Abstract
Background
Motor coordination, the ability to produce context-dependent organized movements in spatial and temporal domains, is impaired after neurological injuries. Outcome measures assessing coordination mostly quantify endpoint performance variables (ie, temporal qualities of whole arm movement) but not movement quality (ie, trunk and arm joint displacements).
Objective
To develop an outcome measure to assess coordination of multiple body segments at both endpoint trajectory and movement quality levels, based on observational kinematics, in adults with neurological injuries.
Methods
A 3-phase study was used to develop the Comprehensive Coordination Scale (CCS): instrument development, Delphi process, and focus group meeting. The CCS was constructed from common tests used in clinical practice and research. Rating scales for different behavioral elements were developed to guide analysis. For content validation, 8 experts (ie, neurological clinicians/researchers) answered questionnaires about relevance, comprehension, and feasibility of each test and rating scale. A focus group conducted with 6 of 8 experts obtained consensus on rating scale and instruction wording, and identified gaps. Three additional experts reviewed the revised CCS content to obtain a final version.
Results
Experts identified a gap regarding assessment of hand/finger coordination. The CCS final version is composed of 6 complementary tests of coordination: finger-to-nose, arm-trunk, finger, lower extremity, and 2- and 4-limb interlimb coordination. Constructs include spatial and temporal variables totaling 69 points. Higher scores indicate better performance.
Conclusions
The CCS may be an important, understandable and feasible outcome measure to assess spatial and temporal coordination. CCS measurement properties are presented in the companion article.
Introduction
The performance of daily actions depends on reciprocal interactions between perception and action 1 and the emergence of task solutions based on natural laws of physics governing the interaction of the body with the physical world. 2 Solutions should satisfy constraints at several levels of analysis (ie, behavioral, biomechanical, perceptual, environmental) and optimize cost functions (eg, time, smoothness). A major component of an effective task solution is the ability to produce coordinated movement, defined as a movement that is organized in both spatial and temporal domains in a context-dependent way. 3 Coordinated movement can change over time depending on internal and external demands. 4
Neurological injuries often result in deficits at different levels of movement production. Impairment of coordination affecting limbs and axial muscles is a primary consequence of cerebellar, 5 vestibular, and/or sensory deficits. 6 Coordination deficits also occur secondarily to pain, perceptual, cognitive, and sensorimotor impairments, such as weakness, decreased range of motion, and spasticity.
Coordination is a complex phenomenon that can be measured at specific time points as well as dynamically throughout movement. 3 Coordination is also a multidimensional construct measured at different levels of movement description, including the motor performance level and the movement quality level. 7 At the motor performance level, movement of the endpoint (ie, hand, foot) can be characterized by measures of smoothness, linearity, precision, and speed. At the movement quality level, joint rotations and segment displacements can be characterized in terms of amplitudes/ranges and interjoint coordination.3,7 Unlike the motor performance level, descriptions of movement quality characterize the use of unwanted movements that are considered to be compensatory. 7
Current clinical assessments of coordination for adults with neurological injuries, both time and criterion based, generally quantify motor performance but do not fully consider how well movements are performed with objective quantification of movement quality and compensations. 8 For example, the finger-to-nose coordination test reliably assesses the time of task execution but not the qualitative aspects of the movement due to the lack of clear scoring criteria for dysmetria and tremor. 9 Similarly, the Box and Blocks, 10 Purdue Pegboard, 11 9-Hole-Peg, 12 Jebsen-Taylor Hand Function, 13 Action Research Arm Test, 14 and the Lower Extremity Motor Coordination Test (LEMOCOT) 15 are examples of performance-based measures of coordination/dexterity that also do not consider the quality of movement. 16 One of the drawbacks of timed scores is that faster times may be obtained by the use of undesirable, compensatory movements that can lead to problems in functional recovery. 7 Movement quality measures are thus essential to provide information about how the task is performed, the compensatory strategies used, missing task elements, and other specific deficits. 8
Two- and 3-dimensional kinematic analysis can be used to assess movement quality.16-18 However, this technology may be inaccessible for use in clinical practice due to its cost and complexity. Alternatively, observational kinematics can be used to address the gap in objectively assessing coordination in clinical settings. Observational kinematics consists of visually judging the quality of body motions by breaking down a movement into its component parts. 19 Clinicians can accurately and reliably assess repetitive standardized tasks for upper limb (UL) movements 19 and gait 20 using observational kinematics, which is not dependent on evaluator experience 21 as a supplement to standard clinical assessments.
Coordination is a basic aspect of movement involving both temporal and spatial domains. Thus, a measure of coordination that considers both endpoint performance and movement quality is needed to evaluate the multidimensional nature of coordination, to make inferences about underlying impairments, to accurately capture small changes over time and to distinguish between recovery and compensation. The goal of this study was to develop a comprehensive clinical outcome measure of sensorimotor coordination of multiple body segments (trunk and limbs) based on observational kinematics in individuals with neurological injuries. This article describes the first step in the process of developing and establishing the measurement properties of such an outcome measure. To facilitate its use in clinical practice and in research, we established feasibility benchmarks: (1) be easy to administer (completed in <30 minutes), (2) have understandable standardized instructions, and (3) require minimal material (common items found in hospital settings that can be assembled in <5 minutes). Preliminary results have appeared in abstract form. 22
Methods
The development of the Comprehensive Coordination Scale (CCS) was guided by the steps proposed by Streiner et al 23 for clinical outcome measure construction and content validity. The first step focused on the construction of the outcome measure and consisted of 3 phases: phase 1—instrument development; phase 2—Delphi process, and phase 3—focus group meeting. The second step, reported in the companion article, focused on testing the reliability and validity of the outcome measure.
Phase 1: Instrument Development
A literature review was conducted to identify unilateral and bilateral tests of UL, lower limb (LL), and trunk coordination currently used in clinical practice or research studies to assess adults with neurological injuries. Database selection included Embase and Pubmed. Two thematic blocks, their synonyms and variants were considered. The blocks consisted of MeSH (medical subject headings) terms “motor coordination” OR “motor performance” AND “assessment”. The search retrieved 2761 publications after duplicates were removed. After reading titles and abstracts, 624 articles were retained, and 62 tests used to assess motor coordination in adult populations were extracted. Tests were selected that assessed single-joint, multijoint, and/or multi-effector discrete, serial or rhythmical movements with different degrees of cognitive demands, without the use of specialized equipment or instrumentation. For example, discrete movements have distinct temporal characteristics (ie, longer reaction and movement times) then serial movements 24 and activate different brain areas such as frontal lobes, cerebellum, and basal ganglia, compared with rhythmical,25,26 bilateral, 27 or multi-effector movements. 28 Tests were included if quantitative and qualitative assessment in both temporal and spatial domains would be possible using observational kinematics. Tests excluded were those that involved a single joint (ie, one-finger tapping), stereotypical repetitive and nonfunctional (and often rhythmic) movements (ie, body-rocking, finger-flicking) or were used only in pediatric populations. If 2 similar tests were found (eg, heel-to-shin and LEMOCOT), the test with the best psychometric properties was chosen.
The first draft of the CCS included 5 tests assessing complementary aspects of motor coordination that were already familiar to neurological clinicians: (1) UL: Finger-to-Nose Test (FTN); (2) UL and trunk: Arm-Trunk Gain (ATG); (3) LL: Lower Extremity MOtor COordination Test (LEMOCOT); (4) UL: interlimb coordination (ILC) between ULs (ILC2); and (5) UL and LL: ILC between UL and LLs (ILC4). The original scoring was adapted by identifying distinct behavioral elements of the limb or segment movement that encompassed both temporal and spatial aspects of performance and movement quality. The initial CCS included 12 elements describing specific motor behaviors distributed across 5 tests. Coordination is assessed separately for each limb in 3 tests, FTN (5 elements: trunk movement, arm movement, endpoint smoothness, endpoint accuracy, movement stability), ATG (2 elements: endpoint accuracy, shoulder-elbow coordination), and LEMOCOT (2 elements: trajectory smoothness, endpoint accuracy), whereas bilateral coordination is assessed using the ILC2 (2 elements: synchronicity/timing, compensation) and ILC4 (1 element: complexity/timing) tests. For each behavioral element, rating scales from 3 (normal coordination) to 0 (impaired coordination) were developed. Qualitative descriptions of each element evaluated on each rating scale were also developed to guide the observational kinematic analysis. Two constructs of coordination were identified based on the levels of movement description: Performance level: timing/accuracy variables related to endpoint movement and Movement Quality level: variables related to joint/segment movement.
FTN is a common test of UL coordination, often used in cerebellar syndromes. 29 In the CCS, the traditional form was selected 30 in which participants perform alternate UL movements for 10 seconds by touching their nose and then a stationary 2.54-cm-target placed at the distance of the individual’s arm length at the level of the participant’s nose on the wall in front of them. Traditionally, performance on FTN is scored as the execution time and the amount of dysmetria and tremor.
The number of repetitions in the FTN test is moderate to highly correlated with dexterity (r = 0.82-0.84), global UL function (r = 0.74-0.79), functional independence (r = 0.74), and social participation (r = 0.78) and poorly correlated (r = 0.19-0.56) with muscle strength. 30 While the time measure has excellent test-retest and interrater reliability (intraclass correlation coefficient [ICC] > 0.9), scoring of dysmetria and tremor is less reliable (ICC = 0.26-0.40). 9 In another version, the time difference to alternately touch the knee and nose 5 times between the more- and less-affected ULs is calculated. 31 FTN time was correlated with impairment severity (Fugl-Meyer Scale–Upper Extremity: r = −0.67, P < .01) and a 10.6-second cutoff time discriminated between mild and moderate-to-severe impairment in patients with chronic stroke. Rodrigues et al 31 also found that shoulder horizontal abduction, shoulder flexion, elbow extension, and trunk kinematics explained more than 80% of the variance in the time taken to perform this FTN version.
Five behavioral elements were initially included in the CCS outcome measure. Four elements quantify endpoint trajectory smoothness (no jerkiness or discontinuous movements) and accuracy (endpoint touches the targets without dysmetria), as well as trunk stability and arm movement. In addition, the time to complete 5-movement cycles in seconds is measured. Correct performance is characterized by fast and smooth whole arm movements in which the endpoint (hand or finger) accurately touches the targets, shoulder level arm elevation is maintained throughout the test and no trunk displacement occurs.
ATG measures the coordination between arm and trunk movements. ATG has been characterized kinematically in healthy elderly individuals 32 and in patients with deafferentation, 33 vestibular deficits 34 and chronic stroke. 35 It is a recently validated test, 35 based on a fundamental principle of motor control, called “motor equivalence” 36 used to analyze the ability to adapt arm and trunk joint rotations to stabilize the endpoint position. To date, ATG has not been implemented in clinical settings. This test consists of maintaining the finger in the same position in space in front of the body while moving the trunk forward with eyes closed. To maintain the hand in the same position, the influence of the trunk movement on the hand position should be neutralized by appropriate changes in the arm joint angles.37-39 In the initial position, the arm is elevated in front of the body to ~70° of shoulder flexion and ~30° of shoulder abduction. The elbow is held at ~120° extension and the forearm is pronated so that the palm faces the floor. The tip of the extended index finger is positioned over, but not touching, the target. The target is placed on an adjustable support at the level of the subject’s mid-sternum in the midline at a distance equal to 2/3 arm length.
Two behavioral elements evaluate aspects of endpoint accuracy and shoulder-elbow interjoint coordination in the ATG. Correct performance is characterized by maintaining the arm in elevation, with the hand over the target while the trunk flexes forward. Maintaining the finger position above the target requires the coordinated action of the shoulder and elbow joint rotations to compensate for the forward trunk movement.
LEMOCOT measures LL coordination. The test was validated in people with stroke and Charlevoix-Saguenay populations with autosomal recessive spastic ataxia.16,40 The LEMOCOT consists of moving the toes of one foot as fast as possible from one round 5-cm target placed on the floor to another. The proximal target is located under the heel of the tested leg when the knee is flexed to 90° and the distal target is located sagittally at a distance of 30 cm. The LEMOCOT has good test-retest reliability (ICC = 0.83-0.88) and is moderately correlated with physical and functional LL tests (r = 0.62-0.79). It discriminates between individuals discharged to long-term care and other living environments. 15 The original version of the test rates endpoint performance by counting the number of targets touched by the toe in 20 seconds.
In the CCS version, 2 behavioral elements assess the smoothness and accuracy of the LL movement. The number of repetitions completed in 10 seconds is measured. A time of 10 seconds was chosen to equalize the time taken to perform each test and to minimize participant fatigue by limiting the time taken to complete the entire CCS. Correct performance is characterized by fast and smooth movements in which the toes accurately touch the targets.
ILC2 assesses the ability to produce stable spatiotemporal movement of both ULs, commonly used in neurological assessments. 41 Nevertheless, the psychometric properties of this test are not available. The test consists of the performance of rapid anti-phase pronation-supination movements of both forearms for 10 seconds. Seated participants maintain their forearms on their thighs with both arms held close to the trunk, while performing antiphase pronation/supination movements. These movements involve nonhomologous muscles and are considered more difficult than in-phase movements since homologous muscle activity is more easily coupled, and therefore more stable. 42 Patients with Parkinson’s disease show reduced maximal velocity and amplitude than controls and performance is associated with akinesia and rigidity. 41 One temporal aspect is assessed by rating synchronicity/timing. A second behavioral element assesses the spatial aspects of compensatory movements (eg, excessive shoulder abduction and/or lateral trunk flexion). Correct performance is characterized by alternate, synchronous movements of both arms (i.e., done at the same time and with the same timing) throughout the test, using the full range of forearm pronation and supination while maintaining elbows close to the trunk in a relaxed fashion.
ILC4 assesses the coordination of all 4 limbs intrinsic to many activities such as walking. Seated participants produce repetitive, symmetrical antiphase flexion-extension movements of both wrists and ankles. Participants start with both hands on the thighs, while both feet are dorsiflexed. Then, wrist and ankle actions are simultaneously reversed (ie, wrists move into extension and ankles into plantarflexion). The test is based on a model of coordination proposed by Kelso et al. 43 It has not been implemented clinically. Stable 4-limb coordination patterns arise as a result of cooperative dynamics based on spontaneous self-organization. 43 Less stable coordination patterns for this test were related to higher and more widespread brain activation in elderly compared to young subjects. 28 Activation of additional brain areas in elderly individuals may reflect compensation for age-related declines in neural functioning and/or difficulties in recruiting specialized neural mechanisms, including cognitive processes. One temporal aspect is assessed by rating synchronicity/timing. Correct performance is characterized by alternate and opposite movements between both hands and both feet made synchronously (ie, simultaneously). The number of repetitions completed in 10 seconds is measured.
Phase 2: Delphi Study
A Delphi study, using a structured questionnaire with open-ended questions was done to identify whether established benchmarks were met and to gather individual suggestions and perspectives about CCS by a panel of 8 experts. 44 Clinicians and researchers in neurological rehabilitation were purposefully recruited according to the following criteria: have expertise in either neurological rehabilitation and/or motor control and/or development/validation of scales; be working in an active treatment or research environment, and have at least 2 years of clinical/research experience. Efforts were made to recruit >50% clinicians, individuals of different professional backgrounds, both sexes, and from different centers, universities, and institutions. Selected experts were contacted by email by a third party not involved in the study. The email included a brief study rationale and described the nature of their participation. Experts signed an informed consent form approved by the Ethics Committee of the Centre for Interdisciplinary Research in Rehabilitation, according to the Helsinki agreement.
A questionnaire containing 4 sections was sent by email. Experts were asked to complete the questionnaire within a 2-week period and send the answers by email to a third party. The third party de-identified all questionnaires received. Sections I and II gathered sociodemographic and professional information, respectively. In section III, experts were asked to rate (1) the relative importance of each test, test element, and rating scale; (2) the level of comprehension of the instructions; and (3) the feasibility of each test. Experts rated each of the 12 test elements by drawing a line across a 10-cm horizontal line visual analogue scale (VAS) where the left extreme border indicated 0% (completely disagree) and the right extreme right border indicated 100% (completely agree). In section IV, 3 additional open-ended questions based on Coutu et al. 45 were asked on whether (1) the CCS fulfilled the purpose of evaluating coordination in patients with diverse neurological problems, (2) the tests and test elements were clear and specific, and (3) the CCS was feasible for use in clinical practice. Experts were also encouraged to provide additional suggestions.
Quantitative and qualitative data from the survey questionnaire were used to inform the next phase of development. To quantify responses, all questionnaires were printed and the distance between the line drawn across the horizontal VAS and the left border of the scale was measured in mm with a ruler for each question and reported as a percentage of the total line length. All written answers were completely transcribed. Hierarchical content analysis 46 was done by 2 independent members of the research team and general consensus was obtained.
Phase 3: Focus Group Meeting
A focus group meeting was held with 6 of the 8 experts who participated in phase 2. One expert was ill and the other could not participate for other reasons. The focus group aimed to obtain consensus on the wording of the instructions and the rating scales of each test element to ensure they were completely understandable by clinicians. 47 Experts were also asked to identify potential gaps, suggest additional behavioral elements and verify if the CCS covered all aspects of coordination. 23 The meeting was led by an experienced facilitator (MD) and cofacilitator, both having clinical and research expertise. The role of the facilitator was to balance the contribution of each expert, remain neutral about the proposed ideas and clarify key points through interactions and discussions, 48 whereas the cofacilitator summarized key discussion components and recorded suggested modifications to the instructions or rating scales for later analysis. The discussion was audio-taped.
The summarized unidentified responses from phase 2 were presented and the focus group discussed each response starting from those elements with cutoff scores of less than 70% on the VAS and continuing until a consensus was reached.
Following the focus group meeting, a final version of the CCS was created and shared by email with all experts to confirm that all benchmarks were achieved and to obtain additional feedback. Finally, 3 different experts, recruited with the same inclusion criteria as phase 2, reviewed all documents (first CCS draft, questionnaire results, focus group consensus, and final CCS version) to make comments and/or suggestions, if necessary. 47
Results
The expert group was composed of 8 individuals (3 occupational therapists [OTs], 5 physical therapists [PTs]) aged between 30 and 58 years, who had more than 5 years of experience. Three participants were academic researchers and 5 were clinicians, 2 of whom also held an academic position. The 3 additional experts contacted in the second round were 1 OT and 2 PTs, aged between 33 and 64 years. Two were academic researchers and 1 was a faculty lecturer. Clinical experience ranged from 5 to 25 years. All academic researchers had dedicated research programs for individuals with neurological disorders, and H-indexes ranging from 7 to 64. Three researchers had previous experience in the development of neurological outcome measures.
The FTN, LEMOCOT, ILC2, and ILC4 were all perceived to be important for assessing coordination in clinical practice and research, with a score of >70% on the VAS for importance. The focus group agreed that all the proposed behavioral elements and rating scales were important to assess and should be retained in the final version. Only the “arm movement” element of the FTN test that analyzes the ability of the individual to keep the arm elevated while moving the shoulder and elbow, had a low perceived importance rating (mean ± SD: 50.5% ± 33.0%). Experts discussed the difference between this element and movement smoothness. After discussion, they agreed to retain this element since it analyzes the stability of the arm while the smoothness element analyzes the quality of the endpoint movement trajectory.
A lower percentage on the VAS was found for the ATG test prior to the focus group. The averages for importance, level of comprehension, and feasibility for the whole test were 36.3% ± 28.2%, 38.4% ± 32.5%, and 61.5% ± 26.4%, respectively. The importance of the behavioral elements evaluated for this test was also considered low. The endpoint accuracy element was rated at 43.5% ± 31.7% and shoulder-elbow coordination element received a rating of 60.1% ± 31.5%. In the discussion of this element, it was pointed out that the ATG is the only test that measures the coordination between movements of the arm and the trunk. The experts recognized the unique contribution of the ATG test and agreed to retain it but to change the name to the Arm-Trunk Coordination (ATC) Test. To improve the level of comprehension, the wording of the instructions and element descriptions were modified (Table 1).
Results of the Questionnaire Completed by 8 Experts About Each of the 5 Tests Included in the First Version of the Comprehensive Coordination Scale.
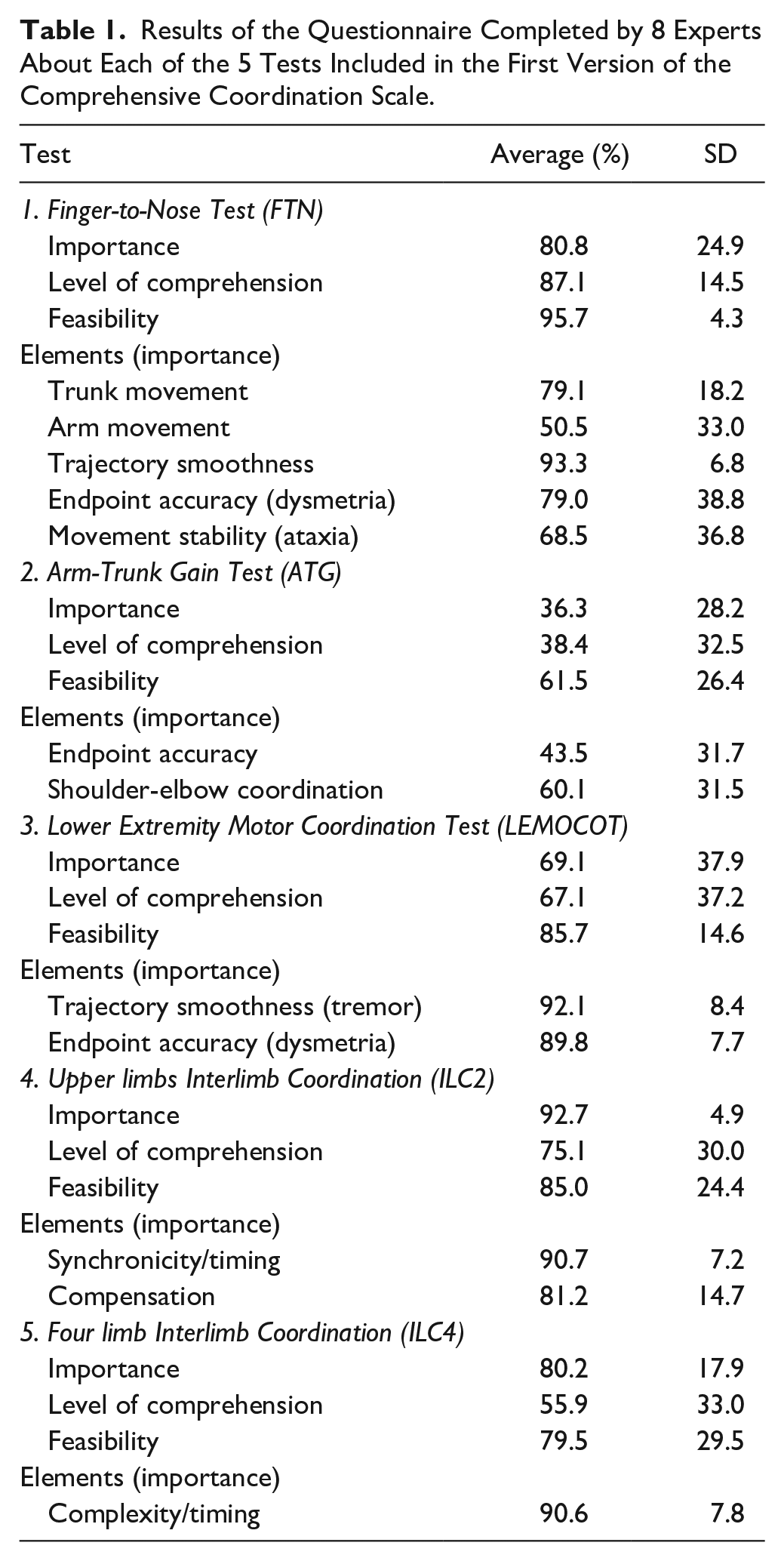
Experts identified a gap in the first version of the CCS regarding the assessment of hand/finger coordination. None of the 5 selected tests captured hand/finger coordination, which was perceived as being important for daily life activities. After considering different tests, their practicality and equipment needed, a consensus was reached to add the Finger Opposition Test (FOT) to assess the selectivity and timing of finger movements.
FOT is a classical clinical test of fine motor function requiring coordinated alternating activity in distal finger flexor and extensor muscles. The task consists of performing a sequence of finger-thumb opposition movements as rapidly as possible in the following order: index, middle, ring, little, ring, middle, repetitively, while maintaining a constant rhythm, performed for 10 seconds for consistency across all tests. During the performance of the task, the rest of the hand should remain open. In patients with stroke, the Barthel Index score at discharge could be predicted by the initial Barthel Index score and the speed of finger tapping at admission (r2 = 0.49) and FOT was a possible marker for predicting the activity of daily living after stroke. 49 Repetitive finger-thumb opposition for at least 20 seconds has high interrater reliability of 83% and has been used to identify upper motor neuron injury due to demyelinating lesions. 50 In the CCS version, one element assesses finger movement selectivity and one element assesses the timing of sequential finger-thumb opposition movements.
All tests had high perceived feasibility, except the ATG (61.5% ± 26.4%). Three of the 5 tests scored above the 70% level for ease of understanding (FTN, LEMOCOT, and ILC2). However, experts perceived that some of the instructions were difficult to follow, especially regarding the description of the initial positions. Experts recognized the importance of providing detailed instructions to ensure reliability and suggested using pictures to facilitate comprehension and standardization. A consensus was reached on the wording of all instructions, test names, test elements, and rating scales to improve readability and understandability for nonexpert clinicians.
Experts also perceived that tests could be applied individually or all together, depending on the patient profile and assessment goals. For example, patients with hemiparesis could be tested only on the paretic side using the unilateral test or tested only for UL coordination using the 4 UL tests.
To facilitate CCS administration, experts suggested changing the order of the tests, starting with UL assessment (FTN, ATC, FOT, ILC2), followed by LL (LEMOCOT) assessment and then the assessment of all 4 limbs (ILC4). Principal discussions and consensus points reached by the experts are summarized in Figure 1.

Consensus reached and main changes recommended at the focus group meeting.
Based on the results of the Delphi study and the focus group, a final version of the CCS was compiled and sent to three additional experts for review. No additional recommendations or suggestions were made. All experts agreed that the established benchmarks were met. The final version of the scale is presented in the Supplemental Appendix.
Discussion
The CCS was developed to objectively measure the underlying elements involved in the production of voluntary coordinated movements that may be impaired after a neurological insult in adults both at the movement performance and quality levels. The CCS was constructed from tests used in clinical practice and research for adults with neurological injuries. The final version of the CCS is composed of six tests: the FTN, ATC, FOT, ILC2, LEMOCOT, ILC4, all perceived as being important and complementary to assess the complexity of motor coordination. For each behavioral element, rating scales ranging from 0 (impaired coordination) to 3 (normal coordination) quantify different spatial and temporal aspects of motor performance and movement quality, totaling 69-points, with higher scores indicating better motor coordination.
Alternative behavioral strategies to enable the performance of tasks are observed after central nervous system injury that spans diverses impairments. It is important to identify alternative motor patterns since they have a strong potential to influence neural remodeling after a lesion. Alterations in synaptic connectivity patterns, intrahemispheric and interhemispheric inhibitory and excitatory activity, and changes in functional activation associated with maladaptive plasticity have been described. 51 A better description and quantification of the elements contributing to coordinated movement may help clinicians distinguish between motor recovery and compensation and assist in the development of individualized treatment plans to improve motor recovery. 8
The tests and instruments currently used in clinical practice to evaluate motor coordination normally assess the ability and time to perform a specific task.9,13,14,52 The qualitative aspects of the movement elements used to perform the tasks, however, are not regularly assessed. The CCS has well-defined constructs at both the endpoint performance and movement quality levels developed to address this gap. The scale differentiates between spatial and temporal aspects of coordination at the end-effector level (ie, performance level) by scoring endpoint (ie, hand, foot) movement. In addition, it provides a measure of the quality of movement of the arm/trunk/leg joints underlying the endpoint movement in order to quantify movement compensations. Temporal and spatial symmetry between effectors can be assessed in the interlimb coordination tests and by comparing unilateral scores of both sides. However, weightbearing symmetry is not assessed by the scale. Common barriers to the use of standardized assessment in neurological populations are the lack of time, cost, unavailability of the assessment tools, and lack of knowledge of how to administer an outcome measure. 53 The CCS could help address these barriers, as unlike complex and costly motion analysis systems, it requires little material, involves minimal training, and is easy to use. The advantage of the CCS is that it is the only clinical outcome measure that provides a comprehensive assessment of coordination at 2 levels of description fundamental to the understanding of underlying motor deficits. The CCS could be used in hospital-based settings, outpatient clinics and research studies to assess sensorimotor coordination in individuals with neurological injuries as a means to identify underlying deficits to be targeted in treatment.
This study has some limitations. First, it is not possible to exclude the possibility of bias in the choice of the tests included in the scale, since a systematic review was not done. However, all relevant scales identified in the literature review were included in the original CCS, and one additional test was added following the focus group consultation to obtain the final version. Detailed descriptions of the path and decision-making processes in the development of the CCS are described. To reduce the risk of bias, a Delphi study and a focus group meeting were conducted with experts purposefully selected for their clinical and research expertise. After choosing tests and constructs and developing scales for each motor element, final decisions were made by individuals not involved in scale development. Second, validity and reliability testing have to be done in different populations before the CCS can be used in clinical settings. The CCS was constructed to be administered in distinct neurologic populations in the clinical context. The generalizability of the final scores of the CCS should be tested and compared between different populations and age ranges. As motor coordination may be differently impaired in different neuropathologies, it is likely that ratings of specific elements of a test may better reflect specific impairments than the total score. Future reliability studies of the CCS are necessary to determine the applicability of the total score and individual test subscores in different populations.
Conclusion
This study demonstrated that the CCS may be an important, understandable, and feasible outcome measure for assessing coordination at and between multiple body segments. The final version of the CCS is composed of six different tests: the FTN, ATC, FOT, ILC2, LEMOCOT, ILC4. The CCS is a first step in filling the gap of clinically identifying and quantifying coordination in individuals with neurological problems. The next step in the validation of the CCS is to determine its measurement properties in a specific population, which is the topic of the companion paper.
Supplemental Material
sj-pdf-1-nnr-10.1177_1545968320981955 – Supplemental material for Development of a Comprehensive Outcome Measure for Motor Coordination; Step 1: Three-Phase Content Validity Process
Supplemental material, sj-pdf-1-nnr-10.1177_1545968320981955 for Development of a Comprehensive Outcome Measure for Motor Coordination; Step 1: Three-Phase Content Validity Process by Sandra R. Alouche, Roni Molad, Marika Demers and Mindy F. Levin in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Rejean Prevost for his assistance, all experts who provided their valuable feedback, and Sandrine Garneau for the figures.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SRA was supported by Grant No. 2018/04544-9, Sao Paulo Research Foundation (FAPESP), Brazil. MD was supported by the Fonds de la Recherche du Québec en Santé (FRQS). MFL is a Distinguished James McGill Professor.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
