Abstract
Background
Studies on short-term upright quiet standing tasks have presented contradictory findings about postural control in people with Parkinson’s disease (pwPD). Prolonged trial durations might better depict body sway and discriminate pwPD and controls.
Objective
The aim of this study was to investigate postural control in pwPD during a prolonged standing task.
Methods
A total of 26 pwPD and 25 neurologically healthy individuals performed 3 quiet standing trials (60 s) before completing a constrained prolonged standing task for 15 minutes. Motion capture was used to record body sway (Vicon, 100 Hz). To investigate the body sway behavior during the 15 minutes of standing, the analysis was divided into three 5-minute-long phases: early, middle, and late. The following body sway parameters were calculated for the anterior-posterior (AP) and medial-lateral (ML) directions: velocity, root-mean-square, and detrended fluctuations analysis (DFA). The body sway area was also calculated. Two-way ANOVAs (group and phases) and 1-way ANOVA (group) were used to compare these parameters for the prolonged standing and quiet standing, respectively.
Results
pwPD presented smaller sway area (P < .001), less complexity (DFA; AP: P < .009; ML: P < .01), and faster velocity (AP: P < .002; ML: P < .001) of body sway compared with the control group during the prolonged standing task. Although the groups swayed similarly (no difference for sway area) during quiet standing, they presented differences in sway area during the prolonged standing task (P < .001).
Conclusions
Prolonged standing task reduced adaptability of the postural control system in pwPD. In addition, the prolonged standing task may better analyze the adaptability of the postural control system in pwPD.
Introduction
People with Parkinson’s disease (pwPD) usually present impaired balance. This symptom increases instability, leading to a high number of falls in this population.1,2 However, short-term upright quiet standing task studies (e.g. standing as still as possible for 60-120 s) have presented contradictory findings, indicating that this population has a smaller3,4 or larger5-7 body sway than their neurologically healthy peers. Nevertheless, the studies showing smaller body sway in pwPD were related either to the margin of stability and/or perturbations. In addition, these contradictory findings may be a result of the short duration of the standing trials (from 15 to 60 s). A study indicated that prolonged trial durations (long-term natural stance) might better depict body sway and discriminate those with Parkinson’s disease and controls. 7 In addition, to detect the failure of the postural control to properly respond over a long time requires a prolonged standing task, 8 especially for complex analysis (eg, detrended fluctuation analyses [DFA]) of the postural system. 9 Indeed, a better postural control adaptability representation may be observed during a prolonged standing trial in pwPD. Longer-term natural stance is defined as remaining in an upright stance for a continuous period of time without rest (e.g. more than 5 minutes). 10 However, it is important to highlight that no previous work has studied the effects of long standing periods on body sway in pwPD.
The dopaminergic degeneration in the brain, especially in the substantia nigra, caused by Parkinson’s disease impairs postural reflexes and reduces stability limits, making postural adjustments more difficult.11-13 The reduced postural responses in pwPD can be explained by muscle stiffness and excessive antagonist muscle contraction during standing. 14 This rigidity also impairs postural adjustments. 15 In addition, brain structure impairments affect the compensatory movements to regain body balance in pwPD. 16 These Parkinson’s disease–related symptoms can lead to a decrease in postural control complexity (e.g. interaction of many physiological functions that assist in absorbing perturbations and allow subjects to adapt) 17 and, consequently, in the postural control adaptability (eg, the capacity of the postural control system to sense and adapt to changes in external and internal constraint). 8 However, postural control complexity and adaptability have been little explored in pwPD. 18 Another aspect that can impair the postural control adaptability in pwPD is the individual’s intensified perception of fatigue.19,20 Thus, it is probable that pwPD would perform prolonged standing with minimal effort and reduced postural adjustments, avoiding any increase in musculoskeletal discomfort, as in older people. 21 Therefore, studying upright prolonged standing may clarify the postural control adaptability in pwPD.
Although prolonged standing is a usual daily activity and a healthy behavior, 22 this posture can increase the risk of musculoskeletal system failure.23,24 A prolonged upright posture requires resistance to fatigue, 25 especially in older people. Young adults constantly shift body weight between feet to vary internal load and release musculoskeletal discomfort and fatigue. 26 On the other hand, older people reduce body sway in a prolonged postural task, not performing weight shifting, to avoid instabilities and reduce energetic cost.8,21,27,28 This behavior suggests less mobility21,29 and a conservative strategy to avoid support base changes and falls. 28 This strategy adopted by older people suggests that postural control is impaired for weight transfer, choosing a less complex and more predictable behavior.9,30 Based on this context, because pwPD tend to sway within and outside their base of support, they may be unable to perform compensatory movements to regain body sway, which increases instability and falls.7,31 In addition, we expected that pwPD would exacerbate the behavior presented by older people during prolonged standing, further reducing body sway and complexity of the postural control system.
The purpose of this study is to investigate the postural control in pwPD during a prolonged standing task. Both pwPD and neurologically healthy individuals (control group) performed prolonged standing in a semirestricted stance for 15 minutes. Prior to the prolonged protocol, these individuals performed 3 quiet standing trials (60 s). Body sway was measured in both the prolonged and quiet standing trials. Because of reported PD-related symptoms, we expected that pwPD would show smaller body sway (sway area) and complexity (DFA values) than the control group in the prolonged standing task, mostly in the last 5 minutes, considering that fatigue is intensified in Parkinson’s disease, leading to a reduction in postural adjustments. In addition, we expected postural control adaptability in pwPD to be different between quiet and prolonged standing trials (measured by the DFA) and that short standing trials would show higher body sway, 32 whereas prolonged standing trials would show reduced body sway.
Method
Participants
The study included 26 pwPD and 25 neurologically healthy individuals (Table 1). The inclusion criteria for both groups were as follows: >60 years old, not having orthopedic problems that hindered performance of the experimental protocol, and not presenting cognitive decline (score greater than 24 points on the Mini Mental State Examination [MMSE]). 33 For pwPD, the following inclusion criteria were applied: (1) diagnosis of idiopathic PD according to UK Brain Bank criteria, 34 (2) Hoehn and Yahr stage I to III, 35 and (3) stable medication for at least 3 months prior to the visit. All pwPD took levodopa medicine. The equivalent daily dose of levodopa was made according to the study of Lozano et al. 36 Participants who did not complete the entire experimental protocol were also excluded from the sample. The local university’s ethical committee approved the study (No. 97193318.1.0000.5398). Informed consent was obtained from all participants. pwPD were evaluated under effects of dopaminergic medication (e.g. 1 hour after taking antiparkinsonian medication).
Means and SDs of General Characteristics and Clinical Parameters for pwPD and Control Groups, and F Values. a
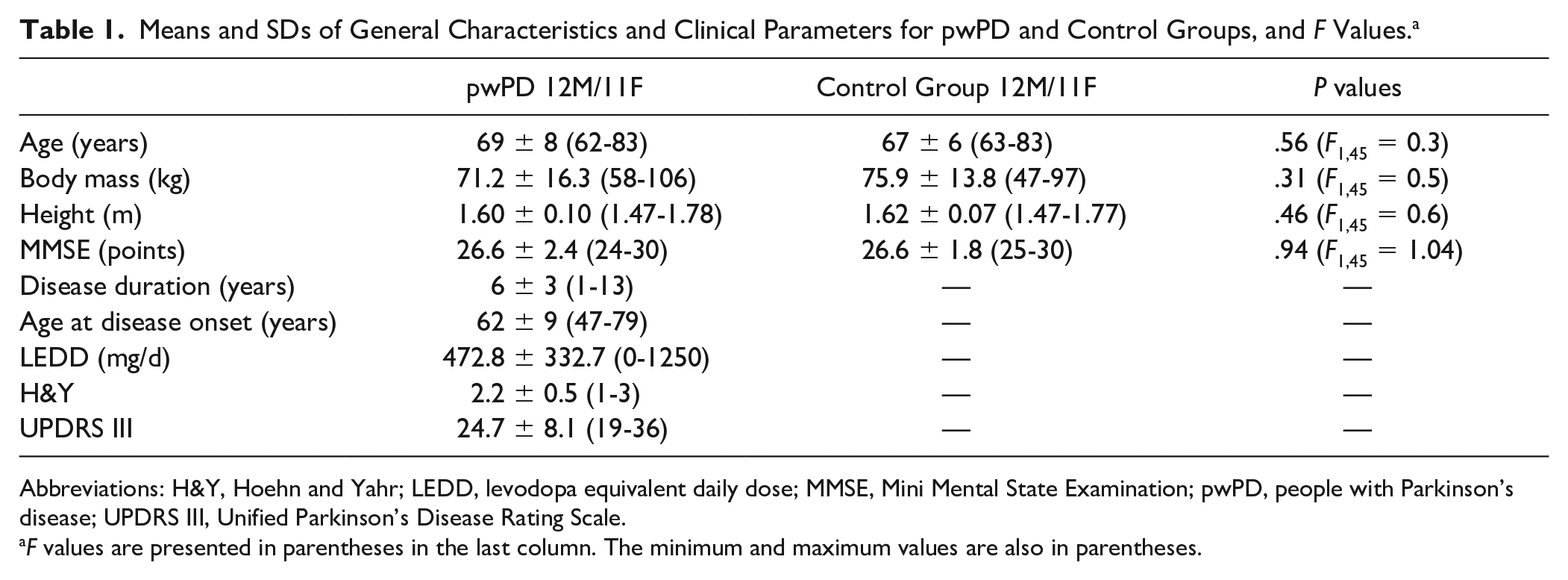
Abbreviations: H&Y, Hoehn and Yahr; LEDD, levodopa equivalent daily dose; MMSE, Mini Mental State Examination; pwPD, people with Parkinson’s disease; UPDRS III, Unified Parkinson’s Disease Rating Scale.
F values are presented in parentheses in the last column. The minimum and maximum values are also in parentheses.
The body sway data were measured by 4 cameras (Vicon Motion System, Bonita System Cameras) with a 100-Hz sampling frequency. A single passive and reflective marker positioned on the second lumbar vertebra (L2) was used to measure the body sway. This is a validated protocol used by previous studies.37,38 Body sway was measured during the prolonged standing and during the short-duration quiet standing tasks.
Clinical and Cognitive Assessment
Clinical and cognitive assessments were performed by an experienced evaluator. The clinical aspects and motor impairments of pwPD were assessed using the Hoehn and Yahr Scale and Unified Parkinson’s Disease Rating Scale, 39 respectively. The cognitive aspect of both groups was assessed using the MMSE.
Short-Duration Quiet Standing Task
The participants performed 3 short-duration quiet standing trials (60 s) before the prolonged standing task. Each of the 3 standing trials was conducted consecutively and standardized for every participant. For short-duration quiet standing, the participant was asked to remain as still as possible, with arms alongside the body and looking fixedly at a projected point on a monitor positioned at the height of the head.
Prolonged Postural Task
Participants stood in an upright stance for 15 minutes with feet parallel, hip width apart, barefoot, staring at a display 2 m in front of them. To maintain the foot position between trials, the position was drawn on a sheet. The participants were instructed to remain as comfortable as possible (freely move the body), but not to step off the sheet placed on the floor. The participants watched a TV documentary during the task. A semirestricted stance was used to mimic tasks during daily life activities.
Data Analysis
A MATLAB script (MathWorks Natick, MA, 2015) was used for data analysis. Body sway displacement was low-pass filtered with a fourth-order Butterworth filter with 5-Hz cutoff frequency. The following parameters of body sway for anterior-posterior (AP) and medial-lateral (ML) directions were analyzed: velocity (dividing the total body sway by the time in each direction) and root mean square (RMS; corresponding to the mean variability of the displacement in the trial). 40 In addition, the sway area (95% of the ellipse) and nonlinear variables were analyzed using DFA.
For the DFA, a fourth-order low-pass Butterworth filter with a 5-Hz cutoff frequency was used. DFA is calculated from the integration of the original time series, which is then divided into windows of equal size. The RMS fluctuation is calculated using the equation below, where F(n) is the fluctuation magnitude in window n, N is the total number of points in the series, y(k) is the time series integrated at interval i, and yn(k) is the y coordinate of the straight-line segments.
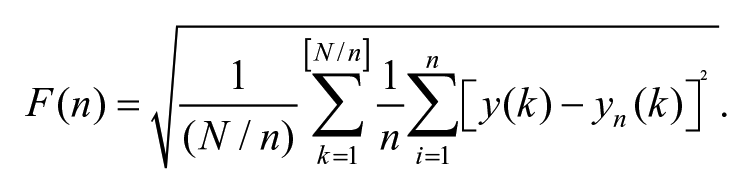
This calculation is performed over all time series to investigate the relationship between fluctuations and windows. 8 These fluctuations are plotted on a log-log graph, and the slope coefficient is calculated (scaling exponent α). The α can be interpreted as white (α < 0.5), brown (α > 1.5), or pink noise (0.5 < α < 1.0). 41 White noise is the absence of dependence from past or future states, representing an uncorrelated signal in long-range correlations and not a complex system, being not adaptable. On the other hand, pink noise represents a strong long-range correlation and represents a complex and adaptable system. In addition, brown noise presents random drifts and is dependent on the previous state, but also presents randomness from point to point. 42
Body sway during the prolonged postural task was divided into three 5-minute-long phases: early, middle, and late. Next, each phase was analyzed by 10 nonoverlapped 30-s moving windows. Subsequently, the average of each variable in the 3 periods was calculated. For the short-duration tasks, the first 10 s were removed.
Statistical Analysis
The level of significance was P <.05 for all analyses, and SPSS 21.0 software was used for statistical analysis. The Shapiro-Wilk, Mauchly, and Levene tests confirmed that all variables presented normal distribution, sphericity, and data homogeneity, respectively. For prolonged standing, 2-way analysis of variance (ANOVA) was applied to compare the body sway parameters across phases (early, middle, and late) and groups (pwPD and control group). For the short duration, 1-way ANOVA was applied to compare the same parameters between groups (pwPD and control group). The Tukey post hoc test was used, with significance levels adjusted when a significant interaction between factors was indicated in both analyses. The Cohen d was estimated and used as the effect size for group comparisons in short-duration trials. 43
Results
Two pwPD did not complete the prolonged standing task; they stopped the test before the end of 15 minutes (1 participant stood for 5 minutes and the other for 9 minutes). We did not include these participants in the study. We also had measurement problems with 1 pwPD and 2 control individuals (the software turned off, deleting their data). Thus, 23pwPD and 23 control individuals were evaluated. None of these participants reported musculoskeletal discomfort or fatigue during the prolonged standing task. Age, height, body mass, and cognition were similar between the groups (Table 1).
Short-Duration Quiet Standing Task
During the short quiet standing trials, the statistical analysis showed significant differences between groups (P < .05). pwPD showed higher values for AP velocity and RMS and AP and ML DFA compared with the control group (Table 2).
Means and SDs of Body Sway Parameters During the Short Standing Task for pwPD and Control Groups. a
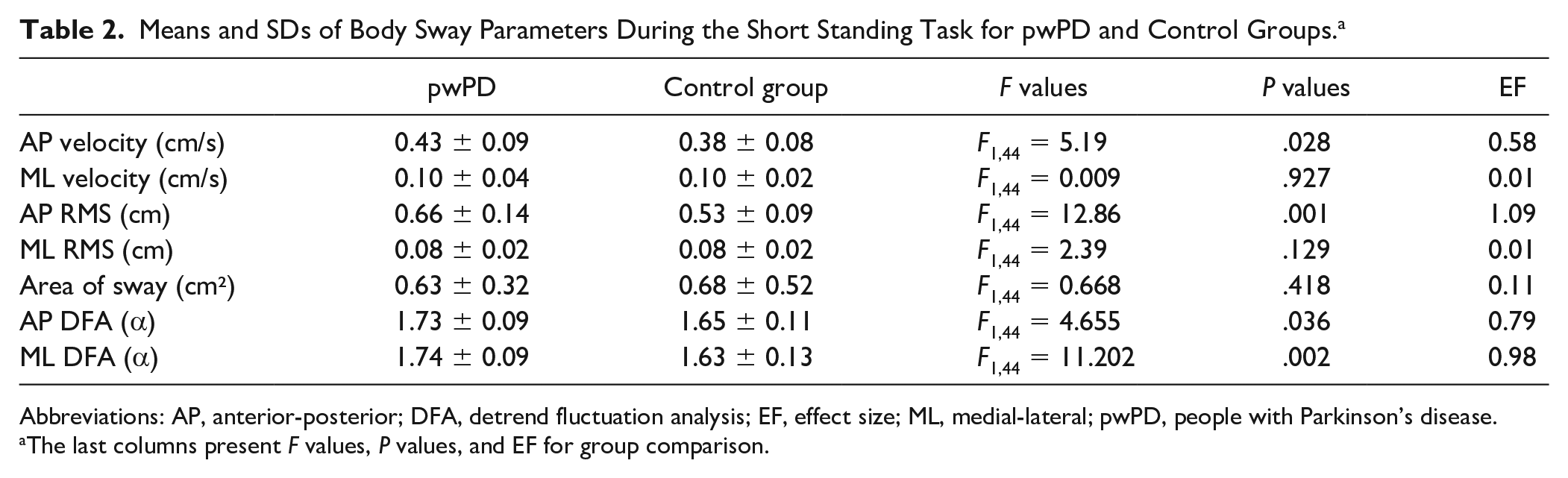
Abbreviations: AP, anterior-posterior; DFA, detrend fluctuation analysis; EF, effect size; ML, medial-lateral; pwPD, people with Parkinson’s disease.
The last columns present F values, P values, and EF for group comparison.
Prolonged Standing Task
Figure 1 illustrates the body sway (AP: Figures 1A and 1B; ML: Figures 1C and 1D; sway area: Figures 1E and 1F) from a representative pwPD individual and a representative control group individual during the prolonged standing task.
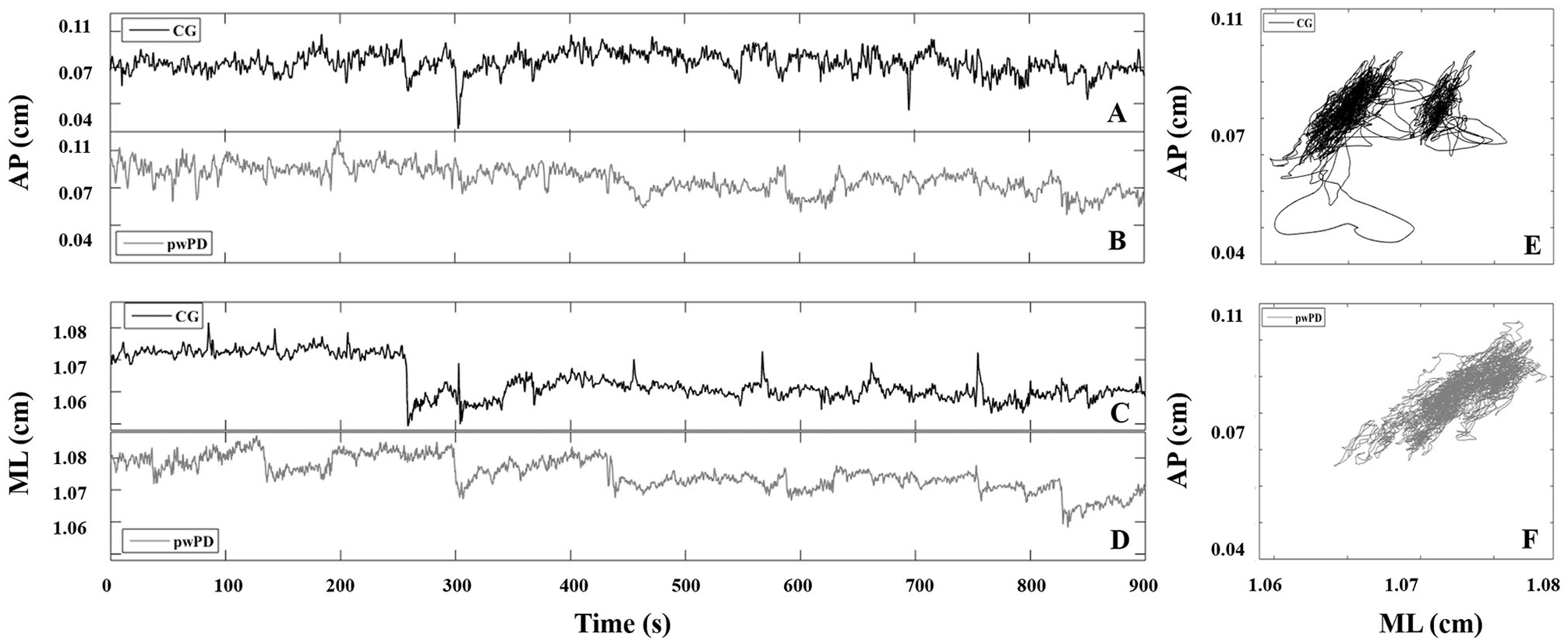
A typical example of the AP (A, B) and ML (C, D) body sway and area of sway (E, F) for pwPD and the CG. pwPD (B, D, F) are represented by gray lines and the CG (A, C, E) by black lines.
We found no main phase effects (early, middle, and late: P > .05). On the other hand, pwPD (main group effects) showed higher values of AP and ML velocity (P < .002 and P < .001, respectively), RMS (P < .001), and DFA values (P < .009 and P < .015, respectively) and lower sway area compared with the control group during prolonged standing (Table 3).
Means and SDs of Body Sway Parameters During the Prolonged Standing Task According to Group and Phase. a
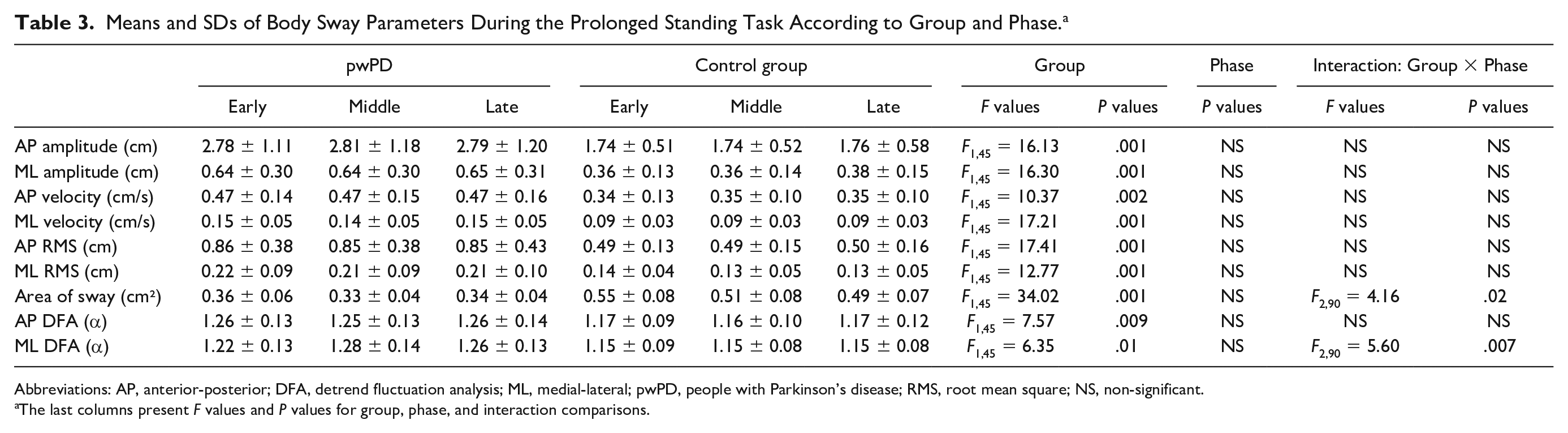
Abbreviations: AP, anterior-posterior; DFA, detrend fluctuation analysis; ML, medial-lateral; pwPD, people with Parkinson’s disease; RMS, root mean square; NS, non-significant.
The last columns present F values and P values for group, phase, and interaction comparisons.
There was a group versus phase interaction for sway area and ML DFA values. The Tukey post hoc analysis indicated that the prolonged standing task led to increased ML DFA values during middle (P < .001) and late (P < .015) phases compared with the early phase in pwPD (Figure 2). For the control group, the prolonged standing task reduced sway area during the late phase compared with the early phase (P < .05).
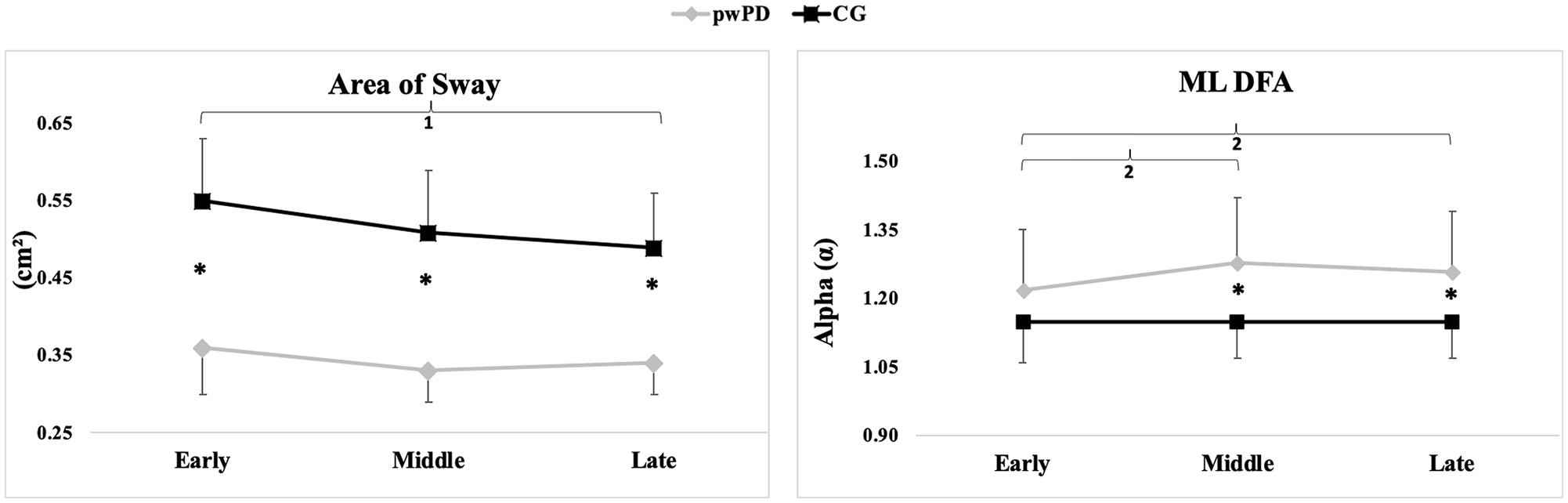
Group and phase interaction for area of sway (A) and ML DFA (B; means and SDs).a
Discussion
The main hypothesis of the study was that pwPD would present reduced body sway and sway complexity during prolonged standing compared with neurologically healthy individuals. Our results confirmed this hypothesis. pwPD presented reduced body sway (smaller sway area) and postural control complexity (higher DFA values) compared with the control group during prolonged standing. In addition, pwPD presented faster sway than the control group. These findings suggest a failure of the postural control to generate an adequate response and reduced adaptability in pwPD during prolonged standing. 8 On the other hand, we found a small effect in the late phase during prolonged standing in both groups, which contradicts our first hypothesis. The second hypothesis, that postural control adaptability in pwPD would be different between short and prolonged standing trials, was confirmed. Although short and prolonged standing trials presented some similar parameters for short standing trials, area of sway was different between groups for prolonged standing. Both groups presented small complexity over short standing trials. These findings may indicate that the prolonged standing test was more sensitive for pwPD to describe postural control adaptability. In the following paragraphs, we present interpretations for the prolonged standing effect on postural adaptability in pwPD and explanations for the different effects between short and prolonged standing trials.
Postural Adaptability in pwPD During the Prolonged Standing Task
In prolonged standing, pwPD presented smaller sway area and complexity as well as faster and larger sway. This effect remained for all prolonged standing intervals (phases of task). Young adults showed greater sway size variability (RMS) during the prolonged standing (30 minutes of standing) to avoid musculoskeletal discomfort and fatigue.21,44,45 Aging reduces the adaptability of postural control, decreasing postural shifts and, consequently, body sway (sway area). 8 Because pwPD present reduced postural responses as a result of reflex impairments11-13 and brain structure alterations, 46 they have enhanced prolonged standing effects, reducing the postural changes. Less postural changes and sway area are conservative strategies during prolonged standing.28,47 In addition, faster sway during prolonged standing in pwPD is an aging-related effect,48,49 meaning a decline in neural processing and delayed muscle activation, which affects feedback and feedforward control and decreases sway. 50 In addition, although we did not measure the proprioceptive sensitivity, it is necessary to consider the peripheral neuropathies, occasioned by the aging process and Parkinson’s disease, in the reduction in body sway. 21
A smaller sway area and faster sway are suboptimal postural changes necessary for standing for a prolonged period. 22 For pwPD, this suboptimal response may cause an inadequate response to an external perturbation and may result in a fall. This behavior (smaller sway area and faster sway) explains the reduced sway complexity during the prolonged standing task in pwPD, with body sway tending toward a Brownian noise, suggesting less adaptable postural behavior.41,51 We can also suggest that pwPD cannot explore the limits of stability during prolonged standing because of their inability to deal with complex balance challenges, as with neurologically healthy older adults. 8 Duarte and Zatsiorsky 27 addressed 3 postural changes: shifting (fast average sway position displacement from one region to another), fidgeting (fast and relatively large sway displacement followed by a return to the same position), and drifting (slow, continuous average sway position displacement). Although we did not calculate these postural changes, we suggest the lower complexity in pwPD to be a result of less shifting and fidgeting, but more drifting. Thus, pwPD did not move the sway position from one region to another (see Figure 1F), maintaining a continuous sway displacement, caused by higher sway variability (indicated by the RMS) around the same average position, leading to a smaller sway area. Less weight transfer reinforces the nature of postural control in pwPD: reduced adaptability, which is less complex and more predictable. Although neurologically healthy individuals also presented aging effects on postural control, they showed more adaptable control during prolonged standing, matching their DFA values with other studies, 8 fidgeting to adapt to the prolonged standing task (see Figure 1E). This is a compensatory adjustment during a prolonged standing task.21,44,45
During prolonged standing in pwPD, postural control presents dysfunctional feedback from continuous and intermittent control. Continuous feedback control regulates the control variable state (center of mass position) continuously updating the output (motor response),52,53 using information from 3 time domains (present, past, and future) regarding the control variable error to shape the output. 54 Intermittent feedback control regulates preprogrammed corrective responses using triggered episodic ballistic events, which better account for the variable feedback delays from neural processing.55-57 Parkinson’s disease impairs the pedunculopontine nucleus and reticulospinal pathways, which are responsible for controlling continuous and intermittent feedback. 58 A system failure in pwPD leads to compensatory overdrive of another system. For example, a continuous control failure, such as increased variability sway, requires a compensatory overdrive from the intermittent system, such as faster sway, or a failure in intermittent control reduces sway and sway changes, requiring more from the continuous system. These 2 systems increase interactions during prolonged standing; however, this interaction seems to be dysfunctional in pwPD, increasing postural instability and falling risk. The continuous and intermittent control are regulated by feedback from the somatosensory system, 59 but pwPD have sensory and perceptual deficits,3,60 exaggerating mechanical body adjustments during challenging postural tasks. 13
The last important issue is the small effect in the late phase during prolonged standing in both groups. Smaller sway area for the control group and increased ML DFA values for pwPD were observed, which gives small support to our hypothesis. Previous studies found effects after prolonged standing in older and younger adults.21,28,45 In these studies, prolonged standing was performed for at least 30 minutes, whereas we measured only 15 minutes. Considering the ability of pwPD to stand for such a long time, we were conservative (yet even so, 2 pwPD were unable to finish the prolonged standing task). It is probable that more participants would have failed if the prolonged standing task had lasted more than 15 minutes. On the other hand, we suggest that postural control changes in the late phase could reflect fatigue: whereas the control group constrained body sway, pwPD further reduced adaptability. These changes increase the falling risk, but can also be understood as adjustments to deal with the task. Fatigue effects on postural control were observed in different populations,61-63 although these studies induced fatigue by exercise and not by a standing task. Thus, the effects of fatigue on postural control may be interpreted according to the task. However, our explanation should be interpreted with caution because fatigue perception/symptoms were not measured.
Short Versus Prolonged Standing Task in pwPD
The sway nature during short and prolonged standing trials is not the same. During short standing trials, the participants were required to stand as still as possible, whereas during prolonged standing, the participants could freely move the body but not the feet. In addition, prolonged standing is a more challenging task than short standing because the postural control needs to continuously shift the bodyweight, changing the body scheme, aiding balance and preventing falls. 28 pwPD possibly tightly controlled the posture for 60 s without great effort because short-duration quiet standing with all sensory information available does not demand postural adaptation. Postural control has short latency responses, and long latency responses are only available during prolonged standing. As a consequence, for short-duration quiet standing, when only the short latency responses are possible, the aging effect equalizes both groups, inducing similar velocity (AP and ML), sway area, ML amplitude and RMS, and AP DFA values. Therefore, prolonged standing trials may afford a better representation of postural control in pwPD and may better discriminate between neurologically healthy individuals and those with Parkinson’s disease.
Limitations
Despite the interesting results, our data should be interpreted with caution. First, we did not analyze the postural changes (shifting, fidgeting, and drifting), which could improve our understanding of postural control during a prolonged standing task in pwPD. However, this is the first exploratory study on prolonged standing in pwPD. Thus, we suggest future studies should evaluate postural changes in prolonged standing. Second, the participants performed the prolonged standing with feet position fixed. This instruction could increase the musculoskeletal discomfort and fatigue. Although there are no reports about this aspect, it should be considered in our findings. Third, during the prolonged standing task, it is possible that watching the documentary could provide an additional attentional demand differently between healthy people and pwPD, which may influence the postural responses during the trial. Finally, we did not statistically compare short versus prolonged standing variables. Our interpretation of the second hypothesis was based on the differences in conditions affecting body sway parameters in pwPD. Therefore, future studies should confirm our findings.
Conclusion
We can conclude that the prolonged standing task reduced postural control adaptability in pwPD compared with neurologically healthy individuals. pwPD presented reduced body sway and increased sway velocity during prolonged standing. In addition, the prolonged standing task may better analyze the adaptability of postural control in pwPD. Therefore, we recommend that (1) pwPD should be careful when standing for prolonged periods and (2) postural control adaptability in pwPD should be investigated during prolonged standing tasks and not during short-duration quiet standing trials. However, prolonged standing trials do not yet have clear clinical applicability. Future studies should investigate if prolonged trials are more sensitive to analyze postural control adaptability compared with short standing trials in pwPD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES; Finance Code 001).
