Abstract
Peripheral nerve injury is a major clinical problem and often results in a poor functional recovery. Despite obvious clinical need, treatment strategies have been largely suboptimal. In the nervous system, exosomes, which are nanosized extracellular vesicles, play a critical role in mediating intercellular communication. More specifically, microRNA carried by exosomes are involved in various key processes such as nerve and vascular regeneration, and exosomes originating from Schwann cells, macrophages, and mesenchymal stem cells can promote peripheral nerve regeneration. In this review, the current knowledge of exosomes’ and their miRNA cargo’s role in peripheral nerve regeneration are summarized. The possible future roles of exosomes in therapy and the potential for microRNA-containing exosomes to treat peripheral nerve injuries are also discussed.
Introduction
Peripheral nerve injury (PNI) is a major public health burden, however, current treatments have remained suboptimal, often resulting in poor functional recovery.1-3 The annual incidence of PNI is estimated between 13 and 23 per 100 000 persons in developed countries.2,4 In the United States alone, more than 20 million patients suffer from PNI caused by trauma and medical disorders, and approximately $150 billion health care dollars are spent on nerve injuries each year.5,6 Clinically, PNI is highly heterogeneous, and often involve a combination of partial or complete motor and sensory dysfunction. Pathophysiologically, a number of critical factors are involved in regeneration after nerve injury, including the activity of macrophages and Schwann cells, inflammatory reaction, and vascular regeneration.7-9 In general, regenerating axons are only able to grow approximately 1 mm per day,10-13 making regeneration across large nerve gaps particularly challenging. Autologous nerve grafting remains the gold standard treatment for treating nerve gap injuries; however, it has many shortcomings, such as the need for additional surgical procedures to retrieve a healthy functioning nerve.14,15 While tissue engineering techniques to produce synthetic conduits have been shown to be beneficial for PNI, outcomes are still far from optimized.16-20
Recently, exosomes, nanosized extracellular vesicles released by nearly all cell types, have been shown to improve the nerve regeneration process.21-23 To date, a sizeable body of literature have demonstrated that exosomes play a critical role in intracellular communication, transferring information via their cargo, which includes DNAs, messenger RNAs, microRNAs (miRNAs), and proteins.21,24,25 Several studies have shown that exosomes are enriched with miRNAs that target transcription factors and genes of several cellular pathways, including angiogenesis, cellular transport, apoptosis, and proteolysis.26,27 In the nervous system, exosomes have also been shown to play an important role in regulating regeneration, with accumulating evidence indicating that exosome miRNAs are critical for vascular regeneration, 28 and that miRNAs carried by exosomes from Schwann cells, macrophages, and mesenchymal stem cells (MSCs) promote peripheral nerve regeneration. 29
This review summarizes the current knowledge of exosomes, their miRNA cargo, and their roles in peripheral nerve regeneration. We will also highlight the future potentials of using exosomes and miRNAs for optimizing treatments of PNIs.
Life Cycle and Characteristics of Exosomes
In 1970, Johnstone et al first described “exosomes” as extracellular vesicles carrying unwanted materials.30-32 Since then, a number of extracellular vesicles (EVs) have been reported and can now be generally classified as microvesicles (MVs), apoptotic bodies, or exosomes depending on their size and biogenesis. According to the current classification scheme, exosomes are small extracellular nanosized (30-100 nm) vesicles with a lipid bilayer membrane released by all cells types. 33 The biogenesis of exosomes involves the endocytic pathway. Briefly, small vesicles are formed by inner budding of the plasma membrane and then fused together to form the early endosome. Intraluminal vesicles (ILVs) begin to form by invagination of the limiting endosomal membrane in the maturation process of early endosomes, which is a critical step of exosome biogenesis. Endosomal sorting complex required for transport (ESCRT), a complex multimolecular machinery that regulates the formation of ILVs, plays an important role in exosome secretion. In the process of ILVs formation, cargo molecules such as RNAs are encapsulated into the ILVs, and then ILVs are further encapsulated to form multivesicular bodies (MVBs). MVBs have 1 of 2 fates. Some of MVBs are transferred to lysosomes for degradation (dMVBs); and other MVBs fuse with the plasma membrane and release ILVs into the extracellular space as exosomes (sMVBs). Once exosomes are released, they are internalized by target neuronal tissue via the soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE) complex. 23
Molecular Contents and Function of Exosomes
Exosomes contain 2 classes of molecules: constitutive molecules and cargo molecules. The makeup of constitutive molecules depends on the cell type from which exosomes were derived, and is unique for exosomes, determining their structure and function. Cargo molecules include proteins, lipids, and various genetic materials, and are resorted, encapsulated, and transported by exosomes. Cargo molecules are highly heterogenous, and the composition of exosome cargoes depends on cell of origin and physiological or pathological conditions during the formation of exosomes. Overall, protein cargoes include tetraspanins (CD9, CD63, CD81, and CD82), Rab GTPases, flotillin, Alix, TSG101, and heat shock proteins (Hsc70, Hsp90); lipid cargoes mainly work to regulate exosomal sorting of small RNAs and proteins; genetic cargoes include DNA, mRNA, miRNA, ribosomal RNA (rRNA), circular RNA, and long noncoding RNA (lnRNA). 34
Exosomes differ depending on their cell of origin not only in size but also in their RNA, protein, and lipid composition. miRNAs are short nucleotides sequences and can regulate gene expression in various cell types, and recent studies showed miRNAs in exosomes are critical factors in intracellular communication between source and target cells. 35 In the nervous system, miRNA-containing exosomes can be secreted by a variety of cells, including Schwann cells, microglia, oligodendrocytes, astrocytes, and neurons. These exosomes can mediate different types of communication between cells by surface-to-surface interaction and shuttling of miRNA between cells, influencing various cell functions such as maturation. 36 Eldh et al 37 have revealed that miRNA content is not only different between exosomes and cells of original but also different depending on the growth conditions of origin cells. Baglio et al 38 published an interesting study in which they found that MSCs and their exosomes have a different miRNA composition. This study revealed that adipose mesenchymal stem cells (ADSC)– and bone marrow mesenchymal stem cells (BMSC)–derived exosomes have highly similar miRNA expression profiles enriched (such as miR-4485, miR-150-80, miR-486-5p, and miR-6087), but different compared to their source cell, demonstrating that exosomes do not merely reflect the cellular contents, and that tissue-specific microenvironment can influence miRNA content of ADSC- and BMSC-derived exosomes.
Proteins in exosomes also play an important role in exosome biogenesis as well as cell-type specific actions. 39 Both actin and β-tubulin are contained in the exosomes and are known to be membrane and cytoskeletal proteins which are required for axonal growth. 40 Previous studies also demonstrated that heat shock protein 70 is key in metabolic support and protection of neurons.41,42 Galectin-3, which is associated with myelin phagocytosis, also has been identified in EVs and shown to be upregulated by Schwann cells following nerve injury.39,43 Kramer-Albers et al 44 have found myelin proteins such as myelin-associated glycoprotein (MAG) and proteolipid protein (PLP) encapsulated in exosomes, and these proteins are also critical for nerve remyelination. More recently, Zhao et al 45 showed that circular ribonucleic acids (circRNAs) are enriched and stable in exosomes and involved primarily with glutamatergic synapse and the cyclic guanosine monophosphate-protein kinase G signaling pathway to improve the recovery of central nerve system injures. In addition to proteins and RNAs, exosomes are also enriched with certain raft-associated lipids such as cholesterol, ceramide, phospoglycerides, and long and saturated fatty-acyl chains.27,46 Studies have also shown that exosomes contain high dose of IL-6, IL-8, and other cytokines. 28 Altogether, these evidences demonstrate that multiple factors are enriched in exosomes, and play an important role for regeneration and remodeling of the nervous system.
Techniques for Isolating Exosomes
Although exosomes have great potential to improve the diagnosis and treatment of clinical disease, its clinical applicability is still limited because of the lack of standardized isolation and purification technologies. 47 To date, techniques for isolation of EVs are based on the molecular weight and size of the particles or chemical precipitation. Several strategies have been reported for exosome isolation including differential ultracentrifugation, density gradient separation, ultrafiltration, size exclusion chromatography, immunoisolation, and flow cytometry.48-50 Currently, differential ultracentrifugation is the most commonly used technique for exosomes purification and has been recommended as a gold standard for isolation of populations of exosomes with relatively homogenous size.48-50 However, this approach is time-consuming, lacks automatization, and is limited in terms of purity and possible compromises to EV integrity and functionality. Although immunoisolation (IA) can offer high efficiency and specificity for exosome capture with minimal impact on EV integrity and functionality,47,50 this method is clearly suboptimal for processing of routine clinical samples and have intrinsic limits that impair their use in biomarker discovery and development of novel diagnostics. Ultrafiltration is another option that not only provides a straightforward procedure but also minimizes the impact on EV integrity and functionality. This procedure maybe more suitable for producing clinical grade exosomes in terms of both time efficiency and quality.
miRNA-Containing Exosomes Play an Important Role in Cell-Cell Communication in the Nervous System
Cell-to-cell communication is essential for physiological homeostasis, particularly in the nervous system. Information exchange performed by exosomes is a newly described form of intercellular communication and plays an important role in the development and physiology of neural systems. 51 Exosomes packed with information such as signaling proteins, coding RNA, and miRNAs are released from source cells and taken up by target cells, thereby facilitating multilevel transfers of information (Figure 1).
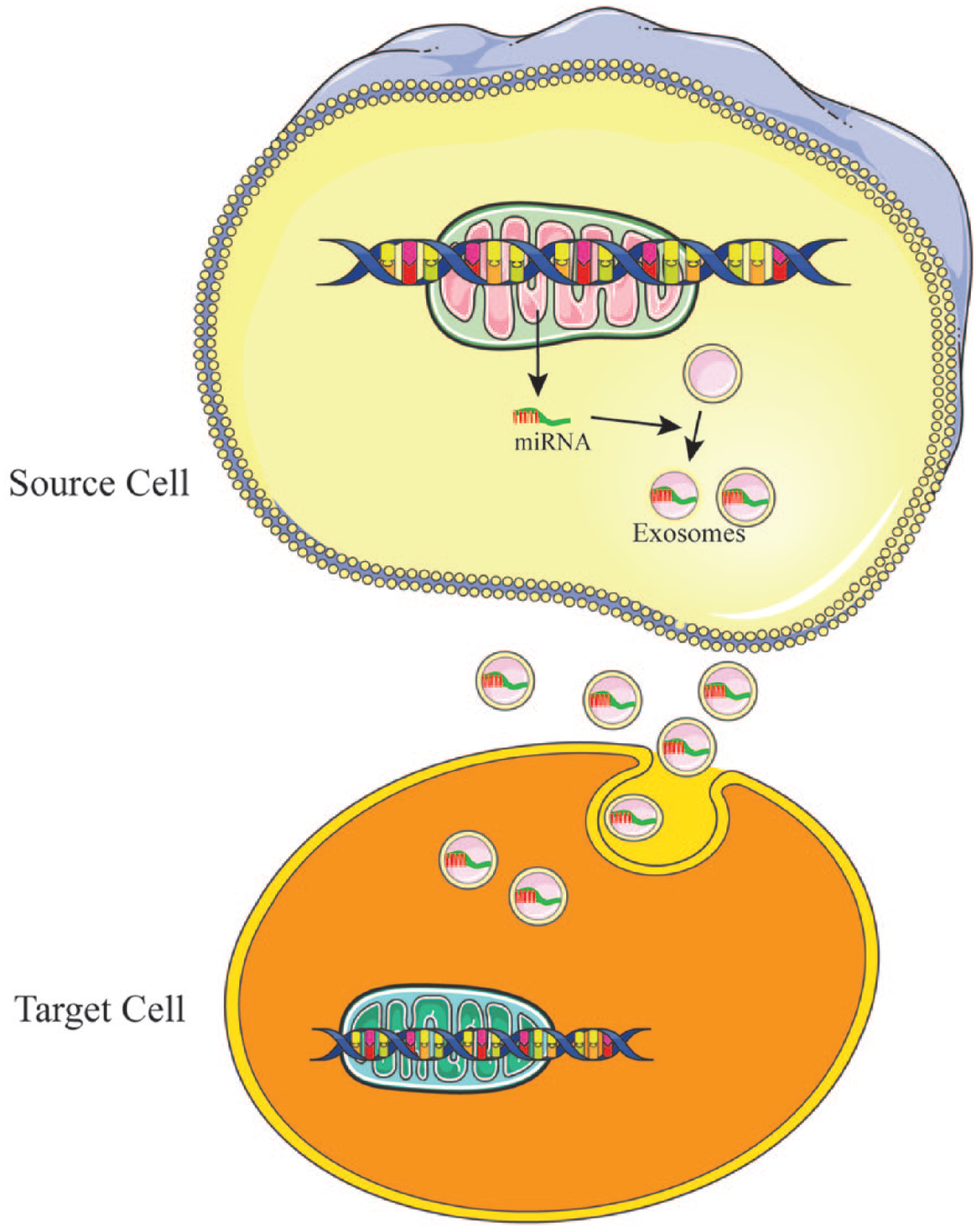
Schematic representation of how microRNA (miRNA)-containing exosomes play an important role in cell to cell communication. Exosomes packed with information such as miRNAs are released from source cells and taken up by target cells, thereby facilitating multilevel transfers of information and playing an important role in the development and physiology of neural systems.
The function of exosomes depends on proteins, lipids and genetic contents. In 2007, Valadi et al 52 demonstrated that exosomes contain both mRNA and miRNA, and can deliver these genetic molecules to other cells. These RNAs are termed exosomal shuttle RNA (esRNA). Recently, it was demonstrated that exosomes with enriched microRNA content is critical in cell-to-cell communication in the nervous system, 53 and that miR-132 carried by exosomes act as intercellular signals mediating neural regulation of brain vascular integrity, suggesting a novel role of exosomes in neurovascular communication.54,55 Recent studies have also implicated that miRNAs contained in MSC-derived exosomes play a role in intercellular signaling. miRNA loading within exosomes is not random and depends on their source cell, which responds to the microenvironment. Overall, miRNA packaged in exosomes from MSCs serve as paracrine control molecules from MSCs can affect the response of target cells. 56
Exosomes and their miRNA cargo serve as an important intercellular communication approach and have also been described in the central nervous system (CNS). In the CNS, miRNAs could be shuttled by exosomes from microglia57,58 and astrocytes59-61 to axons. Similarly, in the peripheral nerve system, miRNAs could be shuttled by exosomes to axons. Simeoli et al 62 found that exosomes containing miRNA-21 also contribute to sensory neuron-macrophage communication after damage to peripheral nerves. Sensory neuron-derived exosomes are readily phagocytized by macrophages in which an increase in miR-21-5p expression promotes a pro-inflammatory phenotype. Overall, a number of studies have demonstrated that exosomes containing miRNA cargo play an important role in cell-to-cell communication in the nervous system. These findings present an interesting direction to further explore the novel mechanisms and therapies for peripheral nerve regeneration (Figure 2).
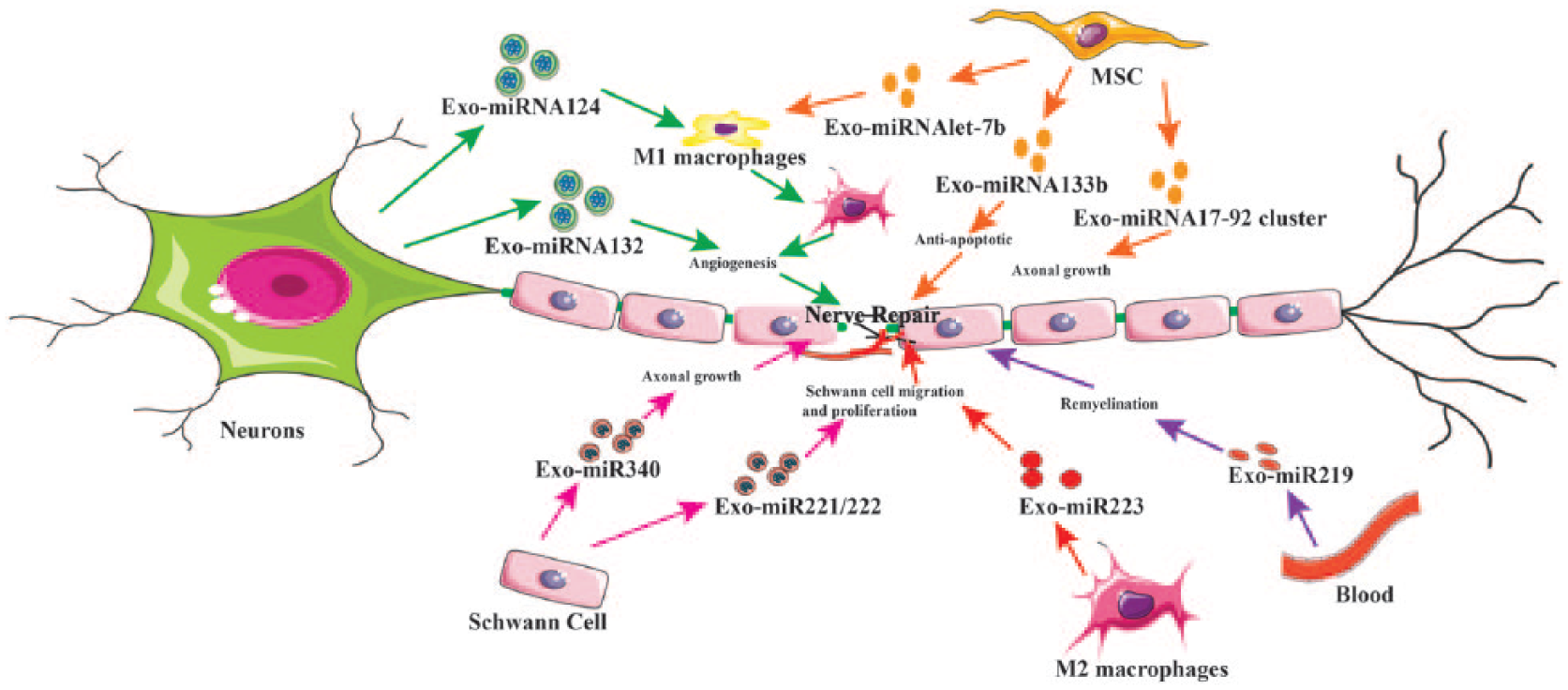
Schematic representation of possible routes of exosomal microRNA (miRNA) to improve peripheral nerve regeneration. During peripheral nerve injury, a complex sequence of events is triggered inside the neuron’s cell body and in neighboring cells. Schwann cells, inflammation, trophic factors, and angiogenesis are all involved in the nerve regeneration process. Encapsulated miRNAs in the exosomes play an important role in the peripheral nerve regeneration. Schwann cell exosomes that encapsulate miRNA340 and miR221 could be used to enhance nerve regeneration. Evidence has shown that miRNA containing exosomes from differentiated MSC can also directly enhance axonal regeneration, or indirectly promote recovery by regulating the inflammation response to promote nerve repair. Macrophages also secrete miRNA containing exosomes that can improve Schwann cell migration and proliferation, axon regeneration and angiogenesis to enhance peripheral nerve regeneration. MSCs, mesenchymal stem cells; Exo, exosomes.
miRNA-containing exosomes promote the vascular regeneration and contribute to nerve repair
Vascular integrity is critical in maintaining nerve system microenvironment homeostasis, which is critical for repair, development, and function of the nervous system. Studies have demonstrated that the neural cells can regulate brain vascular integrity 54 and that MSC-generated exosomes can improve functional recovery following nerve injury by promoting endogenous angiogenesis and neurogenesis in rats. 63 Xu et al 55 showed that neurons transfer miR-132 to endothelial cells via exosomes to maintain brain vascular integrity. This study also showed that miR-132 regulates the expression of vascular endothelial cadherin (VE-cadherin), an important adherent junction protein, by directly targeting eukaryotic elongation factor 2 kinase. MSC-derived exosomes can also function as paracrine promoters of angiogenesis and may also have some clinical therapeutic value, 64 providing a promising strategy to improve survival and angiogenic potency of nerve system.65,66 This effect may be attributed to activation of the PI3K/Akt signaling pathway on endothelial cells. 67 Anderson et al 68 also showed that MSCs-derived exosomes’ promotion of angiogenesis may also be mediated by the nuclear factor–κB (NF-κb) signaling pathway.
Together, these studies illustrate that neurons and MSCs can promote angiogenesis through exosome mediated cell-to-cell communication in the nervous system, which makes exosomes a valuable tool for peripheral nerve regeneration. However, further studies are still needed to determine whether exosomes and their miRNA cargo can improve angiogenesis in the peripheral nerve repairing process.
Schwann Cell–Derived miRNA-Containing Exosomes to Promote Peripheral Nerve Regeneration
Schwann cells are critical for maintaining homeostasis in normal nerves and facilitate the regeneration process of peripheral nerves after injury. These cells provide the nutrition to support axon development, maturation and regeneration, and they are the main cell type responsible for myelinating axons in the peripheral nervous system (PNS). In the central nervous system (CNS), miRNA containing exosomes have been widely reported to originate from supporting cells such as microglia,57,58 astrocytes, 59 and other neurons.60,61 Similarly, in the PNS, miRNAs can be shuttled by exosomes from Schwann cell to neurons to improve the peripheral nerve repair. Schwann cell miRNA expression levels are drastically changed following injury, suggesting that local genetics may play an important role in the nerve regeneration process.69,70 Studies have shown that miRNA can augment Schwann cell proliferation and axon myelination both during development and following injury.71-74 Li et al 75 found that Schwann cell miRNA340 can boost debris clearance following nerve crush injury in a rat sciatic model, and that the dysregulation of the miR-340 expression in the injury site perturbed cell debris removal and axonal regrowth. Yu et al 76 also reported significant expression changes of 77 miRNAs at 4 time points in the proximal nerve stump following rat sciatic nerve injury using microarray analysis. These miRNAs mainly involve cell proliferation, cell locomotion, and cellular homeostasis. 76 Furthermore, upregulation of miR-221 and miR-222 cluster (miR-221/222) was also found to correlate with injury-induced Schwann cell phenotypic modulation. 76 Substantial amounts of miRNAs secreted from axon or nerve terminal have been reported to direct migration of Schwann cells. 77 Many injury-regulated Schwann cell miRNAs are likely drivers of Schwann cell dedifferentiation/proliferation, thereby reinforcing transcriptional programs that promote remyelination by altering cytoskeletal organization and vesicle transport, which has the potential to coordinate axonal growth. 78 In Schwann cells, the expression of many of these proliferative miRNAs is positively regulated by Sox10, a known player in Schwann cells’ development, differentiation, and maturation remyelination. 79 Furthermore, studies into specific miRNAs have shown that their overexpression can enhance neurite outgrowth. 80 Together, these results demonstrate that SC miRNAs are important modulators of the SC regenerative response after nerve damage.
Interestingly, recent studies showed that exosomes from glial cells communicate with axons and may play an important role in axonal growth and axonal regeneration. 41 Studies have reported that an abundance of miRNAs in the axon or nerve terminal are likely due to direct transfer from Schwann cells. 77 Schwann cell exosomes have been shown to be internalized by peripheral nerve axons and enhance neurite outgrowth in vitro, and direct injection of exosomes can promote axon growth following in vivo nerve crush injury. 81 Schwann cell–derived exosomes markedly increase axonal regeneration in vitro and enhance regeneration after sciatic nerve injury in vivo,41,82,83 while fibroblast exosomes have no effect. 41 Exosomes shift the growth cone morphology to a pro-regeneration phenotype and decrease the activity of the GTPase RhoA, which is involved in growth cone collapse and axon retraction. 82 Altogether, this study demonstrated a novel mechanism by which Schwann cells communicate with neighboring axons during nerve regeneration, and that Schwann cell exosomes partake in an important mechanism by which these cells locally support axonal maintenance and regeneration after nerve damage. 82 Overall, Schwann cell–derived exosomes have been shown to be valuable in peripheral nerve regeneration, providing a promising avenue for the development of novel therapeutic strategies. However, to obtain exosomes from Schwann cells, harvesting Schwann cells via sacrifice of normal nerve tissue is currently necessary. This disadvantage will remain a major challenge for clinical translation until novel technologies allow for in vitro assembly of exosomes that are functionally equivalent to Schwann cell–derived exosomes without the need to harvest Schwann cells. Further investigation is required to identify optimal exosomes content and efficient methods to harvest Schwann cell–derived exosomes.
Macrophages-Derived miRNA-Containing Exosomes’ Effect on Peripheral Nerve Regeneration
After peripheral nerve injury, degenerative debris and inflammatory alterations at the injury site may block the elongation of regenerating axons to reach target organs. Macrophages play an important role in modulating the inflammation reaction. Almost immediately after nerve damage, damaged myelin will activate the macrophages to release pro-inflammation cytokines and chemokines. These cytokines mediate macrophage phagocytosis of axons and myelin debris, and also facilitate the migration and accumulation of additional macrophages at the site of nerve injury.
Recent studies have shown that exosomes harvested from various circulating immune cells can increase nerve remyelination and attenuate oxidative stress in the CNS. 80 More specifically, macrophage-derived exosomes contain miRNA that can promote the nerve regeneration, 84 and macrophage-derived microvesicles (MVs) are involved in this regeneration process both in vitro and in vivo. 84 MVs can improve Schwann cell migration and proliferation, and upregulate the expression levels of nerve growth factor (NGF) and Laminin in vitro. 84 Furthermore, the relative expression levels of miR-223 were also increased in M2 macrophages and M2-derived MVs in a rat sciatic nerve injury model. Schwann cells co-incubated with miR-223 inhibitor depressed cell migration and proliferation and downregulated the expression levels of NFG and Laminin protein. 84 MVs containing miR-223 are believed to be critical players in nerve regeneration, and M2-derived MVs provided a new therapeutic strategy. 84 These findings demonstrate that exosomes also play a critical role in the inflammatory response following peripheral nerve injury, and provide an interesting direction for future research in exosome’s role in peripheral nerve regeneration.
MSCs-Derived Exosomes and Their miRNA Cargo’s Role in Promoting Peripheral Nerve Regeneration
MSCs are multipotent stem cells isolated from mesenchymal tissues (eg, bone marrow, adipose, dental pulp, umbilical cord blood, etc). 85 Studies have demonstrated that multipotent MSCs can significantly improve functional recovery following nerve injury. 46 Lopez-Verrilli et al 83 also have explored that MSCs of different sources, including menstrual MSCs (MenSCs), BMSCs, and umbilical cord MSCs, have been shown to promote neuronal growth and survival. Recently exosomes have been shown to be major paracrine effectors of MSCs, 86 and can be released by MSCs to mediate cell-to-cell communication and maintain dynamic and homeostatic microenvironments for tissue repair. 87 MSCs secreted exosomes can activate PI3k/Akt, ERK and STAT3 signaling pathways to induce the expressions of growth factors such as insulin-like growth factor–1 (IGF-1), NGF, and stromal-derived growth factor-1 (SDF-1). 88 Exosomes secreted from human ADSCs also have protective potential against neuron damage through activating the PI3K/Akt signaling pathway. 89 Furthermore, several studies have also demonstrated that exosomal miRNAs (such as miR-199b, miR-218, miR-148a, miR-135b, and miR-221) isolated from MSC culture can influence neuron differentiation, proliferation, and axonal outgrowth. 90
Many studies have found that MSCs derived exosomes from various sources can promote nerve regeneration, 83 and this phenomenon is likely associated with exosomal miRNA. RNA-seq identified at least 386 annotated miRNAs in MSC derived exosomes, but only miR148a, miR532-5p, miR378, and let-7f were enriched in EVs compared with MSCs. 26 Overall, accumulating evidence suggests that MSC-derived exosomes can deliver their cargo miRNAs into recipient neurons to promote axonal growth.61,91,92 Xin et al 93 also found that the miR-17-92 cluster can promote neurogenesis and axonal outgrowth in vitro, and that tailored exosomes enriched with the miR-17-92 cluster increases neural plasticity and functional recovery by activating the PI3K/protein kinase B/mechanistic target of rapamycin/glycogen synthase kinase 3β signaling pathway. 94
MSC-derived exosomes can also promote axonal growth by regulating the Argonaut 2 protein, a key miRNA effector molecule. 23 Mead et al 95 demonstrated that BMSC-derived exosomes also have significant neuroprotective and neuritogenic effects via their microRNA cargo and that knockdown of argonaute-2, a key miRNA effector molecule, diminishes therapeutic effects of exosomes derived from BMSC, demonstrating that exosomes’ successful delivery of their miRNA cargo is critical for its nerve regenerative function. Zhang et al showed that distal axons of embryonic cortical neurons contain miRNA machinery proteins, Dicer and argonaut 2 (Ago2) proteins, and are enriched with miRNAs that can locally regulate axonal growth.23,94,96
The function of MSC-derived exosomes depends on the condition of the origin cell, which may impact the miRNA content of exosomes and subsequently influence their biological function. 30 Some studies have shown that MSCs with overexpression of miRNAs are more able to improve function recovery than naïve MSCs.93,97 A recent study also found that overexpression of microRNA133b (miR-133b) in MSCs leads improves recovery from middle cerebral artery occlusion (MCAO) in rats, and that this improvement is mediated by exosomal shuttling of miR-133b, which also stimulates further exosome release from astrocytes. 93 Zhang et al. also showed that tailored MSC further enhance axonal growth compared to native MSC, as exosomes of tailored MSCs carry an elevated level of miR-17-92 cluster, which activates the PTEN/mTOR signaling pathway in recipient neurons. 23 Recent research has also shown that exosomes derived from hypoxic preconditioned MSCs can upregulate the miRNA content in exosomes 98 and that these exosomes can promote angiogenesis, cell proliferation, and differentiation.99,100
Altogether, these findings show a potential role for MSC-derived exosomes and their miRNA cargo in regeneration and remodeling of the nervous system. Furthermore, modifying MSCs to overexpress miRNAs can give rise to miRNA-enriched exosomes, and may be a promising approach to accelerate peripheral nerve regeneration.
MSCs-Derived Exosomes and Their miRNA Cargo’s Immunomodulatory Effects During Peripheral Nerve Regeneration
Inflammation plays an important role in the peripheral nervous regeneration, 101 and many studies have shown that the inflammatory reaction and cytokines are key facilitators of this process. While inflammation is necessary to clear debris for nerve regeneration, it can also cause problems such as neuropathic pain and hamper the nerve regeneration. Therefore, a suitable level of inflammation is critical for optimal nerve repair.
It is well known that MSCs and MSC-derived exosomes possess immunomodulatory properties.102,103 Recent studies have shown that MSC derived exosomes can deliver miRNA to regulate inflammation in the cardiac, wound, and bone repair.104,105 Wen et al 86 also showed that human BMSCs–derived exosomes and their small RNA cargo can suppress immune reactions by inhibiting peripheral blood mononuclear cell (PBMC) proliferation and increasing regulatory T cell (Treg) function. 86 Fatima et al 106 reported that exosomes harvested from stem cells can mediate the transmission of anti-inflammatory RNAs to injury sites and potentially orchestrate the resolution of the inflammatory responses to better facilitate healing processes. MSC-derived exosomes can also influence brain remodeling by regulating the immune reaction. 107
Lipopolysaccharide (LPS)-preconditioned MSCs (LPS pre-MSCs) can also enhance their paracrine effects through increasing exosome secretion, with LPS pre-MSCs showing an enhanced ability than untreated MSC-derived exosomes (un-Exo) to promote M2 macrophage activation due to their upregulation of anti-inflammatory cytokines via exosomal shuttling of let-7b. 108 This study further investigated the mechanisms for the immunotherapeutic potential of these exosomes and miRNA let-7b expression, and demonstrated that the TLR4/NF-κB/STAT3/AKT regulatory signaling pathway plays an important role in the regulation of macrophage plasticity. 108 Although a number of studies showed that MSCs can boost peripheral nerve regeneration,46,109 some disadvantages, such as immunogenicity, retention, and neoplasticity have also been reported. Therapy strategies using exosomes can overcome the limitation of the MSC transplantation without compromising biological function. Therefore, exosomes may be exploited to develop exciting new therapies for peripheral nerve injury and repair. Few studies have explored whether endogenous niche resident MSCs play a role in nerve repair or normal maintenance of neural tissue by secreting exosomes or MVs. Whether the essence of the endogenous MSC can be captured by its secreted products and used therapeutically would be an exciting area of investigation for novel therapies of peripheral nerve injuries. This is particularly important as the therapeutic efficacy of endogenous MSCs depends on their ability to rapidly respond to the injury microenvironment, while isolated exosomes form MSC would not. Therefore, understanding the role of endogenous MSC-derived exosomes or MVs in the nerve repairs may lead to the development of therapies that are more targeted and less subject to the complexity of the biological milieu after injury.
The Future
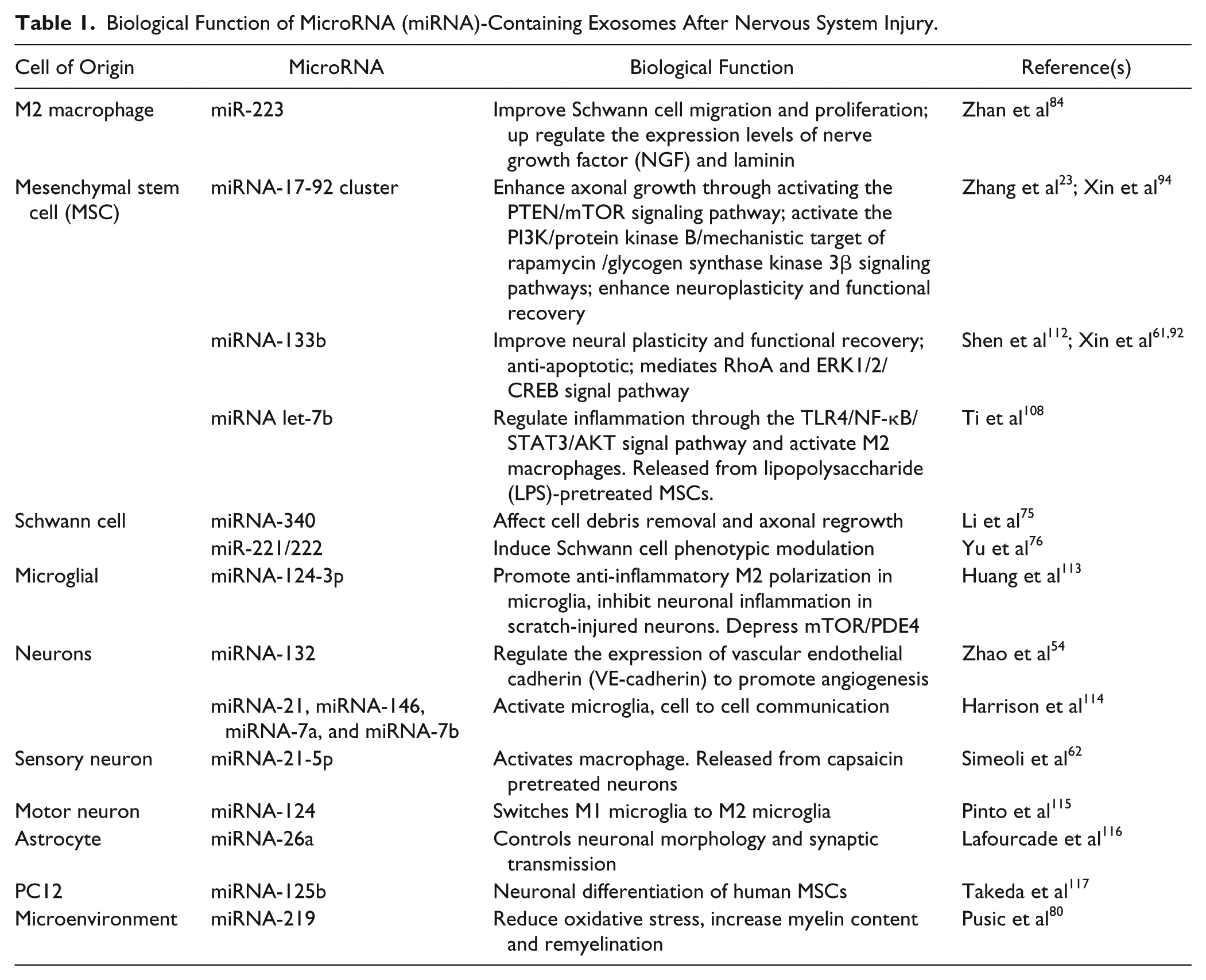
During peripheral nerve injury, a complex sequence of events is triggered inside the neuron’s cell body and in neighboring cells. 81 Schwann cells, inflammation, trophic factors, and angiogenesis are all involved in the nerve regeneration process. Schwann cell– and MSC-based therapies have been attempted but are not ideal due to the need to sacrifice functioning tissue (for Schwann cells) and potential risk of malignant transformation (for MSCs). Studies outlined in this review illustrate that exosomes-mediated communication between cells provide new directions to understand the intercellular regulation of nerve regeneration110,111 (Table 1). These observations identified the possibility of developing novel therapies by using exosomes to either activate or regulate key events in nerve regeneration. Thus, modifying exosomes and their miRNA cargoes is a promising therapies strategy for peripheral nerve injuries. 42 The use of patient-specific Schwann cell exosomes could be used to enhance nerve regeneration; however, it also requires sacrificing a normal nerve to harvest Schwann cells. Evidence has shown that miRNA containing exosomes from differentiated MSCs can also directly enhance axonal regeneration, or indirectly promote recovery by regulating the inflammation response to promote nerve repair. Exosomes harvested from MSCs transfected with overexpression miRNA cluster plasmids could further improve nerve regeneration. Macrophages also secrete miRNA containing exosomes that can improve Schwann cell migration and proliferation and upregulate the expression levels of NGF and laminin to enhance peripheral nerve regeneration.
Biological Function of MicroRNA (miRNA)-Containing Exosomes After Nervous System Injury.
In the future, Schwann cell–, MSC-, or macrophage-derived exosome treatments may be combined with nerve conduit technologies or directly injected into nerve stumps, eliminating the need for live cell transplantation. In addition, existing cells can be modified to overexpress a target miRNA in their exosomes to further enhance their abilities to promote nerve regeneration. These multifactorial approaches provide exciting new prospects for the future of peripheral nerve injury and repair.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Maryland Stem Cell Research Fund (2018-MSCRFD-4271) (to XJ) and R01HL118084 from the National Institutes of Health (to XJ).
