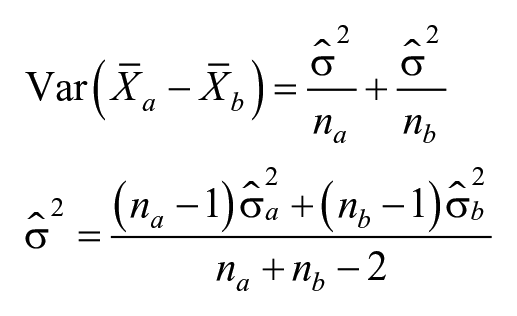Abstract
Background
Adequate sleep is vital for health and quality of life. People with stroke and a concomitant sleep disorder may have poorer outcomes than those without a sleep disorder.
Objective
To systematically evaluate the published literature to determine the impact of sleep disorders on physical, functional recovery at the activity and participation level after stroke.
Methods
A systematic review was conducted using PubMed, CINAHL, Scopus, and PsycINFO. Studies were selected that reported outcomes on physical, functional recovery at the activity and participation levels in participants with stroke and a diagnosed sleep disorder. A meta-analysis was performed on included studies that reported Barthel Index (BI) and modified Rankin Scale (mRS) scores. Results: A total of 33 studies were included in the systematic review with 9 of them in the meta-analysis. The mean mRS score was 0.51 points higher in participants with stroke and sleep disorders versus participants with stroke without sleep disorder [95% CI: 0.23-0.78]. The mean BI score was 10.2 points lower in participants with stroke and sleep disorders versus participants with stroke without sleep disorder [95% CI: −17.9 to −2.6].
Conclusions
People with stroke and a sleep disorder have greater functional limitations and disability than those without a sleep disorder. Rehabilitation professionals should screen their patients with stroke for potential sleep disorders and further research is needed to develop sleep and rehabilitation interventions that can be delivered in combination. PROSPERO registration number: CRD42019125562.
Introduction
Sleep is vital for overall health and quality of life.1,2 Abnormal sleep is associated with obesity, diabetes, cardiovascular disease, hypertension, poor mental health, and dementia.3 -9 Sleep disorders are also associated with mood disorders10,11 and cognitive disability.12 -14
Disordered sleep is both a risk factor and consequence of stroke.15 -21 For example, in a recent meta-analysis, Li and colleagues 15 reported that obstructive sleep apnea (OSA) is associated with an increased risk of stroke (relative risk of 2.10). Sleep apnea also appears to be the most common sleep disorder after stroke. 22 An estimated 40% to 92% of people with stroke have sleep disordered breathing (SDB). 18 In addition, other sleep disorders such as insomnia 23 and periodic limb movements 24 may also occur after stroke.
Sleep disorders may be more relevant than previously recognized in the context of stroke recovery and rehabilitation. Sleep appears to have an important moderating influence on motor learning, which is a central mechanism of rehabilitation interventions after stroke. This impact is likely a result of consolidation during sleep after motor skill practice.25 -28 In people with chronic stroke, Siengsukon and Boyd 29 found that sleep enhanced off-line motor learning of both implicit and explicit motor tasks. Other research has found that after a night’s sleep, people with stroke perform newly learned motor tasks with less error. 30 Sleep also appears to be important for motor learning of functional tasks in older adults. 31 It is thought that consolidation of procedural memory occurs during rapid eye movement (REM) sleep by enhancing long term potentiation (LTP) and neuroplasticity. 27 In stroke this is supported by the findings of Siengsukon and colleagues 30 who found that time in REM sleep was related to the magnitude of off-line motor learning. Additionally, other factors that are negatively affected by disordered sleep in people with stroke such as cognition, 32 attention,32,33 anxiety, 34 and depression 34 make active participation in stroke rehabilitation challenging and are associated with worse rehabilitation outcomes after stroke.
There is evidence to suggest that individuals with sleep disorders across the stages of recovery after stroke appear to have worse outcomes than those who do not have sleep disorders.35 -37 Individuals with stroke and SDB are more dependent both early and late after stroke compared to those without SDB,35,36,38 have longer lengths of stay in the hospital, 36 and poorer quality of life.37,39 Insomnia and periodic limb movements with sleep may also negatively impact recovery after stroke.40,41 Balance and quality of life have been found to be different in patients with stroke with insomnia versus those without insomnia.23,40 People with stroke and restless leg syndrome are more dependent and have poorer quality of life early and later after stroke24,42 More recently, Fleming and colleagues 43 found that sleep quality was associated with poor functional and motor outcomes during in-patient rehabilitation after stroke. In this study, the authors did not identify any specific sleep disorder in the participants. These findings indicate that sleep likely plays an important role during rehabilitation after stroke and abnormal sleep may negatively impact outcomes during rehabilitation as well.
Sleep has a profound impact on many aspects of health and life. It also appears that sleep may impact recovery after stroke. However, there are conflicting findings as some studies have reported no differences in functional outcomes between those with and without a sleep disorder after stroke rehabilitation.44 -46 The growing body of literature in this area has not been previously synthesized. Therefore, the purpose of this systematic review was to evaluate the published literature to determine the impact of sleep disorders on physical, functional recovery at the activity and participation level after stroke.
Methods
A systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement. 47 The protocol of this systematic review was registered with PROSPERO, an international database of prospectively registered systematic reviews (PROSPERO registration number: CRD42019125562).
Information Sources and Search Strategy
Four databases—PubMed, CINAHL, Scopus, and PsycINFO—were searched for potentially relevant articles. Search strategies for each database were developed, tested, and refined in collaboration with a medical librarian (ARS). Key concepts (sleep disorders and stroke) were broadly defined, using combinations of controlled vocabulary (where available) and keywords, to maximize the probability of finding all relevant articles. All searches were run on November 30, 2018, with no restriction on publication date. Search results were limited to English-language articles. Complete search strategies are presented in Supplemental Appendix 1. In addition to these database searches, 2 other data sources were included: (1) contents of the journal Sleep Medicine Research were searched separately, as its contents are not fully indexed in any of the included databases and (2) articles listed in the reference lists of included articles were reviewed for eligibility in this study.
Study Eligibility
Studies were included if the participants had a diagnosis of a stroke, at least some of the participants had a diagnosis of a sleep disorder as defined by the American Academy of Sleep Medicine (AASM), and reported outcomes that included measures of physical function at the activity level or participation level of the International Classification of Functioning, Disability, and Health (ICF). Specifically, we included outcome measures that assessed physical aspects of mobility, self-care, domestic life, major life areas, and community, social and civic life aspects of activity and participation. Examples of outcome measures include the Barthel Index (BI), gait speed, and the Stroke Impact Scale (SIS). Any type of study design was acceptable except if the study was a randomized controlled trial where one group received an intervention to treat the sleep disorder and another group did not. In this case the study was included only if data prior to the intervention comparing the groups was available. This was done because the intervention (e.g. continuous positive airway pressure) may have had an impact on functional recovery. Other inclusion criteria were published in English and in a peer reviewed journal.
Exclusion criteria were (1) the study included health conditions other than stroke and data on stroke not separated out; (2) description of sleep disturbance but no AASM-recognized diagnosed sleep disorder; (3) the outcome solely measured the impact on cognition, memory, or attention; (4) the study solely examined how having a sleep disorder prior to stroke might predict having a stroke; and (5) conference proceeding.
Study Selection
References identified through the above information sources and search strategies were downloaded to Covidence. Each title and abstract were screened independently by 2 researchers working in pairs (RG/MH and SR/JT) for possible inclusion. If there was a difference of opinion regarding including or excluding a study, the title and abstract were reviewed by a third researcher (GF) who made the final decision to include or exclude the study after discussion with the group. Next, the full text of the remaining articles was read independently by 2 researchers working in pairs (RG/MH and SR/JT). If there was a difference of opinion between the 2 researchers regarding including or excluding the study, a third researcher (GF) reviewed the article and discussed the differences with the group to reach consensus. Finally, reference lists of the remaining studies were screened for possibly relevant articles; full text of these articles were the read and assessed for inclusion or exclusion by one researcher (GF).
Data Extraction
Each included article was read independently by two researchers working in pairs (RG/MH and SR/JT) and the following data were extracted: purpose, sample size, time since stroke, stroke severity, setting, type of sleep disorder and how it was diagnosed, and outcome measures at the activity and participation level used. If there was a difference in the data extracted between the two researchers, then a third researcher (GF) reviewed the article and its data and discussed the differences with the group to reach consensus.
Risk of Bias of Individual Studies
One researcher (GF) reviewed all included studies and rated them for potential bias using a modified version of the Newcastle Ottawa Quality Assessment Scale for Cohort Studies (NOQASCS). The NOQASCS assesses potential bias across 3 main areas: selection, comparability, and outcome. Selection consists of representativeness of the cohort (were the participants with stroke and a sleep disorder truly representative of the average person with stroke for the setting in which the study was conducted), selection of the nonexposed cohort (were the participants with stroke without a sleep disorder drawn from the same community as the participants with stroke with a sleep disorder), ascertainment of the exposure (was the sleep disorder diagnosed using a valid method recommended by the AASM), and demonstration that the outcome of interest was not present at the start of the study. We did not include this last aspect of selection from the NOQASCS in our rating of potential bias as our purpose was to explore the impact of sleep disorders on activity and participation at inception, not to determine if a sleep disorder developed after stroke.
Comparability of the cohorts was assessed by ascertaining if the participants with stroke and a sleep disorder were similar to the participants with a stroke and without a sleep disorder in important characteristics such as age, gender, and stroke severity at the start of the study. Outcome consists of assessment of outcome (was the assessor independent and blind to sleep disorder diagnosis), follow-up (was follow-up long enough for change to have happened since initial assessment), and adequacy of follow-up (was follow-up complete, all subjects accounted for or follow-up rate greater than 80% or description of those lost to follow-up suggest no difference from those who follow up).
Data Synthesis
Initially, due to an expected heterogeneity of sleep disorders, methods of diagnosing sleep disorders, methodologies, and outcome measures used we did not anticipate being able to synthesize the data using a meta-analysis. We planned on only using a narrative approach to summarize the evidence for the impact of each type of sleep disorder on recovery after stroke on the outcomes of interest: mobility, self-care, domestic life, major life areas, and community, social and civic life aspects of activity and participation. However, on completion of the search and data extraction we determined that performing a meta-analysis was also possible, as many studies reported scores on either the BI and/or modified Rankin Scale (mRS). Because of this we were able to also perform a meta-analysis examining the effect of sleep disorder on recovery as assessed by the BI and mRS.
To be included in the meta-analysis, studies had to report means, standard deviations and sample sizes for either the mRS or BI in both a stroke + sleep disorder group and a control group (people with stroke without a sleep disorder). Effect sizes were extracted for each dependent variable (mRS and BI) at each study timepoint when sleep disorder and control groups were compared. Because the purpose of the study was to determine the impact of sleep disorders on poststroke function (ie, cross-sectional impact) we included data from every available time point to maximize the precision of the pooled effect size estimate and to minimize risk of bias from selective data inclusion. These effect sizes were calculated as the mean score in the sleep disorder group minus the mean score in the control group. Variances for each effect size were calculated using the following formulae48,49:
where
The overall meta-analysis for each dependent variable calculated a pooled effect size estimate across all available studies and time points. Meta-regression models were then conducted to assess the association between study effect size and each independent variable of interest, including sleep disorder type (SDB vs restless leg syndrome [RLS]), stroke chronicity (<3 months poststroke vs ≥3 months) and mean level of the dependent variable. One study of insomnia was excluded from the sleep disorder type meta-regression because there was only a single BI comparison for this sleep disorder type.
All models included a random effect for study to model between-study variance. In cases where multiple effect sizes were included from different measurement time points within the same study, a second random effect was added for time point to account for within-study correlation. For the BI, this strategy had to be modified because the available dataset was insufficient to model the required number of covariance parameters. Instead of being nested within-study, the second random effect had to pool information across studies to estimate the covariance between time points. To determine the extent to which this might have influenced the BI results, we also performed a sensitivity analysis that only included a single time point from each study (thus eliminating the need for a second random effect). For this sensitivity analysis, the 3-month poststroke time points were selected because they were nearest the middle time point for these studies.
Results
Article Selection
All database searches were conducted on November 30, 2018. Together, these searches identified 2773 articles. Through the title and abstract screening and full text review, this was narrowed to 29 articles. A review of the reference lists of these 29 articles identified 4 additional articles that met the inclusion criteria, resulting in a total of 33 articles that were included in this review. The primary reasons studies were excluded were not reporting an outcome measure at the activity or participation level, wrong population (no diagnosed sleep disorder), and study focus on sleep disorder as a predictor of stroke. See Figure 1 for the article selection process.
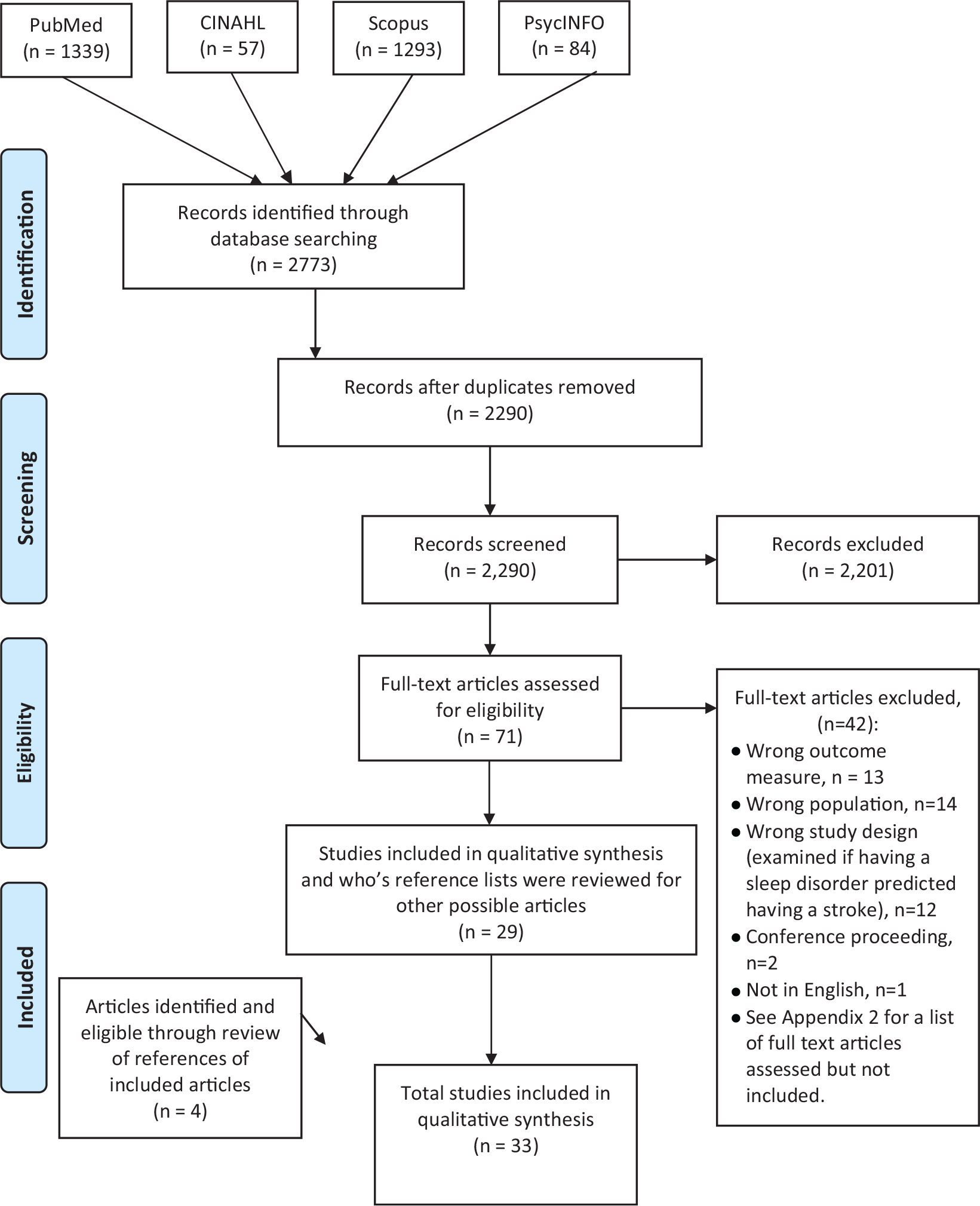
Article selection.
Twenty-three studies35,36,38,39,44 -46,50 -65 examined the impact of SDB on recovery after stroke. In these studies, there were a total of 1,998 participants, with a prevalence of SDB of 57.4%. One study by Brooks and colleagues 52 only had participants with SDB (45 participants). Ten studies23,24,40 -42,66 -70 examined the impact of other sleep disorders. These included RLS24,41,42,66,67 with a prevalence of 18.0% in 5 studies and insomnia23,40,68 -70 with a prevalence of 44.5% in 5 studies. The majority of participants across all of the studies exhibited mild to moderate stroke severity as measured by the National Institutes of Health Stroke Scale (NIHSS) or other similar scale.
The most commonly used outcome measures at the physical functioning level of activity was the BI, which was used in 19 studies, and the mRS, which was used in 15 studies. Other measures of physical functioning at the level of activity that were used were the Functional Independence Measure (FIM), Utrecht Scale for Evaluation of Rehabilitation (USER), Functional Ambulation Categories (FAC), Berg Balance Scale (BBS), 9-hole peg test, and Purdue Peg test. Measures of participation included the 36-Item Short Form Survey (SF-36), EuroQol-5, 8-Item Short Form Survey (SF-8), and the Stroke Specific Quality of Life (SSQoL).
Sleep Disordered Breathing
Activity
The impact of SDB, which in these studies included OSA, central sleep apnea (CSA), and Cheyne Stokes respirations, on recovery of physical function at the activity level was mixed. Seven studies35,36,38,39,51,61,63 found that participants with SDB had worse outcomes at the activity level during the acute/subacute stage of recovery (<3 months poststroke) compared with participants without SDB. However, 8 studies35,44,45,55,56,58 -60 found no difference in outcomes of physical function at the activity level in the acute/subacute stages of recovery. The BI and the FIM were the most commonly used outcomes to measure physical functioning at the activity level. A variety of methods were used to diagnose SDB, including polysomnography (PSG), nocturnal oximetry, and home sleep apnea testing system (HSATS). There were also differences across studies in the criteria used to diagnose SDB (eg, Apnea Hypopnea Index [AHI] > 15 vs AHI > 30). See online Table 1 with specific information on each study.
During the chronic stage of recovery (≥3 months poststroke), the findings were also mixed. Five studies35,50,54,57,61 found that participants with SDB had worse outcomes at the activity level compared to participants without SDB, while 5 studies44,51,56,64 found no difference in activity levels. The BI was also commonly used and there were variety of methods used to diagnose SDB including (HSATS, PSG, and nocturnal oximetry). There were also differences across studies in the criteria used to diagnose SDB. See online Table 1 with specific information on each study.
Participation
The impact of SDB on participation during the acute/subacute stage of recovery poststroke was also mixed. One study 39 found that participants with SDB had significantly worse outcomes on measures of participation and 2 studies39,44 found no difference in participation outcomes between participants with and without SDB. A HSATS was used to diagnose SDB in most of the studies. There were also differences across studies in the criteria used to diagnose SDB. See online Table 1 with specific information on each study.
No studies were found that examined the impact of SDB on participation in the chronic stage of recovery.
There were four studies52,53,62,65 that had participants with stroke both with and without SDB in which the authors did not directly compare activity or participation outcomes between the groups. Brooks and colleagues 52 found no relationship between AHI and FIM and a little to moderate relationship (r = 0.30) with the BI during inpatient rehabilitation. Yan-fang and colleagues 65 reported that AHI at admission to the hospital was an independent predictor of independence measured by the BI at 3-month poststroke. Pace and colleagues 62 reported that there was a significant difference in admission AHI between participants with a good outcome (mRS ≤2) and a poor outcome (mRS >2) at discharge. Cadilhac and colleagues 53 reported that dependency (mRS ≥2) at 1 month, 6 months, and 3 years poststroke showed no significant effect on AHI ≥15. They also reported that dependency (mRS ≥2) at 1 month poststroke was found to be significantly associated with having an AHI ≥15 at 3 years poststroke. See online Table 1 with specific information on each study. studies. The median score on the NOQASCS in studies that examined the impact of SDB on recovery was 4/7 with a range of 2/7 to 7/7, see online Table 2.
Other Sleep Disorders: Restless Leg Syndrome, Insomnia, and Excessive Daytime Sleepiness
Restless Leg Syndrome
Five studies24,41,42,66,67 examined the impact of restless leg syndrome (RLS) on recovery after stroke. Two studies24,41 found that participants with RLS had significantly worse outcomes at the activity level compared to participants without RLS during the acute/subacute stage of recovery after stroke. While 1 study 67 found that there was no significant difference in activity levels between participants with and without RLS, another study 24 found that participants with RLS had significantly worse outcomes at the activity level compared to participants without RLS during the chronic stage of recovery after stroke. During the acute/subacute stage of recovery, 1 study 42 found that participants with RLS had significantly worse outcomes at the participation level compared to participants without RLS. During the chronic stage of recovery, 1 study 42 found that participants with RLS had significantly worse outcomes at the participation level compared to those without RLS. One study did not report on differences between groups but reported finding no significant correlations between RLS measures and the BI. 66 An interview using the RLS criteria established by the International RLS Study Group was commonly used to diagnose RLS. See online Table 3 for specific information on each study. The median score on the NOQASCS in studies that examined the impact of RLS on recovery was 4/7 with a range of 3/7 to 5/7, see online Table 2.
Insomnia
Five studies23,40,68 -70 examined the impact of insomnia on recovery after stroke. The findings were also mixed. During the acute/subacute stage of recovery after stroke, 3 studies40,68,69 found no significant difference in activity between participants with stroke with and without insomnia, while 1 study 40 found that participants with insomnia had significantly worse outcomes on measures of activity compared to participants without insomnia. During the chronic stage of recovery after stroke, 1 study 23 found no significant difference in activity between those with and without insomnia. Measures of activity in these studies include the BI, BBS, 9-hole peg test, and Purdue peg board test. For participation, 1 study 69 during the acute/subacute stage of recovery after stroke and 1 study 23 during the chronic stage of recovery after stroke found that participants with stroke and insomnia had significantly worse outcomes in participation compared to participants with stroke and without insomnia. Measures of participation in these studies included the SF-8, and SSQoL. Another study 70 that did not directly compare differences between participants with and without insomnia reported a small relationship between the mRS and scores on the Insomnia Severity Index (ISI). Insomnia was diagnosed by using a researcher developed questionnaire or the ISI. See online Table 3 for specific details on each study. The median score on the NOQASCS in studies that examined the impact of insomnia on recovery was 2/7 with a range of 1/7 to 3/7, see online Table 2.
Meta-Analysis Results for Impact of Sleep Disorder on Activity and Participation
For the mRS meta-analysis, there were 18 total time point comparisons among 9 studies with available data from 957 total participants (Table 1; Figure 2a). The overall meta-analysis estimated that the mean mRS score was 0.51 points higher in sleep disorders versus control groups [95% CI: 0.23, 0.78]. From the meta-regression analyses, this effect size did not significantly differ based on sleep disorder type, stroke chronicity or mean mRS score (Table 1; right column). Within subgroups, the sleep disorder versus control difference was statistically significant for sleep apnea studies but not RLS studies (2 studies with 5 total time point comparisons). This difference was also statistically significant for both early time points (<3 months poststroke) and later time points (Table 1).
Summary of Random Effects Meta-Analysis Results. a
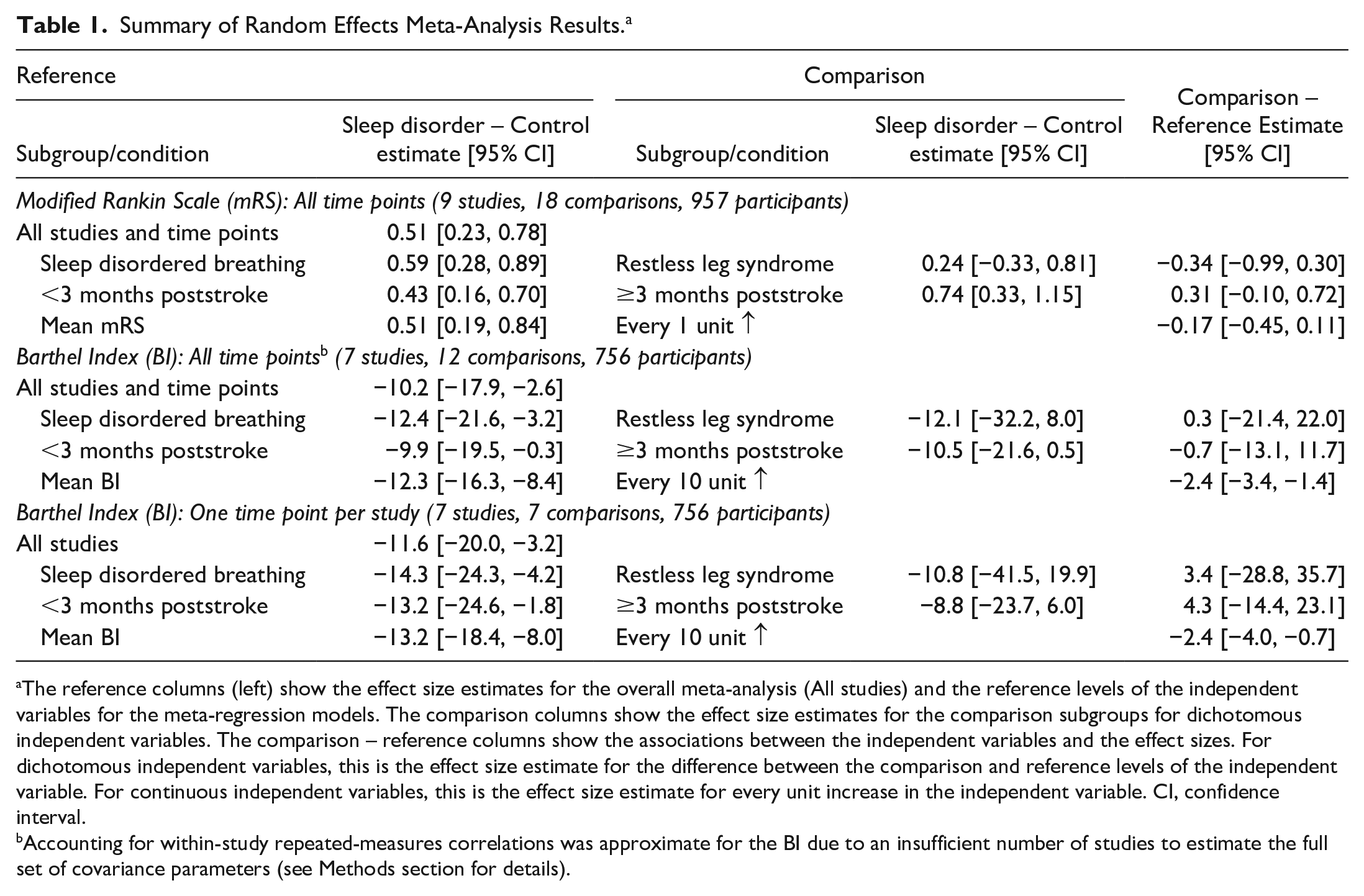
The reference columns (left) show the effect size estimates for the overall meta-analysis (All studies) and the reference levels of the independent variables for the meta-regression models. The comparison columns show the effect size estimates for the comparison subgroups for dichotomous independent variables. The comparison – reference columns show the associations between the independent variables and the effect sizes. For dichotomous independent variables, this is the effect size estimate for the difference between the comparison and reference levels of the independent variable. For continuous independent variables, this is the effect size estimate for every unit increase in the independent variable. CI, confidence interval.
Accounting for within-study repeated-measures correlations was approximate for the BI due to an insufficient number of studies to estimate the full set of covariance parameters (see Methods section for details).
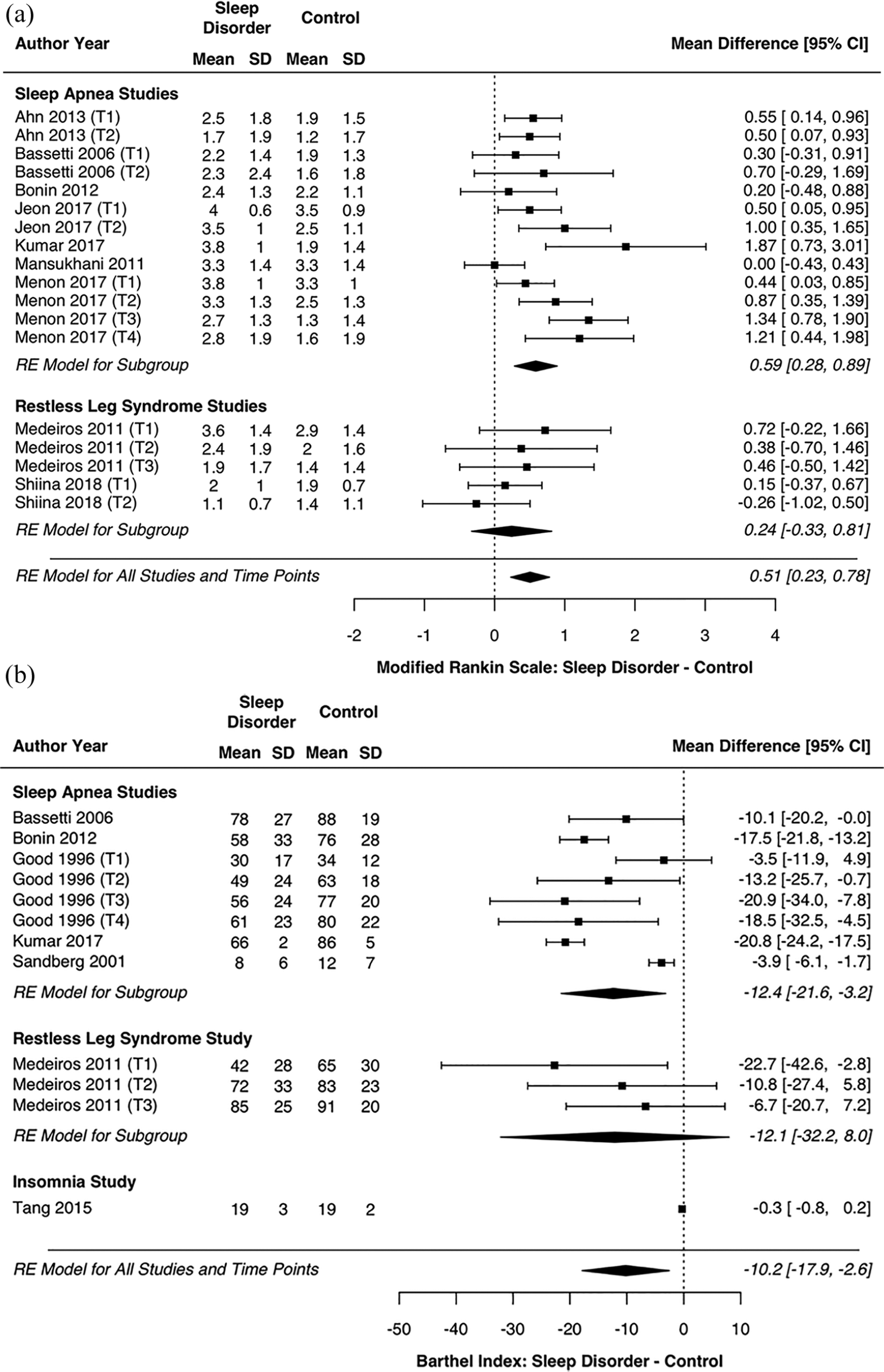
(a) Forest plot of modified Rankin Scale mean differences by sleep disorder type. (b) Forest plot of Barthel Index mean differences by sleep disorder type. SD, standard deviation; CI, confidence interval; (T), time point; RE, random effects.
For the BI meta-analysis, there were 12 total time point comparisons among 7 studies with available data from 756 total participants (Table 1; Figure 2b). The overall meta-analysis estimated that the mean BI score was 10.2 points lower in sleep disorders versus control groups [95% CI: −17.9, −2.6]. From the meta-regression analyses, this effect size did not significantly differ based on sleep disorder type or stroke chronicity but did vary based on mean BI score (online Table 4; right column). Studies and time points with a higher mean BI showed greater between-group differences. Within subgroups, the sleep disorder versus control difference was statistically significant for sleep apnea studies but not the RLS study (1 study with 3 total time point comparisons). The sleep disorder versus control difference was also statistically significant for early time points (<3 months poststroke) but not quite for later time points (P = .0594), despite a slightly larger point estimate (online Table 4). In the sensitivity analysis that only included one–time point comparison from each study, none of the above findings were meaningfully altered (Table 1; bottom rows).
Discussion
Based solely on a descriptive analysis of the findings of the systematic review there appeared to be mixed evidence to support the notion that sleep disorders negatively affect recovery at the physical function level of activity and participation after stroke. With approximately 50% of the studies showing a significant difference in outcomes between those with and without a sleep disorder and the other ~50% showing no difference. However, the meta-analysis demonstrated that people with stroke and a sleep disorder are likely to have worse outcomes at the activity level as measured by the BI and mRS. People with stroke and a sleep disorder are likely to score approximately 10 points lower on the BI and 0.5 points higher on the mRS (higher score indicates greater disability) than people with stroke without a sleep disorder. This is true even when controlled for baseline severity.
Although the meta-analysis found that when all sleep disorder types were pooled there was a negative effect on recovery, these findings are more applicable to people with stroke with SDB compared to RLS and insomnia. The meta-analysis results showed that people with stroke and SDB had worse outcomes on the BI and mRS compared to people with stroke without a sleep disorder. There was not enough power to detect a difference between those with RLS and those without a sleep disorder or a difference between those with SDB and RLS. People with a sleep disorder had worse outcomes on the BI before 3 months poststroke than those without a sleep disorder and there was a trend toward worse outcomes on the BI at ≥3 months poststroke. Disability as measured by the mRS was worse both prior to and ≥3 months poststroke. We are also more confident in the findings as they relate to SDB as the risk of bias in these studies was lower than in those with RLS or insomnia.
Our findings provide further evidence of the wide-ranging negative effects of disturbed sleep on recovery after stroke. Birkbak and colleagues 71 reported an increased risk of recurrent stroke and mortality in people with stroke or transient ischemic attack with SDB. People with stroke with disturbed sleep but no diagnosed sleep disorder demonstrate poorer functional mobility and greater limitations in ability to complete activities of daily living.72,73
Our results are consistent with those found in the other populations with sleep disorders. Sleep apnea and insomnia in the general population is associated with greater incidence of work related disability.74 -77 People with multiple sclerosis with sleep apnea or RLS have greater disability,78,79 higher levels of fatigue, 80 and lower quality of life 81 than people with multiple sclerosis without these sleep disorders. In people with traumatic brain injury, insomnia is associated with poorer quality of life as well. 82 Older adults with RLS have greater functional disability than those without RLS.83,84 In addition, the AASM and Sleep Research Society (SRS) concluded that poor or insufficient sleep is associated with deficits in performance, increased occurrence of errors, and higher likelihood of accidents (as well as worse physical and psychiatric health) and thus jointly recommended amounts of sleep for healthy adults.85,86
The prevalence of sleep disorders among people with stroke found in this review indicate that some sleep disorders are more highly prevalent in people with stroke when compared with the general population. For example, in the general population, SDB is reported to be 9% to 38%, 87 chronic insomnia disorder at 10% 88 to 20%, 89 RLS at 5% to 10% (increasing with age), 88 EDS at 12.7% 89 and insufficient sleep disorder at 30%. 90 The higher prevalence of SDB in people with stroke is likely related to risk factors: high BMI, sedentary lifestyle, and hypertension are all known to increase risk for both stroke and obstructive sleep apnea. 88
The many causes of sleep disorders and the fact that they may coexist 91 make it challenging to determine exact mechanisms of how disordered sleep negatively affects recovery poststroke. In stroke, sleep disordered breathing may be due to loss of or limited pharyngeal muscle activity due to paresis or to damage to pontomedullary structures resulting in impaired neural output.92,93 Obesity, which is common in people with stroke, is also a risk factor for SDB 94 that further complicates the understanding of how SDB affects recovery after stroke. Insomnia is considered a stress-related event 95 and this coupled with factors such as anxiety and depression that are common after stroke can lead to chronic insomnia. Although we cannot determine the exact mechanisms by which sleep disorders impact functional recovery from this systematic review, we believe it is multi-factorial. Disordered sleep during the early stages of recovery likely has a negative influence on motor learning, attention, and executive function all of which are important components of rehabilitation. During the chronic stage of recovery, the general negative health consequences of sleep disorders likely contributes. The bidirectional relationship between sleep and activity96,97 is an especially important consideration in the context of stroke recovery. Inactivity, which is common after stroke, may promote disturbed sleep while disturbed sleep may lead to inactivity.
These findings provide important opportunities for rehabilitation professionals. Tools such as the Sleep Disorders Symptom Checklist-25, 98 Berlin Questionnaire, 99 Epworth Sleepiness Scale, 100 STOP-Bang,101,102 Insomnia Severity Index, 103 and Cambridge Hopkins Restless Leg Syndrome questionnaire 104 should be used to screen clients with stroke to identify possible sleep disorders and assist with referral to an appropriate health care professional. Research is needed that pairs sleep interventions such as continuous positive airway pressure, 105 cognitive behavioral therapy for insomnia,106,107 or sleep hygiene education 108 with rehabilitation interventions. Concomitant delivery of these interventions may have increased benefit compared with delivery in isolation.
There are some important limitations in this systematic review and meta-analysis that should be kept in mind when interpreting and applying the findings. There was not a consistent method for diagnosing the different sleep disorders. For example, studies used different AHI cutoff points and different devices to diagnose SDB. Most of the studies included participants with mild to moderate stroke. Perhaps a sleep disorder in people with severe stroke has an even greater negative impact on recovery compared with what we report. There was limited to no follow-up in many of the included studies, approximately 50% of the studies had no follow-up. It is not clear if the negative impact of the sleep disorder was compounded over time or stayed the same. We also cannot completely rule out the possibility that the observed associations could be confounded by other factors causing increased risk of both sleep disorders and worse outcomes (eg, obesity, diabetes, cardiovascular disease, poor mental health, dementia). The results of the meta-analysis should be interpreted with caution as there is potential for bias in some of the included studies based on the NOQASCS scale. Many of the studies included were not designed to specifically answer the specific purpose of this study and there is no single validated tool specifically designed to measure risk of bias across the wide variety of modified study designs that are included here. We believe the risk of bias in these studies is not sufficient to explain the entirety of the observed effect. This meta-analysis provides a preliminary estimate of the effect of sleep disorders on recovery after stroke that should be confirmed with future studies.
This is the first systematic review and meta-analysis that that has examined the impact of sleep disorders on physical functioning and participation in people with stroke. People with stroke and a sleep disorder have greater functional limitations and disability than those without a sleep disorder. Rehabilitation professionals should screen their patients with stroke for potential sleep disorders and further research is needed to develop sleep and rehabilitation interventions that can be delivered in combination.
Supplemental Material
Supplemental_tABLE – Supplemental material for The Impact of Sleep Disorders on Functional Recovery and Participation Following Stroke: A Systematic Review and Meta-Analysis
Supplemental material, Supplemental_tABLE for The Impact of Sleep Disorders on Functional Recovery and Participation Following Stroke: A Systematic Review and Meta-Analysis by George D. Fulk, Pierce Boyne, Makenzie Hauger, Raktim Ghosh, Samantha Romano, Jonathan Thomas, Amy Slutzky and Karen Klingman in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.