Abstract
Background
Upper limb (UL) impairment in stroke survivors is both multifactorial and heterogeneous. Stratification of motor function helps identify the most sensitive and appropriate assessments, which in turn aids the design of effective and individualized rehabilitation strategies. We previously developed a stratification method combining the Grooved Pegboard Test (GPT) and Box and Block Test (BBT) to stratify poststroke UL motor function.
Objective
To investigate the resilience of the stratification method in a larger cohort and establish its appropriateness for clinical practice by investigating limitations of the GPT completion time.
Methods
Post hoc analysis of motor function for 96 community-dwelling participants with stroke (n = 68 male, 28 female, age 60.8 ± 14 years, 24.4 ± 36.6 months poststroke) was performed using the Wolf Motor Function Test (WMFT), Fugl-Meyer Assessment (F-M), BBT, and GPT. Hypothesis-free and hypothesis-based hierarchical cluster analyses were conducted to determine the resilience of the stratification method.
Results
The hypothesis-based analysis identified the same functional groupings as the hypothesis-free analysis: low (n = 32), moderate (n = 26), and high motor function (n = 38), with 3 exceptions. Thirty-three of the 38 participants with fine manual dexterity completed the GPT in ≤5 minutes. The remaining 5 participants took 6 to 25 minutes to place all 25 pegs but used alternative movement strategies to complete the test. The GPT time restriction changed the functional profile of the moderate and high motor function groups leading to more misclassifications.
Conclusion
The stratification method unambiguously classifies participants by UL motor function. While the inclusion of a 5-minute cutoff time for the GPT is preferred for clinical practice, it is not recommended for stratification purposes.
Keywords
Introduction
Stroke currently affects over 80.1 million people globally and contributes to 5.5 million deaths annually. 1 One of the most common deficits after stroke is upper limb (UL) hemiparesis. UL impairment affects approximately 75% to 80% of stroke survivors,2,3 and can compromise an individual’s ability to carry out activities of daily living (ADLs), resulting in reduced quality of life.4,5 UL impairment spans a wide spectrum and is influenced by a number of factors, such as the location of the lesion, size, and severity of the stroke.6 -8 Moreover, the level of UL impairment may vary according to fluctuations in function, recovery, compensation, and the presence of multiple impairments.9,10 With such heterogeneity, a variety of functional assessment tools with sensitivity across the poststroke spectrum are needed to unambiguously quantify UL motor function and guide clinical decisions about individualized rehabilitation therapy. There is no single assessment tool with sufficient sensitivity across the spectrum of residual voluntary motor capacity in chronic stroke. 11 UL stratification could limit the risk of patients receiving inappropriate rehabilitation due to over or underestimated baseline motor function. UL stratification may also reduce bias from unbalanced study arms in clinical research trials and supports more accurate estimations of the efficacy of rehabilitation strategies. 11
The Wolf Motor Function Test (WMFT) and the Fugl-Meyer Assessment (F-M) are commonly used as primary outcome measures in intensive UL stroke trials.12 -16 However, the WMFT is susceptible to floor effects for patients with low motor function, 17 whereas the F-M assessment is susceptible to ceiling effects for patients with high motor function.18,19 Moreover, the F-M and WMFT take a maximum of 30 and 45 minutes to complete, respectively, which limits their applicability in a time-limited clinical setting.
We previously developed a quick and objective stratification method for UL function based on the participant’s ability to complete 2 common tests of dexterity: the Box and Block Test (BBT), which assesses gross manual dexterity, and the Grooved Pegboard Test (GPT), which assesses fine manual dexterity. By assessing participants’ ability to complete both tests, we were able to objectively separate patients into low, moderate, and high motor function subgroups. 19 These tests are relatively easy to administer in clinical practice, requiring minimal training and together usually take less than 15 minutes to test bilaterally. However, before we can utilize this method of stratification in clinical practice, we need to test the stratification method’s resilience in a larger cohort. In particular, we wanted to address in detail BBT scores between 1 and 10 by including additional participants with low motor function. This study also affords us the opportunity to test the viability of the proposed GPT protocol timings.
The GPT records the time taken for participants to pick up, orient, and place 25 grooved pegs. Variable timing protocols have been reported including scoring the number of pegs placed per second in subacute stroke patients 20 and using a 3-minute completion time in neuropsychological evaluation. 21 The manufacturer’s guidelines specify a maximal 5-minute cutoff time limit should be applied (per side); however, this guideline has not been developed or validated through research. Therefore, the present study examined the use of a 5-minute cutoff time in a cohort of community-dwelling stroke patients.
The aim of this study was to evaluate the resilience of our previously developed stratification method in a larger cohort of stroke survivors. A secondary aim was to investigate the stratification method’s applicability for clinical practice by investigating the appropriateness of the specified 5-minute GPT cutoff time in a cohort of community-dwelling stroke participants. It was hypothesized that the stratification method would unambiguously classify participants into subgroups with low, moderate, and high motor function; and the 5-minute GPT cutoff time would allow sufficient sensitivity to use the GPT as a stratification tool in clinical practice.
Methods
Participants
Ninety-six participants (60.8 ± 14.0 years mean ± SD, range 18-90 years) were recruited from the rehabilitation units of St Vincent’s and Prince of Wales Hospitals, Sydney. A summary of the participants’ demographics are summarized in Table 1. All participants were hemiparetic with UL involvement following unilateral stroke in the territory of the middle cerebral artery. The inclusion criteria were the following: (1) minimum age of 14 years, (2) ability to understand and communicate in English, and (3) cognitive competency assessed as a Mini-Mental State Examination score of ≥24. Participants were excluded if they presented with any comorbidities significantly affecting UL sensorimotor function or with unstable blood pressure. Informed participant consent was obtained prior to enrolment. This study was approved by the St Vincent’s Hospital Human Research Ethics Committee (HREC/10/SVH/20). Data included in this study were previously collected during baseline assessments for 3 studies which investigated UL assessment and rehabilitation strategies in chronic stroke survivors.11,16,19 Sixty-seven participants from this study were included in our original analysis. 19
Patient Demographics and Characteristics.

Assessments
The following assessments were used to investigate UL motor function, 19 that is, the residual functional ability of the UL. The WMFT assessed the more affected UL and includes 15 timed tasks (eg, time taken to pick up a pencil from a table, scored in seconds with a maximum score of 120 seconds per task) and 2 strength-based tasks (grip strength and weighted forearm lift). 17 The UL motor subscale of the F-M comprises 33 items that investigate the function, range of motion, speed of movement, grasp, and reflex activity of the more-affected UL. Each item is subjectively scored on an ordinal scale (0 to 2), where 0 represents inability to perform the activity, and 2 represents full ability. 18
The BBT (Patterson Medical, Inc) was used to assess gross manual dexterity and consists of moving as many 2.5 × 2.5 cm blocks with the thumb and index finger over a 20-cm partition in 60 seconds. 22 The GPT (Lafayette Instrument) assesses fine manual dexterity and visual-motor coordination, and requires the participant to pick up, orientate, and place 25 identical grooved pegs. 23 There were no time restrictions placed on the GPT in the present study. All assessments were administered by researchers from Neuroscience Research Australia, who had completed ongoing standardized training. All assessments followed standardized protocols and were filmed for cross-checking and verification when required.
Stratification Method
In this study, we replicated the stratification methodology developed by Thompson-Butel et al, 19 which was based on clinical observations made during a trial investigating the efficacy of UL rehabilitation in a cohort of stroke survivors. Our original hypothesis was that the floor effects of the BBT and the GPT could be used to identify a time-efficient method to objectively differentiate UL motor function. In brief, all participants first attempted the BBT to determine gross manual dexterity. Those who moved ≤1 block were classed with low motor function. Those who moved >1 block in the BBT then attempted the GPT to assess fine manual dexterity. Participants able to move >1 block on the BBT but unable to place all 25 pegs in the GPT were classed with moderate motor function, and those able to move >1 block on the BBT and successfully place all 25 pegs in the GPT were classed with high motor function (Figure 1).
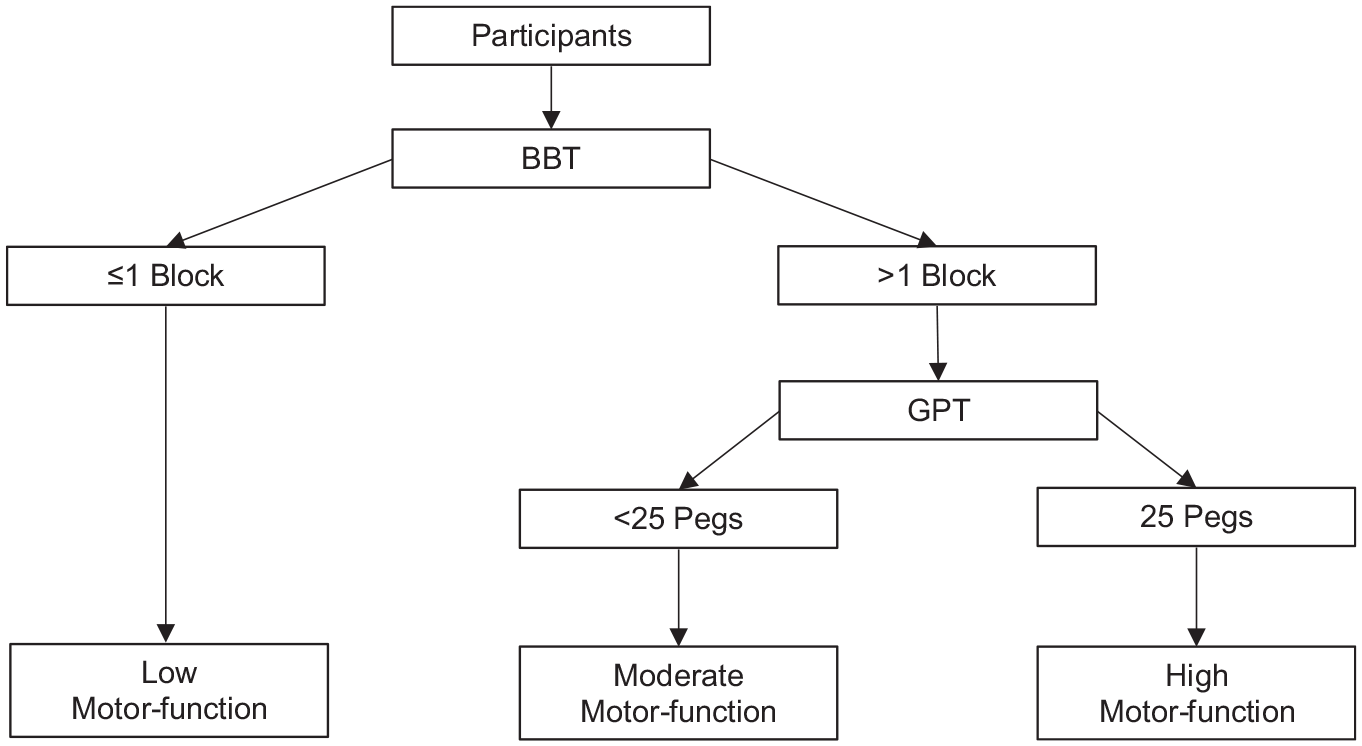
Schematic representation of original stratification method. The floor effects of the Box and Block test (BBT) and Grooved Pegboard Test (GPT) are used to stratify participants’ upper limb motor function. Participants able to move ≤1 block on the BBT are classified with low motor function, those able to move >1 block on the BBT but are unable to complete the GPT are classified with moderate motor function, and those able to move >1 block on the BBT and are able to complete the GPT are classified with high motor function.
Data Analysis
Three series of hierarchical cluster analyses were conducted to evaluate the resilience of the stratification method using IBM SPSS Statistics (V25). In each series, we compared hypothesis-based and hypothesis-free cluster analysis.24,25 In the hypothesis-based analysis, we specified 3 groups of motor function a priori in accordance with clinical observation by inputting dichotomous scores of 2 functional UL assessments into the hierarchical cluster analysis. We then input the raw data from 6 functional UL assessments in hypothesis-free analyses, specifying 2, 3, 4, and 5 groups. Greater than 5 clusters were not chosen as it would provide little relevance and/or benefit to clinicians and allied health therapists in regard to the participant’s assessment and rehabilitation options. We then compared the results from the hypothesis-free analysis to determine which grouping of participants, that is, 2, 3, 4, or 5, best matched the 3 groups of motor function in the hypothesis-based analysis. There is no widely accepted minimum sample size for a cluster analysis; however, a sample size of 2k cases (where k = number of variables) is suggested. 26 This equates to a minimum sample size of 64 cases in this study.
Unrestricted GPT Completion Time
A hypothesis-based hierarchical cluster analysis using the dichotomous scores for the BBT and GPT was first performed to investigate whether the same 3 clusters of motor function would be confirmed in a larger cohort using our original stratification method. 19 In brief, a hypothesis-based hierarchical cluster analysis generated 3 groups using dichotomous scores derived from the BBT and GPT, representing the inability (a score of 0, representing floor effects) or ability (a score of 1) to move >1 block on the BBT and to place all 25 grooved pegs in the GPT, respectively (ie, 0 = BBT, 0 = GPT: low motor function; 1 = BBT, 0 = GPT: moderate motor function; 1 = BBT, 1 = GPT: high motor function). As this was our hypothesis from our previous stratification method, only 3 groups were examined.
We then specified 2, 3, 4, and 5 clusters to perform a hypothesis-free analysis using raw data from the BBT count, GPT time (without time restriction), WMFT summed timed, WMFT grip strength, WMFT forearm lift, and F-M score. All tests were included to provide data from a comprehensive suite of UL assessments. An arbitrary score of 5000 was assigned to participants who were unable to complete the GPT in order to distinguish unsuccessful attempts from successful attempts. 19 Similarly, those unable to complete a WMFT timed task were assigned a score of 121 seconds. All raw data scores were standardized on a 0 to 1 scale. The resultant hypothesis-free clusters were compared to the 3-cluster hypothesis-based analyses to determine resilience of the stratification method.
5-Minute GPT Cutoff Time
The effect of a 5-minute GPT cutoff time on participant stratification was examined. In this set of hypothesis-based and hypothesis-free analyses, the GPT completion time was restricted to 5 minutes. Participants unable to place all 25 pegs in ≤5 minutes were considered to have not completed the test and were therefore given a raw score of 5000, and a dichotomous score of 0 for the GPT.
Redefined Classification for Low Motor Function
A final set of hypothesis-based and hypothesis-free analyses were performed where low motor function was defined as 0 blocks moved on the BBT as opposed to ≤1 block in the above analyses.
Results
All 96 participants completed the BBT, WMFT, and F-M. Only those with fine manual dexterity completed the GPT (n = 38), that is, placed all 25 pegs, while 26 participants were able to move >1 block in the BBT but were unable to place all 25 pegs. Both the GPT and BBT displayed floor effects, with 32 participants unable to move >1 block in the BBT, and 58 participants unable to complete the GPT (Figure 2). UL motor function was heterogeneous, with rank-ordered distributions of the timed WMFT and F-M showing a clear overlap between participants with moderate motor function and high motor function (Figure 3A and B). The timed WMFT tasks displayed floor effects, the WMFT forearm lift displayed both floor and ceiling effects, and the F-M displayed only ceiling effects. Six participants were unable to complete any of the 15 timed WMFT tasks within the given 120 seconds time limit (Figure 3A), and hence were assigned a score of 121 seconds per task. In contrast, the F-M displayed ceiling effects with 6 participants scoring the maximum of 66 across all 33 tasks and an additional 6 participants scoring 65. As shown in Figure 3B and D, this test was more sensitive to participants with low motor function, showing a greater distribution of scores.
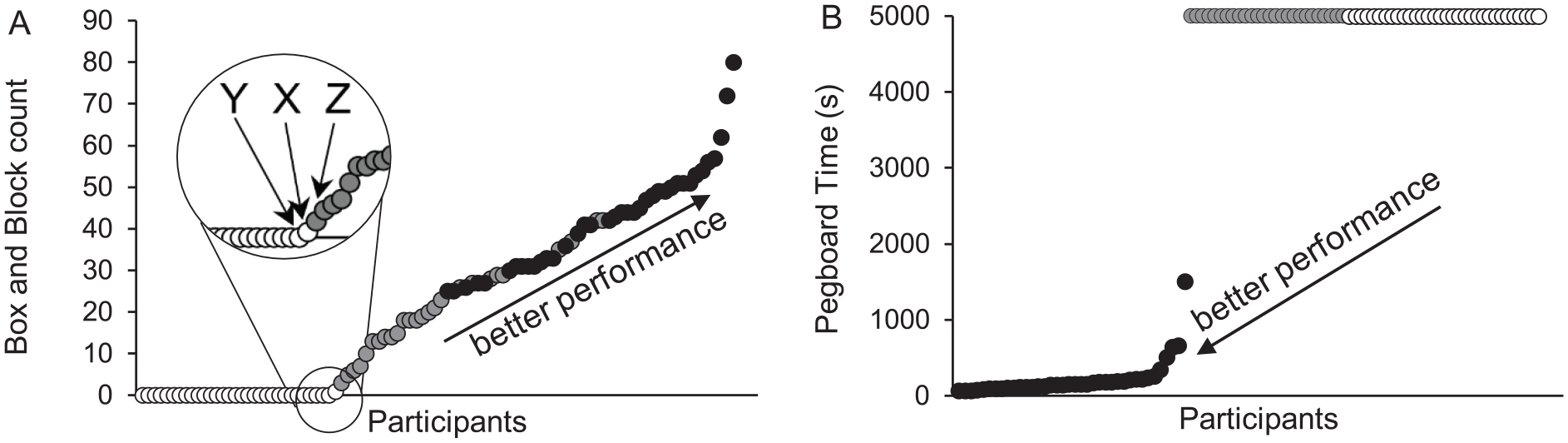
Rank-ordered BBT (Box and Block Test) scores and GPT (Grooved Pegboard Test) completion times. (A) The floor effects of the BBT distinguished between low (white circles) and moderate motor function (gray circles). Participants X, Y, and Z moved 1, 0, and 5 blocks on the BBT, respectively. (B) The floor effects of the GPT distinguished between moderate and high motor function (black circles).
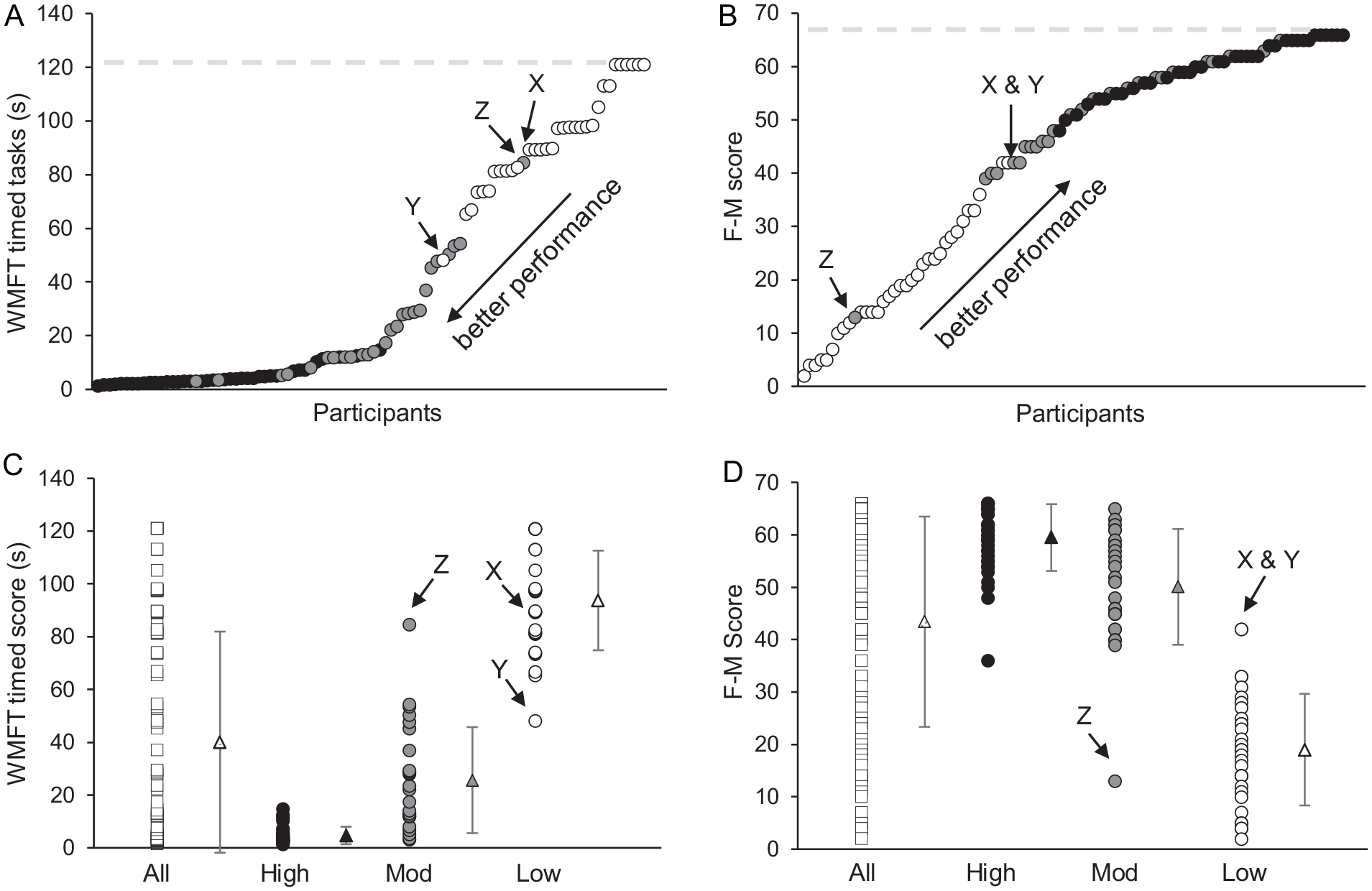
Timed WMFT (Wolf Motor Function Test) and F-M (Fugl-Meyer Assessment) results. Rank-ordered scores for (A) WMFT timed tasks and (B) F-M scores show a continuous distribution of scores with no clear distinction between low (white circles), moderate (gray circles), and high motor function (black circles). The maximum score for each test is represented by horizontal dashed lines. The mean, range, and standard deviation for (C) WMFT timed tasks and (D) F-M scores. Participants X, Y, and Z have outlying WMFT and F-M scores and were misclassified by the hypothesis-free hierarchical cluster analysis.
Hierarchical Cluster Analyses
The 3-cluster hypothesis-free analyses identified the most clinically relevant functional groupings for the unrestricted GPT completion time; the 5-minute GPT cutoff time; and the redefined classification for low motor function analyses. In brief, the 2-cluster analyses combined patients with low and moderate motor function, while the 4-cluster analyses subdivided the high motor function group into high and very high motor function. The very high motor function group included 2 participants who took >5 minutes on the GPT. The 5-cluster analyses subdivided moderate into lower moderate and higher moderate groups. A detailed breakdown of the 2-, 4-, and 5-cluster analyses can be found in the supplementary material.
Unrestricted GPT Completion Time
The hypothesis-based cluster analysis used the floor effects of the BBT to distinguish participants who moved ≤1 block (low motor function) from those who could move >1 block (moderate and high motor function; Figure 2A). The floor effects of the GPT grouped participants according to those who could not place all 25 pegs (low and moderate motor function) and those that could (high motor function; Figure 2B). When the BBT and GPT were analyzed together, the hypothesis based-cluster analyses identified 3 groups of motor function: low (n = 32), moderate (n = 26), and high (n = 38). The resultant 3 groups identified by the hypothesis-based hierarchical cluster analysis were compared with the 2, 3, 4, and 5 group clusters generated by the hypothesis-free hierarchical cluster analysis.
The 3, 4, and 5 hypothesis-free cluster analyses identified the same 3 levels of motor function (ie, low, moderate, and high) as identified by the hypothesis-based analysis. The 3-cluster hypothesis-free analysis allocated participants into the same 3 groups as the hypothesis-based analysis with 3 exceptions (Table 2).
Hypothesis-Free Hierarchical Cluster Analyses a .
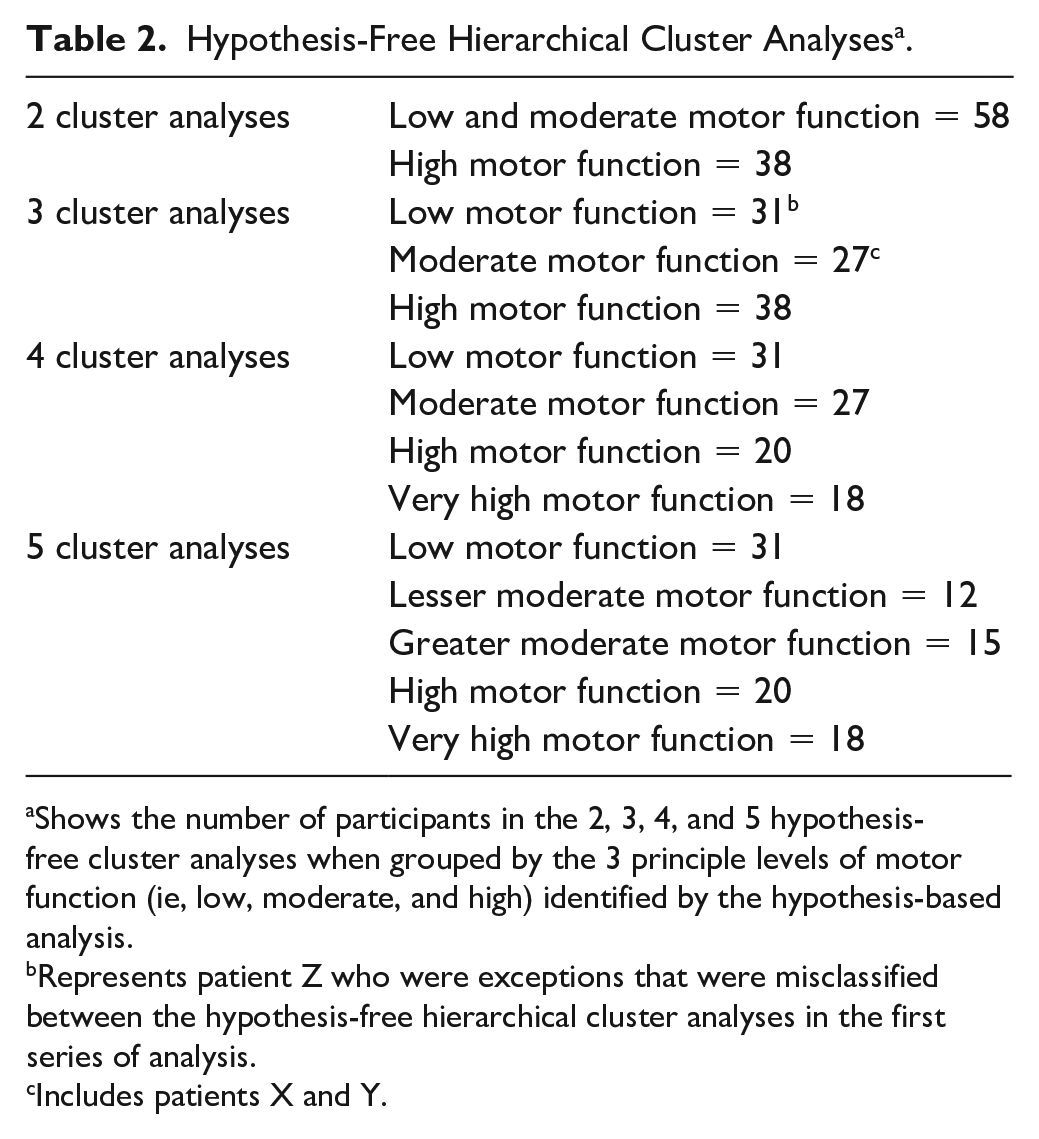
Shows the number of participants in the 2, 3, 4, and 5 hypothesis-free cluster analyses when grouped by the 3 principle levels of motor function (ie, low, moderate, and high) identified by the hypothesis-based analysis.
Represents patient Z who were exceptions that were misclassified between the hypothesis-free hierarchical cluster analyses in the first series of analysis.
Includes patients X and Y.
Grooved Pegboard Times
GPT completion times ranged from 1 to 25 minutes, with 87% of participants completing in ≤5 minutes. The remaining 13% (n = 5) of participants took 6 to 25 minutes (Figure 4), and clinical observations during assessment typically saw these participants using alternative movement strategies to complete the test, that is, manipulation of shoulder, body, or chair orientation. Many of these participants were only able to correctly place the peg if it was initially picked up in the correct orientation or by dragging it to the end of the table to aid in manipulation.
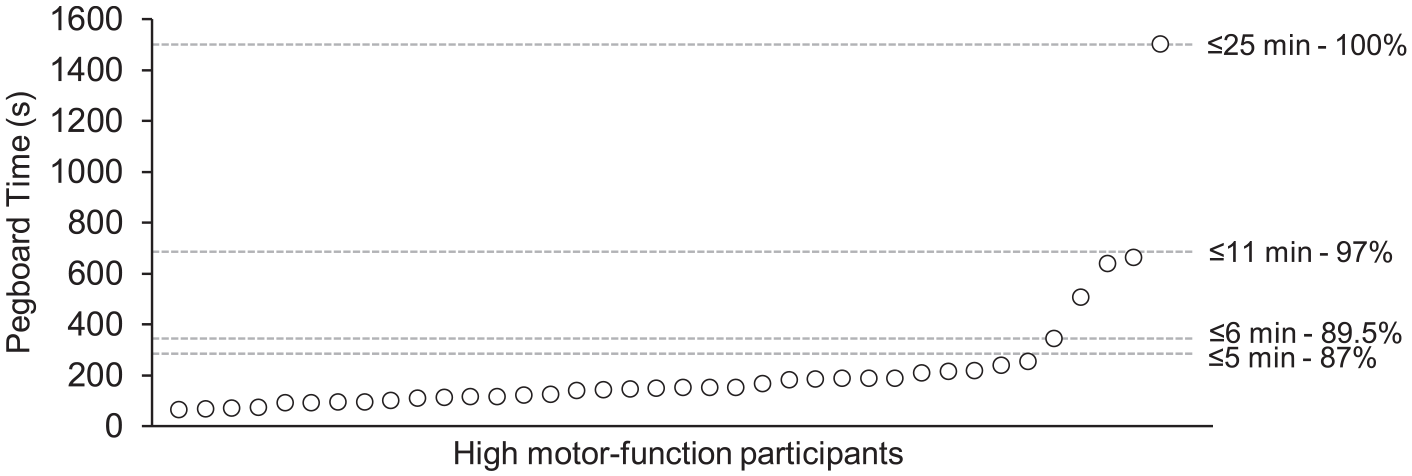
Rank-ordered GPT (Grooved Pegboard Test) times. The majority of participants with high motor function were able to complete the GPT within the 5-minute timeframe. Many of those participants that took >5 minutes used alternative movements (eg, change in body position) to complete the test.
5-Minute GPT Cutoff Time
To better understand the effects of the manufacturer’s maximal 5-minute GPT cutoff time limit on motor function stratification, we reran the hierarchical cluster analyses including the amended scores for the 5 participants who took >5 minutes to complete the GPT. The hypothesis-based cluster analysis was re-analyzed to create 3 clusters of motor function: low (n = 32), moderate (n = 31), and high (n = 33). Note that the main difference between these groups (5-minute GPT cutoff time) and the first hypothesis-based analysis (unrestricted GPT completion time) is that 5 participants who took >5 minutes were classified with moderate motor function, instead of high motor function. The hypothesis-free cluster analyses were also repeated with the amended scores to create 2, 3, 4, and 5 clusters. In each analysis the 5 participants with GPT scores >5 minutes were classified with moderate motor function. The 3-cluster analysis identified the same levels of motor function (ie, low, moderate, and high) as the hypothesis-based analysis with 6 exceptions. These 6 participants were classified as having moderate motor function in the hypothesis-based analysis but were classified as low motor function in the hypothesis-free analyses. Participant Z (with pain-avoidant behavior; see supplementary information) was one of these 6 participants. Group numbers for the hypothesis-free 3-cluster analysis were the following: low (n = 38), moderate (n = 25), and high (n = 33). When compared to the first hypothesis-free cluster analysis (unrestricted GPT completion time), the main changes are as follows: the 5 participants who took >5 minutes to complete the GPT have shifted from high to moderate motor function; 6 participants on the lower end of the spectrum of moderate motor function shifted into the low motor function group (including participant Z); and 2 participants (X and Y) did not shift into moderate motor function and remained classified as low motor function.
Redefined Classification for Low Motor Function
We performed one final set of hierarchical cluster analyses, defining low motor function as moving 0 blocks and compared the resultant hypothesis-free and hypothesis-based clusters. The 3-cluster hypothesis-free analysis allocated participants into the same 3 groups as the hypothesis-based analysis with only 2 exceptions, participants Y and Z. This final set of groupings were the most congruent with clinical observation.
Discussion
Unambiguous stratification of UL motor function is critical in poststroke rehabilitation as it can provide practitioners with objective assessments that support the implementation of appropriate and individualized rehabilitation strategies. This analysis assessed the resilience of our previous stratification method 19 with a larger cohort and examined the unrestricted completion time of the GPT for use in clinical practice. In separating 96 poststroke participants by UL motor function into low, moderate, and high motor function groups, the 3-cluster hypothesis-free hierarchical cluster analysis (with no GPT time restriction) matched the previous stratification method with only 3 exceptions. The results from this study build on our previous analysis 19 and suggests that when used in combination, 2 simple dexterity measures stratified participants equally as well as 6 different assessments covering multiple aspects of UL motor function (timed WMFT, WMFT grip strength, WMFT forearm lift, F-M, GPT, and BBT) that typically require 45 to 60 minutes to complete, depending on each participant’s level of motor function.
The 4 and 5 hypothesis-free clusters identified the same 3 principal levels of motor function as the 3-cluster hypothesis-based analysis with minor variations. The 4-cluster analysis identified an additional subcategory of very high motor function, and the 5-cluster analysis included a subdivision of the moderate group (separating those with lesser moderate and greater moderate motor function), although the clinical relevance (ie, findings that will have direct clinical implications and will inform and/or directly impact clinical practice) of such groups is questionable. For example, participants classified into the 3 clusters of motor function, that is, low, moderate, and high, would require distinctly different approaches in the delivery and content of rehabilitation would therefore have direct clinical implications for informing clinical practice, whereas further subdivisions of motor function may not be necessary to identify appropriate rehabilitation options. These results suggest that in the time-limited clinical setting, it would be of little value to conduct additional assessments (ie, the WMFT and F-M) with limited practical application given that moderate, high, and very high motor function participants receive similarly intensive and challenging rehabilitation (eg, Constraint-Induced Movement Therapy).12,27
The 2-cluster hypothesis-free analysis grouped participants with low and moderate motor function together, primarily reflecting an inability to complete the GPT. This grouping has little clinical relevance as participants with low motor function are usually treated differently to those with moderate and high motor function. The rehabilitation strategies for low motor function patients are often more passive, for example, functional electrical stimulation and mechanical assisted training, 28 whereas the majority of rehabilitation strategies, such as Constraint-Induced Movement Therapy and virtual reality, are usually targeted to groups with moderate to high motor function who have more residual voluntary function.12,28 Thus, the 3-cluster analysis is more relevant in clinical practice as it provides a clear delineation between levels of motor function promoting appropriately targeted rehabilitation without unnecessary levels of classification.12,16,29
The findings of this study corroborate previous literature showing similar floor and ceiling effects for the WMFT and F-M assessments and highlight current limitations of UL clinical assessment. 11 Despite the sensitivity of the F-M to quantify low motor function, 19 we found considerable overlap of scores for stroke survivors with moderate and high motor function (Figure 3B). This overlap was also seen in the timed WMFT (Figure 3A). Our method confirms the use of the BBT and GPT in combination to provide a more discrete, standardized, and objective method to stratify patients in a time efficient manner.30,31
Clinical observation during the assessments suggested reasons for the misclassification of 3 participants (X, Y, and Z). Participants X and Y were considered to demonstrate borderline function (ie, between low and moderate motor function) with participant X moving 1 block in the BBT (score of 1) and participant Y moving blocks using compensatory movement patterns that did not meet the movement requirements of the BBT, that is, participants must use only the thumb and index finger to move the 2.5 cm blocks, and without throwing the block. 19 In our previous analysis, 19 one borderline participant with minimal hand dexterity moved a single block, prompting the low motor function criterion to be defined as ≤1 block instead of 0 blocks as originally hypothesized. 19 However, the inclusion of additional low motor function participants in this analysis has shown greater distinction between scores for participants with low and moderate motor function, resulting in the classification of participants X and Y as moderate. The third set of hierarchical cluster analyses, where low motor function was defined as moving 0 blocks demonstrated the above findings, with only 2 misclassifications; participants Y and Z. This may also have occurred due to the inclusion of additional low motor function participants. This suggests that this stratification definition of 0 blocks moved on the BBT may be more appropriate for this group.
For such borderline cases, the F-M could be used to guide the rehabilitation strategy as it is more sensitive to participants with low motor function due to the greater weighting of gross motor tasks such as grasp and range of motion, while the WMFT displays floor effects, with many participants unable to complete any of the tasks. 19
For the purpose of stratifying poststroke UL motor function our results are not consistent with the GPT manufacturers guidelines of a 5-minute cutoff time. 32 While 87% of participants completed the GPT in <5 minutes, the remaining 13% took longer indicating greater functional ability (ie, fine dexterity) than those who could not complete the test. By suggesting these participants have little or no fine dexterity (moderate motor function), the amended hypothesis-free analysis (ie, 5-minute GPT cutoff time) shifted the functional profile of 5 additional moderate motor function participants into the low motor function group, which is not clinically appropriate. In addition, 2 of the participants who took >5 minutes to complete the GPT were identified as having a profile of very high motor function in the 4-cluster analyses (unrestricted GPT completion time and redefined classification of low motor function). This suggests that implementing the time restriction may incorrectly classify those with overall high motor function as well as underestimate the functional profile of some participants with moderate motor function. Therefore, a 5-minute cutoff time for the GPT is not recommended for stratification purposes.
Even without a 5-minute cutoff time for the GPT, the stratification method is quick and easy to administer, taking 3 to 11 minutes for both tests for the majority of participants; including 3 minutes for a bilateral BBT, but longer if the GPT is attempted. This suggests good clinical value for the majority of participants given clinical appointments in Australia 33 and elsewhere are often time limited. 34 The stratification method could further be condensed by only testing the more affected side. However, performing the full stratification method bilaterally would yield additional dexterity information, providing an index of age-appropriate motor function 35 and reveal any subtle ipsilateral impairments that may be present. 36
A clinical observation during assessment of the GPT was the use of alternative movement strategies to complete the test. Participants are expected to use only their UL to complete the task to sequentially pick up, orientate, and place the pegs. 32 In this study, some participants who required >5 minutes also used trunk movements to complete the task. This may represent a compensatory motor strategy employed by participants with moderate to high motor function who possess gross motor function but fewer fine motor skills. The GPT may benefit from specific instructions to minimize the use of alternative methods, as none are currently provided in the GPT manual. Assessor training should be implemented to specify unacceptable strategies, such as not placing a peg on the table to grasp in a better position, and to identify the use of alternative methods.
Limitations and Future Directions
This study builds on the analysis of previous work 19 investigating UL stratification, and while the present study includes 67 of the original participants it examines a number of new and clinically relevant concepts. First, the addition of 29 new participants provided a greater distribution of BBT scores, especially between 1 and 10 blocks (Figure 5). This filled a data gap in the previous study 19 and led to a redefinition of the stratification criteria, that is, low motor function defined as moving 0 blocks in the BBT, instead of ≤1. In addition, the current study examines the utility of the 5-minute GPT cutoff time and suggests the potential need for stricter guidelines for peg pick up, orientation, and placement technique, as monitoring the quality of movement, that is, use of alternative movement strategies, is not part of the GPT testing requirements.
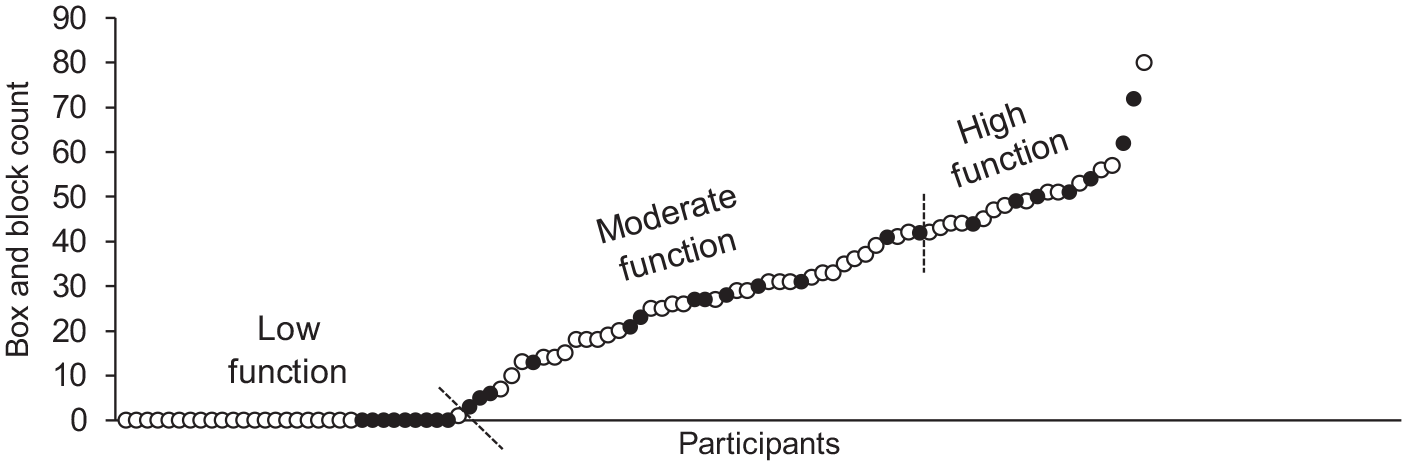
Rank-ordered BBT (Box and Block Test) scores. The rank ordered distribution of BBT scores for the original 67 participants (white circles) were limited between 1 and 10 blocks. The more comprehensive distribution from the additional 29 participants (black circles) in the current analysis provides a clearer distinction between low and moderate motor function. Dotted lines separate low from moderate motor function, and moderate from high motor function.
We acknowledge that the stratification method may misclassify those who are on the borderline of motor function levels. However, both our previous 19 and present analyses suggest that the likelihood of this occurring is relatively small. It is important to note that these borderline cases are identifiable during testing where participants may show gross motor ability (eg, picking up the blocks using compensatory movement patterns) but do not meet the criteria for the test (ie, picking up the blocks using only the thumb and index finger). In such cases clinicians can use the F-M to obtain additional information to inform their rehabilitation strategy. Furthermore, the stratification method may be limited when assessing participants with pain-avoidant behavior (see supplementary information). Also, this study did not accommodate cognitive deficits and/or severe visual impairments and is not generalizable to acute or subacute stroke survivors. Alternative stratification strategies should be explored for use with these patient populations.
The 5-minute GPT cutoff time was not appropriate for stratification purposes. Standardized GPT rules for peg pick up, orientation, and placement should be investigated further to clarify accurate completion of the test.
Since the data collection for this study was completed, a consensus-based recommendation for use of the Action Research Arm Test in stroke trials has been published by the Stroke Recovery and Rehabilitation Roundtable. 37 Future research examining its use and interplay with the assessments used in this study is warranted.
Conclusion
This proposed stratification method was able to unambiguously stratify stroke survivors with low, moderate, and high motor function. Based on the present results, a BBT score of 0 can be used to denote low motor function; a BBT score of ≥1 but incomplete GPT denotes moderate motor function; and a BBT score of ≥1 and complete GPT denotes high motor function. While 87% of participants completed the GPT in 5 minutes, the use of a 5-minute cutoff time is not recommended for stratification purposes. Despite this, the stratification method is a time-efficient assessment tool for the majority of participants that provides an objective stratification in clinical practice where time is restricted.
Supplemental Material
sj-docx-1-nnr-10.1177_1545968321992048 – Supplemental material for Revisiting Poststroke Upper Limb Stratification: Resilience in a Larger Cohort
Supplemental material, sj-docx-1-nnr-10.1177_1545968321992048 for Revisiting Poststroke Upper Limb Stratification: Resilience in a Larger Cohort by Benjamin J. Varley, Christine T. Shiner, Liam Johnson, Penelope A. McNulty and Angelica G. Thompson-Butel in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We gratefully acknowledge the contributions of Gavin Lin and Terry Trinh, who assisted in the data collection; Akbar Yasin, who assisted in preliminary data analysis; and Michael Steele for his statistical advice.
Author Contributions
Study conception and design: PAMcN, AGTB
Data acquisition: CTS, AGTB
Data analysis: BJV, AGTB
Interpretation of data: BJV, CTS, LJ, PAMcN, AGTB
Drafting of manuscript: BJV, CTS, LJ, AGTB
Critical revision and approval of final manuscript: BJV, CTS, LJ, PAMcN, AGTB
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by 3 grants: the National Health and Medical Research Council of Australia (APP630440) and the New South Wales Office of Science and Medical Research, Australia, and a clinical stipend from the National Health and Medical Research Council, Centre of Research Excellence in Stroke Rehabilitation and Brain Recovery (APP1077898).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
