Abstract
Objective
Patients show substantial differences in response to rehabilitation therapy after stroke. We hypothesized that specific genetic profiles might explain some of this variance and, secondarily, that genetic factors are related to cerebral atrophy post-stroke.
Methods
The phase 3 ICARE study examined response to motor rehabilitation therapies. In 216 ICARE enrollees, DNA was analyzed for presence of the BDNF val66met and the ApoE ε4 polymorphism. The relationship of polymorphism status to 12-month change in motor status (Wolf Motor Function Test, WMFT) was examined. Neuroimaging data were also evaluated (n=127).
Results
Subjects were 61±13 years old (mean±SD) and enrolled 43±22 days post-stroke; 19.7% were BDNF val66met carriers and 29.8% ApoE ε4 carriers. Carrier status for each polymorphism was not associated with WMFT, either at baseline or over 12 months of follow-up. Neuroimaging, acquired 5±11 days post-stroke, showed that BDNF val66met polymorphism carriers had a 1.34-greater degree of cerebral atrophy compared to non-carriers (P=.01). Post hoc analysis found that age of stroke onset was 4.6 years younger in subjects with the ApoE ε4 polymorphism (P=.02).
Conclusion
Neither the val66met BDNF nor ApoE ε4 polymorphism explained inter-subject differences in response to rehabilitation therapy. The BDNF val66met polymorphism was associated with cerebral atrophy at baseline, echoing findings in healthy subjects, and suggesting an endophenotype. The ApoE ε4 polymorphism was associated with younger age at stroke onset, echoing findings in Alzheimer’s disease and suggesting a common biology. Genetic associations provide insights useful to understanding the biology of outcomes after stroke.
Introduction
Patients receiving motor therapy during outpatient rehabilitation show substantial variability in treatment response. These high rates of inter-subject variability complicate prescription of therapy in clinical practice and reduce statistical power in clinical trials. Behavioral and neuroimaging measures provide insights into treatment response to rehabilitation therapy after stroke but do not completely explain differences in treatment response.1,2 Measures of genetic variability might further explain inter-subject differences in response to rehabilitation therapy.
Behavioral gains resulting from rehabilitation therapy occur because of brain plasticity, which is related to changes in gene expression. 3 Though numerous genes support neural repair and plasticity, two may be particularly important: brain derived neurotrophic factor (BDNF) and ApolipoproteinE (ApoE), each of which has a polymorphism with effects on neural function.
These findings suggest the hypothesis that these BDNF and ApoE genetic variants predict response to motor rehabilitation therapy after stroke. This hypothesis was tested in the context of the Interdisciplinary Comprehensive Arm Rehabilitation Evaluation (ICARE) study, a phase 3 randomized trial of patients with moderate motor impairment due to stroke. 4 Patients were randomized to task-oriented upper extremity training, dose-equivalent occupational therapy, or standard of care. The primary outcome measure was the 12-month change in Wolf Motor Function Test (WMFT), a measure of arm motor function.
The primary hypothesis of the current study was that the val66met BDNF polymorphism and the ApoE ε4 polymorphism are significantly associated with poorer response to motor rehabilitation therapy over 12 months among ICARE study enrollees. Two secondary hypotheses were examined. First, the primary hypothesis was further examined with respect to treatment group, hypothesizing that the most intensive ICARE intervention would show the largest polymorphism-related difference. Second, both the BDNF 5 and the ApoE ε4 polymorphism 6 have each been associated with regional and global forms of brain atrophy in healthy subjects; however, data are scant in patients with stroke. This motivated the additional hypothesis that polymorphism-related differences in brain atrophy are present on brain images acquired acutely post-stroke. Together, these hypotheses aim to understand the biological impact of these two common genetic polymorphisms for patients in the context of motor rehabilitation after stroke.
Methods
Overview
Subjects enrolled in the ICARE study (clinicaltrials.gov ID NCT00871715) were offered participation in the current genetics substudy. This study was approved by the UC Irvine IRB. All subjects signed a separate consent in addition to consent related to participating in the parent ICARE study. ICARE 4 was a phase 3, pragmatic, single-blind trial that enrolled 361 patients with stroke from 7 US sites. Patients were randomized to 30 h of task-oriented upper extremity training (Accelerated Skill Acquisition Program), 30 h of dose-equivalent occupational therapy, or standard of care. The primary outcome measure was the 12-month change in log-transformed WMFT time, a measure of arm function (activities limitation) consisting of 15 timed arm movements and hand dexterity tasks. The ICARE study found no differences between treatment groups in the primary endpoint, 4 and so the current study combined subject groups in primary analysis. Sample size in the current substudy was derived from the parent study. 4
Entry criteria for the ICARE study included ischemic or hemorrhagic stroke in the past 14–106 days causing upper extremity hemiparesis; age ≥21; and absence of substantial ataxia, sensory deficits, neglect, and pre-stroke disability. After consent was signed for this substudy, buccal swabs were obtained.
Buccal Swab Sampling and DNA Testing
Two buccal swab samples were obtained. Subjects had no food or beverage for >15 min then rinsed his/her mouth with water several times, after which a single cytobrush was twirled firmly along the buccal mucosa for 30 s; this was then repeated. Swabs were shipped to UC Irvine for genotyping.
BDNF and ApoE Genotyping
Genomic DNA was isolated from cheek cells collected via the cytobrushes, using Gentra Puregene standard buccal cell protocol (Qiagen). DNA concentration and purity were determined using the GE Nanovue Spectrophotometer. Genotyping reactions were run on Applied Biosystem 7900 Fast Real-Time PCR System and scored using the algorithm and software supplied by the manufacturer (Applied Biosystems). As quality control during genotyping, six known controls (2 of each genotype) and 3 no template controls were run on each plate along the unknown samples. If insufficient DNA was extracted from a given sample, the enrollment site was notified and asked to obtain a repeat specimen; this occurred in 12 instances, and for each a repeat was obtained at the next scheduled ICARE study visit.
BDNF alleles were determined using Applied Biosystems TaqMan SNP Genotyping Assay c_11592758_10 (Applied Biosystems). The BDNF val66met SNP (rs6265) consists of a nucleotide change that results in the amino acid change from valine to methionine at codon 66. In the current study, patients were classified as BDNF val66met polymorphism carriers (on one or both alleles) or not.
ApoE alleles were determined by interrogating bases at two codons, 112 Cys/Arg (rs429358), and 158 Arg/Cys (rs7412), using two separate assays: TaqMan Assays C_3084793_20 and C_904973_10, respectively. In the current study, patients were classified as ApoE ε4 polymorphism carriers (on one or both alleles) or not.
Imaging Analysis
Images were acquired as part of standard clinical care. Cerebral atrophy was measured using the ventricle-brain ratio (VBR), which has been validated in studies of mild cognitive impairment7,8 and dementia8,9 and is sensitive to neuropsychological performance.10,11 VBR was measured in the cerebrum and calculated by extracting the volume of the lateral ventricles and the brain (limited to the cerebrum), then taking the ratio. When an MRI was available, ventricular volume was measured using ALVIN 12 and then manually refined, and brain volume was manually segmented using MRICron. When an MRI was not available but a CT was, the skull was stripped using SPM, then ventricular volume and brain volume were manually segmented using MRICron. All VBR measurements were verified by a neuroimaging expert (BL).
Data Analysis
Parametric statistical methods were employed, using raw or transformed values. ANCOVA models were used to test whether carriers of the BDNF val66met polymorphism or ApoE ε4 polymorphism differed in relation to the ICARE primary endpoint, 12-month change in log WMFT time. Corollary analysis used baseline WMFT score as the dependent measure. Analyses were then repeated, according to treatment group.
Analysis of imaging data examined whether carriers of the BDNF val66met or the ApoE ε4 polymorphism had greater brain atrophy, measured as VBR and analyzed using the ln transform; secondary measures were the two VBR subcomponents, ventricular volume and brain volume. Imaging data from ICARE enrollees were compared with findings from a cohort of age-matched healthy controls whose images were available from prior studies.13,14
The covariates of interest were those pre-specified in the ICARE trial 4 : randomization site, number of days post-stroke at time of study enrollment, and arm motor impairment (arm motor Fugl-Meyer (FM) score). Markov chain Monte Carlo imputation was used to account for missing data. 4
Results
Subjects
Of the 361 subjects in ICARE, 216 were also enrolled in the current genetics substudy. This difference in number of subjects was due in part to variable time for site initiation of this substudy, for example, one site did not enroll into this genetics substudy until ICARE patient # 108. The 216 subjects in this genetics substudy, vs. the other 145 not studied genetically, did not differ in race, sex, or age, although genetics substudy enrollees had slightly milder strokes (FM score 42.5±9.5 vs. 40.3±.0, P=.02, mean±SD) and earlier times to randomization (43.7±22.0 vs. 48.8±22.6 days post-stroke, P=.01). Genotype could not be determined for BDNF in 7, for ApoE in 2, and for either polymorphism in 1 patient, leaving 206 subjects with complete genetics data.
Baseline characteristics of subjects with stroke.
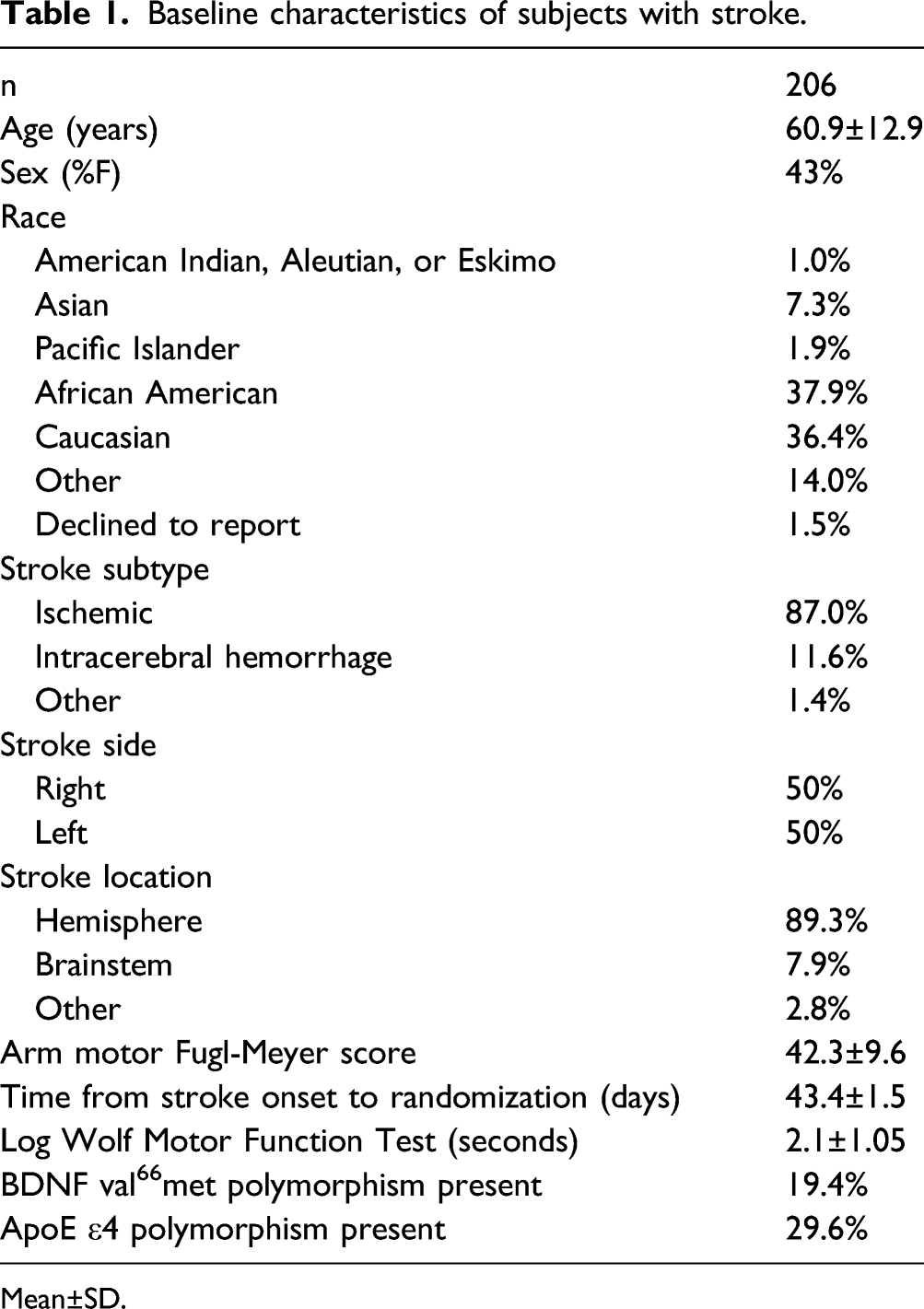
Mean±SD.
Genetic Factors in Relation to Behavioral Outcomes
Correlates of motor status.
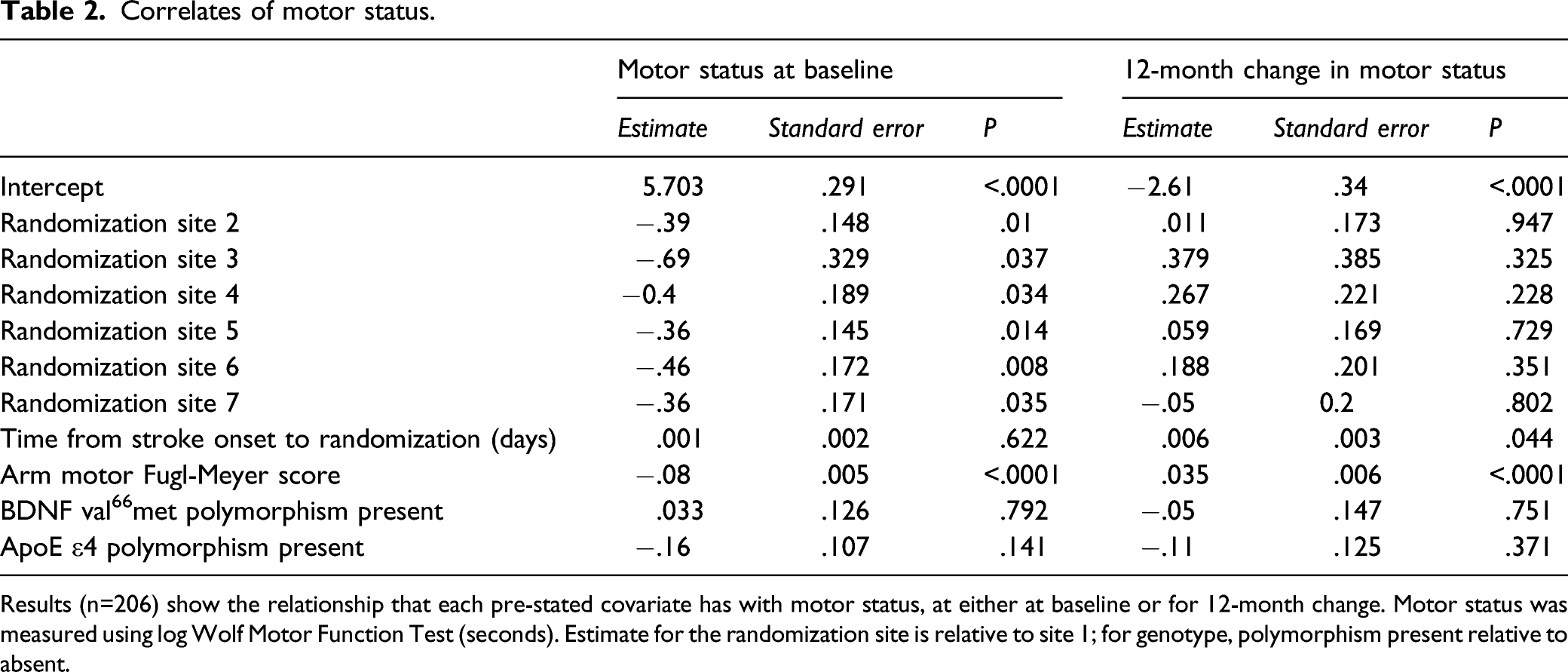
Results (n=206) show the relationship that each pre-stated covariate has with motor status, at either at baseline or for 12-month change. Motor status was measured using log Wolf Motor Function Test (seconds). Estimate for the randomization site is relative to site 1; for genotype, polymorphism present relative to absent.
Correlates of 12-month change in motor status as a function of treatment group.
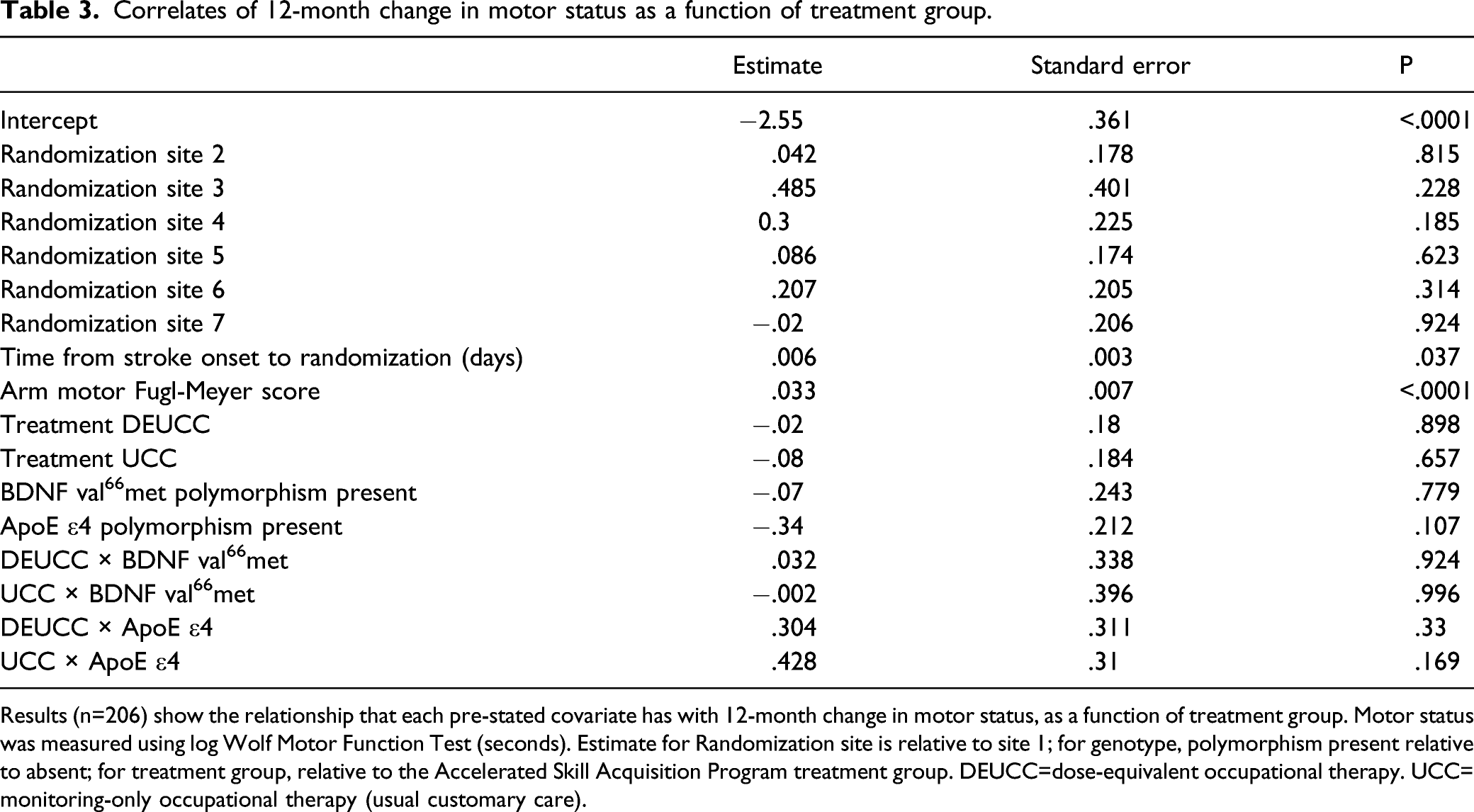
Results (n=206) show the relationship that each pre-stated covariate has with 12-month change in motor status, as a function of treatment group. Motor status was measured using log Wolf Motor Function Test (seconds). Estimate for Randomization site is relative to site 1; for genotype, polymorphism present relative to absent; for treatment group, relative to the Accelerated Skill Acquisition Program treatment group. DEUCC=dose-equivalent occupational therapy. UCC= monitoring-only occupational therapy (usual customary care).
Genetic Factors in Relation to Neuroimaging Measures
Neuroimaging data were available in 127 of the subjects enrolled in this genetic substudy. These 127 subjects, vs. the 234 without neuroimaging, did not significantly differ in age, days post-stroke when enrolled, baseline FM score, or 12-month change in WMFT (P>.4). Imaging consisted of a head CT in 66 and a brain MRI in 61, subjects. Scans were acquired 5±11 days post-stroke, which did not vary according to genotype. Mean VBR did not differ between those studied using CT vs. MRI (.027 vs. .027, P=.49). VBR did not correlate with log WMFT at baseline (P=.50) or its 12-month change (P=.33).
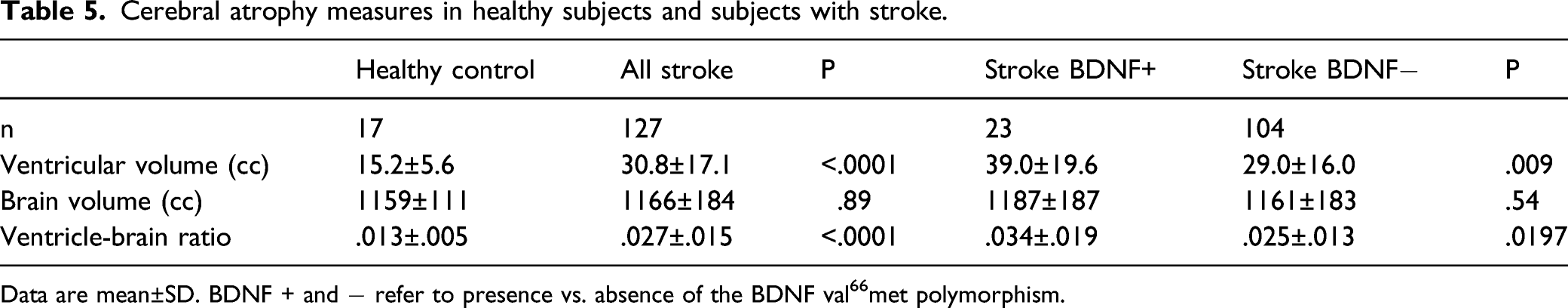
Presence of the BDNF val66met polymorphism was associated with significantly greater cerebral atrophy (P=.01), as carriers had a 1.34-greater VBR in covariate-adjusted analyses (Figure 1, Table 4). Presence of the ApoE ε4 polymorphism was not associated with a significant difference in VBR (P=.53). The association of the BDNF val66met polymorphism with VBR was driven by ventricular enlargement, as ventricular volume varied significantly (P=.0098) with BDNF val66met polymorphism status, while brain volume did not (P=.74). This finding among patients with stroke was further explored by comparing results with 17 age-matched (60.7±12.4 vs. 60.8±5.7 years, P=.98) healthy control subjects. VBR was 2.02-fold greater in subjects with stroke as compared to healthy controls (P<.0001; Table 5), and this too was driven by differences in ventricular volume rather than brain volume. Brain atrophy varied in relation to the BDNF val66met polymorphism. Among patients with stroke, presence of the BDNF val66met polymorphism was associated with significantly larger degree of cerebral atrophy (mean±SD), measured as the ventricle-brain ratio (VBR); larger VBR values indicate greater atrophy. These atrophy findings were driven by differences in ventricular volume. Presence of the ApoE ε4 polymorphism was not associated with a significant difference in cerebral atrophy. * P=.01. Correlates of cerebral atrophy. Results are for 127 subjects, adjusted for covariates. Cerebral atrophy was measured as the VBR (ventricle-brain ratio). Estimate for Randomization site is relative to site 1; for genotype, polymorphism present relative to absent. VBR was ln-transformed when used as a dependent measure, and so the estimate for the BDNF val66met polymorphism indicates that subjects with this genotype had a 1.34-greater VBR in covariate-adjusted analyses. Cerebral atrophy measures in healthy subjects and subjects with stroke. Data are mean±SD. BDNF + and − refer to presence vs. absence of the BDNF val66met polymorphism.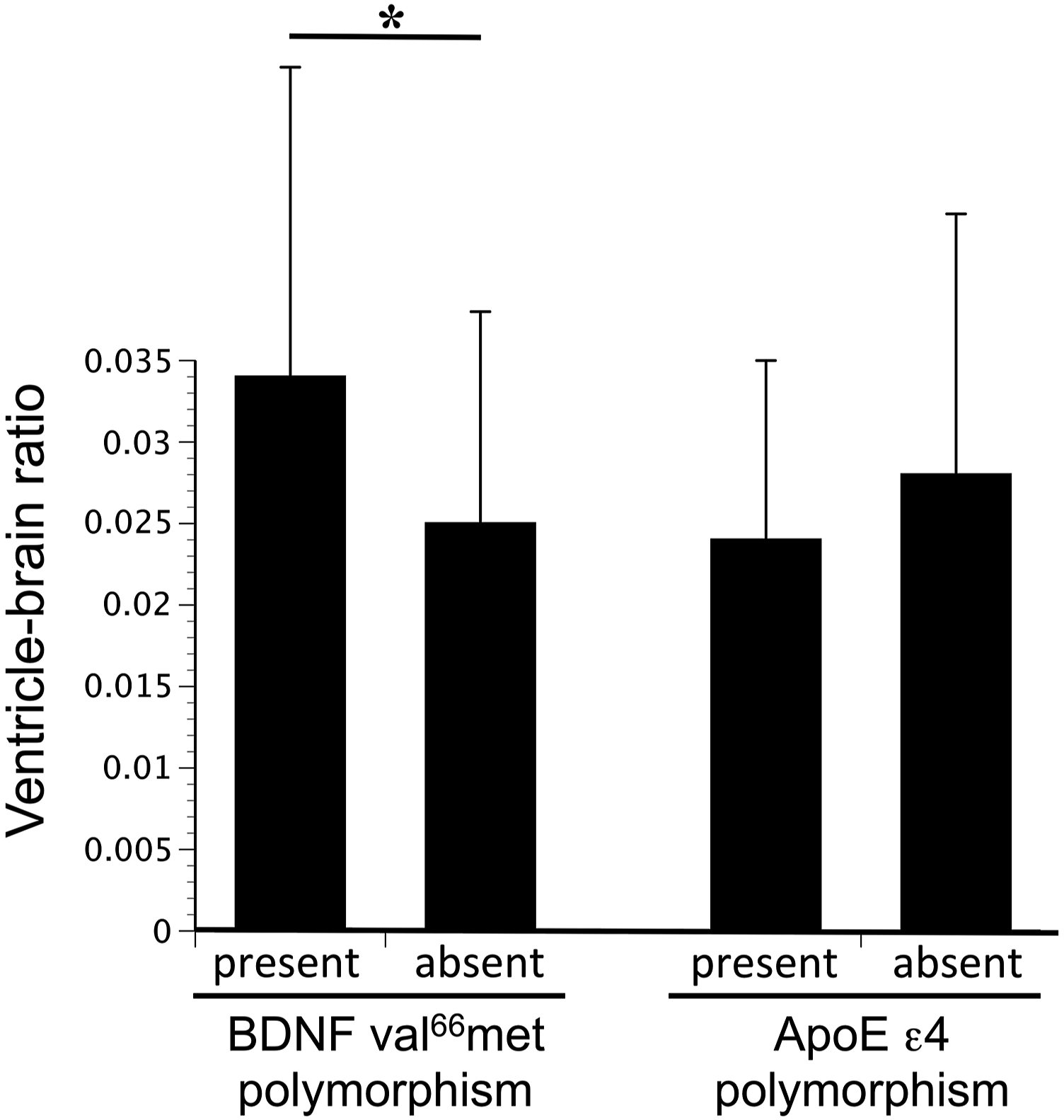
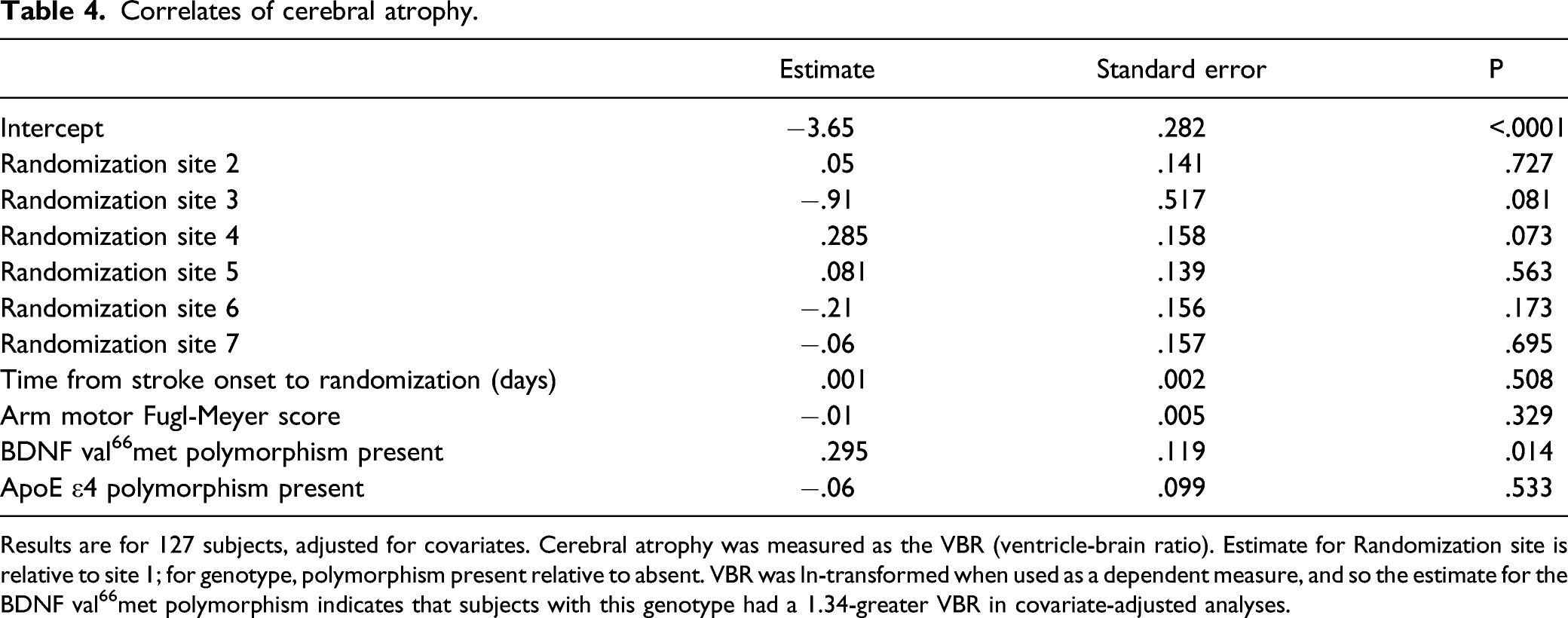
The relationship identified between cerebral atrophy and the BDNF val66met polymorphism was further examined using an additional covariate. Primary analysis, above, used the pre-stated covariates of interest specified by the ICARE study 4 (site, days post-stroke at enrollment, and baseline FM score), and so did not include age. Because brain atrophy is known to vary with age, this measure was added to the original model in a post hoc analysis: while age was significantly related to VBR (P=.007), BDNF val66met polymorphism status nonetheless remained significantly associated with VBR after adjusting for age (P=.04).
Discussion
Patients show substantial variability in response to rehabilitation therapy after stroke. Genetic factors might be important determinants. In the context of the ICARE study, 4 the current study examined two candidates, the BDNF val66met polymorphism and the ApoE ε4 polymorphism. Neither polymorphism was associated with a difference in recovery of arm function (WMFT) over 12 months of follow-up. Carrier status for the BDNF val66met polymorphism was associated with 1.34-greater brain atrophy (P=.01) at baseline, and carriers of the ApoE ε4 polymorphism were 4.6 years younger than non-carriers at time of stroke onset (P=.02). While genetic measures were not related to response to motor rehabilitation, as hypothesized, they provide insights useful to understanding outcomes after stroke.
There may be specific genetic profiles that are associated with diminished capacity for neural plasticity after stroke. BDNF is a widely expressed neurotrophin in the CNS and plays a key role in synaptic plasticity. The val66met polymorphism in the gene for BDNF is associated with reduced activity-dependent BDNF secretion, 16 reduced short-term plasticity in the human motor system, 17 and impaired motor skill acquisition, 18 suggesting a negative influence on motor recovery after stroke. However, current data did not support that this polymorphism is associated with poorer response to motor rehabilitation therapy post-stroke (12-month WMFT change). Prior studies have been mixed. This polymorphism has been associated with poorer outcome in several prior studies (at 1 month, 3 months, and 1 year 19 ) and in a meta-analysis, 20 with better outcome (at rehabilitation ward discharge), 21 or with no relationship to outcome (at 30 days and 3 months 22 ) after stroke. The polymorphism has also been associated with poorer response to subacute rehabilitation therapy 23 or chronic language therapy, 24 and with no relationship to response to subacute language 25 or chronic motor 26 therapy. Although knock-in mice expressing the human val66met polymorphism have greater motor deficits acutely after stroke, 27 they also have been reported to show better motor recovery28,29, suggesting that stroke recovery mechanisms may differ according to polymorphism status.29,30 Current results do not support a relationship between this BDNF polymorphism and response to motor rehabilitation therapy in the subacute period post-stroke.
ApoE is an abundant brain lipoprotein. The ApoE ε4 allele is associated with impairment of some neural repair processes31,32 and poorer microvascular flow during hypoperfusion. 33 ApoE ε4 is associated with poorer outcome after hemorrhagic stroke34,35 and traumatic brain injury. 36 However, most studies of patients with ischemic stroke have not found an association with spontaneous recovery after stroke: one study reported that the ApoE ε4 (+) allele was associated with poorer outcome (1-month change in NIHSS and 3-month mRS 37 ), but most have not seen an association with outcome (at 3 months, 6 months, and 1 year 38 ). The current study extends this body of literature, as here the primary outcome was domain-specific (arm motor function) rather than a global measure (NIHSS or mRS), the focus was on response to rehabilitation therapy rather than spontaneous stroke recovery, and results were collected in the context of a phase 3 stroke rehabilitation trial.
Several reasons might explain the observed lack of association between these two polymorphisms and response to rehabilitation therapy. These polymorphisms might simply have no effect on the processes underlying motor recovery from 6 weeks to 1 year post-stroke--motor system plasticity is highest in the initial weeks post-stroke. Findings might have differed if another primary outcome measure was used. Effects of the val66met BDNF polymorphism on motor system plasticity can be overcome with extended practice 39 : the polymorphism is associated with reduced short-term plasticity in motor cortex, 17 but these effects are overcome by 12 days of intense motor training. 40 Consistent with this, the polymorphism was associated with poorer recovery of global impairment 37 and dysphagia 41 at 1 month but not 3 months post-stroke. Effects of the ApoE ε4 polymorphism might be larger on cognitive rather than motor endpoints. Alternatively, ApoE ε4 effects might be more pronounced in patients with more severe forms of stroke 37 compared to ICARE enrollees.
Presence of the ApoE ε4 polymorphism was associated with younger age of stroke, by 4.6 years. This finding is consistent with a meta-analysis that found ApoE ε4 polymorphism to be associated with younger age at ischemic stroke onset. 42 The biology underlying this association in patients with stroke is uncertain, but might overlap with effects of this polymorphism in the setting of Alzheimer’s disease, where a single ApoE ε4 allele is associated with dementia age of onset that is 3.1 43 –8.8 44 years younger.
Imaging genetics studies can be more sensitive than gene-behavior studies. 45 Both the ApoE ε4 and val66met polymorphisms have been associated with brain atrophy, across many clinical settings, however their association in patients with stroke has received limited study. For example, knock-in mice expressing the human val66met polymorphism have significantly smaller hippocampal volumes. 46 Similarly, healthy human subjects with the val66met BDNF polymorphism have smaller volumes in numerous brain regions, including hippocampus, dorsolateral prefrontal cortex, temporal and occipital cortex, and frontal and parietal white matter. 5 A global effect on brain size has also been described. 5 Here, BDNF val66met polymorphism carriers showed a 1.34-greater degree of cerebral atrophy at baseline (Table 4), driven by ventricular enlargement. The lateral ventricles occupy a much smaller space than the parenchyma of the cerebrum, and therefore changes in ventricular volume may be a more sensitive sign of cerebral atrophy due to loss of tissue and ex vacuo dilatation. This relationship was substantial, being one-third of the effect that stroke status had compared to healthy controls, although it must be noted that genotype data were not available for control subjects. Scan acquisition times averaged 5 days post-stroke, possibly underestimating cerebral atrophy: findings were driven largely by pre-stroke atrophy, but any acute post-stroke brain swelling might have actually reduced the observed ventricular volume. This polymorphism was associated with cerebral atrophy but not poorer behavioral recovery; this constellation of findings might be a consequence of comparing a global imaging measure (brain atrophy) with a systems-based behavioral measure (motor status). Alternatively, this might suggest that cerebral atrophy is an intrinsic property of the met allele and thus can serve as an endophenotype of val66met BDNF polymorphism effects in the stroke population, as has been proposed for this polymorphism in relation to measures of motor system activation after stroke. 26
The ApoE ε4 polymorphism has often been associated with atrophy of various temporal lobe regions 6 and other gray matter areas,6,47 and globally (ventricular volume). 48 Here, an association with VBR following stroke was not observed for this polymorphism, possibly because ApoE ε4 effects may be strongest in subjects with dementia, or in specific brain areas such as hippocampus. In addition, ApoE ε4 effects on brain atrophy are most pronounced when measured as change in brain volume over time, 49 but the current study only had access to images from a single timepoint.
This study had several strengths, including the sample size, availability of serial behavioral measures, a novel focus on response to rehabilitation therapy, and data collection in the context of a phase 3 trial. There were also important weaknesses. Entry criteria focused on specific times post-stroke and a specific level of motor deficit severity, factors that might have biased results and that might limit the extent to which results generalize to the broader stroke population. Neuroimaging consisted of MRI for some subjects and CT for others, and was not available in everyone. Use of VBR to measure cerebral atrophy has limitations, for example, regional measures of brain atrophy were not available. Images were acquired five days post-stroke and so acute stroke events might have affected measurement of cerebral atrophy. Because the three treatment groups in the ICARE study did not differ in outcomes, the current study might reflect spontaneous recovery rather than response to motor rehabilitation therapy.
Conclusion
This study does not support that either the val66met BDNF polymorphism or the ApoE ε4 polymorphism explains inter-subject differences in response to rehabilitation therapy targeting arm motor function after stroke. Presence of the BDNF val66met polymorphism was associated with a significantly greater degree of cerebral atrophy, echoing findings in healthy subjects, and suggesting a potential imaging endophenotype of this polymorphism’s effects. The ApoE ε4 polymorphism was associated with younger age at stroke onset, directly matching findings in Alzheimer’s disease and so suggesting that the biology of this polymorphism might be shared across conditions. Although neither polymorphism was related to response to rehabilitation therapy, findings do provide insights useful to understanding outcomes after stroke.
Footnotes
Acknowledgments
We thank Cherryl Nugas for her technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SC serves as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, SanBio, Fujifilm Toyama Chemical Co., NeuExcell, Elevian, Medtronic, and TRCare. CW serves as a consultant for Enspire DBS Therapy, Inc, and receives royalty payments from Human Kinetics, Inc (for 6th edition of Motor Control and Learning), and DemosMedical Publishers (for 2nd edition of Stroke Recovery and Rehabilitation).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This study was supported by grants R01 NS058755, K24 HD074722, UL1 TR001414, and U01NS056256 (NIH).
