Abstract
Background
The likelihood of regaining independent walking after stroke influences rehabilitation and hospital discharge planning.
Objective
This study aimed to develop and internally validate a tool to predict whether and when a patient will walk independently in the first 6 months post-stroke.
Methods
Adults with stroke were recruited if they had new lower limb weakness and were unable to walk independently. Clinical assessments were completed one week post-stroke. The primary outcome was time post-stroke by which independent walking (Functional Ambulation Category score ≥ 4) was achieved. Cox hazard regression identified predictors for achieving independent walking by 4, 6, 9, 16, or 26 weeks post-stroke. The cut-off and weighting for each predictor was determined using β-coefficients. Predictors were assigned a score and summed for a final TWIST score. The probability of achieving independent walking at each time point for each TWIST score was calculated.
Results
We included 93 participants (36 women, median age 71 years). Age < 80 years, knee extension strength Medical Research Council grade ≥ 3/5, and Berg Balance Test < 6, 6 to 15, or ≥ 16/56, predicted independent walking and were combined to form the TWIST prediction tool. The TWIST prediction tool was at least 83% accurate for all time points.
Conclusions
The TWIST tool combines routine bedside tests at one week post-stroke to accurately predict the probability of an individual patient achieving independent walking by 4, 6, 9, 16, or 26 weeks post-stroke. If externally validated, the TWIST prediction tool may benefit patients and clinicians by informing rehabilitation decisions and discharge planning.
Introduction
Almost two thirds of people have difficulty walking early after stroke. 1 Recovery of independent walking is affected by stroke severity,2-4 lower limb motor impairment,2-10 balance and trunk control,6,7,10,11 age,2-4,8-10,12 sensory impairment,2,8 and visual impairments such as homonymous hemianopia or visuospatial neglect.2,4,8,10 A patient’s potential for walking recovery is important for patients and their and families, and also to establish rehabilitation plans and determine discharge location.13,14 However, accurate walking prediction is difficult when based solely on clinical impression,15,16 leading to high variability in decision-making between clinicians, and potentially inequitable access to rehabilitation. 17
Emerging evidence supports the use of biomarkers and prediction tools to improve the accuracy of stroke recovery predictions and personalize stroke rehabilitation.5,10,18,19 Most walking prediction studies produce regression equations that predict independent walking at a single time point of 6 months post-stroke or discharge from hospital, which is of limited use in planning rehabilitation and discharge.7,8,20-23 Predicting walking ability at discharge from hospital is also problematic, because discharge decisions are based on cultural factors and local health services, in addition to walking ability. 23 A simple tool that predicts both whether and when an individual patient will recover independent walking after stroke is more likely to be useful to clinicians in routine practice.
In an exploratory study, we found that trunk control and hip extensor strength at 1 week post-stroke were the most important predictors for whether a patient will walk independently by 6 or 12 weeks, or remain dependent at 12 weeks post-stroke. 6 These clinical factors most strongly predicted walking recovery, despite the availability of both neurophysiological (transcranial magnetic stimulation) and neuroimaging (magnetic resonance imaging) biomarkers. While these initial results were promising, the sample size was small (n = 41), participants were recruited from only one site, and only two outcome timepoints were analyzed.
The aim of this study was to build on our previous work by investigating additional potential predictors, increasing the number of study sites, and increasing the number of outcome time points. Thus, we developed and internally validated the TWIST tool to predict whether and when a patient is likely to recover independent walking after stroke.
Methods
Participants
Patients were consented into the study between 19/02/2018 and 16/03/2020. Adults aged 18 years and over were recruited within three days of new ischemic or hemorrhagic stroke from the acute stroke services of two hospitals in a regional network. Patients were included if they had new lower limb weakness (Medical Research Council (MRC) strength grade 24 less than 5/5 in any lower limb muscle group) and were unable to walk independently (Functional Ambulation Category (FAC) 25 score less than 4). Previous stroke was allowed if the patient had no residual lower limb weakness and was walking independently prior to the new stroke. Participants were excluded if they had cerebellar or bilateral stroke, used a walking frame pre-stroke, had a pre-stroke FAC score less than 4, had communication or cognitive deficits precluding informed consent, were medically unwell at the time of recruitment, or had a life expectancy less than 6 months.
The study was approved by the national ethics committee. All participants provided written informed consent in accordance with the Declaration of Helsinki. Anonymized data will be provided upon reasonable request to the corresponding author.
Clinical and Demographic Data
Demographics and stroke characteristics.
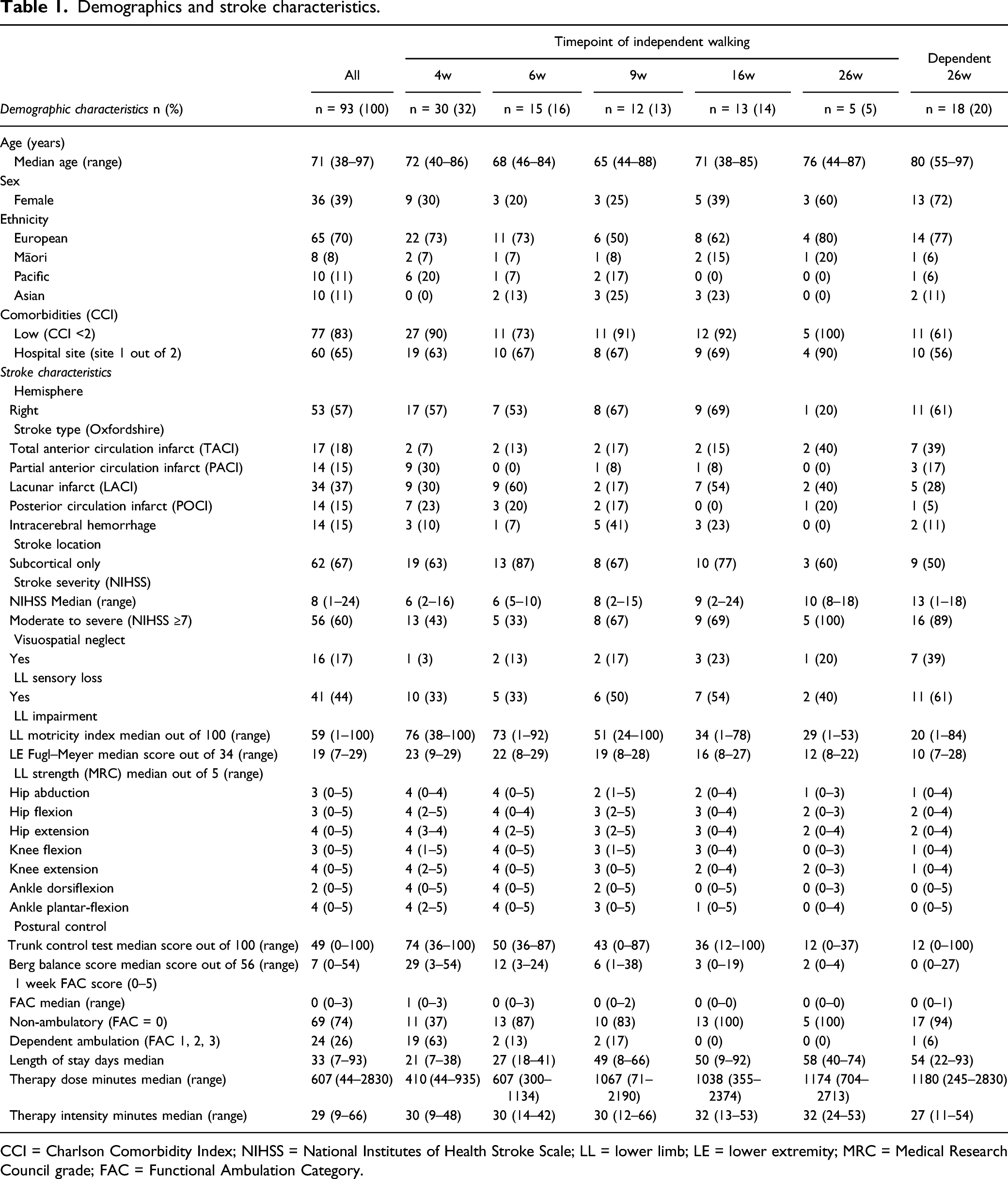
CCI = Charlson Comorbidity Index; NIHSS = National Institutes of Health Stroke Scale; LL = lower limb; LE = lower extremity; MRC = Medical Research Council grade; FAC = Functional Ambulation Category.
Functional Ambulation Category score was re-assessed at one week post-stroke. Participants who still required assistance or supervision to walk on level surfaces (FAC < 4) were enrolled in the study. Enrolled participants completed the following clinical assessments at one week post-stroke: Lower limb motor impairment (lower extremity Fugl–Meyer scale) 27 ; lower limb muscle strength (MRC strength grades for hip abduction, flexion, and extension, knee flexion and extension, and ankle dorsi- and plantar-flexion) 24 ; trunk control (Trunk Control Test) 28 ; sitting and standing balance (Berg Balance Test) 29 ; lower limb power (Motricity Index) 30 ; visuospatial neglect (Star Cancellation Test)31,32; and lower limb sensory loss (monofilaments, plantar surface of the first toe) (Table 1).33,34
Assessment of Walking Outcomes (FAC)
Functional Ambulation Category was re-assessed at 2, 4, 6, 9, 12, 16, 20 and 26 weeks post-stroke. The FAC score was dichotomized at each assessment time point to classify participants as being able to walk independently (FAC ≥ 4) or dependent (FAC < 4).7,9 The primary outcome was time post-stroke by which independent walking (FAC ≥ 4) was achieved.
Follow-up assessments were conducted in person while the participant was still in hospital, and at 12 and 26 weeks post-stroke. The remaining follow-up assessments were completed via telephone. A standardized interview schedule was used for all phone consultations. Information about walking ability was verified with a family member if at home, and the participant’s physiotherapist from the community rehabilitation team, or staff at the residential care facility. All assessments were completed by an experienced, blinded, research physiotherapist not involved in patient care, and therapists involved in routine clinical care were also blinded to the results of the research assessments.
Inpatient Therapy Measurement
Time spent participating in therapy was recorded from acute stroke admission to discharge from inpatient care. The number of minutes spent in active lower limb therapy (lower limb, trunk, walking, or balance training) was recorded immediately after the therapy session by the treating therapist. Lower limb therapy dose was defined as the total number of lower limb therapy minutes during the inpatient stay. Lower limb therapy intensity was calculated as the mean number of minutes of lower limb therapy per available therapy day (Monday to Friday). Therapy dose and intensity were collected to characterize the sample and were not considered as possible variables for the prediction tool. This is because the tool is designed to be used at one week post-stroke, when patients are beginning rehabilitation, and total therapy dose and intensity are not yet known.
Data Processing
Participants’ data were removed from analyses if: they had a second stroke or other significant medical event after enrollment, affecting walking ability; they died before achieving independent walking; or were lost to follow-up before achieving independent walking (Figure 1). Study Flowchart. Reasons for exclusion were classified as “unsuitable for research” or “unsuitable for the TWIST prediction tool” if it were to be used a clinical context. Approximately 2/3 of the patients who were suitable for research at day 3–5 post-stroke were unsuitable for the TWIST prediction tool because they were independently mobile or had no lower limb weakness. Patients were classified as unable to consent if they met all other inclusion criteria but had significant communication or cognitive deficits precluding informed consent to participation in a research study.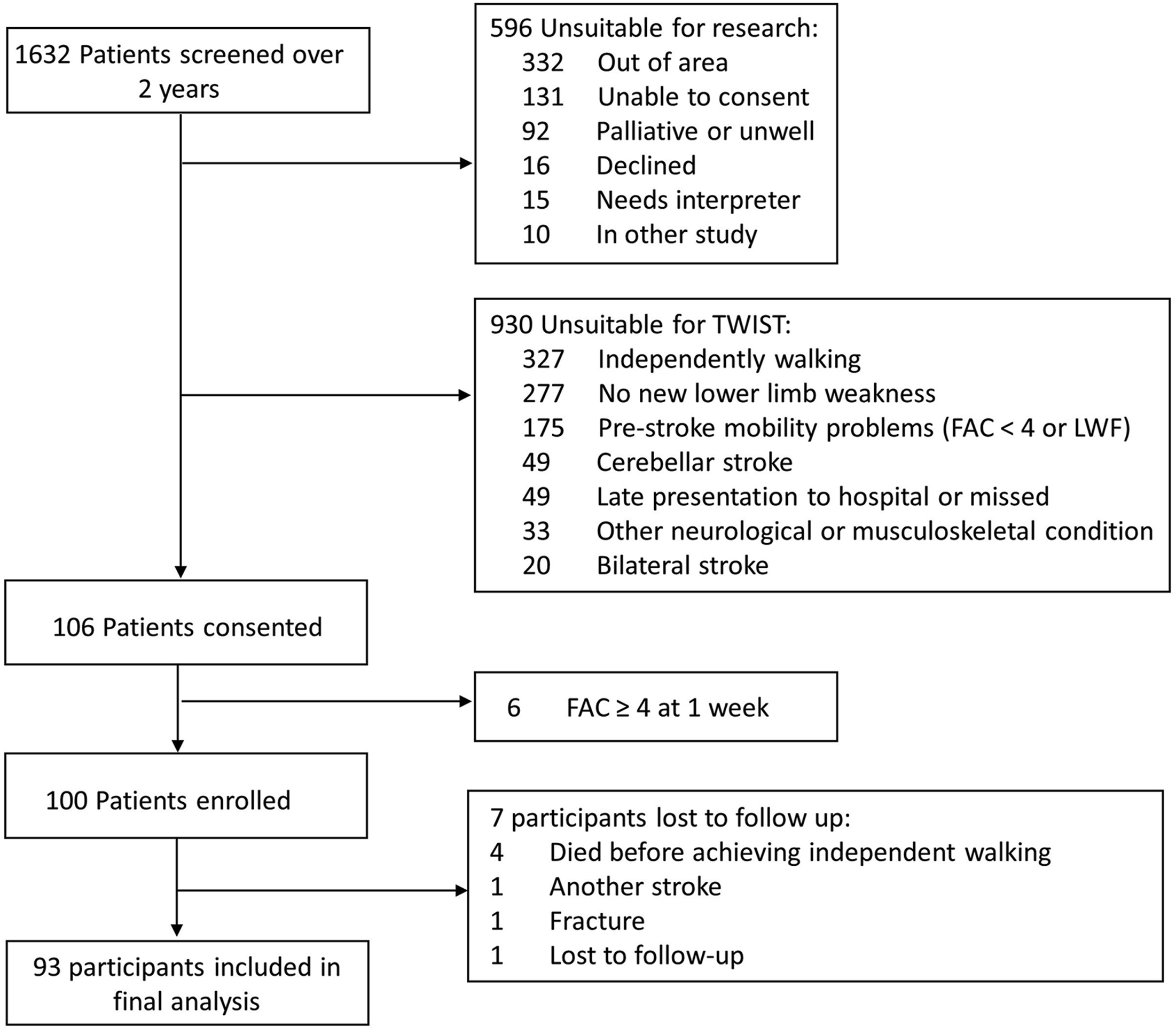
TWIST Timepoint Selection
Participants were categorized as walking independently (FAC ≥ 4) at 2, 4, 6, 9, 12, 16, 20, or 26 weeks, or dependent (FAC < 4) at 26 weeks. Any timepoint with fewer than 10 participants was combined with the subsequent timepoint to ensure at least 10 participants per timepoint. The timepoints included in the final analysis were 4, 6, 9, 16 and 26 weeks post-stroke (Table 1).
TWIST Variable Selection
The target recruitment sample size was 100. Allowing for attrition, we expected at least 10 participants would be available per predictor variable entered into the model, and at least 10 participants would achieve independent walking at each timepoint post-stroke.
Of the 93 participants analyzed in the final dataset, 75 achieved independent walking within 26 weeks post-stroke. The events per variable ratio was set at 10. Therefore, a maximum of seven potential predictor variables could be used for model derivation. 35 Seven variables were selected a priori for model derivation with the following criteria: established predictor in previous literature 10 ; commonly used in clinical care; ease of use in non-ambulatory patients early after stroke. 35
The following factors are consistently identified in the literature as predictors for walking recovery after stroke: age,2-4,8-10,12 lower limb strength,2-10 sitting balance and trunk control,6,7,10,11 and vision/visuospatial neglect.2,4,8,10 For lower limb strength, MRC grades for hip flexion, knee extension and ankle dorsiflexion at one week post-stroke were selected based on previous literature 10 and ease of use in this clinical population. For balance measures, the Trunk Control Test 6 and the Berg Balance Test at one week post-stroke were included to capture the full range of trunk and balance assessment from bed mobility to dynamic standing balance. The Star Cancellation Test at one week post-stroke was used to determine the presence or absence of visuospatial neglect. Visuospatial neglect was considered present if the Star Cancellation Test laterality index score was < .46 or > .54.
Potential predictor variables that were considered but not included were as follows: NIHSS, Motricity Index, MRC grade for hip extension strength, lower extremity Fugl–Meyer Assessment, and LL sensory loss. NIHSS is not routinely collected at one week post-stroke, and in this dataset was collected too early at day 3 post-stroke. The Motricity Index tests hip flexion, knee extension and ankle dorsiflexion strength, which were included as predictors individually. Hip extension strength was considered as it was one of two predictors identified in the original TWIST tool; however, it can be difficult to obtain in non-ambulatory people post-stroke, and therefore it was excluded from this analysis. LE Fugl–Meyer Assessment is not routinely used in clinical practice, and there is limited evidence for LL sensory loss as an important clinical predictor for walking recovery.
The seven predictors put forward for model derivation were as follows: age, MRC grades for hip flexion, knee extension and ankle dorsiflexion, Trunk Control Test, Berg Balance Test, and the presence or absence of visuospatial neglect.
TWIST Model Development
The dependent variable was the time at which independent walking was achieved. Backwards step-wise multivariate Cox hazard regression analysis was conducted for the seven predictor variables and significance set at P < .05. The surviving variables were age, knee extension MRC grade, Berg Balance Test, and the presence or absence of visuospatial neglect (Supplemental Figure 1, Supplemental Table 1).
Ordinal and continuous variables were converted to categorical variables by determining a cut-off score. ROC curves were generated for each ordinal variable (age, knee extension MRC grade, Berg Balance Test) for each timepoint. The ROC coordinates with the highest specificity and sensitivity for each variable at each time point determined the cut-off value. Cut-off values were rounded up to the nearest integer for knee extension MRC grade and Berg Balance Test, and to the nearest 5 years for age. Two cut-off values were identified for age and Berg Balance Test and both were included in subsequent analyses (Supplemental Figure 1).
Cox multivariate regression analysis was conducted with age, knee extension MRC grade, and Berg Balance Test coded to the defined cut-offs and visuospatial neglect coded as present/absent. Variables and variable cut-offs that were not significant (P ≥ .05) were removed from the final model (Supplementary Figure 1).
The TWIST scoring system was developed by weighting variables based on the β-coefficients in the final model, rounded to the nearest integer. Each variable was assigned a score of 0/1 or 0/1/2 and variables were summed to assign participants a TWIST score.36,37
Model Performance and Internal Validation
Participants’ TWIST scores were entered as the only predictor into a binary logistic regression analysis with bootstrapping (1000 samples and 95% confidence intervals) for each time point. Model calibration was assessed with calibration plots and the Hosmer–Lemeshow goodness of fit test for each time point, with P > .05 indicating acceptable calibration. Discrimination was assessed by constructing an ROC curve for the TWIST score at each time point and calculating the C statistic.
The binary logistic regression model at each time point was used to calculate the probability of achieving independent walking (FAC ≥ 4) for each TWIST score, with 95% confidence intervals. Overall accuracy, sensitivity, specificity, positive predictive value and negative predictive value were also calculated for the model at each time point, with 95% confidence intervals.
The probabilities for each TWIST score at each time point were entered into a k-means cluster analysis to identify three levels of likelihood for achieving independent walking (unlikely, possible, likely). An example plain language description of the likely recovery of independent walking was constructed for each TWIST score, to illustrate how the TWIST predictions could be communicated by clinicians if externally validated in subsequent research.
Results
There were 106 participants consented at day three to five post-stroke. One hundred participants still had an FAC score < 4 at one week post-stroke and were enrolled in the study, with 93 retained in the final analysis (median age 71 (38–97) years; 39% female, 74% non-ambulatory (FAC = 0); 60% with moderate-severe stroke based on NIHSS score (Figure 1, Table 1). Most participants (n = 57, 61.3%) achieved independent walking by nine weeks post-stroke, with 18 (19.4%) achieving independent walking between nine weeks and 26 weeks post-stroke. The remaining 18 (19.4%) did not achieve independent walking by 26 weeks post-stroke.
TWIST Timepoint Selection
Fewer than 10 participants achieved independent walking at each of 2, 12, 16, 20 and 26 weeks post-stroke. Participants at these timepoints were combined with those who achieved independently walking at next timepoint until at least 10 were in the group. Final timepoints were 4, 6, 9, 16, and 26 weeks post-stroke (Table 1).
TWIST Model Development
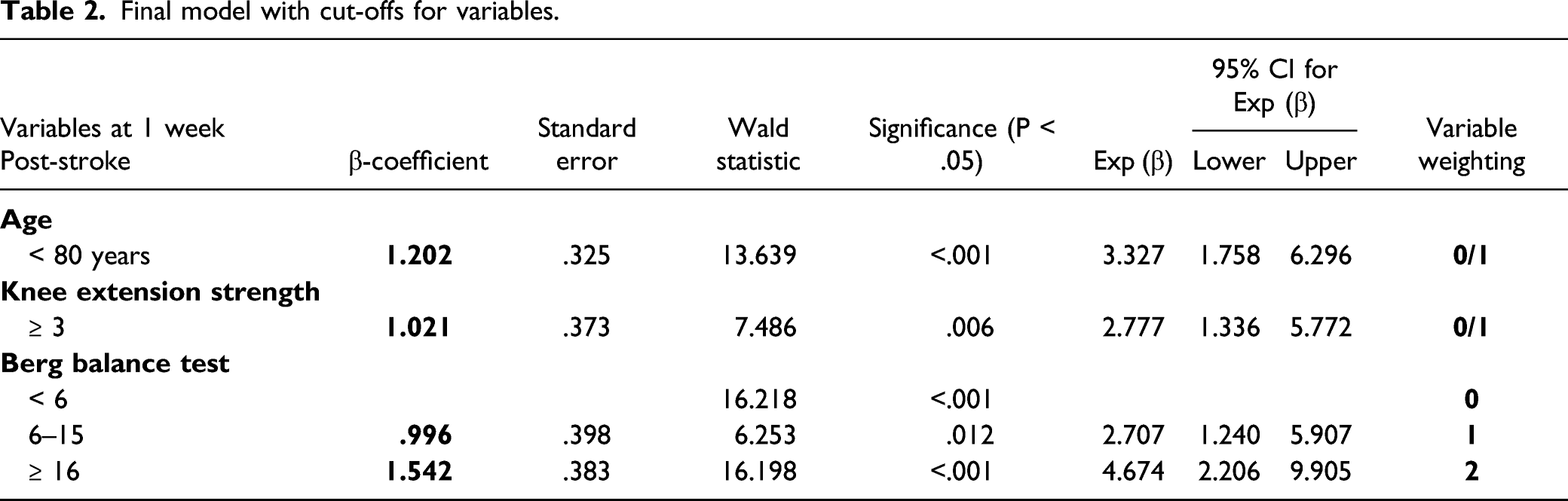
Seven variables were entered into Cox multivariate regression analysis and four survived: Age (P < .001), MRC grade for knee extension strength (P < .001), Berg Balance Test (P < .001), and the presence/absence of visuospatial neglect (P = .044) (Supplemental Table 1).
The following cut-offs were identified with ROC curve analyses: Age < 75 years, 75–79 years, and ≥ 80 years; Berg Balance Test score < 6, 6–15, and ≥ 16 out of 56; and knee extension strength MRC grade ≥ 3 out of 5. When the four variables were entered into Cox multivariate regression analysis with the defined cut-offs, the age cut-off of < 75 years (P = .649) and the presence or absence of visuospatial neglect (P = .366) were no longer significant and were removed from the model (Supplemental Figure 2, Supplemental Table 2). Age ≥ 80 (P < .001), knee extension strength MRC grade ≥ 3 out of 5 (P = .007), Berg Balance Test score < 6, 6–15, and ≥ 16 out of 56 (P = .002, P = .042 and P < .001, respectively) were significant in the multivariate Cox hazards regression and were retained in the final model (Supplemental Figure 2, Supplemental Table 2).
Final model with cut-offs for variables.
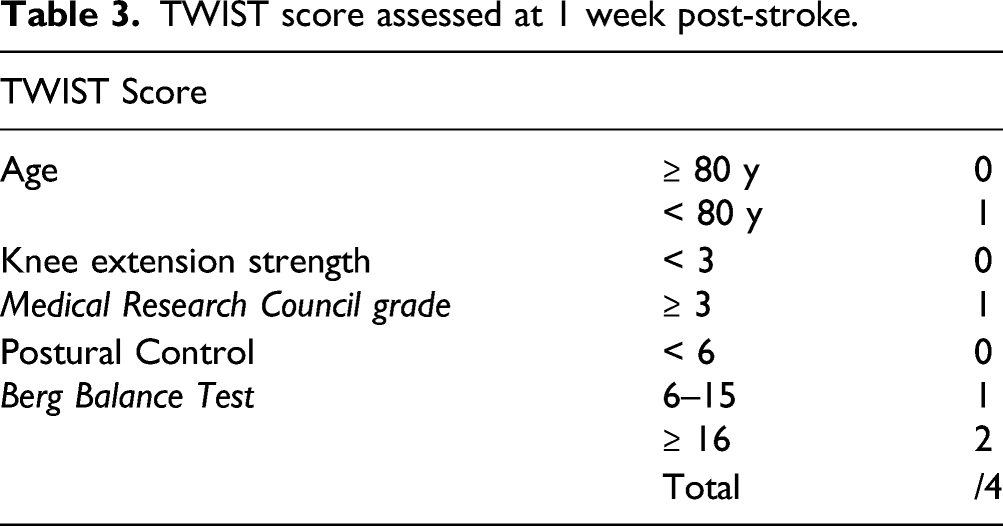
TWIST score assessed at 1 week post-stroke.
Model Performance and Internal Validation
Hosmer–Lemeshow tests at each timepoint were not significant (all P > .2) indicating acceptable goodness of fit. Calibration plots showed good agreement between predicted and observed outcomes with calibration slopes ranging from .97 (6 weeks) to 1.05 (26 weeks) (Supplemental Figure 3). Discrimination, measured with the C statistic, ranged from .87 (95% CI 0.80–.95) at 26 weeks to .94 (95% CI 0.89–.98) at 6 weeks.
The model accurately predicted independent walking at all five time points, ranging from 83% (95% CI 74–90%) at 16 weeks to 86% (95% CI 77–92%) at 4 and 6 weeks. Positive predictive value ranged from 84% (95% CI 66–93%) at 4 weeks to 98% (95% CI 89–100%) at 16 weeks. Negative predictive value ranged from 60% (95% CI 48–70%) at 16 weeks to 87% (95% CI 79–92%) at 4 weeks (Supplemental Table 3).
There were five TWIST scores for each of five timepoints, giving a total of 25 probabilities of achieving independent walking. K-means cluster analysis identified three likelihood clusters based on these 25 probabilities. The three clusters had probabilities ranging from 0–20% (unlikely), 35–68% (possible), and 79–100% (likely). Most TWIST score and timepoint combinations predict either “unlikely” (9/25) or “likely” (11/25) to be independently walking. “Possible” achievement of independent walking is represented by the smallest number of TWIST score and timepoint combinations (5/25). The probabilities for each TWIST score at each timepoint are presented in Figure 2A and 2B. Probabilities are color-coded according to cluster. An example plain language interpretation of the TWIST predictions is provided (Table 4). TWIST score interpretation. A. Probability of achieving independent walking at each time point for each TWIST score (% (95% CI)). Red shading means the person is unlikely to be walking independently (probability 0–10%); Orange shading means the person has some chance of walking independently (probability 35–68%); Green shading means the person is likely to be walking independently (probability 79–100%). B. Time predicted to achieve independent walking after stroke for each TWIST score. Example plain language interpretation of the TWIST predictions.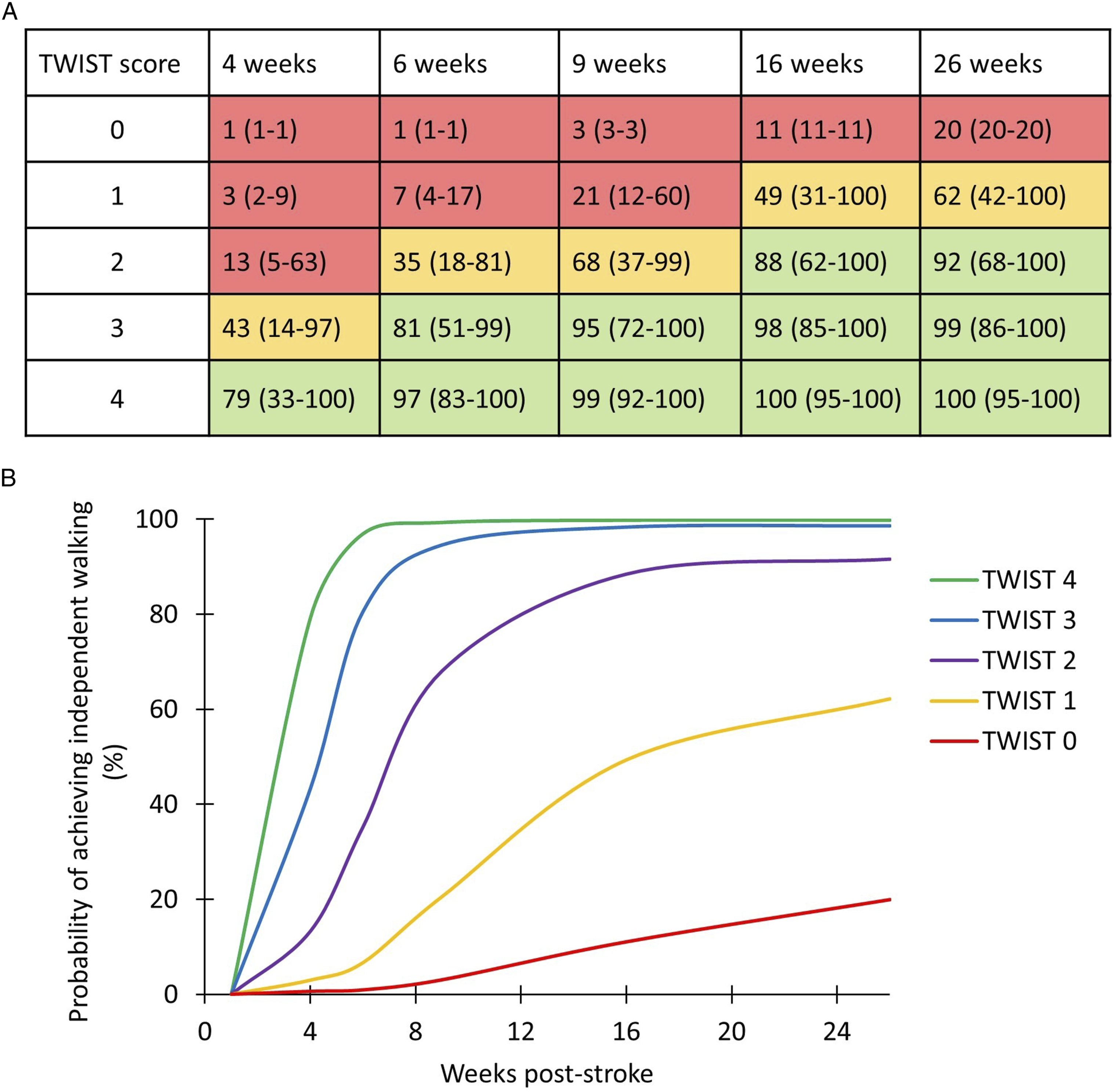

Discussion
This is the first study to develop a tool that at one week post-stroke accurately predicts whether a patient is likely to be walking independently by 4, 6, 9, 16, or 26 weeks post-stroke, or remain dependent at 26 weeks. The TWIST prediction tool is at least 83% accurate for all timepoints. Accurate walking predictions are of value for several reasons. Knowing both whether and when a patient is likely to walk independently is expected to assist rehabilitation and discharge planning by clinical teams.9,23 It will also give patients and their families realistic expectations of the amount and duration of support they will need after discharge and empower them to be more involved in decision-making.38-40
The TWIST prediction tool consists of three routine, quick and simple tests conducted at the bedside at one week post-stroke: age (≥ 80 or < 80 years), knee extension MRC grade (≥ 3 or < 3 out of 5) and Berg Balance Test score (< 6, 6 to 15, or ≥ 16 out of 56). Age and knee extension MRC grade are awarded a score of 0 or 1, and the Berg Balance Test is awarded a score of 0, 1, or 2 based on whether the cut-offs of 6 or 16 are reached (Table 2). The scores are then summed to calculate the TWIST score which can range from zero to four points (Table 3). Almost all participants (97%) with a TWIST score of four points achieved independent walking by six weeks post-stroke, with 79% achieving independent walking by four weeks. In contrast, most participants (80%) with a TWIST score of zero remained dependent at six months post-stroke. The results are presented graphically and in a simple to use format that allows a prediction to be given for an individual patient (Figure 2, Table 4). 23
Age was a significant predictor of walking recovery, consistent with prior evidence that older age is a negative predictor for stroke recovery.3,39,41-43 Advanced age may affect a person’s ability to engage in intensive rehabilitation and to use post-stroke compensatory strategies for walking. Elderly patients are more likely to have lower exercise tolerance, have a higher risk of falls, have more comorbidities, 44 and experience age-related changes to muscle mass, bone density, balance/vestibular mechanisms, joint health, eyesight, and cognition.45-47 In addition, age-related white matter changes may negatively affect stroke outcomes for the elderly and increase the risk of post-stroke cognitive impairment.47,48 Age has not previously been dichotomized for predicting independent walking, but age of 80 years has been identified as an important age cut-off for upper limb outcome. 43
This study also supports previous findings that lower limb strength and postural control are predictors for the recovery of independent walking.6,7,49,50 Knee extension against gravity (MRC grade 3/5 or more) is important for stability during the stance phase of gait. Other muscle groups have previously been identified as important for walking performance,6,51-54 but relate more to walking speed and quality of movement. In contrast, independent walking is more reliant on the ability to maintain stability during the single support phase of gait. While the presence of active knee extension is a predictor for independent walking, the absence of knee extensor muscle activity (MRC = 0) at one week post-stroke does not preclude a person from achieving independent walking (Table 1).
Substantial postural control is needed to successfully use compensatory strategies for lower limb weakness. For example, hip circumduction, high stepping to clear the foot, limiting weight shift to the paretic side and the use of a walking aid all require postural compensation. 6 Therefore, both the Trunk Control Test and Berg Balance Test were included in this study as possible predictors. The Trunk Control Test assesses rolling to each side, moving from lying to sitting, and static sitting for 30 seconds. 28 In contrast, the Berg Balance Test assessments range from static sitting balance to more demanding standing balance activities such as picking up an object from the floor, functional reach, tandem stance, and single leg stance. 29 The Berg Balance Test was found to be a stronger predictor of independent walking than the Trunk Control Test. Two cut-off scores were identified for the Berg Balance Test, at 6 and 16 points out of a maximum of 56. A Berg Balance Test score less than 6 points indicates the patient is unable to stand or transfer independently or with minimal assistance. Participants with a Berg Balance Test score of 16 or more experienced the earliest recoveries (4 weeks), followed by those with a score of 6 to 15 (Table 1).
Visuospatial neglect was initially included as a predictor in the model but became non-significant once all variables with their cut-offs were included. Visuospatial neglect causes poor hemispatial awareness and midline orientation. 55 The influence of visuospatial neglect on postural control can affect a patient’s ability to weight shift during gait, reducing the ability to step with the paretic leg, and increasing the load on the paretic leg during stance phase. 55 Visuospatial neglect also presents a safety issue when maneuvering around obstacles, likely leading to a longer period of supervision than a person without visuospatial neglect might need. Most patients recover fully from visuospatial neglect within 6 months 57 ; thus, visuospatial neglect is more likely to have an influence on earlier recovery timepoints than later. This may be one of the reasons visuospatial neglect did not make it into the final model. Another possibility is that the Star Cancellation Test is not sensitive enough to predict time taken to achieve independent walking. The Star Cancellation Test assesses visuospatial neglect within a self-centered frame of reference (ego-centric). However, many people with stroke have object-centered (allocentric) visuospatial neglect. People with more severe allocentric neglect are less likely to make a full recovery from visuospatial neglect, potentially contributing to ongoing difficulties with walking safety. 56 For these reasons it would be advisable to continue to explore visuospatial neglect as a predictor for walking recovery, taking neglect subtypes into consideration.
Therapy dose and therapy intensity were measured during inpatient stay, with physiotherapists recording time engaged in active lower limb, trunk, walking and balance training. However, therapy dose and intensity were not included as predictors in the model. The amount of therapy a patient will complete cannot be accurately known in advance. Therapy intensity was around 30 minutes per day for all groups. However, the inverse relationship between total therapy dose and stroke severity is evident as those who took longer to achieve independent walking participated in more therapy than those who achieved independent walking by four weeks post-stroke. Therapy dose and intensity were measured and reported to characterize the standard care provided at the hospital sites used for the development of the TWIST tool. This information may assist future researchers to draw conclusions about the performance of TWIST when different amounts of therapy are completed.
An important issue when developing a prediction tool is how predictions will be communicated and influence rehabilitation planning. The TWIST tool predicts the likelihood of a patient walking independently at five timepoints within the first six months post-stroke. TWIST is designed to give clinicians, patients and families realistic timeframes for walking recovery, and should not be used to ration care or determine whether a patient is eligible for inpatient rehabilitation. 23 Care needs to be taken by clinicians when communicating the TWIST predictions. Table 4 provides a suggested interpretation of the TWIST tool which may be a useful starting point for clinicians when planning their conversations with patients and families. It is important to emphasize that TWIST predicts the recovery of independent walking, not overall stroke recovery. Patients who are unlikely to achieve independent walking can still achieve many important functional goals such as bed mobility, sitting balance, transfers, toileting, and walking short distances with assistance. Providing rehabilitation to help patients achieve these goals can have a large impact on independence, quality of life, discharge destination, and the level of support required.
The TWIST prediction tool has several strengths. The clinical assessments are routine, quick to administer, and can be completed at bedside at one week post-stroke by a range of qualified health professionals. The TWIST tool has a clear and accessible format that allows clinicians to combine the scores from each assessment and make a prediction for an individual patient. These strengths support future translation into clinical practice.
This study has some limitations. These include a relatively small sample size with only 5 participants achieving independent walking between 16 and 26 weeks. This is reflected in the lower specificity and negative predictive value at 26 weeks post-stroke. We took steps to mitigate the small sample size with a priori selection of variables entered into the regression analysis, reducing the number of potential variables from 23 to 7. Pre- and post-stroke cognitive impairment may influence engagement in rehabilitation and subsequent walking recovery, but cognition was not assessed. Future studies could include cognition and visuospatial neglect as potential predictors for walking recovery. People who used a walking frame prior to the stroke, or who had cerebellar stroke, were not included meaning that TWIST cannot be used for these patients. The use of a walking frame prior to stroke reflects pre-existing comorbidity related to ambulation and affects the expected level of walking independence post-stroke. Future studies could include participants who used walking frames prior to stroke and those with cerebellar stroke. The timeframe was limited to 26 weeks and it is possible that some participants subsequently achieved independent walking after this time. The TWIST tool has not yet been externally validated. This means it is not yet ready for use in clinical practice.
Conclusions
TWIST predicts with high accuracy the probability of a patient achieving independent walking by 4, 6, 9, 16, or 26 weeks post-stroke. The assessments are simple routine clinical tests administered at bedside one week post-stroke. If externally validated, the TWIST prediction tool may benefit patients and clinicians, and aid rehabilitation and discharge planning.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683221085287 – Supplemental Material for The TWIST Tool Predicts When Patients Will Recover Independent Walking After Stroke: An Observational Study
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683221085287 for The TWIST Tool Predicts When Patients Will Recover Independent Walking After Stroke: An Observational Study by Marie-Claire Smith, Alan P. Barber, Benjamin J. Scrivener and Cathy M. Stinear in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
The authors thank Luke Skinner, Gemma Nolan and Christine Mangold for assistance with data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Neurological Foundation of New Zealand 1735-PG; Auckland Academic Health Alliance 3 715 213
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
