Abstract
Background
Intermittent theta-burst stimulation (iTBS) has been suggested to improve poststroke rehabilitation. The cerebellum is considered crucial for motor control. However, the effects of cerebellar iTBS with routine physical therapy on balance and motor recovery in subacute and chronic stroke patients have not been explored.
Objective
To measure the short-term effects of cerebellar iTBS with physiotherapy on the balance and functional outcomes in subacute and chronic stroke patients with hemiparesis.
Methods
Thirty hemiparetic patients were recruited for this randomized, double-blinded, sham-controlled trial, and randomized into either the treatment or sham group. Both groups participated in physiotherapy 5 times per week for 2 weeks, and cerebellar iTBS or sham iTBS was performed daily, immediately before physiotherapy. The primary outcome was the Berg balance scale (BBS) score. Secondary outcomes included the trunk impairment scale (TIS) score, Fugl-Meyer assessment scale score for lower extremities (FMA-LE), Barthel index (BI), and corticospinal excitability, as measured by transcranial magnetic stimulation. The outcomes were measured before and 1 week and 2 weeks after the intervention.
Results
Compared with those at baseline, significant increases were identified in all clinical scores (BBS, TIS, FMA-LE, and BI) in both groups after the 2-week intervention. The BBS and TIS scores improved more in the iTBS group than in the sham group.
Conclusions
Cerebellar iTBS with physiotherapy promotes balance and motor recovery in poststroke patients. Therefore, this method can be used in low-cost, fast, and efficient protocols for stroke rehabilitation (Chinese Clinical Trial Registry: ChiCTR1900026450).
Introduction
Stroke is a leading cause of death and disability worldwide. 1 Poststroke care and treatments are costly and time consuming, leading to a heavy burden on families and society. 2 Balance and motor dysfunctions after stroke have a substantial impact on gait function, independence in activities of daily living (ADL), and quality of life in stroke patients. 3 Therefore, rehabilitation of the balance function is a critical determinant of functional independence and overall recovery after stroke. However, currently, no standard, effective, and highly efficient rehabilitation protocol is available for poststroke balance dysfunction. The cerebellum is considered a crucial structure involved in balance and motor control. 4 Previous research has established that the cerebellum has strong anatomical and functional connections with the cerebral primary motor cortex (M1) through cerebellar-thalamus-M1 circuits. 5 A recent meta-analysis suggested that cerebellar stimulation may affect corticospinal excitability. 6 Notably, greater activation in the contralesional cerebellum is associated with better motor function. 7
Noninvasive brain stimulation (NIBS) is rapidly becoming a key factor in the rehabilitation of nervous system diseases, and this method promotes neural plasticity and regulates the excitability of brain structures. 4 NIBS includes both transcranial magnetic stimulation (TMS) and transcranial electric stimulation. NIBS has been proposed as a strategy to facilitate rehabilitation of poststroke gait disorders. To date, M1 stimulation has primarily been investigated. 8 However, the cerebellum is also a crucial node controlling support, balance, and locomotion, and it is crucially involved in locomotor adaptation and learning processes. 9 To date, limited data exist on the effect of cerebellar NIBS on poststroke rehabilitation. 10 Kim et al 11 suggested that inhibitory 1-Hz repetitive TMS (rTMS) improved performance in the 10-minute walk test and Berg balance scale (BBS) in posterior circulation stroke. Picelli et al 12 found that cerebellar cathodal transcranial direct current stimulation (tDCS) in combination with cathodal transcutaneous spinal direct current stimulation was useful in improving performance in the 6-minute walk test in chronic brain stroke patients with walking impairment. Intermittent theta-burst stimulation (iTBS) is a specific mode of TMS, that can lead to long-term potentiation. 13 Notably, cerebellar iTBS considerably promotes the neural activity and synaptic plasticity of the cerebellum. 13 Cerebellar iTBS improves posture and gait in posterior circulation stroke. 14 Koch et al 15 found that cerebellar iTBS promotes the excitability of the posterior parietal cortex and improves balance and gait function in chronic stroke patients. In addition, no research on the effects of cerebellar iTBS with physiotherapy on balance and motor recovery in subacute and chronic stroke patients has been conducted.
The present study is the first to explore the short-term effects and potential mechanisms of cerebellar iTBS coupled with routine physical therapy on balance, motor function, and corticospinal excitability in subacute stroke patients. This research seeks to provide a deeper understanding of the relationship between the cerebellum and poststroke rehabilitation.
Methods
Trial Design
The pilot study was designed as a randomized, double-blind, sham-controlled clinical trial, and both the participants and assessors were blinded. The subjects received either 10 sessions of cerebellar iTBS or sham iTBS combined with physical therapy at an allocation ratio of 1:1.
Participants
A total of 30 subacute and chronic stroke inpatients (Supplemental Appendix A) in our rehabilitation center were included in this trial after they provided written informed consent (Supplemental Appendix B). The inclusion criteria for the subjects were as follows: (1) being between 18 and 80 years old; (2) having had subacute or chronic stroke (>2 weeks from stroke onset) 16 ; (3) having presented their first symptomatic unilateral ischemic or hemorrhagic stroke as proven by magnetic resonance imaging or computed tomography; and (4) having presented with a motor deficit in the lower limbs as determined by a score <34 points on the Fugl-Meyer assessment scale score for lower extremities (FMA-LE) and a score of <56 points on the BBS. 17 The exclusion criteria were as follows: (1) nervous system diseases other than stroke (including traumatic brain injury); (2) cerebellar or brainstem stroke; (3) intracranial metallic devices or skull defects; (4) a cardiac pacemaker; (5) difficulty in understanding or executing commands; (6) a history of seizures; or (7) pregnancy.
Intervention
All subjects who met the inclusion criteria were randomly assigned to either the treatment (n=15) or sham group (n = 15) according to the random number table. Both groups received standard stroke physical therapy. Each session lasted 50 minutes, and subjects participated in 1 session per day, 5 times per week (Monday to Friday) for 2 weeks in total. The patients also received cerebellar iTBS or sham iTBS applied over the cerebellar hemisphere contralateral to the affected cerebral hemisphere each day, immediately before physical therapy.
Cerebellar iTBS
We used a CCY-I rapid magnetic stimulator connected with a 70-mm figure-of-8 coil (YIRUIDE Medical) to stimulate the cerebellum. The targeted area was located 3 cm lateral to the midline and 1 cm below the inion over the contralesional lateral cerebellum. 18 Sham iTBS was administered at the same location (coil held at 90° from the scalp). 15 The intensity of iTBS was set at 80% of the active motor threshold (AMT). The AMT was defined as the lowest intensity that produced a motor-evoked potential (MEP) of >200 μV in at least 5 out of 10 trials with 10% maximal voluntary contraction using visual feedback.18,19 Overall, each subject received 600 stimuli for each stimulation session. The coil was positioned tangentially to the scalp, with the handle pointing superiorly. 18 We used the figure-of-8 coil as this approach has been adopted in previous investigations in which cerebellar TMS was shown to be effective in modulating the excitability of the contralateral motor cortex.20,21
Standard Physical Therapy Program (Supplemental Appendix C)
All participants in this trial underwent the standard physical therapy program immediately after receiving cerebellar or sham iTBS, including trunk control training, sit-to-stand training, balance exercises, and gait training. This program was administered by 2 specifically trained physical therapists who were also blinded.
Outcome Measures
The outcomes were measured at baseline (T0), after 1 week (T1) and after 2 weeks (T2) of the intervention (Figure 1). The primary outcome was the BBS. The BBS is the most widely used clinical scale for assessing balance performance in individuals with neurological conditions, including static and dynamic balance. 22 The sum of the scores for the 14 items (each item was rated from 0 to 4) yielded a balance score ranging from 0 to 56. 23 A recent meta-analysis indicated that the instrument demonstrates good reliability and validity. 22
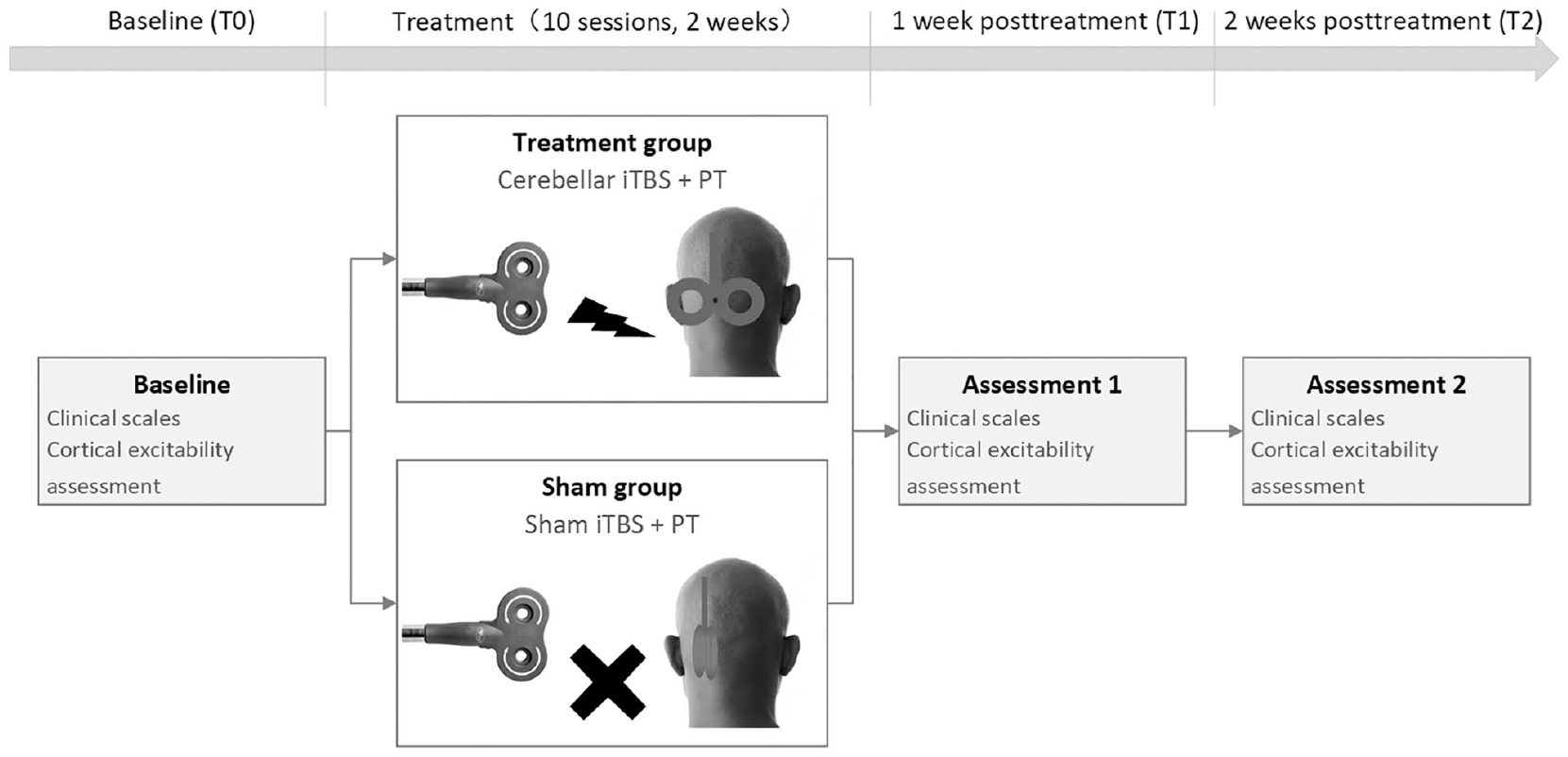
Schematic presentation of the experimental design. The clinical scales and cortical excitability were assessed at baseline (T0) and after 1 week (T1) and 2 weeks (T2) of the intervention. iTBS, intermittent theta-burst stimulation; PT, physical therapy.
The secondary exploratory measures included the trunk impairment scale (TIS) scores, FMA-LE scores, Barthel index (BI) scores, and cortical excitability measured by TMS. The TIS is a tool used to measure motor impairment of the trunk after stroke, with the goal of quantifying the quality of trunk movement and serving as a guide for treatment. 24 The TIS scores ranged from 0 to 23, including those for static and dynamic sitting balance as well as trunk coordination. The FMA-LE can predict lower extremity motor recovery in individuals with stroke. 25 This assessment exhibits good internal consistency and reliability, discriminative validity, and responsiveness to interventions. 26 The BI is considered to be the best of the ADL measurement scales and consists of 10 items with scores ranging from 0 to 100. 27
We used a questionnaire (Supplemental Appendix D) to measure the adverse effects after each iTBS session systematically.
Corticospinal Excitability Assessment
Corticospinal excitability was assessed by the cortical silent period (CSP), resting motor threshold (RMT), and MEP amplitude from the affected side. Single TMS of M1 of the affected hemisphere was performed with a figure-of-8 coil connected to the same stimulator for cerebellar stimulation. The TMS coil was positioned over M1 with the handle directed backward and laterally and at an angle of approximately 45° to the mid-sagittal line of the head. 28 The abductor pollicis brevis (APB) was selected as the target muscle for the corticospinal excitability assessment. We placed the active electrode on the muscle belly of the APB and positioned the reference electrode 2 cm above the wrist line. The RMT was defined as the lowest intensity that produced MEPs of >50 μV in at least 5 out of 10 trials with the APB relaxed. 18 To measure the MEP amplitude, the average of 10 trials was recorded using an RMT intensity of 120% at T0, T1, and T2. 28 The CSP was collected according to previously established methods. 29 The CSP is defined as the duration of the electromyogram quiescence that occurs during voluntary contraction secondary to a superimposed pulse. 30 The duration of the CSP was determined during an isometric voluntary contraction of the APB at approximately 30% of maximum force. 20 Ten CSP trials were obtained with a minimum 10-second rest interval between each trial to prevent fatigue. 29
Sample Size
The sample size was calculated using a G*power of 3.1.9.4. 31 The effect size (f) was calculated as 0.28 based on the BBS scores from a group of individuals, as reported in a study by Koch et al. 15 To achieve improvements with α = .05 and β at 80%, the necessary sample size of n = 12 per group was revealed by a power analysis. The final sample size required was 15 per group to allow for a 20% dropout rate.
Randomization and Blinding
Participants were recruited by an independent therapist and then randomly assigned to either the treatment group or the sham group using shuffled opaque envelopes containing a code for the type of stimulation to be received. Clinical scales and cortical excitability were assessed by a neurophysiologist who was blinded to the condition assigned to the patients. Thus, the patients, physical therapists, and evaluators were all blinded, whereas the therapist who performed the cerebellar stimulation was not blinded.
Statistical Analysis
The data were processed with SPSS version 24.0 (IBM Corp). All primary analyses were conducted by the intention-to-treat (ITT) principle. We tested the normality of the data using the Shapiro-Wilk test. For continuous variables, the data are presented as the means (± standard deviations, SDs); for the ordinal variables, the data are presented as the medians (interquartile ranges, IQRs). Numbers (n) and percent (%) were used for categorical variables.
The independent-samples t test was used to compare the groups in terms of the baseline homogeneity and across the different conditions. If the data did not follow a normal distribution, we used a nonparametric test (the Wilcoxon Mann-Whitney U test or chi-square test) to analyze the differences between groups.
We separately analyzed the results for the clinical scales (BBS, TIS, FMA-LE, and BI scores) and cortical excitability assessments (RMT and CSP) using a repeated-measures mixed analysis of variance with the group as the between-individual factor (cerebellar iTBS and sham iTBS) and the time as the within-individual factor (T0, T1, and T2) after the test for homogeneity of variance and Mauchly’s test for sphericity were conducted. The nonparametric test was used to analyze the differences in MEP amplitudes between groups.
Statistical significance was accepted at P < .05.
Ethics Committee
Ethical approval was obtained from the biomedical ethics committee of West China Hospital at Sichuan University. The protocol of this study was registered with the Chinese Clinical Trial Registry (registration number: ChiCTR1900026450).
Results
Patient Data
The CONSORT (Consolidated Standards of Reporting Trials) patient flowchart is shown in Figure 2. We screened a total of 423 patients for eligibility, and 30 patients met our inclusion criteria and were randomized into 1 of the 2 groups (treatment or sham group). No significant differences in age, sex, type of stroke, lesion site, National Institutes of Health Stroke Scale (NIHSS; used to assess the degree of neurological deficit in stroke patients), baseline clinical scale scores, and baseline cortical excitability existed between the groups (Table 1). After 2 weeks of treatment, 2 patients in the treatment group and 3 patients in the sham group dropped out, but all patients completed the first assessment and were included in the final analysis.
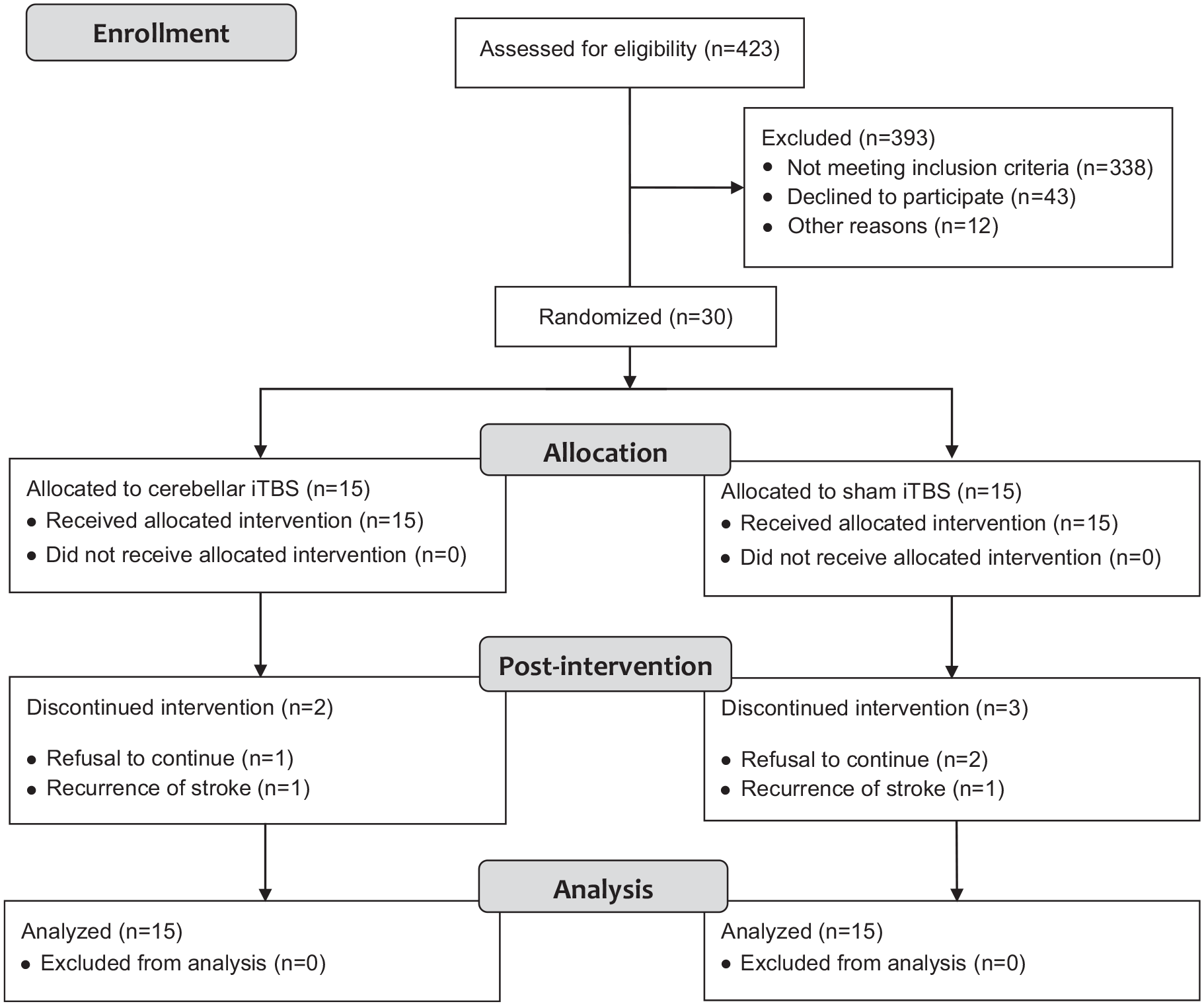
CONSORT (Consolidated Standards of Reporting Trials) patient flowchart.
Participant Characteristics.
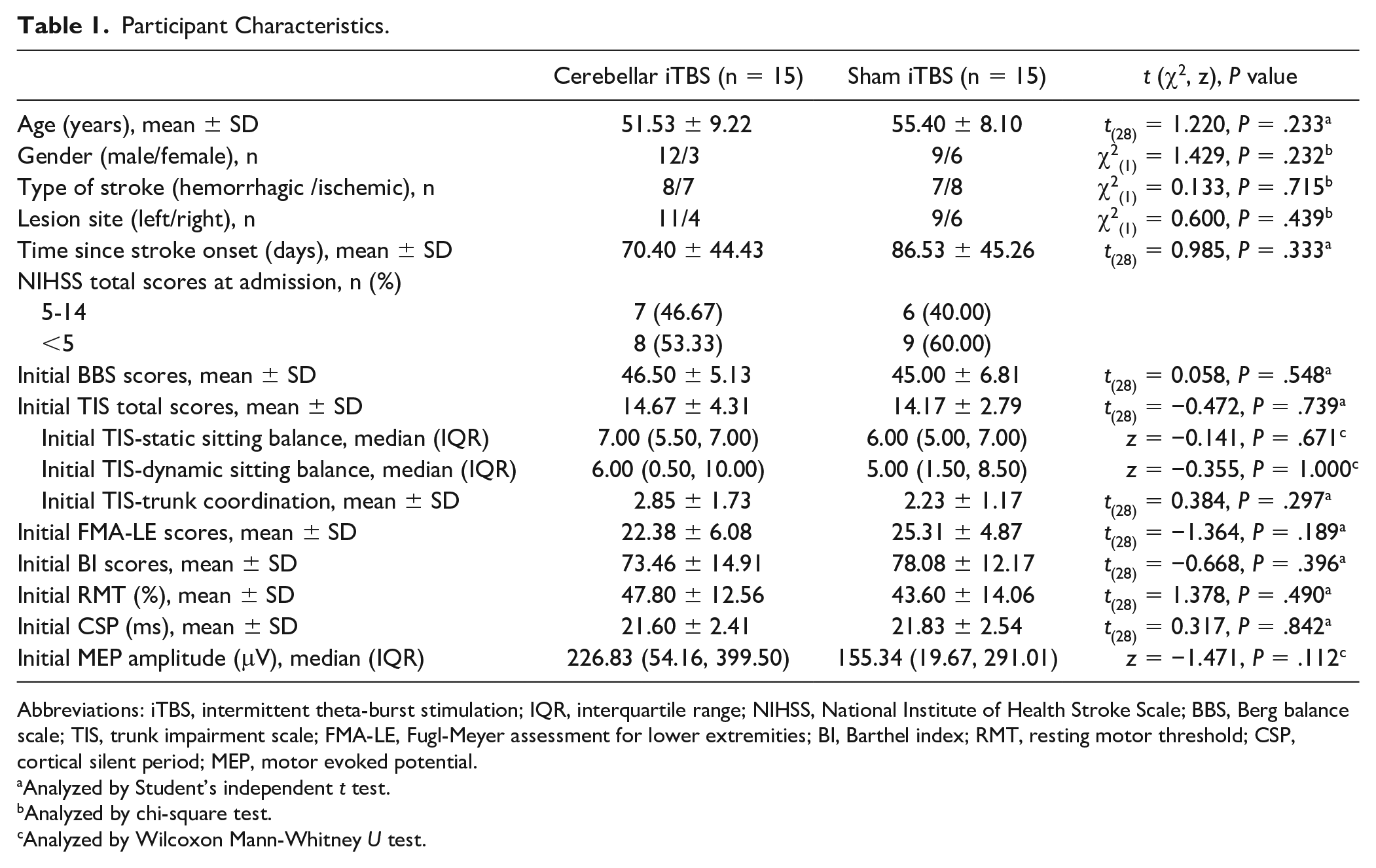
Abbreviations: iTBS, intermittent theta-burst stimulation; IQR, interquartile range; NIHSS, National Institute of Health Stroke Scale; BBS, Berg balance scale; TIS, trunk impairment scale; FMA-LE, Fugl-Meyer assessment for lower extremities; BI, Barthel index; RMT, resting motor threshold; CSP, cortical silent period; MEP, motor evoked potential.
Analyzed by Student’s independent t test.
Analyzed by chi-square test.
Analyzed by Wilcoxon Mann-Whitney U test.
All patients tolerated the treatment program. None of the patients in either group experienced serious adverse effects. Overall, 1 patient in the treatment group reported a mild headache, but he estimated his pain at a level of 2/10 on the numerical pain rating scale and did not require treatment to resolve the headache.
Primary Functional Outcome
The analysis of variance revealed a significant group × time interaction (F = 5.098, P = .045), and post hoc analysis revealed that 2 weeks of cerebellar iTBS associated with physiotherapy led to an improvement in the BBS score compared with that of the sham group. Additional analyses revealed that the BBS score in the treatment group was higher at T1 versus T0 (P = .003) and at T2 versus T0 (P = .001); however, no significant difference was found between T2 and T1 (P = .412) (mean ± SD; T0, 46.50 ± 5.13; T1, 49.92 ± 3.53; T2, 50.75 ± 4.05). The analysis also demonstrated that the BBS score improved in the sham group at T2 compared with T0 (P = .001) (mean ± SD; T0, 45.00 ± 6.81; T1, 46.75 ± 7.44; T2, 47.67 ± 6.58) (Figure 3).
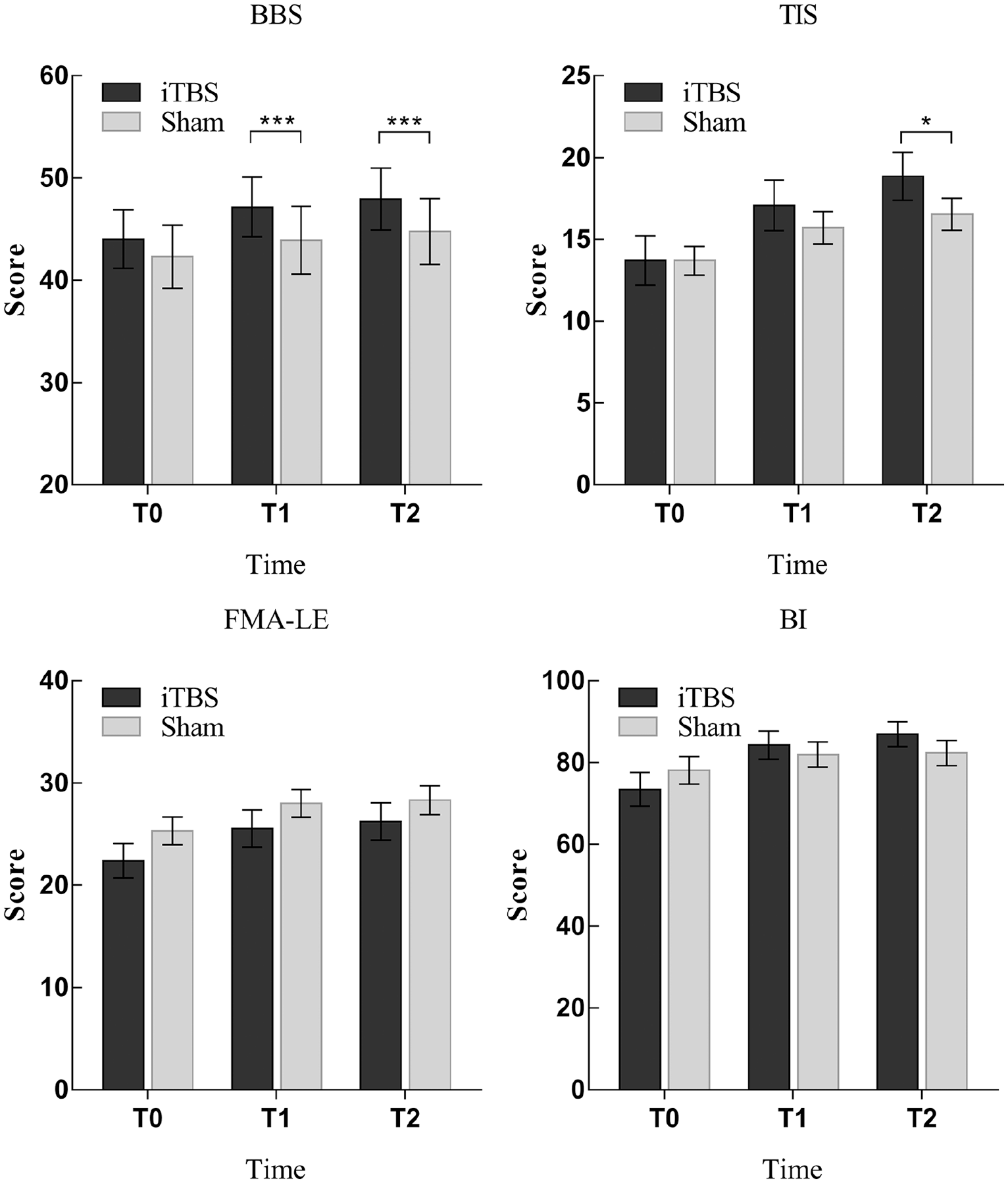
Mean scores of clinical scales in the treatment and sham groups at baseline (T0), 1 week of treatment (T1), and 2 weeks of treatment (T2). The data are expressed as the mean ± SE. *P < .05, ***P < .001. iTBS, intermittent theta-burst stimulation; BBS, Berg balance scale; TIS, trunk impairment scale; FMA-LE, Fugl-Meyer assessment for lower extremities; BI, Barthel index.
Secondary Functional Outcomes
Trunk Function
Compared with the sham group, the treatment group exhibited TIS total scores increased by 2.308 (95% CI 0.235-4.381; P = .023) at T2. No significant difference between the 2 groups was evident at T1 (P = .338). Analysis of the TIS total scores revealed a small improvement at T1 and T2 in the treatment group (T1 vs T0, P = .002; T2 vs T0, P = .001), and at T2 in the sham group only (T1 vs T0, P = .094; T2 vs T0, P = .010). No significant increases in the TIS subscales were identified in the treatment group compared with the sham group (P > .05) (Figure 3).
Lower Extremity Motor Function
No significant difference in FMA-LE scores was noted between the 2 groups (P > .05). However, a similarly small increase was noted in both groups after treatment compared with before treatment (F = 24.35, P < .0001) at both T1 and T2 versus T0 (T1 vs T0, P < .0001; T2 vs T0, P < .0001) and at T2 versus T1 (T2 vs T1, P = .004) (Figure 3).
Activities of Daily Living
No difference in the BI scores was observed compared with those of the sham group. Similar to FMA-LE scores, the BI scores exhibited a small increase in both groups (F = 22.21, P < .0001) both at T1 and T2 compared with the baseline (T1 vs T0, P < .0001; T2 vs T0, P < .0001) and at T2 versus T1 (T2 vs T1, P = .013) (Figure 3).
Corticospinal Excitability
No significant difference in all corticospinal excitability assessments (RMT, CSP, and MEP amplitude) was noted between the 2 groups (P > .05). After 2 weeks of cerebellar iTBS, the MEP amplitude increase over the affected hemisphere was lower in the treatment group compared with the sham group (P = .028). No significant differences were found in other markers of cortical excitability (RMT and CSP) for either group (P > .05) (Table 2).
Corticospinal Excitability Assessments.
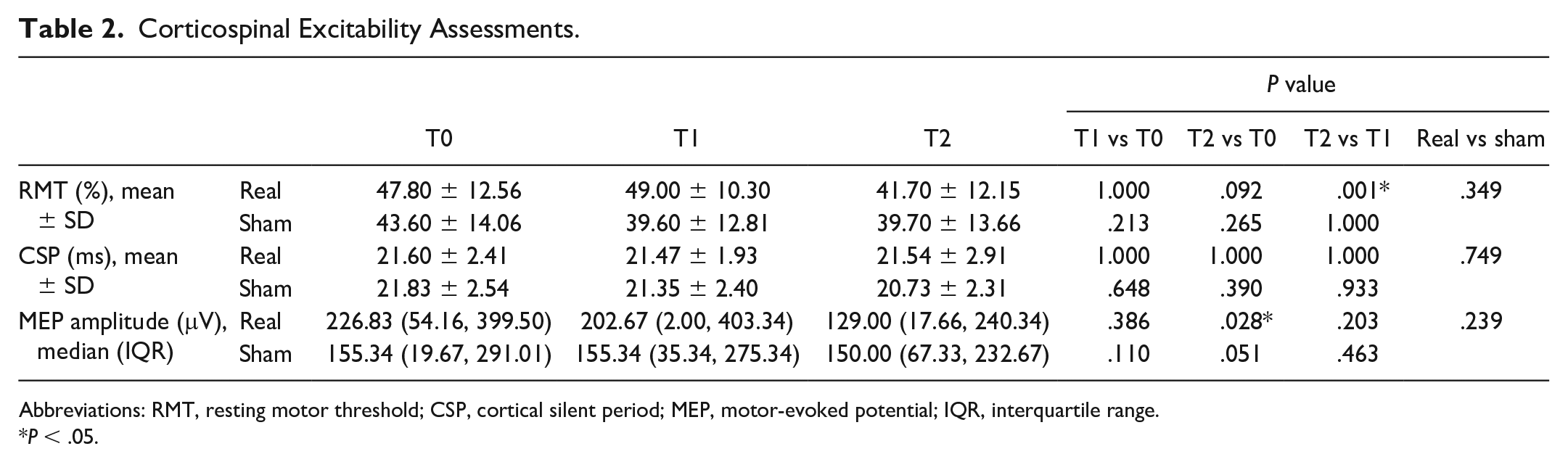
Abbreviations: RMT, resting motor threshold; CSP, cortical silent period; MEP, motor-evoked potential; IQR, interquartile range.
P < .05.
Discussion
The present randomized, double-blind, sham-controlled pilot study was designed to explore the short-term effects and potential mechanisms of cerebellar iTBS coupled with physiotherapy on balance function and its association with corticospinal excitability in subacute and chronic stroke patients to inform a future larger study. Our results demonstrated that cerebellar iTBS coupled with routine physical therapy promotes balance and motor recovery in poststroke stroke patients with hemiparesis by influencing cerebello-cortical plasticity.
Balance Function and Cerebellar iTBS Coupled With Physiotherapy
Balance function is associated with walking ability, fall risk, and functional independence after stroke.32-34 The current study showed that 2 weeks of treatments of cerebellar iTBS combined with physiotherapy increased the BBS score from 46.5 to 50.75. This finding is consistent with that of Koch et al 15 who reported that the BBS score increased from 35 to 47 points after 3 weeks of cerebellar iTBS in chronic stroke patients. Controversially, Kim et al 11 reported that inhibitory low-frequency rTMS (1Hz) improved performance in the BBS in posterior circulation stroke. Possible reasons for the differential results of the studies, applying excitatory versus inhibitory NIBS include the timing (chronic vs acute) or intensity and mode of stimulation (subthreshold TBS vs suprathreshold rTMS). In the current study, we applied iTBS before physiotherapy as its repeated pulses can induce lasting changes in cortical excitability via synaptic associative plasticity and thus modify behavior, possibly improving the balance function in stroke. 35 In addition, Wee et al 36 reported that the BBS negatively correlated with the length of hospital stay; thus, improvements in balance recovery can reduce the length of stay, reduce the amount of medical resources used, and alleviate the economic burden of families. Therefore, these findings indicate that cerebellar iTBS can be a low-cost, effective, and highly efficient tool for balance rehabilitation after stroke.
Trunk Function and Cerebellar iTBS Coupled With Physiotherapy
Trunk dysfunction is common in poststroke patients and leads to a lack of trunk control, a loss of core stability, and an increased risk of falls.37,38 In this study, the TIS scores increased in both groups. A possible explanation for this result might be that trunk control training based on the Bobath concept 39 was included in physiotherapy, and this training can provide increased improvement in trunk function, balance, and gait in stroke patients compared with routine exercises. However, cerebellar iTBS coupled with physiotherapy improved overall trunk function more in poststroke stroke patients compared with the sham group. This discrepancy may be attributed to the effects of cerebellar iTBS, which may increase the efficacy of physiotherapy by activating the contralesional cerebellum. 7
Lower Extremity Motor Function and Cerebellar iTBS Coupled With Physiotherapy
Lower extremity motor function did not significantly differ between the 2 groups. This finding is consistent with that of Koch et al 15 who explored whether cerebellar iTBS could improve balance and gait functions in patients with chronic stroke. Moreover, the analysis of FMA scores revealed a similar mild increase at T1 and T2 for both the cerebellar iTBS and sham iTBS groups. A possible explanation for this finding might be that the improvement in lower extremity motor function was provided by physiotherapy, which was applied in both groups rather than cerebellar iTBS. 39 Another possible explanation for this finding is that the period of cerebellar stimulation was too short to improve the lower extremity motor function.
ADL and Cerebellar iTBS Coupled With Physiotherapy
Similarly, no significant differences were found in ADL between the cerebellar iTBS and sham iTBS groups. Regarding BI scores, we also found a similar mild increase in the 2 groups after 2 weeks of treatment, and a similar result was also reported by Koch et al. 15 A possible explanation for these results may be the lack of adequate cerebellar stimulation. We could extend the period and/or increase the intensity of cerebellar stimulation in future studies.
The Effects of Cerebellar iTBS on Corticospinal Excitability
The results of this study show that cerebellar iTBS can reduce the MEP amplitude of the affected hemisphere, indicating an inhibition in corticospinal excitability in the affected hemisphere. 40 However, this result should not necessarily prevent the exploration of the potential mechanisms of cerebellar iTBS coupled with physiotherapy on the balance function, as it is exclusively based on MEP changes. According to the previous study, we infer that the potential mechanism of balance recovery after the treatment of cerebellar iTBS coupled with physiotherapy is related to the cerebello-thalamocortical circuit, given that the cerebellum can regulate various motor functions by affecting M1 and the corticospinal output pathways through the cerebello-thalamocortical circuit. 41 Cerebellar Purkinje cells form an inhibitory postsynaptic connection with deep cerebellar nuclei (DCN), whereas DCN form an excitatory synaptic connection with the motor cortex through the thalamus ventralis. 42 An animal study has suggested that both long-term depression (LTD) and long-term potentiation (LTP) exist in the cerebellum. When 2 inputs to a Purkinje cell, one from a climbing fiber and the other from a set of granule cell axons, are repeatedly associated, the input efficacy of the granule cell axons in exciting the Purkinje cell is persistently depressed; thus, LTD occurs. When mossy fibers receive iTBS or long-term, high-frequency (100 Hz) electrical stimulation, the activation of the granulosa cells can be persistently enhanced, causing LTP. 43 Because the intensity of iTBS is very low (80% AMT), iTBS may activate interneuron subsets rather than the output pathway of the dentate nucleus directly. 18 In the current study, therefore, cerebellar iTBS may have induced plasticity changes at different levels, such as cerebello-thalamic synapses and thalamocortical synapses, leading to an increase in corticospinal excitability by inducing LTP. 18 Moreover, cerebellar stimulation can activate the parietal-frontal neural network; thus, cerebellar plasticity induced by iTBS may be related to changes in neural activity in the posterior parietal cortex. 15
Implications for Neurorehabilitation
The present results suggest that cerebellar iTBS can improve balance and motor recovery in stroke patients. Overall, iTBS is relatively easy to apply and can be tolerated by different clinical populations.15,44 Therefore, implementation is theoretically feasible in a clinical setting but not suitable during home practice because the magnetic stimulator is relatively expensive for a single family. However, before clinical embedding is considered on a large scale, more information on the safety and risks of iTBS, especially regarding its application across multiple sessions, is needed. 44
Limitations
We acknowledge that the present study has some limitations. First, although the sample size of the present study was estimated rigorously, we recommend that studies with parallel designs, larger samples sizes and multiple centers are conducted in the future. Second, the measurements of corticospinal excitability can be used to explain only a portion of the potential mechanisms of cerebellar iTBS; future studies should include more detection tools (such as TMS-EEG, functional near-infrared spectroscopy, and functional magnetic resonance imaging) to evaluate the changes in multiple brain regions. Third, therapy regimen, depression, and mood could affect motivation and impact the physiotherapy effects for rehabilitation, thus we should consider these effects during clinical application and discuss them in detail in future work. Fourth, the current pilot study was designed to explore the short-term effects of cerebellar iTBS coupled with physiotherapy. More, follow-up studies are needed to demonstrate its long-term effects. However, before long-term trials are initiated, a shorter study is needed for optimization. We will extend the period of the intervention to explore the long-term efficacy of iTBS in future formal trials.
Conclusions
This is the first study to examine the effects of cerebellar iTBS coupled with physical therapy on balance recovery and corticospinal excitability in subacute and chronic stroke patients. Significant improvement in balance and trunk function outcomes was observed after cerebellar iTBS, but no evidence was found that cerebellar iTBS affects lower extremity motor function and corticospinal excitability. However, the combination of cerebellar iTBS with physiotherapy rather than cerebellar magnetic stimulation alone is responsible for the improvement in functional outcomes. Therefore, the functional recovery of stroke patients may be accelerated and improved if we apply cerebellar iTBS in the subacute and chronic stages of stroke.
Supplemental Material
Appendix – Supplemental material for Cerebellar Theta-Burst Stimulation Combined With Physiotherapy in Subacute and Chronic Stroke Patients: A Pilot Randomized Controlled Trial
Supplemental material, Appendix for Cerebellar Theta-Burst Stimulation Combined With Physiotherapy in Subacute and Chronic Stroke Patients: A Pilot Randomized Controlled Trial by Ling-Yi Liao, Yun-Juan Xie, Yi Chen and Qiang Gao in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We are grateful to the stroke survivors who participated in the project, and to the nursing team in the Department of Rehabilitation Medicine at West China Hospital at Sichuan University.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This study was supported by grants from the 1·3·5 project for disciplines of excellence–Clinical Research Incubation Project, West China Hospital, Sichuan University (Grant Number: 2020HXFH051).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
