Abstract
Background. Chronic poststroke oropharyngeal dysphagia (CPSOD) is associated with impaired oropharyngeal sensory/motor function. We aimed to assess effect of sensory (SES) and motor (NMES) transcutaneous electrical stimulation (TES) on safety of swallow and clinical outcomes in patients with CPSOD in a one-year follow-up randomized controlled trial. Methods. Ninety patients (74.1 ± 11.5 y, modified Rankin score 2.6 ± 1.7) with CPSOD and impaired safety of swallow were randomized to (a) compensatory treatment (CT), (b) CT + SES, and (c) CT + NMES. Patients were treated with up to two cycles (6 months apart) of 15 × 1 hour TES sessions over two weeks and followed up with 4-5 clinical and videofluoroscopic assessments during one year. Key results. Baseline penetration–aspiration scale (PAS) was 4.61 ± 1.75, delayed time to laryngeal vestibule closure (LVC) 396.4 ± 108.7 ms, and impaired efficacy signs 94.25%. Swallowing parameters significantly improved between baseline and 1-year follow-up in SES and NMES groups for prevalence of patients with a safe swallow (P < .001), mean PAS (P < .001), time to LVC (P < .01), and need for thickening agents (P < .001). Patients in the CT presented a less intense improvement of signs of impaired safety of swallow without significant changes in time to LVC. No differences between groups were observed for 1-year mortality (6.1%), respiratory infections (9.6%), nutritional and functional status, QoL, and hospital readmission rates (27.6%). No significant adverse events related to TES were observed. Conclusions and inferences. Transcutaneous electrical stimulation is a safe and effective therapy for older patients with CPSOD. After 1-year follow-up, TES greatly improved the safety of swallow and reduced the need for fluid thickening in these patients.
Introduction
Oropharyngeal dysphagia (OD) is common in poststroke classified by the WHO (ICD-9:438.82; ICD-10:I69.391). 1 Its prevalence is 45.06% 2 and 41.7% 3 in the acute and chronic phases, respectively. Chronic poststroke OD (CPSOD) is associated with respiratory infections, pneumonia and malnutrition and an independent risk factor for prolonged hospitalization, institutionalization, poorer functionality, and 3-month mortality after stroke. 2 Chronic poststroke oropharyngeal dysphagia is associated to significantly higher economic costs. 4
Pathophysiology of CPSOD has been associated with (a) bi-hemispheric reduction of efferent pathway’s excitability with loss of motor dominance5,6 and (b) reduced pharyngeal sensitivity 7 with disrupted cortical conduction/integration of pharyngeal sensory inputs at stroke site. 8 These dysfunctions and the uncoupling between sensory/motor swallowing circuitry cause the slow oropharyngeal swallow response (OSR) in these patients.6,9 Delayed time to laryngeal vestibule closure (LVC) is the key impairment causing unsafe swallows and aspirations 9 in CPSOD, and aspiration pneumonia is the main cause of death, the first year PS. 10
Chronic poststroke oropharyngeal dysphagia is an underdiagnosed and undertreated complication. Classical treatment is compensatory and consists of fluid viscosity and food texture modification to reduce aspiration risk.11,12 The effect of compensatory treatments (CTs) is to avoid aspirations rather than improving OSR, while new treatments aim to improve swallowing function through peripheral or central neurostimulation. Pharyngeal electrical stimulation (PES) aims at activating pharyngeal afferents to promote swallowing recovery; however, a recent multicentric randomized clinical trial (RCT) found improvements in swallowing parameters in both active and control arms but without significant statistical differences between them, showing a neutral result for this therapy. 13 On the other hand, transcutaneous electrical stimulation (TES) is a noninvasive peripheral neurostimulation strategy approved for OD treatment by the food and drug administration, the ministry of food and drug safety (South Korea), and the ministry of health, labour and welfare (Japan) and consists of the application of electrical current through electrodes placed in the anterior part of the neck. We hypothesize TES might induce cortical neuroplastic changes responsible for the recuperation of swallowing function.14,15 The National Institute for Health and Care Excellence (NICE) declared TES as a safe and potentially beneficial therapy for PSOD patients although the actual evidence is limited in quality/quantity.16,17 We previously found that 10 days one-hour treatment with sensory or motor TES improved time to LVC and safety of swallow in a group of CPSOD, 18 but studies from other groups have shown mixed results,19,20 and the long-term effect of TES in CPSOD patients has not yet been studied. NICE recommends that research on TES for OD should address patient selection, variations in technique, the retreatment needs, and long-term outcomes. 16
Our main aim was to assess the effect of TES on safety of swallow and OSR, after the treatment and at 1-year follow-up, in patients with CPSOD. Secondary aims were to assess the effect of TES on the efficacy of swallow, functional and nutritional status and clinical outcomes, and the safety and tolerance of TES after treatment and at 1-year follow-up.
Patients and Methods
Patients
Ninety CPSOD patients (≥3 months from stroke) were consecutive included from October 01, 2014 to June 16, 2016. They were clinically assessed for OD with the volume–viscosity swallowing test (V-VST) 21 in Hospital de Mataró. Inclusion criteria were as follows: age ≥ 18 years old, clinical indications of OD, capacity to comply with the protocol, confirmed stroke diagnosis, no OD history before stroke, and score 0 or 1 on NIHSS question 1a. The protocol was approved by our Ethical Committee (CEIC:36/14) and conducted according to the Declaration of Helsinki. We obtained the written informed consent from each patient before enrollment. ClinicalTrials.Gov ID:NCT02379182. This article includes the CONSORT 2010 checklist of information to include when reporting a randomized controlled trial as a supplementary file and accomplishes all the CONSORT 2010 criteria.
Study Design
Prospective, randomized, controlled, three-arm, open-label, blinded-analysis RCT to evaluate and compare the effect of TES at two stimulation levels (sensory and motor) using VitalStim™ (Chattanooga, DJO Global, Lewisville, TX, United States) on the videofluoroscopic (VFS) signs of impaired safety and efficacy of swallow and the OSR in patients with CPSOD. We designed a RCT with 3 arms: a control group only with compensatory strategies and two active treatment groups. On the first visit (screening), patients were clinically assessed with the V-VST
21
and those with signs of impaired safety underwent VFS evaluation. Those with a penetration–aspiration score (PAS) ≥ 2
22
and/or pharyngeal residue were randomized into one of the three treatment arms using a specific software (GraphPad QuickCalcs, San Diego, CA, United States): (1) control group, standard compensatory care at our institution with postural changes, fluid thickening, and texture-modified foods if required, according to VFS examination
11
; (2) sensory group, compensatory treatment plus TES at sensory level intensity; and (3) motor group, compensatory treatment plus TES at motor level intensity.
18
We collected sociodemographic data, concomitant medication, functional capacity according to the Barthel index
23
and the modified Rankin scale,
24
comorbidities according to the Charlson comorbidity index,
25
stroke severity according to the NIHSS,
26
nutritional status with the Mini Nutritional Assessment-Short Form (MNA-SF)
27
and the body mass index (BMI), and quality of life with EQ-5D.
28
Patients were evaluated on the screening/pretreatment visit (day 1) and 1 week, 6 and 12 months following the first cycle of treatment (Figure 1), and treated with up to two cycles (6 months apart) of 15 1-hour stimulation sessions during two weeks. The second treatment cycle of TES was only performed on those patients who still had signs of impaired safety of swallow at follow-up VFS3. The study protocol is in Supplementary Material. Study design. Abbreviations: OD, oropharyngeal dysphagia; V-VST, volume–viscosity swallowing test; VFS, videofluoroscopy; TES, transcutaneous electrical stimuli.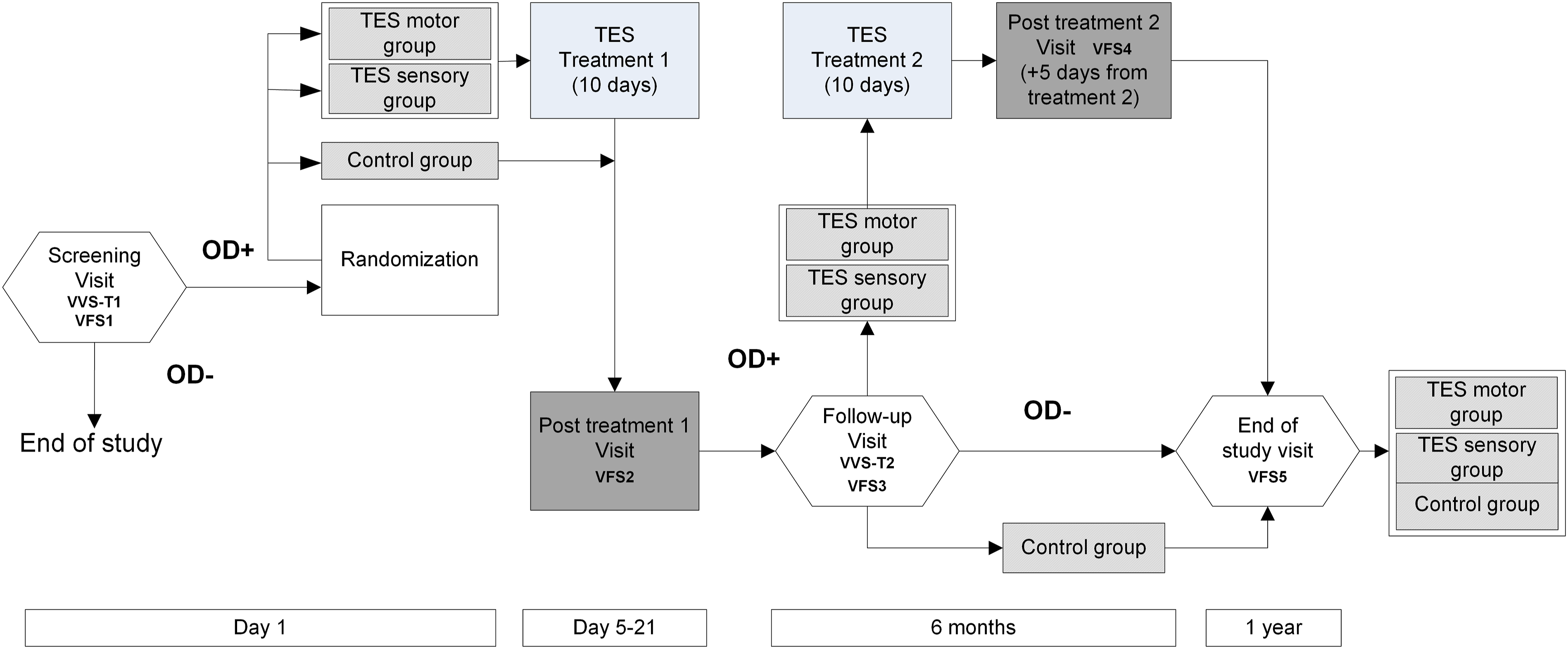
Our study hypothesis was that both TES treatments would improve safety of swallow compared to the control group with CT that NMES treatment will also improve efficacy of swallow and that these effects would remain after 1-year follow-up without relevant adverse events related to TES.
Primary and Secondary Outcome Measures
The primary study endpoint was the effect of the active treatment (TES groups) compared to control group on the safety of swallow (change in PAS scores at post-treatment and study end visits). The secondary endpoints were (compared to the control group) as follows: incidence of all adverse events; change in pharyngeal residue prevalence at post-treatment and study end visits; change in NIHSS at follow-up and study end visits; change in mRS and BI at follow-up and study end visits; and clinical outcomes.
Blinding of Study Intervention
The study was open-labeled for the clinician who administered the therapy and for the patient, who knew if he/she was assigned to the control or one of the treatment groups. However, researchers measuring the VFS responses and clinicians following the patients and providing the recommendations on fluid thickening and texture-modified diets and those performing VFS and data analysis were blinded to the study arm.
Study Intervention
Group 1 (Control)
Standard compensatory clinical care of CPSOD at our center, including adaptation of fluids with thickening agents, texture-modified diet, oral hygiene recommendations, postural changes if necessary (chin down and head rotation to the affected side), and nutritional advice. 11
Group 2 (SES)
Transcutaneous electrical stimulation at sensory level
18
plus the compensatory clinical care described in the control group. SES treatment consisted of the application, at rest, of biphasic pulses (0.7 ms duration) at 80 Hz using VitalStim device (Chattanooga Group, Hixson, TN, United States) two 1-hour sessions per day the first week and one 1-hour session per day the second week (15 sessions). Sessions were applied from Monday to Friday for two weeks. First, the motor threshold was determined by triplicate at the beginning of each session and was defined as the intensity needed to elicit muscular contraction underneath electrodes’ placement. Treatment intensity was set to 75% of the motor threshold, and the electrode placement was in the thyrohyoid position (Figure 2A) aiming to stimulate either cutaneous sensory afferents coming from C2–C3 spinal nerves, as from deeper sensory branches involving the glossopharyngeal cranial nerve (IX) and the superior laryngeal branch of the vagus cranial nerve (X).15,18,29 Every 10 minutes, the patient was asked if the initial intensity sensory perception was maintained, and in the case of reduction due to a common physiological habituation phenomena, it was readjusted to a sensory perception similar to the previous one.
18
Electrode placement. (A) Thyrohyoid placement for the sensory stimulation group and (B) suprahyoid placement for the motor stimulation group.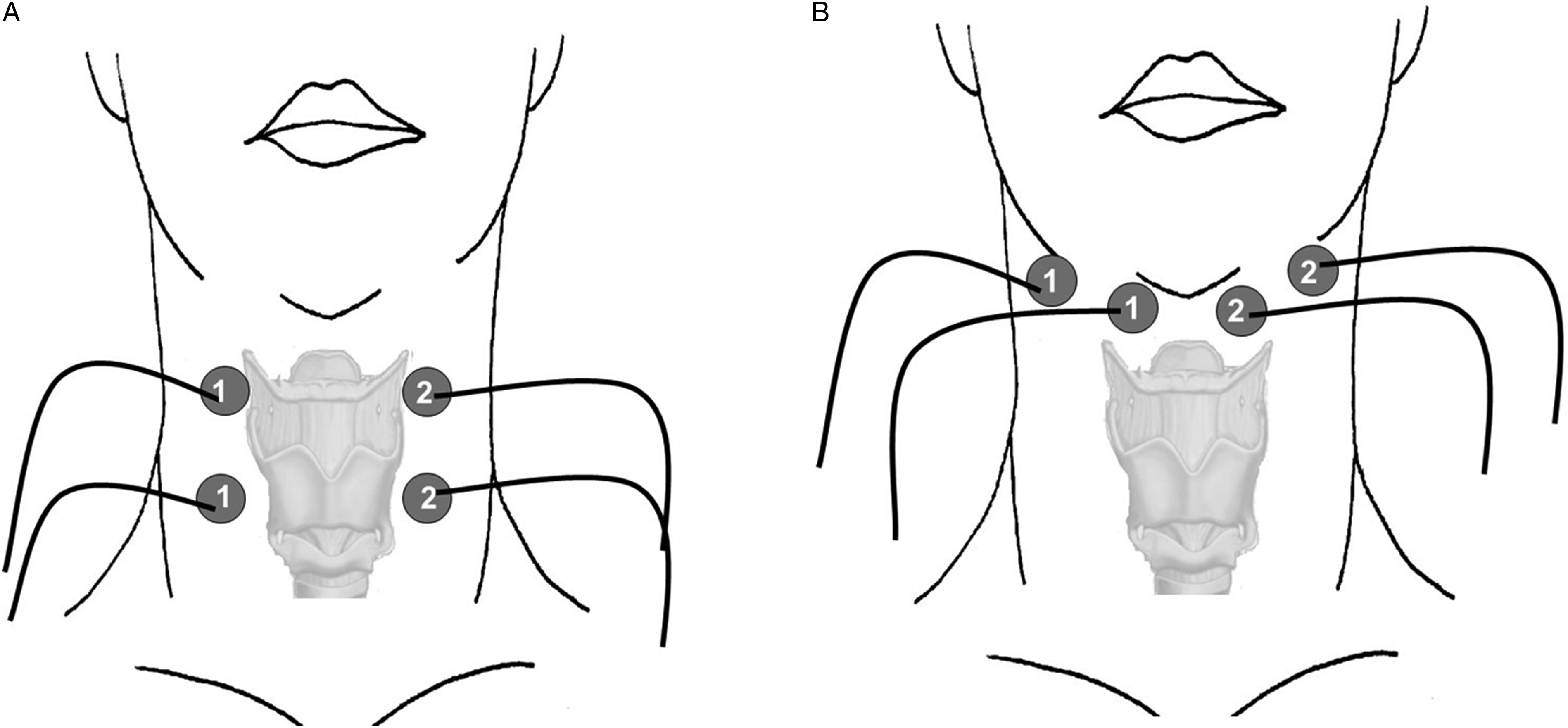
Group 3 (NMES)
Transcutaneous electrical stimulation at motor level (NMES) plus the compensatory clinical care described in the control group. The treatment schedule was the same as in the SES group, but the current intensity was set to 100% of the motor threshold, and the electrodes were located in a suprahyoid configuration (Figure 2B) to target the muscular mouth floor complex formed by the anterior belly of the digastric, the mylohyoid, and the geniohyoid muscles. This arrangement pulls the hyoid bone upward and toward the mandible during muscular contraction, facilitating airway protection and upper esophageal sphincter opening (UESO). 18 Initial current intensity was also readjusted every 10 minutes if necessary.
Patients from both TES groups did not perform any active exercise or maneuver during the sessions. At 6 months follow-up, and after a positive V-VST test with signs of impaired safety of swallow, those patients with a PAS ≥ 2 during VFS3 received a second cycle of treatment with the same protocol of stimulation and with a pre-post swallowing evaluation with VFS (Figure 1).
Swallowing Assessment
V-VST
The V-VST is a validated clinical assessment tool that uses three volumes (5, 10, and 20 mL) and viscosities (nectar (120 mPa·s), liquid (20 mPa·s) and pudding (4500 mPa·s)) to evaluate clinical signs of impaired efficacy (impaired labial seal, piecemeal deglutition, oral residue, and pharyngeal residue), and safety of swallow (wet voice, cough, and decrease in oxygen saturation ≥3% measured with a finger pulse oximeter). 21 Diagnostic sensitivity and specificity for OD are .94 and .88, respectively, and the reliability of V-VST is also high with an overall kappa value of .628 (95 % CI = .45–.78). 30
VFS
Videofluoroscopic was performed at baseline, pre- and post-TES and at 12 months follow-up (Figure 1). The algorithm and equipment used during VFS exploration has been previously published.6,9,18,31,32 Stopping criteria for the VFS were defined as an observed swallow with PAS ≥ 5. Boluses of 120 mPa·s and 4500 mPa·s were obtained with 3.5 g and 8 g, respectively, of Resource Thickener (Nestle Nutrition, Barcelona, Spain) mixed in 100 mL 1:1 mineral water and the radiological contrast Gastrografin (Bayer Hispania, Sant Joan Despí, Spain). Signs of impaired safety and efficacy of swallow and PAS were assessed in all boluses. 31 The prevalence of VFS signs of impaired efficacy and safety of swallow were measured as the ratio of the number of patients presenting these signs to the total number of patients studied. Impaired safety of swallow was defined if a patient had a PAS ≥ 2 during VFS examination at any volume or viscosity. Mean maximum PAS score per study group was calculated from the worst PAS score over the whole VFS exploration from each of the patients from each study arm. 32 Timing of OSR and strength of bolus propulsion were measured with the 5 mL 120 mPa·s bolus.
Clinical Outcomes
Hospital readmissions, prevalence of respiratory infections (including pneumonia), 33 nutritional status (BMI and MNA-sf), quality of life, and mortality were registered. Clinical complications were determined from the electronic medical records of the patients. 11 Functional, nutritional status, and QoL were measured over the whole study. 11
Adverse Events (AE)
Adverse events occurring during the study were monitored according to the guideline of categories described by the WHO and the Uppsala Monitoring Centre. 34
Statistical Analysis
Power calculation: accepting an alpha risk of .05 and a beta risk of .2 in a two-sided test, 30 subjects are necessary in each group to recognize as statistically significant, a difference greater than or equal to 2 points in the PAS score. The common standard deviation is assumed to be 2.5 (data from previous studies). It has been anticipated to have a drop-out rate of 15%.
Statistical methods: categorical data are presented as relative and absolute frequencies and continuous data as mean ± standard deviation. When comparing the effect of each treatment within the same group between different study points, the McNemar test for categorical variables and the Wilcoxon test for continuous were used. When comparing different treatments between groups, categorical variables were compared with the chi-square test and continuous variables with ANOVA test for nonparametric samples (Kruskal–Wallis) with a post hoc analysis (Dunn’s multiple comparison test). To compare between pairs, the effect of each treatment was normalized with respect to its own baseline state (% change), and Mann–Whitney U test was used. P-value < .05 was considered as statistically significant. Statistical analysis was performed with GraphPad Prism 6.0 (San Diego, CA, United States). Results on intergroup comparisons, between baseline and post-treatment 1 and between baseline and one-year post-intervention visit are shown as Supplementary Material.
Results
Patient Flow Chart
A total of 204 patients were screened and 114 excluded because they presented a safe swallow (PAS < 2) and no residue in the VFS, were not able to follow the protocol, or did not want to participate. The remaining 90 were randomized, 30 to each group and 29 patients from each treatment group completed the first period of treatment. Finally 26, 25 and 26 patients from the control, sensory and motor group, respectively, completed the study at 1-year follow-up (Supplementary Material).
Patient Characteristics
Baseline demographic, clinical, and neurological characteristics of study population.
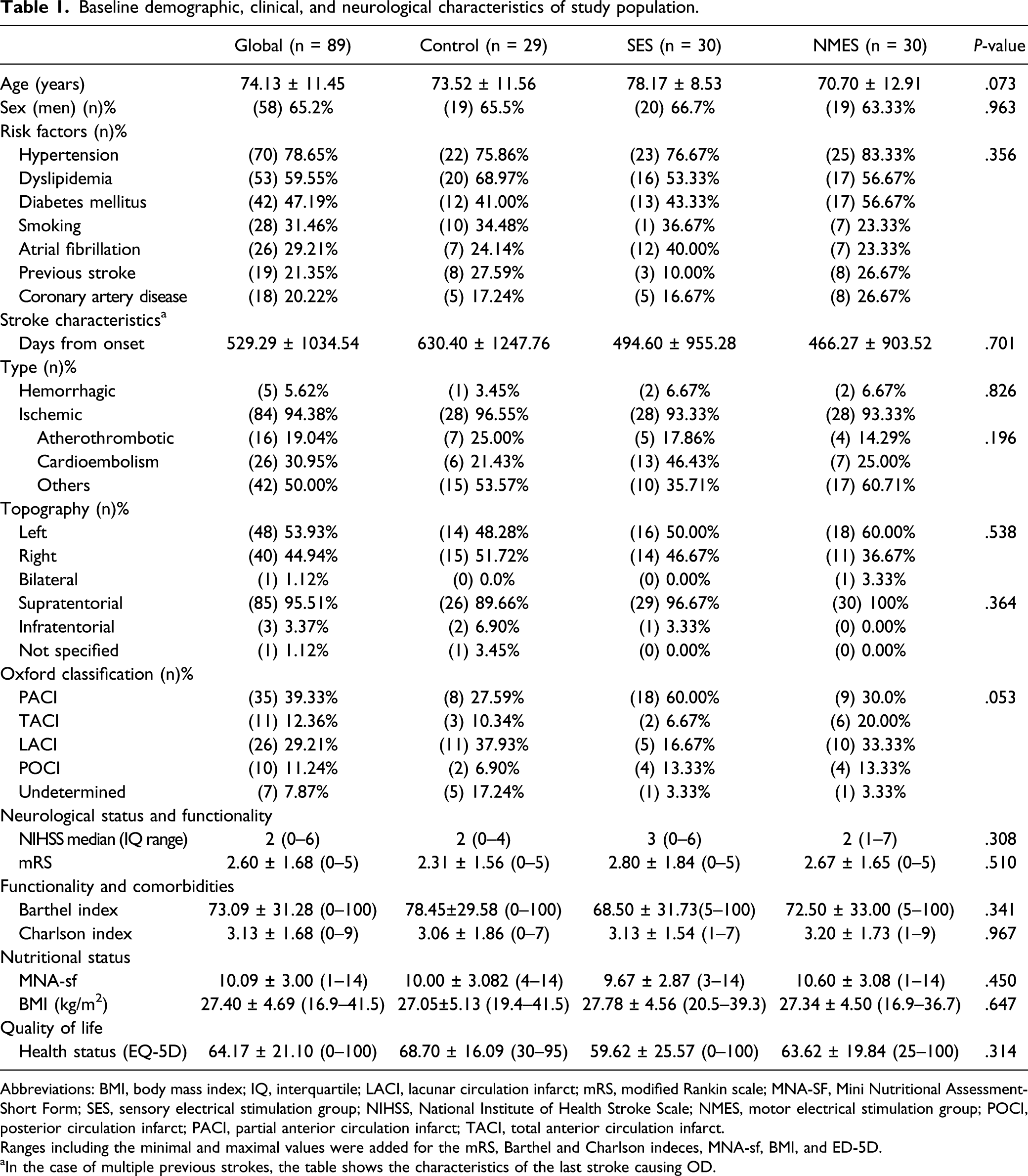
Abbreviations: BMI, body mass index; IQ, interquartile; LACI, lacunar circulation infarct; mRS, modified Rankin scale; MNA-SF, Mini Nutritional Assessment-Short Form; SES, sensory electrical stimulation group; NIHSS, National Institute of Health Stroke Scale; NMES, motor electrical stimulation group; POCI, posterior circulation infarct; PACI, partial anterior circulation infarct; TACI, total anterior circulation infarct.
Ranges including the minimal and maximal values were added for the mRS, Barthel and Charlson indeces, MNA-sf, BMI, and ED-5D.
aIn the case of multiple previous strokes, the table shows the characteristics of the last stroke causing OD.
Baseline Swallowing Evaluation
VFS
Baseline videofluoroscopic swallowing evaluation.
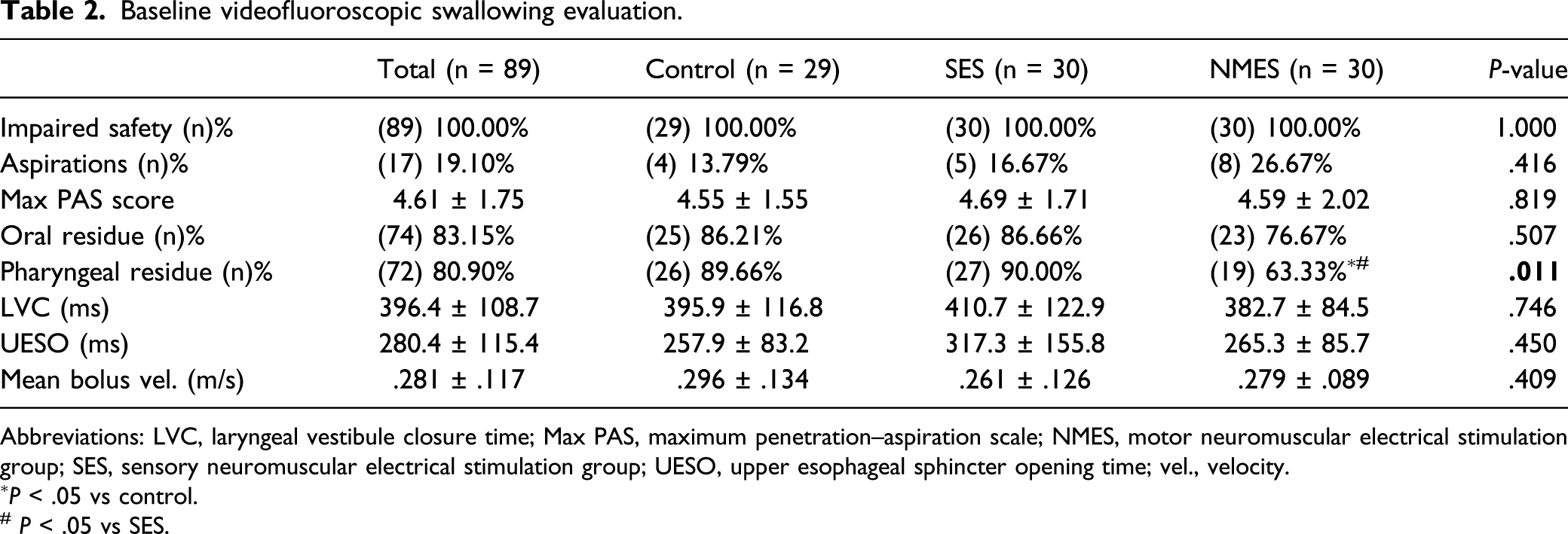
Abbreviations: LVC, laryngeal vestibule closure time; Max PAS, maximum penetration–aspiration scale; NMES, motor neuromuscular electrical stimulation group; SES, sensory neuromuscular electrical stimulation group; UESO, upper esophageal sphincter opening time; vel., velocity.
∗P < .05 vs control.
# P < .05 vs SES.
Treatment 1 Characteristics
Treatment Intensity in SES and NMES
Current intensity applied in the first treatment session in the SES group was 7.47 ± 3.73 mA and at the end (15 sessions) was 9.05 ± 4.63 mA (P = .007). In the NMES group, the current intensity applied in the first session was 11.86 ± 5.11 mA ending with 15.14 ± 5.72 mA (P = .151). Initial intensity of the NMES group was significantly higher than that of the SES group (P < .0001).
Short-Term Effect of the TES Treatment 1 (Baseline VFS vs Post-treatment 1 VFS)
VFS
After the first period of treatment, we found similar improvements in VFS signs of impaired safety of swallow in both active TES treatment groups with a reduction in prevalence of patients with unsafe swallows from 100.0% to 86.67% in the SES group (P = .112) and from 100.0% to 80.0% in the NMES group (P = .024). In contrast, we did not find differences in the control group (100% unsafe swallows). The PAS score was significantly reduced from 4.69 ± 1.71 to 3.38 ± 1.90 (P < .01) in the SES group and from 4.59 ± 2.02 to 3.76 ± 2.31 (P < .05) in the NMES group, and remained unchanged (from 4.55 ± 1.55 to 4.51 ± 1.68; ns) in the control group. No changes in the prevalence of signs of impaired efficacy of swallow were observed in any group.
Prevalence of patients from the SES group that could be safely hydrated with liquid viscosity was increased from 3.3% to 37.9% (P < .001) in SES and from 10.0% to 34.5% (P = .02) in the NMES group. We also found a decrease in the prevalence of patients that had to be hydrated at pudding viscosity in the SES group (from 43.3% to 17.2%; P = .024), and in the NMES group (from 50.0% to 27.6%; P = .063). In contrast, the need of thickening agents in the control group remained unchanged over the time period (liquid 13.8%; nectar from 41.4% to 44.8%; and pudding from 44.8% to 41.4%).
OSR
We also found a significant reduction in the time to LVC in both active TES groups from 410.70 ± 122.90 ms to 353.10 ± 128.80 ms (P < .01) in the SES group and from 382.70 ± 84.48 ms to 310.30 ± 73.02 ms (P < .001) in the NMES group. This improvement on swallow physiology was not observed in the control group (395.90 ± 116.80 ms to 375.20 ± 126.20 ms; ns). We also found some improvements in the UESO time, but only in the SES group (317.3 ± 155.8 ms to 262.10 ± 102.30 ms; P < .05). No significant changes were found in the mean bolus velocity nor in the bolus propulsion forces in any of the study groups.
Long-Term Effect of the TES Treatments T1 + T2 (Baseline vs 1-year Follow-up VFS)
VFS
1-year follow-up videofluoroscopic swallowing evaluation.
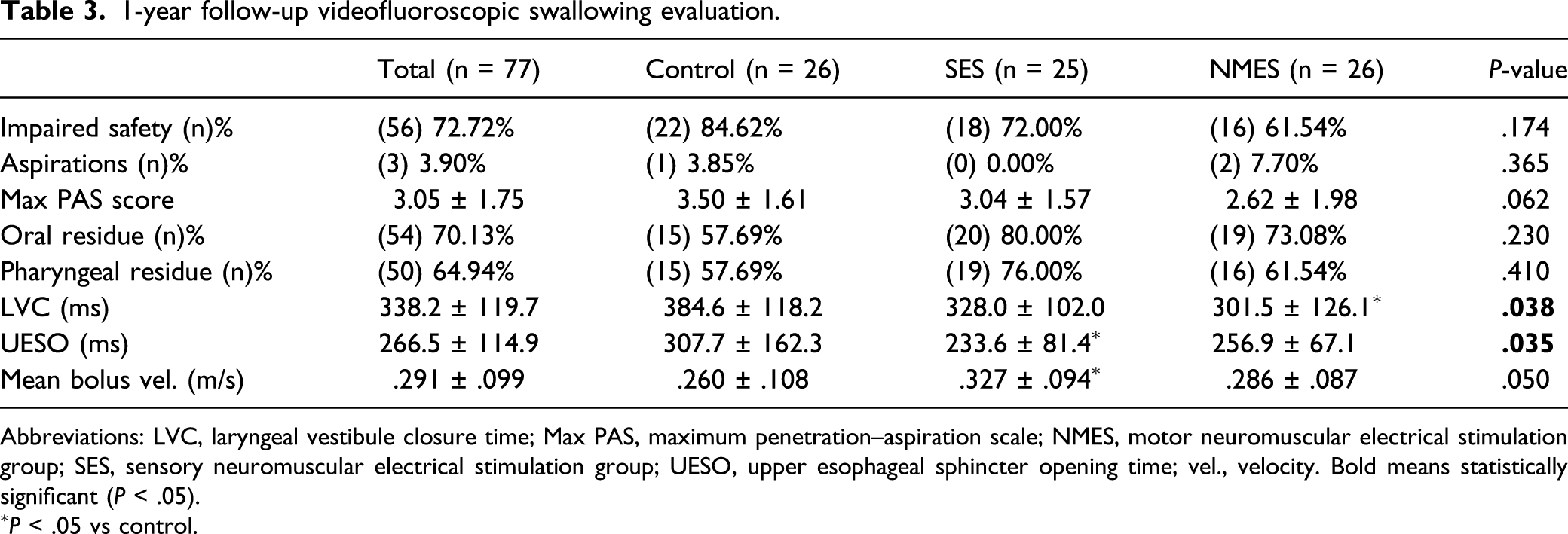
Abbreviations: LVC, laryngeal vestibule closure time; Max PAS, maximum penetration–aspiration scale; NMES, motor neuromuscular electrical stimulation group; SES, sensory neuromuscular electrical stimulation group; UESO, upper esophageal sphincter opening time; vel., velocity. Bold means statistically significant (P < .05).
∗P < .05 vs control.
At 1-year follow-up, we found a significant increase in the percentage of patients that could drink liquid viscosity safely. Only 3.33% of the patients from the SES group were able to be safely hydrated with liquid viscosity in baseline VFS, but after the treatments and 1-year follow-up period, this percentage increased to 52.0% (P < .001). Similarly, in the NMES group, there was a significant increase in prevalence of patients that could swallow liquid viscosity safely (from 10% to 61.5%; P < .001). Finally, we found a decrease in the percentage of patients that had to be hydrated with pudding viscosity in the SES group (from 43.3% to 32.0%; P = .389) that was significant in the NMES group (from 50% to 11.5%; P < .01). In contrast, the need for thickening agents in the control group remained stable over the 1-year follow-up ending up with 30.8%, 34.6%, and 34.6% of patients being able to be hydrated with liquid, nectar, and pudding viscosity, respectively. At the end of the study, 1 year following inclusion, none of the patients needed feeding tubes.
OSR
Time to LVC was greatly reduced in both active groups from 410.7 ± 122.9 ms to 328.0 ± 102.0 ms (P = .01) in the SES group and from 382.7 ± 84.48 ms to 301.5 ± 126.1 ms (P = .004) in the NMES group but not in the control arm of the study (395.9 ± 116.8 ms vs 384.6 ± 118.2 ms; P = .870). We also found some improvements in time to UESO but only in the SES group (317.3 ± 155.8 ms to 233.6 ± 81.4 ms; P = .062). No significant changes were found in the mean bolus velocity between baseline and 1-year follow-up in any of the study groups.
Clinical Outcomes
Clinical outcomes evolution between baseline visit (V1) and final visit (12 months).
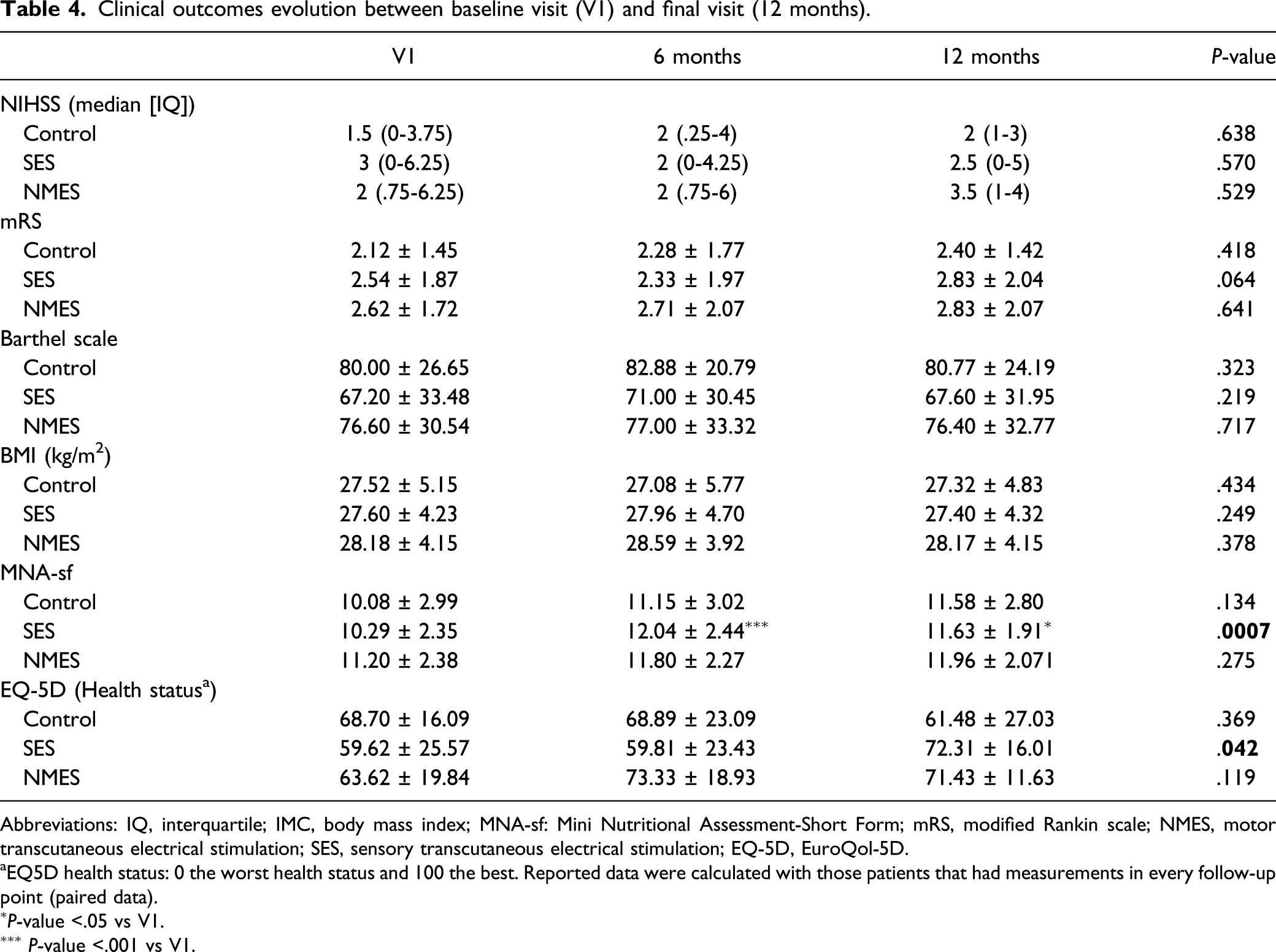
Abbreviations: IQ, interquartile; IMC, body mass index; MNA-sf: Mini Nutritional Assessment-Short Form; mRS, modified Rankin scale; NMES, motor transcutaneous electrical stimulation; SES, sensory transcutaneous electrical stimulation; EQ-5D, EuroQol-5D.
aEQ5D health status: 0 the worst health status and 100 the best. Reported data were calculated with those patients that had measurements in every follow-up point (paired data).
∗P-value <.05 vs V1.
∗∗∗ P-value <.001 vs V1.
Adverse Events (T1 and T2)
Over the study, 36% of patients with CPSOD had at least 1 AE. In total, there were 50 AEs of which 48.0% were in patients from the control group, 22.0% in the SES group, and 30.0% in the NMES group (P = .019). Of the 50 AEs, only 4% were directly related to the treatment where 2 patients had skin irritation in the electrode zone: 1 NMES patient (first treatment session) and 1 SES patient (second treatment session). Nevertheless, they finished treatment without major discomfort and without requiring any intervention. Most prevalent AEs were respiratory infections without pneumonia (12%), falls (10%), gastrointestinal problems (8%), bronchitis (6%), musculoskeletal pain (6%), and urinary infection (6%). A table with the AEs divided by study groups has been added as (Supplementary Material).
Discussion
In this RCT, we found that TES is a safe and effective therapy for elderly patients with CPSOD and that the therapeutic effect starts immediately after treatment and remained for one year. Both SES and NMES greatly improved the safety of swallow in these patients through significant improvements in the biomechanics of the swallow response, mainly reducing time to LVC and dramatically reducing the need for fluid thickening to maintain a safe swallow. However, we did not find any significant difference when performing intergroup comparisons between both TES strategies on safety and efficacy of swallow in poststroke patients. On the other hand, the control group, treated only with compensatory strategies, also showed some significant but weaker improvement in the severity of OD according to the PAS, but it was not associated with any improvement in the swallow function or in the prevalence of patients with CPSOD with unsafe swallows. Regarding clinical outcomes, we only found some specific improvements in nutritional status and quality of life in the SES group. Finally, no significant related AEs or SAEs were found during the 1-year follow-up of this study for any of the TES groups, showing that TES therapy is safe.
Chronic poststroke oropharyngeal dysphagia population is a phenotype of patients growing in Europe, due to the increasing incidence of stroke events (from 1.1 million/year in 2000 to an estimated 1.5 million/year in 2025), high prevalence of stroke survivors and of those with OD among them (50–81%), and even among those with mild strokes (45%). 35 At baseline, our CPSOD study population had advanced age, low disability scoring related to stroke severity and mild impaired functionality related to stroke (mRS) on recruitment (chronic stage), and severe swallowing dysfunction with high prevalence of aspirations. They also presented an impaired general functional status and a high prevalence of comorbidities, the majority of them were malnourished or at risk of malnutrition, and their health status was quite poor according to EQ5D. We defined this population as elderly people who survived to a mild stroke and developed CPSOD. These clinical characteristics are quite similar to those presented by elderly patients with CPSOD in our previous studies2,3,9 as we and others have found that stroke survivors are at high risk of malnutrition and respiratory infections, both closely associated with OD.
The standard of care for the majority of these older CPSOD in Europe is very poor as 80% of them are not diagnosed and do not receive any treatment for OD. For many years, dysphagia therapy for CPSOD has been focused on compensatory strategies including fluid thickening, texture-modified foods, and postures/maneuvers. 9 These strategies improve safety of swallow but not the impaired swallow biomechanics nor the damaged neural swallow networks. 36 Using a similar compensatory strategy as the one used in this study, we improved nutritional and functional status and reduced hospital readmissions, respiratory infections, and mortality in a cohort of older patients with OD. 11 Advances in CPSOD treatments include improvements in these compensatory strategies but are mainly focused on peripheral and central, noninvasive stimulation strategies. Treatment therefore is changing from compensation to promoting brain plasticity, both to recover swallow function and to improve brain-related swallowing dysfunction, 36 and to our knowledge, this is the first RCT with an active treatment (TES) for CPSOD and one-year follow-up showing a strong positive therapeutic effect on swallowing function. This new piece of information covers some of the needs claimed by NICE guidelines on needs of retreatment and long-term outcomes.16,17 In this study, we used two specific strategies combining electrode positions with two specific intensity current parameters of TES previously assayed in our pilot studies. 18 On the one hand, SES aims to increase the sensory input to the pharyngeal sensory afferents to promote cortical plasticity, 14 although there is little evidence in experimental data showing this promotion, and, on the other hand, we used NMES to stimulate the efferent pathways to improve the mechanics of LV closure and UESO. 18 We recently found that similar older patients with CPSOD presented very severe impairments in both sensory and motor pathways involved in swallow function, further supporting our therapeutic approach using TES in the present study. 6
In our control group, we found no improvements in the prevalence of patients with impairments in safety of swallow in VFS; however, we found a mild improvement in the PAS score not associated with any change in the biomechanics of OSR (neither in the time to LVC nor to UESO) as happened with the TES groups. This further suggests that TES could modulate higher-level central nervous system mechanisms such as neuroplasticity,14,15,18 with no significant differences in this study between SES and NMES, but with strong significant differences between both TES groups and the control group. Our approach using SES is quite new as most studies are focused in NMES. Neuroplasticity is thought to be related to the activation of peripheral sensory nerves leading to cortical activation and facilitation of deglutition and airway protection.18,37 We have recently found significant improvements in both the afferent or efferent circuits, measured with pharyngeal sensory and motor evoked potentials, following sensory stimulation strategies in patients with CPSOD. 38 The similar effects of SES and NMES in our study also highlight the role of the sensory afferent pathway as a key therapeutic target in neurorehabilitation strategies for CPSOD. The information carried through the pharyngeal sensory pathway is essential for repeated coupling with motor control modulation of swallowing muscles through the whole process of deglutition. 36 All these positive results open up the possibility of developing new studies with peripheral stimulation strategies such as TES including neurophysiological evaluations of both motor and sensory pathways to better understand their specific mechanisms of action.
According to NICE, current evidence on effectivity of TES in stroke patients is limited in quality and quantity. 16 A recent systematic review and meta-analysis on the comparative efficacy of 4 noninvasive neurostimulation therapies (repetitive transcranial magnetic stimulation -rTMS-, transcranial direct current stimulation -tDCS-, TES, and PES) for stroke patients with OD found that TES is effective for acute and subacute patients. In this review, they remarked on the favorable cost and feasibility of TES compared with rTMS and tDCS and found that PES showed no beneficial effects compared with sham stimulation. 37 Another meta-analysis concluded that TES was more effective than traditional therapy (combination of diet modification, position adjustment, effortful swallowing, and training) for OD adult patients of several etiologies except for patients with CPSOD in which TES and traditional therapy showed similar results. 39 We previously found positive therapeutic effects of TES in two subacute studies with poststroke and older patients with OD. After 10 days stimulation with TES at SES or NMES configuration, there were significant improvements in safety of swallow and an improvement of the OSR.18,32 Taken all this information together with our results, we believe our study will help NICE to complete their guidance on TES for CPSOD as this guideline only includes information on short-term or subacute studies, and our study also shows the positive results of a long-term RCT with 1-year follow-up.
Our study presents some limitations; first of all, our population is mainly constituted by older stroke survivors with low stroke severity according to the NIHSS, but this is the usual population that is admitted at our hospital (general hospital) as can be found in our previous publications.2,3,6,8,9,18,38 However, despite this low stroke severity, our patients had severely reduced functional capacity and severe chronic swallowing impairments as shown by the mean PAS score with a highly affected swallow response (delayed LVC) although none of them were fed by feeding tubes. Another limitation is that we did not collect data on compliance with the compensatory treatment in patients’ daily life, although this was not an objective of our study. Then, our criteria to define impaired safety of swallow (PAS ≥ 2) could be interpreted as little restrictive and an indicator of a population with presbyphagia; however, this criteria has been used in our previous publications6,38 and finally our participants had a severe dysphagia (mean PAS = 4.61 ± 1.75; severe delayed time to LVC = 396.4 ± 108.7) with most patients (86.5%) with a mean PAS > 3 at baseline VFS evaluation. Also, our VFS algorithm only included one swallow per bolus volume and viscosity and this might not represent the intraindividual variability across the swallowing evaluation; however, an algorithm including multiple swallows per volume and viscosity could increase patient’s fatigue and radiation exposure. Ultimately, the retreatment component of the trial is complicated as although the majority of the patients were retreated at 6 months, showing the need of this booster treatment to achieve an optimal result, some of them were not because they already presented a safe swallow and thus, there were some patients with 1 treatment cycle and others with 2 cycles. However, the control group did not receive a booster treatment of their compensatory care at 6 months. This should be taken into account when interpreting data and future studies with larger sample size are required to check differences between these subgroups of patients.
Finally, our study adds evidence for the inclusion of TES in the management guidelines of CPSOD in clinical practice. NICE guidelines concluded that further research on TES for OD in adults should address patient selection, variations in technique, the need for retreatments, and long-term outcomes. 16 In our study, we provide new evidence on two treatment configurations, the need for retreatment in a subgroup of patients, and the long-term clinical outcomes, as well as the long-term effect of the therapy in elderly patients with CPSOD. With this new piece of evidence, we propose to move a step forward from research to clinical practice for the management of CPSOD by using TES in the specific configurations evaluated and by improving the current compensatory treatment with new evidence including fluid adaptation with new xanthan gum–based agents, 35 intensive nutritional support with texture modified diets and caloric and protein supplementation, 40 and comprehensive oral health care. 11 In addition, our study opens a door to the evaluation of similar protocols of TES in more severe stroke survivors with OD such are those needing feeding tubes or tracheostomies.
Summary/Conclusions
In conclusion, TES is a safe and effective therapy for elderly CPSOD. Both SES and NMES greatly improved the safety of swallow and the biomechanics of swallow response and reduced the need for fluid thickening in CPSOD. The effects started immediately after the first treatment series and lasted for one year, thus TES is a safe and effective therapy to move treatments from compensation to the recovery of swallowing function in elderly patients with PSOD. However, a large multicenter RCT is recommended to further confirm whether TES is effective in treating poststroke dysphagia and to explore their potential effects on brain plasticity.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211023187 – Supplemental Material for Effect of Transcutaneous Electrical Stimulation in Chronic Poststroke Patients with Oropharyngeal Dysphagia: 1-Year Results of a Randomized Controlled Trial
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211023187 for Effect of Transcutaneous Electrical Stimulation in Chronic Poststroke Patients with Oropharyngeal Dysphagia: 1-Year Results of a Randomized Controlled Trial by Viridiana Arreola, Omar Ortega, Daniel Álvarez-Berdugo, Laia Rofes, Noemí Tomsen, Christopher Cabib, Desiree Muriana, Elisabet Palomera and Pere Clavé in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We would like to thank Jane Lewis and Laura Chercoles for English editing assistance and Dr Ernest Palomeras and Dr Nicolau Guanyabens from the Neurology Department of the Hospital de Mataró. This work has been conducted within the framework of a doctoral thesis in the Department of Surgery at the Autonomous University of Barcelona.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research grant from DJO Global. The sponsor had no role in the development of this study. Financial support was also received by grants from Fondo de Investigaciones Sanitarias, Instituto de Salud Carlos III (14/00453, 18/00241); Proyectos de Investigación Clínica Independiente, Instituto de Salud Carlos III (ICI20/00117); CIBERehd, Instituto de Salud Carlos III; and Programa de Estabilización de Investigadores y de Intensificación de la Actividad Investigadora en el Sistema Nacional de Salud (INT15/00026, INT16/00111).
Supplementary Material
Supplementary material for this article is available online on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
