Abstract
Background. The precise mechanism of 2-channel neuromuscular electrical stimulation (NMES) treatment is unknown, and controversy remains over its efficacy. The sequential 4-channel NMES was newly developed based on normal contractile sequences of swallowing-related muscles. Objective. To assess the clinical efficacy of sequential 4-channel NMES during swallowing. Methods. In this prospective RCT, 52 inpatients with dysphagia (acute, subacute, and chronic state) after stroke, brain tumor, or encephalitis were enrolled. Participants who underwent a videofluoroscopic swallowing study (VFSS) and clinical evaluation were enrolled and were randomly assigned to the 4-channel NMES or sham group. The 4-channel NMES and sham groups swallowed thin and honey-like fluids under NMES (sequential stimulation on suprahyoid and infrahyoid) and sham stimulation, respectively. The procedures were evaluated with the VFSS. Pre- and post-treatment evaluations were performed with the videofluoroscopic dysphagia scale (VDS), penetration–aspiration scale (PAS), Likert scale, and kinematic analysis. Results. The 4-channel NMES group showed significantly greater improvements than the sham group with respect to oral VDS, pharyngeal VDS, total VDS, and PAS (P < .05). Furthermore, the Likert scale for satisfaction, easiness, and discomfort for swallowing showed favorable results for the 4-channel NMES group (P < .05). In the kinematic analysis, the peak speed point, distance, and velocity of hyoid movement were significantly greater in the 4-channel NMES group (P < .05). Conclusions. Sequential 4-channel NMES activating the suprahyoid, thyrohyoid, and other infrahyoid muscles during swallowing showed significant clinical improvement with respect to VDS, PAS, and kinematic analysis. Therefore, sequential 4-channel NMES is a potential new functional electrical stimulation system for the treatment of dysphagia.
Introduction
Dysphagia is a common and serious problem in patients with neurologic diseases and has a prevalence of 37–78%.1-3 Decreased laryngeal elevation caused by weakness in pharyngeal muscles is a main cause of dysphagia, which can result in aspiration and pharyngeal residue during swallowing. 4
Till now, diverse treatments including modifying food and fluid, altering posture and changing swallowing strategies with some rehabilitative techniques, and 2-channel neuromuscular electrical stimulation (NMES) have been used for the treatment of dysphagia. Especially in acute poststroke patients with dysphagia, 2-channel NMES has been reported to improve swallowing ability and discharge disposition.5-8 Most clinical studies regarding 2-channel NMES have focused on rehabilitative mechanism (cumulative effects) by muscle strengthening or facilitation of the swallowing reflex. 9 Motor stimulation has a muscle strengthening effect, and sensory stimulation facilitates the swallowing reflex. However, the precise mechanism of 2-channel NMES treatment is unknown, and controversy remains over its efficacy and methods of stimulation. 10 In previous meta-analysis studies in patients with poststroke dysphagia (acute, subacute, or chronic stage), traditional swallow treatment (eg, thermal stimulation, effortful swallowing, and postural adjustments) with NMES was seen to be more effective than that without NMES or usual care without swallowing treatment, but the effectiveness of treatment with NMES alone is unclear.9,11,12 Furthermore, in recent structured studies, NMES could not accelerate swallowing recovery in acute and subacute stage of poststroke dysphagia.13,14
A recent study for young and older healthy populations showed that sequential contractions of the suprahyoid and infrahyoid muscles cause a circular movement of the hyoid bone during swallowing.15,16 Hypothetically, NMES could be used not only for rehabilitation but also for the compensatory mechanism.17,18 The compensatory mechanism of NMES means that, unlike the simultaneous stimulation of the suprahyoid and infrahyoid muscles via the conventional 2-channel NMES, the stimulation of these muscles via the sequential 4-channel NMES may lead to a better modification of the abnormal hyoid and laryngeal motion in patients with dysphagia. Therefore, the 4-channel NMES treatment in the present study corresponds to a compensatory application. Compensatory application using 2-channel NMES has resulted in hyolaryngeal descent and the cancellation of positive effects, thereby resulting in no overall clinical improvement.19-21 However, in a pilot study, use of sequential 4-channel NMES resulted in significant improvements in clinical, kinematic, and pressure parameters during swallowing. 18
We hypothesized that compensatory application with sequential 4-channel NMES on the suprahyoid and infrahyoid muscles would improve the hyoid and laryngeal motion during swallowing, as well as the swallowing function in patients with acute, subacute, and chronic dysphagia. Here, we investigated the clinical efficacy of sequential 4-channel NMES for compensatory application during swallowing in a double-blinded randomized clinical trial (RCT) in these patients.
Methods
Study Design
This study was a multicenter, prospective, double-blinded RCT from September 12, 2018 to February 21, 2020. It was performed at the rehabilitation unit of 3 teaching hospitals (Seoul National University Bundang Hospital, Daegu Fatima General Hospital, and the Jeju University Hospital). The study protocol was approved by the institutional review boards of each hospital (E-1806/475-001, 2019-07-006, DFH19DPOS033), and all methods were performed in accordance with relevant guidelines and regulations. This study was also approved by the Ministry of Food and Drug Safety in the Republic of Korea (Approval number: 917) and registered on clinicaltrial.gov (registration number, NCT03670485). All patients or their representatives provided written informed consent prior to participation.
Participants
The study’s inclusion criteria were as follows: patients who were age ≥19 years and had symptoms of dysphagia; underlying diseases affecting cerebral function such as stroke, traumatic brain injury, brain tumor, and encephalitis; dysphagia as confirmed by the videofluoroscopic swallowing study (VFSS); stable vital signs; agreed to participate in the present study; and gave informed consent. If the patient’s Mini-Mental Status Examination (MMSE) score was <15, cognitive abilities were considered impaired and included in the study only if additional consent from the guardian was obtained. The VFSS criteria included definite presence of aspiration (defined as a penetration–aspiration scale [PAS] score ≥6) or presence of penetration (2≤ PAS score ≤5) with residual material at the vallecular pouch or pyriformis sinus to prevent ceiling effects. 22 Patients who had severe cognitive dysfunction who could not perform 1-step follow commands, serious psychiatric disorders, cervical surgery, respiratory difficulties, allergic reactions to electrodes of NMES, and those who were pregnant or breast-feeding were excluded. As this study evaluates the compensatory applications between the 4-channel NMES and sham groups, there were no restrictions with regard to the stage of dysphagia and hospital settings.
A total of 52 participants (26 for the 4-channel NMES group and 26 for the sham group) were initially enrolled. In the sham group, 1 participant dropped out due to a medical condition and 2 participants were additionally excluded because their underlying diseases were not brain lesion. Therefore, 26 participants in the 4-channel NMES group and 23 participants in the sham group completed the clinical trial (Figure 1(a)). Flow of patients through the trial. (a) Confirmatory study and (b) pilot study.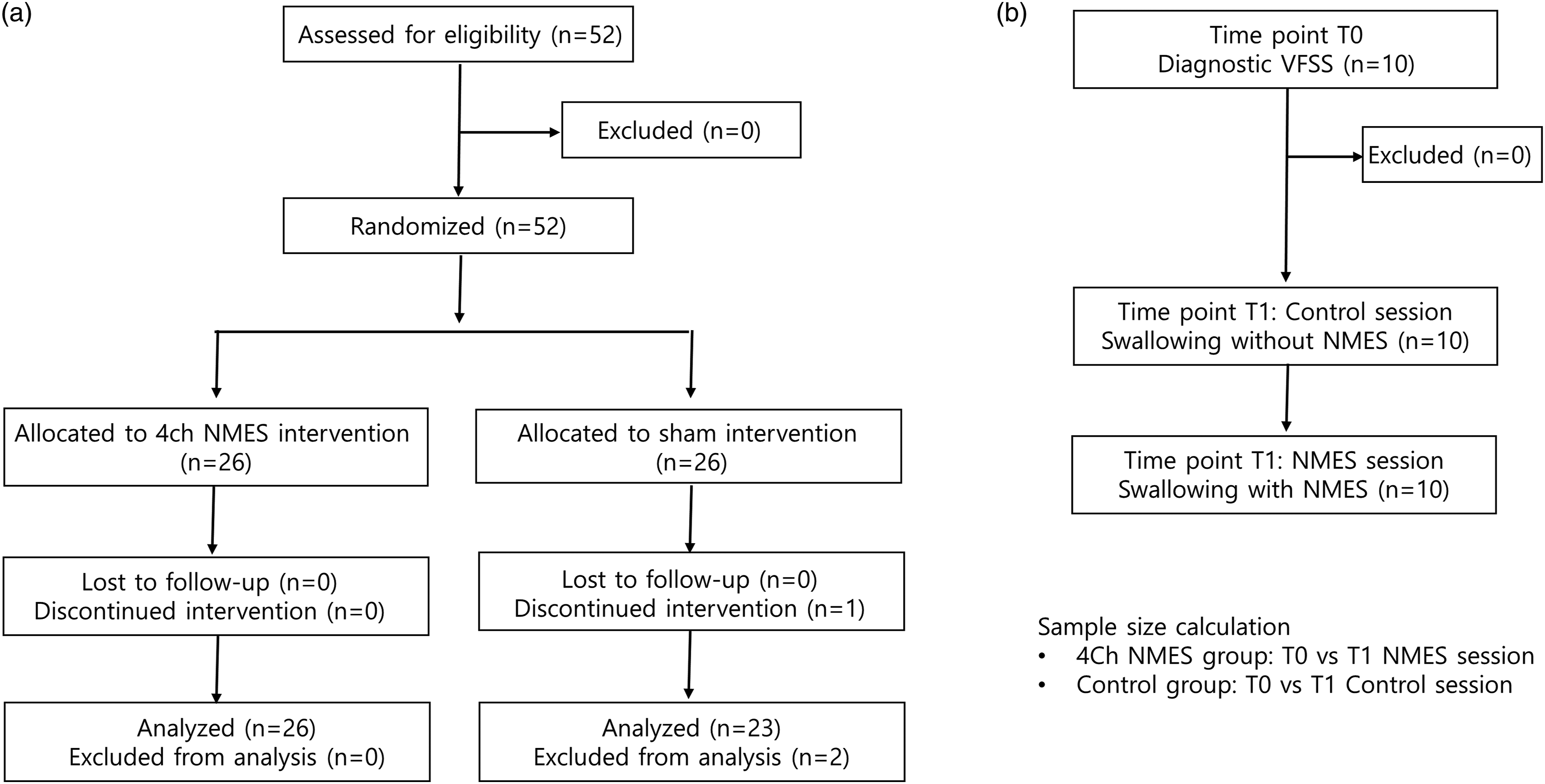
Randomization and Masking
Block randomization was used to randomly assign an equal number of patients to the 4-channel NMES or sham group via computerized random allocation sequences that were prepared by a statistician. The randomization schedule and group allocation were only accessible by the statistician and occupational therapists (EG Jang). All participants were blinded to the allocation, and electrodes were attached to 4 designated areas to guarantee masking. In addition, the investigators involved in the outcome assessment were blinded to the group allocation. This study conforms to all CONSORT guidelines and reports the required information accordingly (Figure 1(a)).
Equipment: Sequential 4-Channel NMES
The sequential 4-channel NMES has 4 channels that were adjustable for latency, amplitude, and duration of current (Supplementary Figure 1(a); STF-1000, Stratec Co, Ltd; Anyang, South Korea). The device uses 4 pairs of electrodes that were round in shape and 22 mm in size, and 2 types of electrodes with different gap sizes were used: .5 cm (type 1 electrode) and 1 cm (type 2 electrode). The type 1 electrode was used for channels 1, 2, and 4; and the type 2 electrode was used for channel 3 (Supplementary Figure 1(b); 1 Bio Medic Co, Ltd; Bucheon-si, Gyeonggi-do, South Korea).
The electrode locations were determined using anatomical landmarks and manual palpation (Supplementary Figure 1(c)). Channel 1 (right) and 2 (left) electrodes were placed superior to the hyoid bone and posterior to the mandible with a 1-cm interval from the midline to target the digastric and mylohyoid muscles. Channel 3 electrodes were placed on the bilateral superior pole of the thyroid cartilage to target the thyrohyoid muscles, and channel 4 electrodes were placed medial to the sternocleidomastoid muscle and inferior to the thyroid cartilage to target the sternohyoid, omohyoid, and sternothyroid muscles. The electrical stimulation algorithm was based on a previous study.15,18 The pulse frequency, pulse duration, and interphase interval are 80 Hz, 300 µs, and 100 µs, respectively. Channels 1 and 2 start their electrical stimulations first, and channels 3 and 4 start their stimulations 150 and 250 ms later, respectively. The stimulations of channels 1, 2, 3, and 4 lasted 1200, 1200, 1050, and 950 ms, respectively. All stimulations ended simultaneously in a single sequence. 15
Procedure
All participants were evaluated with initial VFSS in a standardized manner.4,23 After randomization, all participants underwent a preparation process before the intervention, where the stimulation intensity was gradually increased to the maximum tolerance level and the application of sequential stimulation of the 4-channel NMES were familiarized for 1–2 sessions. The maximal tolerable stimulation intensity levels were recorded for each electrode pair and used during the intervention. For synchronous stimulation of NMES, patients push the start button and begin swallowing when the NMES pulls the suprahyoid muscles.18,24
For the intervention, the 4-channel NMES group underwent the 4-channel NMES while swallowing 5 mL of thin (IDDSI, level 0) and extremely thick fluids (IDDSI, level 4) twice, whereas the sham group had electrodes attached at the same locations but did not undergo the NMES during swallowing. 23 All procedures were evaluated with VFSS. A second VFSS evaluation was performed within 1 week after the initial evaluation to minimize the natural recovery of dysphagia, and a 5-point Likert scale was used to assess satisfaction, easiness, and discomfort during swallowing after the second VFSS. During the interval between the initial and follow-up VFSS, all participants received conventional swallowing therapies.
Outcomes
All VFSS interpretations were performed by blinded 3 researchers (SY Lee, D Park, and JS Ryu) who certified the modified barium swallow impairment profile (MBSImPTM). Thus, our results are mean scores by 3 researchers.
The primary outcome was the videofluoroscopic dysphagia scale (VDS), and the secondary outcomes were PAS, 5-point Likert scale to assess satisfaction, easiness, and discomfort during swallowing and kinematic variables. VDS is a 14-item scale representing oral and pharyngeal functions that can be observed by VFSS. 25 PAS is an 8-point, equal-appearing interval scale for describing any penetration and aspiration events. 22 The Likert scale uses 5 points to describe outcomes as follows: (1) very negative, (2) negative, (3) neutral, (4) positive, and (5) very positive.26,27 For a kinematic analysis of VFSS, we used the automated kinematic analysis software (AKAS®).28,29 This program traced a 2-dimensional moving trajectory of the hyoid bone automatically, then detected and calculated the timing and distance variables among 4 points (A, the starting point of hyoid bone; B, the most upward point; C, the most anterior point [equilibrium state of supra and infrahyoid muscles] and; D, the most inferior location [the end of swallowing]; Supplementary Figure 2). The AKAS’s variables included the maximal velocity (pixel/frame) and the peak speed point between A and C and between C and D (frame). Also, it included the X–Y axes as well as the Euclidean and trajectory distances between A and C (pixel), instantaneous acceleration (pixel/frame2) and its point (frame), mean velocity of the Euclidean and trajectory distances between A and C, and the time duration within 20% of the A–C distance from C (duration of C, number of pixels).
Statistical Analysis
The calculation of the sample size was based on the pilot study of the sequential 4-channel NMES prototype (Figure 1(b)). 18 In this study, the initial diagnostic VFSS was performed (time point: T0) and follow-up VFSS was performed without NMES and then with NMES (T1). The difference in VDS between T0 and T1 with and without NMES was calculated in the 4-channel NMES and sham groups, respectively. The average values and standard deviations of the differences in VDS of the 4-channel NMES and sham groups were −4.00 ± 6.72 and 1.19 ± 3.74, respectively. With an α of less than .05 in the two-tailed tests and a power of 80%, the target sample size of each group was 19 patients. Considering a dropout rate of 25%, the final sample size was determined to be 26 patients per group.
All statistical analyses were conducted in accordance with the statistical analysis plan. SPSS 23.0 software (SPSS Inc.; Chicago, IL, USA) were used. The Student’s t-test or Mann–Whitney U test (if the assumptions of parametric statistical analysis were not satisfied) was used to compare the differences of the clinical, VFSS, and AKAS variables between the 4-channel NMES and sham groups in accordance with the statistical analysis plan. The chi-square and paired t-test or Wilcoxon signed-rank test (if the assumptions of parametric statistical analysis were not satisfied) were used to compare the pre-treatment and post-treatment evaluations. To correct for multiple comparisons, FDR (False Discovery Rate)-Benjamini–Hochberg procedure was used for P-value correction, so the level of statistical significance was less than .021–.026 for these analyses. The results are presented as the mean ± standard deviation. P-values <.05 were considered statistically significant.
Results
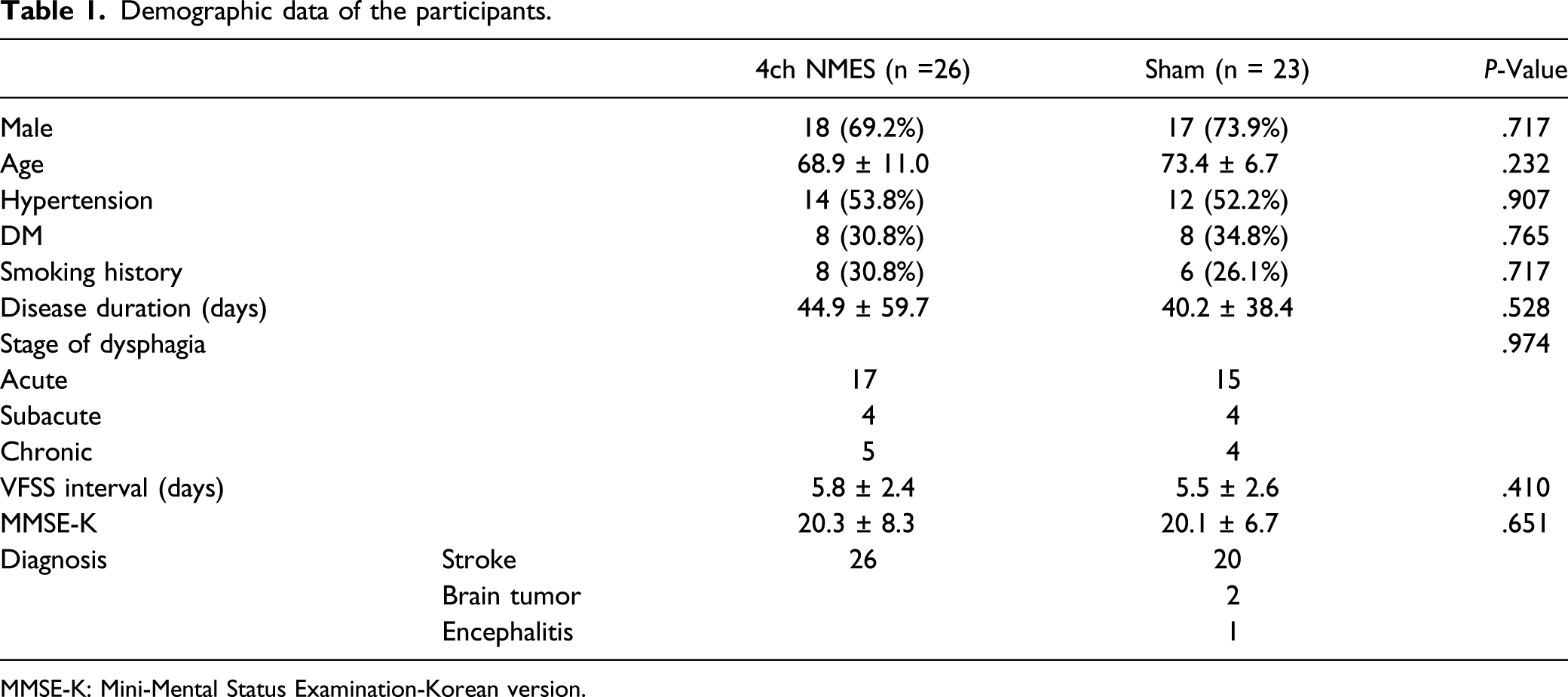
Demographic data of the participants.
MMSE-K: Mini-Mental Status Examination-Korean version.
The comparisons between pre- and post-treatment evaluations in the 4ch NMES and the sham groups.
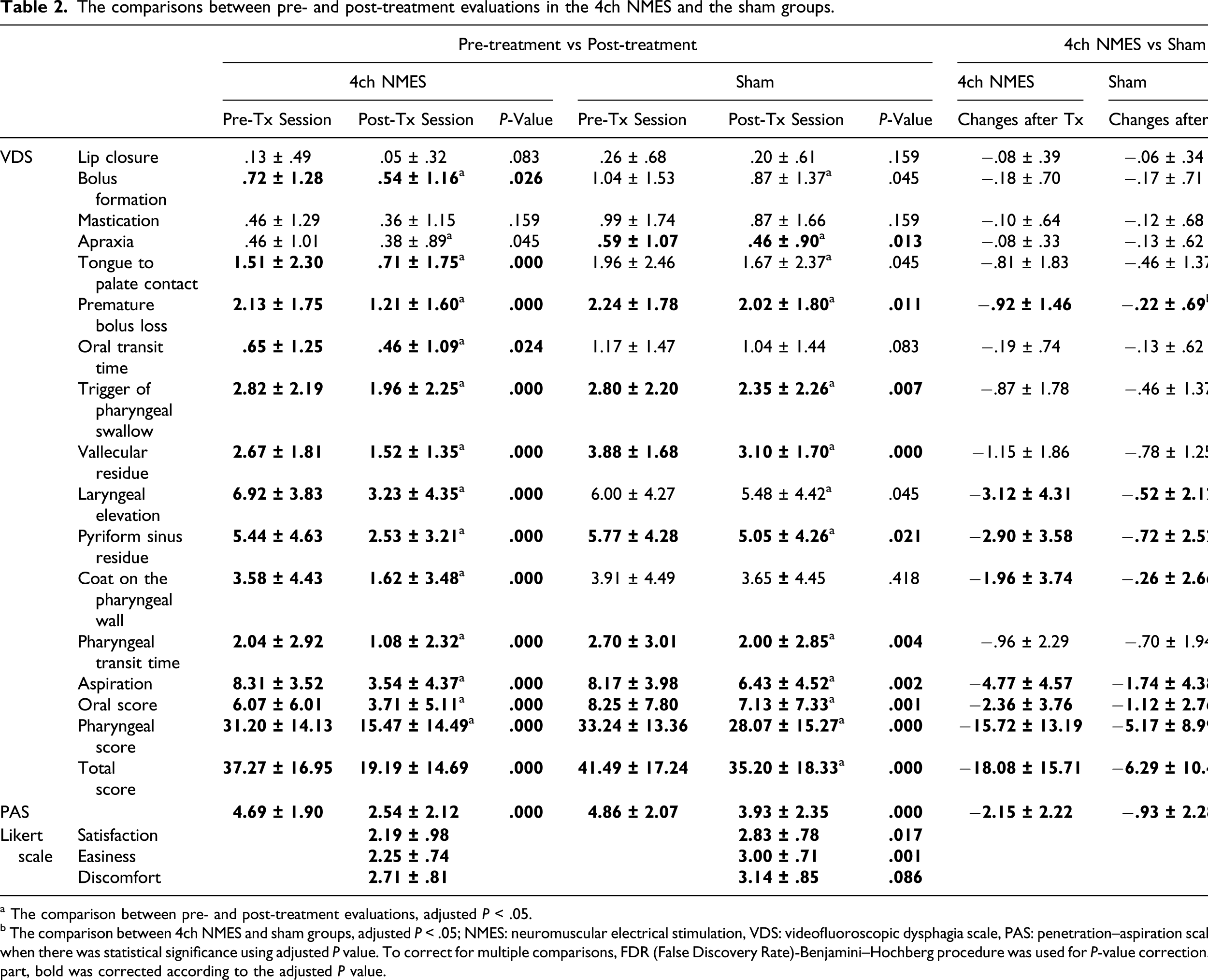
a The comparison between pre- and post-treatment evaluations, adjusted P < .05.
b The comparison between 4ch NMES and sham groups, adjusted P < .05; NMES: neuromuscular electrical stimulation, VDS: videofluoroscopic dysphagia scale, PAS: penetration–aspiration scale. Bold was used when there was statistical significance using adjusted P value. To correct for multiple comparisons, FDR (False Discovery Rate)-Benjamini–Hochberg procedure was used for P-value correction. For the colored part, bold was corrected according to the adjusted P value.
When we compared the pre- and post-VFSS changes between the 2 groups, the improvements of the 4-channel NMES group were significantly greater than that of the sham group with respect to oral, pharyngeal, and total VDS as well as PAS scores (P < .05, Table 2, Figure 2(a–b). In subcategories of VDS, premature bolus loss in oral VDS, laryngeal elevation, pyriform sinus residue, coating on the pharyngeal wall, and aspiration in pharyngeal VDS were significantly improved in the 4-channel NMES group than in the sham group. Also, the Likert scales for satisfaction, easiness, and discomfort for swallowing showed favorable results for the 4-channel NMES group (Table 2). Comparison of improvements before and after the 4-channel NMES and sham stimulation. (a) Oral, pharyngeal, pharyngeal, and total VDS scores were significantly improved in both groups, and the differences between pre- and post-evaluations were greater in the 4-channel NMES group. (b) PAS scores were also significantly improved in the 4ch NMES group (*: the comparison between the pre- and post-evaluations, p < .05; #: the comparison between the 2 groups, p < .05). The movement trajectory of the hyoid bone became larger and clear in direction with NMES in thin (c), extremely thick (d), and whole fluids (d). Abbreviations: NMES: neuromuscular electrical stimulation; VDS: videofluoroscopic dysphagia scale; PAS: penetration–aspiration scale.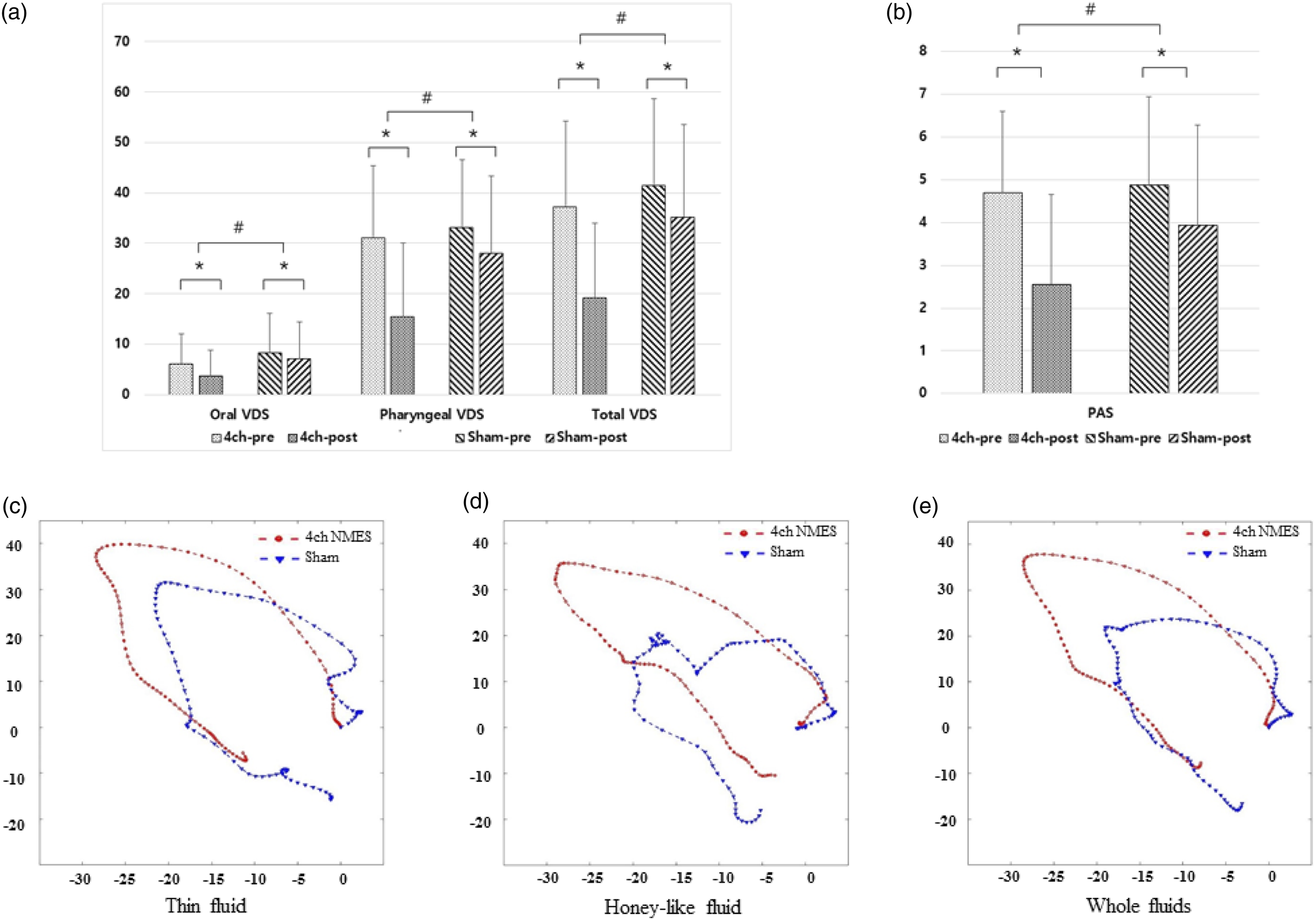
Comparisons of kinematic analysis data of the thin and extremely thick fluids swallowing between the 4ch NMES and the sham groups.
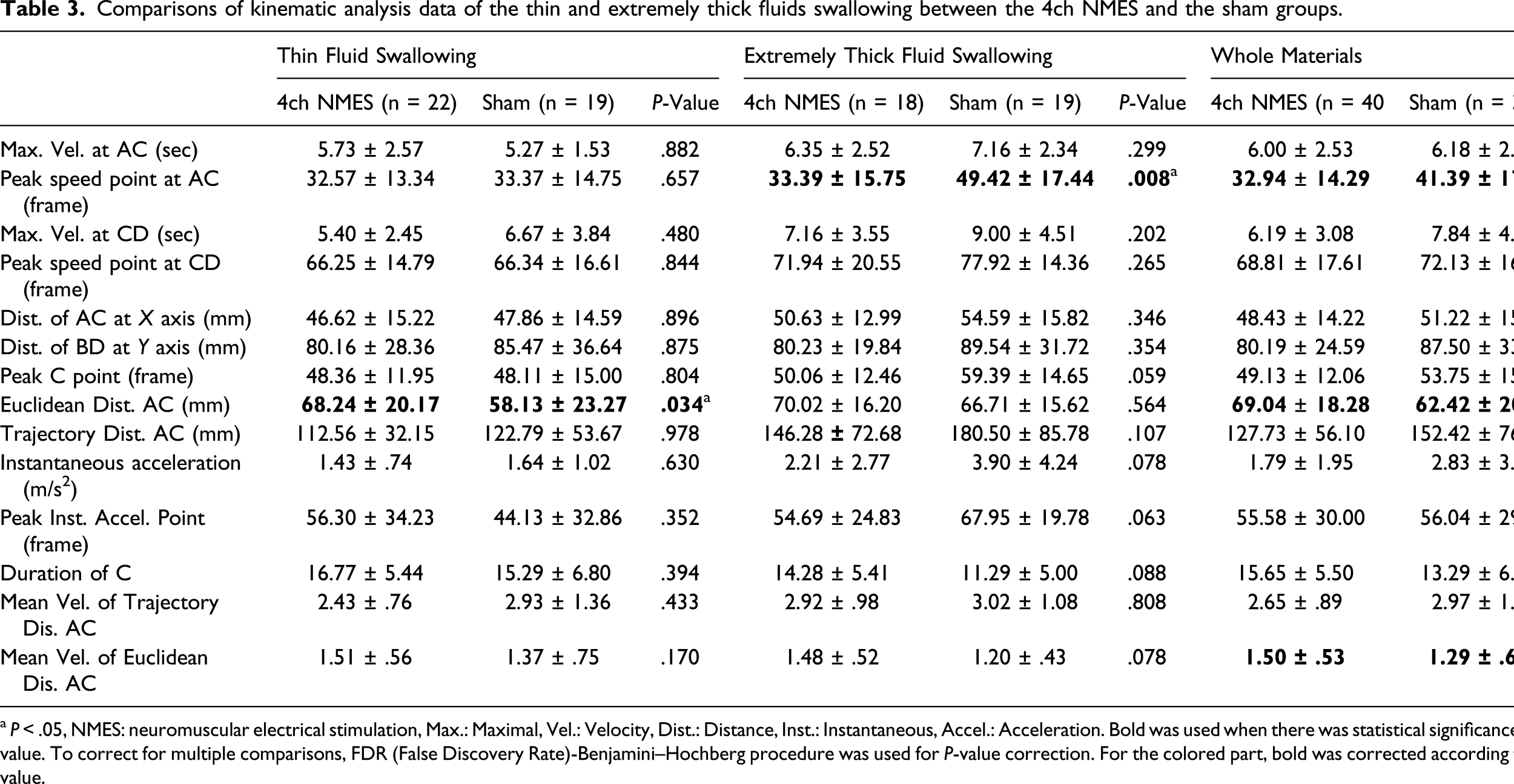
a P < .05, NMES: neuromuscular electrical stimulation, Max.: Maximal, Vel.: Velocity, Dist.: Distance, Inst.: Instantaneous, Accel.: Acceleration. Bold was used when there was statistical significance using adjusted P value. To correct for multiple comparisons, FDR (False Discovery Rate)-Benjamini–Hochberg procedure was used for P-value correction. For the colored part, bold was corrected according to the adjusted P value.
Discussion
In the EMG analysis of normal swallowing, the suprahyoid, thyrohyoid, sternohyoid, sternothyroid, and omohyoid muscles were sequentially activated with intervals of 150 and 350 ms.15,30 These sequential contractions accomplish the circular motion of the hyoid bone. 31 Likewise, the thyrohyoid muscle assists laryngeal elevation, and other infrahyoid muscles assist the upper esophageal sphincter opening by producing prolonged anterior motion of the hyoid bone.15,31 Therefore, to obtain a physiological motion, sequential stimulations with proper intervals and at least 3 channels are required.
When applying 2-channel NMES for the compensatory mechanism, we must select 2 regions for stimulation because of the limitation of the channels. In a previous study using compensatory application for dysphagia patients, the efficacy varied according to the stimulation sites.
21
Stimulation of the suprahyoid (Figure 3(a), (ⓐ)) and throat regions (Figure 3(a), (ⓑ)) during swallowing showed better results on the NIH-Swallowing Safety Scale but did not change the PAS score; however, stimulation of only the bilateral suprahyoid muscles did not show any improvement (Figure 3(a)).
21
In other studies that used 2-channel NMES in the normal population, stimulation significantly decreased laryngeal elevation and safety of swallowing than in non-stimulated swallowing.19,20 Therefore, based on previous studies of 2-channel NMES, we devised more effective and functional stimulation methods using 4-channel NMES. These methods included sequential stimulation to stimulate the normal contractile sequences of swallowing-related muscles
15
and stimulating the other infrahyoid muscles (Figure 3(a), (ⓒ, ⓓ)) that anchor and pull the hyolaryngeal complex in the anteroinferior direction so that the strength of the summated vector of the stimulated muscles (Figure 3(a), red line) was sufficient. In the present study, sequential 4-channel NMES was used as a compensatory application. These sequential stimulations for swallowing-related muscles (yellow arrows) were intended to improve the coordination and contractile strength of these muscles, thereby producing sufficient strength of the summated vector (red arrow) (Figure 3(b)). Schematic drawings of the changes in muscular contractions stimulated by 2-channel (a) and 4-channel NMES (b). The arrows imply force vectors. Blue arrows (dashed line) indicate unstimulated vectors, yellow arrows (dotted line) indicate stimulated vectors by NMES, and red arrows (solid line) indicate the summated vector of stimulated vectors. In these images, the size of the vector is not quantified. Abbreviation: NMES: neuromuscular electrical stimulation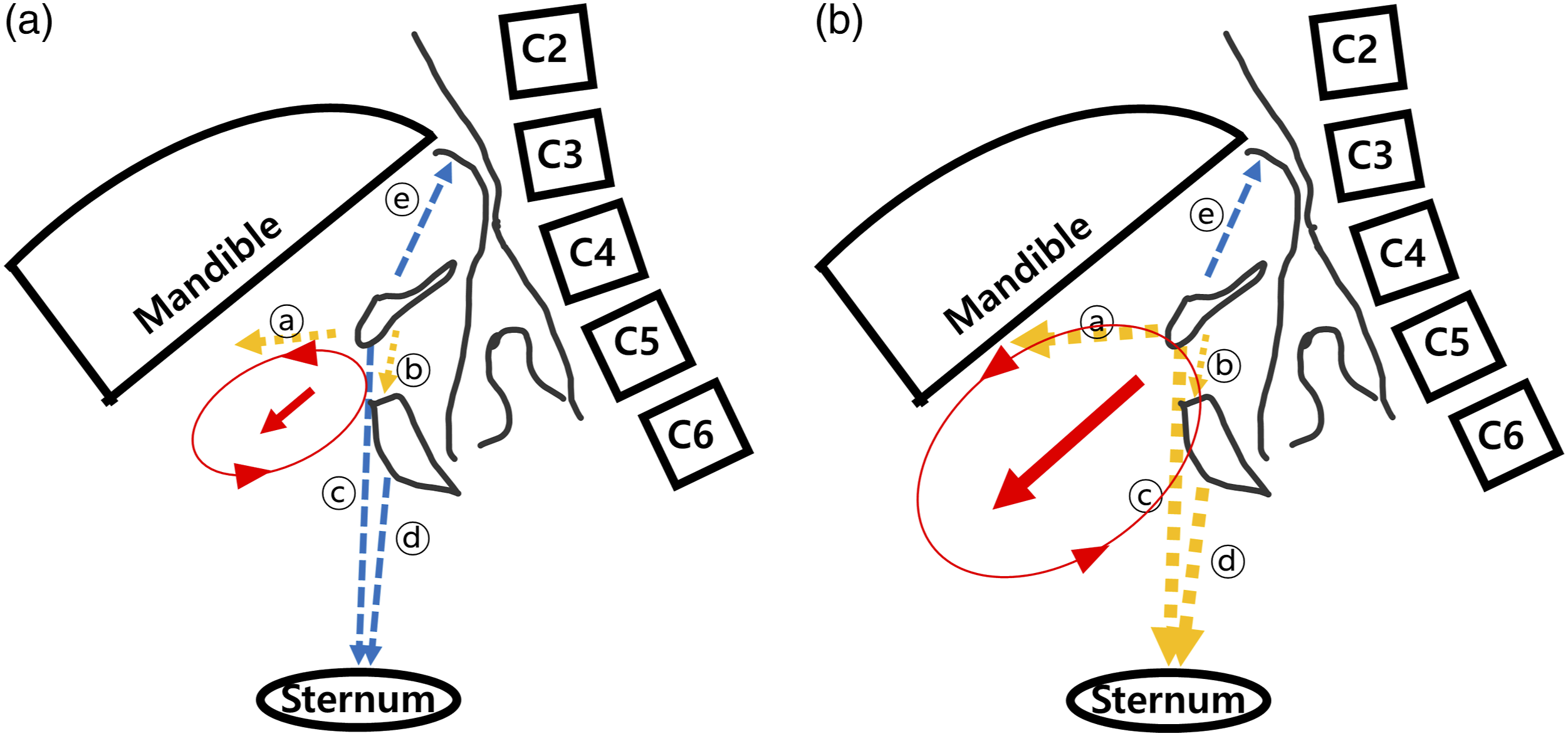
Here, the 4-channel NMES group showed significant improvement with respect to oral, pharyngeal, and total VDS as well as PAS scores than the sham group (Table 2, Figure 2(a and b)). In subcategories, our results showed significant improvements in premature bolus loss, which was correlated with pre-swallow aspiration; improved laryngeal elevation and decreased aspiration in VDS subscale and PAS, which implied decreased aspiration during swallow; and significantly decreased piriformis sinus residue and coating on the pharyngeal wall in VDS, which implied decreased risk of post-swallow aspiration. Post-deglutitive overflow aspiration has been frequently observed in patients with pharyngeal retention, and the risk of aspiration increases markedly with the amount of residue. 32 Therefore, our present study demonstrated that the sequential 4-channel NMES decreased the aspiration risk in the whole swallowing processes.
The 4-channel NMES significantly improved the oral VDS scores in the oral phase. Tongue-base pressure is important for tongue contact with the posterior pharyngeal wall and for squeezing out the bolus through the pharynx. 33 Alterations in tongue coordination, strength, and pressure generation may result in a disruption of bolus movement from the oral cavity to the pharynx and result in increased risk of aspiration before or after swallowing. 34 In the present study, the first 2 channels were widely attached to the suprahyoid lesion (submandibular area). Thus, the sequential 4-channel NMES seem to induce effective contraction of the genioglossus and tongue muscles, which is highly correlated with tongue motion and can improve the strength of the tongue base, thereby resulting in superior efficacy in oral VDS scores.
The analysis of the hyoid bone is reflective of swallowing function.35,36 In this current study, sequential 4-channel NMES significantly increased the Euclidean distance and velocity of the hyoid movement, therefore normalizing the hyoid excursion (Figure 2(c–e)). 18 Also, the peak speed point was significantly faster in the 4ch NMES group, which implied that the 4-channel NMES group reached the peak velocity early with the assistance of NMES. Initially, the NMES pull the hyoid bone and the patients resist. When the patients start swallowing, the contractions of the swallowing-related muscles are augmented by the NMES. In summary, these results suggest that the sequential 4-channel NMES facilitated the movement of hyolaryngeal structures during swallowing, thereby improving swallowing function.
Previously, though functional NMES has been applied as a walking aid, NMES has limitations in daily applications due to muscle fatigue. 37 Eating is completed within 15 repetitive swallowing motions; therefore, we do not need to consider muscle fatigue, and application to daily swallowing is possible and promising. In the present study, the cognitive function was relatively preserved (the average value of MMSE> 20), and we trained the participants to use the device. Therefore, before applying this device at home or a long-term facility, the clinician should check the patients’ cognitive function and their ability to use the device. In addition, follow-up studies are required to develop daily application of sequential 4-channel NMES at home or in long-term facilities.
In the present study, the 4-channel NMES group showed favorable outcomes in subjective measurements for satisfaction, easiness, and discomfort. With the help of the 4-channel NMES, the patients showed improved easiness during swallowing and decreased discomfort maybe due to less required effort during swallowing. Also, the decreased residual sensation after swallowing might have contributed to the decreased discomfort and satisfaction. In the present study, no adverse events were observed. As sequential 4-channel NMES stimulated swallowing-related muscles with a normal contractile algorithm, thereby inducing physiological motions, less discomfort developed. However, it seems to be necessary that a long-term study should be conducted on the clinical effectiveness, patient satisfaction, and risk for the daily application of 4-channel NMES in the future.
This study had some limitations. First, this study was designed as double-blind to keep the blinding as much as possible. However, during the intervention, the participants know the presence or absence of NMES and the blindness is broken. However, since the study was completed immediately after this test, it was described as double-blind. Second, we used the Likert scale only once after the study was completed, and comparisons could not be made with swallowing without NMES. Third, this study was only performed during hospitalization. Fourth, although there were no statistically significant differences between groups, there was a potential for bias due to differences in age and the severity, acuity, and etiology of dysphagia between groups. Fifth, as patients were required to be able to swallow at least 5 mL of thin and very thick liquid twice in order to participate in the study, the results do not apply to individuals with severe dysphagia who are unable to swallow thin liquids. Therefore, follow-up studies are required to evaluate the effect of treatment with 4-channel NMES on dietary tolerance of solid and liquid food of different consistencies, which is a practical consideration in dysphagia treatment.
Conclusion
A new method using sequential 4-channel NMES on the suprahyoid, thyrohyoid, and infrahyoid muscles during swallowing showed significant clinical improvement with respect to VDS, PAS, and kinematic analysis. Since no side effects were observed during the investigation, 4-channel NMES seems to be a safe and well-tolerated treatment method for dysphagia. Therefore, sequential 4-channel NMES can be used as a new treatment method for the compensatory mechanism in patients with dysphagia.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211029891 – Supplemental Material for Compensatory Effects of Sequential 4-Channel Neuromuscular Electrical Stimulation for the Treatment of Acute, Subacute, and Chronic Dysphagia in a Prospective, Double-Blinded Randomized Clinical Trial
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211029891 for Compensatory Effects of Sequential 4-Channel Neuromuscular Electrical Stimulation for the Treatment of Acute, Subacute, and Chronic Dysphagia in a Prospective, Double-Blinded Randomized Clinical Trial by So Young Lee, Donghwi Park, Joonyoung Jang, Eun Gyeong Jang, Jun Chang Lee, Yulhyun Park, Seon Cho, Won-Seok Kim, Jihong Park, Bo Ryun Kim, Kyoung-Ho Seo, Sungwon Park and Ju Seok Ryu in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgment
The authors gratefully appreciate MRCC team of Seoul National University Bundang Hospital for their work in the area of statistics in this study.
Author Contributions
Conceptualization: Ju Seok Ryu.Enrollment of participants: Joonyoung Jang, So young Lee, Bo Ryun Kim, Donghwi Park, and Kyoung-Ho Seo.Procedures: Joonyoung Jang, Eun Gyeong Jang, Seon Cho, So Young Lee, Bo Ryun Kim, Donghwi Park, Kyoung-Ho Seo, Sungwon Park, and Ju Seok Ryu.AKAS development and analysis: Jun Chang Lee.VFSS analysis: So young Lee, Donghwi Park, and Ju Seok Ryu.Writing: Ju Seok Ryu.
Declaration of Conflicting Interests
No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the authors or upon any organization with which the authors are associated.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI18C1169). This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2016R1D1A1B03935130).
Data sharing
All data in this study are available after de-identification upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
