategies and relearning motor control to normalize scapular movement and stability.5,10 We hypothesize that multidisciplinary rehabilitation, but not usual care, can modify abnormal cerebral sensorimotor representations of the affected upper extremity. As a secondary objective, we explore how cerebral changes relate to changes in clinical symptoms: functional capability of the upper extremity, and persistent pain, which strongly relates to scapular dyskinesia in NA.5,7,11
To reach our objectives, we use a combination of task-based functional MRI (fMRI) and the hand laterality judgment task, for its ability to activate sensorimotor representations of the upper extremity12 -14 and its sensitivity to cerebral and behavioral adaptations in NA.9,15 In this validated task, patients judge the laterality (left or right) of hand stimuli, by mentally rotating their own body part to match the stimulus. This involves “motor imagery” (i.e., mental simulation of movement) and engages sensorimotor processes similar to motor planning.14,16 The embodied nature of the task is confirmed by the fact that subjects incorporate their own body posture.17,18 Motor imagery activates a fronto-parieto-occipital network involving key motor areas such as the supplementary motor area and premotor cortex, as well as visuomotor areas in the posterior parietal cortex and along the dorsal visual stream.16,17,19,20 This activation is typically more pronounced for imagined movements rotating away from the body midline, which pose complex biomechanical constraints, compared to movements rotating toward the body midline that are easier to perform as they pose fewer biomechanical constraints.12,13,17 We have previously shown, in a companion cross-sectional comparison to healthy participants, that NA patients with persistent motor dysfunction had decreased activity related to their affected limb in two visuomotor brain regions in the occipito-parietal cortex, especially when biomechanically complex (imagined) movement was involved. 9 In the current study, we focus on these same visuomotor regions of interest (ROIs), to determine whether multidisciplinary rehabilitation (compared to usual care) can lead to increased task-related activity in these regions.
Methods
This study is part of a randomized controlled trial (RCT) investigating the effect of multidisciplinary rehabilitation on residual complaints and cerebral mechanisms in NA, for which the primary outcome measure is functional capability of the upper extremity (quantified with the Shoulder Rating Questionnaire; SRQ). 8 All MRI-compatible RCT participants were included in this sub-study. In this sub-study, the primary objective was to determine if patients with NA have altered cerebral activity related to motor planning of their affected arm, compared to healthy controls and compared to their non-affected arm. 8 To this end, we focused on behavioral performance and cerebral activity associated with the hand laterality judgment task. We originally expected to find changes in parietal and occipital brain regions, as well as postcentral and precentral gyri. 8 These a priori regions of interest (ROIs) were further specified to the right extrastriate cortex and bilateral parieto-occipital sulcus, where we recently found reduced imagery-related brain activity in NA patients versus healthy controls in the baseline measurement. 9 In addition to these ROI analyses, we performed an exploratory whole-brain search. We also included relevant clinical data collected as part of the RCT to relate cerebral adaptations to changes in symptom severity. The clinical data of the full sample of participants will be published in a parallel study. A previous study compared the baseline task-fMRI of the NA patients to a group of healthy participants. 9 The local medical ethical committee (Medical Ethical Committee region Arnhem-Nijmegen, CMO 2017-3740) approved this study and the study is registered at ClinicalTrials.gov (NCT03441347).
Participants
The full RCT was powered 6 to demonstrate the effect of specialized multidisciplinary rehabilitation compared to usual care on a clinical measure: the functional capability of the upper extremity as measured with the Shoulder Rating Questionnaire Dutch Language Version (SRQ-DLV). 21
We included 47 patients with a right-sided NA of the brachial plexus as part of the RCT. NA was diagnosed by an experienced neurologist (NvA) in patients with a history of acute onset severe, numerical rating scale (NRS) ≥7, upper extremity pain combined with scapular dyskinesia during abduction–anteflexion and muscle paresis of serratus anterior, shoulder exorotation, and long flexor of the thumb. 4 Due to the COVID-19 pandemic and subsequent national measures, inclusion was terminated short of the original goal of 50 patients. Forty-four patients underwent an fMRI scan during the pre-treatment baseline assessment. See Figure 1 for an overview of exclusions and drop-out throughout the study. Drop-outs were mostly caused by recurrent NA attacks, COVID-19-related restrictions or personal reasons. The final data set came from 27 patients in this sub-study: 16 in the multidisciplinary rehabilitation group and 11 in the usual care group (see Table 1 for participant characteristics).
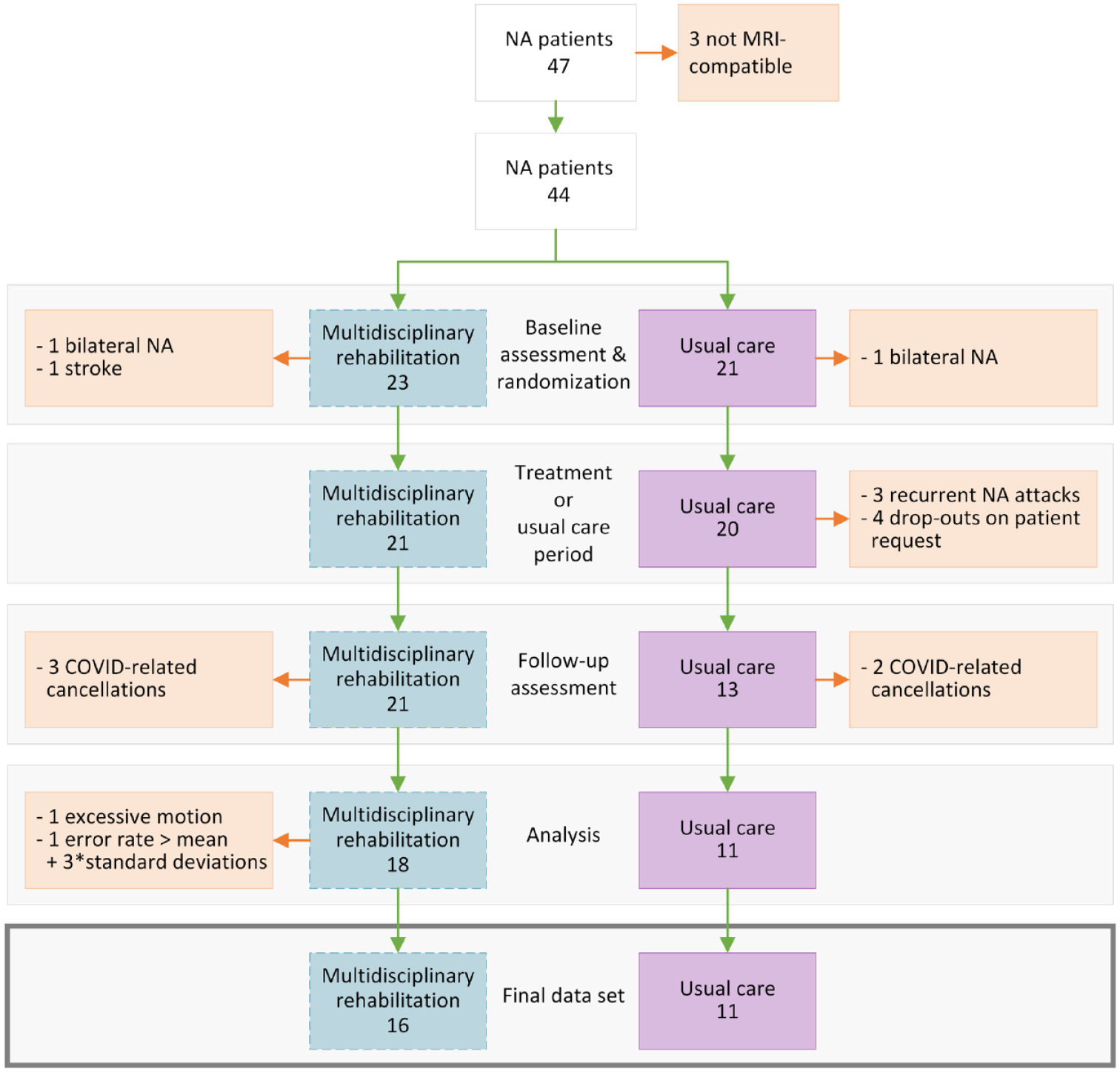
Flow diagram patients showing how many patients were included and excluded at each stage of the study and the reasons. For the three patients excluded at the baseline assessment/randomization stage, it became later apparent that they met an exclusion criterion (two showed bilateral involvement of NA, and for one a history of stroke was missed at the initial screening). Patients were excluded from further analyses if the mean framewise displacement during one or both of the fMRI scans exceeded 0.5 mm (excessive motion), or if their mean error rate was more than three times the standard deviation above the group mean. Five follow-up assessments were cancelled due to government-mandated closing of our facilities during the COVID-19 pandemic.
Participant Characteristics of the Patients that were Included in the Final Analyses.
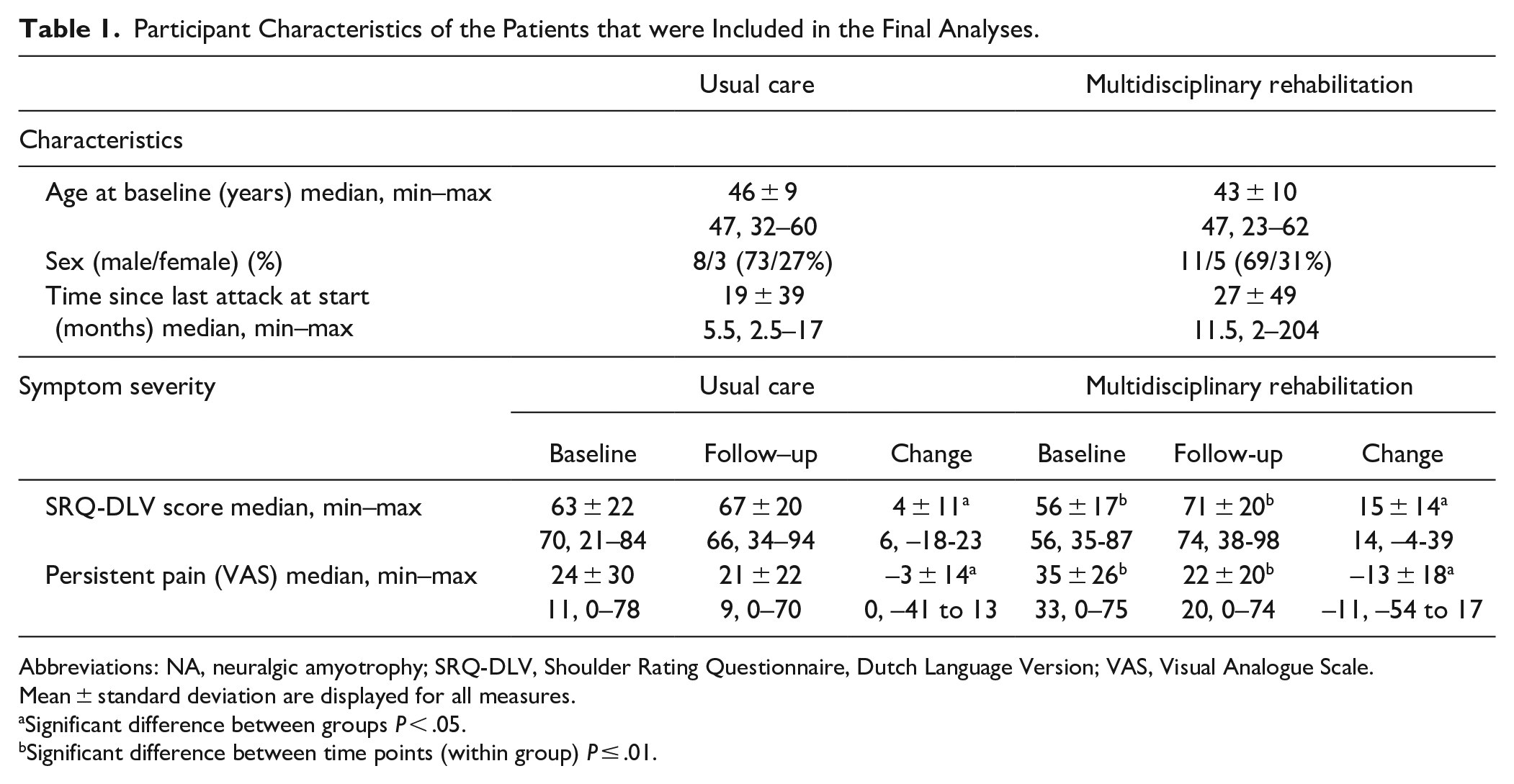
Abbreviations: NA, neuralgic amyotrophy; SRQ-DLV, Shoulder Rating Questionnaire, Dutch Language Version; VAS, Visual Analogue Scale.
Mean ± standard deviation are displayed for all measures.
Significant difference between groups P < .05.
Significant difference between time points (within group) P ≤ .01.
NA patients were recruited through the Radboud University Medical Center’s Neuromuscular Center between March 1, 2018 and March 16, 2020 and were included if they met the following criteria: presenting with clearly lateralized symptoms of the right upper extremity; exhibiting explicit coordinative motor dysfunction (scapular dyskinesia); no longer in the acute inflammatory phase (>8 weeks after attack onset); no prior participation in the multidisciplinary rehabilitation program; no relevant comorbidities; ≥18 years of age; and right hand dominance (score of > +40 on the Edinburgh Handedness Inventory). 22 Patients were excluded from further participation if they experienced a recurrent NA attack during the treatment period. All MRI assessments took place between April 1, 2018 and August 1, 2020.
Procedures
Patients were informed about the procedures of the study and gave written informed consent prior to their participation, in accordance with the declaration of Helsinki. All patients underwent a baseline assessment, followed by a 17-week treatment period and a follow-up assessment. Patients were first randomized into either the multidisciplinary rehabilitation group or usual care group in a 1:1 ratio, considering age and sex (see Lustenhouwer et al 8 for details), after baseline assessment. Patients in the multidisciplinary rehabilitation group received a diagnostic visit at our outpatient Plexus Clinic resulting in a personalized rehabilitation treatment advice and plan. This was followed by a 16-week outpatient multidisciplinary rehabilitation program carried out in 4-weekly treatment sessions followed by two sessions every other week and two monthly sessions. Each of these eight treatment sessions involved 1 hour of physical therapy and 1 hour of occupational therapy, with both disciplines working closely together, exchanging clinical findings and strategies when possible and appropriate to advance the treatment process. The multidisciplinary rehabilitation combines motor learning principles to normalize scapular stability and coordination with self-management strategies for pain and fatigue to enable daily occupations and reduce persisting pain and fatigue.5,8,10 The focus and extent to which each of these components were addressed within the intervention depended on individual patient needs. Patients in the usual care group received their usual care for 17 weeks, which differed per patient, consisting of no (reported) therapy (n = 6), four (n = 1), ten (n = 1), or thirteen (n = 1) times physical therapy, or 29 times physical therapy and 4 times occupational therapy (n = 1). For one patient, the content of usual care was unknown, as she was lost to follow-up. See also Supplemental Table 1 for details.
We performed the same measurements in the same order at baseline and follow-up. All participants first completed the MRI session, which involved a structural scan, a resting-state functional MRI (fMRI) scan (data not included here), and a task-fMRI scan (see below). Following scanning, we performed several clinical assessments (see Lustenhouwer et al 8 ) to quantify residual NA symptoms. Here, we focused on two of the clinical measures. First, patients completed the SRQ-DLV, which measured the functional capability of the upper extremity, where lower scores reflected more impairment (range 17–100). 21 Second, patients indicated how much pain they were currently experiencing on a visual analogue scale (VAS), which ranged from no pain to unbearable pain (0–100). 23
Experimental Design
All patients performed a hand laterality judgment task 12 in the fMRI scanner. Patients had to indicate whether the stimulus (a white line drawing of a hand, presented on a black background), represented a left or a right hand by pressing a button with the big toe of the corresponding foot. They were instructed to respond as quickly and accurately as possible and to use their own hands as reference (such that they could imagine moving their limb to match the hand stimulus), without overt movement of their limb (compliance was checked with EMG over the thenar eminence of both hands). At the start of each block, participants assumed one of four possible postures (both hands palms facing down or up, or one hand palm down [left/right] and the other palm up [right/left]). Stimuli varied in LATERALITY (left, right), BIOMECHANICAL COMPLEXITY (complex, easy), and POSTURAL CONGRUENCY (congruent, incongruent) (see Figure 2). BIOMECHANICAL COMPLEXITY relates to the (imagined) movement needed to match the orientation of the own hand to that of the stimulus. Biomechanically complex trials (laterally oriented hands, rotated away from the body midline) typically elicit longer reaction times (RTs) and more brain activity in the fronto-parieto-occipital motor imagery network than biomechanically easy trials (medially oriented hands, rotated toward the body midline), even though the degree of rotation is matched.12,13 The POSTURAL CONGRUENCY of the stimulus with the current posture of the participant’s corresponding limb (Figure 2B) tests whether participants perform first-person motor imagery: i.e. whether stimuli that are incongruent with their own body posture elicit longer RTs and more activation in parietal areas compared to congruent postures.16,17
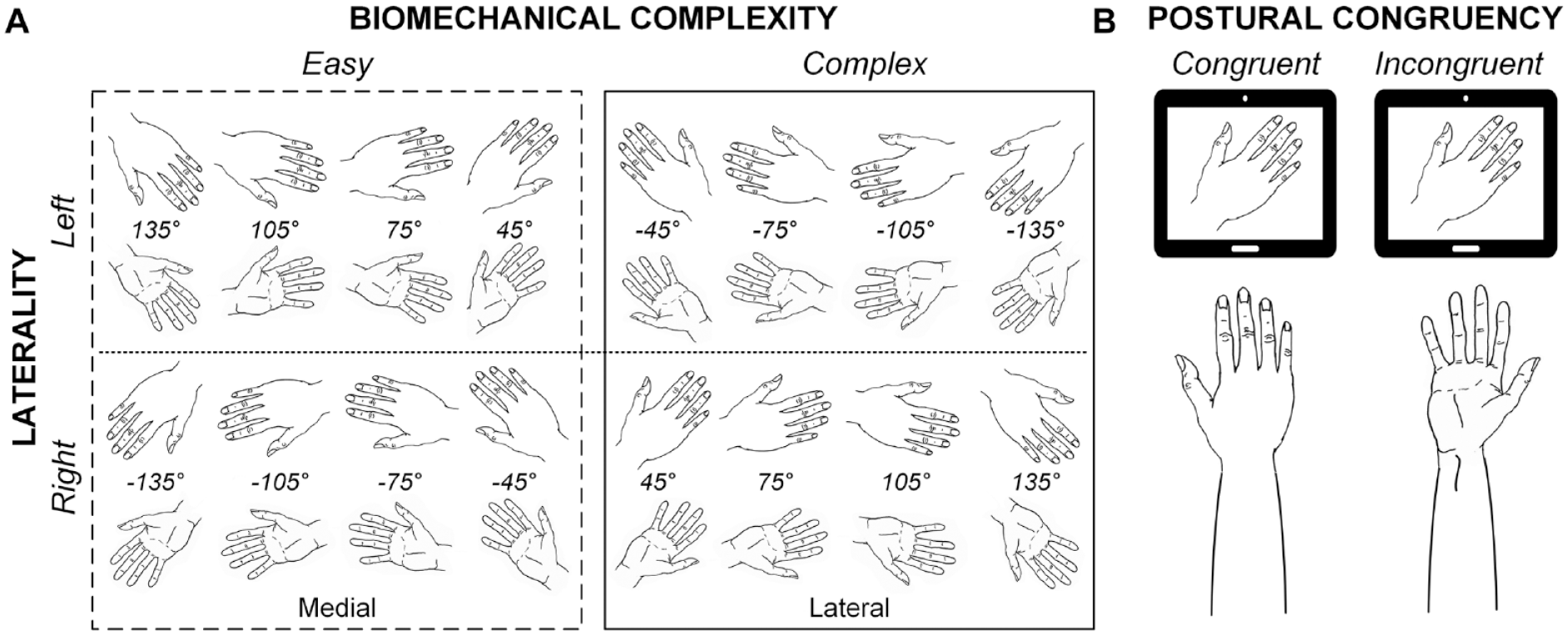
Experimental task design. (A) Overview of stimuli. Stimuli differed in laterality (left or right), view (back or palm of the hand), and were rotated at −135° to 135° with 30° increments. Stimuli with a medial orientation (rotated toward the body midline) are associated with biomechanically easy movement, whereas stimuli with a lateral orientation (rotated away from the body midline) are associated with biomechanically complex movement. (B) POSTURAL CONGRUENCY at the start of each block, patients were instructed to assume a limb posture (with the hand palm either up or down). This posture could be either congruent (example on the left), or incongruent with the stimulus view (example on the right).
Trials started with a white fixation cross, followed by presentation of the stimulus in the center of the screen, until a response was recorded (max 4000 ms). The inter-trial interval varied randomly between 2000 and 3000 ms. The task consisted of 256 trials in 32 blocks of 8, and took 20–30 minutes, depending on performance. The order of the trials was pseudo-randomized, ensuring an even distribution of trial types across blocks.
Prior to scanning, patients performed four blocks of eight practice trials at a desktop computer. For the MRI-scan, patients lay on the scanner bed in a supine position, with their extended arms resting on the scanner bed, their thighs, or support pillows, and their head fixated in the MRI head coil with a piece of tape across their forehead to minimize movement. 24 Stimuli were presented on a screen visible through a mirror attached to the head coil. We monitored alertness with an eye-monitor.
We acquired images on a 3T Siemens PrismaFit scanner (Siemens Healthcare, Erlangen, Germany), equipped with a 32-channel head coil. We acquired a T1-weighted anatomical scan with a Magnetization Prepared Rapid Gradient Echo (MPRAGE, TR = 2300 ms, TE = 3.03 ms, TI = 1100 ms, flip angle = 8°, voxel size = 1.0 × 1.0 × 1.0 mm, slices = 192, FOV = 256 mm, scanning time = 5:21 minutes). During the task, we acquired functional images using a multiband 6 sequence (MB6, TR = 1000 ms, TE = 34 ms, acceleration factor = 6, flip angle = 60°, voxel size = 2.019 × 2.019 × 2.000 mm, slices = 72, FOV = 210 mm, scanning time = 20–30 minutes varying with task performance).
Analyses
Preprocessing of Task-fMRI Data
MRI data were preprocessed using FSL 5.0.11 (FMRIB’s Software Library, http://www.fmrib.ox.ac.uk/fsl). 25 We first removed non-brain structures from the structural image, 26 realigned the functional images, 27 applied smoothing (Full Width at Half Maximum (FWHM) = 3 mm), and grand mean scaling. Next, we registered the functional images to the structural image and standard MNI152 space28 -31 and removed motion-related noise with the FSL tool ICA-AROMA. 32 After manual inspection and appropriate reclassification of the components generated by ICA-AROMA (n = 100), we applied non-aggressive denoising. 33 We performed nuisance regression on the denoised images, including 24 motion parameters, 34 and cerebrospinal fluid and white matter regressors. Finally, we applied a temporal high pass filter of 0.01 Hz and additional smoothing with a 5.2 mm FWHM Gaussian kernel (total smoothing of 6 mm FWHM). See Lustenhouwer et al 9 for detailed description of preprocessing.
First Level Task-fMRI Analyses
We performed further analyses with FSL version 6.0.1. 25 We modeled blood-oxygen-level dependent (BOLD) activation on the first level (subject and session specific) with a general linear model (GLM) that included factors BIOMECHANICAL COMPLEXITY, POSTURAL CONGRUENCY, and LATERALITY. 27 All regressors of interest included correct responses only. Events were set at stimulus onset and the duration was the subject- and session-specific median RT across trials. We additionally included three regressors of non-interest: limb-repositioning between blocks (7000 ms duration), incorrect, and missed trials (duration of median RT). BOLD activation was modeled by convolving all regressors with a canonical hemodynamic response function and its temporal derivative. 35 We performed pre-whitening followed by voxel-wise fitting of the GLM. 27 The resulting contrast of parameter estimate (COPE) images were registered to MNI152 standard space.28 -30
Main Hypothesis Testing
We tested for possible group differences in the change in cerebral and behavioral responses from baseline to follow-up for the affected (right) versus the unaffected (left) limb. We focused on a priori defined ROI’s in the right extrastriate cortex and bilateral parieto-occipital sulcus during biomechanically complex trials, 8 following our previous findings showing decreased activity in NA versus healthy controls in these regions. 7
Group Level Task-fMRI Analyses
To investigate the effect of factors TIME (baseline, follow-up), LATERALITY (right, left), and GROUP (usual care, multidisciplinary rehabilitation) on imagery-related activity, we first calculated the patient-specific difference in activity between timepoints (follow-up minus baseline COPE image) for the contrast of interest (i.e., right > left, complex trials only). We then performed non-parametric threshold-free cluster enhancement (TFCE) based permutation testing (5,000 permutations), 36 correcting for multiple comparisons with a conservative Family Wise Error of P < .05. The GLM in this analysis included factor GROUP, with the patient-specific follow-up minus baseline COPE images as input. We performed two ROI analyses for the right extrastriate cortex and bilateral parieto-occipital sulcus where we previously found reduced imagery-related brain activity in NA patients versus healthy controls in the baseline measurement 9 (see Figure 3A and B; in both NA patients and previous work in other disorders, abnormalities occurred in the hemisphere ipsilateral to the (most) affected limb9,19). As patients may also show functional reorganization in other brain regions,37,38 we additionally performed an exploratory analysis at the whole brain level, including all trial types.
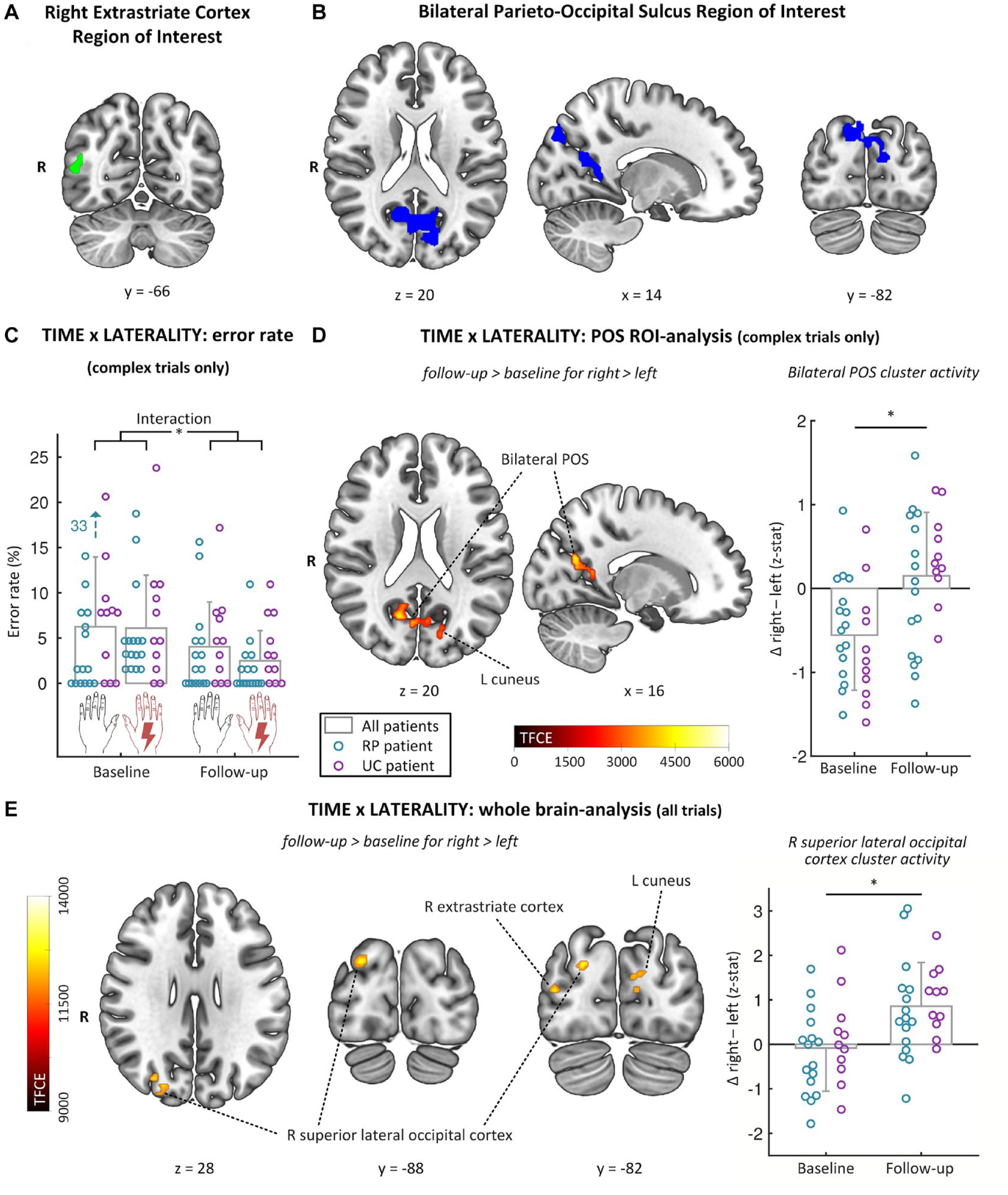
Time × Laterality showing the significant interaction between TIME (baseline, follow-up) and Laterality (right, left). Panels A and B show the masks for the region of interest analyses. Masks are taken from Lustenhouwer et al,
8
where NA patients had decreased activity compared to healthy participants during complex imagined movement of the affected limb. A. Right extrastriate cortex region of interest B. Bilateral parieto-occipital sulcus region of interest C. Time × Laterality: error rate showing the error rate for complex trials with the left (unaffected) and right (affected) upper extremity at baseline and follow-up (collapsed over group). The decrease in error rate between baseline and follow-up was significantly greater for the affected limb than for the unaffected limb. D. Time × laterality: parieto-occipital sulcus ROI-analysis showing TFCE map of increase in brain activity (across groups) from baseline to follow-up for the contrast right > left for complex trials only in the bilateral parieto-occipital sulcus region of interest. The bar graphs show the z-stat for the same contrast at baseline and follow-up for the largest of the 5 sub-clusters within the region of interest.
Behavioral Task Performance
Statistical testing was performed using IBM SPSS statistics 25 (IBM Corp, Armonk, NY, USA), statistical tests were two-tailed and alpha-level was set at P = .05. For each patient, we evaluated task performance by calculating median RTs (on correct trials) and error rates (ERs) (number of incorrect trials divided by number of valid trials) for all relevant conditions. ERs were normalized through an arcsine transformation before further analyses. 39
We performed two separate three-factor mixed ANOVAs on median RTs and normalized ERs with the same repeated factors TIME and LATERALITY, and between-subject factor GROUP, using biomechanically complex trials only as with the main fMRI hypothesis.
Clinical outcomes
To interpret potential group differences in cerebral adaptations in the context of clinical improvement, we compared the effects of multidisciplinary rehabilitation to usual care on the SRQ-DLV-score and persistent pain (VAS). For the SRQ-DLV, we performed a 2-factor mixed ANOVA with repeated-factor TIME and between-factor GROUP. As pain was not normally distributed, we performed a Mann–Whitney U test on the difference in pain between baseline and follow-up. We defined a minimal clinically important difference (MCID) on the SRQ-DLV of >12 points. 40
Brain-Behavior-Symptom Correlations
We correlated any significant changes between brain activity, symptoms, and behavioral task performance, for the affected upper extremity only. We additionally tested (one-tailed) whether the significant correlations between brain activity, behavior, and symptoms that we previously reported in the full sample of patients at baseline (n = 39) 7 were also present at follow-up in this smaller sample (see Supplemental Materials).
Confirmation of Embodied Processes
To verify that patients employed sensorimotor representations of their own limb when performing the task, we tested the influence of factors BIOMECHANICAL COMPLEXITY and POSTURAL CONGRUENCY on behavioral task performance (RTs) and related brain activity at both baseline and follow-up across groups.
Data Availability
The data are available through the corresponding author on reasonable request.
Results
There were no significant group differences in age (t(25) = −0.84, P = 0.41), sex (χ2(1) = 0.05, P = 0.82), or disease characteristics at baseline (time since last attack: U = 75.00 z = −0.64 P = 0.54; persistent pain: U = 62.00 z = −1.64 P = 0.20; SRQ-DLV: t(25) = −0.90, P = 0.38), see Table 1.
There was an overall learning effect from baseline to follow for the task: across groups, patients made significantly fewer errors at follow-up (3.3% ± 3.3%) compared to baseline (6.2% ± 5.2%) (TIME: F(1,25) = 25.16, P < 0.001, part. η2 = 0.50), and tended to be faster (TIME: F(1,25) = 3.71, P = 0.07, part. η2 = 0.13).
Main Hypothesis Tests
Task-Related Cerebral Activity
Neither the whole-brain analysis nor the ROI-analyses revealed any significant group differences in change in activity between baseline and follow-up for the main contrast of interest (no GROUP × TIME × LATERALITY interaction for contrast right > left, complex trials only). Interestingly, there was a significant two-way interaction between TIME and LATERALITY in the parieto-occipital sulcus ROI, independent of GROUP. Specifically, across both groups, NA patients showed a significant increase in brain activity from baseline to follow-up when performing imagery of biomechanically complex movements with their affected limb, compared to their unaffected limb (right > left, complex trials only). This increase was observed in five sub-clusters within the parieto-occipital sulcus ROI (see Figure 3D and Supplemental Table 3).
When we included biomechanically easy trials, we found similar results as above in the whole brain analysis: there was no significant three-way interaction with GROUP, but there was a significant interaction between TIME and LATERALITY. Specifically, NA patients showed a significant increase in activity from baseline to follow-up when performing imagery with their affected limb, compared to their unaffected limb, in four clusters within the occipito-parietal cortex. Three sub-clusters were located in the right, ipsilateral dorsal visual stream (two in the superior lateral occipital cortex and one in the extrastriate cortex), and one cluster in the left cuneus, partly overlapping with the parieto-occipital sulcus ROI) (see Figure 3E and Supplemental Table 3).
Behavioral Task Performance
We found no significant three-way GROUP × TIME × LATERALITY interactions for ERs (F(1,25) = 1.48, P = 0.23, part. η2 = 0.06) or RTs (F(1,25) = 0.004, P = 0.95, part. η2 ≤ 0.001). Similar to the cerebral activity, we did find a significant two-way TIME × LATERALITY interaction for ERs (F(1,25) = 5.11, P = 0.03 part. η2 = 0.17; see Figure 3C). Specifically, post-hoc testing revealed that ERs for complex trials decreased for both the affected (F(1,26) = 17.58, P < 0.001, part. η2 = 0.40) and unaffected (F(1,26) = 5.53, P = 0.03, part. η2 = 0.18) limb from baseline to follow-up, but the significant interaction effect indicates that this decrease was significantly greater for the affected limb.
Clinical Outcomes
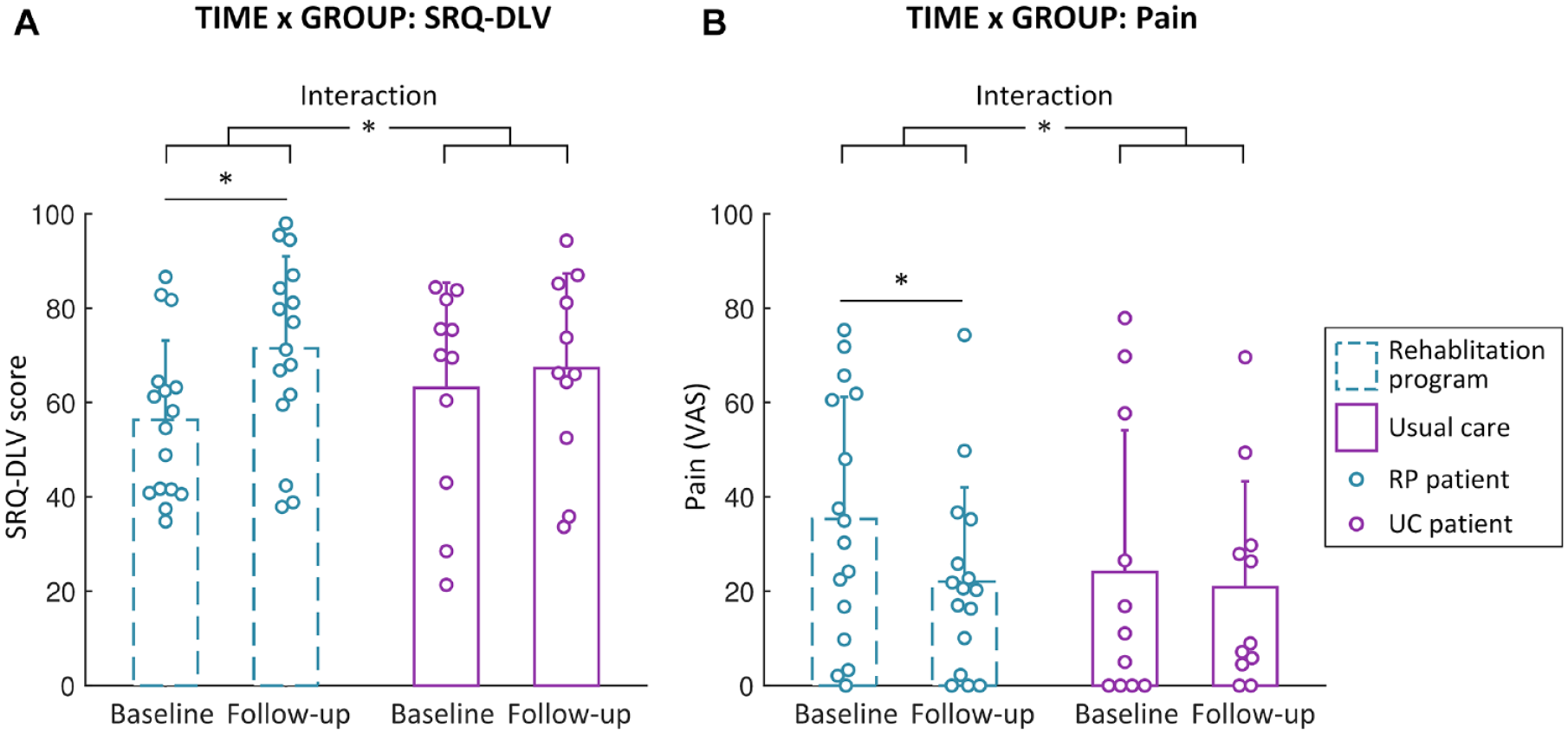
Patients in the multidisciplinary rehabilitation group showed significantly greater increase in functional capability of the upper extremity (SRQ-DLV: GROUP × TIME interaction F(1,25) = 15.38, P = 0.04 part. η2 = 0.17) and a greater reduction of persistent pain than patients in the usual care group (Δ pain: U = 46.00, Z = −2.07 P = 0.04). Post-hoc testing revealed that, whereas patients in the multidisciplinary rehabilitation group showed a significant increase in functional capability (F(1,15) = 19.87, P < 0.001, part. η2 = 0.57) that exceeded the MCID (>12 points), and a significant decrease in pain (z = −2.59, P = 0.01, r = −0.46) from baseline to follow-up, patients in the usual care group did not (SRQ-DLV: F(1,10) = 1.62, P = 0.23, part. η2 = 0.14; pain: z = −0.42, P = 0.68, r = −0.09) (Figure 4, Table 1).
Effect of group on clinical outcome bars show the interaction between GROUP and TIME for (A) SRQ-DLV score and (B) Pain. Patients in the multidisciplinary rehabilitation group showed a significant improvement from baseline to follow-up in both SRQ-DLV score (i.e., functional capability of the upper extremity) and persistent pain, whereas patients in the usual care group did not show significant clinical improvements.
Brain-Symptom-Behavior Correlations
We did not find any significant correlations between changes in affected limb-specific brain activity (in clusters > 10 voxels), behavioral task performance, or clinical improvement (rs < 0.29, P > 0.14). We did find significant correlations between behavioral task performance with the affected limb and related activity in the parieto-occipital sulcus at baseline, and with persistent pain at both timepoints, similar to our previous findings 7 (see Supplemental Materials and Supplemental Figure 2).
Confirmation of Embodied Processes
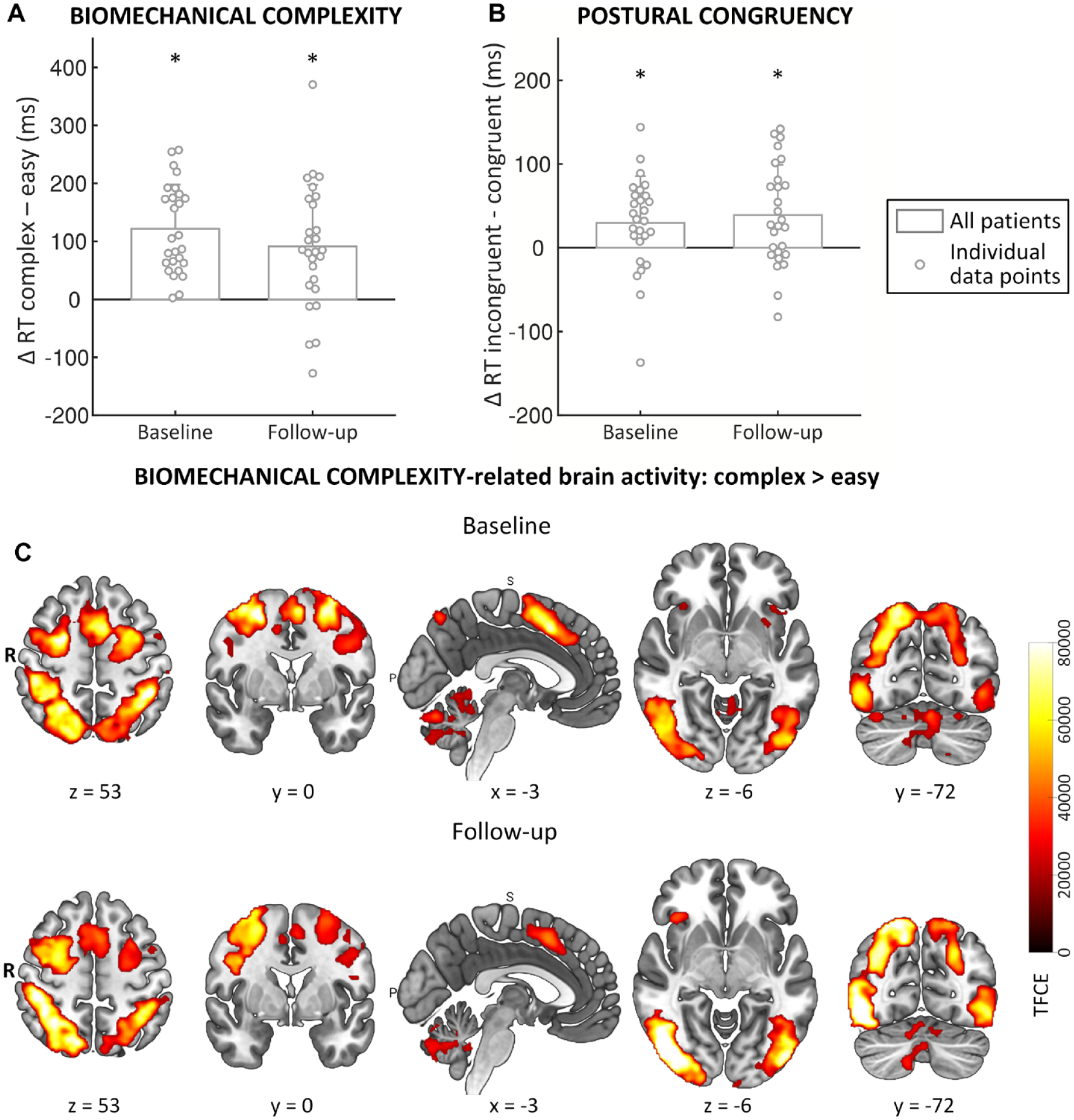
Across time-points and groups, behavioral and cerebral responses confirmed that patients employed embodied processes during the motor-imagery task, consistent with previous work:14,15 significantly slower RTs (BIOMECHANICAL COMPLEXITY: F(1, 26) = 47.58, P < 0.001, part. η2 = 0.65, Figure 5) and increased activity in a motor-imagery related brain network for biomechanically complex versus easy (imagined) movements (Figure 5, Supplemental Table 2), and significantly slower RTs when the posture of the own limb was incongruent versus congruent with the stimulus view (POSTURAL CONGRUENCY: F(1, 26) = 14.46, P = 0.001, part. η2 = 0.36) (Figure 5).
General task effects The behavioral task effects of (A) biomechanical complexity (complex > easy) and (B) postural congruency (incongruent > congruent) on reaction times are shown in separate bar plots for baseline and follow-up, collapsed over group. Bars show the mean difference between conditions (reaction times for complex minus easy trials (A), and incongruent minus congruent trials (B)), error bars are ±1 standard deviation; individual data points are plotted on top of the bars. (C) Biomechanical complexity-related brain activity (complex > easy) for all patients (collapsed over groups) at baseline and follow-up separately. Figures are TFCE-maps, and match the behavioral data shown in A.
Discussion
Despite the fact that clinical improvement was significantly greater after multidisciplinary rehabilitation than after usual care, we found no significant group differences in changes in task performance or in brain activity from baseline to follow-up (18 weeks later). Across groups, NA patients did show improved task performance and increased activity in visuomotor occipito-parietal brain areas, where activity was decreased compared to healthy participants at baseline, 9 specifically for imagined movements with their affected upper extremity. These findings indicate that the abnormal cerebral sensorimotor representations that occur in response to peripheral nerve damage in NA 9 can recover toward normality.
We hypothesized that multidisciplinary rehabilitation, but not usual care, could modify abnormal cerebral sensorimotor representations of the affected upper extremity, as it targets cerebral motor control, whereas usual care typically does not. Instead, we found significant behavioral and cerebral changes in both groups, occurring along with improvements in clinical outcome after multidisciplinary rehabilitation. There are several possible explanations for this apparent discrepancy between clinical and cerebral group effects. First, we may have lacked power to discern potential group differences in cerebral and behavioral changes because of our small sample size, or the clinical effect may have been too modest to reveal cerebral differences. That is, the mean SRQ-DLV change in the multidisciplinary rehabilitation group of 15 points was clinically relevant, 40 and was significantly larger than the change in the usual care group (4 points; Table 1). Hence, the differential change in SRQ-DLV between the two groups (11 points) approaches the MCID, which may have been too subtle to reveal cerebral changes in brain plasticity.
Alternatively, multidisciplinary rehabilitation may not have elicited greater change in cerebral representations of the upper extremity than usual care. Most patients participated relatively close to their latest NA attack (22/27 patients completed the study within 24 months after onset). Natural recovery processes associated with peripheral nerve recovery 5 and training through performing daily activities could have contributed to changes in cerebral sensorimotor representations regardless of the specific rehabilitation program. This may explain why patients in the usual care group also showed increased cerebral activity and improved task performance, despite the fact that more than half of these patients did not report any formal therapy. The fact that multidisciplinary rehabilitation did elicit greater clinical improvement may be understood by taking a closer look at the program. First, occupational therapy is a key component, which often lacks in usual care. It improves clinical outcome by enabling daily activities through training of self-management strategies,5,10 but it does not target cerebral motor control. Second, although the specific physical therapy does target cerebral motor control, it was not developed to specifically train the visuomotor processes that we have recently found to be altered in NA. 9 Multidisciplinary rehabilitation may in fact mediate clinical outcome through different cerebral mechanisms, which we did not test with our motor imagery paradigm.
Our findings provide valuable insights into (cerebral) recovery in NA, despite the lack of significant cerebral group differences. In our parallel cross-sectional study, we showed that compared to healthy participants, NA patients had decreased brain activity during motor imagery of the affected upper extremity in two visuomotor brain areas: the bilateral parieto-occipital sulcus and the right extrastriate cortex. 9 In the current longitudinal study, we show that patients’ activity in the parieto-occipital sulcus increased in the direction of normality (see Supplemental Figure 2), 9 although we could not directly compare patients to healthy participants at follow-up, as the healthy group was only measured once. The parieto-occipital sulcus is involved in processing and integration of self-relevant information, and hence contributes to a multisensory representation of the spatial location of the own body.41 -43 Accordingly, increased activity in this region in NA over time may reflect a better encoding of hand position relative to the rest of the body, which is necessary for computing optimal motor plans. Brain activity in the right extrastriate cortex ROI did not change significantly from baseline to follow-up. However, an unconstrained whole brain analysis, considering both biomechanically complex and easy trials, revealed significant increases in activity related to the affected limb in several ipsilateral occipito-parietal areas. Although located more posteriorly and medially than the ROI in the extrastriate cortex, these areas are part of the same dorsal visual stream. The dorsal visual stream has strong connections with the motor system, and forms an interface between perception and action by integrating visual and proprioceptive information to form sensorimotor representations of the upper extremity.44 -47 Accordingly, the increased activity in the dorsal visual stream observed in NA patients over time may reflect improved integration of multisensory (visual and proprioceptive) information into motor plans. Combined with our previous findings showing that NA patients have reduced activity compared to healthy controls in the parieto-occipital sulcus and along the dorsal visual stream 9 , the longitudinal findings reported here suggest that the activation pattern of NA patients moves toward that of healthy people, indicating a trend toward normalization of visuomotor processing in NA along with peripheral recovery. This identifies visuomotor processing as a potential target for treatment strategies to further facilitate clinical recovery in NA.
Patients also improved their overall behavioral task performance (i.e. overall decrease in errors from baseline to follow-up), which could raise the question of whether the reported cerebral changes can be explained by general learning effects. 47 Such an explanation can however be ruled out, since both cerebral and behavioral changes involve interactions between time and laterality, which makes them specific to the affected upper extremity.
The generalizability of our findings to the wider NA population may have limits, despite the heterogeneity of our sample. We excluded patients with severe comorbidities, bilateral or unilateral left-sided involvement, left hand dominance or ambidexterity, and patients who suffered a recurrent attack during the study. Although it is likely that cerebral adaptations also occur in those patients, the underlying mechanisms and their response to rehabilitation and peripheral neve recovery are unknown and could be different. In a similar vein, drop-out was greater in the usual care than in the multidisciplinary rehabilitation group, which is a potential source of bias that should be acknowledged.
Conclusions and Clinical Implications
We observed no cerebral group differences even though multidisciplinary rehabilitation elicited greater clinical improvement than usual care. This shows that specific rehabilitation can accelerate recovery in NA patients, and underlines the importance of a multidisciplinary approach. Importantly, patients in both groups showed increased cerebral activity in visuomotor brain regions, where their activity was decreased compared to healthy people at baseline. 9 This indicates that altered cerebral sensorimotor representations of the upper extremity can recover toward normality in NA. Rehabilitation may be further optimized by implementing strategies that target visuomotor processes, and sensorimotor integration in particular, as both initial (mal)adaptation and recovery occur in visuomotor brain areas involved in forming sensorimotor representations. Visuomotor strategies that have been successfully applied in other neurological disorders include mirror visual feedback, 48 action observation, 49 and graded motor imagery. 50 Future research could focus on identifying rehabilitation strategies to effectively target cerebral mechanisms in NA and other peripheral nerve disorders. To better understand cerebral (treatment) effects in NA, future studies could employ different paradigms (other tasks such as motor execution or explicit motor imagery, and/or alternative neuroimaging techniques such as EEG), and compare cerebral mechanisms between patients that respond well to existing treatment and those who do not.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221145149 – Supplemental material for Cerebral Adaptation Associated with Peripheral Nerve Recovery in Neuralgic Amyotrophy: A Randomized Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683221145149 for Cerebral Adaptation Associated with Peripheral Nerve Recovery in Neuralgic Amyotrophy: A Randomized Controlled Trial by Renee Lustenhouwer, Ian G.M. Cameron, prof. Nens van Alfen, Ivan Toni, Alexander C.H. Geurts, Baziel G.M. van Engelen, Jan T. Groothuis and Rick C. Helmich in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank our participants for their time and commitment to the study. We also thank Melissa Bakkenes, Eline van de Ven, and Elze Wolfs for their contributions to data collection, Paul Gaalman for his technical support, Renske Janssen, Jos IJspeert, Judith Kanters, Yvonne Veenhuizen, Thomas Verheggen, Allan Pieterse, and Jessica ten Broek-Pastoor for providing the multidisciplinary rehabilitation program, Ellis Gielink-Kersten for her secretarial support, and Saskia Lassche, Fran Smulders, and Juerd Wijntjes for their role in patient inclusion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Prinses Beatrix Spierfonds [W.OR16-05]. Several authors of this publication are members of the Netherlands Neuromuscular Center (NL-NMD) and the European Reference Network for rare neuromuscular diseases (EURO-NMD).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
