Abstract
Background
Cognition affects poststroke recovery, but meta-analyses of cognition have not yet provided a comparison of observational and intervention evidence.
Objective
To describe the trajectory of poststroke cognition and the factors that moderate it across intervention and observational cohorts.
Methods
Six databases were searched up to January 2020. Studies describing quantitative changes in cognition in adults poststroke were included. Interventions were classified into pharmacological, therapist-led, nonroutine/alternative, and usual care. Summary estimates were compared via hierarchical mixed-effects models. Age, recovery stage, stroke etiology, cognitive domain targeted in studies, and intervention types were investigated as moderators of cognition. Recovery stage and intervention were further analyzed in a multiplicative metaregression model.
Results
A total of 43 intervention trials and 79 observation cohorts involving 28 222 stroke participants were included. Heterogeneity was significant (τ2 = 0.09; CI = 0.01-0.21, P < .001) with no evidence of publication bias. Cognitive recovery was greater in intervention trials (g = 0.47; CI = 0.37-0.58) than observational cohorts (g = 0.28; CI = 0.20-0.36) across all moderators analyzed. Nonroutine/alternative and pharmacological trials achieved the best overall results (g = 0.57, CI = 0.42-0.73, and g = 0.52, CI = 0.30-0.74, respectively), followed by therapist-led (g = 0.46; CI = 0.17-0.74), and usual care (g = 0.28; CI = 0.11-0.45) interventions. Medium recovery effects (ie, g ≥ 0.5) were observed in examining first-ever stroke, executive function, visuo-perceptual, consciousness, and psychomotor skills, 61 to 180 days poststroke, in participants aged 65 to 70 years.
Conclusion
Cognitive recovery is possible using different controlled interventions in all recovery stages, with smaller benefits ≥2 years poststroke. Longer-term studies are needed to determine the role of nonroutine/alternative therapies and the association between cognitive recovery and performance in everyday activities.
Introduction
Cognition is a fundamental skill for learning, planning, and executing new and known tasks in the context of stroke recovery and rehabilitation. 1 The study of cognition, however, is a complex endeavor because the construct comprises many processes (eg, attention, processing speed, memory), 2 all of which can work separately or in unison to meet the demands of different functional tasks. 3 For stroke survivors, cognitive deficits—especially in executive function—are a common and persistent manifestation of disability, affecting up to 80% of this population.4,5 The study of cognition is a top research priority in stroke 6 because survivors with cognitive impairment are often discharged to a new and more challenging life, facing major limitations in common activities such as driving, cooking, working, and overall participation in society.7-10 The consequences of cognitive disability are well documented in the literature, bringing a substantial and negative impact to the economy and global health but, more importantly, to the livelihoods of stroke survivors and their families.11-14
Several prospective cohort studies have described poststroke cognition past the 3-year mark.15-17 Although these investigations continue to reveal strong associations between cognition and individual factors (eg, age, diabetes, hypertension, ethnicity, socioeconomic status), they only provide suggestions on what might be driving cognitive recovery. Intervention trials, on the other hand, have investigated a variety of treatments and their effect on cognition using pharmacological,18,19 activity-based, 20 return-to-work, 21 behavioral, 22 and combined approaches (eg, acupuncture plus pharmacotherapy). 23 , 24 These trials often yield positive outcomes, though they are also frequently deemed underpowered to determine causal effects. 14
A growing interest in describing cognitive recovery poststroke has reignited recently with new systematic studies examining the effects of psychological and cognitive remediation interventions on cognition.25-27 These studies support the notion of small to medium effects on recovery of poststroke cognition while also highlighting the need to determine the timing at which interventions may be more effective.
Despite the existence of very recent evidence looking at treatment of poststroke cognitive impairment, to date, no systematic review has summarized and compared the full range of existing interventions. Recent clinical and public health reports have recommended the adoption of a comparative approach that permits pooling across study designs.28-32 This approach would allow for the examination of poststroke cognition under different intervention conditions and in comparison to observational studies and natural history trajectories. In addition, such information would critically inform health professionals interested in designing programs targeting cognition at different stages of poststroke recovery.
Given the available knowledge, it is opportune to analyze the trajectory of cognitive function after stroke—and the factors that drive cognitive recovery—with a wider lens that allows for a global comparison of different study designs (ie, intervention and observational), intervention types, intervention timepoints, and moderating factors.
We aimed to quantitatively summarize the short- and long-term changes in poststroke cognition and to examine the factors that moderate these changes over time, across intervention and observational studies, by means of meta-analysis and metaregression. In addition, we sought to ascertain the interaction between intervention and time (ie, recovery stage) on cognition by comparing outcomes sourced from different intervention trials at different times of recovery.
Methods
This review adheres to all PRISMA guidelines. 33 Our PROSPERO protocol (CRD42017054449, August 23, 2017) included two parts: (1) a systematic scoping review, 34 describing the types of outcomes (frequencies, odds ratios, mean scores, median scores, etc) reported in stroke cognitive research; the cognitive tools administered; and the use of linking rules to classify cognitive outcomes with the definitions provided by International Classification of Functioning and Disability (ICF)3,35 and (2) the present study, in which we make use of this classification method and describe the quantitative changes in cognition poststroke.
Search Strategy and Study Selection
Six online libraries were examined up to January 14, 2020: PsycInfo, EMBASE, MEDLINE, PubMed, Web of Science, and CINAHL. The following MeSH terms were used: cerebrovascular accident, cognition, executive function, attention, memory, longitudinal studies, and treatment outcome. An extensive list of additional index terms, keywords, synonyms, truncation, and wildcards were included and used consistently across all searches. For instance, the term cognit* was truncated across all searches to include words with different endings such as cognition and cognitive as well as other associated keywords, such as cognit* training, cognit* rehabilitation, and cognit* intervention. Search limits (English language, peer-reviewed articles, excluding preclinical studies, reviews, study protocols, and conference proceedings) were embedded in our strategy to limit the number of off-topic results. The full search for each online library can be found in our online protocol.
Study Selection and Data Extraction
We included randomized trials, nonrandomized trials, before-after trials, and observational cohort studies in adults with ischemic or hemorrhagic stroke 36 using objective (therapist-scored) instruments of cognition, obtained for 2 or more evaluation time points. Two independent reviewers (JPS and TT) screened all titles and abstracts using Covidence. 37 Potentially relevant studies were retrieved and screened in full by the same 2 reviewers. Conflicts were resolved by consensus, after thorough review of each article. Records not available online were requested from corresponding authors or excluded after 1 month of no response. Studies including stroke in addition to other types of brain injury (eg, traumatic brain hemorrhage, aneurysm, anoxic brain injury) were included only if cognitive outcomes over time were provided separately for the stroke subgroup.
Data from each eligible report were independently extracted in pairs formed by VN, KS, and JPS. Data from studies reporting cognitive outcomes in figures were extracted with the Engauge software. 38 Figures were then replicated to compare them against the original ones. Items extracted from each study included the following: author, sample size, study origin (ie, country), year of publication, study type (intervention or observational), study design (prospective/retrospective cohort, case-control, case-series, randomized/nonrandomized, and before-after), 39 sample age and sex, stroke etiology (ie, ischemic, hemorrhagic, both), assessment time (days poststroke), instruments used, and sample attrition (if reported). Extraction inaccuracies were identified by data matching, using algorithmic-based comparison. Conflicting datapoints were re-extracted and checked with the same code until no more conflicts were found.
Recoding of Cognitive Domains
Domains were independently mapped by JPS and TT to a general or specific mental function of the ICF, using linking rules, 35 as explained in detail in our previous publication. 34 This process allowed consistency in the assignation of cognitive domains evaluated by each tool, despite discrepancies found between the domains reported in each article. Studies looking at specific interventions were included as long as the cognitive domain was addressed and defined as a cognitive function. For instance, studies of neglect were included only if the evaluation included attention as a cognitive function rather than a visual skill (ie, as evaluated by a visual field test). Similarly, studies looking at language were included using the same criteria (ie, they were eligible for this review, so long as language was defined as a cognitive function rather than a motor or social skill).
Defining the Types of Intervention
Each group or subgroup within an intervention trial undergoing treatment in addition to usual care was categorized as indicated below:
Therapist-Led Clinical Interventions (One-on-One)
This consisted of all studies, or subgroups within studies, that examined the effects of therapist-led interventions (in addition to usual care) on cognition. These included additional physical or occupational therapy; multidisciplinary, therapist-led clinical interventions; cognitive training; Cognitive Orientation to daily Occupational Performance (CO-OP) 40 ; time-pressure management; and task-oriented interventions.
Nonroutine Interventions and Alternative Therapies
This included studies, or subgroups within studies, with interventions not routinely administered in stroke treatment, such as thrombolysis and stenting; driving, driving simulation, or driving-related tasks; multidisciplinary home-based care or home treatment; video game interventions, computer-based cognitive training, robot therapy, color therapy, or color-rhythm therapy; and intensive exercise, cardiac conditioning, and ergometer-based interventions. This category also included other less common forms of therapy, such as alternative therapies (eg, acupuncture, herbal medicine, or mixed therapies), transcranial magnetic stimulation, and secondary prevention of stroke (eg, self-management strategies and education, with diet and pharmacological treatment).
Pharmacological Intervention
This group consisted of studies, or subgroups within studies, exposed to treatment exclusively with pharmaceuticals such as blood pressure–lowering drugs (eg, nimodipine, perindopril), lipid-lowering drugs (eg, simvastatin), cognitive stimulants (eg, donepezil, methylphenidate, levodopa, or paroxetine), thrombolytics (eg, cerebral care granule), or aspirin.
Usual Care, With or Without Placebo Interventions
Study subgroups within intervention trials receiving usual care or usual care plus placebo. Usual care varied between studies as defined by each study’s specific intervention protocol. These studies were used to quantify cognitive outcomes associated with specified usual care and to provide a comparison group of natural trajectory directly linked with the intervention trials.
Methodological Quality (Risk of Bias) of Included Studies
All studies were independently appraised by JPS, TT, or SO using the Study Quality Assessment Tools of the National Institutes of Health and National Heart, Lung and Blood Institute (NHLBI). 39 These tools were developed to assess the methodological strength of specific study designs (ie, controlled interventions, observational cohorts, case-control studies, and before-after trials with no control group). Each tool contains 12 to 14 items. The number of items vary depending on the presence/absence of randomization, blinding, and/or a control group. The items appraised across all study designs are tailored to identify methodological flaws that might be associated with bias in the following areas: (1) precision of the research aims and presence/absence of a study protocol; (2) sampling and selection methods; (3) outcome assessment; (4) statistical analyses; (5) study power and attrition; (6) blinding; and (7) randomization/concealment procedures (where applicable). In accordance with the guidelines provided by the NHLBI, a final judgment of good, fair, or poor quality was given to each study to indicate susceptibility to bias. 39 Disagreements in quality ratings were resolved by consensus between the 3 reviewers involved in the appraisal phase (JPS, TT, and SO).
Statistical Analysis
Standardized mean differences (SMDs) were calculated as the primary analysis outcome. We used the Hedges g statistic to correct for the influence of studies with small sample size. 41 All summary estimates in models were calculated using the restricted maximum likelihood (REML) method, incorporating nested random effects to take into account the unique differences of each stroke subgroup as well as the correlation (ie, pseudoreplication) of repeated measurements between and within study subgroups. 42 The REML method iteratively compares models based on a transformed set of data that limits the effect of nuisance parameters. The final selected model is the one that best explains the observed data.
Mixed-effects models were fit using intervention (yes/no), age levels, study quality, recovery stage, cognitive domain targeted in the studies, and stroke etiology in separate, metaregression models. All models were first run for the pooled data and then separately for intervention trials and observation cohorts for each of the mentioned variables.
Multivariate, mixed-effects models were fit to study associations between cognition, intervention categories, and time poststroke. Intervention studies (k = 43) were recoded into the 4 groups previously defined: (1) pharmacological, (2) therapist-led, (3) nonroutine/alternative, and (4) usual care (please see our Data Supplement, Section 4.1 for full details on this step). Time poststroke was recoded into 4 recovery stages: acute (1-60 days), subacute (61-180 days), chronic (181-729 days), and long-term (>729 days). 14 Interventions were then examined together with recovery stages in a multiplicative model to study the joint effect of time and intervention on cognitive recovery.
Publication bias was assessed with an adaptation of the Egger regression test by including the standard error of the effect size (ie, calculated SMD of each study subgroup) as a predictor in a REML model incorporating all studies along with random effects, as previously specified, and then intervention and observational studies separately. 43 Analyses were considered biased if the resulting intercept from these models differed from zero with P set at .10. 44
Model convergence and overfitting were evaluated using profile plots, as shown in sections 6 and 8 of our Data Supplement. Heterogeneity was assessed by inspecting forest plots and quantified with the Q and τ2 tests. Data from transient ischemic attack and healthy participants were used as a reference group, if provided separately in the included papers, to quantify the effect of different stroke etiologies. Associations (ie, SMD or effect estimates) were interpreted as small ≥0.2, medium ≥0.5, and large ≥0.8. 45 Post hoc, pairwise comparisons of summary estimates were performed with Tukey contrasts. 46
All analyses were completed using R v.3.5. The package for meta-analysis, metafor, was used to calculate effect sizes, compare models, and build itemized forest plots. 47 A detailed description of our statistical methods, including the R code, can be found in our Data Supplement.
Data Availability
All data are available in this article and in the Data Supplement. The raw data extracted from each paper can be accessed from our Github account on https://github.com/jpsaa/meta-analysis-cognition
Results
General Findings
Our search returned 4885 records. After removing duplicates, 3766 unique titles and abstracts were screened, and 623 reports were fully examined for data extraction. In total, 122 studies met our inclusion criteria, representing 229 study subgroups and 28 822 stroke survivors followed longitudinally. Studies were excluded for not reporting descriptive data (ie, mean and SD; 158 studies), being a cohort duplication (72 studies), not describing stroke cases separately (51 studies), not reporting a time poststroke (51 studies), and not evaluating cognition longitudinally (32 studies). Further details about included and excluded articles can be found in Figure 1.
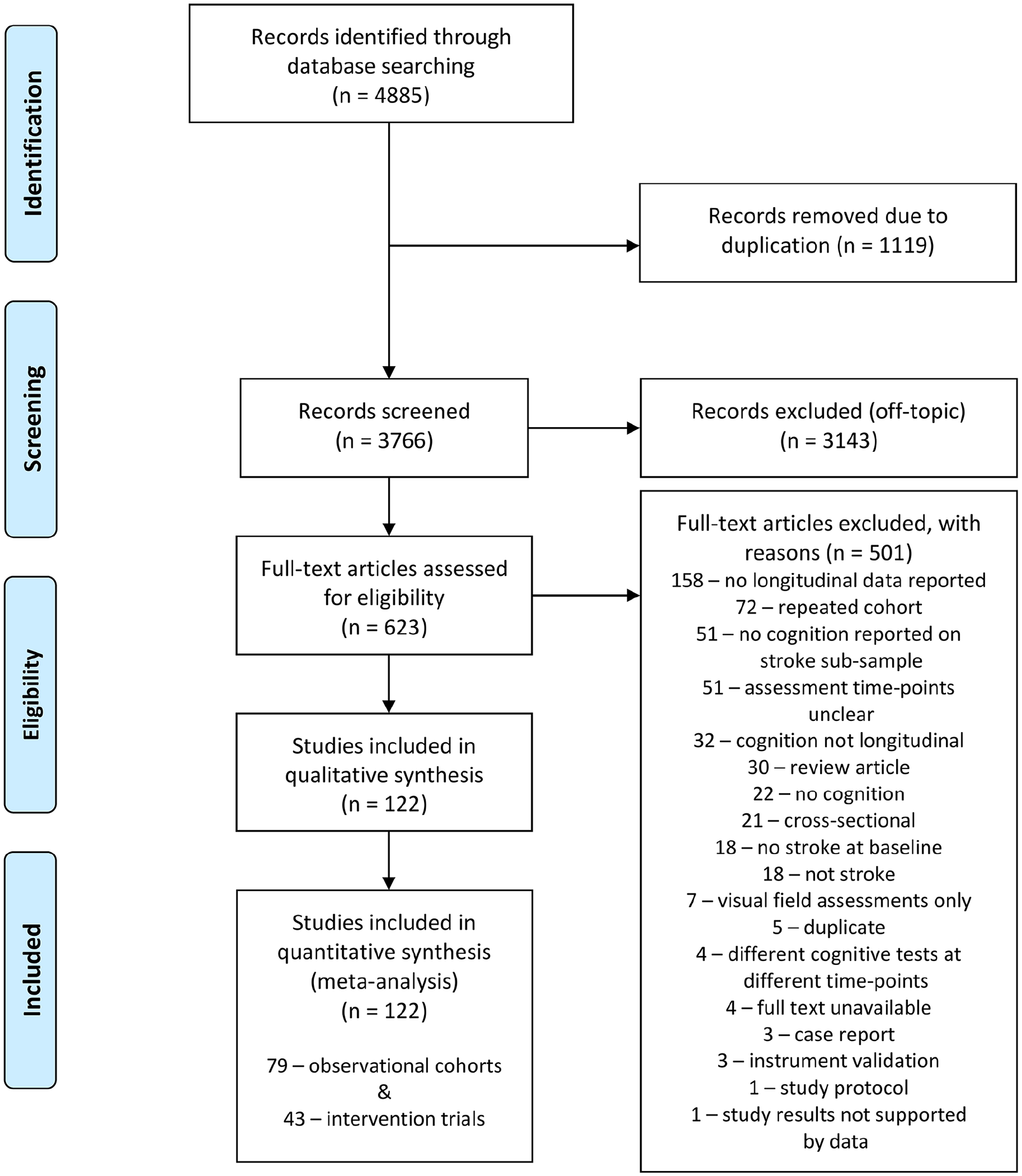
Study selection flow diagram.
The characteristics of the included studies and the types of study design are summarized in Tables 1 and 2, respectively. The median sample size across all studies was 54 participants (range 8-3680), and the median time interval from baseline to the last follow-up was 150 days (range 5.5-1795 days).
Summary of Included Studies.
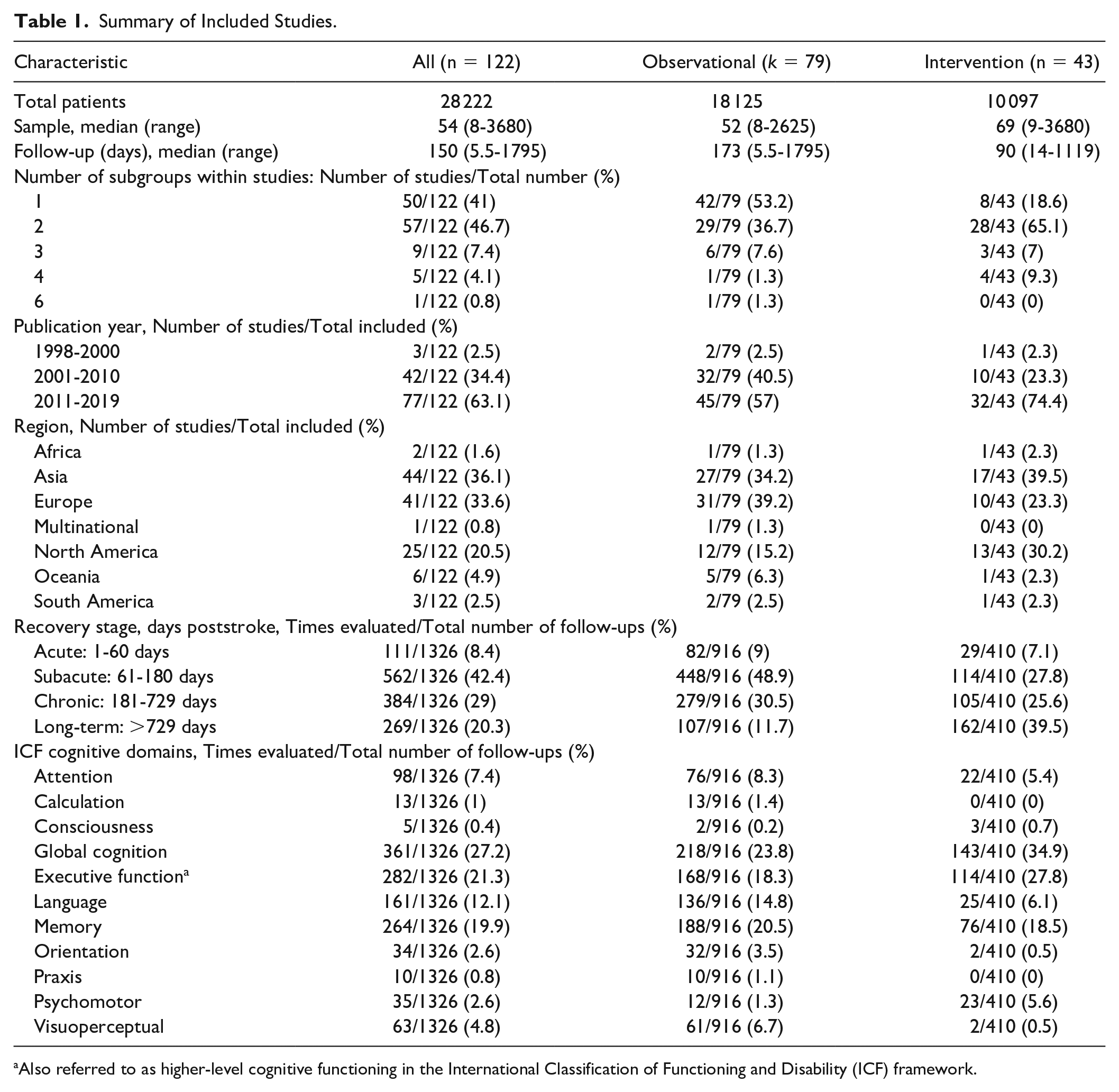
Also referred to as higher-level cognitive functioning in the International Classification of Functioning and Disability (ICF) framework.
Characteristics of the Study Designs Included.
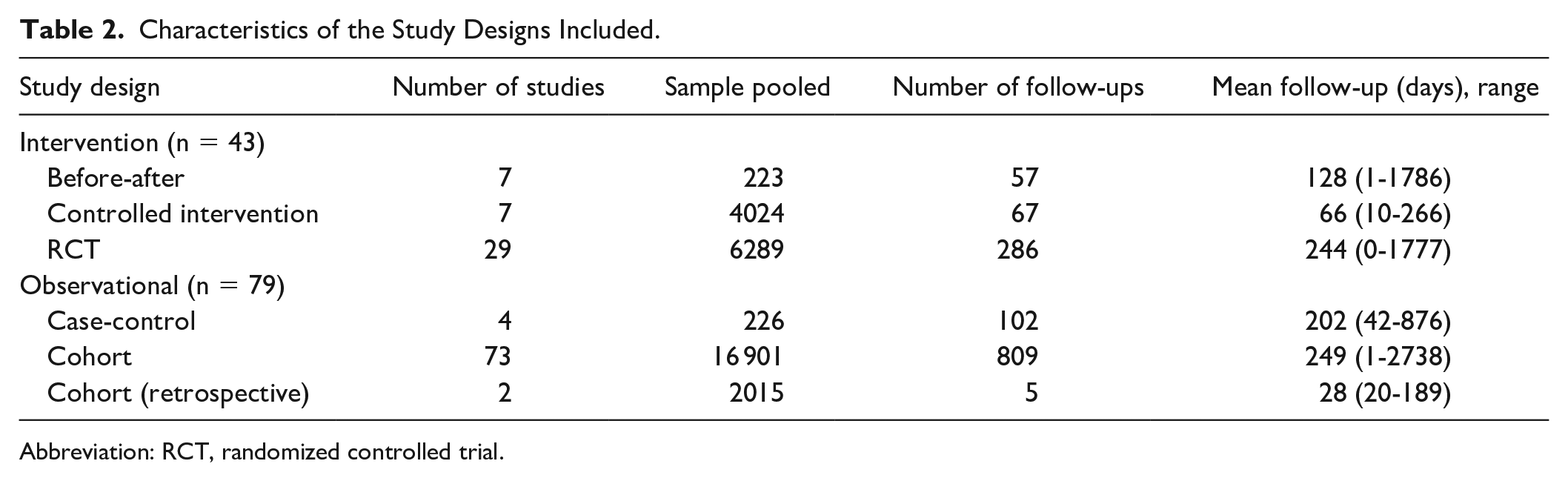
Abbreviation: RCT, randomized controlled trial.
The different evaluation tools included in this review are summarized in Figure 2 using a circular histogram. The cognitive domains primarily targeted across studies were global cognition (75%), memory (31%), executive function (30%), and language (21%).
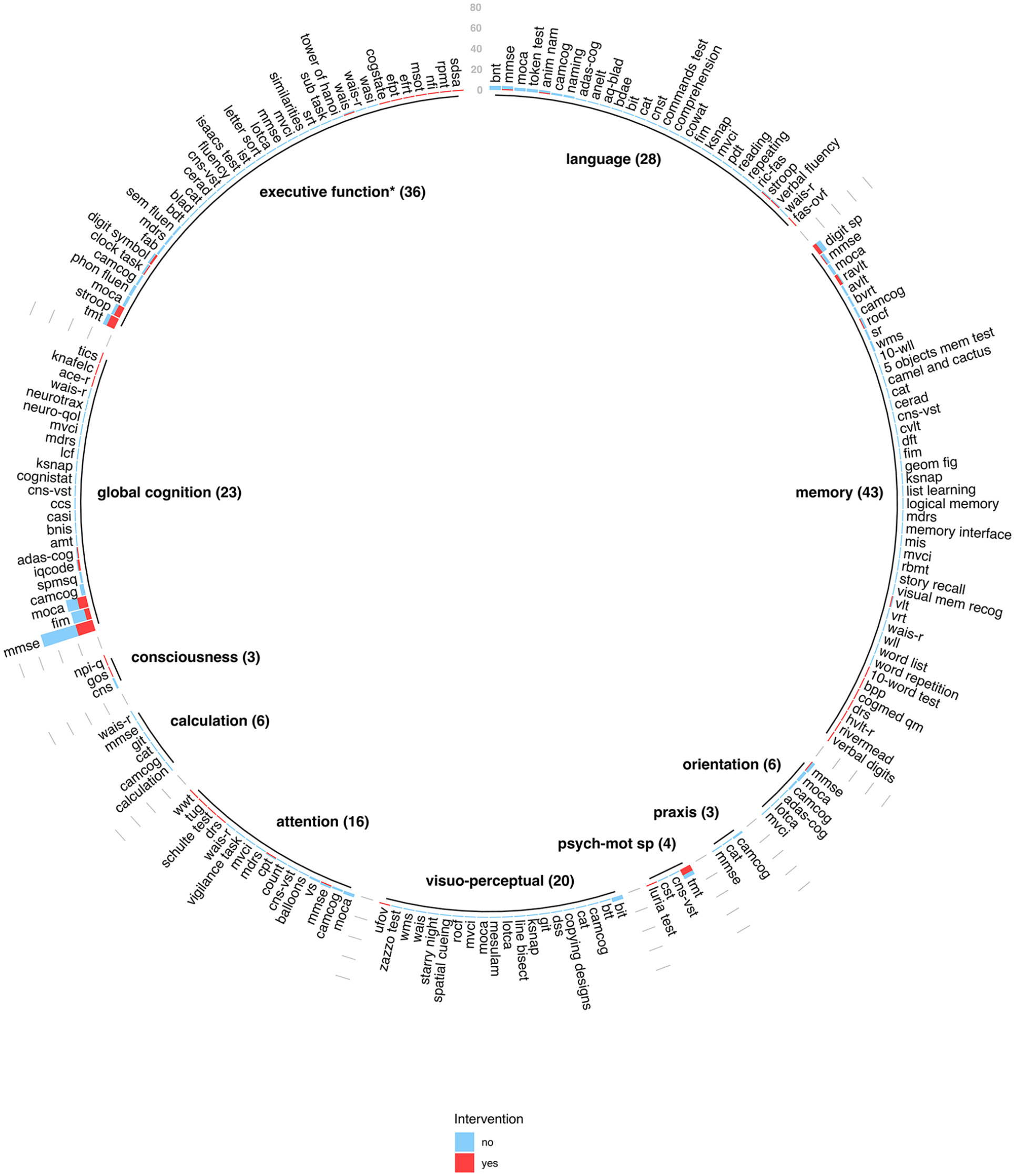
Cognitive domains and instruments included in the meta-analysis.
Figure 3 provides details about the estimates from individual meta-regression analyses performed across all predictor variables (ie, moderators), after adjusting for random effects. The overall recovery across all studies was small (g = 0.35; CI = 0.29-0.42). However, after stratifying by study type, these recovery effect sizes were greater for intervention (g = 0.47; CI = 0.37-0.58) than observational (g = 0.28; CI = 0.20-0.36) studies and across all moderator variables (ie, fixed effects) analyzed (ie, recovery stage, cognitive domain targeted in the study, stroke etiology, age group, methodological quality, and intervention type).
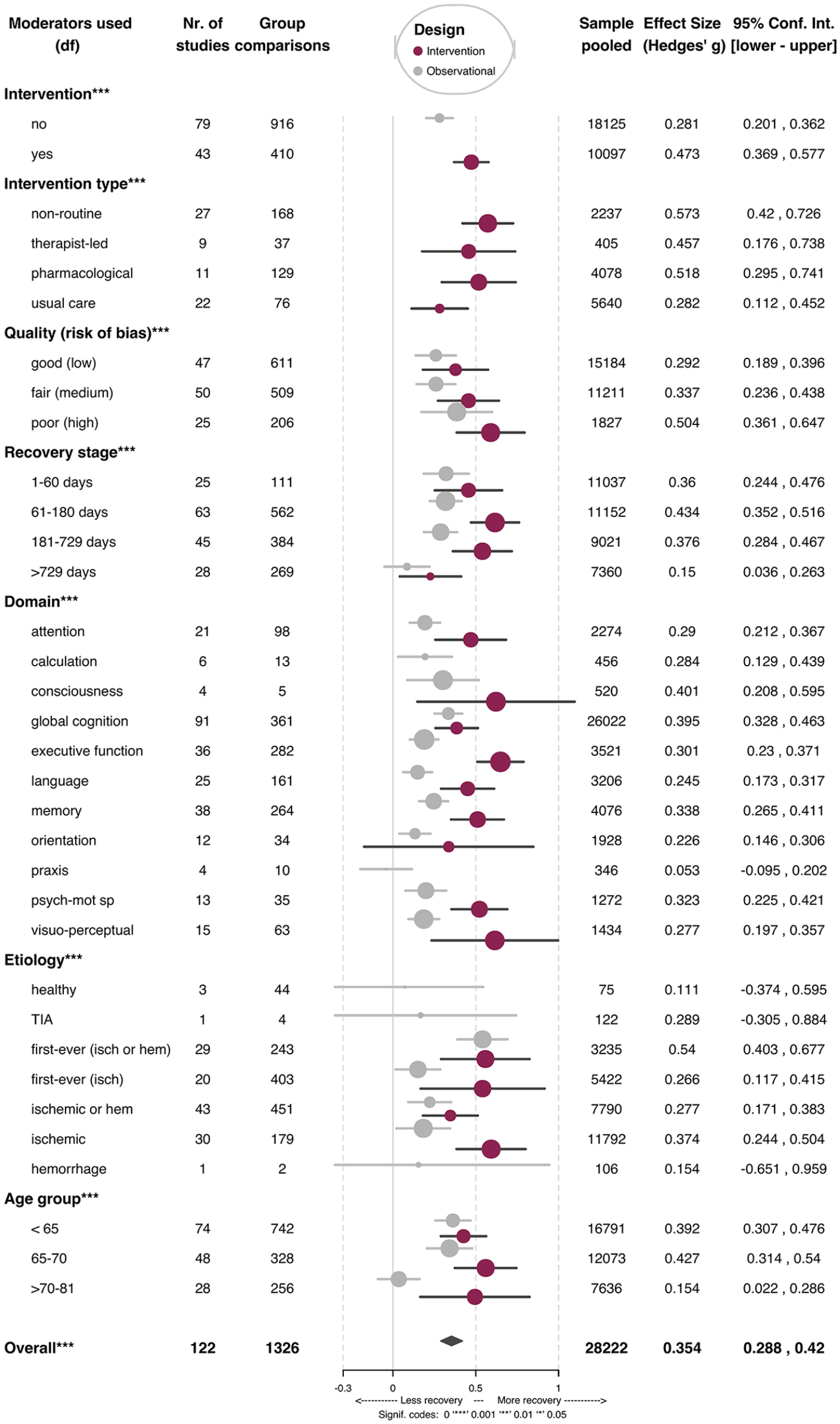
Average effect of moderators analyzed (mixed-effects model).
Intervention Studies
We included a total of 43 intervention trials (10 097 stroke survivors). No significant differences (P = .984) in cognitive recovery were found between randomized controlled trials (RCTs; g = 0.47, CI = 0.34-0.61) when compared with non-RCT intervention studies (g = 0.47; CI = 0.26-0.68). The type of intervention, however, did show significant differences in the calculated recovery estimates. More specifically, pharmacological (g = 0.52; CI = 0.30-0.74) and nonroutine/alternative treatments (g = 0.57; CI = 0.42-0.73) yielded medium overall effects, whereas therapist-led (g = 0.45; CI = 0.18-0.72), and usual care (g = 0.28; CI = 0.11-0.44) had a small effect. These differences were statistically significant when comparing usual care with nonconventional (P = .003) interventions via Tukey contrasts.
Time and Type of Intervention as an Interaction
Mixed-effects modeling using a multiplicative approach combining time and intervention type (Table 3 and Figure 4) yielded large treatment effects (ie, g ≥ 0.89) 1 to 60 days poststroke from studies using nonroutine/alternative treatments in the acute stage, medium-sized effects (ie, g ≥ 0.5) for therapist-led interventions, and small effects (ie, g ≥ 0.46) for pharmacological interventions.
Associations Between Cognition and Intervention Types at Different Recovery Stages (n = 43), Based on the Multiplicative Model. a
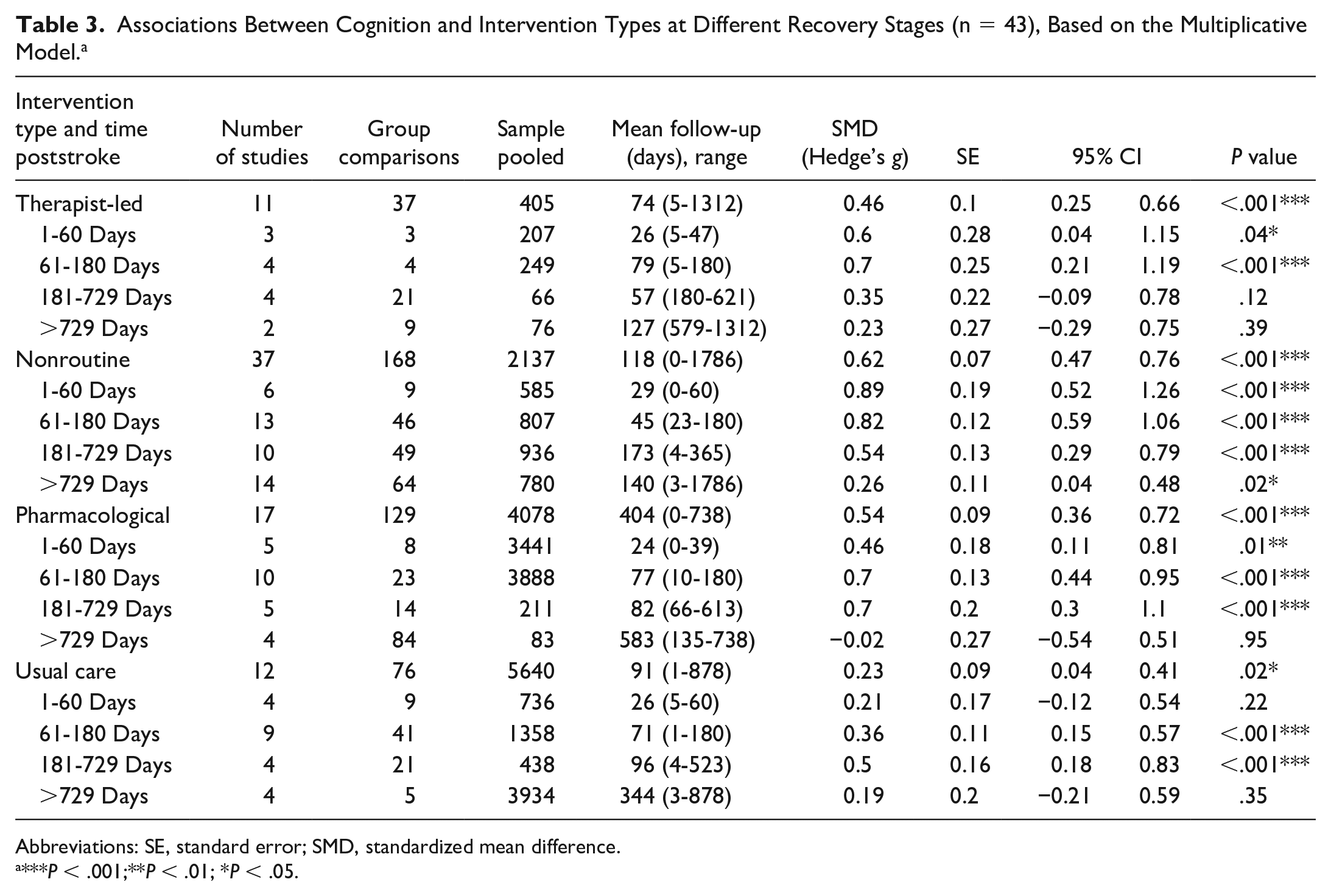
Abbreviations: SE, standard error; SMD, standardized mean difference.
***P < .001;**P < .01; *P < .05.
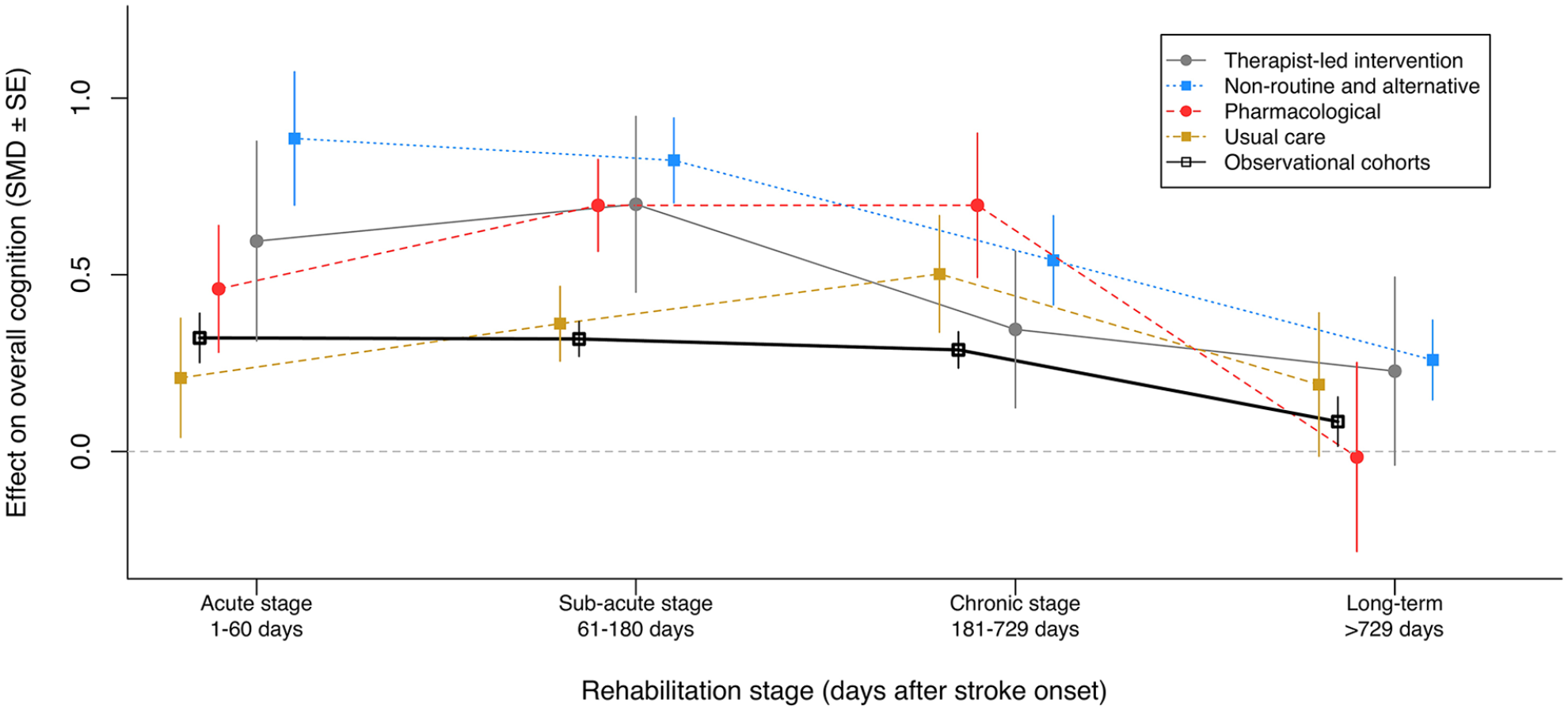
Average associations of intervention with cognitive function after stroke based on the multiplicative model.
Between 2 and 6 months poststroke, nonroutine interventions again returned strong effects on cognition (g ≥ 0.82), whereas therapist-led and pharmacological trials showed medium effects (g = 0.70 for both). Usual care interventions in this phase had a small (g = 0.36) and significant effect on cognitive recovery.
From 6 months to 2 years poststroke, pharmacological, nonconventional, and usual care interventions showed medium effects (ie, g = 0.70, g = 0.54, respectively, and g = 0.50, respectively), whereas therapist-led interventions showed nonsignificant effects on cognitive recovery.
After the 2-year period, controlled interventions showed no significant effects on cognitive recovery when analyzed separately, except for nonconventional/alternative therapies, which had a small and significant effect (g = 0.26).
Recovery Stage
Our overall, pooled analyses showed the largest improvement in the subacute stage (61-180 days poststroke; g = 0.43, CI = 0.35-0.52), followed by the chronic (181-729 days; g = 0.38, CI = 0.28-0.47), acute (1-60 days; g = 0.36, CI = 0.24-0.48), and long-term (>729 days; g = 0.15, CI = 0.04-0.26) stages. These estimates became larger after analyzing intervention studies separately, with the subacute stage showing the largest improvement value (g = 0.62; CI = 0.47-0.76), followed by the chronic (g = 0.54; CI = 0.36-0.72), acute (g = 0.46; CI = 0.25-0.66), and long-term stages (g = 0.23; CI = 0.04-0.41). For details about all analyses looking at the outcomes from intervention trials and observational cohorts, please see Supplemental Table IV.
Cognitive Domains
The contribution of different cognitive domains to overall cognitive recovery was small across all the mental functions analyzed (Figure 2). After examining intervention trials and observational cohorts separately, the latter continued to yield small associations across domains (see Supplemental Table IV for details), whereas intervention trials yielded medium-sized recovery estimates for executive function (g = 0.65; CI = 0.51-0.79), psychomotor speed (g = 0.52; CI = 0.35-0.69), visuoperceptual function (g = 0.62; CI = 0.23-1.00), memory (g = 0.51; CI = 0.35-0.67), and consciousness function (g = 0.62; CI = 0.15-1.10). Global cognition (g = 0.38; CI = 0.26-0.51), attention (g = 0.47; CI = 0.26-0.68), language (g = 0.45; CI = 0.29-0.61), and orientation (g = 0.34; CI = −0.17 to 0.85) exhibited small associations even in the presence of a controlled intervention.
Etiology and Age
Stroke etiology exhibited medium associations with cognitive recovery in first-ever mixed stroke (ie, ischemic or hemorrhagic) studies (g = 0.54; CI = 0.40-0.68) and small associations in studies including only first-ever ischemic stroke (g = 0.27; CI = 0.12-0.42). The latter group showed marked improvement when in the presence of a controlled intervention (g = 0.54; CI = 0.17-0.92) when compared with observational studies (g = 0.15; CI = 0.02-0.29). Studies that included participants with previous stroke showed better estimates if they consisted of ischemic stroke survivors only (g = 0.37; CI = 0.24-0.50), rather than mixed stroke (ie, ischemic or hemorrhagic; g = 0.28, CI = 0.17-0.38). The former also showed greater recovery estimates when undergoing controlled interventions (g = 0.59; CI = 0.38-0.80) when compared with mixed stroke studies that included individuals with previous stroke (g = 0.35; CI = 0.18-0.51).
In pooled analyses, cognitive recovery estimates were larger for stroke survivors aged 65 to 70 years (g = 0.43; CI = 0.31-0.54) and <65 years (g = 0.39; CI = 0.31-0.48) than for older groups (>70-81 years; g = 0.15, CI = 0.02-0.29). As was the case for all moderators, independent modeling by intervention (yes/no) revealed stronger estimates of recovery across all age groups. Notably, older stroke survivors (ie, >70-81 years) showed a significant improvement only when being part of controlled interventions (g = 0.41, CI = 0.05-0.77, vs g = 0.08, CI = −0.06 to 0.21, for observational cohorts).
Methodological Quality
Quality assessment of the included investigations resulted in 47 “good” (39%), 50 “fair” (41%), and 25 “poor” (20%) studies, indicating low, some, and high risk of bias, respectively. Mixed-effects models yielded significantly higher effects (P = .019) in studies with “poor” quality rating (g = 0.50; CI = 0.36-0.65) than in those with “good” methodological strength (g = 0.29; CI = 0.19-0.40). This difference was not significant (P = .062) when comparing studies with “poor” versus “fair” quality ratings (g = 0.34; CI = 0.24-0.44).
Heterogeneity and Publication Bias
Overall, heterogeneity was significant [Q(1278) = 6306.67; τ2 = 0.09; CI = 0.01-0.21; P < .001] after accounting for the correlation within and between study subgroups (ρbetween = 0.42, CI = 0.07-1.00; σwithin = 0.57, CI = −1.00 to 0.84). Similarly, intervention trials [Q(408) = 1498.08; P < .0001] and observational studies [Q(864) = 4258.69; P < .0001] had significant heterogeneity between study subgroups when analyzed separately.
No publication bias was evident in our overall analysis examining intervention and observational studies together (P = .1280). Sensitivity analysis revealed no publication bias (ie, P ≥ .10) for intervention trials (P = .100), and some publication bias for observational studies (P = .086) when analyzed separately.
Discussion
Our findings provide evidence that cognition can, and usually does, improve throughout the poststroke recovery stages. Such improvement is significantly greater during the first 6 months poststroke and begins to wane by the end of the second year. Previous systematic studies on cognitive recovery by Cicerone and colleagues have classified the available evidence for various types of brain injury based on methodological strength.49-52 In the present review, we pooled this evidence and supplied a quantitative estimate of cognitive recovery for brain injury associated with stroke across different recovery phases.
Overall, the pooling of cognitive outcomes from studies with different intervention designs, as recommended by previous reports,28,31,32 revealed that these studies are associated with better cognitive recovery than observational cohorts across all recovery stages, cognitive domains, stroke etiologies, and age groups analyzed. Moreover, the data gathered from observational cohort studies allowed us to characterize with relatively high confidence a benchmark in terms of the progression of cognitive recovery, when no additional interventions other than the standard care offered in each stroke rehabilitation setting are present.
Intervention Type, Stage of Recovery, and Quality of Evidence
One particularly important finding from the present review was that intervention trials demonstrated significant and varying effects on cognition at different times of recovery. These differences became apparent after examining the interaction between intervention type and time after stroke, in a multiplicative model. Our results suggest a small but significant effect on cognitive recovery in the first 1 to 60 days poststroke across interventions and a large effect of nonroutine/alternative therapies after stratifying estimates in this phase by treatment type. Studies with nonroutine/alternative therapies had in common the use of therapeutic methods that require further investigation and validation. Examples of nonroutine therapies with strong effects in this phase were stenting in combination with thrombolysis, 53 virtual reality, 54 and robot-assisted therapy/ergometry for the upper limb. 20 All these treatments were administered in addition to usual care and were appraised as having fair (eg, Shi et al 53 and Rogers et al 54 ) and good (eg, Rabadi et al 20 ) methodological quality. Moreover, in some cases, these interventions were designed to also target recovery in other areas. For instance, Rabadi et al 20 targeted motor, cognitive, and functional recovery. For details about the effect size of each intervention within each study please refer to the itemized forest plots available in our Data Supplement, section 8.
Nonconventional/alternative treatments again achieved large effects on recovery cognition in the subacute phase (61-180 days poststroke), whereas pharmacological interventions had a medium overall effect. Pharmacological studies used a variety of active drugs such as antidepressants (eg, citalopram 55 or duloxetine 56 ) and cognitive stimulant drugs (ie, methylphenidate), 24 which were administered in addition to standard therapy. Pharmacological trials were small for the most part and varied from poor (eg, Simis and Nitrini 55 and Delbari et al 24 ) to fair (eg, Zhang et al 56 ) methodological rigor. The largest trial (n = 3644) in this recovery phase 57 tested a certified Chinese herb–based antioxidant medicine (Cerebralcare Granule) and compared it with aspirin, achieving fair methodological quality and medium-sized treatment effects in favor of the former (g = 0.67 Cerebralcare Granule; g = 0.41 aspirin). Similarly, nonroutine interventions at this stage that provided evidence of strong effects on cognition were again virtual reality, 54 robot-assisted therapy, and ergometry, 20 all coupled with usual care, as in the acute stage. Strong treatment effects were also achieved by a multicenter controlled trial of 360 stroke participants, 58 using acupuncture and herbal medicine in addition to usual care. This last trial was appraised as having good methodological rigor, and although it commenced treatment administration in the acute phase (<60 days poststroke), it did not show large benefits on cognition until the subacute phase (61-180 days poststroke). The findings pertaining to this phase suggest that the period of greatest recovery of cognition might extend beyond the acute stage (ie, >2 months poststroke), especially when using nonalternative approaches. The available evidence on recovery of cognition beyond the acute phase is mixed, with previous meta-analyses indicating both significant and nonsignificant recovery in the chronic phase (ie, up to 1 year poststroke).54,59 Our findings add to the field because they support the notion of an extended recovery period that goes into the chronic recovery stage when using raw outcomes that are appropriately adjusted (ie, by taking into account each study’s unique sample size, repeated measurements, and the presence of multiple comparison/treatment groups within the studies). 60 More research should corroborate these findings by examining recovery estimates with methods that are able to capture the intricacies of multidimensional data (eg, multiple subgroups within studies, multiple follow-ups, and sample attrition).
From 181 days (ie, 6 months) and up to 2 years poststroke, interventions trials revealed medium treatment effects. Pharmacological and nonroutine therapies continued to yield medium-sized effects; however, the difference between these 2 interventions was more marked, favoring the former (g = 0.71 for pharmacological vs g = 0.54 for nonroutine interventions). Interventions with pharmacological approaches resulting in strong effects used methylphenidate and levodopa as the main active drug.24,61 Most studies in this phase were also limited by both a small sample size (n < 20) in the experimental groups and poor methodological quality. An exception to this trend was a randomized trial of 119 survivors undergoing treatment with nimodipine, which showed medium recovery estimates on cognition (g = 0.69) and good methodological quality. 23 Among all nonroutine interventions/alternative therapies, the same study using nimodipine 23 returned even better results when combining this drug with acupuncture (g = 1.60). Similarly, a study of 195 stroke participants using a multifactorial, secondary prevention approach to reduce vascular factors (education + targeted counseling + pharmacological intervention) resulted in a medium treatment effect (g = 0.56). 62 The methodological quality of this study was also good.
The last recovery stage analyzed (ie, ~2 years and up to 5 years poststroke) had interventions with no significant effect on cognition. However, as highlighted in our results, nonroutine interventions were the only intervention category that achieved significant but small treatment effects. Among these interventions, one study achieved strong effects when using a 6-week computerized cognitive training intervention. 63 However, this study was small (n = 26) and was appraised as having poor methodological quality. A second, larger study of 144 stroke participants tested a multidisciplinary home-treatment approach. 64 This study returned a small and significant effect on cognition (g = 0.49), but also presented high risk of bias. The only study in this phase with good methodological quality showing a small but significant effect on cognition (g = 0.30) used a combination of stenting with aggressive pharmacological and behavioral treatment.65,66 Notably, participants in this trial were treated and followed up at regular 4-month intervals from the first week, and up to 3.08 years post–stroke symptom onset.
The representation of different interventions in the present review was considerably less for therapist-led (405 participants) when compared with that of nonroutine (n = 2137), pharmacological (n = 4078), and usual care (n = 5640) studies. The small pooled sample for therapist-led studies is comparable in size to previous systematic reviews examining associations of therapist-led interventions with global cognition, 26 memory, 67 visuoperceptual functions (eg, neglect), 68 attention, 69 and executive function, 4 all of which had pooled samples that ranged from 223 to 630 participants, excluding comparison groups. This finding highlights the need for larger high-quality studies of therapist-led interventions, especially in the chronic and long-term stages.
Effect of Different Cognitive Domains on Overall Cognitive Recovery
Cognitive domains had distinct associations with recovery when analyzed separately in intervention and observational studies. Post hoc exploration of domain-specific estimates yielded medium-sized associations with recovery in most domains when comparing interventions versus observational studies. Among the domains analyzed, executive function showed the largest association with overall cognitive recovery from intervention compared with observational studies, followed by conciousness, visuoperceptual, psychomotor speed functions and memory. Recovery of global cognition, attention, language, and orientation remained small (though higher than for observational cohorts), even in the presence of an intervention. These results are consistent with recent systematic studies examining associations of therapist-led interventions for some of the mentioned domains (ie, attention, memory, visuospatial functions, and global cognition). 26 Differences in domain-specific estimates of recovery with respect to previous systematic reviews may be explained by our statistical approach and the number of studies included in the present review. For instance, we analyzed study subgroups separately (ie, did not pool outcomes from the same study) and included 9 additional trials looking at executive function when compared with a recent systematic review. 26 Our differences may also be influenced by our broader inclusion criteria and our classification of cognitive domains, which adopted the use of standardized, validated rules, 34 and did not rely on profesional judgment of commonly used typologies, as has been the case in previous systematic studies looking at cognitive recovery poststroke.25,26
As for the instruments used, few studies used functionally oriented assessments of cognition.54,70-73 Findings from previous systematic reviews have revealed a similar pattern,26,69,74 highlighting the need to include these instruments. The association of cognition with functional outcomes is extensively documented and represents a common concern for stroke survivors.6,74 Evidently, the introduction of these assessments represents a way to move one step closer to understanding some of the most pressing needs of stroke survivors, such as the return to productive activities, driving safely, and participating in the community.7,74,75 Similarly, studies should aim to include survivors who are often underrepresented. People with aphasia, for instance, are reportedly excluded from studies under the assumption that they cannot consent or be adequately investigated, despite the existence of adapted and validated tools that take these issues into consideration. 76
Effect of Age and Stroke Etiology on Cognitive Recovery
Age groups and stroke etiologies demonstrated varying effects on cognitive recovery depending on the group/etiology evaluated and the presence/absence of a controlled intervention. Our results suggest greater cognitive recovery estimates from intervention studies on participants aged 65 to 70 years with first-ever stroke and on studies that included patients with a history of previous stroke who present with a current, ischemic stroke. These 2 variables, although present in all included studies, have not been formally assessed in recent meta-analyses looking at overall25,26 or domain-specific cognitive recovery.4,67-69 Therefore, further research on stroke type and age groups is recommended in future meta-analyses to corroborate our findings.
Overall, our analyses of observational cohorts and intervention trials represent new evidence relative to recent meta-analyses on the type and time of intervention and on the role of moderators in cognitive recovery.25,26,69 Observational cohorts as a group provide substantial evidence on the trajectory of short- and long-term cognition in the absence of interventions, whereas intervention trials offer approaches to enhance recovery of cognition poststroke. Our formal inclusion of pharmacological and nonroutine interventions such as music-based therapy, meditation, yoga, mindfulness, herbal medicine, acupuncture, and other mixed therapies was central to the present study. These interventions often exhibited stronger associations with recovery than therapist-led interventions, though they have normally been excluded from other systematic reviews of cognition (eg, Merriman et al, 25 Rogers et al, 26 Loetscher et al 69 ). This new evidence improves the understanding of cognitive recovery over time based on meta-analysis of subgroups and meta-regression of factors affecting cognition. The present review should also assist future studies in refining and more clearly articulating the differences between different interventions. This will, in turn, allow for a more rigorous assessment and comparison of potential benefits on cognitive recovery.
Limitations and Recommendations for Future Research
This study had several limitations, including our search strategy, which captured cognition primarily using a set of commonly investigated cognitive domains (ie, overall cognition, memory, executive function, attention). Although other important domains—such as language or perceptual functions—were captured under the umbrella of “cognition,” our results were limited for praxis and perceptual abilities. These 2 domains are defined within the ICF and are also research priorities in stroke rehabilitation; 14 thus, including them more explicitly would likely improve their representation in future studies.
A second limitation was that data for different stroke etiologies (eg, hemorrhagic vs ischemic) were in most cases not provided separately for the follow-up evaluations. Considering this limitation, our team stratified studies by etiology (eg, ischemic or hemorrhagic; first stroke or recurrent). The calculations resulting from etiology strata provided some explanation in the variability of cognition between studies; however, this analysis method was only a workaround for the missing data on etiology. Future reports would benefit from reporting follow-ups stratified by stroke type. This information will, in turn, allow meta-analysts to make more accurate accounts on stroke etiology and its associations with cognitive recovery.
As mentioned in our results, most potentially eligible studies (k = 501) did not report enough data to meet our inclusion and were, therefore, excluded. Our power has also been reduced because we included only studies reporting nontransformed data, and we accounted for all study dropouts, where applicable. Similarly, although our team made sure to get the most accurate and error-free data, it is unlikely that all cognitive descriptive outcomes met the assumption of normality, even when they were reported using parametric descriptors (mean ± SD).
Altogether, these issues made the mapping of cognitive trajectories extremely challenging, reducing our capacity to provide a more accurate description of cognition after stroke. We recommend that future longitudinal studies improve on the reporting of the aforementioned factors (eg, stroke etiology and/or initial severity, summary of cognitive scores over time, and study attrition) to enhance the quality of evidence available for future meta-analyses.
Conclusion
Our systematic review and meta-analyses provide a comprehensive summary and synthesis of the trajectory of global and domain-specific cognition and cognitive recovery poststroke. We identified several interventions associated with improvement in cognitive function and provided a benchmark of recovery with evidence from observational cohorts across different stages of recovery. Stroke etiology, age group, time poststroke, and the cognitive domains targeted in the studies (especially executive function) were identified as significant moderators of cognitive recovery. A broader description that is inclusive of the role of cognition in functional recovery and participation remains an important area of investigation in stroke. The use of the ICF can assist professionals and researchers to organize this knowledge. Collectively, the findings from this review, along with the methods and raw data shared in our Data Supplement, provide new evidence about the nature and extent of cognitive recovery over time and advance current understanding of the type and timing of interventions that may improve cognition after stroke.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211017501 – Supplemental material for Cognitive Recovery After Stroke: A Meta-analysis and Metaregression of Intervention and Cohort Studies
Supplemental material, sj-pdf-1-nnr-10.1177_15459683211017501 for Cognitive Recovery After Stroke: A Meta-analysis and Metaregression of Intervention and Cohort Studies by Juan Pablo Saa, Tamara Tse, Carolyn M. Baum, Toby Cumming, Naomi Josman, Miranda Rose, Sophie O’Keefe, Katherine Sewell, Vinh Nguyen and Leeanne M. Carey in Neurorehabilitation and Neural Repair
Footnotes
Authors’ Note
JPS, LMC, CMB, TT, and TC developed the concept and design. JPS prepared the PROSPERO protocol and planned and completed the searches. JPS and TT did the screening. JPS, VN, and KS extracted the data. JPS, TT, and SO evaluated study quality. JPS performed the statistical analysis and drafted the manuscript and the data supplement. All authors participated in the critical revisions of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: James S. McDonnell Foundation (Cognitive Rehabilitation Collaborative Award 220020413), Commonwealth Science and Industrial Research Organisation (CSIRO) flagship collaborative fund, National Health and Medical Research Council of Australia (NHMRC, Awards 1077898, 1153236, 1113352 and 2004443), and a La Trobe University Postgraduate Research Grant (LTUPRS).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
