Abstract
Background
Functional imaging studies have associated dystonia with abnormal activation in motor and sensory brain regions. Commonly used techniques such as functional magnetic resonance imaging impose physical constraints, limiting the experimental paradigms. Functional near-infrared spectroscopy (fNIRS) offers a new noninvasive possibility for investigating cortical areas and the neural correlates of complex motor behaviors in unconstrained settings.
Methods
We compared the cortical brain activation of patients with focal upper-limb dystonia and controls during the writing task under naturalistic conditions using fNIRS. The primary motor cortex (M1), the primary somatosensory cortex (S1), and the supplementary motor area were chosen as regions of interest (ROIs) to assess differences in changes in both oxyhemoglobin (oxy-Hb) and deoxyhemoglobin (deoxy-Hb) between groups.
Results
Group average activation maps revealed an expected pattern of contralateral recruitment of motor and somatosensory cortices in the control group and a more bilateral pattern of activation in the dystonia group. Between-group comparisons focused on specific ROIs revealed an increased activation of the contralateral M1 and S1 cortices and also of the ipsilateral M1 cortex in patients.
Conclusions
Overactivity of contralateral M1 and S1 in dystonia suggest a reduced specificity of the task-related cortical areas, whereas ipsilateral activation possibly indicates a primary disorder of the motor cortex or an endophenotypic pattern. To our knowledge, this is the first study using fNIRS to assess cortical activity in dystonia during the writing task under natural settings, outlining the potential of this technique for monitoring sensory and motor retraining in dystonia rehabilitation.
Introduction
Handwriting is a highly complex, skilled task involving multiple linguistic, cognitive, sensory, and motor processes. 1 Dystonia, a movement disorder characterized by involuntary sustained or intermittent muscle contractions that cause abnormal postures and repetitive twisting movements, 2 has many different clinical manifestations. The most common task-specific dystonia, a subtype of focal dystonia known as writer’s cramp (WC), is believed to evolve as a consequence of intense repetition of skilled movements in individuals with a genetic susceptibility. 3
Functional imaging studies have associated dystonia with abnormal activity across multiple motor and sensory brain regions, leading to the conception of a circuit disorder and supporting a network model.4-6 This model is consistent with 3 main mechanisms believed to be linked to the physiopathology of dystonia: (a) disorders of the inhibitory process, (b) disorders of sensorimotor integration, 7 and (c) maladaptive plasticity.3,8,9 From that perspective, several studies have shown abnormal cortical activity in patients with focal hand dystonia, suggesting a loss of surround inhibition10,11 and altered somatosensory processing and integration.12-15 Recent investigations have shown that the dorsal premotor cortical projections to the primary motor cortex (M1) appear to contribute to an increase in surround inhibition during rest and a reduction during movement, supporting M1 dysfunction in focal dystonia. Regarding the abnormal sensory function, maladaptive plasticity (ie, abnormal plastic changes of the primary sensorimotor cortex triggered by repetitive movements and peripheral sensory stimulation of the affected hand) is considered to be a possible cause, especially in task-specific dystonias.16-18
Most studies investigating neural correlates of motor control currently use neuroimaging techniques, such as functional magnetic resonance imaging (fMRI) and positron emission tomography (PET),19-21 which impose significant physical constraints on mobility. Recent advances in functional near-infrared spectroscopy (fNIRS) offer a new possibility for investigating cortical areas and the neural correlates of complex motor behaviors in unconstrained settings.22-26 The operating principle of fNIRS relies on the selective light absorption of hemoglobin chromophores, oxyhemoglobin (oxy-Hb), and deoxyhemoglobin (deoxy-Hb) in the near-infrared region of the spectrum. It enables us to measure brain activity by quantifying the light migrating from sources to detectors carefully placed on the scalp, based on the neurovascular-coupling principle, which indicates a relationship between neural activity and changes in regional cerebral blood flow. 27 Compared with traditional techniques, fNIRS has several advantages, such as lower cost, portability, and lower sensitivity to motion artifacts, which enables the study of complex motor tasks in more naturalistic conditions. 28
Long-term effects of existing dystonia medical treatments are limited, and previous studies support neurorehabilitation of focal hand dystonia patients through behavioral treatment, such as sensory and motor retraining.29-32 However, this method is almost exclusively evaluated based on clinical measures, which are often subjective and lack precision. The features of fNIRS make it a potential candidate for monitoring sensory and motor retraining.
In this study, we investigated the feasibility of using fNIRS to measure cortical hemodynamic changes during writing tasks in patients with focal right upper limb dystonia and controls. We hypothesized that patients and controls differ at the level of cortical sensorimotor representation and that dystonia patients show a less specific pattern of activity during the writing task.
Methods
This study was approved by the review boards of all participating institutions (Hospital Israelita Albert Einstein, Universidade Federal de São Paulo and Hospital do Servidor Público Estadual de São Paulo, Approval Number 63155316.8.0000.0071), and all participants provided written informed consent for participation and study data publication.
Participants
We recruited 27 controls and 28 patients with focal right upper limb idiopathic dystonia. After applying the exclusion criteria described in the fNIRS Data Analysis section, we analyzed 21 patients (mean age 45.38 ± 10.93 years; 7 males) and 21 healthy volunteers (mean age 42.14 ± 12.71; 13 males). Participants were paired by age and years of education and were recruited by neurologists at the participating sites. All participants were >18 years of age, right-handed, literate in Portuguese, and with a minimum of 8 years of formal education. The diagnosis of idiopathic focal dystonia was established according to current recommendations. 2 Of the 21 patients with focal right upper limb dystonia, 6 had task-specific dystonia (WC). All participants had normal structural brain MRI. Visual inspection of MRI and clinical diagnosis of dystonia were performed by the team neurologists (DDF, VB, SMCAS, HBF, and PCA), all experienced in movement disorders. Exclusion criteria were as follows: a history of neurological diseases other than idiopathic dystonia, history of limb or brain surgery, moderate to severe cranial trauma, cancer, current use of psychoactive or other drugs that might interfere with the brain’s hemodynamic response, uncontrolled metabolic disorders, contraindications for MRI, and use of botulinum toxin less than 3 months before recruitment.
fNIRS Data Acquisition
fNIRS data were acquired using a continuous wave NIRSport system (NIRx Medical Technologies, Glen Head, NY) with 8 LED sources emitting 2 wavelengths of near-infrared light (760 and 850 nm) and 8 silicon photodiode active detectors, at a sampling rate of 7.81 Hz. Optodes were positioned on the measuring cap with reference to the 10-5 international system. 33 The spatial distribution of the optodes on the cap was chosen to result in channels (ie, source-detector pairs) with standard interoptode distances of approximately 30 mm. In total, 23 channels covered 5 different regions of interest (ROIs): right and left primary motor (M1) and primary somatosensory areas (S1) and the supplementary motor area (Figure 1). ROIs were defined using the MATLAB-based toolbox fOLD (fNIRS Optodes’ Location Decider; Zimeo Morais et al 34 ). fOLD provides a measure of specificity of an fNIRS channel for each anatomical region given a chosen parcellation atlas—in this case, the Juelich atlas. Supplemental Table T1 (supplemental material available online) shows the anatomical regions with highest sensitivity for each channel of our setup. The NIRStar14-2 acquisition software was used to record the raw intensity data.
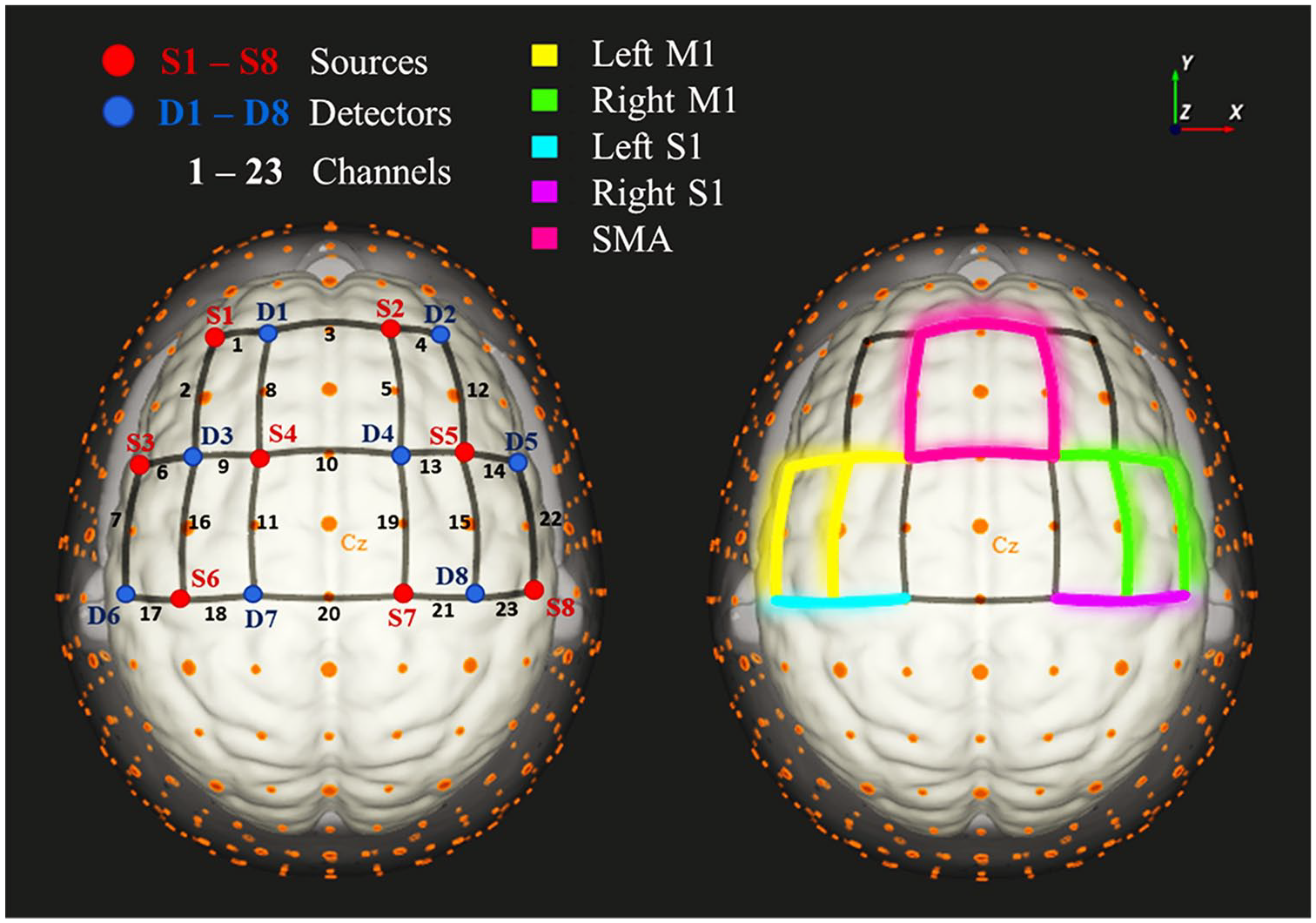
Functional near-infrared spectroscopy setup. On the left: channels are represented by the black edges numbered from 1 to 23; sources are represented in red (S1 to S8) and detectors in blue (D1 to D8). On the right, the 5 regions of interest: Left M1 (M1 L) comprising channels 6, 7, 9, and 16; right M1 (M1 R), channels 13, 14, 15, and 22; supplementary motor area (SMA), channels 3, 5, 8, and 10; left somatosensory (S1 L), channels 17 and 18; right somatosensory (S1 R), channels 21 and 23.
Writing Task
Participants were submitted to a simple right-hand writing task paradigm that consisted of 4 epochs of alternating writing/resting blocks (30 s duration each). For the handwriting condition, participants were required to write, as many times as possible, the first sentence of the song “Happy Birthday to You” in Portuguese (Parabéns para você) until the end of the block. This sentence was chosen because of it being universally known, thus not requiring significant cognitive efforts, allowing us to primarily measure the brain activity derived from the motor task. Participants were instructed to write in their usual writing style as quickly as possible, while maintaining accuracy. We used a traditional ballpoint pen and A4 white paper in order to simulate a more ecological condition. During rest, participants were instructed to stay still and to keep staring at the paper while holding the pen. A short video of a volunteer executing the task can be found as supplementary material.
The timing of writing and resting blocks were controlled and cued with auditory stimuli using the software E-prime. The total duration of the paradigm was approximately 4 minutes. The presence of dystonia during the writing task was clinically characterized by a clear dystonic posture, either hyperflexion or hyperextension of the wrist with a tight grip. The performance was evaluated by the total number of written letters, which was tested for normality (ie, verified if it was Gaussian) using the Shapiro-Wilk test and compared between groups with a Student t-test. Given the lack of standardized handwriting assessments in Portuguese, 2 independent investigators, blinded to volunteers’ clinical status, performed a subjective quality analysis of their handwriting, evaluating whether they were legible or not and whether legibility worsened during the task or not. For the cases where reviewers disagreed, they discussed in order to come up with a consensual rating. Fisher Exact Tests were performed to examine the relation between group and legibility as well as group and handwriting worsening during the task within investigator’s consensual ratings on each of those aspects.
fNIRS Data Analysis
The data were analyzed using NIRSLab v2017.06 (https://www.nitrc.org/projects/fnirs_downstate/). The quality of the signals was evaluated by considering each channel’s coefficient of variation (CV = SD/mean) over the entire experiment. Channels with a CV exceeding 7.5% were rejected. After converting the intensity data to optical density, oxy-Hb and deoxy-Hb concentration changes were calculated using the modified Beer-Lambert equation (according to the parameters from W. B. Gratzer). 35
Channel-wise statistical analysis was carried out using the approach proposed by Baker et al (2013) 36 with an autoregressively whitened robust regression model embedded in NIRSLab v2017.06. This analytical approach deals with physiological noise and motion artifacts statistically within the general linear model (GLM), with no prior preprocessing of the data. 37 In brief, a GLM was applied to each fNIRS channel time series with one regressor of interest, which modeled the 30-s duration of the writing condition. The regressor was convolved with a time-to-peak canonical hemodynamic response function of 6 s. 38 To track decreases in deoxy-Hb concentration as a biomarker of increased neuronal activity, the hemodynamic response function used in the GLM for this chromophore was multiplied by −1. At the individual level, each channel contrast between the writing condition relative to the resting condition (writing > resting) was evaluated using a 1-sample t-test. Group average activation maps were created separately for each group using a 1-sample t-test, Bonferroni corrected for multiple comparisons (resulting P value threshold: <.002). Differences between groups were examined using an independent samples t-test. False-positive results were set at 5% (Bonferroni corrected for multiple comparisons).
We also conducted a group analysis based on ROIs. Eligibility criteria required participants to have at least 50% of channels at each ROI that passed quality control (ie, CV <7.5%). For each ROI, we calculated the mean of group-averaged β estimates for the contrast between the writing condition relative to the resting baseline across the channels of the ROI. Mean activation differences between patients and controls for each ROI were then compared using analysis of covariance controlling for sex. Statistical analysis was conducted using the IBM SPSS Statistics 23 software on changes in both oxy-Hb and deoxy-Hb. WC and non-WC patients were also compared using the same approach. Statistical significance was considered under the threshold P <.05. Partial correlations were calculated controlling for sex in order to investigate the relation between the number of written letters and oxy-Hb and deoxy-Hb levels in each ROI for all participants.
Results
The Shapiro-Wilk test showed no deviation from normality regarding the total number of letters written by both groups. A 1-tailed t-test revealed that controls wrote significantly more than patients (t = 2.901; P = .003). All patients presented with dystonia during the writing task. Investigators agreed in 90.47 % of cases regarding participant’s handwriting legibility; and 83.55% of them agreed with respect to handwriting legibility worsening during the task. The consensual rating revealed that 100% (21/21) of controls and 80.95% (17/21) of patients had legible handwriting and that 14.28% (3/21) of controls and 33.33% (7/21) of patients experienced worsened handwriting during the task. Fisher Exact Tests suggested no association between group and legibility (P = .107) or between group and handwriting worsening (P = .227).
Figure 2 depicts channels exhibiting increased oxy-Hb (above) and decreased deoxy-Hb (below) during the writing condition compared with rest in patients and controls. In controls, a predominant lateralization to the left side was observed in most of the central channels covering the primary motor and somatosensory cortices. A more bilateral pattern of activation was observed for patients. However, in a direct channel-wise comparison between groups, no statistically significant differences were observed in any channel.
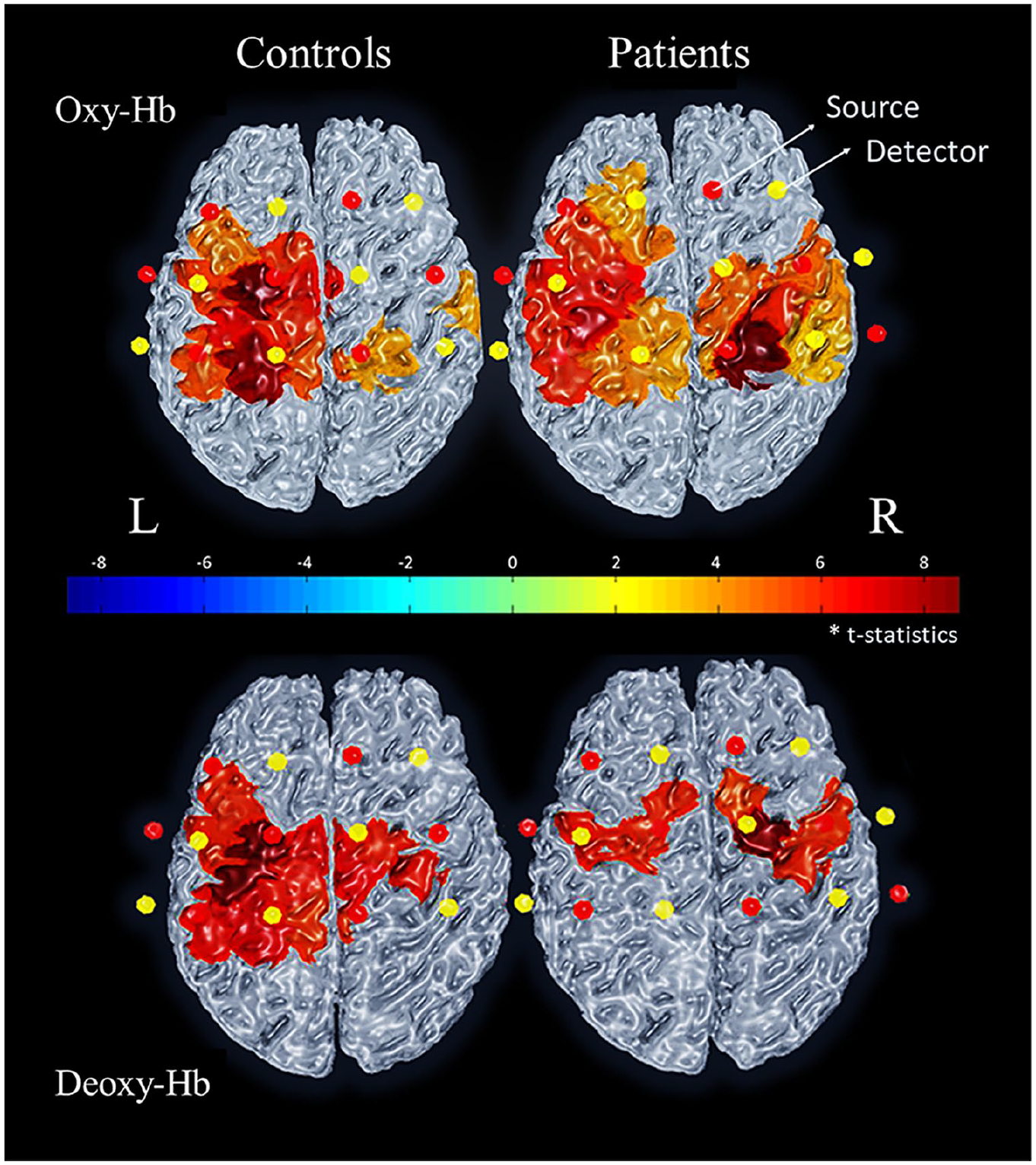
Oxy-hemoglobin (oxy-Hb) and deoxy-hemoglobin (deoxy-Hb) activation maps for the contrast Writing > Resting. Activation maps from the channel-wise analysis showing the mean activation of controls (left) and patients (right) for oxy-Hb (above) and deoxy-Hb (below). Patients’ map shows a more bilateral activation pattern. Red and yellow dots represent sources and detectors, respectively. Results for each channel were Bonferroni corrected for multiple comparisons (P value < .002).
Using an ROI-based group analysis controlled for sex, group differences were observed in 3 of the 5 ROIs (Figure 3). Homogeneity of regression slopes and equality of error variances were verified. Compared to controls, patients exhibited increased oxy-Hb levels in the left M1 [F(1, 39) = 4.24; P = .046], right M1 [F(1, 39) = 7.820; P = .008], and left S1 [F(1, 39) = 4.47; P = .041] ROIs. No differences between patients and controls were observed in this analysis regarding deoxy-Hb levels. Results of statistical analysis for oxy-Hb and deoxy-Hb can be found in Supplemental Tables T2 and T3, respectively. In addition, we did not observe significant differences in oxy-Hb and deoxy-Hb changes between WC patients and non-WC patients for any ROI (Supplemental Tables T4 and T5). Partial correlations also controlled for sex revealed no relationship between the number of letters written and either oxy-Hb levels or deoxy-Hb levels in any ROI (Supplemental Tables T6 and T7). Supplemental Figure S1 depicts the average hemodynamic response of each group observed in the left M1, right M1, and left S1 ROIs.
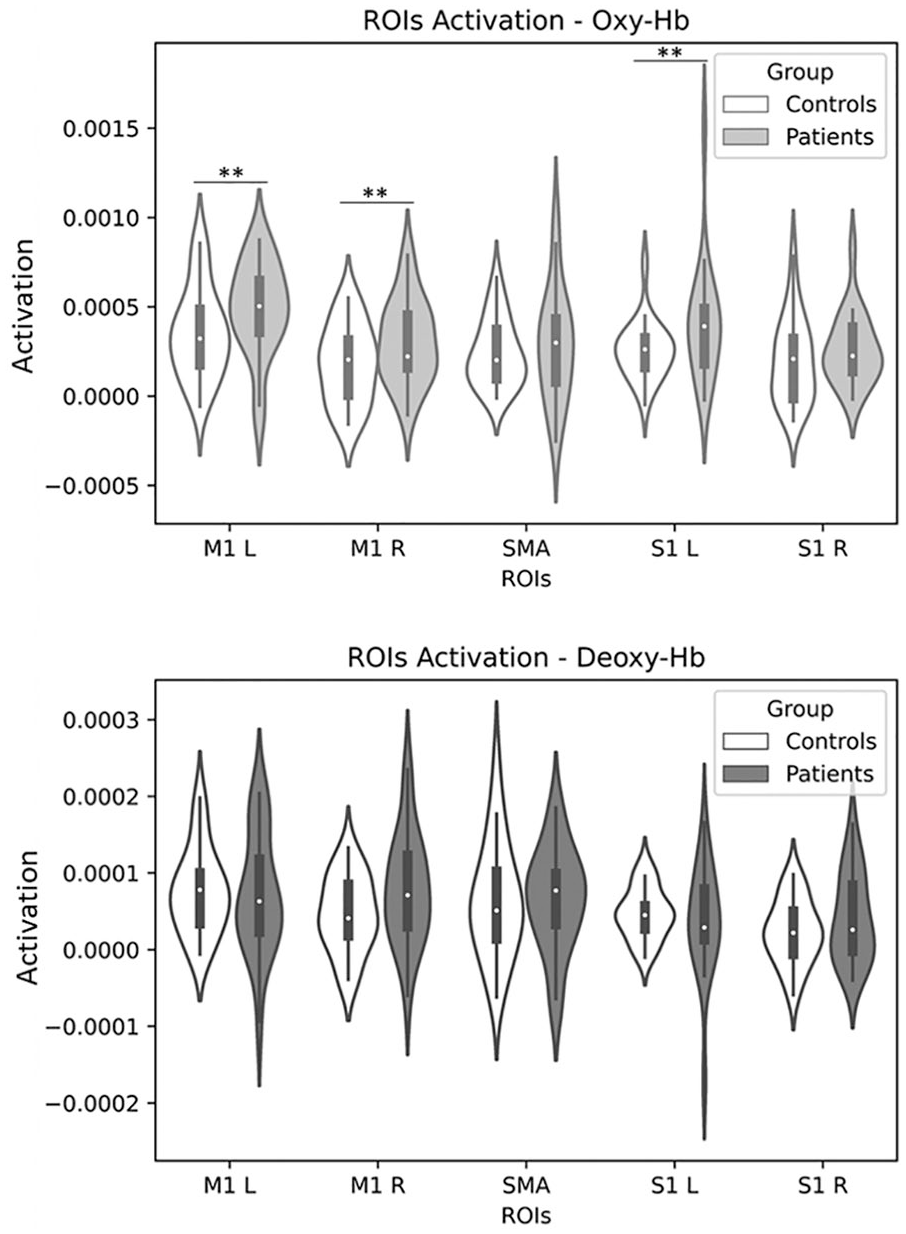
Comparison of the regions of interest (ROIs) activation between patients and controls. Left M1 (M1 L), right M1 (M1 R), supplementary motor area (SMA), left S1 (S1 L), and right S1 (S1 R), where activation is given by the difference between writing and resting β coefficients (eg, coefficients of activation; Activation = βwriting − βresting) for oxy-Hb (above) and deoxy-Hb (below) signals. Patients’ M1 L (P = .046), M1 R (P = .008), and S1 L (P = .041) exhibited statistically significant increased oxy-Hb levels compared with controls.
Two tables with all participants’ coefficients of activation for both oxy-Hb and deoxy-Hb signals, their age, sex, number of letters written, and clinical status (ie, whether WC, non–task-specific dystonia or nondystonic) are available in the supplementary material (Supplemental Tables T8 and T9).
Discussion
To our knowledge, this is the first study to use fNIRS to access the cortical activity of patients with focal upper limb dystonia during the writing task. This task was performed in a naturalistic environment, reflecting a real-life scenario, which is usually not completely reproducible by other brain mapping techniques such as fMRI and PET. Results revealed that patients have higher activation in contralateral M1 and S1, which might be related to the motor overflow and recruitment of muscles not related to the task. Furthermore, the finding of increased activation in patients’ left S1 reinforces the contribution of abnormal somatosensory processing in focal upper limb dystonia, as suggested by previous studies.12,39
Increased activation in patients’ left M1 is in accordance with fMRI findings during imaginative writing. 40 Overactivity in contralateral M1 and S1 have been reported in patients with WC, in musician’s dystonia,41,42 and in DYT1-positive patients. 43 High spatial resolution fMRI studies have shown a disorganized representation of the fingers of the dystonic hand in patients with WC, with shortened interdigit distance and overlapping of the receptive fields. 18
Interestingly, we observed a higher activation of the patients’ right M1 in comparison to controls expressed in oxy-Hb signals. Bilateral activation of M1 during complex tasks, with increased demand for higher accuracy, has been described in healthy participants.44,45 Although the role of the ipsilateral M1 is unclear, it is possible that it modulates the movement in order to achieve higher precision and that the magnitude of interhemispheric inhibition depends on the task complexity. As we chose a low complexity task, with no high skills demand, we were not expecting an ipsilateral activation, which turned out as expected in the control group but not in dystonia patients.
Bilateral activation, though, is not unusual in dystonia and could indicate a possible primary disorder of the cortical motor system that leads to dystonia or an endophenotypic pattern. 18 Abnormal cortical finger representation in patients with dystonia has been observed in the nondystonic limb at rest while receiving peripheral sensory stimulation, 18 and bilateral functional abnormalities have been reported in asymptomatic DYT1 mutation carriers. 46 The later onset of dystonia in the previously nonaffected limb in WC patients, mainly when they try to readapt using the contralateral limb, also corroborates this hypothesis. 47
Activation maps suggest bilateral activity in controls as well as in patients, which is probably related to the interhemispheric inhibition of the motor cortices because of the skillfulness demanded by the writing task. However, at the ROI level, patients presented a significantly increased activation of the ipsilateral motor cortex in relation to controls, which matches our primary hypothesis of a less-specific recruitment of motor areas.
Regarding handwriting evaluation, patients wrote significantly less than controls as expected. However, we did not observe any significant differences in handwriting legibility or in handwriting worsening during the task between patients and controls. Thus, we believe that observed differences in the number of letters written indeed reflects the dystonic symptoms during the task and not possible speed-accuracy trade-offs.
Many studies point out a problem in sensorimotor integration in focal dystonia.7,15,48 According to Blood, 4 the most likely possibility is that the motor system responds to the sensory information sent at too great an amplitude. Among the possible causes for this response amplification are altered neurotransmitter/receptor functions, including dopamine 49 and GABA. 50 It remains uncertain whether misrepresentation of the dystonic limb sensory processing is secondary to dystonia, because dystonic movements are likely to alter the corresponding receptive fields in S1 through maladaptive plasticity, or whether it is the primary cause.16,18 Meunier et al 18 found a dramatic disorganization on the cortical finger representation of the nondystonic limb in patients with idiopathic upper limb dystonia, supporting the hypothesis of a primary cause to abnormal sensory processing. However, in terms of activation magnitude, we did not observe a significant difference between groups in the ipsilateral somatosensory cortex ROI.
We did not observe significant differences in deoxy-Hb signals between patients and controls in any ROI. This may perhaps account for the lower signal-to-noise ratio of deoxy-Hb. 51 Additionally, studies suggest a low symmetry between oxy-Hb and deoxy-Hb signals in the motor cortex, 52 although not much is known about the relation between oxy-Hb and deoxy-Hb hemodynamics in dystonia patients.
Functional brain connectivity and activation patterns have been assessed with fNIRS in patients after stroke during motor rehabilitation tasks 53 and motor preparation for gait rehabilitation. 54 Because fNIRS allows complex movement in unconstrained environments, it may be used with feedback to facilitate learning in motor control 55 and even in neurofeedback settings to control prosthetic limbs. 56 We believe that future studies could use real-time neurofeedback for modulating activity in the sensorimotor cortex in patients with dystonia, using fNIRS signals.
Among the limitations of our study, it is worth mentioning that we did not monitor systemic variables that could better inform the origins of the observed task-related changes in oxy-Hb using, for example, short-distance channels or breath and heart rate monitoring. 26 Also, an intrinsic limitation of fNIRS is the restriction to the cortical area activity. In this concern, there is an increasing acknowledgment of the involvement of subcortical areas as modulators of M1, yet it is still not possible to investigate such relations using fNIRS. Additionally, we should point out that in our analysis based on ROIs, statistical significance was considered under the threshold P <.05 and, therefore, not corrected for multiple comparisons as opposed to the channel-wise analysis. Considering Bonferroni correction for multiple comparisons, only the difference in ipsilateral M1 (P < .01) remains. Therefore, the differences spotted in contralateral M1 and S1 must be interpreted with caution.
Conclusions
Our findings provide preliminary support for the use of fNIRS as a resource for monitoring sensory and motor retraining in patients with focal upper-limb dystonia. fNIRS was able to detect altered sensorimotor processing in these patients during the writing task and proved to be a powerful tool because it allows experiments involving a vast repertoire of complex movements in unconstrained environments. Considering its portability, low cost, and relative robustness to motion artifact, fNIRS holds potential for focal hand dystonia rehabilitation assessment and needs to be further explored for the study of other movement disorders under naturalistic conditions. Nevertheless, further research is needed to confirm the feasibility of this approach for behavioral treatment in dystonia patients.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211019341 – Supplemental material for Motor Cortex Activation During Writing in Focal Upper-Limb Dystonia: An fNIRS Study
Supplemental material, sj-docx-1-nnr-10.1177_15459683211019341 for Motor Cortex Activation During Writing in Focal Upper-Limb Dystonia: An fNIRS Study by Renata Prôa, Joana Balardin, Danilo D. de Faria, Artur M. Paulo, João R. Sato, Carlos A. Baltazar, Vanderci Borges, Sonia M. C. Azevedo Silva, Henrique B. Ferraz and Patricia de Carvalho Aguiar in Neurorehabilitation and Neural Repair
Supplemental Material
Footnotes
Acknowledgements
We thank all volunteers and the support of Sociedade Beneficente Israelita Brasileira Hospital Albert Einstein.
Authors’ Note
A preliminary version of this work was presented at the 3rd Pan American Parkinson’s Disease and Movement Disorders Congress and published as an abstract in the annals. Additional data not included in the supplementary material may be available on request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JB, CAB, and PCA are employed at Hospital Israelita Albert Einstein. DDF and SMCAS are employed at Hospital do Servidor Publico Estadual de São Paulo. HBF and SMCAS are employed at Universidade Federal de São Paulo. JRS is employed at Universidade Federal do ABC. JRS and PCA received research grants from São Paulo Research Foundation (FAPESP, Brazil). SMCAS received honoraria from Roche. VB received honoraria from Aché. HBF received honoraria from Zambon, Roche, Teva, and Torrent. RP and AMP have no disclosures to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Sociedade Beneficente Israelita Brasileira Hospital Albert Einstein.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
