Abstract
Following traumatic peripheral nerve injury, adequate restoration of function remains an elusive clinical goal. Recent research highlights the complex role that the immune system plays in both nerve injury and regeneration. Pro-regenerative processes in wounded soft tissues appear to be significantly mediated by cytokines of the type 2 immune response, notably interleukin (IL)-4. While IL-4 signaling has been firmly established as a critical element in general tissue regeneration during wound healing, it has also emerged as a critical process in nerve injury and regeneration. In this context of peripheral nerve injury, endogenous IL-4 signaling has recently been confirmed to influence more than leukocytes, but including also neurons, axons, and Schwann cells. Given the role IL-4 plays in nerve injury and regeneration, exogenous IL-4 and/or compounds targeting this signaling pathway have shown encouraging preliminary results to treat nerve injury or other neuropathy in rodent models. In particular, the exogenous stimulation of the IL-4 signaling pathway appears to promote postinjury neuron survival, axonal regeneration, remyelination, and thereby improved functional recovery. These preclinical data strongly suggest that targeting IL-4 signaling pathways is a promising translational therapy to augment treatment approaches of traumatic nerve injury. However, a better understanding of the type 2 immune response and associated signaling networks functioning within the nerve injury microenvironment is still needed to fully develop this promising therapeutic avenue.
Introduction
Every year, over 3 million upper extremity traumas and over 4 million lower extremity traumas are estimated to occur in the United States.1,2 Of these, approximately 3% and 2%, respectively, involve peripheral nerve injury (PNI).3,4 PNIs can result in severe motor and sensory deficits, as well as neuropathic pain that have devastating impact on quality of life. 5 In an attempt to ameliorate these symptoms, patients can undergo nerve repair surgery, yet despite these efforts, restoration of function following surgery still produces poor outcomes: in median and ulnar nerve reconstructions, 52% and 43% achieve satisfactory motor and sensory recovery, respectively. 6 These suboptimal surgical outcomes are due to factors that limit nerve regeneration: neuronal death following injury, a decreased capacity for neurons to regenerate axons over time, the slow growth of regenerating axons compounded with long distances between lesion site and target, misdirected axonal regrowth to targets, and potentially irreversible chronic denervation-induced muscle atrophy.7-10 Thus, there is a great need to develop therapies that will enhance nerve regeneration and improve functional recovery following PNI.
The immune system plays a crucial and intricate role throughout the wound healing process. Immune cells, while low in quantity within healthy peripheral nerve, are swiftly recruited to nerve tissue in response to injury: first neutrophils, followed by monocytes/macrophages, and then lymphocytes. These immune cells respond to injury initially by phagocytosing debris and secreting cytokines to further attract leukocytes from circulation and to polarize one another toward pro-inflammatory phenotypes. With time this type 1 immune response, mediated by pro-inflammatory “M1” macrophages and type 1 T-helper cells, evolves into a pro-regenerative, healing phenotype mediated by “M2” macrophages and type 2 T-helper cells. This pro-regenerative, type 2 immune response resolves inflammation to facilitate regeneration and ultimately restore functionality of the nerve. Specifically, some cytokines have emerged as interesting candidates that could play important roles in guiding the immune and resident cells in injured tissue toward pro-regenerative processes. Interleukin 4 (IL-4) is of particular interest as it is the principal cytokine governing wound healing in the type 2 immune response. IL-4 signaling in damaged nerve may play an important role in promoting regeneration and restoring function. In this review, we will focus on the role of IL-4 signaling, both on its endogenous role during the cascade of responses that follow traumatic nerve injury and regeneration and on its role when used as a potential exogenous therapeutic to manage nerve injuries.
Overview of Nerve Injury and Regeneration
At a macroscopic level, nerve includes myelinated axons of motor neurons and large sensory neurons, and the non-myelinated smaller axons of sensory and autonomic neurons, bundled into fascicles to compose a nerve fiber. Following nerve injury, different segments of nerve (axons vs soma) encounter and interact with different environments and cells (Schwann cells, macrophages, fibroblasts, microglial, etc) in a region-specific manner. As such, nerve injury can be viewed in a compartmentalized fashion (Figure 1), which we first review to provide context for the cells potentially involved in IL-4 signaling.
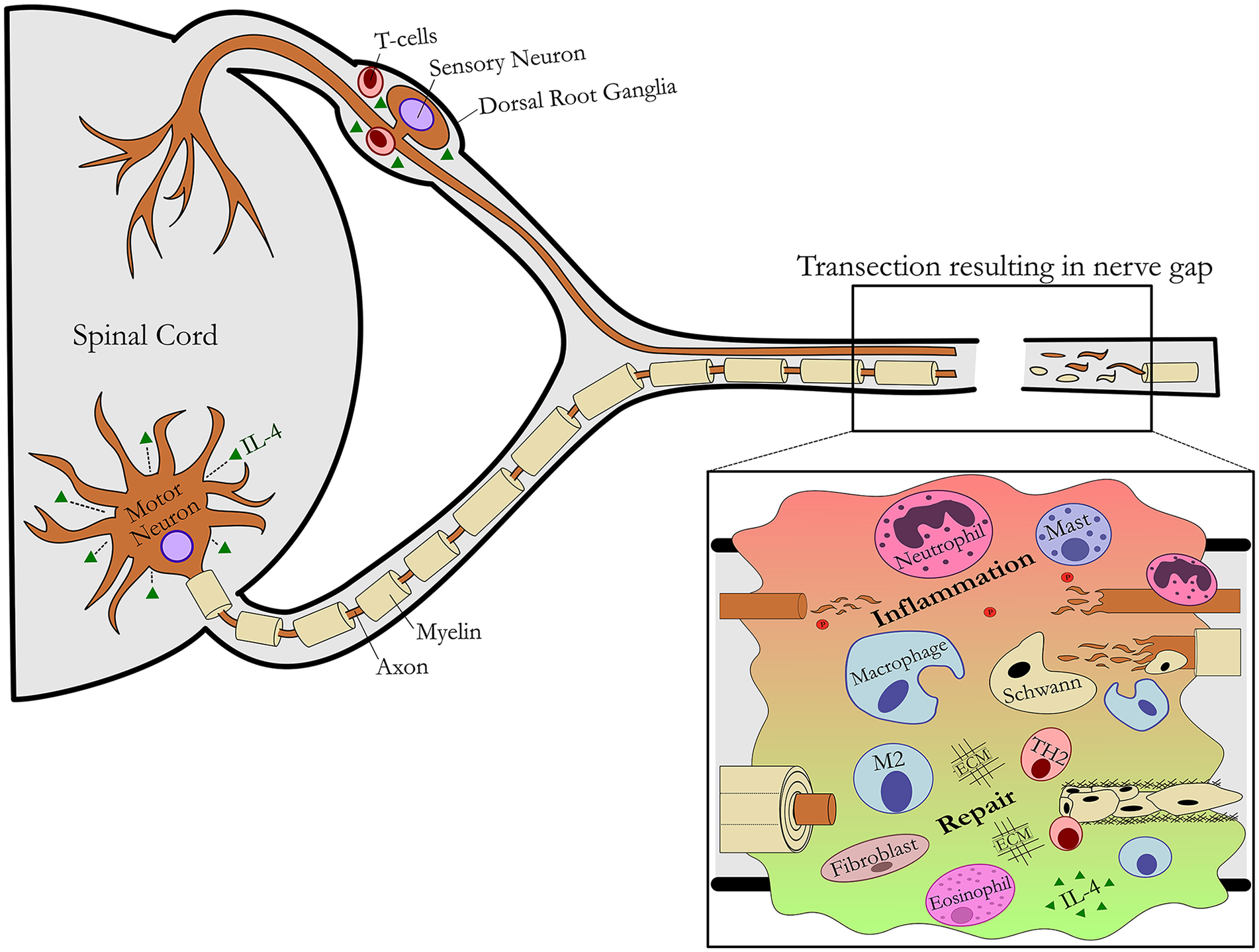
Schematic of nerve injury and regeneration. An overview of the affected central and peripheral nervous system compartments is shown in the context of a nerve transection resulting in a nerve gap.
Proximal Nerve and Neuronal Response
Following injury, a small amount of degeneration occurs in the axons of the proximal nerve, but these axons remain viable, as they are still intact with the neuronal soma. In these proximal axons, cytoskeletal and myelin degeneration occurs only up to the first node of Ranvier. With this, the axons undergo a dormant phase until genes are activated to facilitate axon growth. Proximal to the site of nerve injury, neurons sense compromised axonal function and upregulate a regeneration-associated gene (RAG) program in response (Figure 2). These genes become activated through the axonal trauma, as significant calcium influx, retrograde transport of kinases, and other pathways are triggered leading to activation of the RAG program. The RAG program includes expression of inflammatory signaling pathways, neurotrophic factors, and cytoskeletal elements to promote axon outgrowth from the proximal end of the damaged axon. Additionally, there is release of pro-inflammatory cytokines in the dorsal horn of the spinal cord by injury-activated glial cells, including microglial, where this action is potentiated by increased levels of neurotrophic factors locally. 11 Only neurons that survive the initial injury will be able to upregulate the RAG program and undergo regeneration. Motor neurons are generally spared from cell death if the axonal injury is distal enough from the cell bodies and the organism is mature,12,13 while sensory neuron death can be as high as 40% regardless of injury location and age.13-16
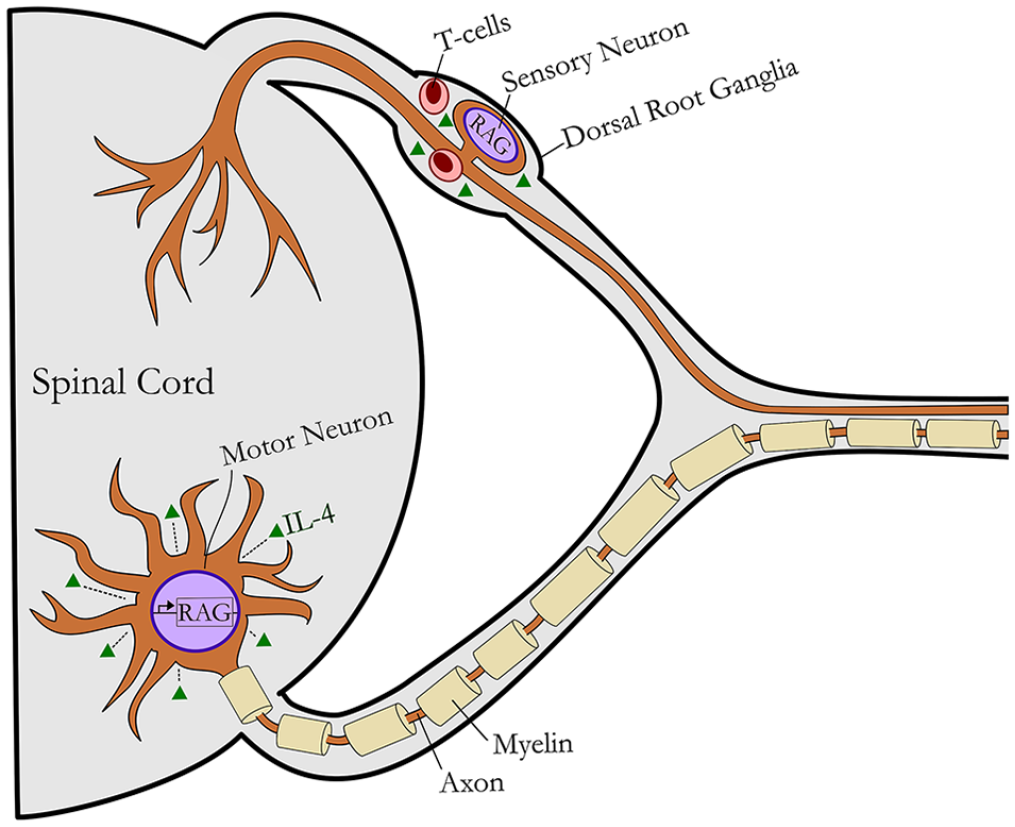
Focused schematic of the response to peripheral nerve injury within the central compartment and the cells implicated in interleukin (IL)-4 signaling. Following axotomy, injury-induced retrograde signals activate a regeneration-associated gene (RAG) program within neurons. This gene program results in release of pro-inflammatory cytokines and axon outgrowth. IL-4 signaling in the ventral horn and DRG further stimulate pro-regenerative processes within neurons, promoting cell survival and recovery. TH2 cells (effector T-cells) have been implicated as a source or regulator for IL-4 signaling post axotomy. IL-4 signaling to motoneurons promotes their survival, although its effects to sensory neurons is less clear despite the known accumulation of T-cells within dorsal root ganglia.
Distal Nerve Response
Following trauma to axons within a nerve, subsequent axonal degeneration occurs in the distal nerve. This axonal degeneration is necessary for nerve regeneration as it activates and recruits non-neuronal cells that clear the distal stump of debris and prepare it to receive newly sprouted axons from the proximal nerve. Schwann cells (SCs) respond to the absence of axonal contact by assuming a dedifferentiated, non-myelinating, phagocytic phenotype. These new pro-regenerative SCs begin expressing neurotrophic and pro-inflammatory cytokines that increase the permeability of the blood-nerve barrier, enabling neutrophils and then macrophages to arrive at the injury site. These hematogenous cells, along with resident macrophages in the nerve, assist SCs in phagocytosing debris and remodeling the extracellular matrix (ECM). Once cellular debris in the distal stump has been cleared, SCs begin aligning themselves along the basal lamina of the endoneurium to form hollow tubes, known as bands of Büngner, into which sprouting axons from the proximal nerve stump may grow to reach their end-organ targets.8,17 Within nerve, macrophages then experience anti-inflammatory signaling that promotes their conversion to a phenotype that promotes SC maturation. Additionally, axons express neuregulin type 1, which interacts with receptor ErbB2 on SCs, signaling to SCs to mature and myelinate. These processes lead to restoration of function.
Nerve Gap Response
In circumstances when nerve is severely injured resulting in a gap between the proximal and distal nerve, the response within the gap area is similar in ways to general, multiphase, wound healing processes. For most soft tissues, at the site of injury, blood coagulation and transient vasoconstriction occur, filling the tissue gap with a blood clot consisting of platelets, macrophages, and other immune cells, ECM, growth factors, and importantly, chemotactic and pro-inflammatory cytokines. These cytokines attract leukocytes from circulating blood, first neutrophils and then monocytes/macrophages, each phagocytosing cellular debris and enhancing inflammation and repair through cytokine release. Last, T lymphocytes arrive and further assist in promoting and ultimately resolving inflammation in the injured tissue. These late pro-regenerative signals promote fibroblast activity, neovascularization and angiogenesis, and long-term tissue remodeling.18,19
Similarly, in nerve gaps, immediately after a nerve has been injured, an array of factors are released including pro-inflammatory chemotactic factors to attract resident and circulating leukocytes (Figure 3). Extrinsic and intrinsic coagulation cascades convert fibrinogen to fibrin, which serves as a dense ECM upon which neutrophils, macrophages, fibroblasts, endothelial cells, and later SCs may migrate to bridge the proximal and distal nerve stumps. Neutrophils and then macrophages arrive at the injury site migrating upon the scaffold and begin phagocytosing debris and remodeling the scaffold in preparation for regeneration. Both tissue resident and later hematogenous macrophages secrete vascular endothelial growth factor (VEGF), a proangiogenic factor, recruiting endothelial cells to the nerve gap.20,21 As blood vessels form, fibroblasts and dedifferentiated SCs migrate along the endothelial basal lamina, forming cords similar in structure to the bands of Büngner in the distal stump. Concurrently, there is an influx of other myeloid cells (ie, eosinophils) and T lymphocytes promoting expression of anti-inflammatory cytokines.21-23 With these elements in place, the healing nerve gap is prepared to receive and guide sprouting axons from the proximal stump to the distal stump. The newly formed matrix of SCs, fibroblasts, and blood vessels in the nerve gap conduct the regenerating axons to the distal nerve stump, where processes proceed to facilitate axon growth to their end-organ targets as described previously.
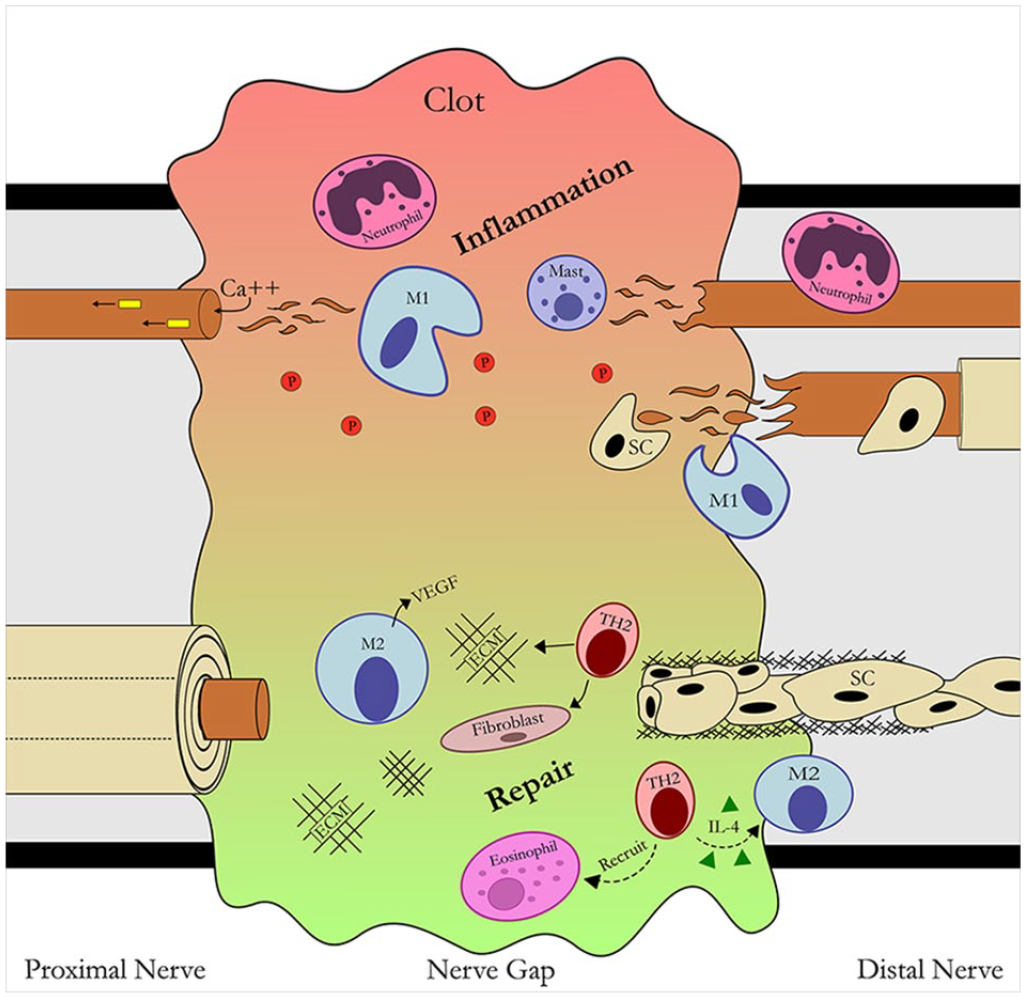
Focused schematic of the inflammatory and pro-regenerative phases of nerve post-axotomy. In the proximal nerve, retrograde signals and calcium influx signal neurons to injury activating a RAG program (see Figures 1 and 2). Within the nerve gap, the pro-inflammatory phase of injury involves leukocyte accumulation including platelets (P), neutrophils, macrophages (M), and inflammatory cytokines forming a clot to promote inflammation and a provisional extracellular matrix (ECM). This inflammation resolves as T-cells arrive in the nerve gap and promote pro-regenerative processes including interleukin (IL)-4 regulation and leukocyte modulation and recruitment. The pro-regenerative phase involves changes to macrophages from type 2 (ie, IL-4) cytokines leading to promotion of neovascularization, angiogenesis, ECM remodeling, and then recruitment of aligned Schwann cell (SC) channels within the gap. In the distal nerve, axons undergo Wallerian degeneration, where SCs dedifferentiate into a pro-inflammatory, phagocytic phenotype recruiting other phagocytic leukocytes. Following this inflammatory phase, these dedifferentiated SCs then align themselves into channels (bands of Büngner) to facilitate axon growth from the proximal nerve.
The Role of Inflammation and Endogenous IL-4 Signaling During Injury and Regeneration
During regeneration, the immune system establishes a balance between pro-inflammatory and anti-inflammatory processes (type 1 and type 2, respectively). The type 1 response is characterized by processes that enhance inflammation, such as the recruitment of leukocytes, expression of pro-inflammatory cytokines, and polarization of macrophages to the M1 phenotype. This inflammation indirectly promotes tissue regeneration by supplying chemotactic cytokines that attract cells required for wound healing. The type 2 response directly promotes tissue regeneration, characterized by the cytokines IL-4 and IL-13. These cytokines classically promote anti-inflammatory, pro-regenerative immune cell phenotypes, such as the M2 macrophage and type 2 helper T-cell (Th2). Considerably more is known regarding type 2 responses and IL-4 signaling in general wound healing, while less is known about these responses and signaling during nerve injury and regeneration, where each compartmentalized region of nerve could have important roles and unique cellular targets.
IL-4 Signaling in the Context of General Wound Healing
Interleukin-4 is a small, globular protein similar in structure to other cytokines. In response to injury, basophils, 24 eosinophils, 25 and mast cells 26 are classically associated as sources of IL-4 for the innate immune response (Figure 4A). But other innate immune cells, including NK T-cells 24 and neutrophils, 27 have been found to express IL-4 as well. In the adaptive immune response, type 2 helper (Th2 CD4+ T-cells) and follicular T-helper cells can be principal sources of IL-4 or regulate IL-4 expression from other cells, such as eosinophils. 28
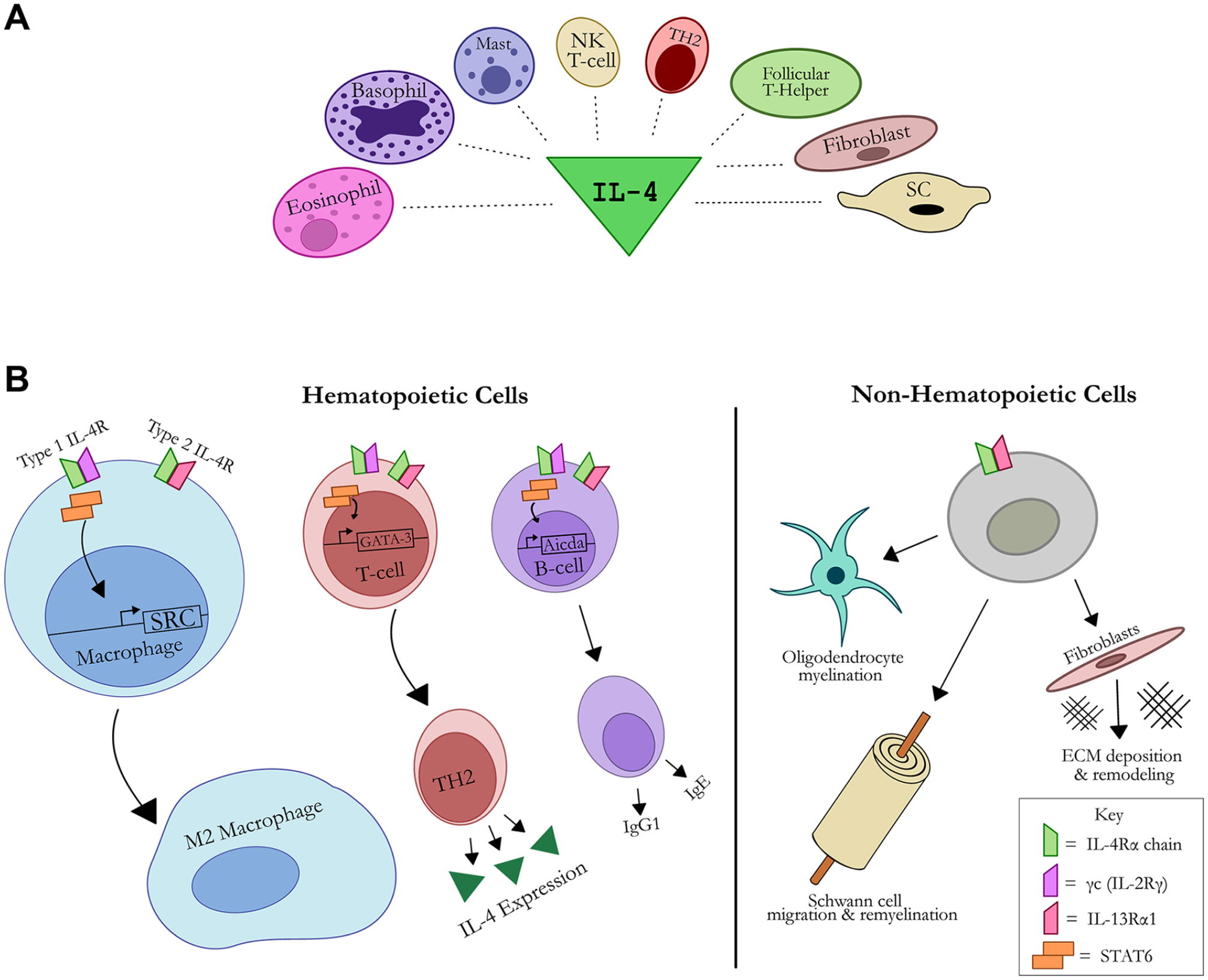
Schematic of interleukin (IL)-4 signaling sources (A) and targets (B). (A) Cell sources of IL-4 signaling include type 2 T-helper (TH2) cells, eosinophils, basophils, mast cells, natural killer (NK) T-cells, follicular T-helper cells, fibroblasts, and Schwann cells (SCs). IL-4 signals via IL-4R, a complex of either the type I (IL-4Rα and γc) or type II (IL-4Rα and IL-13Rα1) chains. The type I receptor complex activates Janus kinases (JAK), whereas type II (IL-13Rα1) activates tyrosine kinase 2 (TYK2) and JAKs, which in turn activate STAT-6 leading to dimerization and transmigration to the nucleus to transduce changes to response genes. (B) Cells of hematopoietic origin (ie, macrophages, T-cells, B-cells) express both type I and type II IL-4 receptors, whereas non-hematopoietic cells (ie, oligodendrocytes, SCs, fibroblasts) only express the type II IL-4 receptor. This signaling promotes cell-specific gene expression, such as promoting M2 macrophage phenotypes. In non-hematopoietic cells specific to nerve, IL-4 signaling promotes myelination in oligodendrocytes, migration and remyelination in SCs, and extracellular matrix (ECM) deposition and remodeling in fibroblasts.
IL-4 regulates target cell function and transcriptional activity through cell surface receptors (Figure 4B). Assembly of the IL-4 receptor complex is initiated when IL-4 binds the extracellular domain of the IL-4Rα chain. This high-affinity association between receptor and ligand enables the IL-4Rα chain to bind either the common gamma chain (γc, also known as the IL-2Rγ) or the IL-13Rα1 chain, respectively—forming functional type 1 IL-4R or the type 2 IL-4R complexes. 29 The IL-4Rα and IL-13Rα1 chains are expressed in both hematopoietic and non-hematopoietic cells. Contrastingly, the γc chain is expressed primarily in hematopoietic cells and is mostly absent in non-hematopoietic cells. In myeloid cells, such as macrophages, eosinophils, and mast cells, IL-13Rα1 and the γc chain are equally expressed, whereas in lymphocytes, there is increased expression of the γc chain compared to IL-13Rα1. 29
Once an IL-4-receptor complex has formed, intracellular Jak kinases become activated leading to downstream activation of STAT6. 29 STAT6 molecules dimerize and translocate to the nucleus, activating transcriptional machinery with diverse effects for each cell type. For example, IL-4-STAT6 signaling in macrophages upregulates Src kinase expression, promoting M2 phenotype differentiation 30 ; in B-cells the Aicda gene encoding AID (activation-induced cytidine deaminase) is upregulated, which is required for class switching 31 ; and in T-cells the GATA-3 transcription factor is upregulated, which promotes the Th2 cell phenotype. 32 Overall, IL-4 associated downstream signaling can be quite diverse based on the cell targeted.
IL-4 Signaling in the Context of Nerve Injury and Regeneration
While the immune system is regarded as a major source providing IL-4 within nerve, IL-4 is indeed expressed within cells specific to nerve after injury (Figure 4A). Systemically, blood plasma levels of IL-4 do not change following sciatic nerve crush in a rodent model. 33 But locally within nerve, an inflammatory and regenerative milieu has been implicated to produce IL-4. In both neuropathic and morphologically normal human sural nerves, immunohistochemical staining for IL-4 were found in myelinating SCs, immunoreactive lymphocytes, and putative macrophages and fibroblasts. 34
Interleukin-4 signaling associated with cell populations specific to nerve after injury and regeneration are still sources of ongoing investigations, but there is increasing evidence that IL-4 targets these cells (Figure 4B). In the central nervous system, which also contains select neuronal bodies of peripheral nerves such as motor neurons, many neuronal populations, astrocytes, and microglia have been found to express IL-4R. Additionally, IL-4R has been found not only on neuron cell bodies but also axons of central nervous system (CNS) neurons. 35 The role of IL-4 on injured neurons and their axons has primarily been studied in the CNS. IL-4 mediates CNS neuroprotection and regeneration following injury through inducing microglia and macrophages to a pro-regenerative phenotype36,37 and additionally promotes oligodendrocyte differentiation and subsequent remyelination. 38 But also, in a series of studies relevant for peripheral nerve injuries, Jones’ group elucidated a critical role for IL-4 in nerve regeneration following peripheral injury affecting cranial nerves. Using facial nerve transection models in mice, they showed that Th2 cells are necessary for facial motor nucleus (FMN) survival post axotomy and that these immune cells’ neuroprotective effects are dependent on IL-4 expression.39-44 Walsh et al found similar results in the CNS, showing an MHCII-independent neuroprotective role for a subset of IL-4 producing T-cells. 45 For spinal nerves, the presence of T-cells (and therefore, potentially IL-4) in the DRG and sympathetic ganglia has been reported following sciatic nerve transection46-49 (Figure 2). But the role of T-cells and IL-4 in promoting survival of sensory and sympathetic neurons, as well as spinal motor neurons, has yet to be elucidated. Furthermore, how T-cells affect the CNS to activate motoneurons is yet not clear, but could involve their actions within the meninges,50,51 as demonstrated for IL-17 affecting brain. 52
IL-4 is directly involved in macrophage M2 polarization within nerve, 53 where these polarized M2 macrophages have a clear role in directing SC regenerative responses. Increased SC proliferation and migration were seen in response to IL-4 induced M2 macrophage-derived microvesicles. 54 Similar results were found with IL-4-stimulated M2 macrophages in an in vitro SC culture. 55 During the pro-regenerative phase of nerve repair, IL-4 signaling has been shown to increase SC mobility, conducing them to form the cellular cords previously mentioned that guide regenerating axons. 56 And finally, during nerve regeneration across a nerve gap, IL-4 signaling was associated with promoting SC myelination within the regenerated nerve gap.22,23 However, while these responses involved IL-4 signaling, it is not yet clear whether direct IL-4 signaling via SCs was involved, as SCs also express the IL-4Rα chain. 57 From these studies, there is potential that IL-4 signaling within SCs was partly involved in observed responses rather than all effects being mediated indirectly to SCs via IL-4 signaling to cells of the immune system (ie, macrophages).
Mesenchymal cells within nerve, such as stromal cells and fibroblasts, play a central role in tissue injury and regeneration. These cells are also receptive to cytokines, stimulating them to take on a variety of phenotypes depending on the tissue and circumstance. In the wound-healing response, type II cytokines play an important role influencing fibroblasts to deposit ECM to enable tissue repair. Early in this process, IL-4 has been shown to specifically induce expression of tenascin by fibroblasts. Tenascin is important in wound healing as it precedes collagen deposition and cellular migration. 58 Furthermore, IL-4 signaling induces increased migration and ECM protein deposition by fibroblasts. 59 However, when type II signaling and fibroblast activity is extended beyond the normal wound-healing timeline, as in allergic diseases, tissue scarring and fibrosis can occur. Whether these processes are recapitulated within nerve is not yet known, but overall, IL-4 signaling within cell populations specific to nerve could clearly be instrumental to injury and regenerative responses.
The Potential of Exogenous IL-4 as a Therapy
Type 2 immunity and therapies targeting IL-4/IL-13 might play a significant role in overcoming severe nerve injuries that do not regenerate, or regenerate to a degree but yield inadequate recovery. In Table 1, the findings from studies of exogenous IL-4 and its effects on rodent models of neuropathies are presented. While many of these studies are not specific to traumatic PNI, the results suggest some findings could be extrapolated and hold promise in the context of PNI. The studies were broadly divided into 2 categories—systemically versus locally administered IL-4 treatments—and into a third category for resveratrol, an IL-4R-modulating drug.
Effects of IL-4 in nerve injury models.
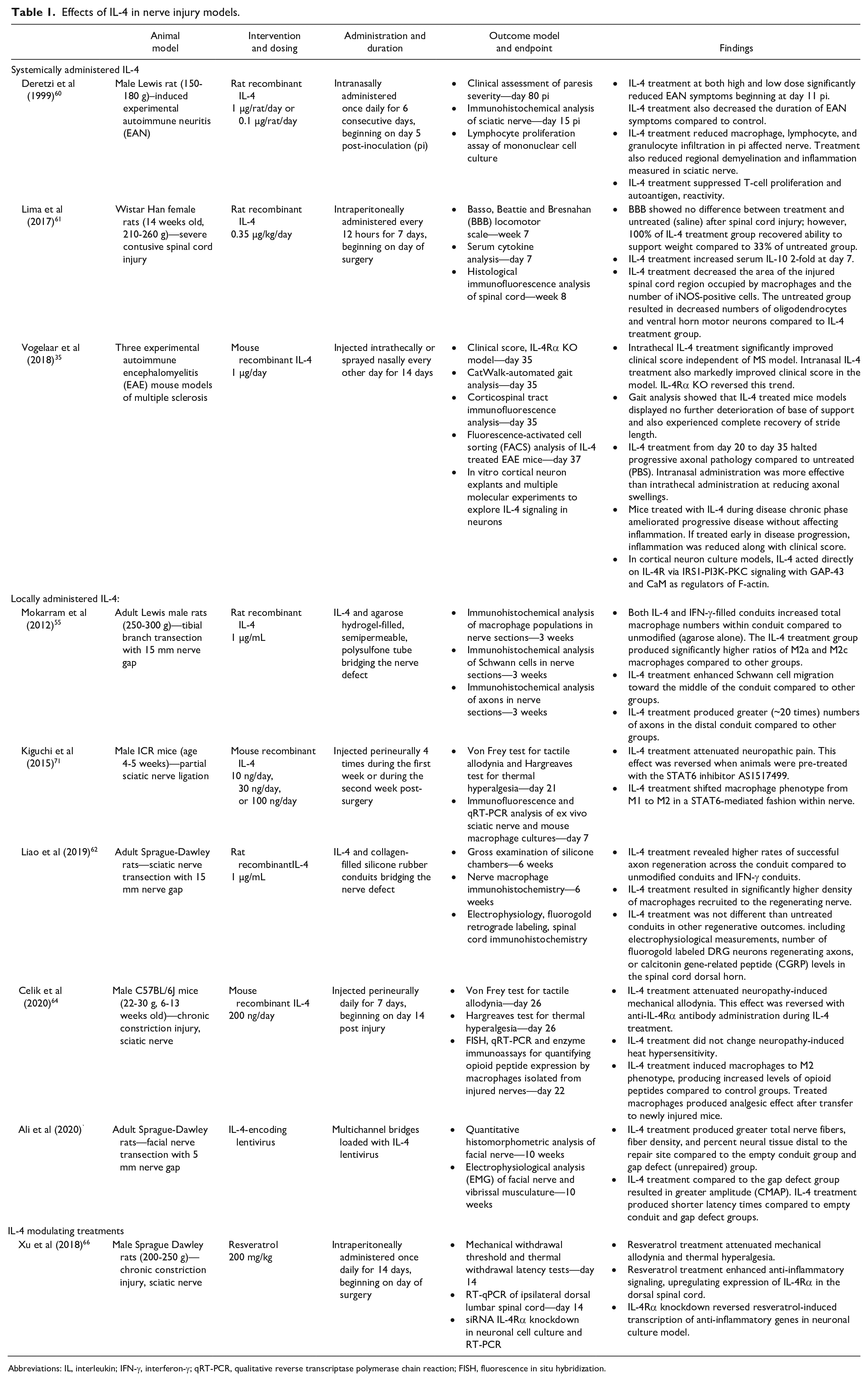
Abbreviations: IL, interleukin; IFN-γ, interferon-γ; qRT-PCR, qualitative reverse transcriptase polymerase chain reaction; FISH, fluorescence in situ hybridization.
Systemic treatment using exogenous IL-4 was associated with improved regenerative outcomes in rodent neuropathy models. Studies using autoimmune models of neuropathy reported significantly reduced severity of symptoms with exogenous IL-4, and that treatment attenuated neuronal inflammation and progressive demyelination.35,60 Vogelaar et al 35 found that IL-4 therapy ameliorated disease progression equally whether treatment begun in early or late stages of disease. Interestingly, when IL-4 therapy was administered at an early phase, symptoms and inflammation from the disease model were both reduced. However, at a late phase, symptoms were reduced while no change in inflammation was observed, suggesting a potential nonimmune mechanism for IL-4 during chronic demyelinating diseases like MS. 35 Finally, systemic IL-4 used in a spinal cord contusive injury model demonstrated decreased inflammation throughout the spinal cord and increased numbers of oligodendrocytes and motor neurons in the ventral horn compared to untreated models. 61 While additional research is needed to confirm the relevance of these findings to PNI, these findings suggest that an IL-4 therapy may need to target neuronal populations to promote regeneration.
Studies considering the effects of IL-4 on PNI models focused on the effects of locally administered IL-4 at the site of peripheral neuropathy. Overall, these studies found an increase in pro-regenerative processes associated with IL-4 treatment. Nerve conduit repair models showed greater axon regeneration and axon density across a nerve gap when using IL-4-filled conduits compared to no drug within a conduit.62,63 Additionally, IL-4 treatment increased SC migration into the conduit and was associated with increased numbers of macrophages in the nerve. Upon further analysis, this greater macrophage population was composed primarily of M2 macrophages and had a decreased M1 macrophage ratio compared to controls. 55 Alternatively, perineural injected IL-4 treatment attenuated neuropathic pain in a dose-dependent fashion in models of neuropathic pain. 64 IL-4 treatment was also associated with increased M2 macrophage populations in the affected nerve. Interestingly, Celik et al 64 reported IL-4-induced M2 macrophages produced increased levels of opioid peptides compared to untreated groups, unveiling a potential mechanism whereby IL-4 treatment reduces neuropathy-associated pain. Compared to the previously discussed systemic IL-4 studies, the findings here carry greater relevance for applications to treat PNI. However, given the heterogeneity of local environments that PNS neuron components project their axons to, and the heterogeneity of the neurons themselves, more research is needed to elucidate the specific effects IL-4 has on the various cells of the PNS environments (nerve gap, proximal and distal stumps, DRG, dorsal horn, ventral horn, etc) and how these effects might work together to promote nerve repair and regrowth.
Finally, an interesting study was done using resveratrol, as opposed to IL-4. Resveratrol is a naturally derived polyphenol that is known for its wide-spread beneficial effects (ie, cardioprotective, antioxidant, anti-inflammatory, anti-tumorigenic, etc) with myriad proposed mechanisms of action. 65 Additionally, resveratrol has been shown to improve outcomes of various kinds of nerve injury. Xu et al 66 found that resveratrol attenuates neuropathic pain through IL-4R-mediated signaling. With resveratrol treatment, they observed an upregulation of anti-inflammatory receptors and signaling in the spinal dorsal horn, leading to reduced mechanical allodynia and thermal hyperalgesia. This resveratrol-induced anti-inflammatory signaling was reversed with an IL-4Rα knock out, suggesting an IL-4R-dependent mechanism for resveratrol-mediated alleviation of neuropathic pain. 66
Translating an IL-4 Therapy
Given the role endogenous IL-4 plays in guiding the regenerative phase of the wound response and the encouraging results from its exogenous use in treating PNI in rodents, there is great clinical translational potential for an IL-4-focused therapy. While exogenous IL-4 attenuates neuropathy and promotes significant nerve regeneration with recovery, the exact cellular mechanisms whereby IL-4 treatment promotes nerve regrowth still needs considerable attention. And, while there seems high potential to translate IL-4 therapies to the clinic given these early results, exogenous IL-4 treatment is not devoid of risks and potential side effects. Although IL-4 is critical for promoting tissue regeneration, chronic IL-4 exposure in tissues promotes excessive tissue remodeling, fibrosis, and chronic inflammation. Chronic inflammatory diseases frequently result in tissue fibrosis and are attributed to a significant proportion of deaths worldwide.67,68 Excessive IL-4 signaling has been implicated in various chronic inflammatory conditions like allergic asthma. As such, many therapies have been developed to block IL-4 signaling and diminish its long-term effects on tissue inflammation and scarring.29,69 Thus, great care needs to be taken in developing an exogenously applied IL-4 treatment to maintain the desired regenerative effects while attenuating unwanted scarring and potential fibrosis.
Based upon these concerns, some risk could be minimized by IL-4 local delivery at the nerve injury site through injections or nerve conduits, rather than systemic administration. If unwanted side effects pose a serious roadblock for treatment design, having an understanding of IL-4 signaling pathways within specific cells would be critical for potentially developing IL-4 therapies specific to the cells and sites required for stimulating nerve regeneration. Given the structure and distribution of the IL-4R subtypes, it might be possible to accomplish this. Non-hematopoietic cells principally express the type 2 IL-4R, whereas hematopoietic cells express both type 1 and type 2. Attempts to mutate IL-4 to preferably activate the type 1 receptor, preventing fibrotic side effects, has been met with setbacks, but recent results are promising and show potential for developing a hematogenous cell-selective “superkine.29,70 Alternatively, it also might be worthwhile to search for additional therapies that modify or promote IL-4 signaling without administering exogenous IL-4 itself. This method could pose fewer side effects compared to native IL-4 therapy and potentially be easier to develop and administer. The recent results from Xu et al using resveratrol to stimulate IL-4-mediated signaling are encouraging for this form of PNI treatment. However, care needs to be taken as some studies have found negative effects for resveratrol as a pro-oxidizing agent. 65 Overall, more research will need to be done to ensure treatment safety in the context of PNI, but therapies targeting IL-4 signaling have demonstrated promise to potentially better manage PNIs.
Conclusion
Interleukin-4 is a promising target for managing PNI and enhancing recovery. It is the principal cytokine governing the pro-regenerative type 2 immune response, suppressing inflammation and stimulating immune cells to a healing phenotype. The cells implicated in nerve regeneration—Schwann cells, macrophages, fibroblasts, neurons, etc—respond to IL-4 signaling, either indirectly via other immune cells or directly, and subsequently promote regrowth. Without IL-4, nerve recovery post-injury is greatly hindered. IL-4 treatments for nerve injury and neuropathy in rodent models have shown encouraging preliminary results and indicate targeting IL-4 signaling as a promising translational therapy for recovery from traumatic nerve injury. More study is needed to characterize the precise signaling network—IL-4 sources, targets, and effects—between the various cells of post-trauma nerve that is necessary for regeneration. With that knowledge, more specific and robust therapies could potentially be developed to optimize nerve regeneration without the unwanted effects of systemically stimulated type II immunity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institutes of Neurological Disorders and Stroke of the National Institutes of Health under award number R01 NS115960 (MDW) to Washington University.
