Abstract
Background
Since people with Parkinson disease (PD) rely on limited prefrontal executive resources for the control of gait, interventions targeting the prefrontal cortex (PFC) may help in managing PD-related gait impairments. Transcranial direct current stimulation (tDCS) can be used to modulate PFC excitability and improve prefrontal cognitive functions and gait.
Objective
We investigated the effects of adding anodal tDCS applied over the PFC to a session of aerobic exercise on gait, cognition, and PFC activity while walking in people with PD.
Methods
A total of 20 people with PD participated in this randomized, double-blinded, sham-controlled crossover study. Participants attended two 30-minute sessions of aerobic exercise (cycling at moderate intensity) combined with different tDCS conditions (active- or sham-tDCS), 1 week apart. The order of sessions was counterbalanced across the sample. Anodal tDCS (2 mA for 20 minutes [active-tDCS] or 10 s [sham-tDCS]) targeted the PFC in the most affected hemisphere. Spatiotemporal gait parameters, cognitive functions, and PFC activity while walking were assessed before and immediately after each session.
Results
Compared with the pre-assessment, participants decreased step time variability (effect size: −0.4), shortened simple and choice reaction times (effect sizes: −0.73 and −0.57, respectively), and increased PFC activity in the stimulated hemisphere while walking (effect size: 0.54) only after aerobic exercise + active-tDCS.
Conclusion
The addition of anodal tDCS over the PFC to a session of aerobic exercise led to immediate positive effects on gait variability, processing speed, and executive control of walking in people with PD.
Introduction
People with Parkinson disease (PD) present gait impairments that lead to increased risk of falls and reduced independence.1,2 Gait patterns in PD include short, wide, and asymmetric steps; slow gait speed; and increased step-to-step variability. 3 Because of degeneration in neural networks associated with movement automaticity, people with PD rely on compensatory prefrontal executive-attentional resources for the control of gait, even during usual walking.4-9 This is particularly problematic because executive processes are slow and limited in capacity, 10 and people with PD often present cognitive impairments, including reduced executive function and attention and slow processing speed. 11 Such cognitive impairments may limit the efficiency of the compensatory executive control of gait in PD. 12 Indeed, executive dysfunction is associated with PD-related gait impairments. 13 Because levodopa improves only certain aspects of gait, 3 complementary therapies are needed to manage gait impairments in PD. The body of evidence presented above supports the idea that the prefrontal cortex (PFC) should be targeted for the development of enhanced interventions for gait in PD.
There is growing interest in using aerobic exercise and transcranial direct current stimulation (tDCS) for gait rehabilitation in PD. Aerobic exercise has been shown to acutely increase PFC activity14,15 and improve spatiotemporal gait parameters and prefrontal cognitive function in PD.16-18 It is possible that aerobic exercise enhances the compensatory prefrontal executive mechanism used for the control of gait in PD. tDCS is a low-cost method of noninvasive brain stimulation that directs, through scalp electrodes, a constant low-amplitude electric current, which modulates excitability in both cortical 19 and subcortical brain areas.20,21 A single session of anodal tDCS applied over the PFC has been shown to improve functional mobility and working memory in PD.22,23 Improved executive function and reduced freezing of gait have also been reported after simultaneous stimulation of the PFC and primary motor cortex, but not after primary motor cortex only or sham stimulation in people with PD.22-24 Proposed action mechanisms of anodal tDCS include increased neuronal excitability and modulation of oxygen supply to cortical and subcortical areas 25 and neuronal synapsis strength. 26 Additionally, anodal tDCS has been shown to increase resting-state PFC activity in young adults. 27 Thus, aerobic exercise and tDCS may contribute to the development of enhanced gait interventions in PD.
Our recent systematic review demonstrated that additional clinical benefits are observed in people with PD when anodal tDCS is combined with physical interventions, including aerobic exercise. 28 However, to the best of our knowledge, the responses of PFC activity while walking and gait parameters to a combined session of aerobic exercise and anodal tDCS applied over the PFC have not been investigated yet. Since people with PD rely on prefrontal executive-attentional resources to control gait,4-9 assessment of PFC activity while walking in response to such combined intervention is necessary to advance our understanding on potential cortical mechanisms underlying gait improvements. Thus, the aim of this study was to investigate the effects of adding anodal tDCS applied over the PFC to a session of aerobic exercise on gait, cognition, and PFC activity while walking in people with PD. We hypothesized that the combination of aerobic exercise with anodal tDCS would promote superior benefits to gait and cognition compared with those of stand-alone aerobic exercise (sham tDCS). Specifically, because anodal tDCS increases neuronal excitability, we expected that the combined session would expand the capacity of the compensatory prefrontal executive mechanism (i.e., increased PFC activation while walking) used for the control of gait in PD.
Methods
Participants
A total of 20 people with mild to moderate (stages 1 to 3 in the Hoehn and Yahr Rating Scale) idiopathic PD participated in this randomized, double-blinded, sham-controlled crossover study. Exclusion criteria were presence of neural implants, use of a pacemaker, history of seizures, disabling musculoskeletal problems, and cognitive decline (Mini Mental State Examination [MMSE] 29 score < 24). This study was approved by the local research ethics committee. All participants provided written informed consent prior to participation, which involved 3 visits to the lab.
Procedures
All visits were carried out with participants in the state “on” of medication, approximately 45 to 60 minutes after the ingestion of PD medication. In the first visit, the following demographic, clinical, and cognitive characteristics were obtained. MMSE was used to assess global cognitive function. 29 The Hospital Anxiety and Depression Scale (HADS) was used to assess anxiety and depressive symptoms. 30 The Movement Disorders Society-United Parkinson’s Disease Rating Scale motor part (MDS-UPDRS-III) was used to assess disease severity. 31 The levodopa equivalent daily dose (LED) was calculated according to Tomlinson’s suggestions. 32
Because PD is characterized by asymmetric neuronal loss (with increased inhibitory GABAergic signaling from the basal ganglia to other encephalic regions, including the brain cortex), we chose to apply anodal tDCS over the most affected hemisphere. 33 Anodal tDCS has been shown to increase extracellular dopamine levels in the striatum 34 and inhibit GABAergic neurons.35,36 We identified the most affected cerebral hemisphere using items 3.3 to 3.8 and 3.15 to 3.17 of MDS-UPDRS-III. The most affected cerebral hemisphere was defined as contralateral to the most affected body side (i.e., more severe scores in the aforementioned MDS-UPDRS-III items).
The second and third visits were 2 sessions of aerobic exercise combined with different tDCS conditions (active or sham anodal tDCS). These visits were 1 week apart, and the order of the tDCS condition was counterbalanced across the sample. Participants were randomized in a 1:1 ratio to start with either active or sham tDCS. After 1 week, participants were crossed over—that is, participants who received sham-tDCS in the second visit received active-tDCS in the third visit, and vice versa (Figure 1A).
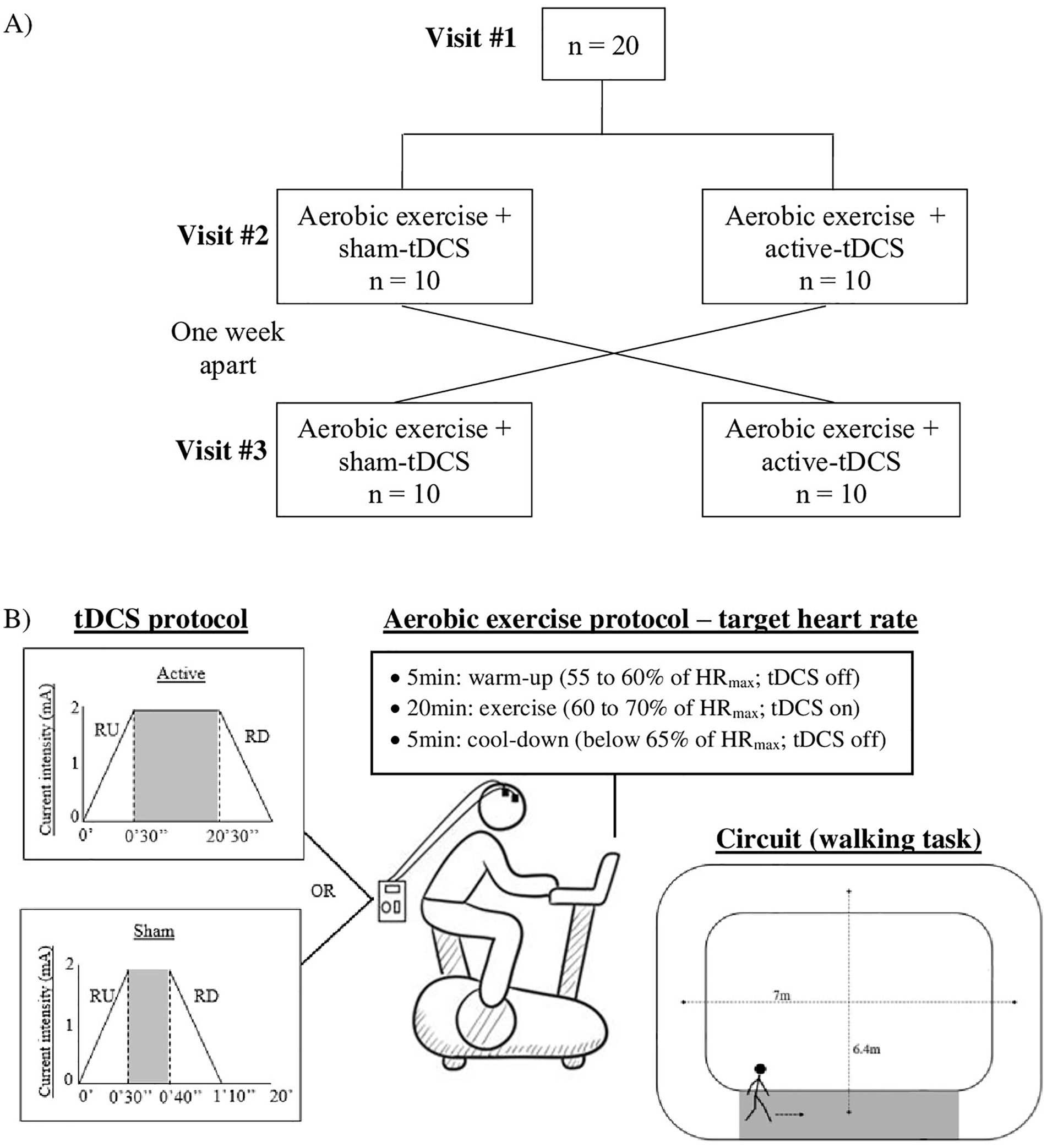
Flow diagram (A) and illustration of the intervention protocol and circuit used for walking task (B). Both tDCS protocols (sham and active) included 30-s periods of RU/RD. The stimulation current of 2 mA was applied for 20 minutes and 10 s in the active- and sham-tDCS sessions, respectively.
Aerobic Exercise and Anodal tDCS
Aerobic exercise was performed on a recumbent stationary bike (model EB597E, Uniforce Fitness, Brazil). Exercise intensity was moderate and personalized by the individual’s maximum heart rate (HRmax). HRmax was estimated using the following formulas: HRmax = 208 − (0.7 × Age) 37 and HRmax = 164 − (0.7 × Age) for participants using β-blocker medication. 38 The aerobic exercise protocol lasted 30 minutes and consisted of 3 stages: (1) 5-minute warm-up, with HR maintained between 55% and 60% of HRmax (tDCS off); (2) 20 minutes of exercise, with intensity between 60% and 70% of HRmax (combined with active-tDCS or sham-tDCS); (3) 5-minute cooldown with HR kept below 65% of HRmax (tDCS off). HR was recorded using a cardiac monitor (model V800 POLAR) during the entire session. Of note, moderate aerobic exercise performed on a recumbent bike has been shown to increase PFC activity. 15 In the central 20 minutes of aerobic exercise, tDCS was delivered by a battery-driven stimulator (Microestim GENIUS, NKL Electronics Products LTDA EPP, Santa Catarina, Brazil) through a pair of saline-soaked sponge electrodes (7 cm × 5 cm). Stimulation targeted the dorsolateral PFC of the most affected cerebral hemisphere. The anodal electrode was placed over the F3/F4 position of the 10-20 international electroencephalography system. The cathodal electrode was positioned on the contralateral supraorbital region (FP2 or FP1). In the active-tDCS session, the stimulation current of 2 mA was applied for 20 minutes and was ramped up at the start and ramped down at the end of the stimulation over a period of 30 s. In the sham-tDCS session, current was applied for only 10 s between the ramp-up/-down periods (Figure 1B). A structured questionnaire applied immediately after the end of the session assessed potential adverse effects of tDCS and participants’ guesses of the tDCS condition. 39
Cognitive and gait assessments (described below), which included measurements of PFC activity while walking, were carried out before (pre-intervention) and after (post-intervention) each intervention session. Post-intervention assessments started 10 minutes after the end of the intervention. This interval was adopted to minimize/exclude the effect of increased blood flow resulting from exercise on the functional near-infrared spectroscopy (fNIRS) signal, which has been reported to last approximately 8 minutes. 40
Cognitive Assessments
Global cognition was assessed with the Montreal Cognitive Assessment (MoCA), which includes questions in several cognitive domains; MoCA scores range from 0 to 30, and higher scores correspond to better global cognition. 41 Executive function was measured with the Trail Making Test. The participant is required to connect, as quickly and accurately as possible, a sequence of 25 consecutive targets/dots printed on a sheet of paper, using a hand-drawn pen. The Trail Making Test consists of 2 parts: in part A, the targets to be connected are all numbers (from 1 to 25); in part B, the targets combine numbers and letters and the participant must alternate between them (1, A, 2, B, 3, C, etc). The time taken to complete each part was recorded using a manual stopwatch. 42 Simple and choice reaction time were assessed using customized tests implemented in MATLAB (The MathWorks, Inc, Natick, Massachusetts, United States); they provide information about processing speed. In the simple reaction time test, participants were instructed to press the spacebar as quickly as possible after the presentation of a stimulus (i.e., appearance of a green rectangle in the center of the computer screen). In the choice reaction time test, 2 stimuli (and corresponding responses) were possible: the green rectangle could be presented in the left- or right-hand side of the screen. Participants were instructed to press a key on the left-hand side of the keyboard when the stimulus was displayed on the left-hand side of the screen and a key on the right-hand side of the keyboard when the stimulus was displayed on the right-hand side of the screen. The presentation side of the stimuli in choice reaction time test was random. In all, 10 trials were performed for each reaction time test. The 5 best nonanticipated trials (out of 10 trials) were averaged for each participant in each test.
Gait Assessment
Initially, participants were prepared for fNIRS recordings. A portable fNIRS system (OctaMon fNIRS system, Artinis Medical Systems, Netherlands) was used to measure changes in oxygenated (HbO2) and deoxygenated (HHb) hemoglobin concentrations in the PFC. It uses continuous wave diodes with wavelengths of 760 and 850 nm and interoptode distance of 35 mm. Data were recorded at a frequency of 10 Hz. An 8-channel arrangement with 10 optodes, consisting of 8 transmitters and 2 detectors (4 transmitters and 1 detector per hemisphere), covered Brodmann areas 9, 10, and 46 in both right and left hemispheres. After the Cz and Fpz positions were determined on the participant’s head, a neoprene head-cap (fiber holder) marked with labels of the international 10-20 electroencephalography system and predetermined locations for the optodes was placed on the participant’s head. The head-cap was adjusted so that the Cz and Fpz positions matched the Cz and Fpz marks on the participant’s head. Procedures for head-cap positioning were similar in visits 2 and 3.
For overground walking, participants performed 5 trials. The participants walked at their normal and comfortable pace on a cone circuit (total distance = 26.8 m) with 2 parallel 7-m long straights. 7 Each trial started with 30 s of standing still, followed by a “ready-go” instruction and 30 s of walking. During the standing still period, participants were instructed to count forward mentally to avoid/minimize mind wandering during the fNIRS baseline period. A 5.74-m long electronic walkway with pressure sensors (GAITRite, CIR Systems Inc, Sparta) was positioned in the center of one of the straights to record spatiotemporal gait measures (Figure 1B). All steps registered by the system were considered for the analysis. Means and variability (SDs) of step length, step width, step time, step velocity, and swing and double support phases were calculated.
Functional Near Infrared Spectroscopy Analysis
The fNIRS system measured optical density of the raw signal and converted this to HbO2 and HHb using the modified Beer-Lambert law in the Oxysoft software (Artinis, Netherlands). The differential path factor was set to 6.61 for all participants. Artifact corrections and filtering were performed according to recent recommendations for the processing of fNIRS signals.43,44
fNIRS data were preprocessed using the time series analysis routine available in the NIRS-SPM software (NIRS-SPM, http://www.nitrc.org/projects/nirs_spm), 45 which included the following steps: (1) filtering, high frequency noise was reduced/removed by a low-pass filter based on canonical hemodynamic response function, 46 and (2) detrending, a wavelet algorithm (wavelet minimum description length detrending algorithm) was used to decompose the signal into global trends, hemodynamic response, and noise components. 47 Then, the preprocessed signal was exported to MATLAB (MATLAB and Statistics Toolbox Release R2015a, The MathWorks, Inc, Natick, MA) for additional processing steps using a custom algorithm. Channels were averaged per region of interest (i.e., stimulated and nonstimulated hemispheres) and normalized by dividing them by the corresponding maximum signal amplitude of the whole experiment. This procedure was used to reduce the differences in amplitude between individuals. 48 The task was divided into 2 periods: rest (10 s of standing still before start of walking, with the exception of the 5 s immediately before the walk, which was excluded from the analysis to avoid anticipatory responses) and task (period between 5 and 25 s after start of walking). The initial 5 s of walking were excluded because of the physiological delay in the hemodynamic response, and the final 5 s of walking were excluded to avoid anticipatory responses to the stop command. The normalized concentrations were averaged over the rest and task periods for each region of interest and each trial. Finally, differences between rest and task periods were calculated to evaluate the relative change in HbO2 and HHb. 44
Statistical Analysis
The Shapiro-Wilk test was used to verify data distribution. Dependent variables that violated the assumption of normality were transformed (log transformation: MoCA, Trail Making Test, step length variability, step time variability, and step width variability; reciprocal transformation: swing time variability and double support time variability). Two-way ANOVAs, with repeated measures for Stimulation ([Aerobic Exercise + Active-tDCS] × [Aerobic exercise + Sham-tDCS]) and Moment (Pre-intervention × Post-intervention), were used to investigate the acute effects of the interventions on outcomes. The level of significance was set at 0.05. Bonferroni post hoc tests (adjusted for multiple comparisons) were used to localize differences when ANOVA revealed significant interactions. Also, the standardized response mean (SRM) was calculated to compare changes from pre-intervention to post-intervention between the intervention sessions; SRM was interpreted as follows: trivial (<0.2), small (>0.2 and <0.5), moderate (>0.5 and <0.8), and large (>0.8). The Wilcoxon test was applied to compare the reported adverse effects and participants’ guesses of tDCS condition between active- and sham-tDCS. Statistical analysis was performed using SPSS 22.0 for Windows (IBM Corporation, Armonk, New York, NY).
Results
All participants enrolled in the study completed the 3 visits. Participants’ characteristics are summarized in Table 1. Overall, participants had mild to moderate disease severity. The Wilcoxon test indicated that there were no significant differences between aerobic exercise + active-tDCS and aerobic exercise + sham-tDCS for adverse effects of tDCS (Table 2). The main sensations reported were mild tingling (60% of sessions) and itching (72.5% of sessions). The Wilcoxon test also revealed no significant difference between the 2 intervention sessions for participants’ guesses of tDCS condition (Table 2).
Demographic, Cognitive, Neuropsychiatric, and Clinical Characteristics of the Sample.
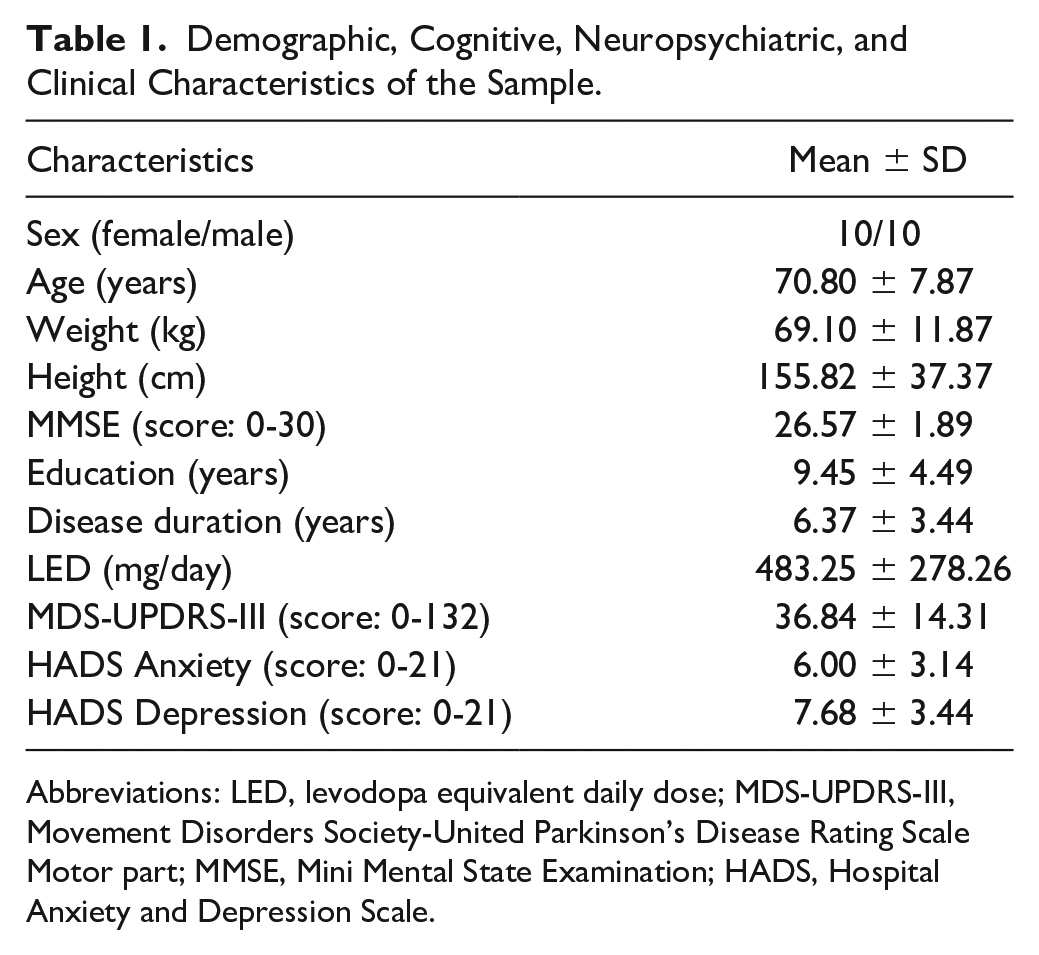
Abbreviations: LED, levodopa equivalent daily dose; MDS-UPDRS-III, Movement Disorders Society-United Parkinson’s Disease Rating Scale Motor part; MMSE, Mini Mental State Examination; HADS, Hospital Anxiety and Depression Scale.
Adverse Effects of tDCS for Each Intervention Session and Participants’ Guesses of tDCS Condition. a
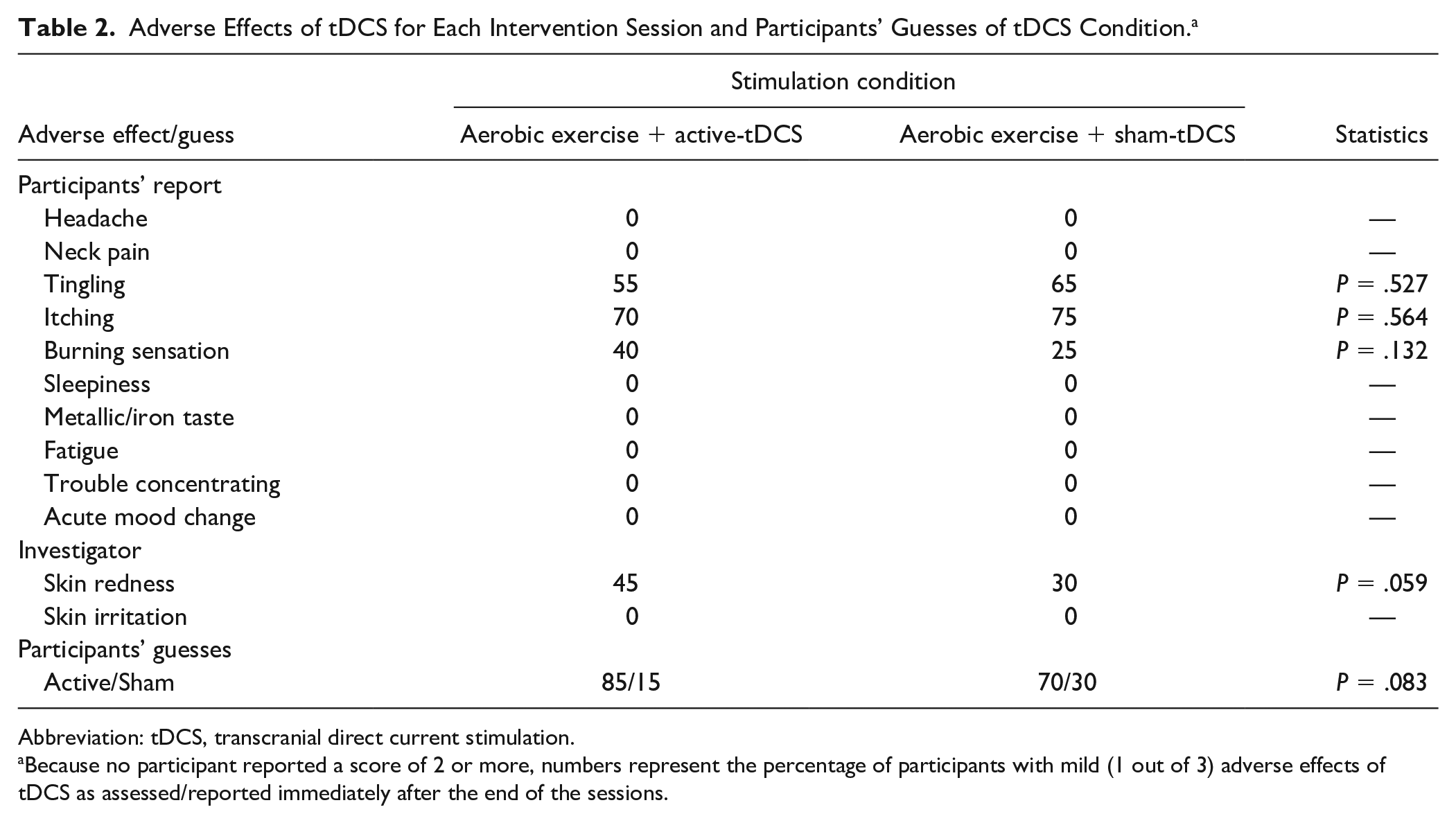
Abbreviation: tDCS, transcranial direct current stimulation.
Because no participant reported a score of 2 or more, numbers represent the percentage of participants with mild (1 out of 3) adverse effects of tDCS as assessed/reported immediately after the end of the sessions.
Cortical activity data of 1 participant were excluded because of excessive noise in the signal. Five participants were unable to complete the Trail Making Test part B (because of understanding difficulties), and therefore, the corresponding data were considered missing. Descriptive and statistical values of all outcome measures are presented in Supplementary Table 1.
Effects on PFC Activation While Walking
Stimulated Hemisphere
ANOVA revealed significant Stimulation × Moment interaction for relative HbO2 levels in the stimulated hemisphere during walking (F1,18 = 4.501; P = .048). Post hoc tests indicated that, compared with the pre-intervention assessment, participants increased relative HbO2 levels in the stimulated hemisphere only after the aerobic exercise + active-tDCS session (P = .031, SRM = 0.54; Figure 2A); no significant change (Pre-intervention × Post-intervention) was observed for aerobic exercise + sham-tDCS (P = .659, SRM = −0.07; Figure 2A). No significant main effects of stimulation or moment were observed for relative HbO2 levels in the stimulated hemisphere during walking.
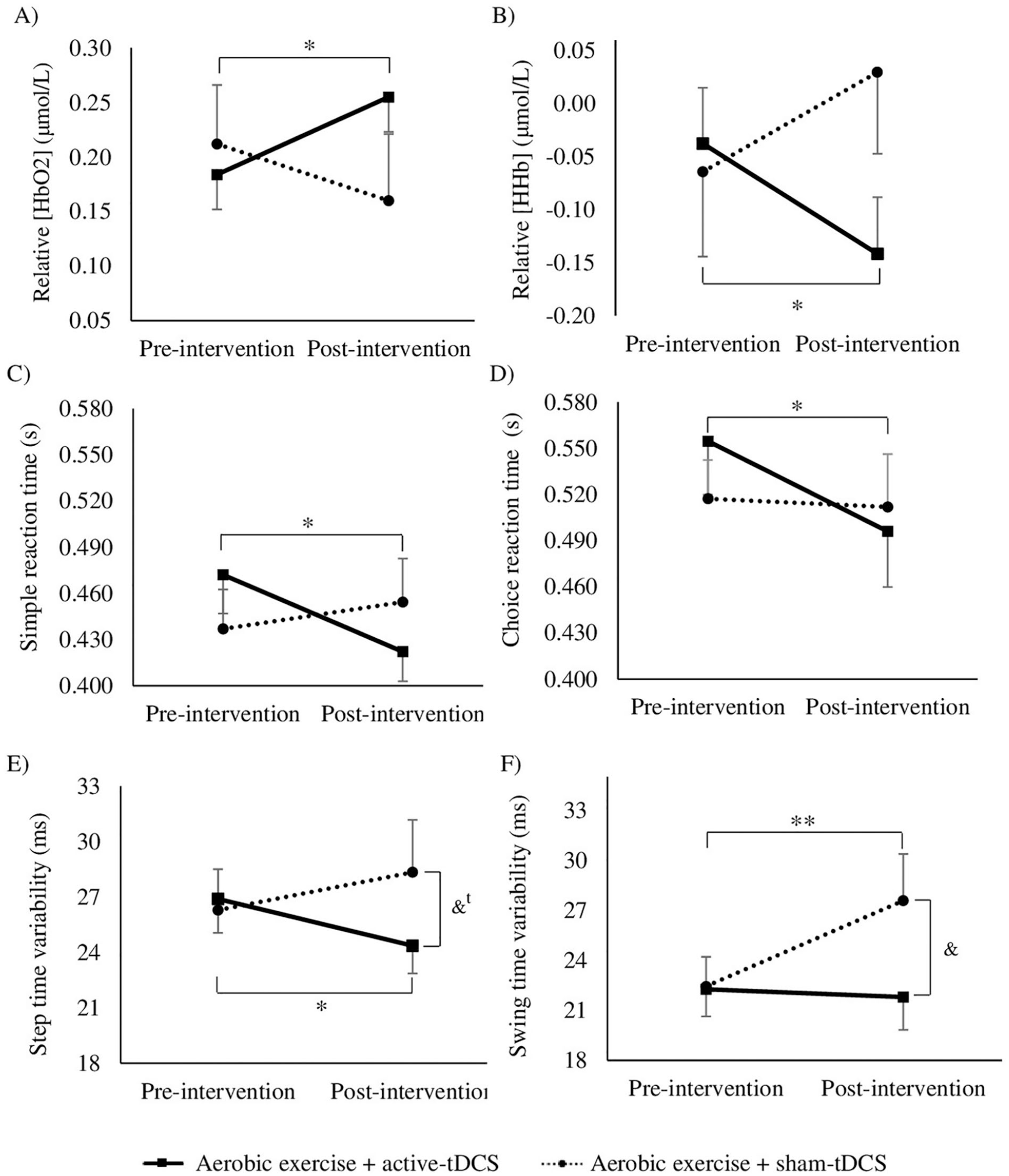
Illustration of stimulation (active- and sham-tDCS) by moment (pre-intervention and post-intervention) interactions. Means and standard errors of relative HbO2 (A) and HHb (B) in the stimulated hemisphere. Means and SDs of simple reaction time (C), choice reaction time (D), step time variability (E), and swing time variability (F).a
ANOVA revealed significant Stimulation × Moment interaction for relative HHb levels in the stimulated hemisphere during walking (F1,18 = 5.192; P = .035). Post hoc tests indicated that, compared with the pre-intervention assessment, participants decreased relative HHb levels in the stimulated hemisphere only after the aerobic exercise + active-tDCS session (P = .032, SRM = −0.53; Figure 2B); no significant changes (Pre-intervention × Post-intervention) were observed for aerobic exercise + sham-tDCS (P = .283, SRM = 0.26; Figure 2B). No significant main effects of stimulation or moment were observed for relative HHb levels in the stimulated hemisphere during walking.
Nonstimulated Hemisphere
No significant findings (i.e., interaction between factors or main effects) were observed for relative HbO2 levels in the nonstimulated hemisphere during walking. For relative HHb levels in the nonstimulated hemisphere during walking, ANOVA indicated that Stimulation × Moment interaction approached statistical significance (F1,18 = 4.294; P = .053; η 2 P = .193). However, post hoc tests revealed no significant changes (Pre-intervention × Post-intervention) for aerobic exercise + active-tDCS (P = .078) or aerobic exercise + sham-tDCS (P = .367).
Effects on Cognition
ANOVA indicated significant Stimulation × Moment interaction for simple reaction time (F1,19 = 5.248; P = .034). Stimulation × Moment interaction approached statistical significance for choice reaction time (F1,19 = 3.711; P = .069), and therefore, we checked the corresponding pairwise comparisons. Post hoc tests indicated that, compared with the pre-intervention assessment, participants decreased simple (P = .004, SRM = −0.73; Figure 2C) and choice reaction times (P = .019, SRM = −0.57; Figure 2D) only after the aerobic exercise + active-tDCS session; for the aerobic exercise + sham-tDCS session, no significant differences between pre-intervention and post-intervention assessments were observed for simple (P = .612, SRM = 0.11; Figure 2C) or choice reaction times (P = .865, SRM = −0.04; Figure 2D).
ANOVA revealed a moment main effect for MoCA score (F1,19 = 8.779, P = .008; Figure 3A), Trail Making Test part A (F1,19 = 37.156, P < .001; Figure 3B), and Trail Making Test part B (F1,13 = 10.423, P = .007; Figure 3C). Participants increased MoCA score and reduced time taken to complete Trail Making Test parts A and B after both intervention sessions.
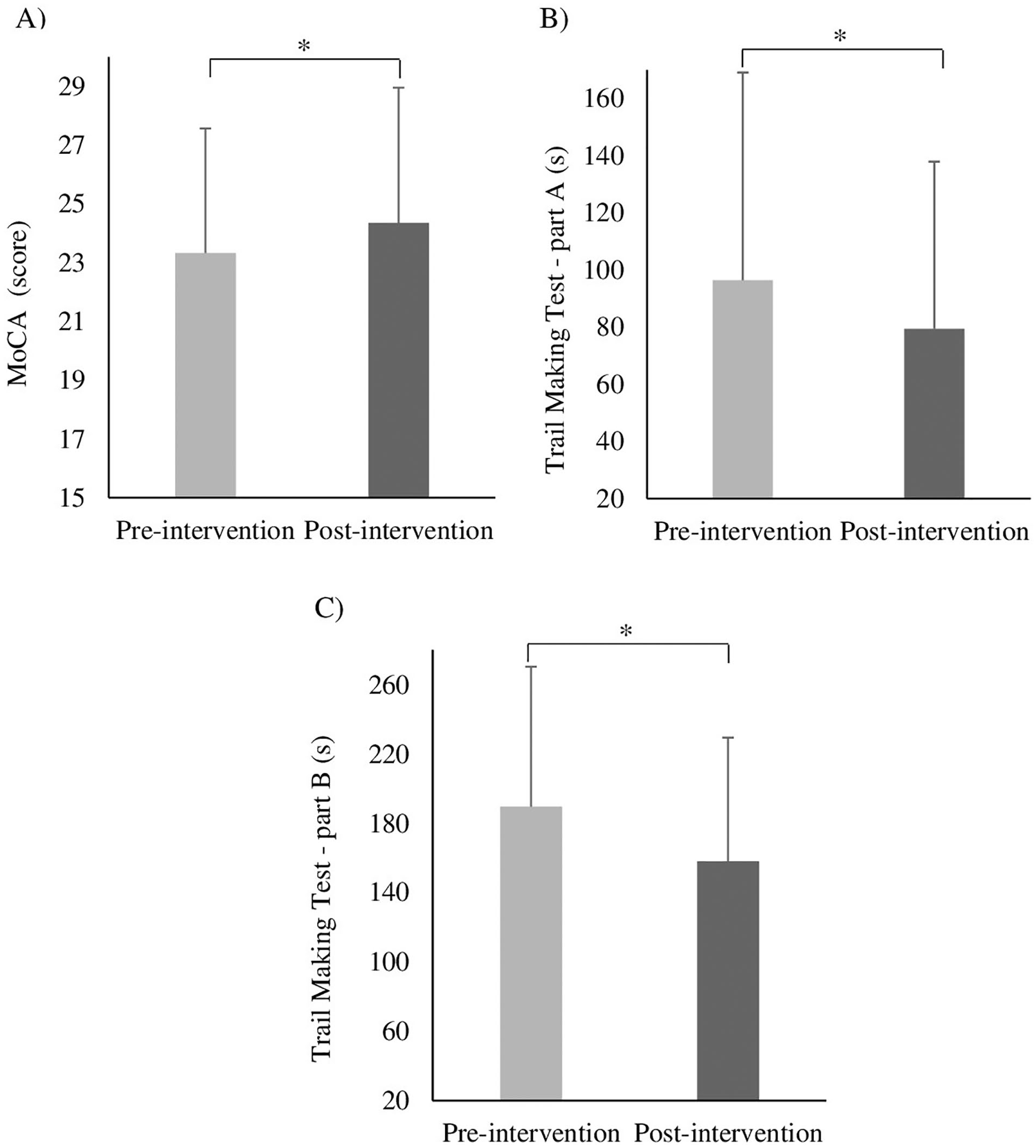
Illustration of moment main effects. Means and SDs of MoCA score (A), Trail Making Test part A (B), Trail Making Test part B (C) for both moments (pre-intervention and post-intervention), regardless of the stimulation condition.
Effects on Gait
ANOVA revealed significant Stimulation × Moment interaction for step time variability (F1,19 = 6.080; P = .023) and swing time variability (F1,19 = 5.105; P = .036). Post hoc tests indicated that, compared with the pre-intervention assessment, participants decreased step time variability (P = .044, SRM = −0.4; Figure 2E) only after the aerobic exercise + active-tDCS session; in the post-intervention assessment, there was a trend for lower step time variability in the aerobic exercise + active-tDCS session compared with the aerobic exercise + sham-tDCS session (P = .063; Figure 2E). Additionally, post hoc tests indicated that, compared with the pre-intervention assessment, participants increased swing time variability (P = .026, SRM = 0.52; Figure 2F) only after the aerobic exercise + sham-tDCS session; in the post-intervention assessment, swing time variability was greater in the aerobic exercise + sham-tDCS session compared with the aerobic exercise + active-tDCS session (P = .042; Figure 2F).
Discussion
This study investigated if the addition of anodal tDCS over the PFC to a session of aerobic exercise would promote greater positive effects on gait, cognition, and PFC activity while walking compared with a stand-alone session of aerobic exercise (sham tDCS) in people with PD. The combined session of aerobic exercise with active anodal tDCS over the PFC had the following effects, which were not observed with the stand-alone session of aerobic exercise: decreased step time variability, shortened simple and choice reaction times (faster responses), and increased PFC activity (increased relative HbO2 levels and decreased relative HHb levels) in the stimulated hemisphere while walking. The corresponding effect sizes ranged from small to moderate. These findings suggest that the addition of anodal tDCS over the PFC to a session of aerobic exercise led to positive effects on gait, processing speed, and executive control of walking in people with PD. Current findings are strengthened by results indicating that our sham protocol effectively blinded participants to the tDCS condition.
The combination of aerobic exercise with anodal tDCS improved the compensatory executive control of gait. Evidence from different research groups suggests that people with PD rely on prefrontal executive-attentional resources for the control of gait,4-7 which is argued to be a compensatory mechanism for impaired motor automaticity. However, the efficiency of such compensatory control mechanism seems to be limited by the pool of executive resources available. 12 In this study, we observed that a single session of aerobic exercise combined with anodal tDCS over PFC increased PFC activity in the stimulated hemisphere during walking and reduced gait variability. These findings suggest that the combined intervention expanded the availability of prefrontal executive-attentional resources to be allocated to the control of walking, leading to a more efficient compensation for impaired movement automaticity. Patients may have been able to recruit additional neurons as a result of increased neuronal excitability promoted by the anodal tDCS. Notably, PFC activity did not increase in the nonstimulated hemisphere after the active-tDCS session, nor with aerobic exercise + sham-tDCS (both hemispheres). Hence, it is possible to interpret that anodal tDCS facilitated the activation of the PFC in the stimulated hemisphere.
The combination of aerobic exercise with anodal tDCS may have modulated cholinergic activity. Acetylcholine is reduced in people with PD compared with healthy individuals,49,50 and it has been shown to be involved in the control of gait. 51 Particularly, increased gait variability is associated with cholinergic dysfunction in PD. 51 Studies assessing the effects of cholinergic augmentation, via pharmacological intervention (i.e., rivastigmine and donepezil) or vagus nerve stimulation, have observed significant reduction of gait variability in people with PD.52,53 Among various gait domains assessed in the current study, only gait variability (i.e., step time variability) responded to the combined intervention. Thus, it is possible to speculate that the addition of anodal tDCS over the PFC to a session of aerobic exercise may have increased cholinergic activity, leading to reduced gait variability. In line with this interpretation, Scelzo et al 54 observed that anodal tDCS increased short latency afferent inhibition of motor-evoked potentials in young adults, suggesting enhancement of cortical cholinergic activity. Specifically, our anodal tDCS protocol may have modulated cholinergic pathways that project from basal forebrain nuclei to the PFC. 51 Future research should examine if such a mechanism, in fact, plays a role when anodal tDCS is applied over the PFC in people with PD.
The combination of aerobic exercise with anodal tDCS over the PFC also improved bradyphrenia. Slower processing speed (as measured by reaction time tests) is often reported in people with PD.55,56 In this study, we demonstrated that the addition of anodal tDCS over the PFC to a session of aerobic exercise promoted faster responses in the simple and choice reaction time tests. These findings suggest that the increased PFC excitability, which is typically promoted by anodal tDCS (but was not measured in the present study), may have facilitated the stimulus identification and/or response to a stimulus.
Other measures of cognitive function responded similarly to both intervention sessions (aerobic exercise + sham-tDCS and aerobic exercise + active-tDCS). Participants increased MoCA score and reduced time taken to complete Trail Making Test parts A and B after both intervention sessions. These findings may represent exercise-related improvement on global cognition, visual attention, and executive function. However, we cannot rule out a potential learning effect in our data because the current study did not include a control session for the exercise (e.g., sham-exercise or rest). To date, benefits to cognition have been reported by previous studies assessing the acute effects of a single session of aerobic exercise in PD.16,18
Clinical Implications and Future Directions
Current findings suggest that the combination of aerobic exercise with anodal tDCS is a promising approach for the development of enhanced gait intervention. Benefits observed with the addition of anodal tDCS to aerobic exercise may have a positive impact on mobility in real life (out of the lab). Considering that increased gait variability is a surrogate marker of fall risk, the observed reduction in gait variability after the combined intervention may represent a reduction in fall risk. Furthermore, faster processing speed (shortened reaction time) can help patients identify and respond more quickly to dangerous stimuli during daily activities, such as obstacles in the pathway and external perturbations to balance. To date, a recent study from our group showed that patients with PD had faster postural responses to external perturbation after a single session of anodal tDCS (1-2 mA for 20 minutes) applied over the primary motor cortex. 57
Since the current study focused on the immediate/acute effects of the combined intervention, studies are needed to examine the duration of benefits following 1 intervention session as well as the potential benefits of multiple sessions. Future studies are also needed to identify the optimal tDCS montage (current parameters and electrode placement) to be applied in combination with aerobic exercise. Because PD leads to a broad cortical dysfunction, 58 perhaps the stimulation of multiple areas may lead to greater benefits. Finally, studies on larger sample sizes are required to identify potential predictors of response to interventions combining aerobic exercise with tDCS in PD.
Strengths and Limitations
A key strength of the current study is the sham protocol, which was effective in blinding participants to the tDCS condition. Also, the concurrent assessment of PFC activity while walking, gait parameters, and cognitive functions provides a more complete picture of the multiple aspects involved in the control of gait and the potential mechanisms underlying gait improvements obtained with the combined intervention. On the other hand, our study had no control session for the exercise (e.g., sham-exercise or rest), involved a small cohort (n = 20) and is limited by assessing and stimulating the PFC only.
Conclusion
The addition of anodal tDCS over the PFC to a session of aerobic exercise was well tolerated and led to immediate positive effects on gait variability, processing speed, and executive control of walking in people with PD. Current findings encourage the combination of aerobic exercise with anodal tDCS for the development of enhanced gait intervention in PD.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211019344 – Supplemental material for Aerobic Exercise Combined With Transcranial Direct Current Stimulation Over the Prefrontal Cortex in Parkinson Disease: Effects on Cortical Activity, Gait, and Cognition
Supplemental material, sj-docx-1-nnr-10.1177_15459683211019344 for Aerobic Exercise Combined With Transcranial Direct Current Stimulation Over the Prefrontal Cortex in Parkinson Disease: Effects on Cortical Activity, Gait, and Cognition by Núbia Ribeiro Conceição, Lilian Teresa Bucken Gobbi, Priscila Nóbrega-Sousa, Diego Orcioli-Silva, Victor Spiandor Beretta, Ellen Lirani-Silva, Alexandre Hideki Okano and Rodrigo Vitório in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank the National Council for Scientific and Technological Development (CNPq; Grant Number: 147763/2017-7; 166467/2017-0) and the São Paulo Research Foundation (FAPESP; Grant Number: 2016/21499-1) for financial support. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior Brasil (CAPES), Finance Code 001.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the the National Council for Scientific and Technological Development (CNPq; Grant Number: 147763/2017-7), the São Paulo Research Foundation (FAPESP; Grant Number: 2014/22308-0; 2016/21499-1), and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior Brasil (CAPES), Finance Code 001.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
